Submitted:
22 May 2025
Posted:
23 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Antibody Constructs, Protein Expression and Purification
2.2. MS-Analysis
2.3. Capillary Gelelectrophoresis (cGE)
2.4. SDS-PAGE Analysis
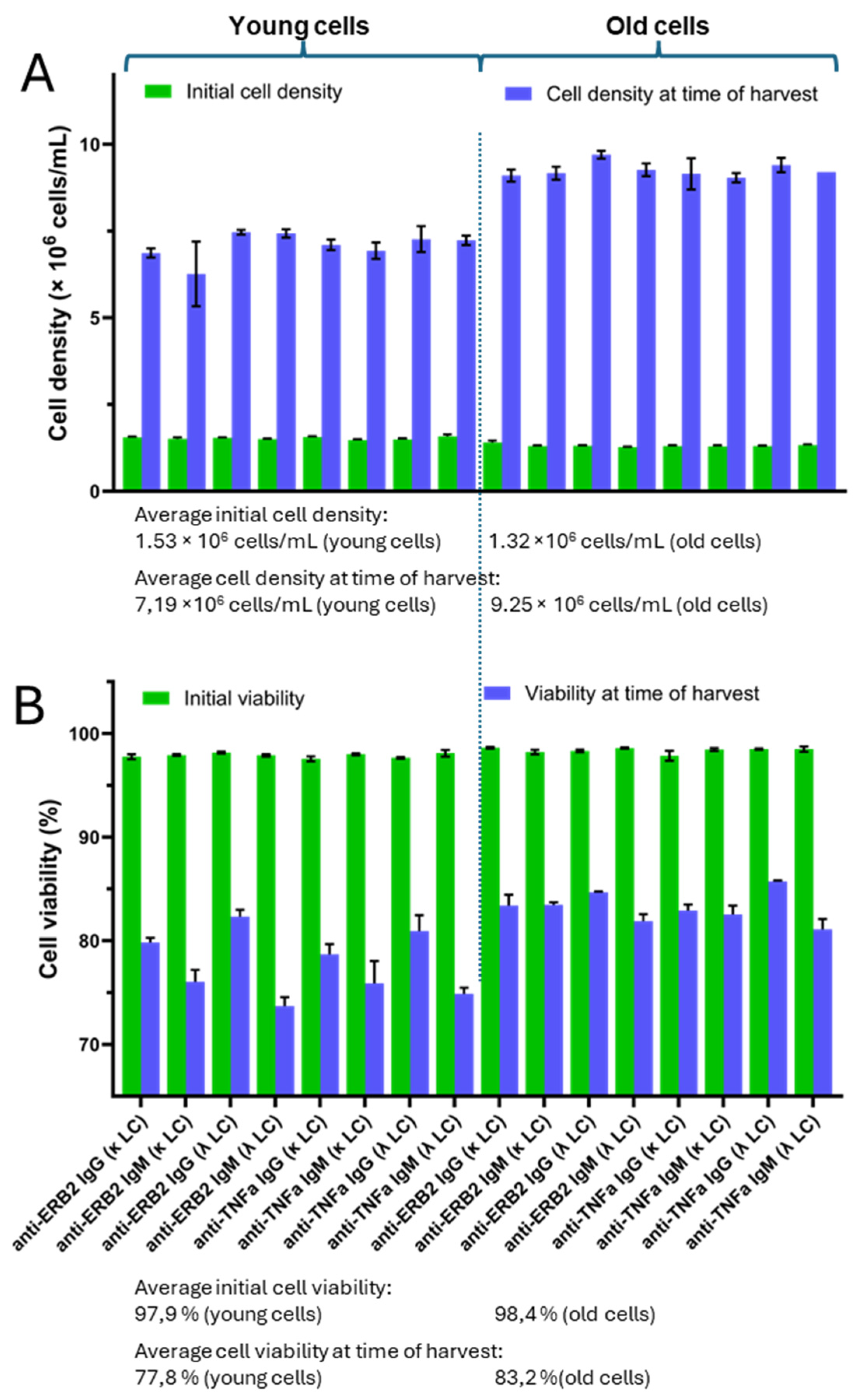
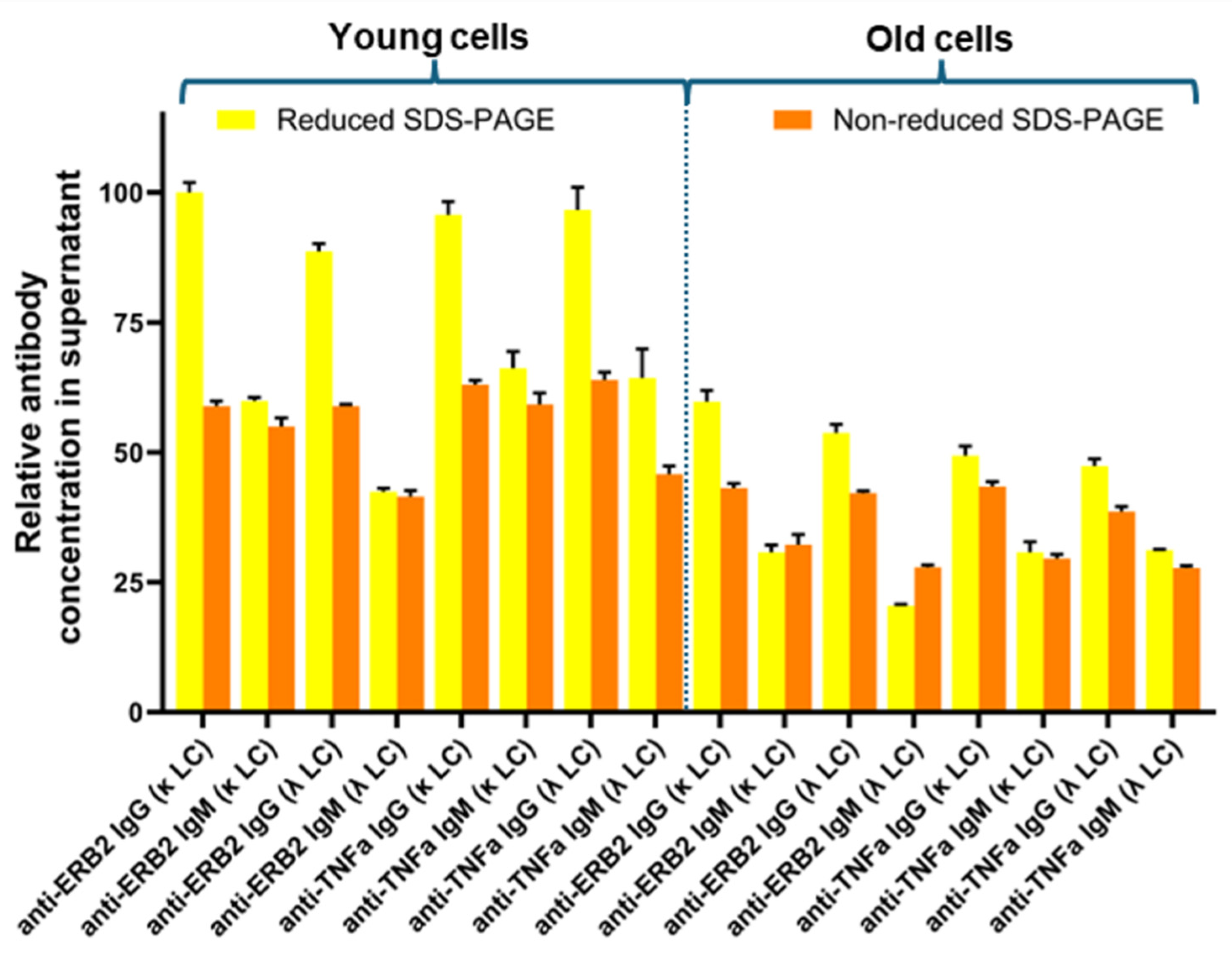
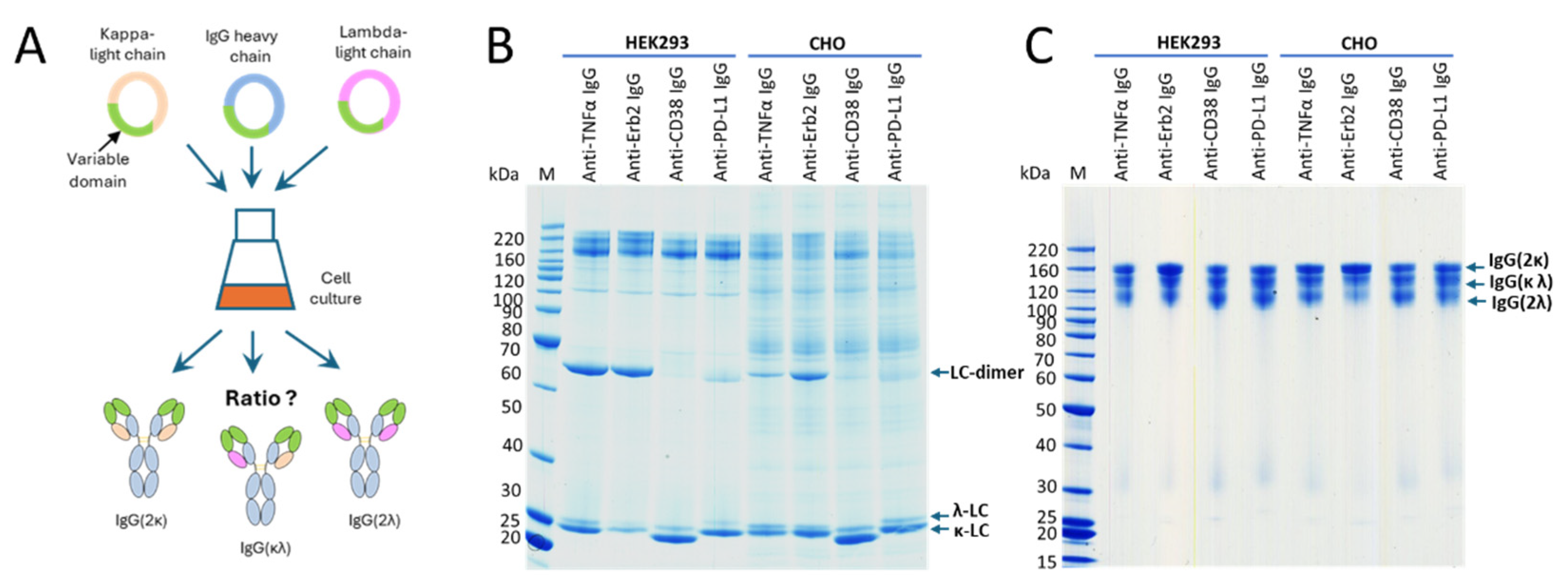
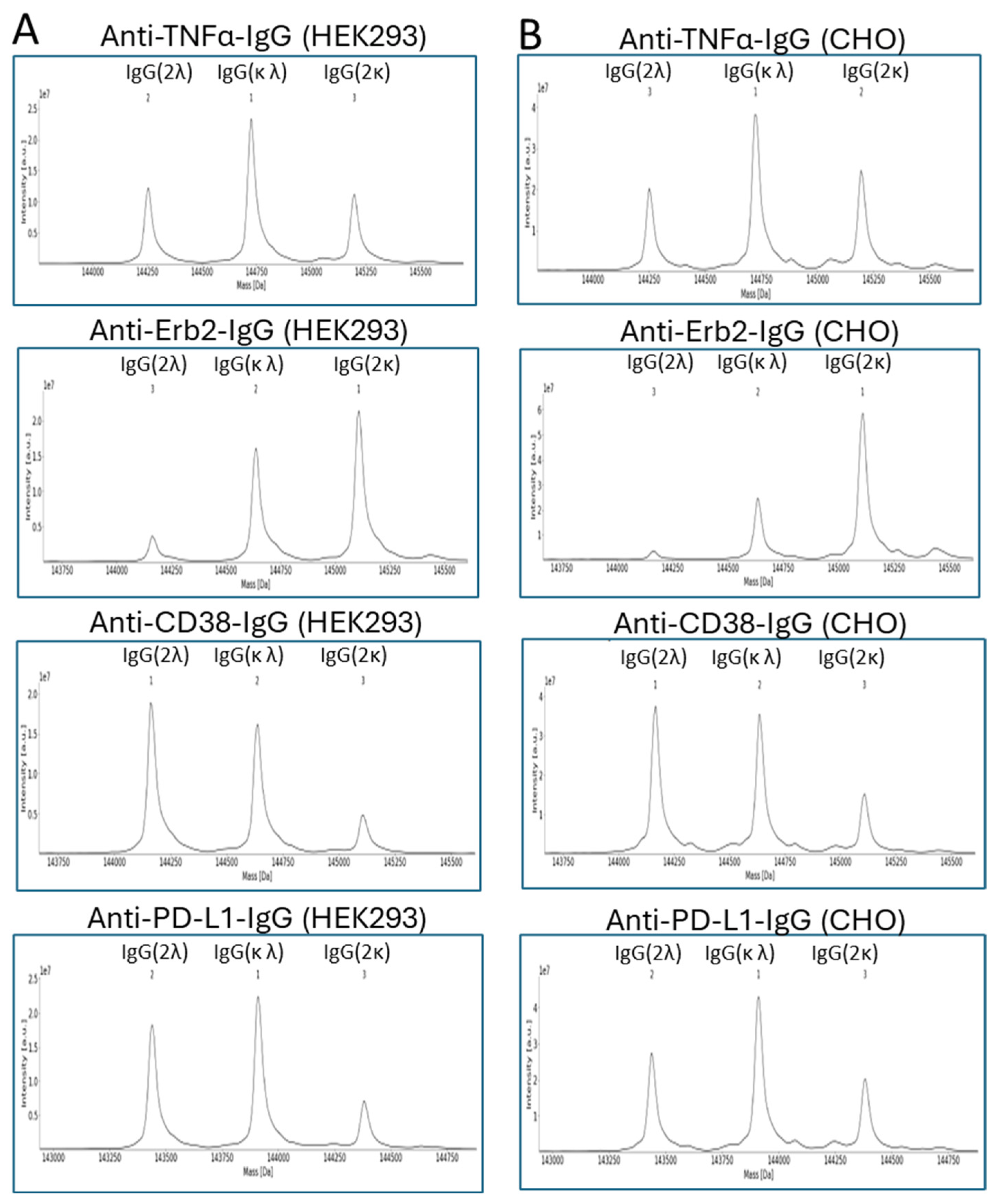
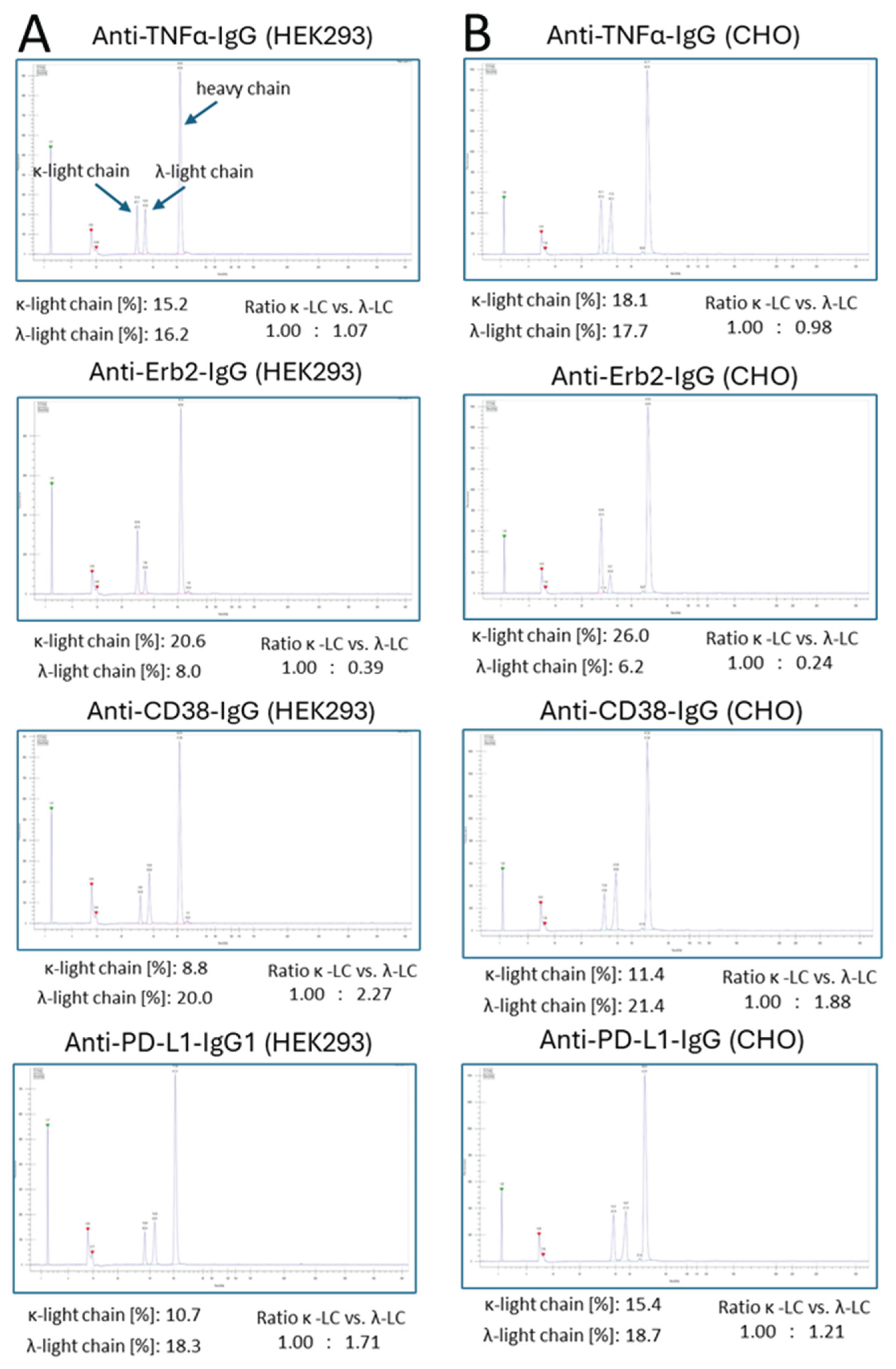
3. Results
4. Discussion
Supplementary Materials
Conflicts of Interest statement
References
- Arun SS, Breuer W, Hermanns W. Immunohistochemical examination of light-chain expression (lambda/kappa ratio) in canine, feline, equine, bovine and porcine plasma cells. Zentralbl Veterinarmed A. 1996;43(9):573-576. [CrossRef]
- Becker W, Scherer A, Faust C, et al. A fully automated three-step protein purification procedure for up to five samples using the NGC chromatography system. Protein Expr Purif. 2019;153:1-6. [CrossRef]
- Crescioli S, Kaplon H, Wang L, Visweswaraiah J, Kapoor V, Reichert JM. Antibodies to watch in 2025. MAbs. 2025;17(1):2443538. [CrossRef]
- Durdik J, Moore MW, Selsing E. Novel kappa light-chain gene rearrangements in mouse lambda light chain-producing B lymphocytes. Nature. 1984;307(5953):749-752. [CrossRef]
- Giannone C, Mess X, He R, et al. How J-chain ensures the assembly of immunoglobulin IgM pentamers. EMBO J. Published online , 2024. 4 December. [CrossRef]
- Gong S, Gautam S, Coneglio JD, Scinto HB, Ruprecht RM. Antibody Light Chains: Key to Increased Monoclonal Antibody Yields in Expi293 Cells?. Antibodies (Basel). 2022;11(2):37. Published 2022 May 18. [CrossRef]
- Haughton G, Lanier LL, Babcock GF. The murine kappa light chain shift. Nature. 1978;275(5676):154-157. [CrossRef]
- Keyt BA, Baliga R, Sinclair AM, Carroll SF, Peterson MS. Structure, Function, and Therapeutic Use of IgM Antibodies. Antibodies (Basel). 2020;9(4):53. Published 2020 Oct 13. [CrossRef]
- Kim JY, Kim YG, Lee GM. CHO cells in biotechnology for production of recombinant proteins: current state and further potential. Appl Microbiol Biotechnol. 2012;93(3):917-930. [CrossRef]
- Lehmann A, Wixted JH, Shapovalov MV, Roder H, Dunbrack RL Jr, Robinson MK. Stability engineering of anti-EGFR scFv antibodies by rational design of a lambda-to-kappa swap of the VL framework using a structure-guided approach. MAbs. 2015;7(6):1058-1071. [CrossRef]
- Li Y, Wang G, Li N, et al. Structural insights into immunoglobulin M. Science. 2020;367(6481):1014-1017. [CrossRef]
- Malm M, Saghaleyni R, Lundqvist M, et al. Evolution from adherent to suspension: systems biology of HEK293 cell line development [published correction appears in Sci Rep. 2021 Mar 2;11(1):5407. doi: 10.1038/s41598-021-85105-9.]. Sci Rep. 2020;10(1):18996. Published 2020 Nov 4. [CrossRef]
- McBride OW, Hieter PA, Hollis GF, Swan D, Otey MC, Leder P. Chromosomal location of human kappa and lambda immunoglobulin light chain constant region genes. J Exp Med. 1982;155(5):1480-1490. [CrossRef]
- Molé CM, Béne MC, Montagne PM, Seilles E, Faure GC. Light chains of immunoglobulins in human secretions. Clin Chim Acta. 1994;224(2):191-197. [CrossRef]
- Popov AV, Zou X, Xian J, Nicholson IC, Brüggemann M. A human immunoglobulin lambda locus is similarly well expressed in mice and humans. J Exp Med. 1999;189(10):1611-1620. [CrossRef]
- Schroeder HW Jr, Cavacini L. Structure and function of immunoglobulins. J Allergy Clin Immunol. 2010;125(2 Suppl 2):S41-S52. [CrossRef]
- Smith K, Shah H, Muther JJ, Duke AL, Haley K, James JA. Antigen nature and complexity influence human antibody light chain usage and specificity. Vaccine. 2016;34(25):2813-2820. [CrossRef]
- Solomon A and Weiss DT. Structural and functional properties of human lambda-light-chain variable-region subgroups. Clin Diagn Lab Immunol. 1995;2(4):387-394. 1995. [CrossRef]
- Stavnezer J, Guikema JE, Schrader CE. Mechanism and regulation of class switch recombination. Annu Rev Immunol. 2008;26:261-292. [CrossRef]
- Strohl WR & Strohl LM (2012) Therapeutic Antibody Engineering, Woodhead Publishing,111-595. [CrossRef]
- Tan E, Chin CSH, Lim ZFS, Ng SK. HEK293 Cell Line as a Platform to Produce Recombinant Proteins and Viral Vectors. Front Bioeng Biotechnol. 2021;9:796991. Published 2021 Dec 13. [CrossRef]
- The Antibody Society. Therapeutic monoclonal antibodies approved or in regulatory review. (access April 2025); www.antibodysociety.org/antibody-therapeutics-product-data.
- Titani K, Wikler M, Putnam FW. Evolution of immunoglobulins: structural homology of kappa and lambda Bence Jones proteins. Science. 1967;155(3764):828-835. [CrossRef]
- van der Burg M, Tümkaya T, Boerma M, de Bruin-Versteeg S, Langerak AW, van Dongen JJ. Ordered recombination of immunoglobulin light chain genes occurs at the IGK locus but seems less strict at the IGL locus. Blood. 2001;97(4):1001-1008. [CrossRef]
- Woloschak GE, Krco CJ. Regulation of kappa/lambda immunoglobulin light chain expression in normal murine lymphocytes. Mol Immunol. 1987;24(7):751-757. [CrossRef]
- Zhou J, Yan GG, Cluckey D, et al. Exploring Parametric and Mechanistic Differences between Expi293FTM and ExpiCHO-STM Cells for Transient Antibody Production Optimization. Antibodies (Basel). 2023;12(3):53. Published 2023 Aug 10. [CrossRef]
| HEK293 expression | CHO expression | HEK293 expression | CHO expression | ||
|---|---|---|---|---|---|
| Non-reduced SDS-PAGE | MS (Signal intensity) | ||||
| Parental mab | Isoform | [%] | [%] | [%] | [%] |
| Anti-TNFα-IgG | IgG(2κ) | 35,7 | 36,4 | 23,9 | 29,7 |
| IgG(2λ) | 31,6 | 30,4 | 26,1 | 24,3 | |
| IgG(κλ) | 32,7 | 33,2 | 50,0 | 46,1 | |
| Anti-Erb2-IgG | IgG(2κ) | 46,4 | 50,3 | 52,0 | 67,4 |
| IgG(2λ) | 25,0 | 19,0 | 8,8 | 4,1 | |
| IgG(κλ) | 28,6 | 30,7 | 39,2 | 28,5 | |
| Anti-CD38-IgG | IgG(2κ) | 28,2 | 27,3 | 12,1 | 17,2 |
| IgG(2λ) | 37,5 | 38,2 | 47,3 | 42,5 | |
| IgG(κλ) | 34,3 | 34,5 | 40,6 | 40,2 | |
| Anti-PD-L1-IgG | IgG(2κ) | 28,9 | 31,8 | 14,8 | 22,3 |
| IgG(2λ) | 36,1 | 30,0 | 38,3 | 30,3 | |
| IgG(κλ) | 35,0 | 38,2 | 46,9 | 47,4 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




