Submitted:
29 April 2025
Posted:
14 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Historical Background and Applications of Ethidium Bromide
3. Structural Features and Chemical Properties
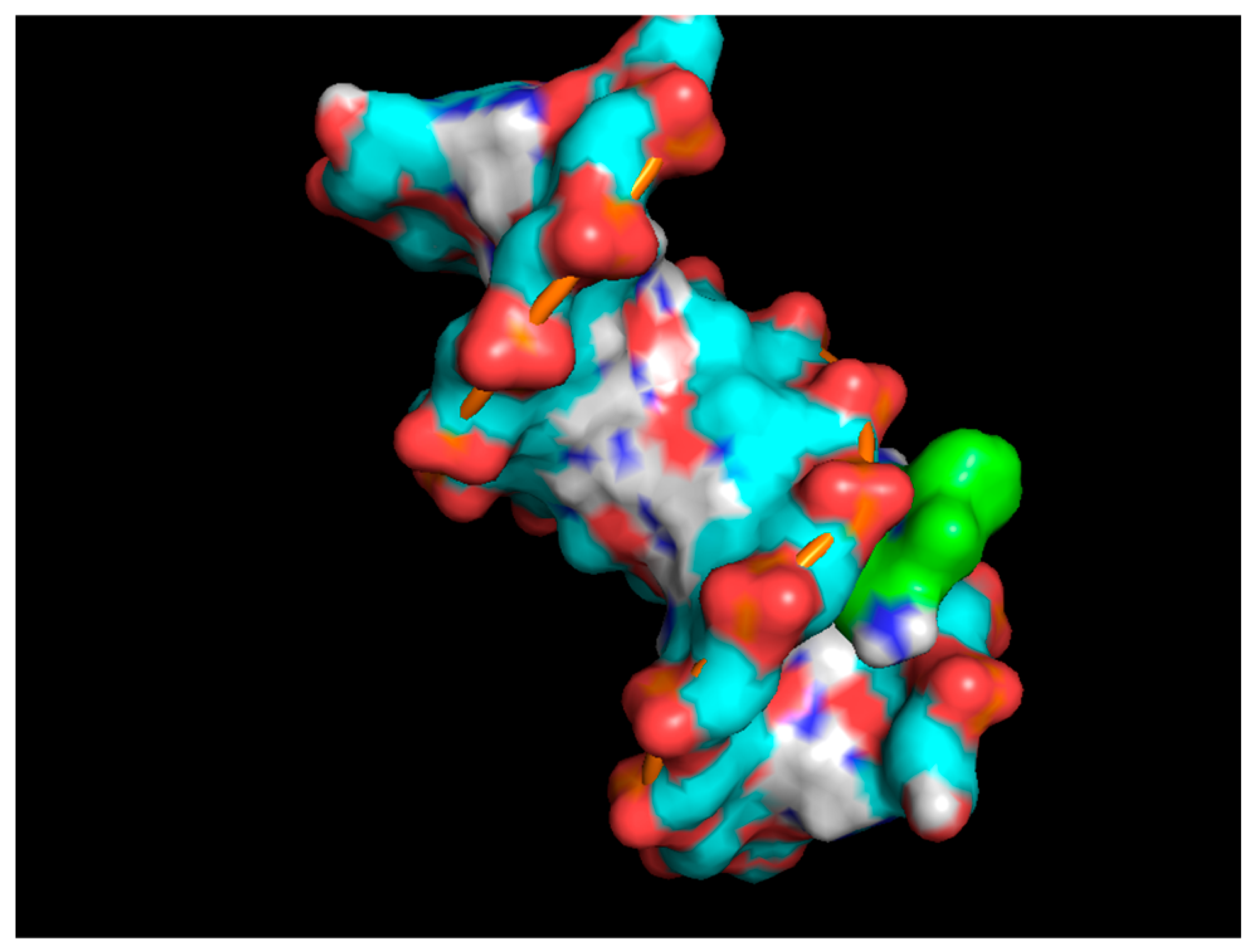
4. Mechanism of Action as a DNA Intercalator and Drug Agent
4.1. Mechanism of EtBr as a Drug
4.2. Mechanism of EtBr as a Dye
5. Toxicological Profile: Mutagenic and Carcinogenic Potential
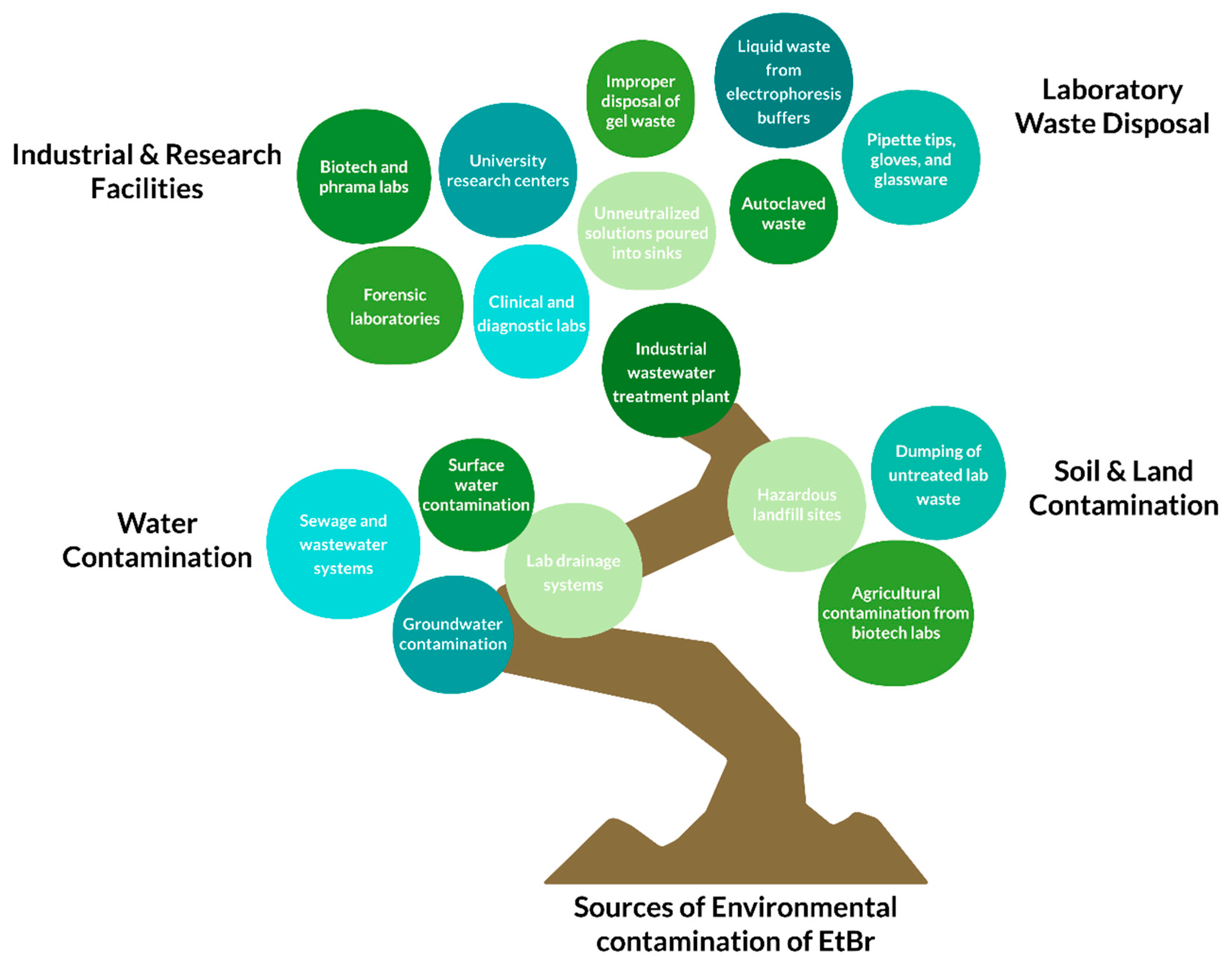
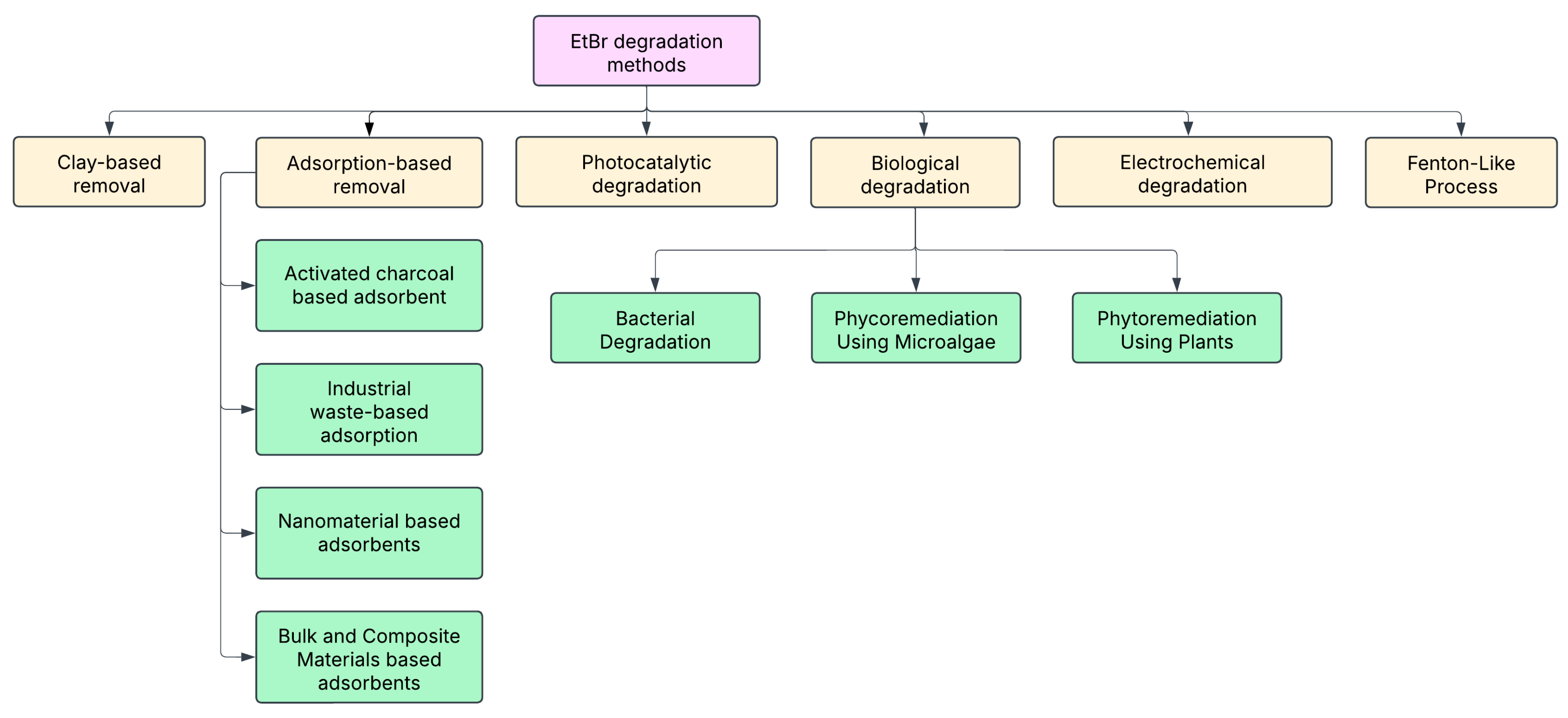
6. Strategies for Safe Handling and Removal of EtBr
6.1. Adsorption-Based Techniques
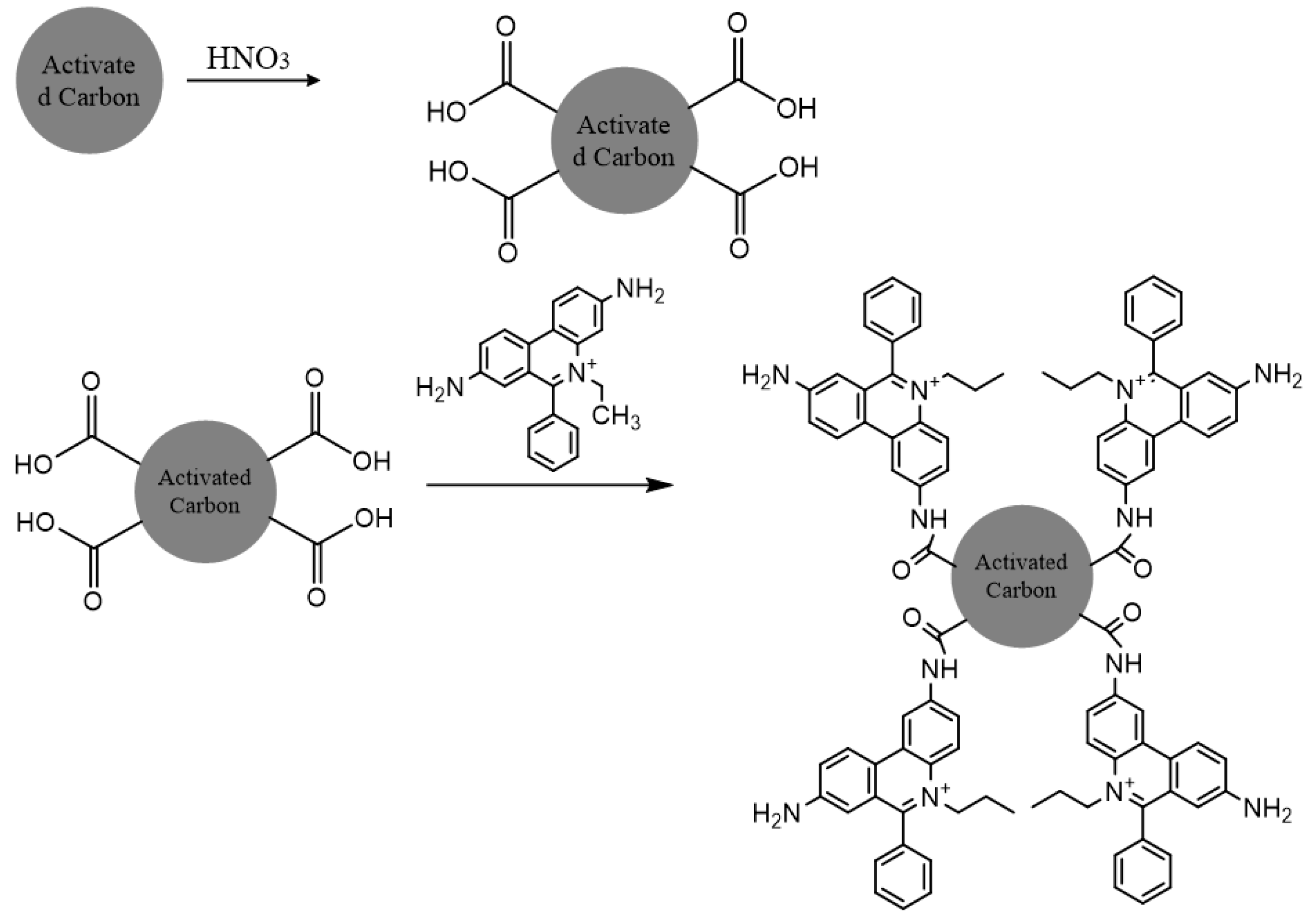
6.1.1. Activated Charcoal-Based Adsorbent
6.1.2. Industrial Waste-Based Adsorption
6.1.3. Nano-Composites
6.1.4. Bulk Composites
6.2. Clay-Based Removal
6.3. Biological Degradation
6.3.1. Bacterial Degradation
6.3.2. Phycoremediation Using Microalgae
6.3.3. Phytoremediation Using Plants
6.4. Photocatalytic Degradation
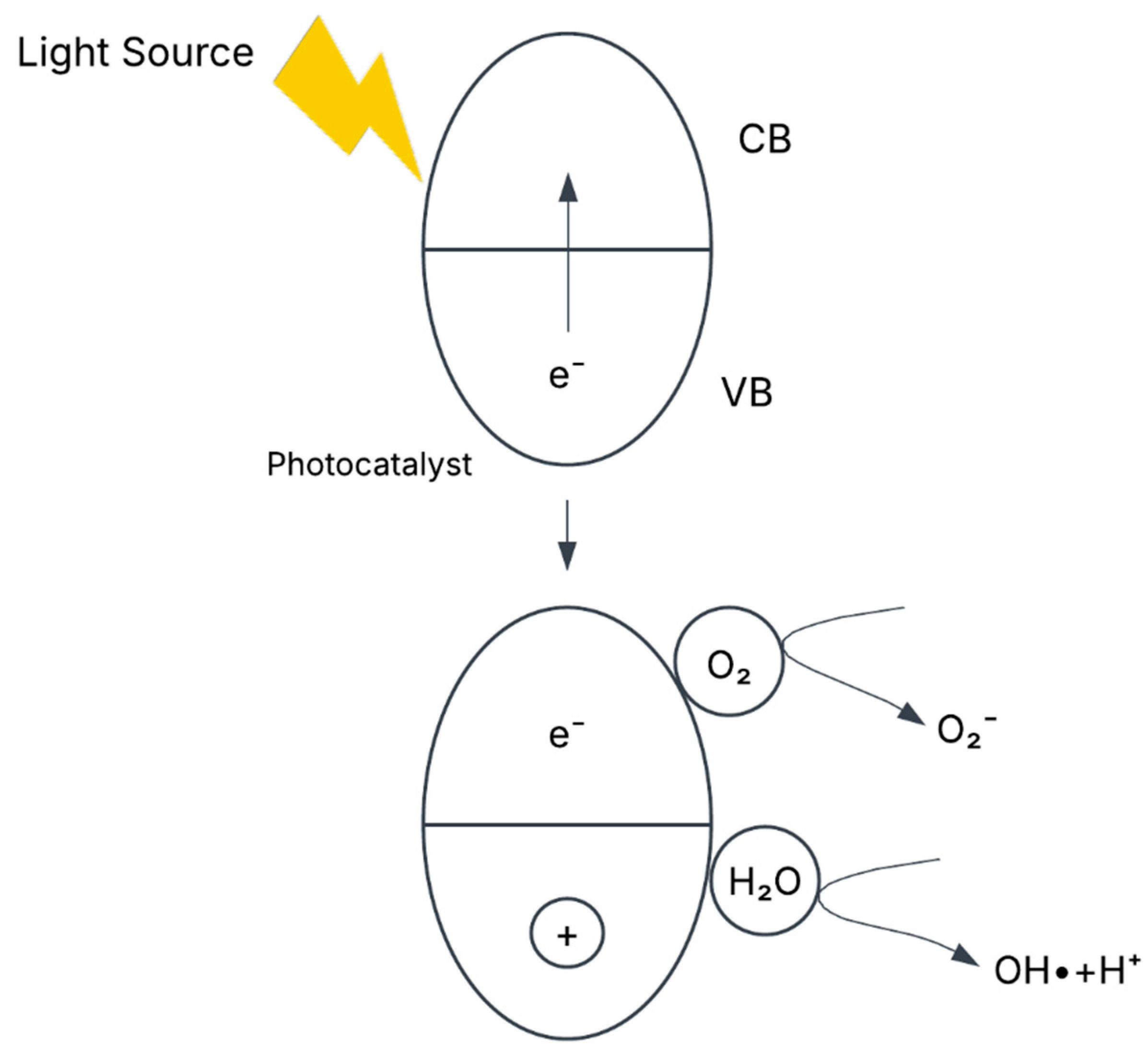
6.4.1. Mechanism of Photocatalytic Degradation
- Excitation of Photocatalyst
- 2.
- Oxidation of EtBr and Hydroxyl Radical Formation
- 3.
- Reduction of Oxygen and Formation of Reactive Oxygen Species (ROS)
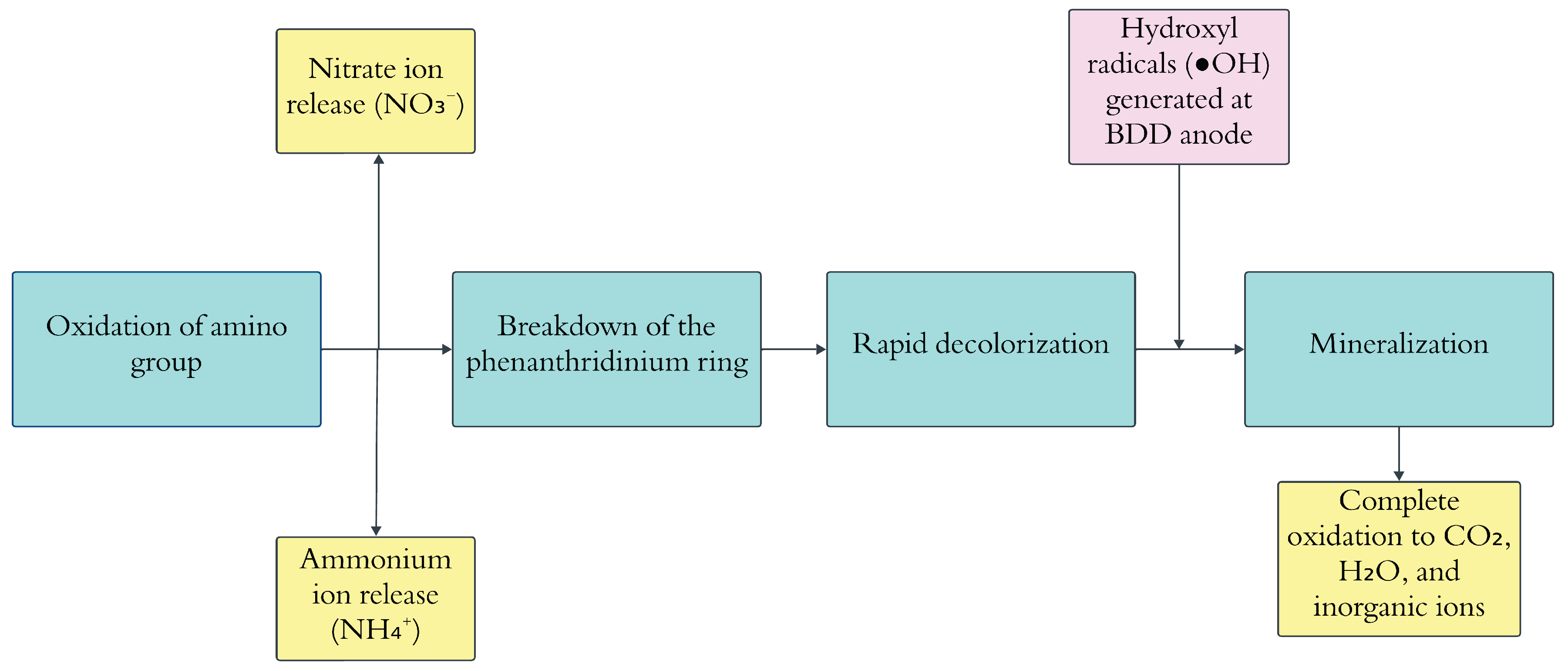
6.5. Electrochemical Degradation
6.6. Fenton-like Process Degradation
- Catalytic Decomposition of H2O2 on Iron Oxide Surface
- 2.
- Hydroxyl Radical Generation
- 3.
- Target Pollutant Oxidation
7. Future Directions
7.1. Mechanistic Elucidation at the Molecular Level
7.2. Long-Term Performance and Regeneration
7.3. Performance in Complex and Real Wastewater Matrices
7.4. Scalability and Economic Viability
7.5. Fate of Degradation Products and Environmental Safety
7.6. Design of Multifunctional and Hybrid Systems
8. Conclusion
Statement of Disclosure
Funding
Abbreviations
References
- Y. Wang et al., “Role of the sodium hydrogen exchanger in maitotoxin-induced cell death in cultured rat cortical neurons,” Elsevier, Accessed: Mar. 12, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/S0041010109001652.
- P. Scaria, R. S.-J. of B. Chemistry, and undefined 1991, “Binding of ethidium bromide to a DNA triple helix. Evidence for intercalation.,” Elsevier, Accessed: Mar. 12, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/S0021925819676118.
- Nafisi, A. Saboury, N. Keramat, … J. N.-J. of M., and undefined 2007, “Stability and structural features of DNA intercalation with ethidium bromide, acridine orange and methylene blue,” Elsevier, Accessed: Mar. 12, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/S0022286006004819.
- C. T. Mcmurray and K. E. Van Holde, “The binding of ethidium bromide to chromatin: model for carcinogen interactions,” 1987, Accessed: Mar. 12, 2025. [Online]. Available: https://ir.library.oregonstate.edu/concern/graduate_thesis_or_dissertations/vt150n10g.
- M. Hogan, N. Dattagupta, J. W.-J. of B. Chemistry, and undefined 1981, “Carcinogen-induced alteration of DNA structure.,” jbc.org, vol. 256, no. 9, pp. 4504–4613, 1981. [CrossRef]
- H. Hatami and M. Sieyahchehreh, “Investigating the effects of ethidium bromide on some hematological parameters in Cyprinus carpio.,” 2012, Accessed: Mar. 12, 2025. [Online]. Available: https://www.cabidigitallibrary.org/doi/full/10.5555/20143091472.
- “Ethidium Bromide – Laboratory Safety.” Accessed: Mar. 12, 2025. [Online]. Available: https://wp.stolaf.edu/chemical-hygiene/ethidium-bromide/.
- P. S. Sadalage and K. D. Pawar, “Adsorption and removal of ethidium bromide from aqueous solution using optimized biogenic catalytically active antibacterial palladium nanoparticles,” Environmental Science and Pollution Research, vol. 30, no. 2, pp. 5005–5026, Jan. 2023. [CrossRef]
- B. Heibati et al., “Adsorption of ethidium bromide (EtBr) from aqueous solutions by natural pumice and aluminium-coated pumice,” J Mol Liq, vol. 213, pp. 41–47, Jan. 2016. [CrossRef]
- R. Sulthana, S. N. Taqui, F. Zameer, U. T. Syed, and A. A. Syed, “Adsorption of ethidium bromide from aqueous solution onto nutraceutical industrial fennel seed spent: Kinetics and thermodynamics modeling studies,” Int J Phytoremediation, vol. 20, no. 11, pp. 1075–1086, Sep. 2018. [CrossRef]
- S. Novania, A. R. P. Widagdo, D. M. Prihatiningrum, S. Fauziyah, and T. H. Sucipto, “Ethidium Bromide Waste Treatment with Activated Charcoal,” EnvironmentAsia, vol. 16, no. 1, pp. 138–145, Jan. 2023. [CrossRef]
- S. A. Shaik, J. B. Shaik, and A. Goswami, “Ethidium bromide adsorption on pyrophyllite nanoclay: Insights from batch, thermodynamic, kinetic, and recyclability studies and optimization through response surface methodology,” Colloids Surf A Physicochem Eng Asp, vol. 692, p. 133900, Jul. 2024. [CrossRef]
- O. Moradi, A. Fakhri, S. Adami, and S. Adami, “Isotherm, thermodynamic, kinetics, and adsorption mechanism studies of Ethidium bromide by single-walled carbon nanotube and carboxylate group functionalized single-walled carbon nanotube,” J Colloid Interface Sci, vol. 395, pp. 224–229, Apr. 2013. [CrossRef]
- P.-H. Chang and B. Sarkar, “Mechanistic insights into ethidium bromide removal by palygorskite from contaminated water,” J Environ Manage, vol. 278, p. 111586, Jan. 2021. [CrossRef]
- P.-H. Chang et al., “Efficient ethidium bromide removal using sodium alginate/graphene oxide composite beads: Insights into adsorption mechanisms and performance,” Chemical Engineering Journal, vol. 500, p. 156379, Nov. 2024. [CrossRef]
- Z. Li, P. H. Chang, W. T. Jiang, and Y. Liu, “Enhanced removal of ethidium bromide (EtBr) from aqueous solution using rectorite,” J Hazard Mater, vol. 384, p. 121254, Feb. 2020. [CrossRef]
- Z. Ge, T. Sun, J. Xing, and X. Fan, “Efficient removal of ethidium bromide from aqueous solution by using DNA-loaded Fe 3 O 4 nanoparticles,” Environmental Science and Pollution Research, vol. 26, no. 3, pp. 2387–2396, Jan. 2019. [CrossRef]
- P. H. Chang et al., “Efficient ethidium bromide removal using sodium alginate/graphene oxide composite beads: Insights into adsorption mechanisms and performance,” Chemical Engineering Journal, vol. 500, p. 156379, Nov. 2024. [CrossRef]
- L. Wang, Z. Li, X. Zhang, G. Lv, and X. Wang, “High capacity ethidium bromide removal by montmorillonites,” Korean Journal of Chemical Engineering, vol. 37, no. 12, pp. 2202–2208, Dec. 2020. [CrossRef]
- C. Zhang, L. Liu, J. Wang, F. Rong, and D. Fu, “Electrochemical degradation of ethidium bromide using boron-doped diamond electrode,” Sep Purif Technol, vol. 107, pp. 91–101, Apr. 2013. [CrossRef]
- E. Xie, L. Zheng, A. Ding, and D. Zhang, “Mechanisms and pathways of ethidium bromide Fenton-like degradation by reusable magnetic nanocatalysts,” Chemosphere, vol. 262, p. 127852, Jan. 2021. [CrossRef]
- C. Zhang, L. Liu, J. Wang, F. Rong, and D. Fu, “Electrochemical degradation of ethidium bromide using boron-doped diamond electrode,” Sep Purif Technol, vol. 107, pp. 91–101, Apr. 2013. [CrossRef]
- B. Heibati et al., “Adsorption of ethidium bromide (EtBr) from aqueous solutions by natural pumice and aluminium-coated pumice,” J Mol Liq, vol. 213, pp. 41–47, Jan. 2016. [CrossRef]
- C. Adán, A. Martínez-Arias, M. Fernández-García, and A. Bahamonde, “Photocatalytic degradation of ethidium bromide over titania in aqueous solutions,” Appl Catal B, vol. 76, no. 3–4, pp. 395–402, Nov. 2007. [CrossRef]
- P. Sukhumungoon, P. Rattanachuay, F. Hayeebilan, and D. Kantachote, “Biodegradation of ethidium bromide by Bacillus thuringiensis isolated from soil,” Afr J Microbiol Res, vol. 7, no. 6, pp. 471–476, 2013.
- V. Gandhi, K. Kesari, A. K.- BioTech, and undefined 2022, “The identification of ethidium bromide-degrading bacteria from laboratory gel electrophoresis waste,” mdpi.comVP Gandhi, KK Kesari, A KumarBioTech, 2022•mdpi.com, 2022. [CrossRef]
- S. Amirijavid, M. Chizari, M. S.-I. J. P. B. Res, and undefined 2014, “Phytoremediation of ethidium bromide by tomato and alfalfa plants,” researchgate.netS Amirijavid, M Chizari, M SadrzadehInt J Plant Biol Res, 2014•researchgate.net, Accessed: Mar. 12, 2025. [Online]. Available: https://www.researchgate.net/profile/Shaghayegh-Amirijavid/publication/275520986_Phytoremediation_of_Ethidium_Bromide_by_Tomato_and_Alfalfa_Plants/links/553e587c0cf294deef7096c2/Phytoremediation-of-Ethidium-Bromide-by-Tomato-and-Alfalfa-Plants.pdf.
- H. Cavalcante de Almeida, A. Luís de Sá Salomão, J. Lambert, L. Cardoso Rocha Saraiva Teixeira, M. Marques, and A. S. Lu ıs de Salom, “Phycoremediation potential of microalgae species for ethidium bromide removal from aqueous media,” Taylor & FrancisHC de Almeida, ALS Salomão, J Lambert, LCRS Teixeira, M MarquesInternational Journal of Phytoremediation, 2020•Taylor & Francis, vol. 22, no. 11, pp. 1168–1174, Sep. 2020. [CrossRef]
- A. Kumar, P. Swarupa, … V. G.-I. J. of, and undefined 2017, “Isolation of ethidium bromide degrading bacteria from Jharkhand,” drive.google.com, Accessed: Mar. 12, 2025. [Online]. Available: https://drive.google.com/file/d/1nPyZDWpkSWorOVxQFF0sycU2dllMsUzB/view.
- V. Jhalora, S. Mathur, R. B.-R. J. of Pharmacy, and undefined 2024, “Screening and Evaluation of Biodegradation Potential of Bacterial Isolates Against Ethidium Bromide,” researchgate.netV Jhalora, S Mathur, R BistResearch Journal of Pharmacy and Technology, 2024•researchgate.net, Accessed: Mar. 12, 2025. [Online]. Available: https://www.researchgate.net/profile/Vandana-Jhalora/publication/381519726_Screening_and_Evaluation_of_Biodegradation_Potential_of_Bacterial_Isolates_Against_Ethidium_Bromide/links/66726b6285a4ee7261d109e8/Screening-and-Evaluation-of-Biodegradation-Potential-of-Bacterial-Isolates-Against-Ethidium-Bromide.pdf.
- K. L.-S. Visualization and undefined 2016, “The making of modern biotechnology: how ethidium bromide made fame,” sciencevision.orgK LalchhandamaScientific Visualization, 2016•sciencevision.org, Accessed: Mar. 12, 2025. [Online]. Available: https://sciencevision.org/storage/journal-articles/February2019/Qqzr9WRH7oWA55dcup5h.
- M. Waring, “Ethidium and Propidium,” Mechanism of Action of Antimicrobial and Antitumor Agents, pp. 141–165, 1975. [CrossRef]
- S. Thititananukij, R. Vejaratpimol, T. Pewnim, and A. W. Fast, “Ethidium bromide nuclear staining and fluorescence microscopy: An alternative method for triploidy detection in fish,” J World Aquac Soc, vol. 27, no. 2, pp. 213–217, 1996. [CrossRef]
- V. Singer, T. Lawlor, S. Y.-M. R. T. and, and undefined 1999, “Comparison of SYBR® Green I nucleic acid gel stain mutagenicity and ethidium bromide mutagenicity in the Salmonella/mammalian microsome reverse,” Elsevier, Accessed: Mar. 12, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/S1383571898001727.
- P. H. Chang, Z. Li, and W. T. Jiang, “Mechanisms of ethidium bromide removal by Ca-montmorillonite,” Desalination Water Treat, vol. 254, pp. 80–93, Apr. 2022. [CrossRef]
- “Ethidium Bromide | C21H20BrN3 | CID 14710 - PubChem.” Accessed: Mar. 12, 2025. [Online]. Available: https://pubchem.ncbi.nlm.nih.gov/compound/14710.
- T. Watkins, G. W.- Nature, and undefined 1952, “Effect of changing the quaternizing group on the trypanocidal activity of dimidium bromide,” nature.comTI Watkins, G WoolfeNature, 1952•nature.com, Accessed: Mar. 13, 2025. [Online]. Available: https://www.nature.com/articles/169506a0.
- F. Hawking, “Drug-resistance of trypanosoma congolense and other trypanosomes to quinapyramine, phenanthridines, berenil and other compounds in mice,” Ann Trop Med Parasitol, vol. 57, no. 3, pp. 262–282, 1963. [CrossRef]
- R. G. Pegram and J. M. Scott, “The prevalence and diagnosis of Trypanosoma evansi infection in camels in southern Ethiopia,” Trop Anim Health Prod, vol. 8, no. 1, pp. 20–27, Dec. 1976. [CrossRef]
- T. Leach, C. R.-P. & therapeutics, and undefined 1981, “Present status of chemotherapy and chemoprophylaxis of animal trypanosomiasis in the eastern hemisphere,” ElsevierTM Leach, CJ RobertsPharmacology & therapeutics, 1981•Elsevier, Accessed: Mar. 13, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/0163725881900693.
- J. B. Lepecq and C. Paoletti, “A fluorescent complex between ethidium bromide and nucleic acids: Physical—Chemical characterization,” J Mol Biol, vol. 27, no. 1, pp. 87–106, Jul. 1967. [CrossRef]
- V. W. F. Burns, “Fluorescence decay time characteristics of the complex between ethidium bromide and nucleic acids,” Arch Biochem Biophys, vol. 133, no. 2, pp. 420–424, Sep. 1969. [CrossRef]
- B. Hudson and R. Jacobs, “The ultraviolet transitions of the ethidium cation,” Biopolymers, vol. 14, no. 6, pp. 1309–1312, Jun. 1975. [CrossRef]
- S. Ramotowska, P. Spisz, J. Brzeski, A. Ciesielska, and M. Makowski, “Application of the SwitchSense Technique for the Study of Small Molecules’ (Ethidium Bromide and Selected Sulfonamide Derivatives) Affinity to DNA in Real Time,” Journal of Physical Chemistry B, vol. 126, no. 38, pp. 7238–7251, Sep. 2022. [CrossRef]
- J. Olmsted and D. R. Kearns, “Mechanism of Ethidium Bromide Fluorescence Enhancement on Binding to Nucleic Acids,” Biochemistry, vol. 16, no. 16, pp. 3647–3654, Aug. 1977. [CrossRef]
- T. Ohta, S. I. Tokishita, and H. Yamagata, “Ethidium bromide and SYBR Green I enhance the genotoxicity of UV-irradiation and chemical mutagens in E. coli,” Mutation Research/Genetic Toxicology and Environmental Mutagenesis, vol. 492, no. 1–2, pp. 91–97, May 2001. [CrossRef]
- R. Y. Ouchi, A. J. Manzato, C. R. Ceron, and G. O. Bonilla-Rodriguez, “Evaluation of the effects of a single exposure to ethidium bromide in Drosophila melanogaster (Diptera-Drosophilidae),” Bull Environ Contam Toxicol, vol. 78, no. 6, pp. 489–493, Jun. 2007. [CrossRef]
- B. A. NEWTON, “The mode of action of phenanthridines: the effect of ethidium bromide on cell division and nucleic acid synthesis,” J Gen Microbiol, vol. 17, no. 3, pp. 718–730, Dec. 1957. [CrossRef]
- R. TOMCHICK and H. G. MANDEL, “BIOCHEMICAL EFFECTS OF ETHIDIUM BROMIDE IN MICRO-ORGANISMS.,” J Gen Microbiol, vol. 36, no. 2, pp. 225–236, Aug. 1964. [CrossRef]
- V. Vacquier, J. B.- Nature, and undefined 1969, “Chromosomal abnormalities resulting from ethidium bromide treatment,” nature.comVD Vacquier, J BrachetNature, 1969•nature.com, Accessed: Mar. 21, 2025. [Online]. Available: https://www.nature.com/articles/222193a0.
- S. Singh and A. Singh, “Ethidium bromide: Is a stain turning into a pollutant? A synthesis on its status, waste management, monitoring challenges and ecological risks to the environment Issue 4 IJRAR1904431,” Oct. 2018.
- Y. Naum, D. P.-E. C. Research, and undefined 1971, “Reversible inhibition of cytochrome oxidase accumulation in human cells by ethidium bromide,” Elsevier, Accessed: Mar. 21, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/0014482771900103.
- N. von Wurmb-Schwark, … L. C.-… M. M. of, and undefined 2006, “A low dose of ethidium bromide leads to an increase of total mitochondrial DNA while higher concentrations induce the mtDNA 4997 deletion in a human,” Elsevier, Accessed: Mar. 21, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/S0027510705005336.
- J. McCann and B. N. Ames, “The Salmonella/microsome mutagenicity test: Predictive value for animal carcinogenicity,” Origins of Human Cancer, vol. 4, pp. 1431–1450, 1977.
- R. Ouchi, A. Manzato, … C. C.-B. of environmental, and undefined 2007, “Evaluation of the Effects of a Single Exposure to Ethidium Bromide in Drosophila melanogaster (Diptera-Drosophilidae),” SpringerRY Ouchi, AJ Manzato, CR Ceron, GO Bonilla-RodriguezBulletin of environmental contamination and toxicology, 2007•Springer, vol. 78, no. 6, pp. 489–493, Jun. 2007. [CrossRef]
- Z. Jiang, J. Li, G. Huang, L. Yan, and J. Ma, “Efficient removal of ethidium bromide from aqueous solutions using chromatin-loaded chitosan polyvinyl alcohol composites,” Environ Sci Pollut Res Int, vol. 31, no. 2, pp. 3276–3295, Jan. 2024. [CrossRef]
- R. Sulthana, S. N. Taqui, F. Zameer, U. T. Syed, and A. A. Syed, “Adsorption of ethidium bromide from aqueous solution onto nutraceutical industrial fennel seed spent: Kinetics and thermodynamics modeling studies,” Int J Phytoremediation, vol. 20, no. 11, pp. 1075–1086, Sep. 2018. [CrossRef]
- B. Heibati et al., “Adsorption of ethidium bromide (EtBr) from aqueous solutions by natural pumice and aluminium-coated pumice,” J Mol Liq, vol. 213, pp. 41–47, Jan. 2016. [CrossRef]
- E. Xie, L. Zheng, A. Ding, and D. Zhang, “Mechanisms and pathways of ethidium bromide Fenton-like degradation by reusable magnetic nanocatalysts,” Chemosphere, vol. 262, p. 127852, Jan. 2021. [CrossRef]
- G. Crini and E. Lichtfouse, “Advantages and disadvantages of techniques used for wastewater treatment,” Environ Chem Lett, vol. 17, no. 1, pp. 145–155, Mar. 2019. [CrossRef]
- B. Sivasankar, Biosperations: Principles and Techniques. PHI Learning Pvt. Ltd., 2005.
- G. S. Simate, N. Maledi, A. Ochieng, S. Ndlovu, J. Zhang, and L. F. Walubita, “Coal-based adsorbents for water and wastewater treatment,” J Environ Chem Eng, vol. 4, no. 2, pp. 2291–2312, Jun. 2016. [CrossRef]
- Saruchi, R. Verma, V. Kumar, and A. A. ALOthman, “Comparison between removal of Ethidium bromide and eosin by synthesized manganese (II) doped zinc (II) sulphide nanoparticles: kinetic, isotherms and thermodynamic studies,” J Environ Health Sci Eng, vol. 18, no. 2, pp. 1175–1187, Dec. 2020. [CrossRef]
- A. E.-E. H. Gad, … A. Y.-E. J. of, and undefined 2020, “Synthesis of high efficient CS/PVDC/TiO2-Au nanocomposites for photocatalytic degradation of carcinogenic ethidium bromide in sunlight,” ejchem.journals.ekb.eg, Accessed: Mar. 12, 2025. [Online]. Available: https://ejchem.journals.ekb.eg/article_69527.
- A. R. Kumar et al., “A state of the art review on the cultivation of algae for energy and other valuable products: Application, challenges, and opportunities,” Renewable and Sustainable Energy Reviews, vol. 138, p. 110649, Mar. 2021. [CrossRef]
- Lavand, Y. M.-J. of T. A. and Calorimetry, and undefined 2016, “Visible-light photocatalytic degradation of ethidium bromide using carbon- and iron-modified TiO2 photocatalyst,” SpringerAB Lavand, YS MalgheJournal of Thermal Analysis and Calorimetry, 2016•Springer, vol. 123, no. 2, pp. 1163–1172, Feb. 2016. [CrossRef]
- Lavand, Y. M.-J. of T. A. and Calorimetry, and undefined 2016, “Visible-light photocatalytic degradation of ethidium bromide using carbon- and iron-modified TiO2 photocatalyst,” SpringerAB Lavand, YS MalgheJournal of Thermal Analysis and Calorimetry, 2016•Springer, vol. 123, no. 2, pp. 1163–1172, Feb. 2016. [CrossRef]
- J. Carbajo, C. Adán, A. Rey, … A. M.-A.-A. C. B., and undefined 2011, “Optimization of H2O2 use during the photocatalytic degradation of ethidium bromide with TiO2 and iron-doped TiO2 catalysts,” Elsevier, Accessed: Mar. 12, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/S0926337310005163.
- S. Swetha, R. B.-C. J. of Catalysis, and undefined 2011, “Preparation and characterization of high activity zirconium-doped anatase titania for solar photocatalytic degradation of ethidium bromide,” ElsevierS Swetha, RG BalakrishnaChinese Journal of Catalysis, 2011•Elsevier, Accessed: Mar. 12, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/S1872206710602008.
- Adán, A. Martínez-Arias, … M. F.-G.-A. C. B., and undefined 2007, “Photocatalytic degradation of ethidium bromide over titania in aqueous solutions,” ElsevierC Adán, A Martínez-Arias, M Fernández-García, A BahamondeApplied Catalysis B: Environmental, 2007•Elsevier, Accessed: Mar. 12, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/S0926337307001907.
- S. Singh, V. Srivastava, I. M.-T. J. of Physical, and undefined 2013, “Mechanism of dye degradation during electrochemical treatment,” ACS Publications, vol. 117, no. 29, pp. 15229–15240, Jul. 2013. [CrossRef]
- Y. Zhu, W. Fan, W. Feng, Y. Wang, … S. L.-J. of H., and undefined 2021, “A critical review on metal complexes removal from water using methods based on Fenton-like reactions: Analysis and comparison of methods and,” Elsevier, Accessed: Mar. 12, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/S0304389421004805.
- A. Shokri, M. F.-E. Challenges, and undefined 2022, “A critical review in Fenton-like approach for the removal of pollutants in the aqueous environment,” Elsevier, Accessed: Mar. 12, 2025. [Online]. Available: https://www.sciencedirect.com/science/article/pii/S2667010022000920.
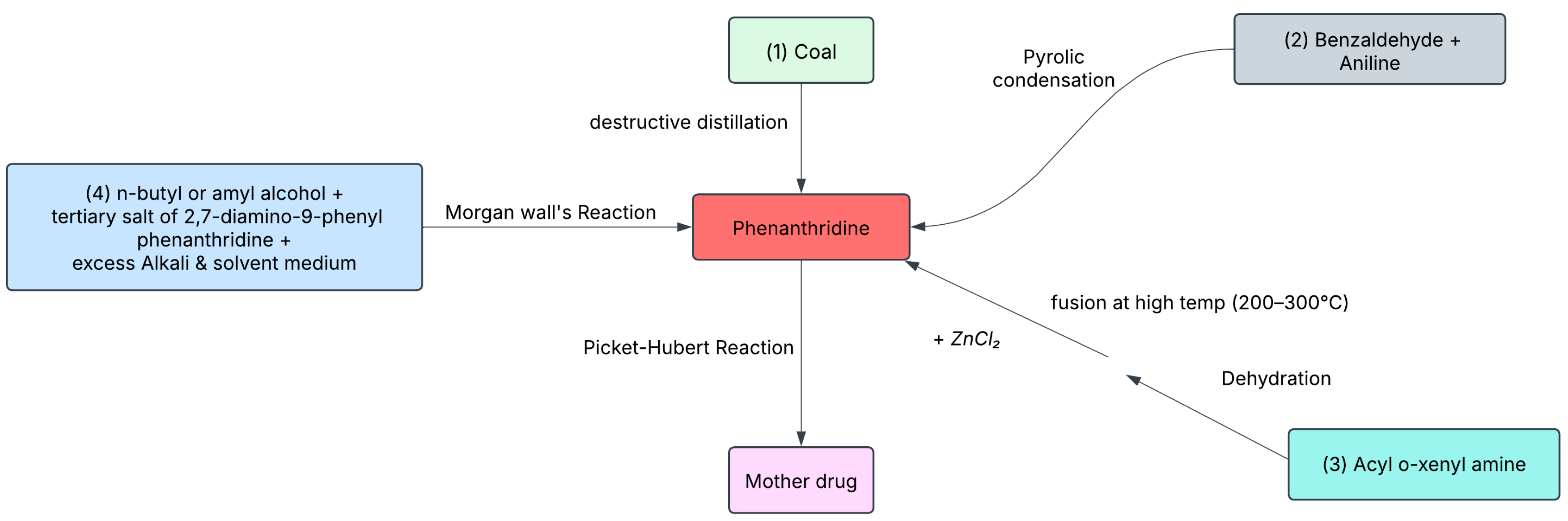
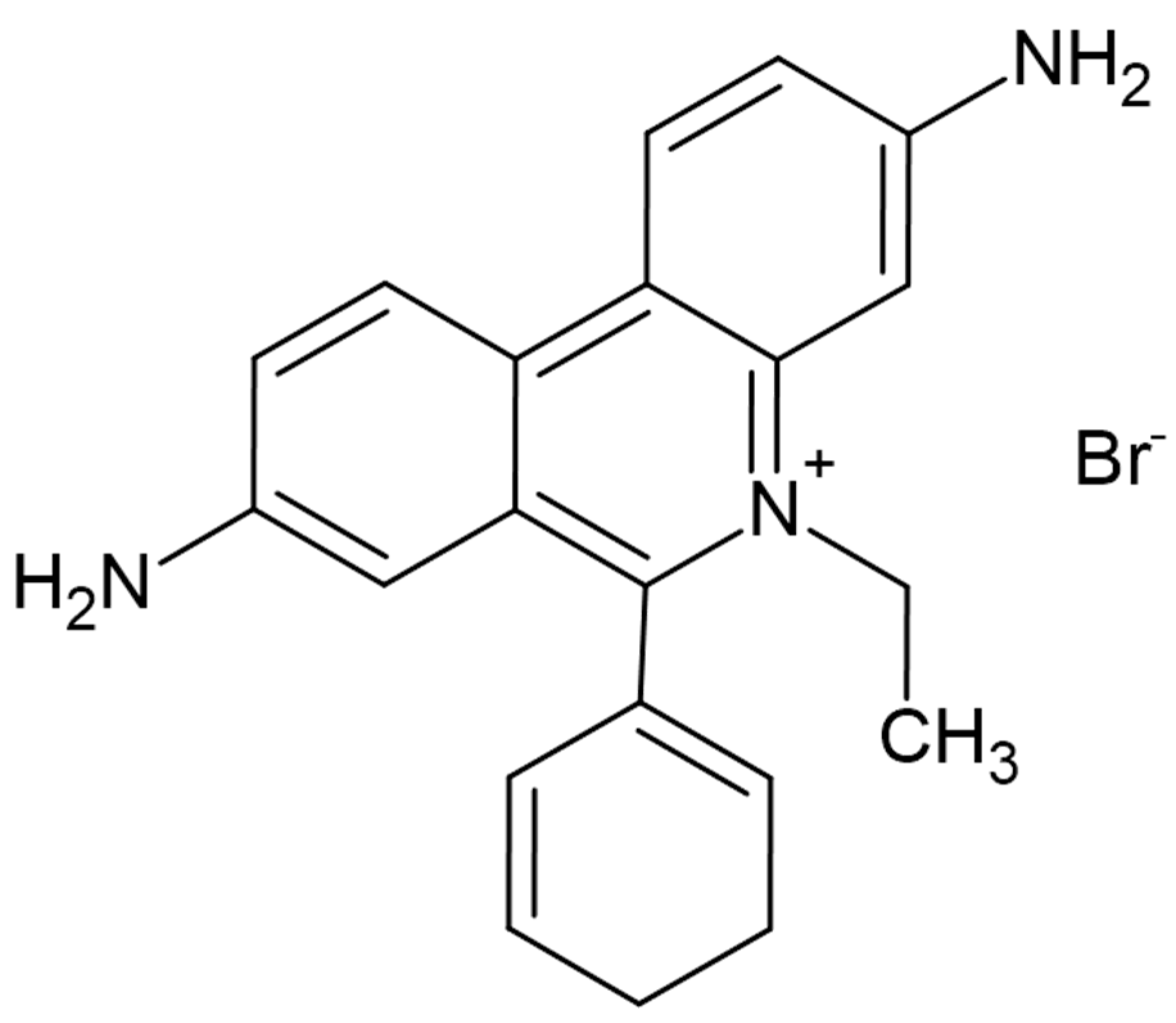
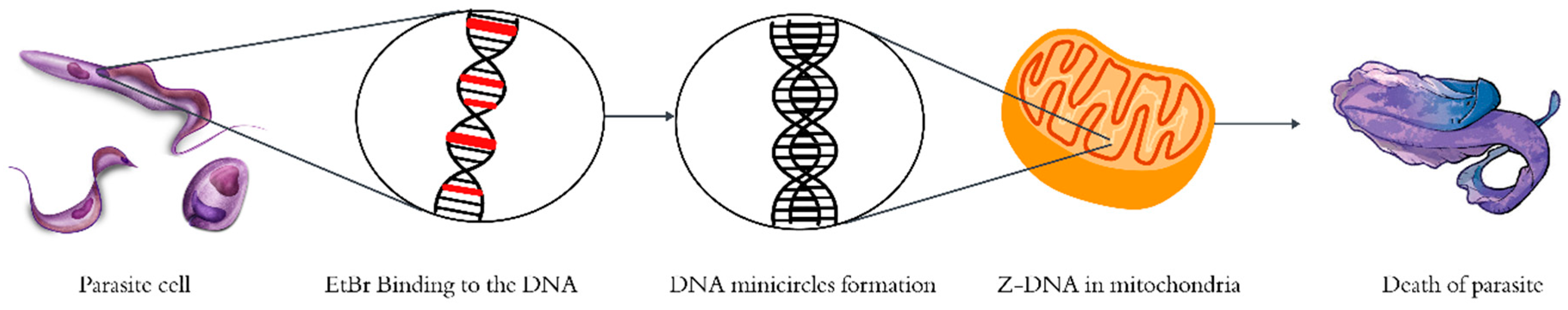
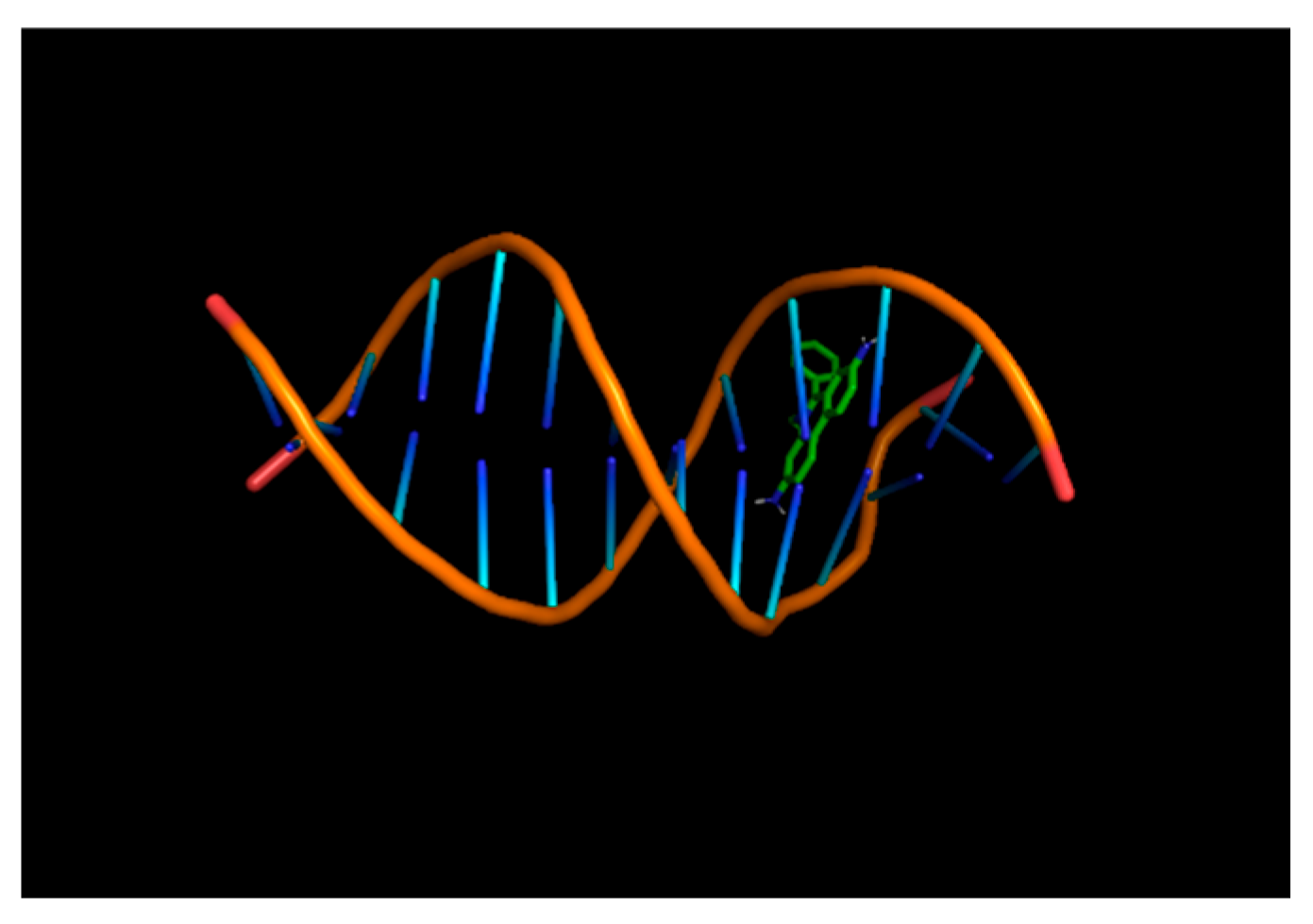
| Year | Description |
|---|---|
| 1891 | Phenanthridine Synthesis: Pictet and Ankersmit synthesize phenanthridine (EtBr precursor) from coal tar using benzaldehyde and aniline. |
| 1930s | Medical Applications: Phenanthridine derivatives, including EtBr, are developed to treat trypanosomiasis. |
| 1950s | Veterinary Use: EtBr is marketed under the trade name Homidium as an antiprotozoal agent for treating trypanosomiasis in cattle. |
| 1952 | EtBr Synthesis: Scientists at Boots Pure Drug Co., UK, synthesize EtBr by replacing a methyl group with an ethyl group in dimidium bromide. |
| 1962 | DNA Inhibition Study: Kandaswamy and Henderson observe inhibition of DNA synthesis in Strigomonas oncopelti using EtBr. |
| 1964 | Intercalation Discovery: Michael Waring uses X-ray diffraction and molecular modeling to demonstrate EtBr’s DNA intercalation mechanism. |
| 1965 | DNA Binding Confirmation: Waring’s further studies confirm the strong binding affinity between EtBr and nucleic acids. |
| 1967 | Parasiticidal Activity: Guy Riou shows EtBr’s inhibition of kinetoplast DNA (kDNA) in trypanosomes, reinforcing its antiparasitic effects. |
| 1972 | Gel Electrophoresis Application: Piet Borst and Cees Aaij successfully use EtBr in ethidium-agarose electrophoresis to detect mitochondrial DNA topoisomers. |
| 1973 | SV40 Virus Research: Philip A. Sharp’s lab at Cold Spring Harbor uses EtBr to stain DNA fragments in SV40 virus research. |
| 1980s | Resistance Issues: Resistance to EtBr emerges in Trypanosoma congolense, leading to its decline as a veterinary drug. |
| 1990s | Widespread Lab Use: EtBr becomes the most widely used nucleic acid stain for DNA visualization in molecular biology research. |
| 2000s-present | Safety Concerns and Alternatives: Due to its mutagenic and carcinogenic nature, alternatives are sought, but EtBr remains in use due to its effectiveness and affordability. |
| Parameter | Value |
|---|---|
| IUPAC Name | 3,8-Diamino-5-ethyl-6-phenylphenanthridinium bromide |
| Molecular Formula | C21H20BrN₃ |
| Molecular Weight | 394.31 g/mol |
| Melting Point | ~260°C (decomposes) |
| Flash Point | Not available (non-volatile solid) |
| Solubility | Soluble in water, ethanol, and DMSO |
| Colour | Dark red or purple solid |
| Taste | Bitter (but highly toxic, should not be tasted) |
| Density | ~1.0–1.4 g/cm³ (approximate) |
| Vapour Pressure | Negligible (solid at room temperature) |
| Sno | Adsorbent used | pH | Temperature (°C) | Adsorption Capacity (mg/g) | Isotherm Model | Kinetic Model | Process Type | Reference |
|---|---|---|---|---|---|---|---|---|
| 1 | palladium nanoparticles | 8 | 47 | - | Freundlich and Temkin isotherm models | pseudo-second-order kinetic model | Non-spontaneous and endothermic | [8] |
| 2 | Activated Charcoal | 8 | 27±2 | - | - | - | - | [11] |
| 3 | fennel seed spent | 2 | 50 | 79.76 | Vieth–Sladek | pseudo-first-order kinetic model | endothermic andalmost spontaneous | [57] |
| 4 | natural pumice and aluminium-coated pumice | 8 | 25 ± 1 | NP: 58.82 ACP: 76.92 | Freundlich isotherm model | pseudo-second-order kinetics model | - | [23] |
| 5 | pyrophyllite nanoclay | 6.8 | 27±2 | 6.53 | Freundlich adsorption isotherm | pseudo-second-order kinetics model | spontaneous, exothermic and entropy-driven | [12] |
| 6 | single-walled carbon nanotube and carboxylate group functionalized single-walled carbon nanotube | - | - | - | Langmuir isotherm model | intra-particle diffusion | endothermic | [13] |
| 7 | palygorskite | ~8 | - | 112.37 | Freundlich isotherm model | pseudo- second-order kinetic model | exothermic | [14] |
| 8 | sodium alginate/graphene oxide composite beads | 11 | - | GO: 1068.55 SA: 449.50 | FreundlichIsotherm model | pseudo-second-order kinetic model | - | [18] |
| 9 | Manganese (II) doped zinc (II) sulphide nanoparticles | 9 | 45 | Langmuir isotherm model | Intra particle diffusion | [63] |
| Photocatalyst | Doping | Synthesis method | Light source | Time (min) | Reference |
|---|---|---|---|---|---|
| TiO2 | Au | Hydrothermal treatment | Sunlight | 120 | [64] |
| TiO2 | - | Combined sol–gel/ reverse microemulsion method | High-pressure Hg lamp (500W) | 30-60 | [70] |
| TiO2 | C and Fe | Microemulsion method | 65 W compact fluorescent lamp | 90 | [66] |
| TiO2 | Zr | Sol-Gel Process | Sunlight | 120 | [69] |
| TiO2 | Fe | Combined sol–gel/ reverse microemulsion method | High-pressure Hg lamp (500W) | >60 | [68] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




