Submitted:
12 May 2025
Posted:
14 May 2025
You are already at the latest version
Abstract
Keywords:
Introduction
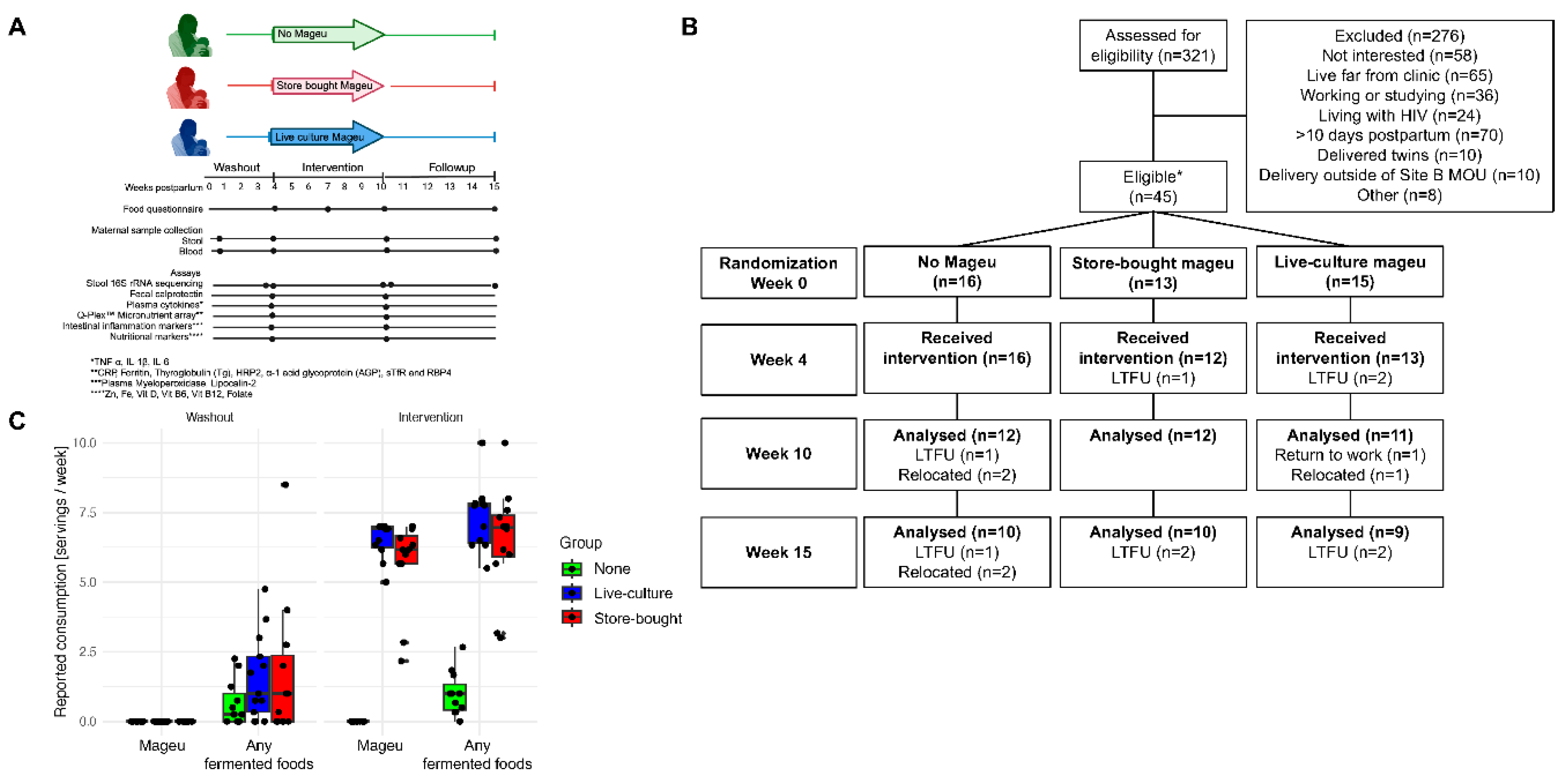
Methods
Results
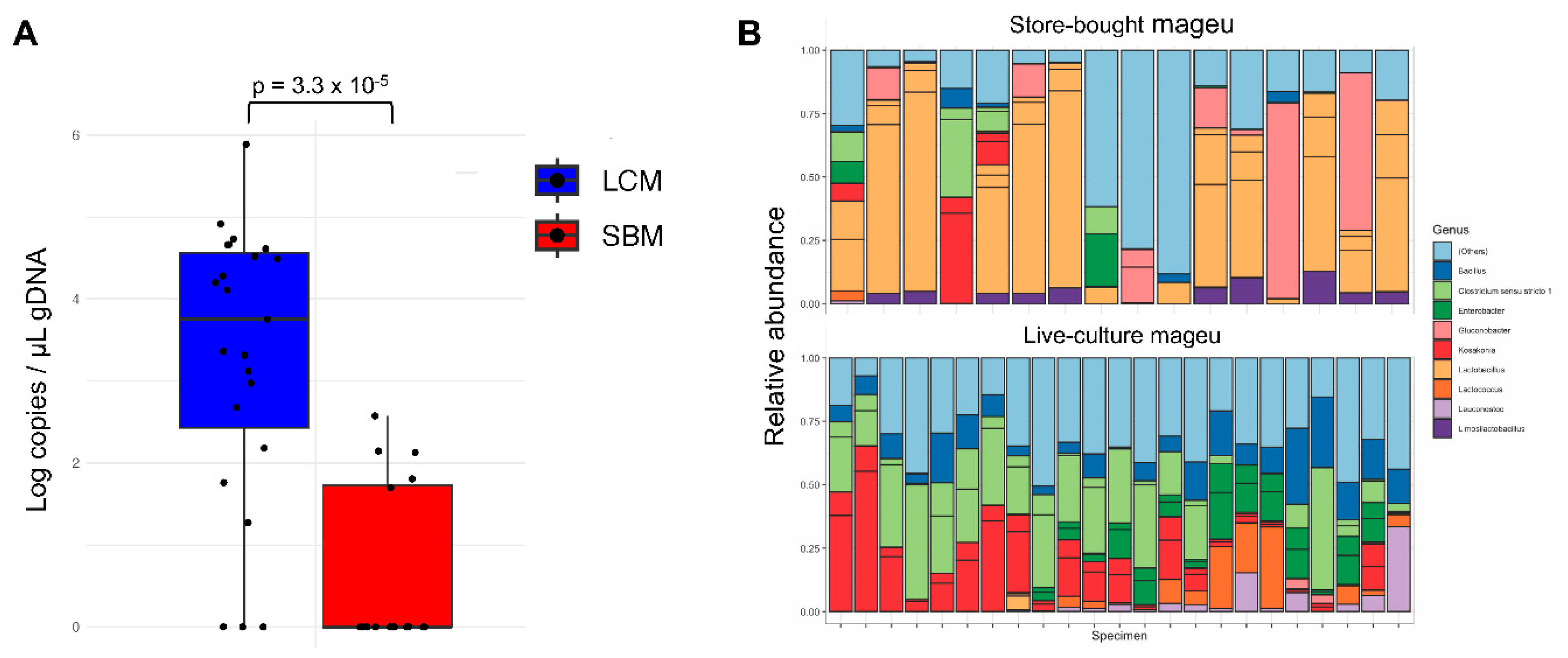
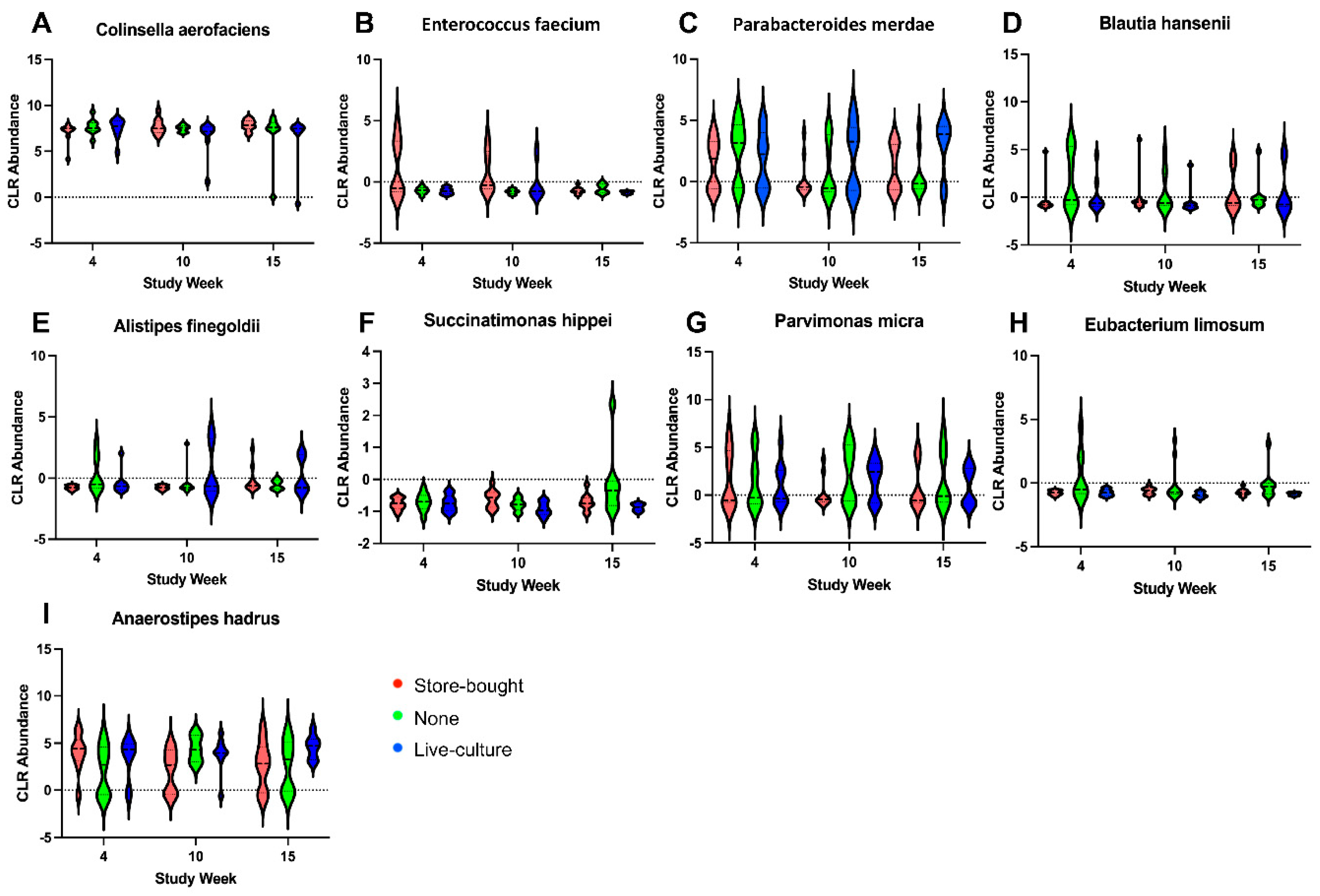
LCM Had a Higher Bacterial Load and a More Complex Bacterial Community Than SBM.
Consumption of Both SBM or LCM Was Generally Safe and Did Not Affect Maternal BMI.
Mageu Consumption Resulted in Higher Plant Protein Intake.
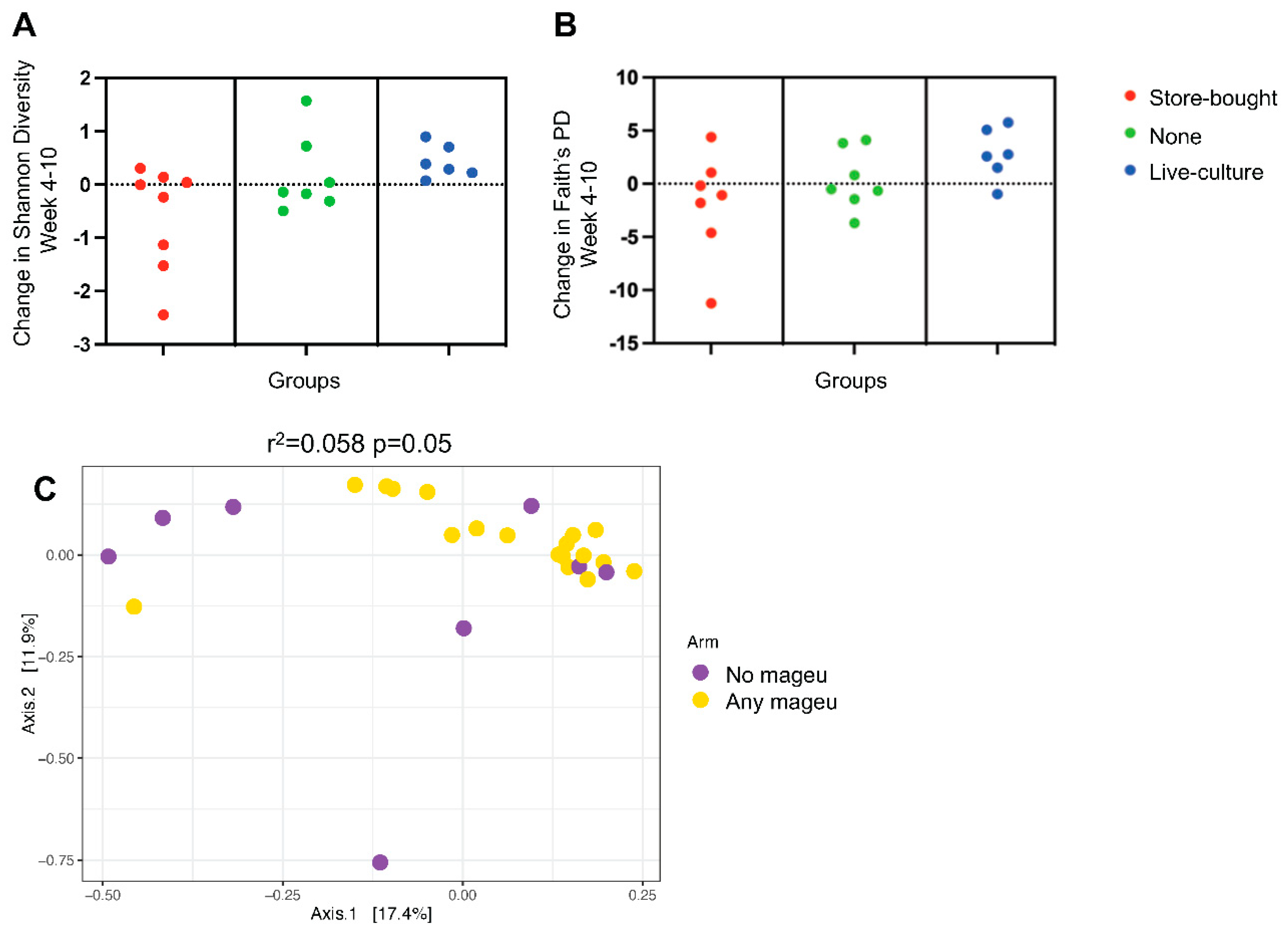
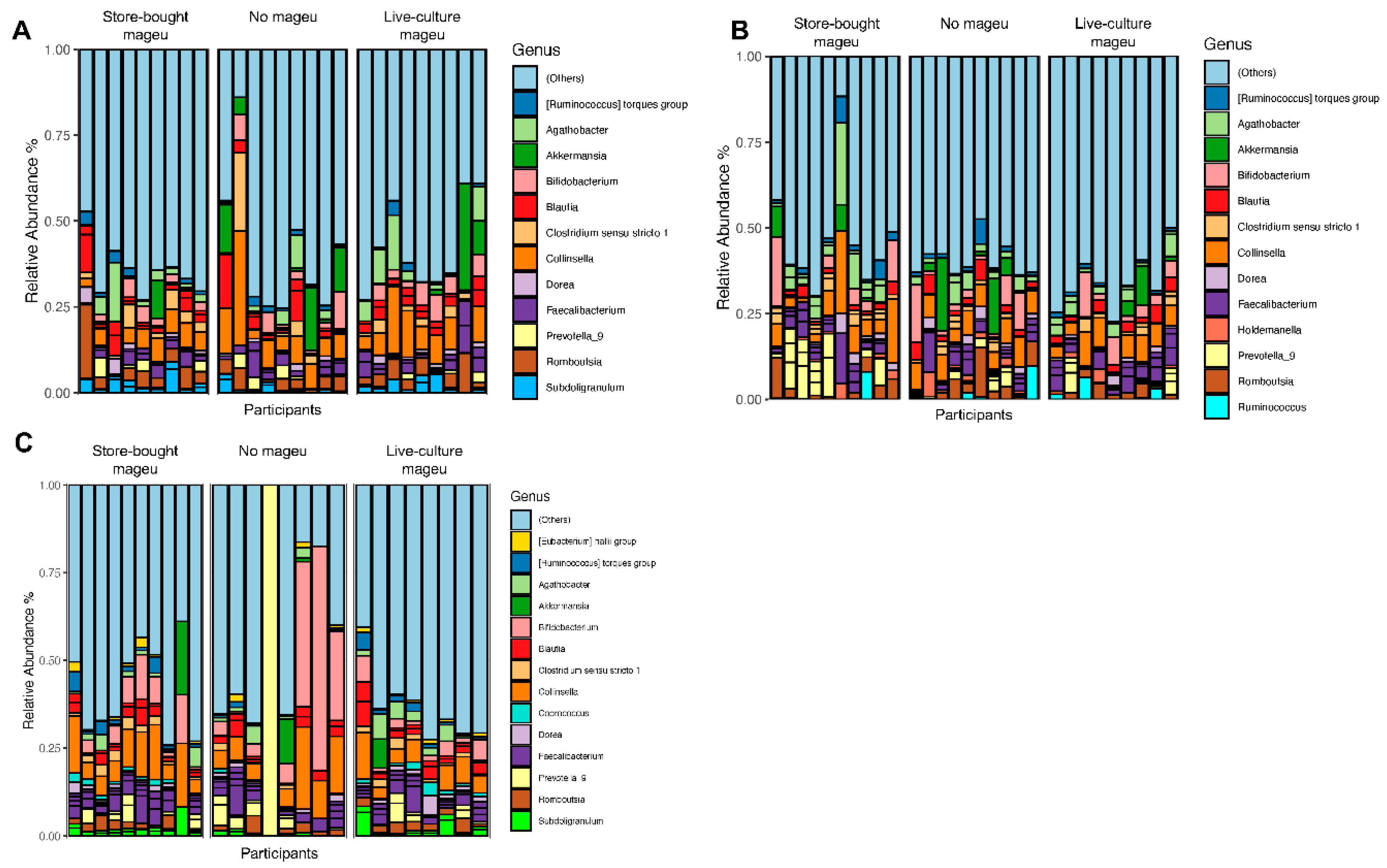
LCM Increased Shannon Gut Microbiota Diversity While SBM and no Mageu Did Not.
LCM Did Not Affect Host Inflammatory Markers But Decreased Circulating Ferritin Compared to no Mageu Users.
Mageu Use Did Not Affect Iron and Nutritional Markers.
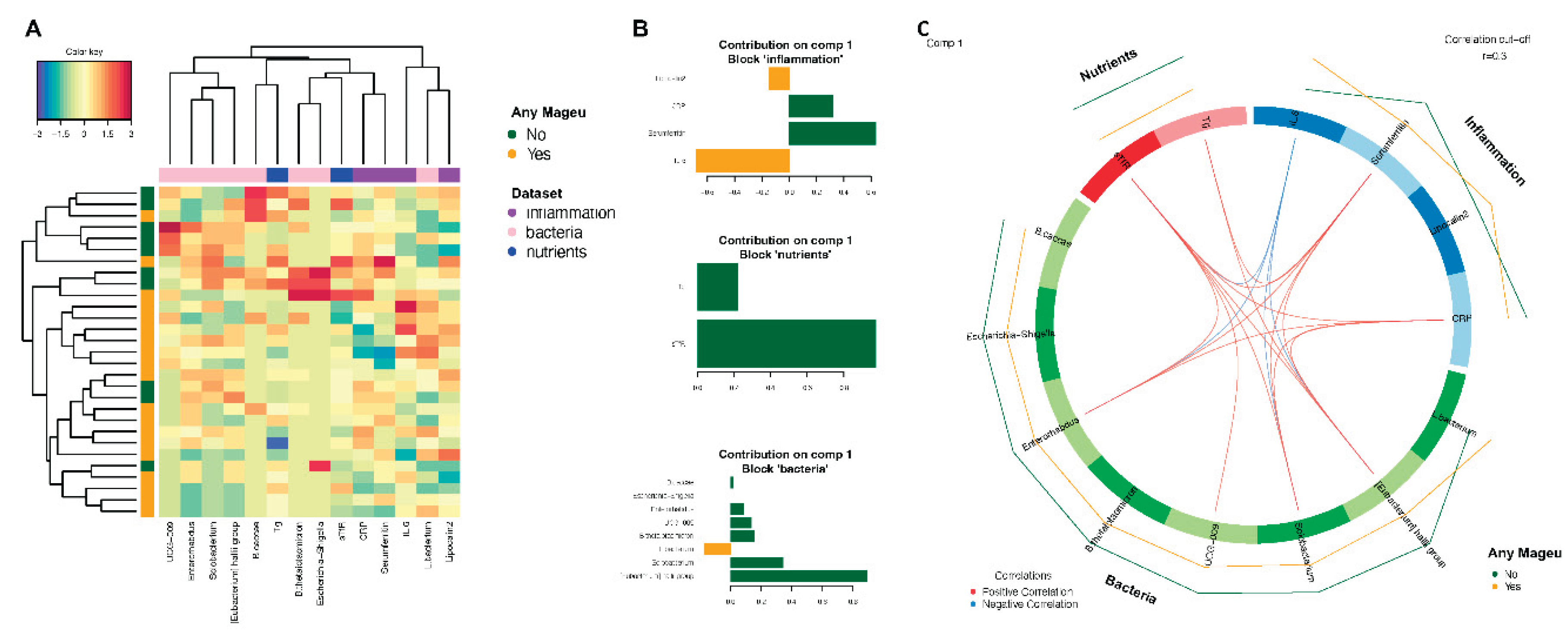
System Analysis Revealed Bacterial, Inflammation, and Nutritional Signatures Unique to Women Randomized to Mageu Compared to No Mageu.
Discussion
Supplementary Materials
Author Contributions
Financial Support
Availability of Data and Materials
Acknowledgements
Declaration of Interests
References
- Nnam, N. M. Improving maternal nutrition for better pregnancy outcomes. Proc Nutr Soc 74, 454–9 (2015). [CrossRef]
- National Department of Health (NDoH), Statistics South Africa (Stats SA) & South African Medical Research Council (SAMRC). South Africa Demographic and Health Survey. (2016).
- Stats SA. General Household Survey 2021 . (2022).
- Labadarios, D., Shisana, O., Rehle, T. & Simbayi, L. SANHANES: a unique survey series in the health landscape. S Afr Med J 104, 675–6 (2014). [CrossRef]
- Rothschild, D. et al. Environment dominates over host genetics in shaping human gut microbiota. Nature 555, 210–215 (2018).
- Strobel, K. M., Juul, S. E. & Hendrixson, D. T. Maternal Nutritional Status and the Microbiome across the Pregnancy and the Post-Partum Period. Microorganisms 11, (2023). [CrossRef]
- Bourdeau-Julien, I. et al. The diet rapidly and differentially affects the gut microbiota and host lipid mediators in a healthy population. Microbiome 11, 26 (2023).
- Wastyk, H. C. et al. Gut-microbiota-targeted diets modulate human immune status. Cell 184, 4137-4153.e14 (2021). [CrossRef]
- Martínez, I. et al. Gut microbiome composition is linked to whole grain-induced immunological improvements. ISME J 7, 269–80 (2013).
- Prendergast, A. J. et al. Stunting Is Characterized by Chronic Inflammation in Zimbabwean Infants. PLoS One 9, e86928 (2014). [CrossRef]
- Savaiano, D. A. & Hutkins, R. W. Yogurt, cultured fermented milk, and health: a systematic review. Nutr Rev 79, 599–614 (2021).
- Khorraminezhad, L. & Rudkowska, I. Effect of Yogurt Consumption on Metabolic Syndrome Risk Factors: a Narrative Review. Curr Nutr Rep 10, 83–92 (2021). [CrossRef]
- Schmidt, K. A. et al. Impact of low-fat and full-fat dairy foods on fasting lipid profile and blood pressure: exploratory endpoints of a randomized controlled trial. Am J Clin Nutr 114, 882–892 (2021). [CrossRef]
- Marco, M. L. et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on fermented foods. Nat Rev Gastroenterol Hepatol 18, 196–208 (2021). [CrossRef]
- Taylor, B. C. et al. Consumption of Fermented Foods Is Associated with Systematic Differences in the Gut Microbiome and Metabolome. mSystems 5, (2020). [CrossRef]
- SaeidiFard, N., Djafarian, K. & Shab-Bidar, S. Fermented foods and inflammation: A systematic review and meta-analysis of randomized controlled trials. Clin Nutr ESPEN 35, 30–39 (2020).
- Adekoya, I. et al. Occurrence of bacteria and endotoxins in fermented foods and beverages from Nigeria and South Africa. Int J Food Microbiol 305, 108251 (2019). [CrossRef]
- Chelule, P. K., Mokgatle, M. M., Zungu, L. I. & Chaponda, A. Caregivers’ knowledge and use of fermented foods for infant and young children feeding in a rural community of odi, gauteng province, South Africa. Health Promot Perspect 4, 54–60 (2014).
- Fadahunsi, I. & Soremekun, O. Production, Nutritional and Microbiological Evaluation of Mahewu a South African Traditional Fermented Porridge. J Adv Biol Biotechnol 14, 1–10 (2017).
- Pswarayi, F. & Gänzle, M. G. Composition and Origin of the Fermentation Microbiota of Mahewu, a Zimbabwean Fermented Cereal Beverage. Appl Environ Microbiol 85, (2019). [CrossRef]
- Holzapfel, W. & Leonie Taljaard, J. Industrialization of Mageu Fermentation in South Africa. in (2004). [CrossRef]
- Rajabally, N. et al. A comparison of Clostridium difficile diagnostic methods for identification of local strains in a South African centre. J Med Microbiol 65, 320–327 (2016). [CrossRef]
- Weisburg, W. G., Barns, S. M., Pelletier, D. A. & Lane, D. J. 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173, 697–703 (1991).
- Zhang, Z., Schwartz, S., Wagner, L. & Miller, W. A Greedy Algorithm for Aligning DNA Sequences. Journal of Computational Biology 7, 203–214 (2000). [CrossRef]
- Harris, P. A. et al. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42, 377–381 (2009).
- World Health Organization. Infant and young child feeding fact sheet. https://www.who.int/news-room/fact-sheets/detail/infant-and-young-child-feeding (2023).
- Minimum Dietary Diversity for Women. (FAO, 2021). [CrossRef]
- U.S. Department of Health and Human Services, N. I. of H. N. I. of A. and I. D. D. of A. Division of AIDS (DAIDS) Table for Grading the Severity of Adult and Pediatric Adverse Events, Corrected Version 2.1. https://rsc.niaid.nih.gov/sites/default/files/daidsgradingcorrectedv21.pdf (2017).
- Moshfegh, A. J. et al. The US Department of Agriculture Automated Multiple-Pass Method reduces bias in the collection of energy intakes. Am J Clin Nutr 88, 324–332 (2008). [CrossRef]
- Steyn, N. P. & Senekal, M. A Guide for the Use of the Dietary Assessment and Education Kit (DAEK). (Medical Research Council, 2004).
- South African Medical Research Council. SAMRC Food Composition Tables for South Africa. (Cape Town, 2017).
- The Future of the Public’s Health in the 21st Century. (National Academies Press, Washington, D.C., 2003). [CrossRef]
- Dabee, S. et al. Comparison of Female Genital Tract Cytokine and Microbiota Signatures Induced by Initiation of Intramuscular DMPA and NET-EN Hormonal Contraceptives - a Prospective Cohort Analysis. Front Immunol 12, (2021). [CrossRef]
- Liu, C. M. et al. BactQuant: An enhanced broad-coverage bacterial quantitative real-time PCR assay. BMC Microbiol 12, 56 (2012). [CrossRef]
- Callahan, B. J. et al. DADA2: High-resolution sample inference from Illumina amplicon data. Nat Methods 13, 581–583 (2016). [CrossRef]
- Quast, C. et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 41, D590–D596 (2013).
- Davis, N. M., Proctor, D. M., Holmes, S. P., Relman, D. A. & Callahan, B. J. Simple statistical identification and removal of contaminant sequences in marker-gene and metagenomics data. Microbiome 6, 226 (2018).
- McMurdie, P. J. & Holmes, S. phyloseq: An R Package for Reproducible Interactive Analysis and Graphics of Microbiome Census Data. PLoS One 8, e61217 (2013).
- Kembel, S. W. et al. Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26, 1463–1464 (2010). [CrossRef]
- Paradis, E. & Schliep, K. ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35, 526–528 (2019). [CrossRef]
- Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15, 550 (2014).
- Lahti, L. & Shetty, S. microbiome R package. Preprint at (2017).
- Oksanen, J. et al. vegan: Community Ecology Package. Preprint at (2019).
- Callahan, B. J., Sankaran, K., Fukuyama, J. A., McMurdie, P. J. & Holmes, S. P. Bioconductor Workflow for Microbiome Data Analysis: from raw reads to community analyses. F1000Res 5, 1492 (2016). [CrossRef]
- Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J Stat Softw 67, (2015).
- Singh, A. et al. DIABLO: an integrative approach for identifying key molecular drivers from multi-omics assays. Bioinformatics 35, 3055–3062 (2019). [CrossRef]
- Dinno, A. Nonparametric Pairwise Multiple Comparisons in Independent Groups using Dunn’s Test. Stata J 15, 292–300 (2015). [CrossRef]
- Dietary Reference Intakes for Energy. (National Academies Press, Washington, D.C., 2023). [CrossRef]
- Dietary Reference Intakes. (National Academies Press, Washington, D.C., 2006). [CrossRef]
- Dietary Reference Intakes for Calcium and Vitamin D. (National Academies Press, Washington, D.C., 2011). [CrossRef]
- Gulhar, R., Ashraf, M. A. & Jialal, I. Physiology, Acute Phase Reactants. (2024).
- Zhernakova, A. et al. Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Science 352, 565–9 (2016).
- Muegge, B. D. et al. Diet drives convergence in gut microbiome functions across mammalian phylogeny and within humans. Science 332, 970–4 (2011). [CrossRef]
- Ruebel, M. L. et al. Associations between Maternal Diet, Body Composition and Gut Microbial Ecology in Pregnancy. Nutrients 13, (2021).
- Strobel, K. M., Juul, S. E. & Hendrixson, D. T. Maternal Nutritional Status and the Microbiome across the Pregnancy and the Post-Partum Period. Microorganisms 11, (2023). [CrossRef]
- Harper, A. et al. Dietary diversity, food insecurity and the double burden of malnutrition among children, adolescents and adults in South Africa: Findings from a national survey. Front Public Health 10, (2022). [CrossRef]
- Daji, G. A. et al. Physicochemical Properties and Bacterial Community Profiling of Optimal Mahewu (A Fermented Food Product) Prepared Using White and Yellow Maize with Different Inocula. Foods 11, 3171 (2022). [CrossRef]
- Senekal, M., Nel, J., Malczyk, S., Drummond, L. & Steyn, N. P. Provincial Dietary Intake Study (PDIS): Micronutrient Intakes of Children in a Representative/Random Sample of 1- to <10-Year-Old Children in Two Economically Active and Urbanized Provinces in South Africa. Int J Environ Res Public Health 17, (2020).
- Turnbaugh, P. J. et al. A core gut microbiome in obese and lean twins. Nature 457, 480–484 (2009).
- Le Chatelier, E. et al. Richness of human gut microbiome correlates with metabolic markers. Nature 500, 541–546 (2013). [CrossRef]
- Zouiouich, S. et al. Markers of metabolic health and gut microbiome diversity: findings from two population-based cohort studies. Diabetologia 64, 1749–1759 (2021).
- Ozato, N. et al. Two Blautia Species Associated with Visceral Fat Accumulation: A One-Year Longitudinal Study. Biology (Basel) 11, 318 (2022). [CrossRef]
- Shibata, M. et al. Mouse Model of Anti-Obesity Effects of Blautia hansenii on Diet-Induced Obesity. Curr Issues Mol Biol 45, 7147–7160 (2023). [CrossRef]
- Endo, A., Tanno, H., Kadowaki, R., Fujii, T. & Tochio, T. Extracellular fructooligosaccharide degradation in Anaerostipes hadrus for co-metabolism with non-fructooligosaccharide utilizers. Biochem Biophys Res Commun 613, 81–86 (2022). [CrossRef]
- Zeevi, D. et al. Structural variation in the gut microbiome associates with host health. Nature 568, 43–48 (2019).
- Qiao, S. et al. Gut Parabacteroides merdae protects against cardiovascular damage by enhancing branched-chain amino acid catabolism. Nat Metab 4, 1271–1286 (2022). [CrossRef]
- Narazaki, M. & Kishimoto, T. The Two-Faced Cytokine IL-6 in Host Defense and Diseases. Int J Mol Sci 19, (2018). [CrossRef]
- Infusino, I., Braga, F., Dolci, A. & Panteghini, M. Soluble Transferrin Receptor (sTfR) and sTfR/log Ferritin Index for the Diagnosis of Iron-Deficiency Anemia A Meta-Analysis. Am J Clin Pathol 138, 642–649 (2012). [CrossRef]
- Engels, C., Ruscheweyh, H.-J., Beerenwinkel, N., Lacroix, C. & Schwab, C. The Common Gut Microbe Eubacterium hallii also Contributes to Intestinal Propionate Formation. Front Microbiol 7, (2016).
- Di Vincenzo, F., Del Gaudio, A., Petito, V., Lopetuso, L. R. & Scaldaferri, F. Gut microbiota, intestinal permeability, and systemic inflammation: a narrative review. Intern Emerg Med 19, 275–293 (2024). [CrossRef]
| Characteristic | All (n=44) |
No Mageu (n=16) | SBM (n=13) |
LCM (n=15) |
|---|---|---|---|---|
| Age at enrollment, years, median (IQR) | 27 (23-33) | 27 (23-32) | 24 (21-28) | 31 (27-33) |
| Formal housing, n (%) | 21 (47.7) | 9 (56.3) | 6 (46.2) | 6 (40.0) |
| Unemployed, n (%) | 41 (93.2) | 16 (100) | 11 (84.6) | 14 (93.3) |
| BMI at week 4, median (IQR)* | 29.6 (26.6-34.9) |
30.5 (25.9-35.3) |
29.8 (27.8-34.3) |
28.7 (27.4-34.5) |
| Primiparous, n (%) | 15 (34.1) | 6 (37.5) | 5 (38.5) | 3 (20.0) |
| Running water inside, n (%) | 37 (84.1) | 14 (87.5) | 12 (92.3) | 12 (80.0) |
| Married, n (%) | 11 (25.0) | 3 (18.8) | 3 (23.1) | 5 (33.3) |
| EER/EAR/AI/RDA | No mageu (n=11) | SBM (n=12) | LCM (n=11) | Adj. p | |
|---|---|---|---|---|---|
| Energy (kcal) | 9939 [9511-10607(EER) [48] | 8228.02 (1410.19) |
9308.90 (1131.41) | 9006.62 (1224.42) |
0.12 |
| Carbohydrate (g) | - | 276.14 (69.89) | 319.13 (54.37) | 314.61 (57.31) | 0.10 |
| Total protein (g) | 63 [55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71] (RDA) [49] | 65.52 (11.16) | 74.08 (8.52) | 71.41 (13.20) | 0.10 |
| Plant protein (g) | - | 24.94 (5.03) | 30.77 (5.30) | 29.01 (5.70) | 0.04 |
| Animal protein (g) | - | 35.65 (7.77) | 36.97 (4.92) | 37.03 (7.95) | 0.87 |
| Total fat (g) | - | 59.75 (6.28) | 62.90 (4.73) | 61.20 (5.84) | 0.41 |
| Saturated fat (g) | - | 16.01 (2.41) | 17.05 (2.10) | 16.82 (2.80) | 0.57 |
| Monounsaturated fat (g) | - | 20.26 (3.38) | 21.27 (2.28) | 20.10 (2.75) | 0.56 |
| Polyunsaturated fat (g) | - | 17.28 (0.43) | 17.53 (0.43) | 17.43 (0.44) | 0.41 |
| Trans fat (g) | - | 0.52 (0.47) | 0.46 (0.34) | 0.49 (0.40) | 0.95 |
| Cholesterol (g) | - | 200.18 (130.34) | 221.54 (135.03) | 270.96 (211.55) | 0.58 |
| Total fibre (g) | 25g (AI) [49] | 15.02 (3.59) | 17.82 (3.34) | 17.12 (3.44) | 0.15 |
| Added sugar (g) | - | 30.55 (27.37) | 20.83 (24.18) | 23.23 (25.22) | 0.65 |
| Calcium (mg) | 800 (EAR) [50] | 309.71 (144.44) | 391.62 (117.84) | 429.64 (140.26) | 0.12 |
| Magnesium (mg) | 350 (EAR) [50] | 206.97 (51.36) | 251.32 (49.72) | 233.44 (57.27) | 0.15 |
| Iron (mg) | 6 (EAR) [49] | 11.90 (2.74) | 14.75 (3.22) | 14.29 (4.47) | 0.14 |
| Zinc (mg) | 9.4 (EAR) [49] | 10.72 (1.80) | 12.25 (2.08) | 11.18 (2.12) | 0.19 |
| Vitamin A (mcg) | 500 (EAR) [49] | 571.91 (180.53) | 675.59 (158.07) | 677.94 (249.60) | 0.37 |
| Vitamin C (mg)* | 60 (EAR) [49] | 19.90 (0.00) | 19.90 (0.00) | 19.90 (0.00) | - |
| Vitamin E (mg) | 12 (EAR) [49] | 9.41 (0.15) | 9.50 (0.14) | 9.48 (0.12) | 0.25 |
| Vitamin B1 (Thiamine) (mg) | 0.9 (EAR) [49] | 1.37 (0.31) | 1.68 (0.42) | 1.55 (0.38) | 0.16 |
| Vitamin B2 (Riboflavin) (mg) | 0.9 (EAR) [49] [49] | 0.97 (0.29) | 1.17 (0.47) | 1.22 (0.55) | 0.40 |
| Niacin (mg)* | 11 (EAR) [49] | 27.90 (0.00) | 27.90 (0.00) | 27.90 (0.00) | - |
| Vitamin B6 (mg) | 1.1 (EAR) [49] | 3.35 (0.35) | 3.58 (0.41) | 3.48 (0.42) | 0.38 |
| Folate (mcg) | 320 (EAR) [49] | 2.39 (1.66) | 2.68 (1.60) | 2.47 (1.25) | 0.90 |
| Vitamin B12 (mcg) | 2 (EAR) [49] | 2.39 (1.66) | 2.68 (1.60) | 2.47 (1.25) | 0.90 |
| Beta-carotene (mg) | - | 364.68 (343.89) | 671.34 (790.70) | 513.76 (557.31) | 0.48 |
| Flavonoids (mg) | - | 108.32 (196.53) | 103.70 (90.27) | 54.44 (58.34) | 0.55 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




