Submitted:
02 May 2025
Posted:
06 May 2025
Read the latest preprint version here
Abstract
Keywords:
Introduction
- 1.
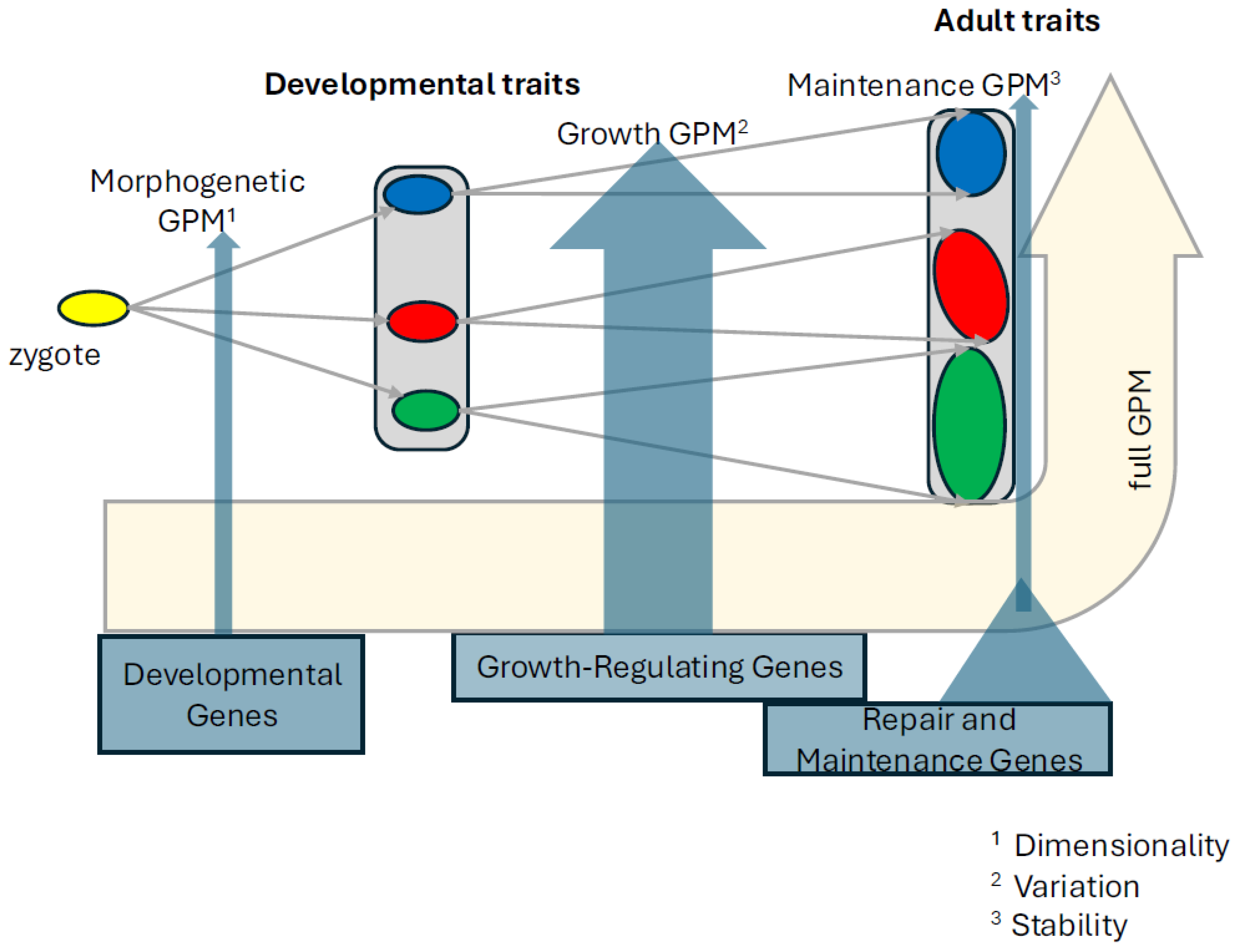
- Not a unitary map: Decomposing the GP map by process type
- a.
- GP maps of morphogenetic processes
- b.
- GP maps of growth processes
- c.
- GP maps of maintenance metabolism
- d.
- Developmental hierarchy and interdependence of the processes
2. GP Maps, (Micro)Evolutionary Theory and the Long-Term Evolutionary Change
3. Conclusion and Outlook
Acknowledgments
References
- Amundson, R. 2005. The changing role of the embryo in evolutionary thought : roots of evo-devo. Cambridge University Press, Cambridge ; New York.
- Arthur, W. 1997. The origin of animal body plans : a study in evolutionary developmental biology. Cambridge University Press, Cambridge, U.K. ; New York, NY, USA.
- Atchley, W.R.; Hall, B.K. A model for development and evolution of complex morphological structures. Biol Rev Camb Philos Soc 1991, 66, 101–157. [Google Scholar] [CrossRef] [PubMed]
- Baumann, G.; Shaw, M.A.; Buchanan, T.A. In vivo kinetics of a covalent growth hormone-binding protein complex. Metabolism 1989, 38, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Bell, G.; Mooers, A. Size and complexity among multicellular organisms. Biological Journal of the Linnean Society 1997, 345–363. [Google Scholar] [CrossRef]
- Bonner, J.T. 1988. The evolution of complexity by means of natural selection. Princeton University Press, Princeton, N.J.
- Bonner, J.T. Perspective: the size-complexity rule. Evolution 2004, 58, 1883–1890. [Google Scholar] [PubMed]
- Bourg, S.; Bolstad, G.H.; Griffin, D.V.; Pélabon, C.; Hansen, T.F. Directional epistasis is common in morphological divergence. Evolution 2024, 78, 934–950. [Google Scholar] [CrossRef]
- Carter, A.J.; Hermisson, J.; Hansen, T.F. The role of epistatic gene interactions in the response to selection and the evolution of evolvability. Theor Popul Biol 2005, 68, 179–196. [Google Scholar] [CrossRef]
- Cheverud, J.M. Phenotypic, Genetic, and Environmental Morphological Integration in the Cranium. Evolution 1982, 36, 499–516. [Google Scholar] [CrossRef]
- Cheverud, J.M. Relationships among ontogenetic, static, and evolutionary allometry. Am J Phys Anthropol 1982, 59, 139–149. [Google Scholar] [CrossRef]
- Cheverud, J.M.; Ehrich, T.H.; Vaughn, T.T.; Koreishi, S.F.; Linsey, R.B.; Pletscher, L.S. Pleiotropic effects on mandibular morphology II: differential epistasis and genetic variation in morphological integration. J Exp Zool B Mol Dev Evol 2004, 302, 424–435. [Google Scholar] [CrossRef]
- Cheverud, J.M.; Routman, E.J. Epistasis and its contribution to genetic variance components. Genetics 1995, 139, 1455–1461. [Google Scholar] [CrossRef]
- Cheverud, J.M.; Routman, E.J. Epistasis as a Source of Increased Additive Genetic Variance at Population Bottlenecks. Evolution 1996, 50, 1042–1051. [Google Scholar] [CrossRef]
- Cheverud, J.M.; Routman, E.J.; Duarte, F.A.; van Swinderen, B.; Cothran, K.; Perel, C. Quantitative trait loci for murine growth. Genetics 1996, 142, 1305–1319. [Google Scholar] [CrossRef] [PubMed]
- Cheverud, J.M.; Rutledge, J.J.; Atchley, W.R. Quantitative Genetics of Development: Genetic Correlations among Age-Specific Trait Values and the Evolution of Ontogeny. Evolution 1983, 37, 895–905. [Google Scholar]
- Clemmons, D.R. Insulin-like growth factor binding proteins and their role in controlling IGF actions. Cytokine Growth Factor Rev 1997, 8, 45–62. [Google Scholar] [CrossRef]
- Cooper, K.L.; Sears, K.E.; Uygur, A.; Maier, J.; Baczkowski, K.S.; Brosnahan, M.; Antczak, D.; Skidmore, J.A.; Tabin, C.J. Patterning and post-patterning modes of evolutionary digit loss in mammals. Nature 2014, 511, 41–45. [Google Scholar] [CrossRef] [PubMed]
- de Bakker, M.A.; Fowler, D.A.; den Oude, K.; Dondorp, E.M.; Navas, M.C.; Horbanczuk, J.O.; Sire, J.Y.; Szczerbińska, D.; Richardson, M.K. Digit loss in archosaur evolution and the interplay between selection and constraints. Nature 2013, 500, 445–448. [Google Scholar] [CrossRef] [PubMed]
- De Beer, G. 1940. Embryos and ancestors. The Clarendon Press, Oxford,.
- DiFrisco, J.; Wagner, G. Body plan identity: A mechanistic model. Evolutionary Biology 2023, 49, 1–19. [Google Scholar] [CrossRef]
- Draghi, J.A.; Parsons, T.L.; Wagner, G.P.; Plotkin, J.B. Mutational robustness can facilitate adaptation. Nature 2010, 463, 353–355. [Google Scholar] [CrossRef]
- Galis, F. 2023. Evolvability of Body Plans: On Phylotypic Stages, Developmental Modularity, and an Ancient Metazoan Constraint in T. Hansen, D. Houle, M. Pavlicev, and C. Pelabon, eds. Evolvability. A Unifying Concept in Evolutionary Biology? MIT Press.
- Gjuvsland, A.B.; Hayes, B.J.; Omholt, S.W.; Carlborg, O. Statistical epistasis is a generic feature of gene regulatory networks. Genetics 2007, 175, 411–420. [Google Scholar] [CrossRef]
- Gjuvsland, A.B.; Vik, J.O.; Beard, D.A.; Hunter, P.J.; Omholt, S.W. Bridgin? the genotype-phenotype gap: what does it take? J Physiol 2013, 591, 2055–2066. [Google Scholar]
- Gould, S.J. 1977. Ontogeny and phylogeny. Belknap Press of Harvard University Press, Cambridge, Mass.
- Haeckel, E. Die Gastrula und die Eifurchung der Thiere. Jena. Z. Naturwiss. 1875, 402–508. [Google Scholar]
- Hallgrímsson, B.; Hall, B.K. 2005. Variation. Elsevier Academic Press, Amsterdam ; Boston.
- Hallgrímsson, B.; Jamniczky, H.; Young, N.M.; Rolian, C.; Parsons, T.E.; Boughner, J.C.; Marcucio, R.S. Deciphering the Palimpsest: Studying the Relationship Between Morphological Integration and Phenotypic Covariation. Evol Biol 2009, 36, 355–376. [Google Scholar] [CrossRef] [PubMed]
- Hammond, J.E.; Baker, R.E.; Verd, B. Modularity of the segmentation clock and morphogenesis. Elife 2025, 14. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.F.; Wagner, G.P. Modeling genetic architecture: a multilinear theory of gene interaction. Theor Popul Biol 2001, 59, 61–86. [Google Scholar] [CrossRef]
- Hendrikse, J.L.; Parsons, T.E.; Hallgrimsson, B. Evolvability as the proper focus of evolutionary developmental biology. Evol Dev 2007, 9, 393–401. [Google Scholar] [CrossRef]
- Holstad, A.; Voje, K.L.; Opedal, Ø.; Bolstad, G.H.; Bourg, S.; Hansen, T.F.; Pélabon, C. Evolvability predicts macroevolution under fluctuating selection. Science 2024, 384, 688–693. [Google Scholar] [CrossRef]
- Houle, D.; Bolstad, G.H.; van der Linde, K.; Hansen, T.F. Mutation predicts 40 million years of fly wing evolution. Nature 2017, 548, 447–450. [Google Scholar] [CrossRef]
- Huxley, J. 1932. Problems of Relative Growth. Dial Press, New York.
- Irie, N.; Sehara-Fujisawa, A. The vertebrate phylotypic stage and an early bilaterian-related stage in mouse embryogenesis defined by genomic information. BMC Biol 2007, 5, 1. [Google Scholar] [CrossRef]
- Lande, R. EVOLUTIONARY MECHANISMS OF LIMB LOSS IN TETRAPODS. Evolution 1978, 32, 73–92. [Google Scholar] [CrossRef]
- Lande, R. Quantitative Genetic Analysis of Multivariate Evolution, Applied to Brain:Body Size Allometry. Evolution 1979, 33, 402–416. [Google Scholar] [CrossRef]
- Lynch, M. Evolution of the mutation rate. Trends Genet 2010, 26, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Marchini, M.; Rolian, C. Artificial selection sheds light on developmental mechanisms of limb elongation. Evolution 2018, 72, 825–837. [Google Scholar] [CrossRef] [PubMed]
- Mayer, C.; Hansen, T.F. Evolvability and robustness: A paradox restored. J Theor Biol 2017, 430, 78–85. [Google Scholar] [CrossRef]
- Meiklejohn, C.D.; Hartl, D.L. A single mode of canalization. 2002, 17, 468–473. [Google Scholar] [CrossRef]
- Meyer, R.D.; Mohammadi, M.; Rahimi, N. A single amino acid substitution in the activation loop defines the decoy characteristic of VEGFR-1/FLT-1. J Biol Chem 2006, 281, 867–875. [Google Scholar] [CrossRef] [PubMed]
- Nijhout, H.F.; Reed, M.C.; Budu, P.; Ulrich, C.M. A mathematical model of the folate cycle: new insights into folate homeostasis. J Biol Chem 2004, 279, 55008–55016. [Google Scholar] [CrossRef] [PubMed]
- Pavlicev, M.; Bourg, S.; Le Rouzic, A. 2023. The Genotype-Phenotype Map Structure and its Role in Evolvability in T. Hansen, D. Houle, M. Pavlicev, and C. Pelabon, eds. Evolvability: A Unifying Concept in Evolutionary Biology? MIT Press.
- Pavlicev, M.; Cheverud, J. Constraints Evolve: Context Dependency of Gene Effects Allows Evolution of Pleiotropy. Annual Review of Ecology, Evolution and Systematics 2015, 46, 413–434. [Google Scholar] [CrossRef]
- Pavlicev, M.; Cheverud, J.M.; Wagner, G.P. Evolution of adaptive phenotypic variation patterns by direct selection for evolvability. Proc Biol Sci 2011, 278, 1903–1912. [Google Scholar] [CrossRef]
- Pavlicev, M.; DiFrisco, J.; Love, A.C.; Wagner, G.P. Metabolic complementation between cells drives the evolution of tissues and organs. Biol Lett 2024, 20, 20240490. [Google Scholar] [CrossRef]
- Pavlicev, M.; Kenney-Hunt, J.P.; Norgard, E.A.; Roseman, C.C.; Wolf, J.B.; Cheverud, J.M. Genetic variation in pleiotropy: differential epistasis as a source of variation in the allometric relationship between long bone lengths and body weight. Evolution 2008, 62, 199–213. [Google Scholar] [CrossRef]
- Pavlicev, M.; Le Rouzic, A.; Cheverud, J.M.; Wagner, G.P.; Hansen, T.F. Directionality of epistasis in a murine intercross population. Genetics 2010, 185, 1489–1505. [Google Scholar] [CrossRef]
- Piasecka, B.; Lichocki, P.; Moretti, S.; Bergmann, S.; Robinson-Rechavi, M. The hourglass and the early conservation models--co-existing patterns of developmental constraints in vertebrates. PLoS Genet 2013, 9, e1003476. [Google Scholar] [CrossRef] [PubMed]
- Raff, R.A. 1996. The shape of life : genes, development, and the evolution of animal form. University of Chicago Press, Chicago.
- Reed, M.; Best, J.; Golubitsky, M.; Stewart, I.; Nijhout, H.F. Analysis of Homeostatic Mechanisms in Biochemical Networks. Bull Math Biol 2017, 79, 2534–2557. [Google Scholar] [CrossRef] [PubMed]
- Reed, M.C.; Thomas, R.L.; Pavisic, J.; James, S.J.; Ulrich, C.M.; Nijhout, H.F. A mathematical model of glutathione metabolism. Theor Biol Med Model 2008, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Riedl, R. 1978. Order in living organisms : a systems analysis of evolution. Wiley, Chichester ; New York.
- Riska, B. SOME MODELS FOR DEVELOPMENT, GROWTH, AND MORPHOMETRIC CORRELATION. Evolution 1986, 40, 1303–1311. [Google Scholar] [CrossRef]
- Riska, B. COMPOSITE TRAITS, SELECTION RESPONSE, AND EVOLUTION. Evolution 1989, 43, 1172–1191. [Google Scholar] [CrossRef]
- Riska, B.; Atchley, W.R. Genetics of growth predict patterns of brain-size evolution. Science 1985, 229, 668–671. [Google Scholar] [CrossRef]
- Riska, B.; Atchley, W.R.; Rutledge, J.J. A genetic analysis of targeted growth in mice. Genetics 1984, 107, 79–101. [Google Scholar] [CrossRef]
- Rohner, P.T.; Berger, D. Developmental bias predicts 60 million years of wing shape evolution. Proc Natl Acad Sci U S A 2023, 120, e2211210120. [Google Scholar] [CrossRef]
- Rohner, P.T.; Berger, D. Macroevolution along developmental lines of least resistance in fly wings. Nat Ecol Evol 2025, 9, 639–651. [Google Scholar] [CrossRef]
- Saito, K.; Tsuboi, M.; Takahashi, Y. Developmental noise and phenotypic plasticity are correlated in. Evol Lett 2024, 8, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Sander, K. 1983. The evolution of patterning mechanisms: Gleanings from insect embryogenesis and spermatogenesis. Pp. 137-154 in B. Goodwin, N. Holder, and C. Wylie, eds. Development and Evolution. Cambridge University Press, Cambridge.
- Sanger, T.J.; Norgard, E.A.; Pletscher, L.S.; Bevilacqua, M.; Brooks, V.R.; Sandell, L.J.; Cheverud, J.M. Developmental and genetic origins of murine long bone length variation. J Exp Zool B Mol Dev Evol 2011, 316, 146–161. [Google Scholar] [CrossRef] [PubMed]
- Schut, P.C.; Cohen-Overbeek, T.E.; Galis, F.; Broek, C.M.T.; Steegers, E.A.; Eggink, A.J. Adverse Fetal and Neonatal Outcome and an Abnormal Vertebral Pattern: A Systematic Review. Obstet Gynecol Surv 2016, 71, 741–750. [Google Scholar] [CrossRef]
- Schut, P.C.; Eggink, A.J.; Cohen-Overbeek, T.E.; Van Dooren, T.J.M.; de Borst, G.J.; Galis, F. Miscarriage is associated with cervical ribs in thoracic outlet syndrome patients. Early Hum Dev 2020, 144, 105027. [Google Scholar] [CrossRef]
- Schwenk, K.; Wagner, P.G. Function and the Evolution of Phenotypic Stability: Connecting Pattern to Process. American Zoologist 2001, 41, 552–563. [Google Scholar] [CrossRef]
- Thompson, D.A.W. 1942. On Growth and Form. Cambridge University Press, Cambridge.
- Tickle, C. Molecular basis of vertebrate limb patterning. Am J Med Genet 2002, 112, 250–255. [Google Scholar] [CrossRef]
- Tickle, C. Vertebrate limb development and possible clues to diversity in limb form. J Morphol 2002, 252, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Tickle, C.; Towers, M. Sonic Hedgehog Signaling in Limb Development. Front Cell Dev Biol 2017, 5, 14. [Google Scholar]
- Tsuboi, M.; Sztepanacz, J.; De Lisle, S.; Voje, K.L.; Grabowski, M.; Hopkins, M.J.; Porto, A.; Balk, M.; Pontarp, M.; Rossoni, D.; Hildesheim, L.S.; Horta-Lacueva, Q.J.; Hohmann, N.; Holstad, A.; Lürig, M.; Milocco, L.; Nilén, S.; Passarotto, A.; Svensson, E.I.; Houle, D.; et al. The paradox of predictability provides a bridge between micro- and macroevolution. J Evol Biol 2024, 37, 1413–1432. [Google Scholar] [CrossRef]
- Ulrich, C.M.; Neuhouser, M.; Liu, A.Y.; Boynton, A.; Gregory, J.F.; Shane, B.; James, S.J.; Reed, M.C.; Nijhout, H.F. Mathematical modeling of folate metabolism: predicted effects of genetic polymorphisms on mechanisms and biomarkers relevant to carcinogenesis. Cancer Epidemiol Biomarkers Prev 2008, 17, 1822–1831. [Google Scholar] [CrossRef]
- Ulrich, C.M.; Reed, M.C.; Nijhout, H.F. Modeling folate, one-carbon metabolism, and DNA methylation. Nutr Rev 2008, 66 (Suppl 1), S27–S30. [Google Scholar] [CrossRef] [PubMed]
- von Dassow, G.; Meir, E.; Munro, E.M.; Odell, G.M. The segment polarity network is a robust developmental module. Nature 2000, 406, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Von Dassow, G.; Odell, G.M. Design and constraints of the Drosophila segment polarity module: robust spatial patterning emerges from intertwined cell state switches. J Exp Zool 2002, 294, 179–215. [Google Scholar] [CrossRef]
- Wagner, A. Robustness and evolvability: a paradox resolved. Proc Biol Sci 2008, 275, 91–100. [Google Scholar] [CrossRef]
- Wagner, G.P.; Altenberg, L. Perspective: Complex Adaptations and the Evolution of Evolvability. Evolution 1996, 50, 967–976. [Google Scholar] [CrossRef] [PubMed]
- Wagner, G.P.; Booth, G.; Bagheri-Chaichian, H. A Population Genetic Theory of Canalization. Evolution 1997, 51, 329–347. [Google Scholar] [CrossRef]
- Wagner, G.P.; Schwenk, K. Evolutionarily Stable Configurations: Functional Integration and the Evolution of Phenotypic Stability. Evolutionary Biology 2000, 31, 155–217. [Google Scholar]
- Wolf, J.B.; Leamy, L.J.; Routman, E.J.; Cheverud, J.M. Epistatic pleiotropy and the genetic architecture of covariation within early and late-developing skull trait complexes in mice. Genetics 2005, 171, 683–694. [Google Scholar] [CrossRef]
- Wolf, J.B.; Pomp, D.; Eisen, E.J.; Cheverud, J.M.; Leamy, L.J. The contribution of epistatic pleiotropy to the genetic architecture of covariation among polygenic traits in mice. Evol Dev 2006, 8, 468–476. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



