Submitted:
01 May 2025
Posted:
02 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
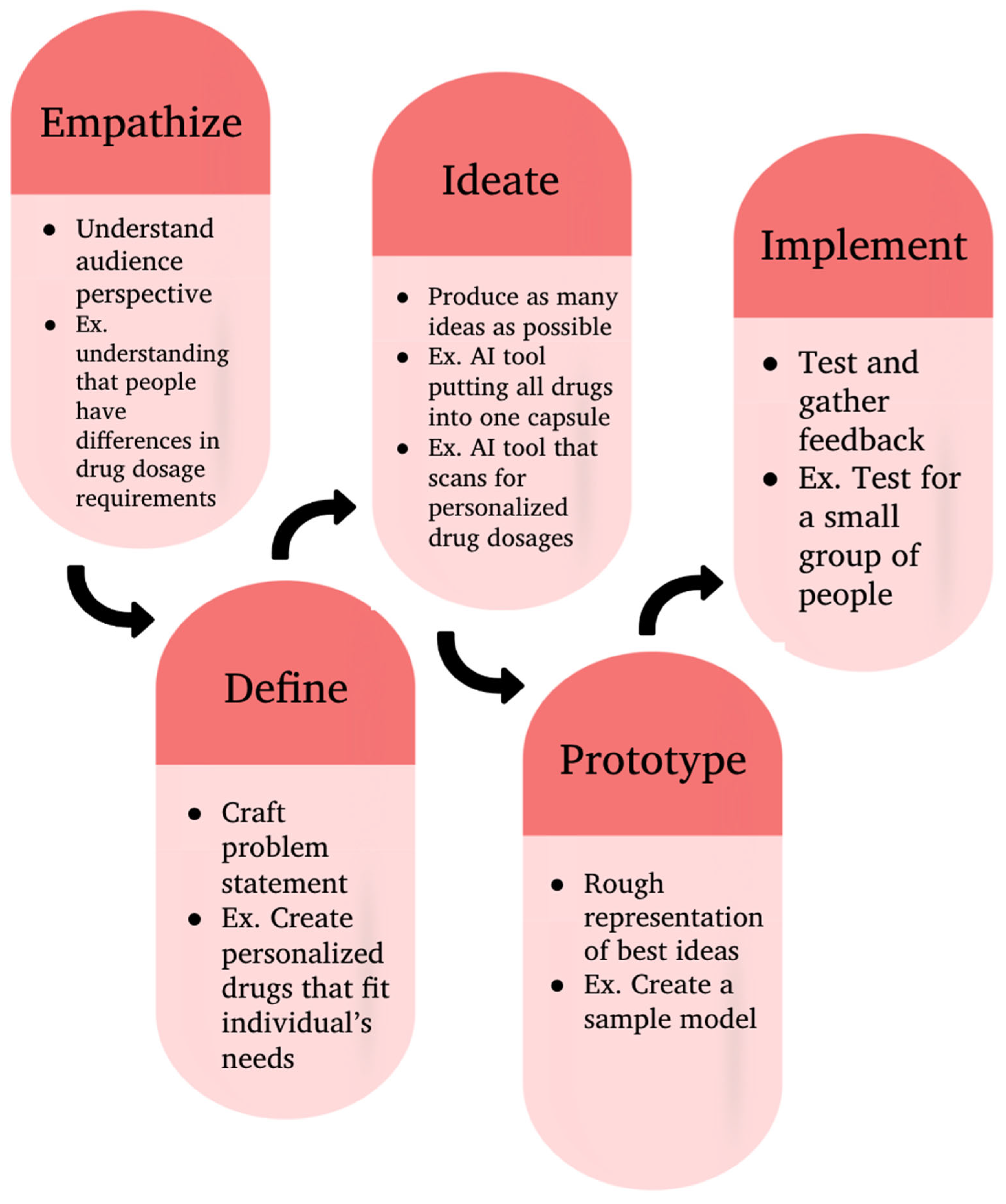
2. Background
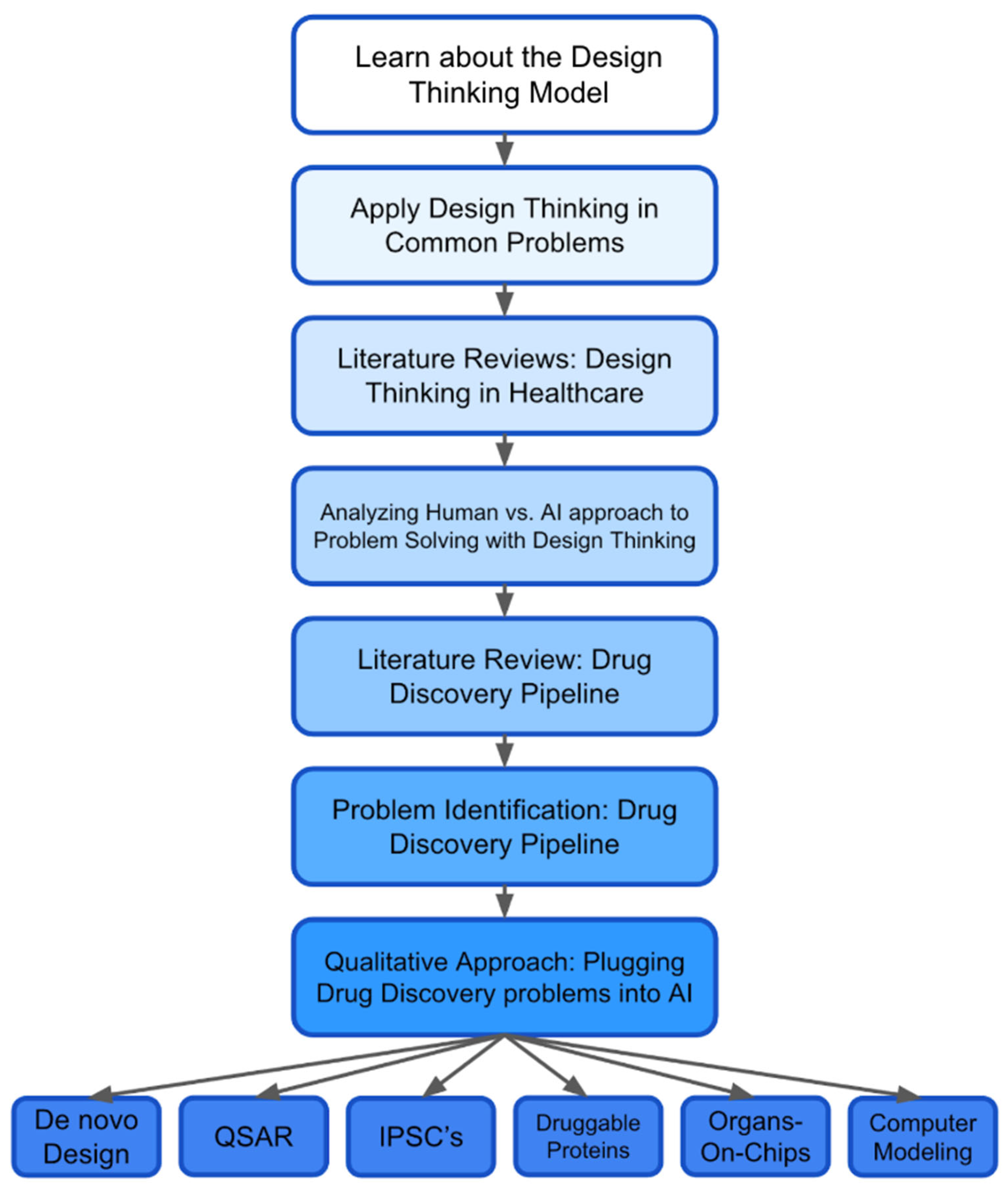
3. Methodology
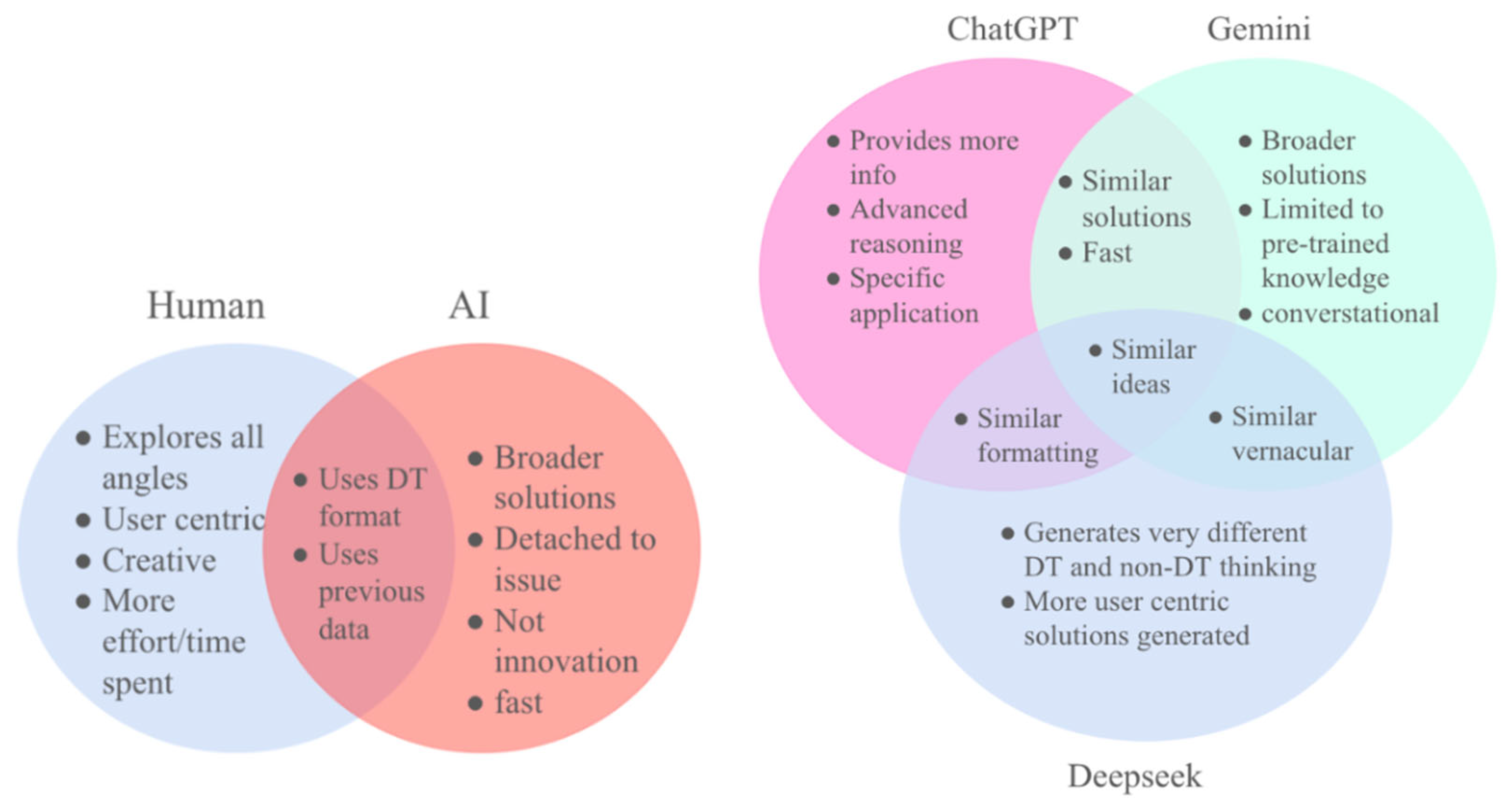
4. Results
- Organ-on-chip


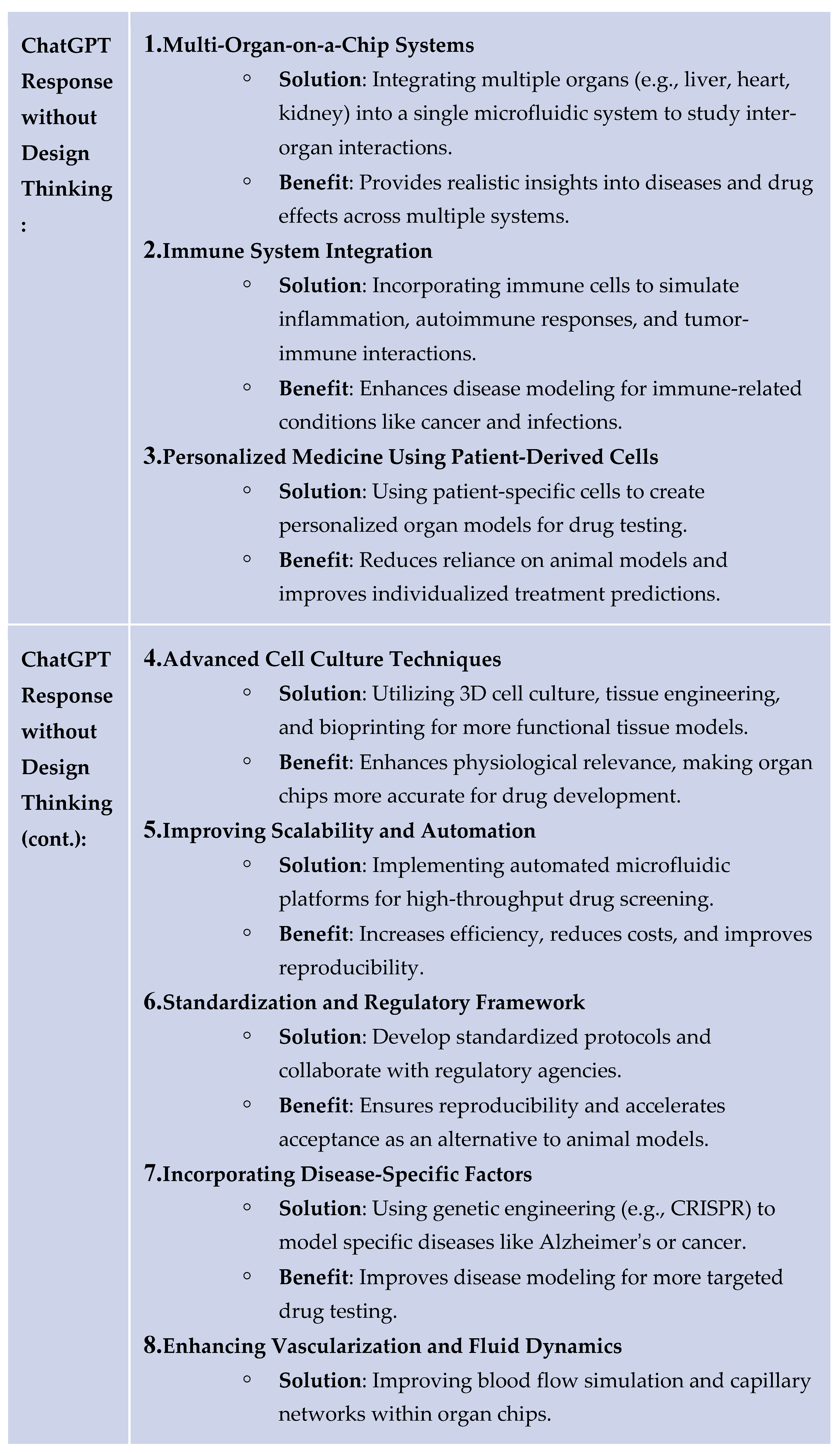













5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| QSAR | Quantitative structure-activity relationship |
| iPSCs | Induced Pluripotent Stem Cells |
References
- McLaughlin, J. E., Wolcott, M. D., Hubbard, D., Umstead, K., & Rider, T. R. (2019). A qualitative review of the design thinking framework in health professions education. BMC Medical Education, 19(1). [CrossRef]
- Health care providers can use design thinking to improve patient experiences. (2017, August 31). https://hbr.org/2017/08/health-care-providers-can-use-design-thinking-to-improve-patient-experiences.
- Fortune, J., Burke, J., Dillon, C., Dillon, S., O’Toole, S., Enright, A., Flynn, A., Manikandan, M., Kroll, T., Lavelle, G., & Ryan, J. M. (2022). Co-designing resources to support the transition from child to adult health services for young people with cerebral palsy: A design thinking approach. Frontiers in Rehabilitation Sciences, 3. [CrossRef]
- Lorusso, L., Lee, J. H., & Worden, E. A. (2021). Design Thinking for Healthcare: Transliterating the Creative Problem-Solving Method into Architectural practice. HERD Health Environments Research & Design Journal, 14(2), 16–29. [CrossRef]
- Altman, M., Huang, T. T., & Breland, J. Y. (2018). Design thinking in health care. Preventing Chronic Disease, 15. [CrossRef]
- Göttgens, I., & Oertelt-Prigione, S. (2021). The Application of Human-Centered Design Approaches in Health Research and Innovation: A Narrative review of current practices. JMIR Mhealth and Uhealth, 9(12), e28102. [CrossRef]
- Roberts, J. P., Fisher, T. R., Trowbridge, M. J., & Bent, C. (2016). A design thinking framework for healthcare management and innovation. Healthcare, 4(1), 11–14. [CrossRef]
- Abookire, S., Plover, C., Frasso, R., & Ku, B. (2020). Health Design Thinking: an innovative approach in public health to defining problems and finding solutions. Frontiers in Public Health, 8. [CrossRef]
- Feuerwerker, S., Rankin, N., Wohler, B., Gemino, H., & Risler, Z. (2019). Improving patient satisfaction by using design thinking: Patient advocate role in the emergency department. Cureus. [CrossRef]
- Wang, N., & He, Q. (2023). Artificial intelligence and Bioinformatics Applications in precision medicine and future implications. In Elsevier eBooks (pp. 9–24). [CrossRef]
- Santa Clara University. (n.d.). Pharmacogenomics, Ethics, and Public Policy. Markkula Center for Applied Ethics. https://www.scu.edu/ethics/focus-areas/bioethics/resources/pharmacogenomics-ethics-and-public-policy/.
- Feldman, K., Suppes, S. L., & Goldman, J. L. (2024). Clarification of adverse drug reactions by a pharmacovigilance team results in increased antibiotic re-prescribing at a freestanding United States children’s hospital. PLoS ONE, 19(1), e0295410. [CrossRef]
- Brown, M. T., & Bussell, J. K. (2011). Medication adherence: WHO cares? Mayo Clinic Proceedings, 86(4), 304–314. [CrossRef]
- Varghese, D., Ishida, C., Patel, P., & Koya, H. H. (2024, February 12). Polypharmacy. StatPearls - NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK532953/.
- Sullivan, T. (2019, March 21). A Tough Road: Cost To Develop One New Drug Is $2.6 Billion; Approval Rate for Drugs Entering Clinical Development is Less Than 12%. Policy & Medicine. https://www.policymed.com/2014/12/a-tough-road-cost-to-develop-one-new-drug-is-26-billion-approval-rate-for-drugs-entering-clinical-de.html.
- Team, C. S. (2023, December 15). Dealing with the challenges of drug discovery. CAS. https://www.cas.org/resources/cas-insights/dealing-challenges-drug-discovery.
- Singh, N., Vayer, P., Tanwar, S., Poyet, J., Tsaioun, K., & Villoutreix, B. O. (2023). Drug discovery and development: introduction to the general public and patient groups. Frontiers in Drug Discovery, 3. [CrossRef]
- Artificial intelligence and machine learning technology driven modern drug discovery and development. (2023). In International Journal of Molecular Sciences (Vol. 24, p. 2026). https://pmc.ncbi.nlm.nih.gov/articles/PMC9916967/pdf/ijms-24-02026.pdf.
- Sallam, M. (2023). ChatGPT Utility in Healthcare Education, Research, and Practice: Systematic Review on the promising perspectives and valid concerns. Healthcare, 11(6), 887. [CrossRef]
- Deng, J., Yang, Z., Ojima, I., Samaras, D., & Wang, F. (2021). Artificial intelligence in drug discovery: applications and techniques. Briefings in Bioinformatics, 23(1). [CrossRef]
- Zhavoronkov, A., Vanhaelen, Q., & Oprea, T. I. (2020). Will artificial intelligence for drug discovery impact clinical pharmacology? Clinical Pharmacology & Therapeutics, 107(4), 780–785. [CrossRef]
- Basile, A. O., Yahi, A., Tatonetti, N. P., & Columbia University Medical Center. (2019). Artificial intelligence for drug toxicity and safety. In Trends Pharmacol Sci (Vols. 40–9, pp. 624–635). [CrossRef]
- Schneider, P., Walters, W. P., Plowright, A. T., Sieroka, N., Listgarten, J., Goodnow, R. A., Fisher, J., Jansen, J. M., Duca, J. S., Rush, T. S., Zentgraf, M., Hill, J. E., Krutoholow, E., Kohler, M., Blaney, J., Funatsu, K., Luebkemann, C., & Schneider, G. (2019). Rethinking drug design in the artificial intelligence era. Nature Reviews Drug Discovery, 19(5), 353–364. [CrossRef]
- Moingeon, P., Kuenemann, M., & Guedj, M. (2021). Artificial intelligence-enhanced drug design and development: Toward a computational precision medicine. Drug Discovery Today, 27(1), 215–222. [CrossRef]
- MacFadyen, J. S. (2013). Design thinking. Holistic Nursing Practice, 28(1), 3–5. [CrossRef]
- Huang, T. T. K., Aitken, J., Ferris, E., & Cohen, N. (2018). Design thinking to improve implementation of public health interventions: an exploratory case study on enhancing park use. Design for Health, 2(2), 236–252. [CrossRef]
- Chen, Y., & Esmaeilzadeh, P. (2024). Generative AI in Medical Practice: In-Depth Exploration of privacy and Security challenges. Journal of Medical Internet Research, 26, e53008. [CrossRef]
- Generative AI vs. Large Language Models (LLMs): What’s the Difference? (n.d.). https://appian.com/blog/acp/process-automation/generative-ai-vs-large-language-models.
- What is LLM? - Large Language Models Explained - AWS. (n.d.). Amazon Web Services, Inc. https://aws.amazon.com/what-is/large-language-model/.
- Bach, S., & Bach, S. (2025, February 6). Large language model training: how three training phases shape LLMs. Snorkel AI. https://snorkel.ai/blog/large-language-model-training-three-phases-shape-llm-training/.
- https://medium.com/@kamalakshi.deshmukh/white-paper-on-chatgpt-a-comprehensive-overview-ee235c026cea.
- Emote. (2021, July 29). David Kelley: From design to design thinking at Stanford and IDEO - Echos. Echos. https://schoolofdesignthinking.echos.cc/blog/2017/06/david-kelley-from-design-to-design-thinking-at-stanford-and-ideo/.
- A new collaboration between the Hasso Plattner Institut and HAI brings the human factor of AI to the forefront | Stanford HAI. (n.d.). https://hai.stanford.edu/news/new-collaboration-between-hasso-plattner-institut-and-hai-brings-human-factor-ai-forefront.
- Lugmayr, A., Stockleben, B., Zou, Y. et al. Applying “Design Thinking” in the context of media management education. Multimed Tools Appl 71, 119–157 (2014). [CrossRef]
- Krolikowski, K. A., Bi, M., Baggott, C. M., Khorzad, R., Holl, J. L., & Kruser, J. M. (2022). Design thinking to improve healthcare delivery in the intensive care unit: Promise, pitfalls, and lessons learned. Journal of Critical Care, 69, 153999. [CrossRef]
- Gifford, A., Butcher, B., Chima, R. S., Moore, L., Brady, P. W., Zackoff, M. W., & Dewan, M. (2023). Use of design thinking and human factors approach to improve situation awareness in the pediatric intensive care unit. Journal of Hospital Medicine, 18(11), 978–985. [CrossRef]
- Aboul-Soud, M. a. M., Alzahrani, A. J., & Mahmoud, A. (2021). Induced Pluripotent stem cells (IPSCs)—Roles in regenerative therapies, disease modelling and drug screening. Cells, 10(9), 2319. [CrossRef]
- Induced pluripotent stem cells and neurological disease models. (2014, February 25). PubMed. https://pubmed.ncbi.nlm.nih.gov/24553870/.
- Bindhya, S., Sidhanth, C., Shabna, A., Krishnapriya, S., Garg, M., & Ganesan, T. (2018). Induced pluripotent stem cells: A new strategy to model human cancer. The International Journal of Biochemistry & Cell Biology, 107, 62–68. [CrossRef]
- Medvedev, S., Shevchenko, A., & Zakian, S. (2010, July 1). Induced Pluripotent Stem Cells: Problems and Advantages when Applying them in Regenerative Medicine. https://pmc.ncbi.nlm.nih.gov/articles/PMC3347549/.
- Gupta, R., Srivastava, D., Sahu, M., Tiwari, S., Ambasta, R. K., & Kumar, P. (2021). Artificial intelligence to deep learning: machine intelligence approach for drug discovery. Molecular Diversity, 25(3), 1315–1360. [CrossRef]
- Animals in research. (n.d.). Humane World for Animals. https://www.humaneworld.org/en/issue/animals-in-research.
- Leung, C. M., De Haan, P., Ronaldson-Bouchard, K., Kim, G., Ko, J., Rho, H. S., Chen, Z., Habibovic, P., Jeon, N. L., Takayama, S., Shuler, M. L., Vunjak-Novakovic, G., Frey, O., Verpoorte, E., & Toh, Y. (2022). A guide to the organ-on-a-chip. Nature Reviews Methods Primers, 2(1). [CrossRef]
- Meyers, J., Fabian, B., & Brown, N. (2021). De novo molecular design and generative models. Drug Discovery Today, 26(11), 2707–2715. [CrossRef]
- Kortemme, T. (2024). De novo protein design—From new structures to programmable functions. Cell, 187(3), 526–544. [CrossRef]
- Torres, S. V., Leung, P. J. Y., Venkatesh, P., Lutz, I. D., Hink, F., Huynh, H., Becker, J., Yeh, A. H., Juergens, D., Bennett, N. R., Hoofnagle, A. N., Huang, E., MacCoss, M. J., Expòsit, M., Lee, G. R., Bera, A. K., Kang, A., De La Cruz, J., Levine, P. M., . . . Baker, D. (2023). De novo design of high-affinity binders of bioactive helical peptides. Nature, 626(7998), 435–442. [CrossRef]
- Cherkasov, A., Muratov, E. N., Fourches, D., Varnek, A., Baskin, I. I., Cronin, M., Dearden, J., Gramatica, P., Martin, Y. C., Todeschini, R., Consonni, V., Kuz’min, V. E., Cramer, R., Benigni, R., Yang, C., Rathman, J., Terfloth, L., Gasteiger, J., Richard, A., & Tropsha, A. (2013). QSAR modeling: Where have you been? Where are you going to? Journal of Medicinal Chemistry, 57(12), 4977–5010. [CrossRef]
- Schneider, G. (2010). Virtual screening: an endless staircase? Nature Reviews Drug Discovery, 9(4), 273–276. [CrossRef]
- James, T., & Hristozov, D. (2021). Deep learning and computational chemistry. Methods in Molecular Biology, 125–151. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



