Submitted:
26 April 2025
Posted:
29 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1 Cell Lines and Plasmids
2.2. Antibodies
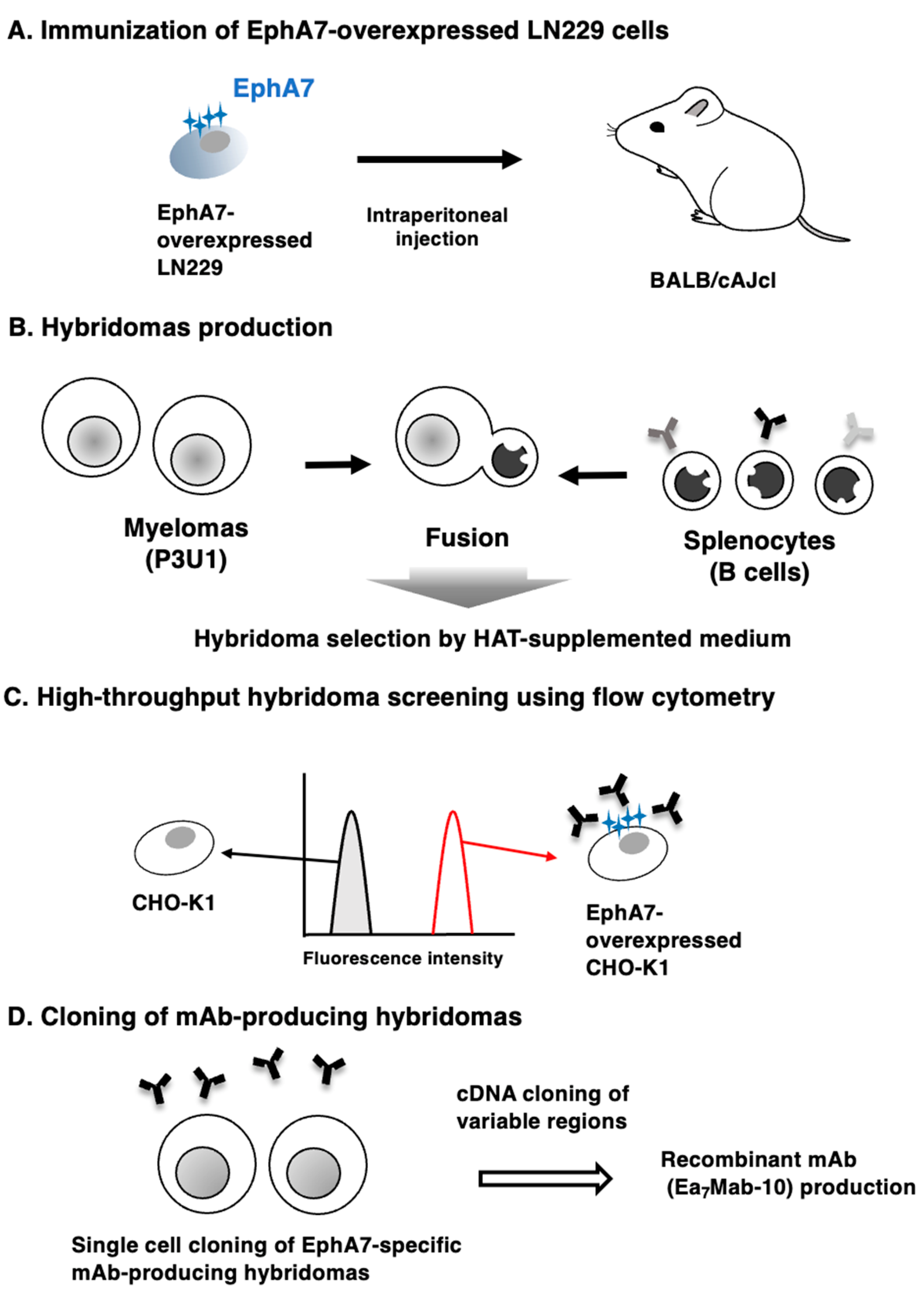
2.3. Hybridoma Production
2.4. Production of Recombinant Antibodies
2.5. Flow Cytometry
2.6. Determination of the Binding Affinity by Flow Cytometry
2.7. Western Blotting
2.8. Immunohistochemistry
3. Results
3.1. Development of an Anti-EphA7 mAb, Ea7Mab-10 Using the CBIS Method
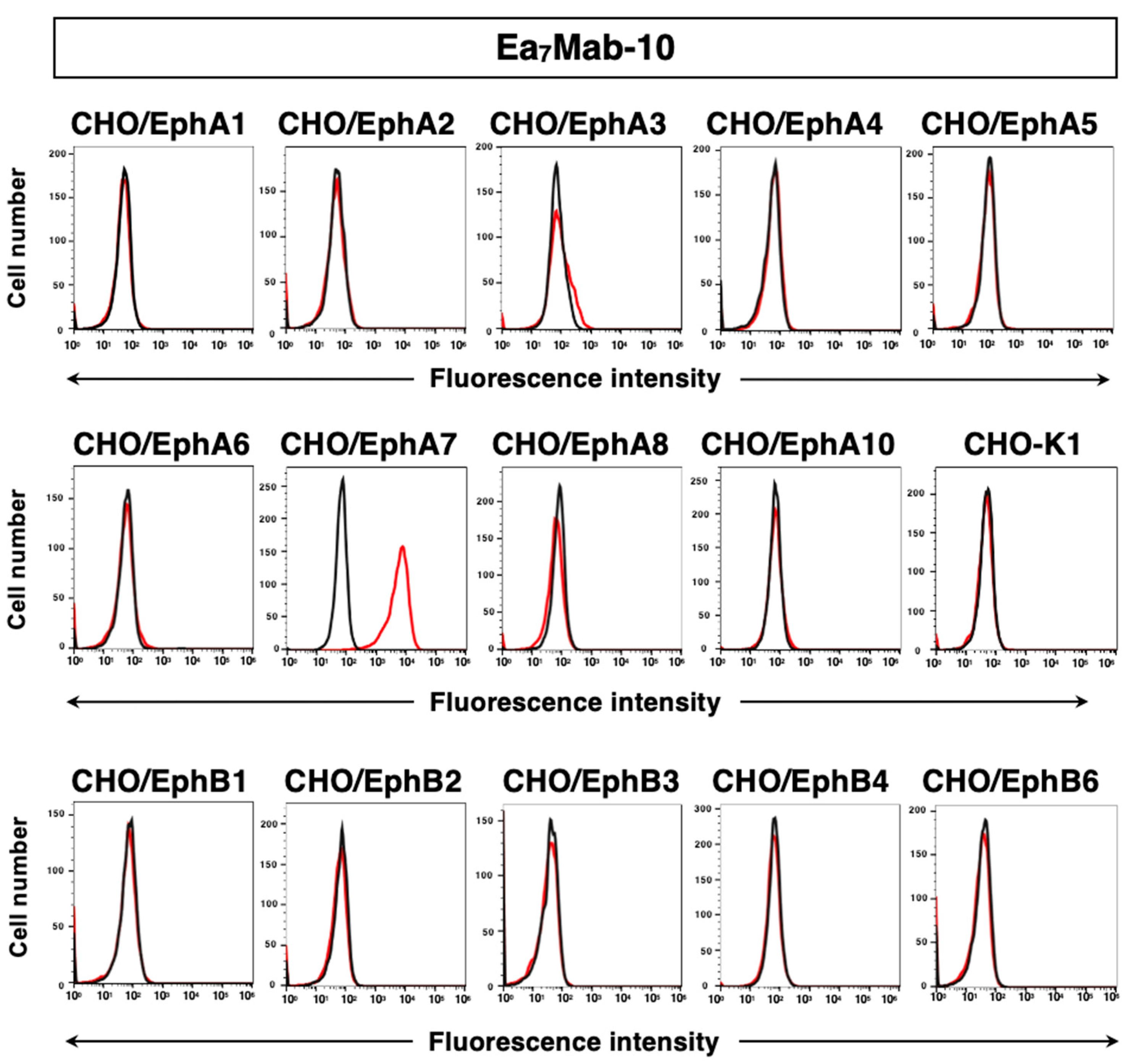
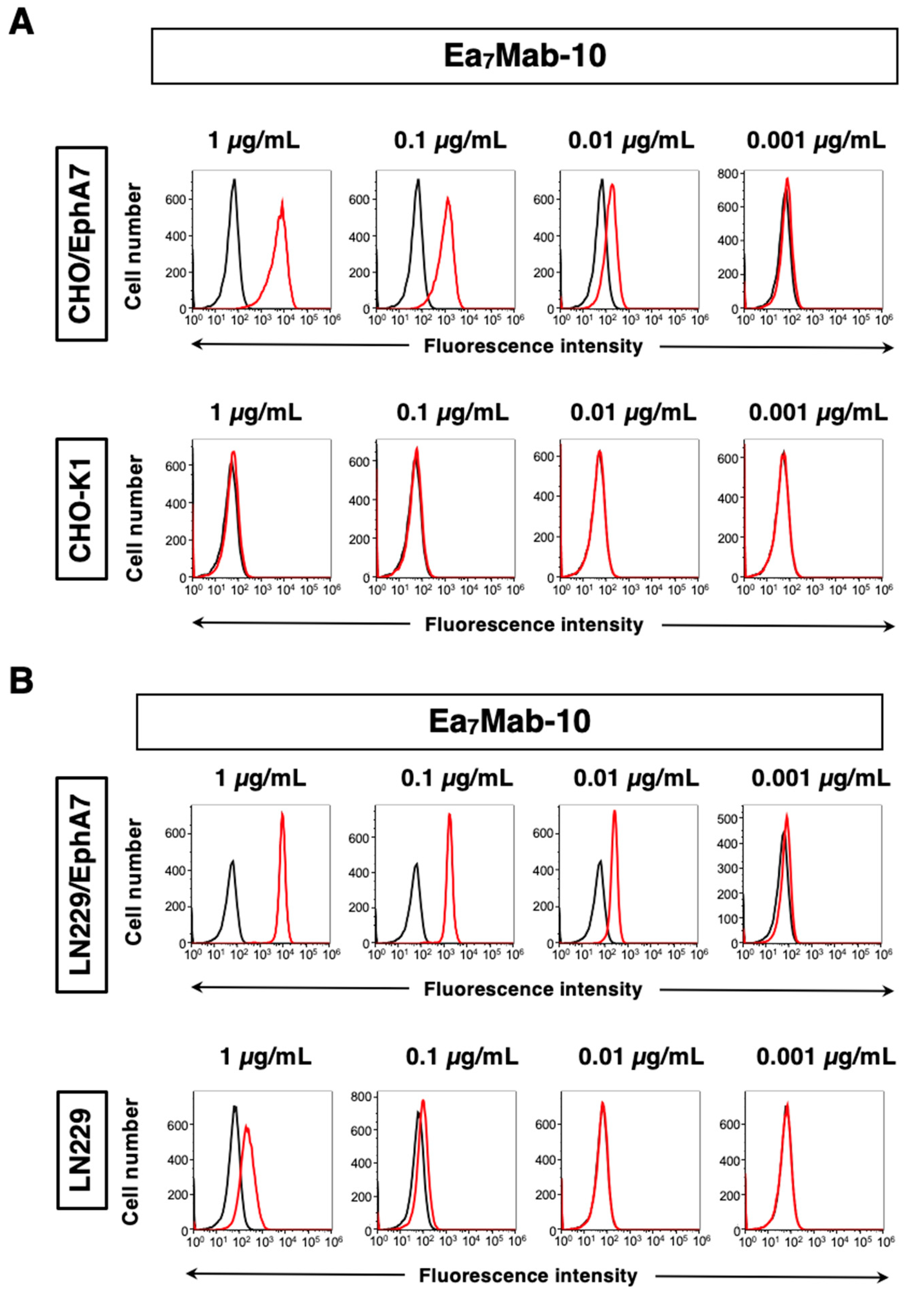
3.2. Investigation of the Reactivity of Ea7Mab-10 Using Flow Cytometry
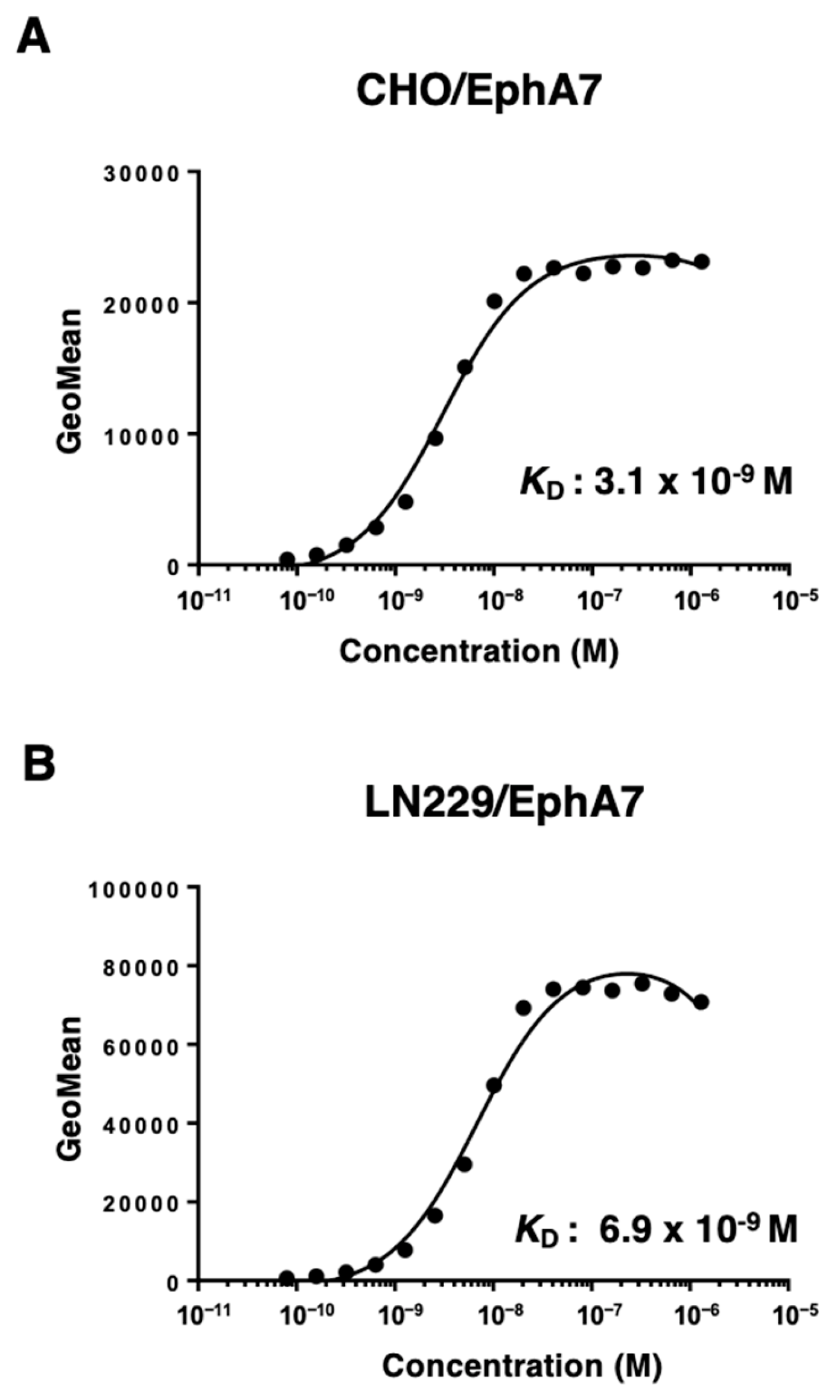
3.3. Calculation of the Binding Affinity of Ea7Mab-10 Using Flow Cytometry
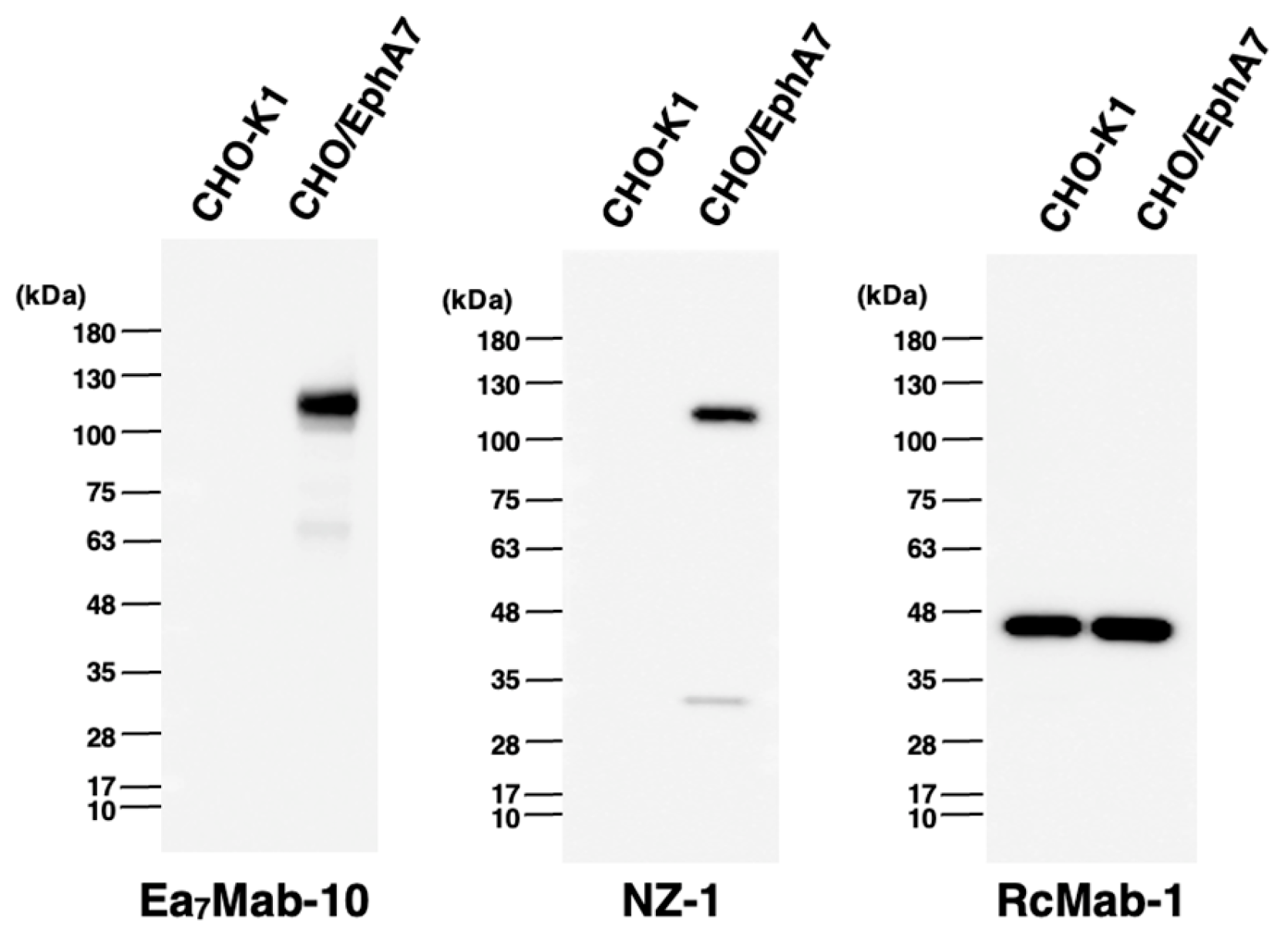
3.4. Western Blotting Using Ea7Mab-10
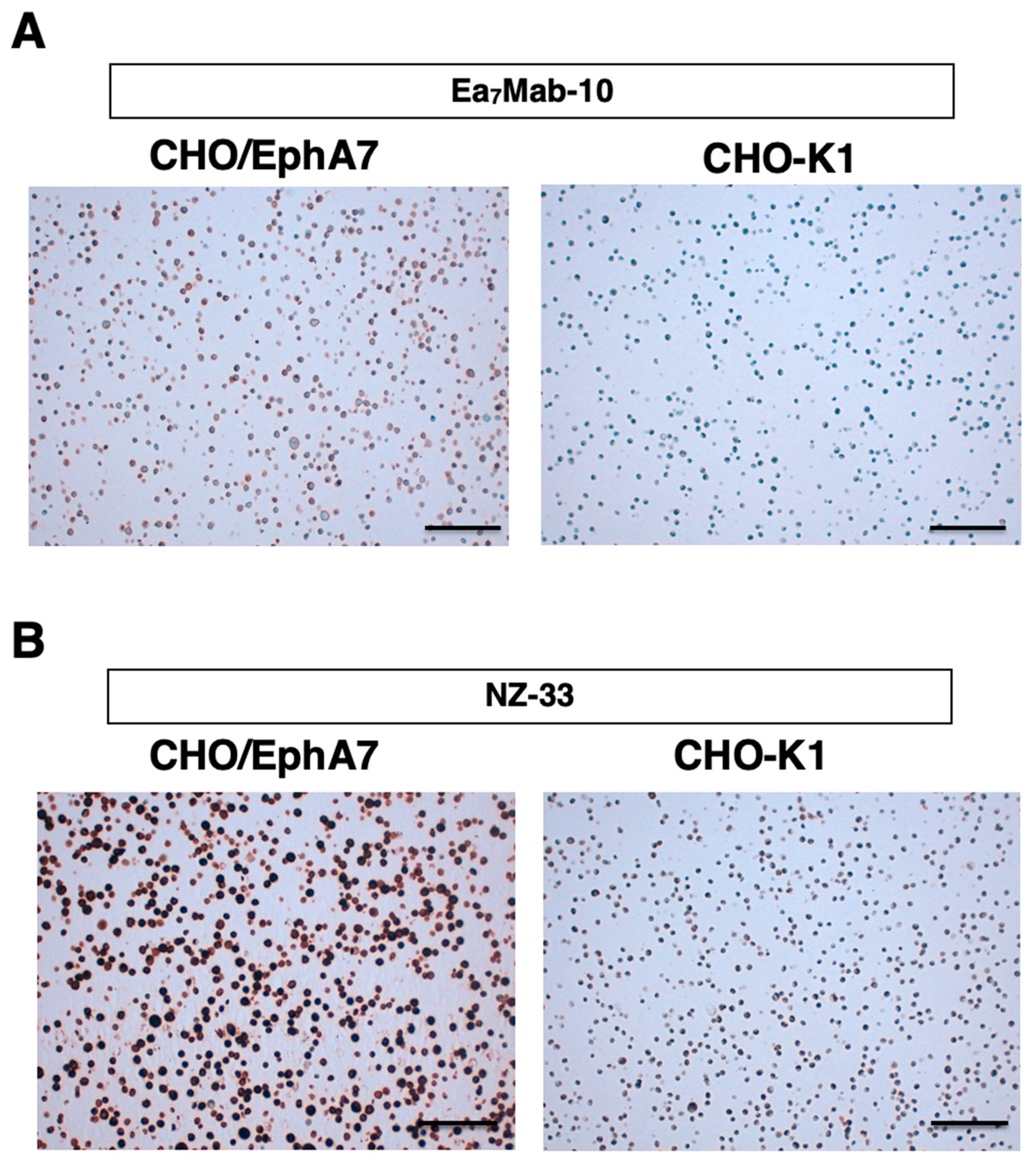
3.5. IHC Using Ea7Mab-10
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pasquale, E.B. Eph receptors and ephrins in cancer progression. Nat. Rev. Cancer 2024, 24, 5–27. [Google Scholar] [CrossRef] [PubMed]
- Pasquale, E.B. Eph receptor signaling complexes in the plasma membrane. Trends Biochem. Sci. 2024, 49, 1079–1096. [Google Scholar] [CrossRef] [PubMed]
- Bush, J.O. Cellular and molecular mechanisms of EPH/EPHRIN signaling in evolution and development. Curr Top Dev Biol 2022, 149, 153–201. [Google Scholar] [PubMed]
- Cayuso, J.; Xu, Q.; Wilkinson, D.G. Mechanisms of boundary formation by Eph receptor and ephrin signaling. Dev. Biol. 2015, 401, 122–131. [Google Scholar] [CrossRef]
- Rasool, D.; Jahani-Asl, A. Master regulators of neurogenesis: the dynamic roles of Ephrin receptors across diverse cellular niches. Transl. Psychiatry 2024, 14, 462. [Google Scholar] [CrossRef]
- Kania, A.; Klein, R. Mechanisms of ephrin-Eph signalling in development, physiology and disease. Nat. Rev. Mol. Cell Biol. 2016, 17, 240–256. [Google Scholar] [CrossRef]
- Zhu, Y.; Su, S.A.; Shen, J.; et al. Recent advances of the Ephrin and Eph family in cardiovascular development and pathologies. iScience 2024, 27, 110556. [Google Scholar] [CrossRef]
- Rudno-Rudzińska, J.; Kielan, W.; Frejlich, E.; et al. A review on Eph/ephrin, angiogenesis and lymphangiogenesis in gastric, colorectal and pancreatic cancers. Chin. J. Cancer Res. 2017, 29, 303–312. [Google Scholar] [CrossRef]
- Guo, X.; Yang, Y.; Tang, J.; Xiang, J. Ephs in cancer progression: complexity and context-dependent nature in signaling, angiogenesis and immunity. Cell Commun. Signal 2024, 22, 299. [Google Scholar] [CrossRef]
- Arora, S.; Scott, A.M.; Janes, P.W. Eph Receptors in Cancer. Biomedicines 2023, 11(2). [Google Scholar] [CrossRef]
- Valenzuela, D.M.; Rojas, E.; Griffiths, J.A.; et al. Identification of full-length and truncated forms of Ehk-3, a novel member of the Eph receptor tyrosine kinase family. Oncogene 1995, 10, 1573–1580. [Google Scholar] [PubMed]
- Lee, H.; Park, E.; Kim, Y.; Park, S. EphrinA5-EphA7 complex induces apoptotic cell death via TNFR1. Mol. Cells 2013, 35, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; Zhang, M.; Liu, L.; et al. ADAM10-cleaved ephrin-A5 contributes to prostate cancer metastasis. Cell Death Dis. 2022, 13, 453. [Google Scholar] [CrossRef]
- Li, S.; Wu, Z.; Ma, P.; et al. Ligand-dependent EphA7 signaling inhibits prostate tumor growth and progression. Cell Death Dis. 2017, 8, e3122. [Google Scholar] [CrossRef]
- Oricchio, E.; Nanjangud, G.; Wolfe, A.L.; et al. The Eph-receptor A7 is a soluble tumor suppressor for follicular lymphoma. Cell 2011, 147, 554–564. [Google Scholar] [CrossRef]
- Peifer, M.; Fernández-Cuesta, L.; Sos, M.L.; et al. Integrative genome analyses identify key somatic driver mutations of small-cell lung cancer. Nat. Genet. 2012, 44, 1104–1110. [Google Scholar] [CrossRef]
- Zhang, Z.; Wu, H.X.; Lin, W.H.; et al. EPHA7 mutation as a predictive biomarker for immune checkpoint inhibitors in multiple cancers. BMC Med. 2021, 19, 26. [Google Scholar] [CrossRef]
- He, Y.; Zhang, C.; Tan, L.; et al. Eph receptors activate myeloid checkpoint receptor LILRB5 to support tumor development. Cancer Immunol Res 2025. [CrossRef]
- Asano, T.; Ohishi, T.; Takei, J.; et al. Anti-HER3 monoclonal antibody exerts antitumor activity in a mouse model of colorectal adenocarcinoma. Oncol Rep 2021, 46(2). [Google Scholar] [CrossRef]
- Itai, S.; Yamada, S.; Kaneko, M.K.; et al. Establishment of EMab-134, a Sensitive and Specific Anti-Epidermal Growth Factor Receptor Monoclonal Antibody for Detecting Squamous Cell Carcinoma Cells of the Oral Cavity. Monoclon. Antib. Immunodiagn. Immunother. 2017, 36, 272–281. [Google Scholar] [CrossRef]
- Itai, S.; Fujii, Y.; Kaneko, M.K.; et al. H(2)Mab-77 is a Sensitive and Specific Anti-HER2 Monoclonal Antibody Against Breast Cancer. Monoclon. Antib. Immunodiagn. Immunother. 2017, 36, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Ubukata, R.; Suzuki, H.; Hirose, M.; et al. Establishment of a highly sensitive and specific anti-EphB2 monoclonal antibody (Eb2Mab-12) for flow cytometry. MI 2025. [CrossRef]
- Tanaka, T.; Kaneko, Y.; Yamamoto, H.; et al. Development of a novel anti-erythropoietin-producing hepatocellular receptor B6 monoclonal antibody Eb(6)Mab-3 for flow cytometry. Biochem Biophys Rep 2025, 41, 101960. [Google Scholar] [PubMed]
- Satofuka, H.; Suzuki, H.; Tanaka, T.; et al. Development of an anti-human EphA2 monoclonal antibody Ea2Mab-7 for multiple applications. Biochemistry and Biophysics Reports 2025, 42, 10998. [Google Scholar] [CrossRef]
- Fujii, Y.; Kaneko, M.K.; Kato, Y. MAP Tag: A Novel Tagging System for Protein Purification and Detection. Monoclon. Antib. Immunodiagn. Immunother. 2016, 35, 293–299. [Google Scholar] [CrossRef]
- Fujii, Y.; Kaneko, M.; Neyazaki, M.; et al. PA tag: a versatile protein tagging system using a super high affinity antibody against a dodecapeptide derived from human podoplanin. Protein Expr Purif 2014, 95, 240–247. [Google Scholar] [CrossRef]
- Ikota, H.; Nobusawa, S.; Arai, H.; et al. Evaluation of IDH1 status in diffusely infiltrating gliomas by immunohistochemistry using anti-mutant and wild type IDH1 antibodies. Brain Tumor Pathol. 2015, 32, 237–244. [Google Scholar] [CrossRef]
- Chen, X.; Yu, D.; Zhou, H.; et al. The role of EphA7 in different tumors. Clin. Transl. Oncol. 2022, 24, 1274–1289. [Google Scholar] [CrossRef]
- Bai, Y.Q.; Zhang, J.Y.; Bai, C.Y.; et al. Low EphA7 Expression Correlated with Lymph Node Metastasis and Poor Prognosis of Patients with Esophageal Squamous Cell Carcinoma. Acta Histochem. Cytochem. 2015, 48, 75–81. [Google Scholar] [CrossRef]
- Wang, L.F.; Fokas, E.; Juricko, J.; et al. Increased expression of EphA7 correlates with adverse outcome in primary and recurrent glioblastoma multiforme patients. BMC Cancer 2008, 8, 79. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



