Submitted:
27 April 2025
Posted:
28 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction to Centrioles and Centrosomes
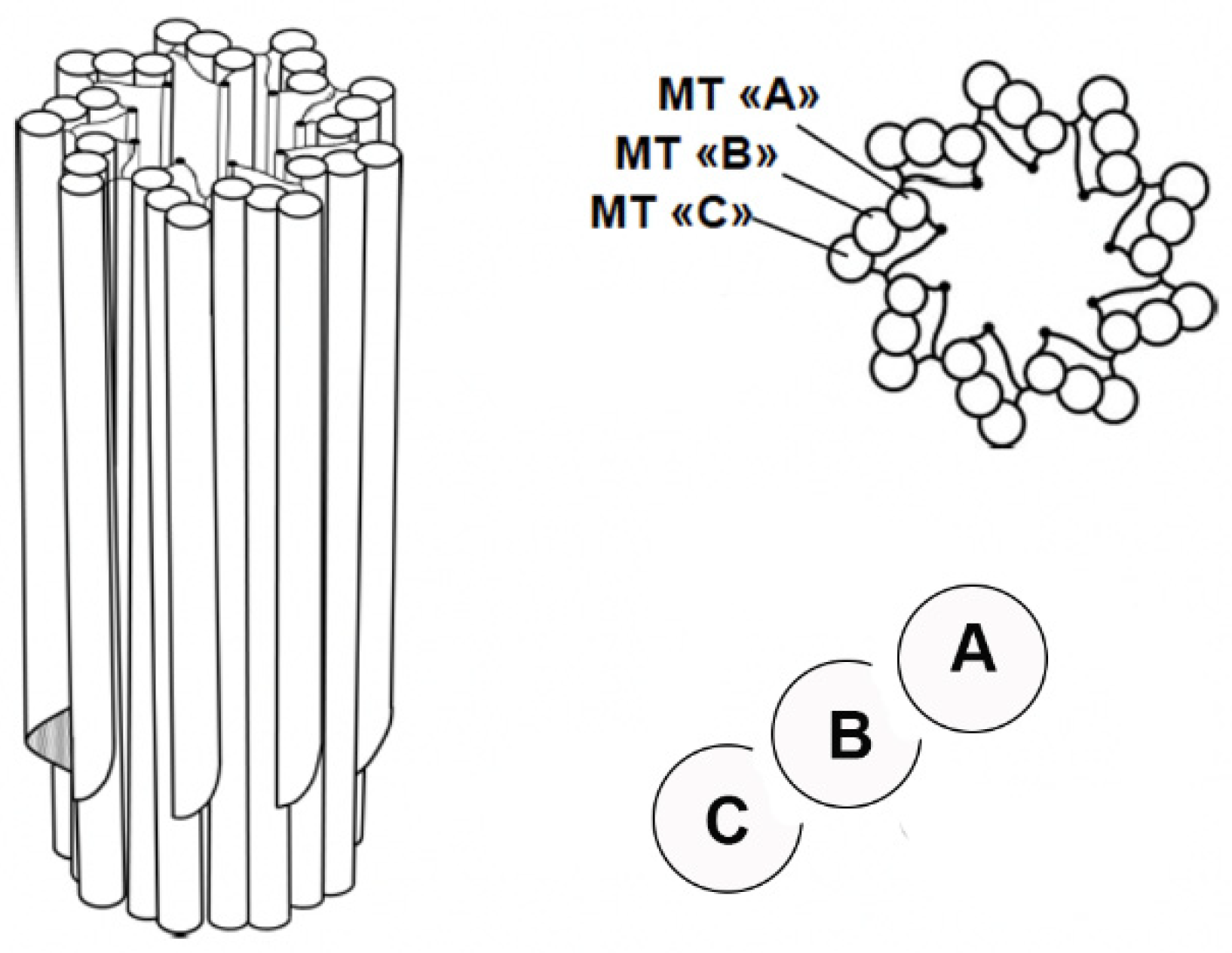
1.1. Structure of Centrioles
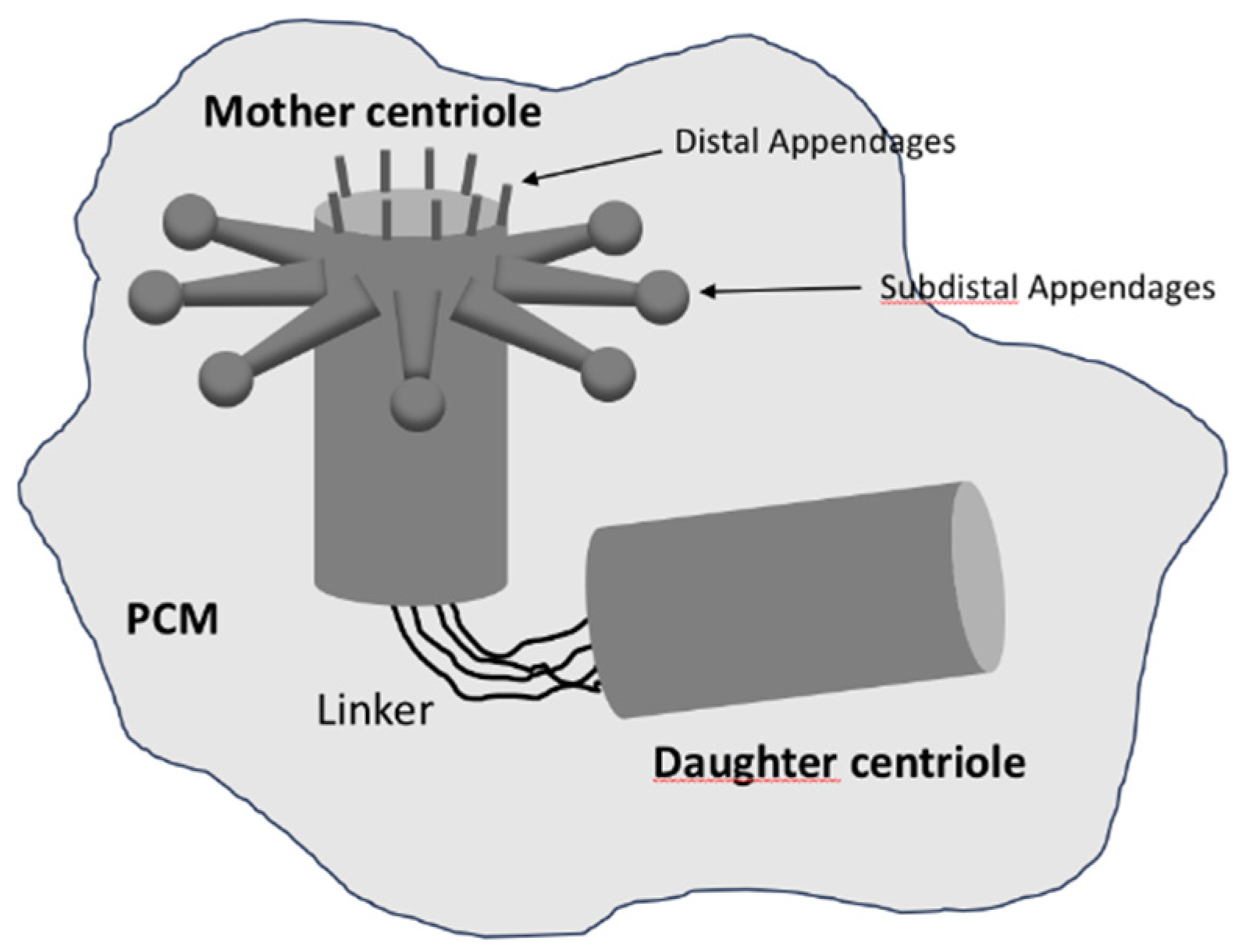
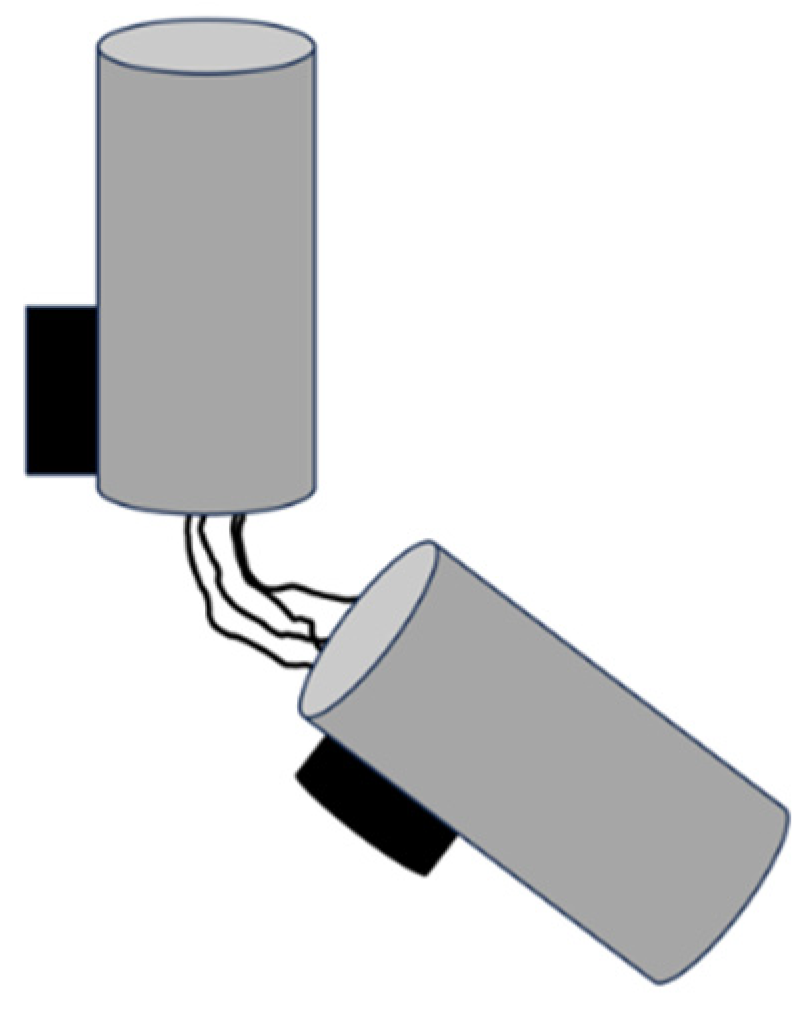
1.2. The Centrosome and Its Function
1.3. Centriole Duplication: A Brief Overview
1.4. The Importance of Centriole Number and Integrity
1.5. Historical Perspective and Discovery of Centrioles
1.6. Relevance of Centriole Research in Disease and Therapeutics
Summary
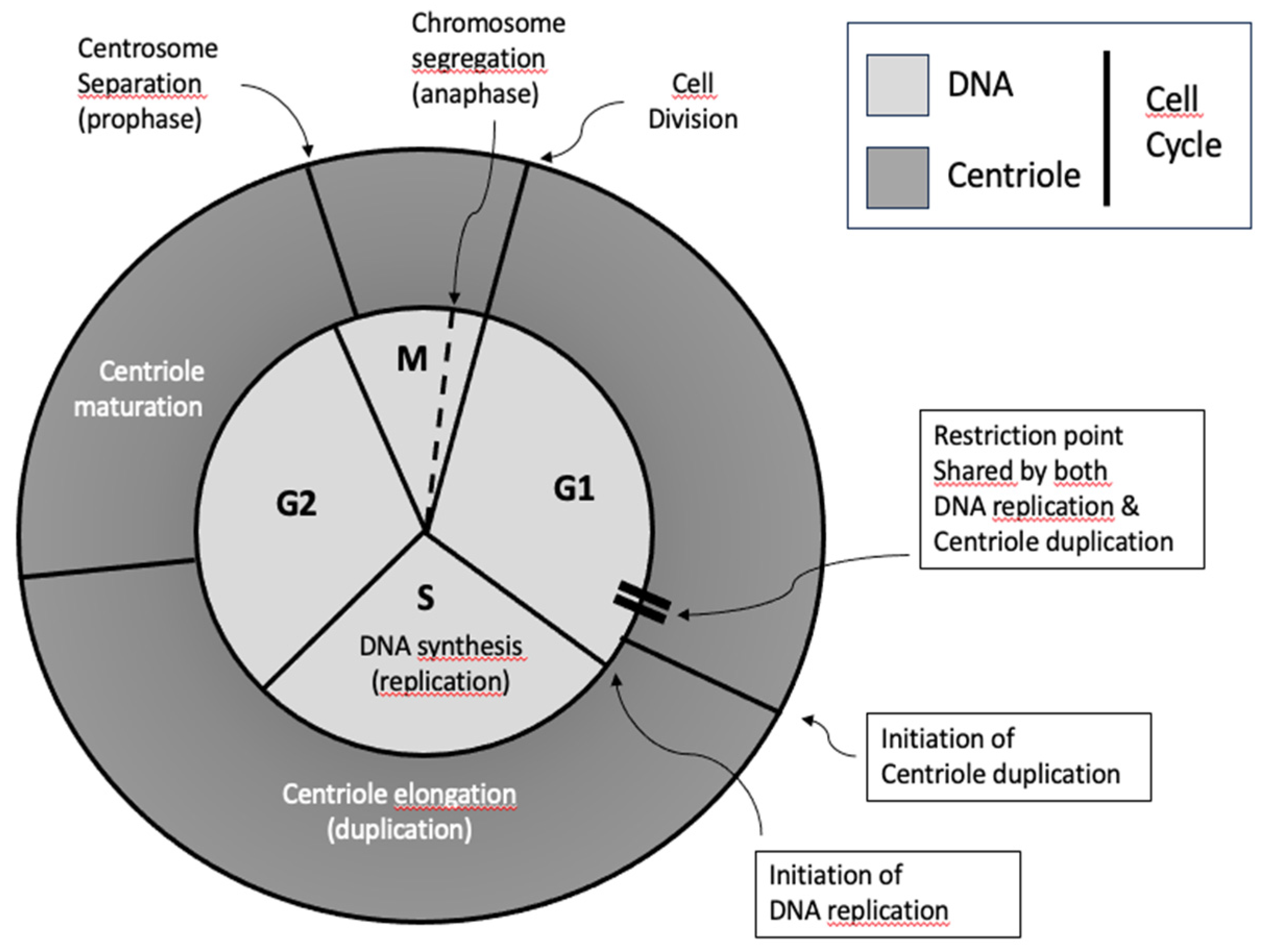
2. Overview of the Centriole Duplication Process
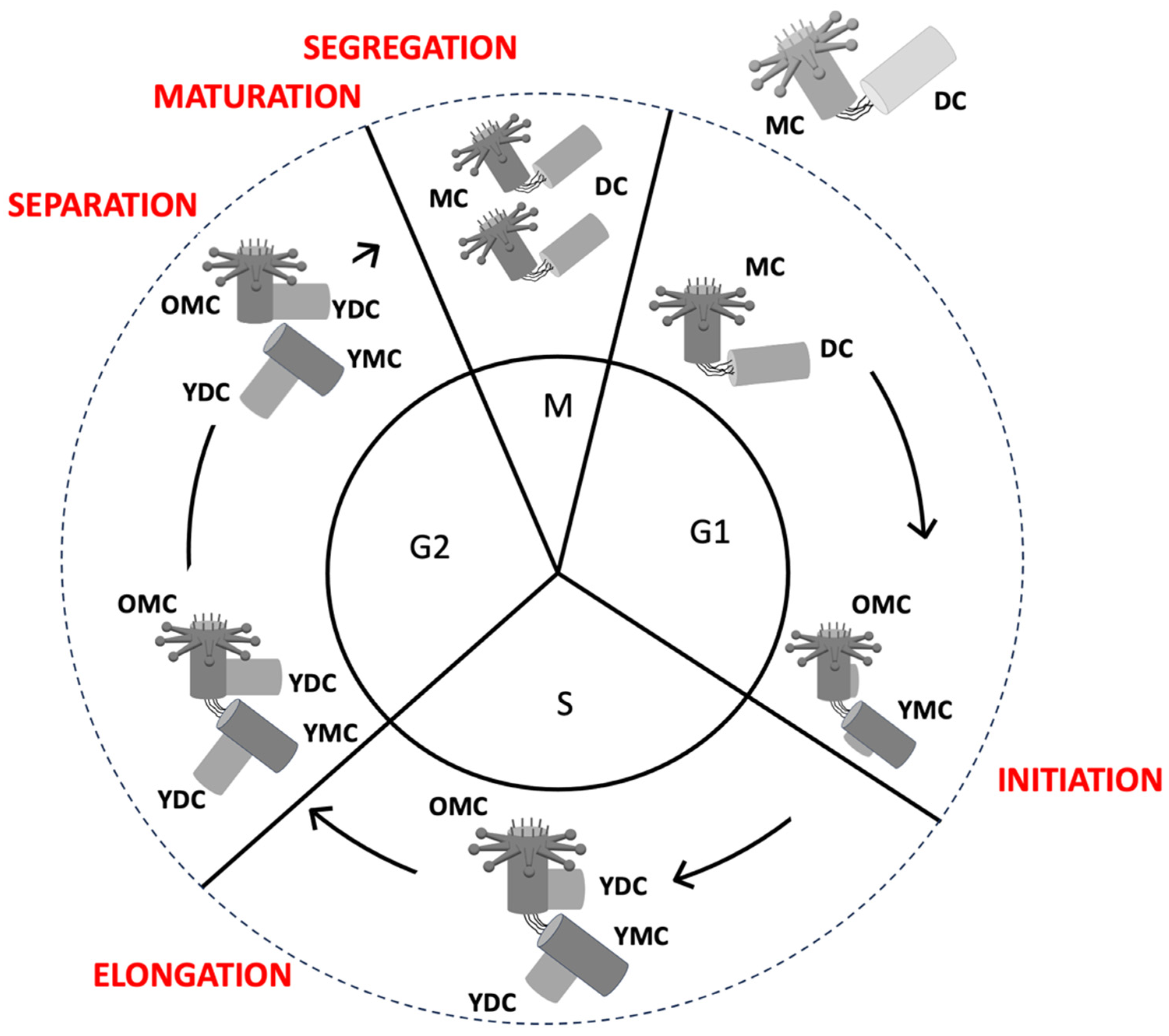
2.1. The Centriole Duplication Cycle
Key Phases of Centriole Duplication:
2.2. Key Stages of Centriole Duplication
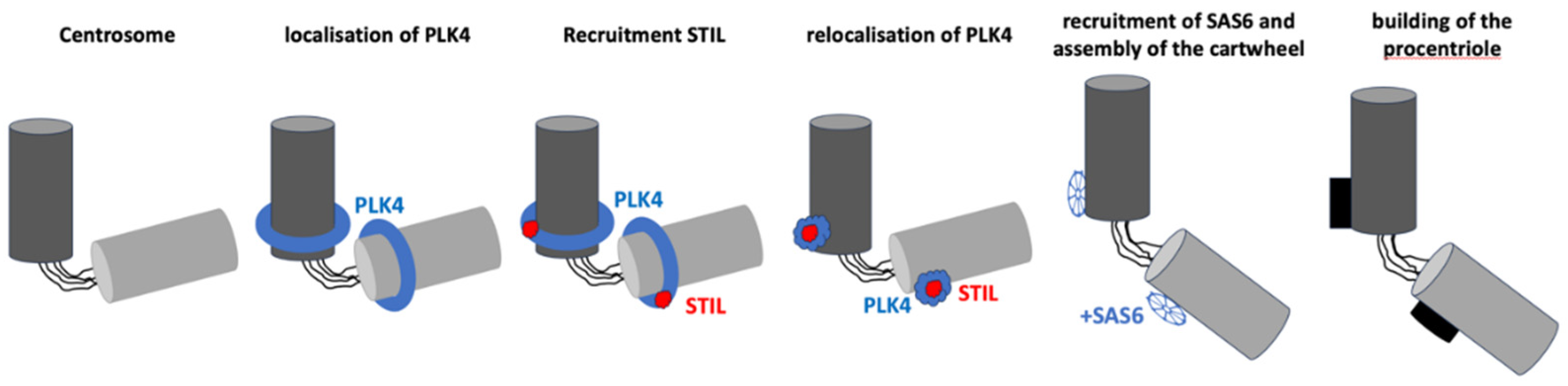
2.2.1. Initiation: Formation of the Procentriole
2.2.2. Elongation: Growth of the Procentriole
2.2.3. Maturation: Preparation for Function
2.2.4. Separation: Distribution to Daughter Cells
2.3. Mechanisms Preventing Overduplicatio
Conclusion
3. Key Molecular Players in Centriole Duplication
3.1. PLK4
3.2. STIL
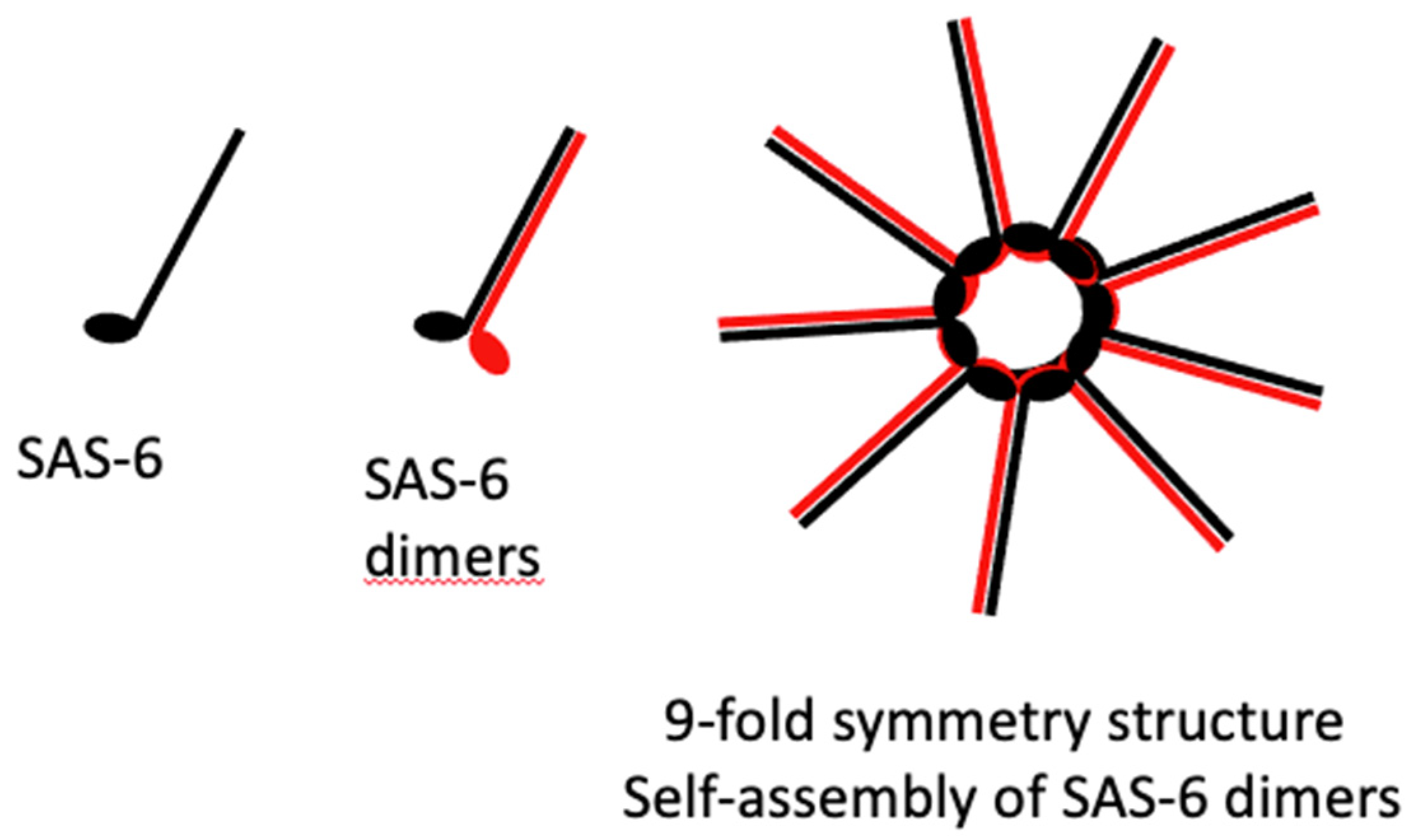
3.3. SAS-6
3.4. CPAP
3.5. CEP135
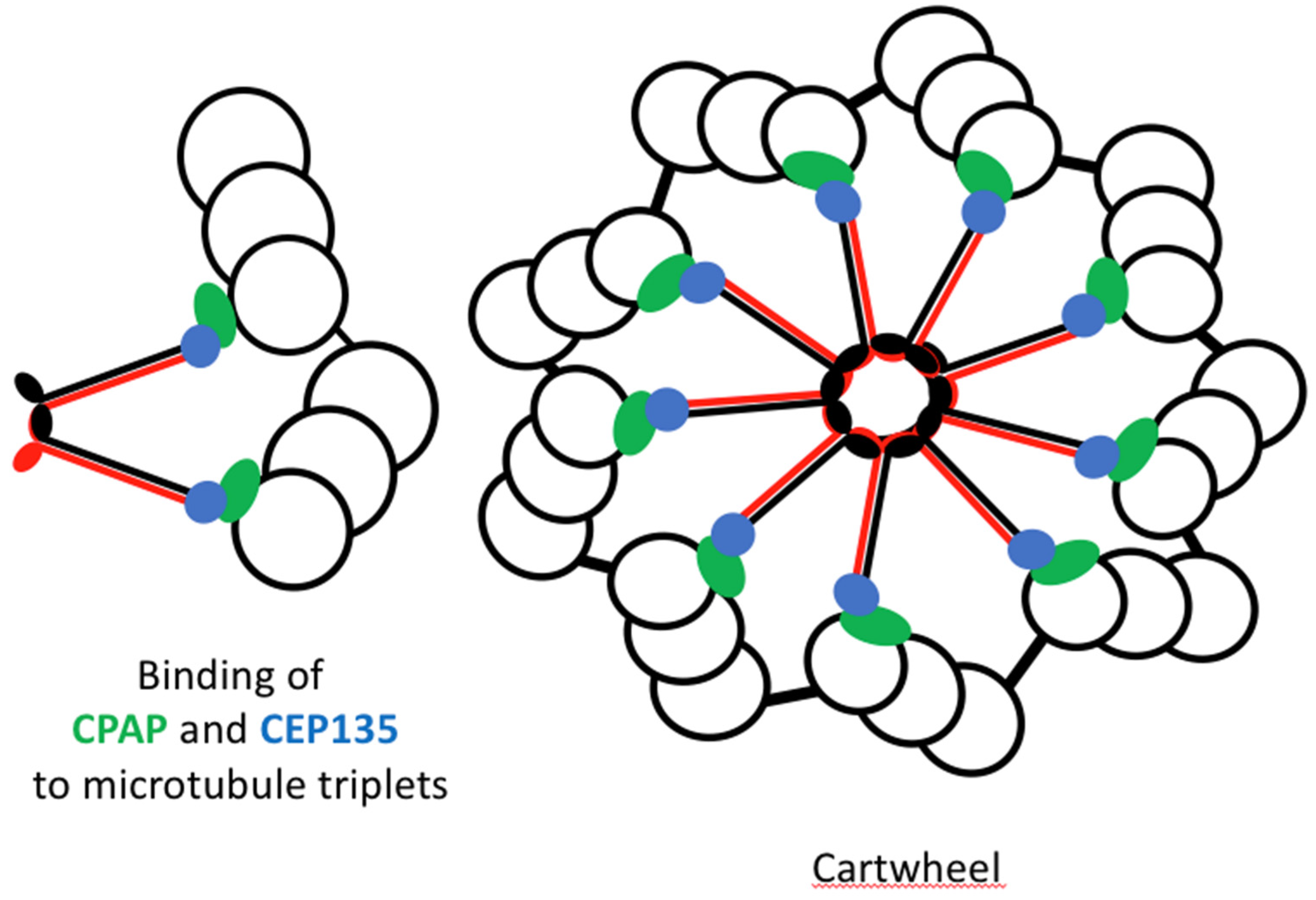
3.6. Other Proteins Involved in Centriole Biogenesis and Maintenance
4. Regulation of Centriole Duplication
4.1. Licensing and Temporal Control
4.1.1. Licensing Mechanisms
4.1.2. Preventing Overduplication
4.1.3. Checkpoints in Centriole Duplication
4.2. Ubiquitination and Proteasomal Degradation
4.3. Negative Regulation by p53 and p21
4.4. Centriole Re-Duplication Block
5. Aberrant Centriole Duplication and Its Consequences
5.1. Centrosome Amplification
5.1.1. Dysregulation of Centriole Duplication Proteins
5.1.2. Causes of Amplification
5.2. Cellular Consequences
5.2.1. Formation of Multipolar Spindles
5.2.2. Cancer and Chromosomal Instability
5.3. Microcephaly and Developmental Defects
6. Centriole Duplication and Disease
6.1. Cancer
6.1.1. Therapeutic Targeting
6.2. Neurological Disorders
6.3. Ciliopathies
7. Recent Advances in Visualizing Centriole
8. Future Directions in Centriole Duplication Research
9. Conclusions
References
- Bornens M. The centrosome in cells and organisms. Science. 2012 Jan 27;335(6067):422-6. [CrossRef]
- Tassin AM, Bornens M. Centrosome structure and microtubule nucleation in animal cells. Biol Cell. 1999 May-Jun;91(4-5):343-54.
- Boveri, T. (1914). Zur Frage der Entstehung maligner Tumoren. Jena: Gustav Fischer.
- Qi F, Zhou J. Multifaceted roles of centrosomes in development, health, and disease. J Mol Cell Biol. 2021 Dec 6;13(9):611-621. [CrossRef]
- Kiermaier E, Stötzel I, Schapfl MA, Villunger A. Amplified centrosomes-more than just a threat. EMBO Rep. 2024 Sep 16. [CrossRef]
- Kitagawa D, Vakonakis I, Olieric N, Hilbert M, Keller D, Olieric V, Bortfeld M, Erat MC, Flückiger I, Gönczy P, Steinmetz MO. Structural basis of the 9-fold symmetry of centrioles. Cell. 2011 Feb 4;144(3):364-75. [CrossRef]
- Le Guennec M, Klena N, Aeschlimann G, Hamel V, Guichard P. Overview of the centriole architecture. Curr Opin Struct Biol. 2021 Feb;66:58-65. [CrossRef]
- Nigg EA, Raff JW. Centrioles, centrosomes, and cilia in health and disease. Cell. 2009 Nov 13;139(4):663-78. [CrossRef]
- Blanco-Ameijeiras J, Lozano-Fernández P, Martí E. Centrosome maturation—in tune with the cell cycle. J Cell Sci. 2022 Jan 15;135(2):jcs259395. [CrossRef]
- Kobayashi T, Dynlacht BD. Regulating the transition from centriole to basal body. J Cell Biol. 2011 May 2;193(3):435-44. [CrossRef]
- Mitchison T, Kirschner M. Dynamic instability of microtubule growth. Nature. 1984 Nov 15-21;312(5991):237-42. [CrossRef]
- Godinho SA, Pellman D. Causes and consequences of centrosome abnormalities in cancer. Philos Trans R Soc Lond B Biol Sci. 2014 Sep 5;369(1650):20130467. [CrossRef]
- Nam HJ, Naylor RM, van Deursen JM. Centrosome dynamics as a source of chromosomal instability. Trends Cell Biol. 2015 Feb;25(2):65-73. [CrossRef]
- Sánchez I, Dynlacht BD. Cilium assembly and disassembly. Nat Cell Biol. 2016 Jun 28;18(7):711-7. [CrossRef]
- Basto R, Lau J, Vinogradova T, Gardiol A, Woods CG, Khodjakov A, Raff JW. Flies without centrioles. Cell. 2006 Jun 30;125(7):1375-86. [CrossRef]
- Vorobjev IA, Chentsov YuS. Centrioles in the cell cycle. I. Epithelial cells. J Cell Biol. 1982 Jun;93(3):938-49. [CrossRef]
- Tsou MF, Stearns T. Mechanism limiting centrosome duplication to once per cell cycle. Nature. 2006 Aug 24;442(7105):947-51. [CrossRef]
- Lingle WL, Barrett SL, Negron VC, D’Assoro AB, Boeneman K, Liu W, Whitehead CM, Reynolds C, Salisbury JL. Centrosome amplification drives chromosomal instability in breast tumor development. Proc Natl Acad Sci U S A. 2002 Feb 19;99(4):1978-83. [CrossRef]
- Vásquez-Limeta A, Lukasik K, Kong D, Sullenberger C, Luvsanjav D, Sahabandu N, Chari R, Loncarek J. CPAP insufficiency leads to incomplete centrioles that duplicate but fragment. J Cell Biol. 2022 May 2;221(5):e202108018. [CrossRef]
- Martin CA, Ahmad I, Klingseisen A, Hussain MS, Bicknell LS, Leitch A, Nürnberg G, Toliat MR, Murray JE, Hunt D, Khan F, Ali Z, Tinschert S, Ding J, Keith C, Harley ME, Heyn P, Müller R, Hoffmann I, Cormier-Daire V, Dollfus H, Dupuis L, Bashamboo A, McElreavey K, Kariminejad A, Mendoza-Londono R, Moore AT, Saggar A, Schlechter C, Weleber R, Thiele H, Altmüller J, Höhne W, Hurles ME, Noegel AA, Baig SM, Nürnberg P, Jackson AP. Mutations in PLK4, encoding a master regulator of centriole biogenesis, cause microcephaly, growth failure and retinopathy. Nat Genet. 2014 Dec;46(12):1283-1292. [CrossRef]
- Sladky VC, Akbari H, Tapias-Gomez D, Evans LT, Drown CG, Strong MA, LoMastro GM, Larman T, Holland AJ. Centriole signaling restricts hepatocyte ploidy to maintain liver integrity. Genes Dev. 2022 Aug 18;36(13-14):843–56. [CrossRef]
- Boveri T 1888 Zellen-Studien Heft 2, Die Befruchtung und Teilung des Eles von Ascaris megalocephala II. Verlag von Gustav Fischer, Jena, Germany.
- Boveri T. 1900 Ueber die Natur de Centrosomen Zellen-Studien 4. Jena, Germany: G Fisher.
- Basto R, Brunk K, Vinadogrova T, Peel N, Franz A, Khodjakov A, Raff JW. Centrosome amplification can initiate tumorigenesis in flies. Cell. 2008 Jun 13;133(6):1032-42. [CrossRef]
- Dzafic E, Strzyz PJ, Wilsch-Bräuninger M, Norden C. Centriole Amplification in Zebrafish Affects Proliferation and Survival but Not Differentiation of Neural Progenitor Cells. Cell Rep. 2015 Oct 6;13(1):168-182. [CrossRef]
- Levine MS, Bakker B, Boeckx B, Moyett J, Lu J, Vitre B, Spierings DC, Lansdorp PM, Cleveland DW, Lambrechts D, Foijer F, Holland AJ. Centrosome Amplification Is Sufficient to Promote Spontaneous Tumorigenesis in Mammals. Dev Cell. 2017 Feb 6;40(3):313-322.e5. [CrossRef]
- D’Assoro AB, Lingle WL, Salisbury JL. Centrosome amplification and the development of cancer. Oncogene. 2002 Sep 9;21(40):6146-53. [CrossRef]
- Turan MG, Orhan ME, Cevik S, Kaplan OI. CiliaMiner: an integrated database for ciliopathy genes and ciliopathies. Database (Oxford). 2023 Jul 26;2023:baad047. [CrossRef]
- Holland AJ, Cleveland DW. Polo-like kinase 4 inhibition: a strategy for cancer therapy? Cancer Cell. 2014 Aug 11;26(2):151-3. [CrossRef]
- Lei Q, Yu Q, Yang N, Xiao Z, Song C, Zhang R, Yang S, Liu Z, Deng H. Therapeutic potential of targeting polo-like kinase 4. Eur J Med Chem. 2024 Feb 5;265:116115. [CrossRef]
- Lacey KR, Jackson PK, Stearns T. Cyclin-dependent kinase control of centrosome duplication. Proc Natl Acad Sci U S A. 1999 Mar 16;96(6):2817-22. [CrossRef]
- Nigg EA, Holland AJ. Once and only once: mechanisms of centriole duplication and their deregulation in disease. Nat Rev Mol Cell Biol. 2018 May;19(5):297-312. [CrossRef]
- Kuriyama R, Dasgupta S, Borisy GG. Independence of centriole formation and initiation of DNA synthesis in Chinese hamster ovary cells. Cell Motil Cytoskeleton. 1986;6(4):355-62. [CrossRef]
- Krämer A, Neben K, Ho AD. Centrosome replication, genomic instability and cancer. Leukemia. 2002 May;16(5):767-75. [CrossRef]
- Ko MJ, Murata K, Hwang DS, Parvin JD. Inhibition of BRCA1 in breast cell lines causes the centrosome duplication cycle to be disconnected from the cell cycle. Oncogene. 2006 Jan 12;25(2):298-303. [CrossRef]
- Durcan TM, Halpin ES, Casaletti L, Vaughan KT, Pierson MR, Woods S, Hinchcliffe EH. Centrosome duplication proceeds during mimosine-induced G1 cell cycle arrest. J Cell Physiol. 2008 Apr;215(1):182-91. [CrossRef]
- Gönczy P, Hatzopoulos GN. Centriole assembly at a glance. J Cell Sci. 2019 Feb 20;132(4):jcs228833. [CrossRef]
- Schmidt TI, Kleylein-Sohn J, Westendorf J, Le Clech M, Lavoie SB, Stierhof YD, Nigg EA. Control of centriole length by CPAP and CP110. Curr Biol. 2009 Jun 23;19(12):1005-11. [CrossRef]
- Sharma A, Olieric N, Steinmetz MO. Centriole length control. Curr Opin Struct Biol. 2021 Feb;66:89-95. [CrossRef]
- Sullenberger C, Vasquez-Limeta A, Kong D, Loncarek J. With Age Comes Maturity: Biochemical and Structural Transformation of a Human Centriole in the Making. Cells. 2020 Jun 9;9(6):1429. [CrossRef]
- Faruki S, Cole RW, Rieder CL. Separating centrosomes interact in the absence of associated chromosomes during mitosis in cultured vertebrate cells. Cell Motil Cytoskeleton. 2002 Jun;52(2):107-21. [CrossRef]
- Agircan FG, Schiebel E, Mardin BR. Separate to operate: control of centrosome positioning and separation. Philos Trans R Soc Lond B Biol Sci. 2014 Sep 5;369(1650):20130461. [CrossRef]
- Habedanck R, Stierhof YD, Wilkinson CJ, Nigg EA. The Polo kinase Plk4 functions in centriole duplication. Nat Cell Biol. 2005 Nov;7(11):1140-6. [CrossRef]
- Holland AJ, Fachinetti D, Da Cruz S, Q, Vitre B, Lince-Faria M, Chen D, Parish N, Verma IM, Bettencourt-Dias M, Cleveland DW. Polo-like kinase 4 controls centriole duplication but does not directly regulate cytokinesis. Mol Biol Cell. 2012 May;23(10):1838-45. [CrossRef]
- Tang CJ, Lin SY, Hsu WB, Lin YN, Wu CT, Lin YC, Chang CW, Wu KS, Tang TK. The human microcephaly protein STIL interacts with CPAP and is required for procentriole formation. EMBO J. 2011 Oct 21;30(23):4790-804. [CrossRef]
- Yoshiba S, Tsuchiya Y, Ohta M, Gupta A, Shiratsuchi G, Nozaki Y, Ashikawa T, Fujiwara T, Natsume T, Kanemaki MT, Kitagawa D. HsSAS-6-dependent cartwheel assembly ensures stabilization of centriole intermediates. J Cell Sci. 2019 Jun 20;132(12):jcs217521. [CrossRef]
- Hatch EM, Kulukian A, Holland AJ, Cleveland DW, Stearns T. Cep152 interacts with Plk4 and is required for centriole duplication. J Cell Biol. 2010 Nov 15;191(4):721-9. [CrossRef]
- Sonnen KF, Gabryjonczyk AM, Anselm E, Stierhof YD, Nigg EA. Human Cep192 and Cep152 cooperate in Plk4 recruitment and centriole duplication. J Cell Sci. 2013 Jul 15;126(Pt 14):3223-33. [CrossRef]
- Tang CJ, Fu RH, Wu KS, Hsu WB, Tang TK. CPAP is a cell-cycle regulated protein that controls centriole length. Nat Cell Biol. 2009 Jul;11(7):825-31. [CrossRef]
- Inanç B, Pütz M, Lalor P, Dockery P, Kuriyama R, Gergely F, Morrison CG. Abnormal centrosomal structure and duplication in Cep135-deficient vertebrate cells. Mol Biol Cell. 2013 Sep;24(17):2645-54. [CrossRef]
- Gould RR, Borisy GG. The pericentriolar material in Chinese hamster ovary cells nucleates microtubule formation. J Cell Biol. 1977 Jun;73(3):601-15. [CrossRef]
- Marshall WF. Basal bodies platforms for building cilia. Curr Top Dev Biol. 2008;85:1-22. [CrossRef]
- Ma D, Wang F, Teng J, Huang N, Chen J. Structure and function of distal and subdistal appendages of the mother centriole. J Cell Sci. 2023 Feb 1;136(3):jcs260560. [CrossRef]
- Woodruff JB, Wueseke O, Hyman AA. Pericentriolar material structure and dynamics. Philos Trans R Soc Lond B Biol Sci. 2014 Sep 5;369(1650):20130459. [CrossRef]
- Uzbekov R, Kireyev I, Prigent C. Centrosome separation: respective role of microtubules and actin filaments. Biol Cell. 2002 Sep;94(4-5):275-88. [CrossRef]
- Giet R, Petretti C, Prigent C. Aurora kinases, aneuploidy and cancer, a coincidence or a real link? Trends Cell Biol. 2005 May;15(5):241-50. [CrossRef]
- Wang X, Yang Y, Duan Q, Jiang N, Huang Y, Darzynkiewicz Z, Dai W. sSgo1, a major splice variant of Sgo1, functions in centriole cohesion where it is regulated by Plk1. Dev Cell. 2008 Mar;14(3):331-41. [CrossRef]
- Korzeniewski N, Zheng L, Cuevas R, Parry J, Chatterjee P, Anderton B, Duensing A, Münger K, Duensing S. Cullin 1 functions as a centrosomal suppressor of centriole multiplication by regulating polo-like kinase 4 protein levels. Cancer Res. 2009 Aug 15;69(16):6668-75. [CrossRef]
- Arquint C, Nigg EA. The PLK4-STIL-SAS-6 module at the core of centriole duplication. Biochem Soc Trans. 2016 Oct 15;44(5):1253-1263. [CrossRef]
- Blow JJ, Laskey RA. A role for the nuclear envelope in controlling DNA replication within the cell cycle. Nature. 1988 Apr 7;332(6164):546-8. [CrossRef]
- Zitouni S, Nabais C, Jana SC, Guerrero A, Bettencourt-Dias M. Polo-like kinases: structural variations lead to multiple functions. Nat Rev Mol Cell Biol. 2014 Jul;15(7):433-52. [CrossRef]
- Vakonakis I. The centriolar cartwheel structure: symmetric, stacked, and polarized. Curr Opin Struct Biol. 2021 Feb;66:1-7. [CrossRef]
- Ohta M, Ashikawa T, Nozaki Y, Kozuka-Hata H, Goto H, Inagaki M, Oyama M, Kitagawa D. Direct interaction of Plk4 with STIL ensures formation of a single procentriole per parental centriole. Nat Commun. 2014 Oct 24;5:5267. [CrossRef]
- Arquint C, Gabryjonczyk AM, Imseng S, Böhm R, Sauer E, Hiller S, Nigg EA, Maier T. STIL binding to Polo-box 3 of PLK4 regulates centriole duplication. Elife. 2015 Jul 18;4:e07888. [CrossRef]
- Cunha-Ferreira I, Rodrigues-Martins A, Bento I, Riparbelli M, Zhang W, Laue E, Callaini G, Glover DM, Bettencourt-Dias M. The SCF/Slimb ubiquitin ligase limits centrosome amplification through degradation of SAK/PLK4. Curr Biol. 2009 Jan 13;19(1):43-9. [CrossRef]
- Čajánek L, Glatter T, Nigg EA. The E3 ubiquitin ligase Mib1 regulates Plk4 and centriole biogenesis. J Cell Sci. 2015 May 1;128(9):1674-82. [CrossRef]
- Grossmann J, Kratz AS, Kordonsky A, Prag G, Hoffmann I. CRL4DCAF1 ubiquitin ligase regulates PLK4 protein levels to prevent premature centriole duplication. Life Sci Alliance. 2024 Mar 15;7(6):e202402668. [CrossRef]
- Guderian G, Westendorf J, Uldschmid A, Nigg EA. Plk4 trans-autophosphorylation regulates centriole number by controlling betaTrCP-mediated degradation. J Cell Sci. 2010 Jul 1;123(Pt 13):2163-9. [CrossRef]
- Cunha-Ferreira I, Bento I, Pimenta-Marques A, Jana SC, Lince-Faria M, Duarte P, Borrego-Pinto J, Gilberto S, Amado T, Brito D, Rodrigues-Martins A, Debski J, Dzhindzhev N, Bettencourt-Dias M. Regulation of autophosphorylation controls PLK4 self-destruction and centriole number. Curr Biol. 2013 Nov 18;23(22):2245-2254. [CrossRef]
- Rodrigues-Martins A, Riparbelli M, Callaini G, Glover DM, Bettencourt-Dias M. Revisiting the role of the mother centriole in centriole biogenesis. Science. 2007 May 18;316(5827):1046-50. [CrossRef]
- Shamir M, Martin FJO, Woolfson DN, Friedler A. Molecular Mechanism of STIL Coiled-Coil Domain Oligomerization. Int J Mol Sci. 2023 Sep 27;24(19):14616. [CrossRef]
- Kratz AS, Bärenz F, Richter KT, Hoffmann I. Plk4-dependent phosphorylation of STIL is required for centriole duplication. Biol Open. 2015 Feb 20;4(3):370-7. [CrossRef]
- Dzhindzhev NS, Tzolovsky G, Lipinszki Z, Schneider S, Lattao R, Fu J, Debski J, Dadlez M, Glover DM. Plk4 phosphorylates Ana2 to trigger Sas6 recruitment and procentriole formation. Curr Biol. 2014 Nov 3;24(21):2526-32. [CrossRef]
- Gopalakrishnan J, Guichard P, Smith AH, Schwarz H, Agard DA, Marco S, Avidor-Reiss T. Self-assembling SAS-6 multimer is a core centriole building block. J Biol Chem. 2010 Mar 19;285(12):8759-70. [CrossRef]
- van Breugel M, Hirono M, Andreeva A, Yanagisawa HA, Yamaguchi S, Nakazawa Y, Morgner N, Petrovich M, Ebong IO, Robinson CV, Johnson CM, Veprintsev D, Zuber B. Structures of SAS-6 suggest its organization in centrioles. Science. 2011 Mar 4;331(6021):1196-9. [CrossRef]
- Nakazawa Y, Hiraki M, Kamiya R, Hirono M. SAS-6 is a cartwheel protein that establishes the 9-fold symmetry of the centriole. Curr Biol. 2007 Dec 18;17(24):2169-74. [CrossRef]
- Gönczy P. Towards a molecular architecture of centriole assembly. Nat Rev Mol Cell Biol. 2012 Jun 13;13(7):425-35. [CrossRef]
- Hilbert M, Noga A, Frey D, Hamel V, Guichard P, Kraatz SH, Pfreundschuh M, Hosner S, Flückiger I, Jaussi R, Wieser MM, Thieltges KM, Deupi X, Müller DJ, Kammerer RA, Gönczy P, Hirono M, Steinmetz MO. SAS-6 engineering reveals interdependence between cartwheel and microtubules in determining centriole architecture. Nat Cell Biol. 2016 Apr;18(4):393-403. [CrossRef]
- Cottee MA, Raff JW, Lea SM, Roque H. SAS-6 oligomerization: the key to the centriole? Nat Chem Biol. 2011 Sep 19;7(10):650-3. [CrossRef]
- Hung LY, Tang CJ, Tang TK. Protein 4.1 R-135 interacts with a novel centrosomal protein (CPAP) which is associated with the gamma-tubulin complex. Mol Cell Biol. 2000 Oct;20(20):7813-25. [CrossRef]
- Garcez PP, Diaz-Alonso J, Crespo-Enriquez I, Castro D, Bell D, Guillemot F. Cenpj/CPAP regulates progenitor divisions and neuronal migration in the cerebral cortex downstream of Ascl1. Nat Commun. 2015 Mar 10;6:6474. [CrossRef]
- Zheng X, Ramani A, Soni K, Gottardo M, Zheng S, Ming Gooi L, Li W, Feng S, Mariappan A, Wason A, Widlund P, Pozniakovsky A, Poser I, Deng H, Ou G, Riparbelli M, Giuliano C, Hyman AA, Sattler M, Gopalakrishnan J, Li H. Molecular basis for CPAP-tubulin interaction in controlling centriolar and ciliary length. Nat Commun. 2016 Jun 16;7:11874. [CrossRef]
- Lin YC, Chang CW, Hsu WB, Tang CJ, Lin YN, Chou EJ, Wu CT, Tang TK. Human microcephaly protein CEP135 binds to hSAS-6 and CPAP, and is required for centriole assembly. EMBO J. 2013 Apr 17;32(8):1141-54. [CrossRef]
- Hussain MS, Baig SM, Neumann S, Nürnberg G, Farooq M, Ahmad I, Alef T, Hennies HC, Technau M, Altmüller J, Frommolt P, Thiele H, Noegel AA, Nürnberg P. A truncating mutation of CEP135 causes primary microcephaly and disturbed centrosomal function. Am J Hum Genet. 2012 May 4;90(5):871-8. [CrossRef]
- Bamborschke D, Daimagüler HS, Hahn A, Hussain MS, Nürnberg P, Cirak S. Mutation in CEP135 causing primary microcephaly and subcortical heterotopia. Am J Med Genet A. 2020 Oct;182(10):2450-2453. [CrossRef]
- Kim TS, Park JE, Shukla A, Choi S, Murugan RN, Lee JH, Ahn M, Rhee K, Bang JK, Kim BY, Loncarek J, Erikson RL, Lee KS. Hierarchical recruitment of Plk4 and regulation of centriole biogenesis by two centrosomal scaffolds, Cep192 and Cep152. Proc Natl Acad Sci U S A. 2013 Dec 10;110(50):E4849-57. [CrossRef]
- Cizmecioglu O, Arnold M, Bahtz R, Settele F, Ehret L, Haselmann-Weiss U, Antony C, Hoffmann I. Cep152 acts as a scaffold for recruitment of Plk4 and CPAP to the centrosome. J Cell Biol. 2010 Nov 15;191(4):731-9. [CrossRef]
- Chang CW, Hsu WB, Tsai JJ, Tang CJ, Tang TK. CEP295 interacts with microtubules and is required for centriole elongation. J Cell Sci. 2016 Jul 1;129(13):2501-13. [CrossRef]
- Löffler H, Fechter A, Matuszewska M, Saffrich R, Mistrik M, Marhold J, Hornung C, Westermann F, Bartek J, Krämer A. Cep63 recruits Cdk1 to the centrosome: implications for regulation of mitotic entry, centrosome amplification, and genome maintenance. Cancer Res. 2011 Mar 15;71(6):2129-39. [CrossRef]
- Fagundes R, Teixeira LK. Cyclin E/CDK2: DNA Replication, Replication Stress and Genomic Instability. Front Cell Dev Biol. 2021 Nov 24;9:774845. [CrossRef]
- Tarapore P, Okuda M, Fukasawa K. A mammalian in vitro centriole duplication system: evidence for involvement of CDK2/cyclin E and nucleophosmin/B23 in centrosome duplication. Cell Cycle. 2002 Jan;1(1):75-81.
- Zhao H, Chen X, Gurian-West M, Roberts JM. Loss of cyclin-dependent kinase 2 (CDK2) inhibitory phosphorylation in a CDK2AF knock-in mouse causes misregulation of DNA replication and centrosome duplication. Mol Cell Biol. 2012 Apr;32(8):1421-32. [CrossRef]
- Okuda M, Horn HF, Tarapore P, Tokuyama Y, Smulian AG, Chan PK, Knudsen ES, Hofmann IA, Snyder JD, Bove KE, Fukasawa K. Nucleophosmin/B23 is a target of CDK2/cyclin E in centrosome duplication. Cell. 2000 Sep 29;103(1):127-40. [CrossRef]
- Fisk HA, Winey M. The mouse Mps1p-like kinase regulates centrosome duplication. Cell. 2001 Jul 13;106(1):95-104. [CrossRef]
- Leidel S., Delattre M., Cerutti L., Baumer K., Gönczy P. SAS-6 defines a protein family required for centrosome duplication in C. elegans and in human cells. Nat. Cell Biol. 2005;7:115–125. [CrossRef]
- Peel N., Stevens N. R., Basto R., Raff J. W. Overexpressing centriole-replication proteins in vivo induces centriole overduplication and de novo formation. Curr. Biol. 2007;17:834–843. [CrossRef]
- Strnad P., Leidel S., Vinogradova T., Euteneuer U., Khodjakov A., Gönczy P. Regulated HsSAS-6 levels ensure formation of a single procentriole per centriole during the centrosome duplication cycle. Dev. Cell. 2007;13:203–213. [CrossRef]
- Lange BM. Integration of the centrosome in cell cycle control, stress response and signal transduction pathways. Curr Opin Cell Biol. 2002 Feb;14(1):35-43. [CrossRef]
- Reed SI. The role of p34 kinases in the G1 to S-phase transition. Annu Rev Cell Biol. 1992;8:529-61. [CrossRef]
- Hatakeyama M, Brill JA, Fink GR, Weinberg RA. Collaboration of G1 cyclins in the functional inactivation of the retinoblastoma protein. Genes Dev. 1994 Aug 1;8(15):1759-71. [CrossRef]
- Lentini L, Iovino F, Amato A, Di Leonardo A. Centrosome amplification induced by hydroxyurea leads to aneuploidy in pRB deficient human and mouse fibroblasts. Cancer Lett. 2006 Jul 8;238(1):153-60. [CrossRef]
- Chan CY, Yuen VW, Chiu DK, Goh CC, Thu KL, Cescon DW, Soria-Bretones I, Law CT, Cheu JW, Lee D, Tse AP, Tan KV, Zhang MS, Wong BP, Wong CM, Khong PL, Ng IO, Bray MR, Mak TW, Yau TC, Wong CC. Polo-like kinase 4 inhibitor CFI-400945 suppresses liver cancer through cell cycle perturbation and eliciting antitumor immunity. Hepatology. 2023 Mar 1;77(3):729-744. [CrossRef]
- Pellizzari S, Bhat V, Athwal H, Cescon DW, Allan AL, Parsyan A. PLK4 as a potential target to enhance radiosensitivity in triple-negative breast cancer. Radiat Oncol. 2024 Feb 16;19(1):24. [CrossRef]
- He G, Siddik ZH, Huang Z, Wang R, Koomen J, Kobayashi R, Khokhar AR, Kuang J. Induction of p21 by p53 following DNA damage inhibits both Cdk4 and Cdk2 activities. Oncogene. 2005 Apr 21;24(18):2929-43. [CrossRef]
- Nakamura T, Saito H, Takekawa M. SAPK pathways and p53 cooperatively regulate PLK4 activity and centrosome integrity under stress. Nat Commun. 2013;4:1775. [CrossRef]
- Tarapore P, Fukasawa K. Loss of p53 and centrosome hyperamplification. Oncogene. 2002 Sep 9;21(40):6234-40. [CrossRef]
- Lambrus BG, Uetake Y, Clutario KM, Daggubati V, Snyder M, Sluder G, Holland AJ. p53 protects against genome instability following centriole duplication failure. J Cell Biol. 2015 Jul 6;210(1):63-77. [CrossRef]
- Lipsick J. A History of Cancer Research: The P53 Pathway. Cold Spring Harb Perspect Med. 2025 Feb 3;15(2):a035931. [CrossRef]
- Sabat-Pośpiech D, Fabian-Kolpanowicz K, Prior IA, Coulson JM, Fielding AB. Targeting centrosome amplification, an Achilles’ heel of cancer. Biochem Soc Trans. 2019 Oct 31;47(5):1209-1222. [CrossRef]
- Delattre M, Gönczy P. The arithmetic of centrosome biogenesis. J Cell Sci. 2004 Apr 1;117(Pt 9):1619-30. [CrossRef]
- Remo A, Li X, Schiebel E, Pancione M. The Centrosome Linker and Its Role in Cancer and Genetic Disorders. Trends Mol Med. 2020 Apr;26(4):380-393. [CrossRef]
- Shukla A, Kong D, Sharma M, Magidson V, Loncarek J. Plk1 relieves centriole block to reduplication by promoting daughter centriole maturation. Nat Commun. 2015 Aug 21;6:8077. [CrossRef]
- Holland AJ, Lan W, Niessen S, Hoover H, Cleveland DW. Polo-like kinase 4 kinase activity limits centrosome overduplication by autoregulating its own stability. J Cell Biol. 2010 Jan 25;188(2):191-8. [CrossRef]
- Kleylein-Sohn J, Westendorf J, Le Clech M, Habedanck R, Stierhof YD, Nigg EA. Plk4-induced centriole biogenesis in human cells. Dev Cell. 2007 Aug;13(2):190-202. [CrossRef]
- Meraldi P, Honda R, Nigg EA. Aurora-A overexpression reveals tetraploidization as a major route to centrosome amplification in p53-/- cells. EMBO J. 2002 Feb 15;21(4):483-92. [CrossRef]
- Inanç B, Dodson H, Morrison CG. A centrosome-autonomous signal that involves centriole disengagement permits centrosome duplication in G2 phase after DNA damage. Mol Biol Cell. 2010 Nov 15;21(22):3866-77. [CrossRef]
- Denu RA, Shabbir M, Nihal M, Singh CK, Longley BJ, Burkard ME, Ahmad N. Centriole Overduplication is the Predominant Mechanism Leading to Centrosome Amplification in Melanoma. Mol Cancer Res. 2018 Mar;16(3):517-527. [CrossRef]
- Denu RA, Burkard ME. Analysis of the “centrosome-ome” identifies MCPH1 deletion as a cause of centrosome amplification in human cancer. Sci Rep. 2020 Jul 17;10(1):11921. [CrossRef]
- Cui FM, Sun XJ, Huang CC, Chen Q, He YM, Zhang SM, Guan H, Song M, Zhou PK, Hou J. Inhibition of c-Myc expression accounts for an increase in the number of multinucleated cells in human cervical epithelial cells. Oncol Lett. 2017 Sep;14(3):2878-2886. [CrossRef]
- Courapied S, Cherier J, Vigneron A, Troadec MB, Giraud S, Valo I, Prigent C, Gamelin E, Coqueret O, Barré B. Regulation of the Aurora-A gene following topoisomerase I inhibition: implication of the Myc transcription factor. Mol Cancer. 2010 Aug 3;9:205. [CrossRef]
- Khodjakov A, Rieder CL. Mitosis: too much of a good thing (can be bad). Curr Biol. 2009 Dec 1;19(22):R1032-4. [CrossRef]
- Zheng S, Guerrero-Haughton E, Foijer F. Chromosomal Instability-Driven Cancer Progression: Interplay with the Tumour Microenvironment and Therapeutic Strategies. Cells. 2023 Nov 26;12(23):2712. [CrossRef]
- Godinho SA, Picone R, Burute M, Dagher R, Su Y, Leung CT, Polyak K, Brugge JS, Théry M, Pellman D. Oncogene-like induction of cellular invasion from centrosome amplification. Nature. 2014 Jun 5;510(7503):167-71. [CrossRef]
- Kulukian A, Holland AJ, Vitre B, Naik S, Cleveland DW, Fuchs E. Epidermal development, growth control, and homeostasis in the face of centrosome amplification. Proc Natl Acad Sci U S A. 2015 Nov 17;112(46):E6311-20. [CrossRef]
- Marthiens V, Rujano MA, Pennetier C, Tessier S, Paul-Gilloteaux P, Basto R. Centrosome amplification causes microcephaly. Nat Cell Biol. 2013 Jul;15(7):731-40. [CrossRef]
- Thornton GK, Woods CG. Primary microcephaly: do all roads lead to Rome? Trends Genet. 2009 Nov;25(11):501-10. [CrossRef]
- Chavali PL, Pütz M, Gergely F. Small organelle, big responsibility: the role of centrosomes in development and disease. Philos Trans R Soc Lond B Biol Sci. 2014 Sep 5;369(1650):20130468. [CrossRef]
- Goundiam O, Basto R. Centrosomes in disease: how the same music can sound so different? Curr Opin Struct Biol. 2021 Feb;66:74-82. [CrossRef]
- Chan JY. A clinical overview of centrosome amplification in human cancers. Int J Biol Sci. 2011;7(8):1122-44. [CrossRef]
- Marteil G, Guerrero A, Vieira AF, de Almeida BP, Machado P, Mendonça S, Mesquita M, Villarreal B, Fonseca I, Francia ME, Dores K, Martins NP, Jana SC, Tranfield EM, Barbosa-Morais NL, Paredes J, Pellman D, Godinho SA, Bettencourt-Dias M. Over-elongation of centrioles in cancer promotes centriole amplification and chromosome missegregation. Nat Commun. 2018 Mar 28;9(1):1258. [CrossRef]
- Sugihara E, Kanai M, Matsui A, Onodera M, Schwab M, Miwa M. Enhanced expression of MYCN leads to centrosome hyperamplification after DNA damage in neuroblastoma cells. Oncogene. 2004 Jan 29;23(4):1005-9. [CrossRef]
- Kelleher FC, Kroes J, Lewin J. Targeting the centrosome and polo-like kinase 4 in osteosarcoma. Carcinogenesis. 2019 Jun 10;40(4):493-499. [CrossRef]
- Méniel V, Megges M, Young MA, Cole A, Sansom OJ, Clarke AR. Apc and p53 interaction in DNA damage and genomic instability in hepatocytes. Oncogene. 2015 Jul 30;34(31):4118-29. [CrossRef]
- Caulin C, Nguyen T, Lang GA, Goepfert TM, Brinkley BR, Cai WW, Lozano G, Roop DR. An inducible mouse model for skin cancer reveals distinct roles for gain- and loss-of-function p53 mutations. J Clin Invest. 2007 Jul;117(7):1893-901. [CrossRef]
- Zhao Y, Wang X. PLK4: a promising target for cancer therapy. J Cancer Res Clin Oncol. 2019 Oct;145(10):2413-2422. [CrossRef]
- Li J, Tan M, Li L, Pamarthy D, Lawrence TS, Sun Y. SAK, a new polo-like kinase, is transcriptionally repressed by p53 and induces apoptosis upon RNAi silencing. Neoplasia. 2005 Apr;7(4):312-23. [CrossRef]
- Sun Y, Xue Y, Liu H, Mu S, Sun P, Sun Y, Wang L, Wang H, Wang J, Wu T, Yin W, Qin Q, Sun Y, Yang H, Zhao D, Cheng M. Discovery of CZS-241: A Potent, Selective, and Orally Available Polo-Like Kinase 4 Inhibitor for the Treatment of Chronic Myeloid Leukemia. J Med Chem. 2023 Feb 23;66(4):2396-2421. [CrossRef]
- Murphy T, Mason JM, Leber B, Bray MR, Chan SM, Gupta V, Khalaf D, Maze D, McNamara CJ, Schimmer AD, Schuh AC, Sibai H, Trus M, Valiquette D, Martin K, Nguyen L, Li X, Mak TW, Minden MD, Yee KWL. Preclinical characterization and clinical trial of CFI-400945, a polo-like kinase 4 inhibitor, in patients with relapsed/refractory acute myeloid leukemia and higher-risk myelodysplastic neoplasms. Leukemia. 2024 Mar;38(3):502-512. [CrossRef]
- Zhu W, Xie B. PLK4 inhibitor exhibits antitumor effect and synergizes sorafenib via arresting cell cycle and inactivating Wnt/β-catenin pathway in anaplastic thyroid cancer. Cancer Biol Ther. 2023 Dec 31;24(1):2223383. [CrossRef]
- Farcy S, Hachour H, Bahi-Buisson N, Passemard S. Genetic Primary Microcephalies: When Centrosome Dysfunction Dictates Brain and Body Size. Cells. 2023 Jul 7;12(13):1807. [CrossRef]
- Kumar A, Girimaji SC, Duvvari MR, Blanton SH. Mutations in STIL, encoding a pericentriolar and centrosomal protein, cause primary microcephaly. Am J Hum Genet. 2009 Feb;84(2):286-90. [CrossRef]
- Lin YN, Lee YS, Li SK, Tang TK. Loss of CPAP in developing mouse brain and its functional implication for human primary microcephaly. J Cell Sci. 2020 Jun 24;133(12):jcs243592. [CrossRef]
- Phan TP, Holland AJ. Time is of the essence: the molecular mechanisms of primary microcephaly. Genes Dev. 2021 Dec 1;35(23-24):1551-1578. [CrossRef]
- Yang J, Hu X, Ma J, Shi SH. Centrosome regulation and function in mammalian cortical neurogenesis. Curr Opin Neurobiol. 2021 Aug;69:256-266. [CrossRef]
- Papuc SM, Erbescu A, Glangher A, Streata I, Riza AL, Budisteanu M, Arghir A. Autistic Behavior as Novel Clinical Finding in OFD1 Syndrome. Genes (Basel). 2023 Jan 27;14(2):327. [CrossRef]
- Mendes M, Chen DZ, Engchuan W, Leal TP, Thiruvahindrapuram B, Trost B, Howe JL, Pellecchia G, Nalpathamkalam T, Alexandrova R, Salazar NB, McKee EA, Rivera-Alfaro N, Lai MC, Bandres-Ciga S, Roshandel D, Bradley CA, Anagnostou E, Sun L, Scherer SW. Am J Hum Genet. 2025 Jan 2;112(1):135-153. [CrossRef]
- Mahjoub MR, Stearns T. Supernumerary centrosomes nucleate extra cilia and compromise primary cilium signaling. Curr Biol. 2012 Sep 11;22(17):1628-34. [CrossRef]
- Karalis V, Donovan KE, Sahin M. Primary Cilia Dysfunction in Neurodevelopmental Disorders beyond Ciliopathies. J Dev Biol. 2022 Dec 13;10(4):54. [CrossRef]
- Schneider P, Fandrey J, Leu T. Primary cilia as antennas for oxygen. Am J Physiol Cell Physiol. 2025 Feb 1;328(2):C381-C386. [CrossRef]
- Luo L, Roy S, Li L, Ma M. Polycystic kidney disease: novel insights into polycystin function. Trends Mol Med. 2023 Apr;29(4):268-281. [CrossRef]
- Chen X, Shi Z, Yang F, Zhou T, Xie S. Deciphering cilia and ciliopathies using proteomic approaches. FEBS J. 2023 May;290(10):2590-2603. [CrossRef]
- Kantsadi AL, Hatzopoulos GN, Gönczy P, Vakonakis I. Structures of SAS-6 coiled coil hold implications for the polarity of the centriolar cartwheel. Structure. 2022 May 5;30(5):671-684.e5. [CrossRef]
- Li S, Fernandez JJ, Marshall WF, Agard DA. Electron cryo-tomography provides insight into procentriole architecture and assembly mechanism. Elife. 2019 Feb 11;8:e43434. [CrossRef]
- Agostini L, Pfister J, Basnet N, Ding J, Zhang R, Biertümpfel C, O’Connell KF, Mizuno N. Structural insights into SSNA1 self-assembly and its microtubule binding for centriole maintenance. bioRxiv [Preprint]. 2024 Nov 15:2024.11.13.623454. [CrossRef]
- Lau L, Lee YL, Sahl SJ, Stearns T, Moerner WE. STED microscopy with optimized labeling density reveals 9-fold arrangement of a centriole protein. Biophys J. 2012 Jun 20;102(12):2926-35. [CrossRef]
- Sonnen KF, Schermelleh L, Leonhardt H, Nigg EA. 3D-structured illumination microscopy provides novel insight into architecture of human centrosomes. Biol Open. 2012 Oct 15;1(10):965-76. [CrossRef]
- Laporte MH, Gambarotto D, Bertiaux É, Bournonville L, Louvel V, Nunes JM, Borgers S, Hamel V, Guichard P. Time-series reconstruction of the molecular architecture of human centriole assembly. Cell. 2024 Apr 25;187(9):2158-2174.e19. [CrossRef]
- Danielsson F, Mahdessian D, Axelsson U, Sullivan D, Uhlén M, Andersen JS, Thul PJ, Lundberg E. Spatial Characterization of the Human Centrosome Proteome Opens Up New Horizons for a Small but Versatile Organelle. Proteomics. 2020 Dec;20(23):e1900361. [CrossRef]
- Cerulo L, Pezzella N, Caruso FP, Parente P, Remo A, Giordano G, Forte N, Busselez J, Boschi F, Galiè M, Franco B, Pancione M. Single-cell proteo-genomic reveals a comprehensive map of centrosome-associated spliceosome components. iScience. 2023 Apr 10;26(5):106602. [CrossRef]
- Damodaran AP, Gavard O, Gagné JP, Rogalska ME, Behera AK, Mancini E, Bertolin G, Courtheoux T, Kumari B, Cailloce J, Mereau A, Poirier GG, Valcárcel J, Gonatopoulos-Pournatzis T, Watrin E, Prigent C. Proteomic study identifies Aurora-A-mediated regulation of alternative splicing through multiple splicing factors. J Biol Chem. 2025 Jan;301(1):108000. [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



