Submitted:
17 April 2025
Posted:
17 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Experimental Materials
2.2. Experimental Methods
2.2.1. Adsorption Experiment
2.2.2. Isothermal Adsorption Model
3. Results
3.1. Clay Mineral Composition and Content
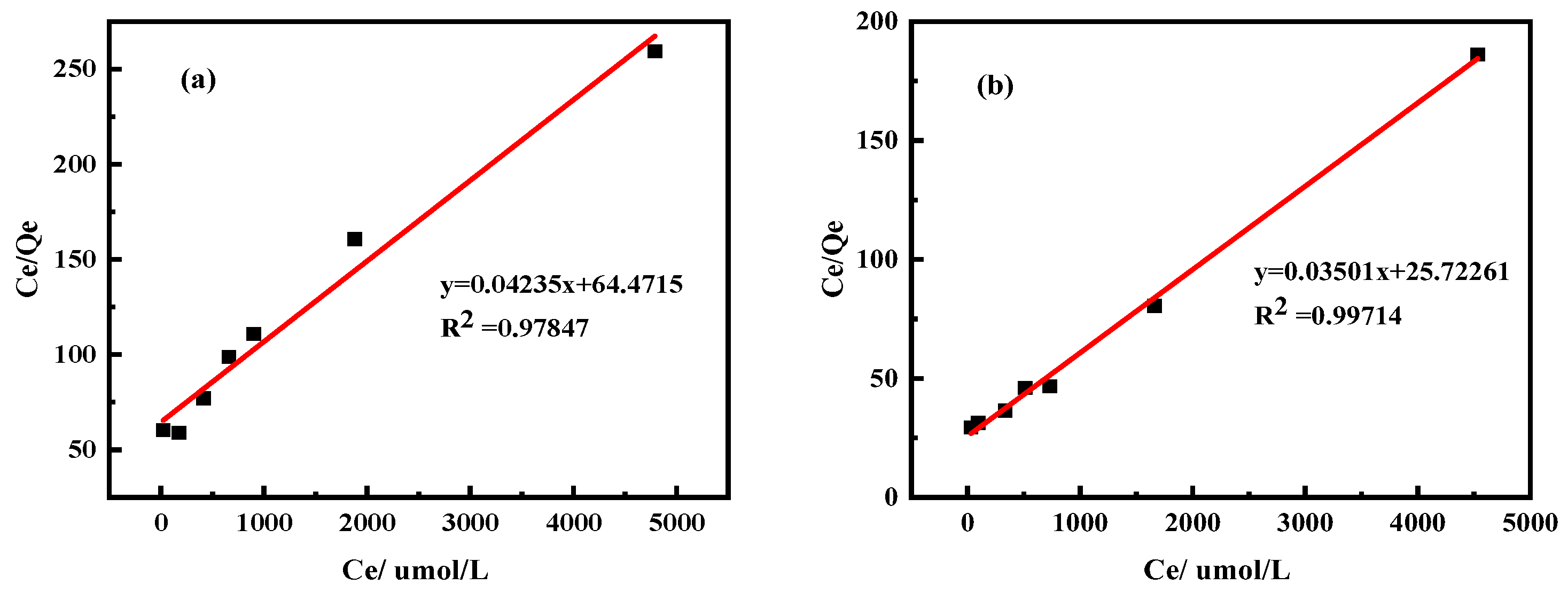
3.2. Effects of Initial Solution Concentration on Adsorption Process
3.2.1. Effects of Kaolinite and Halloysite on Pr Adsorption Properties
3.2.2. Effects of Kaolinite and Halloysite on Nd Adsorption Properties
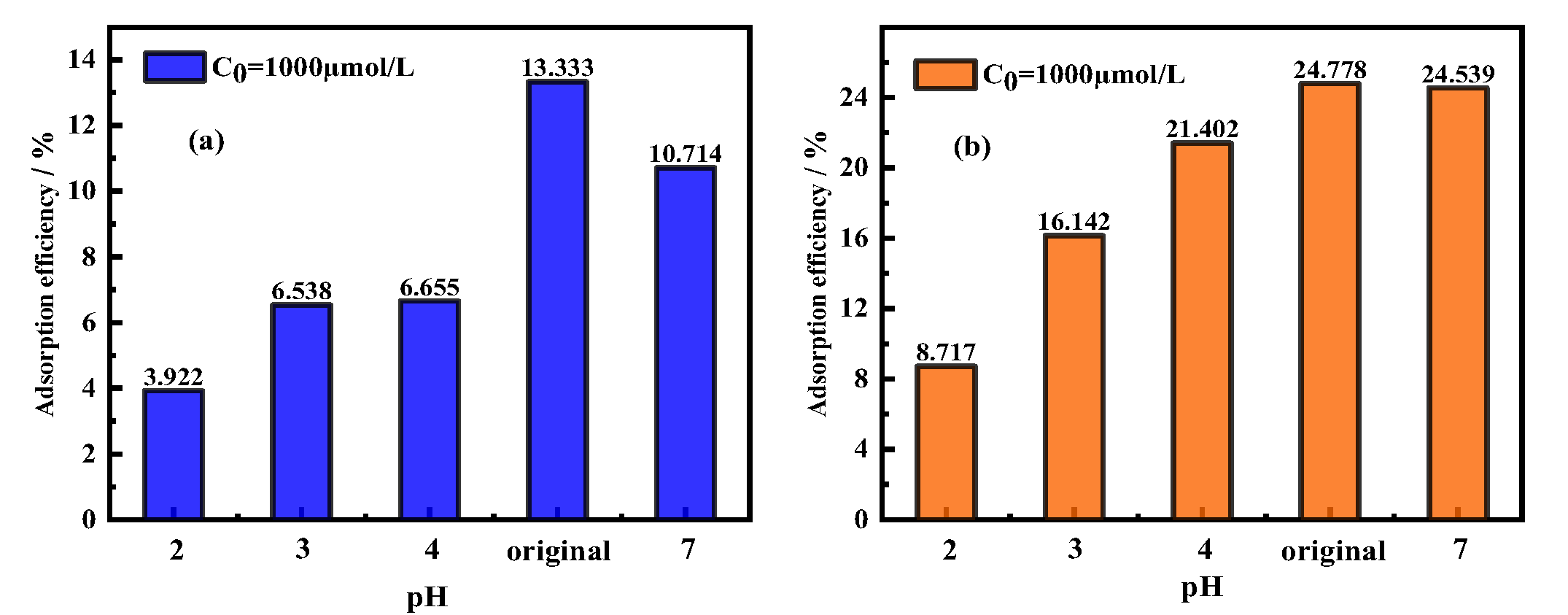
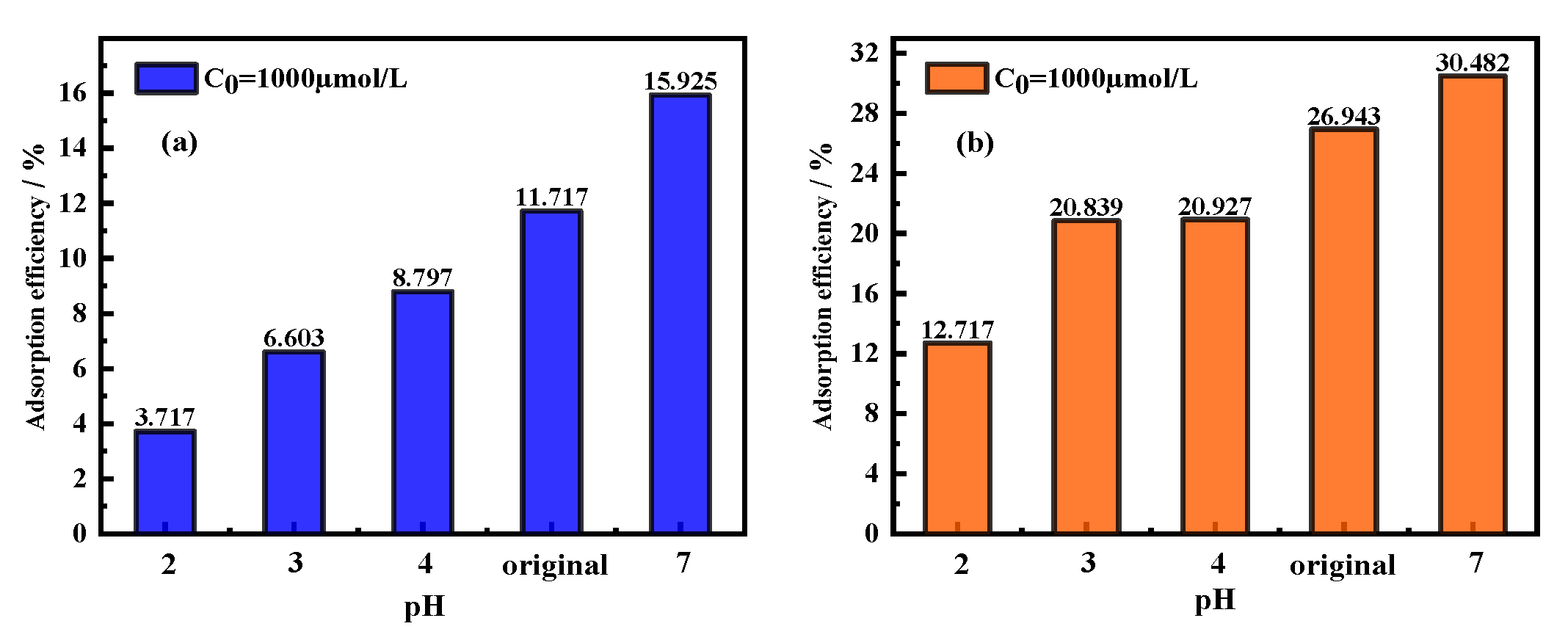
3.3. Effects of pH on Adsorption Efficiency/Adsorption Capacity
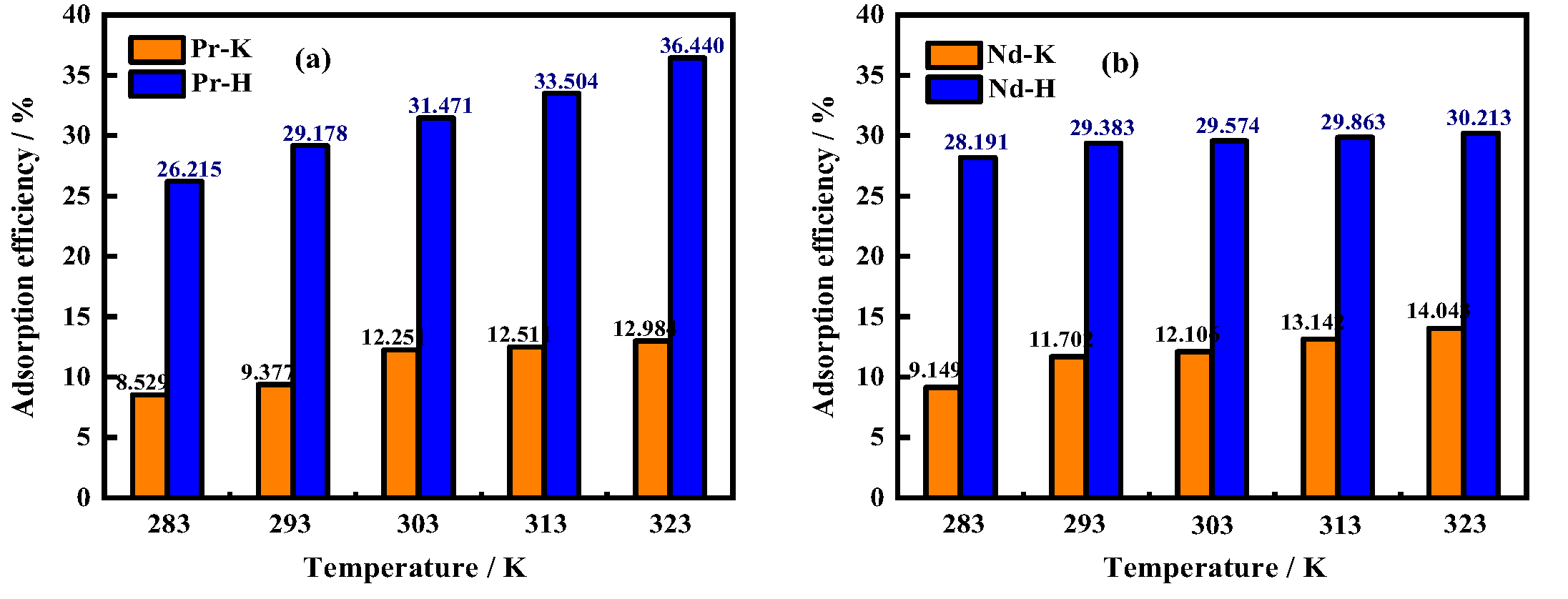
3.4. Effects of Temperature on Adsorption Efficiency/Adsorption Capacity
3.5. Adsorption Mechanism of Rare Earth Elements with Clay Minerals
3.5.1. SEM Analysis Results of Adsorption Process
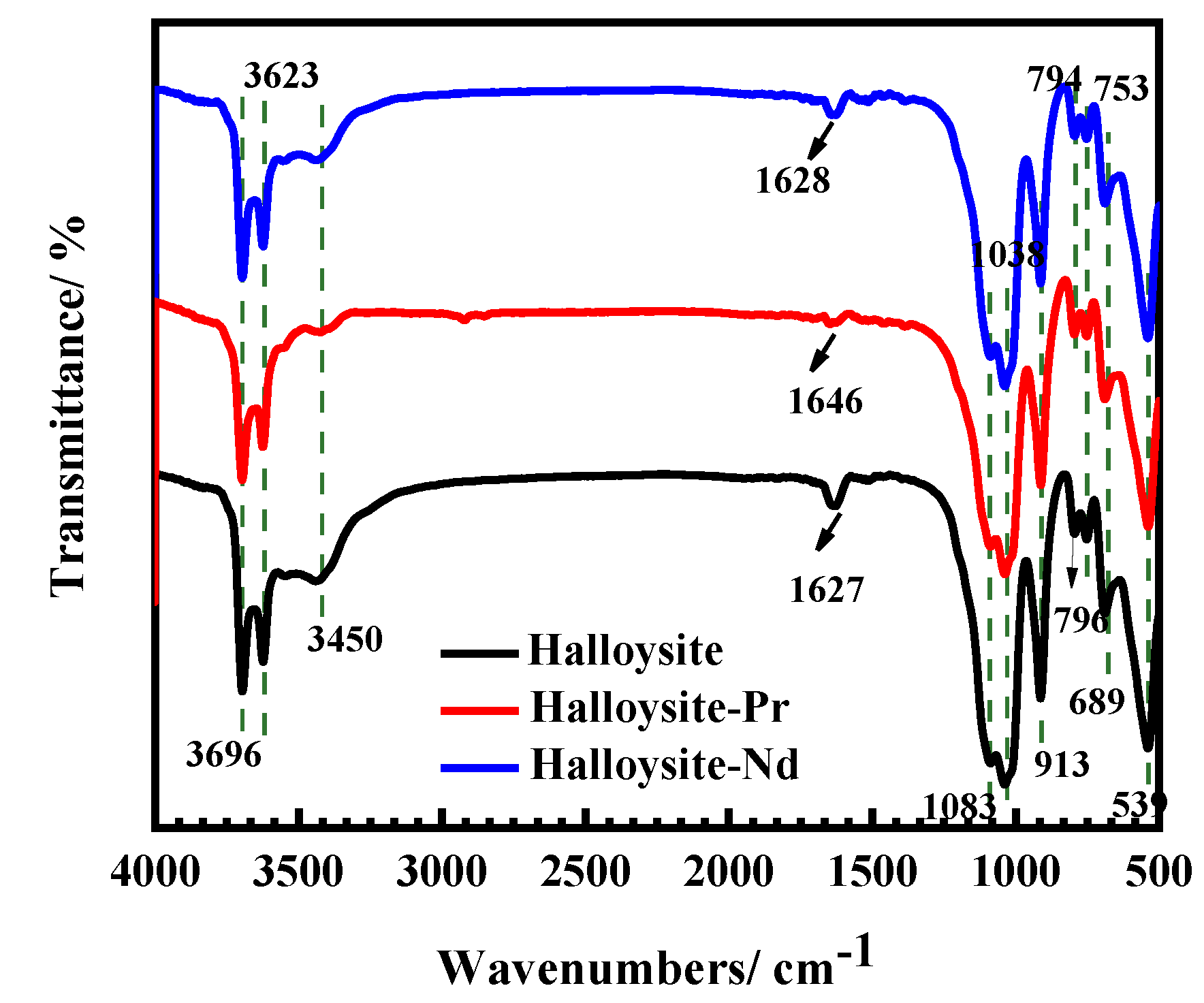
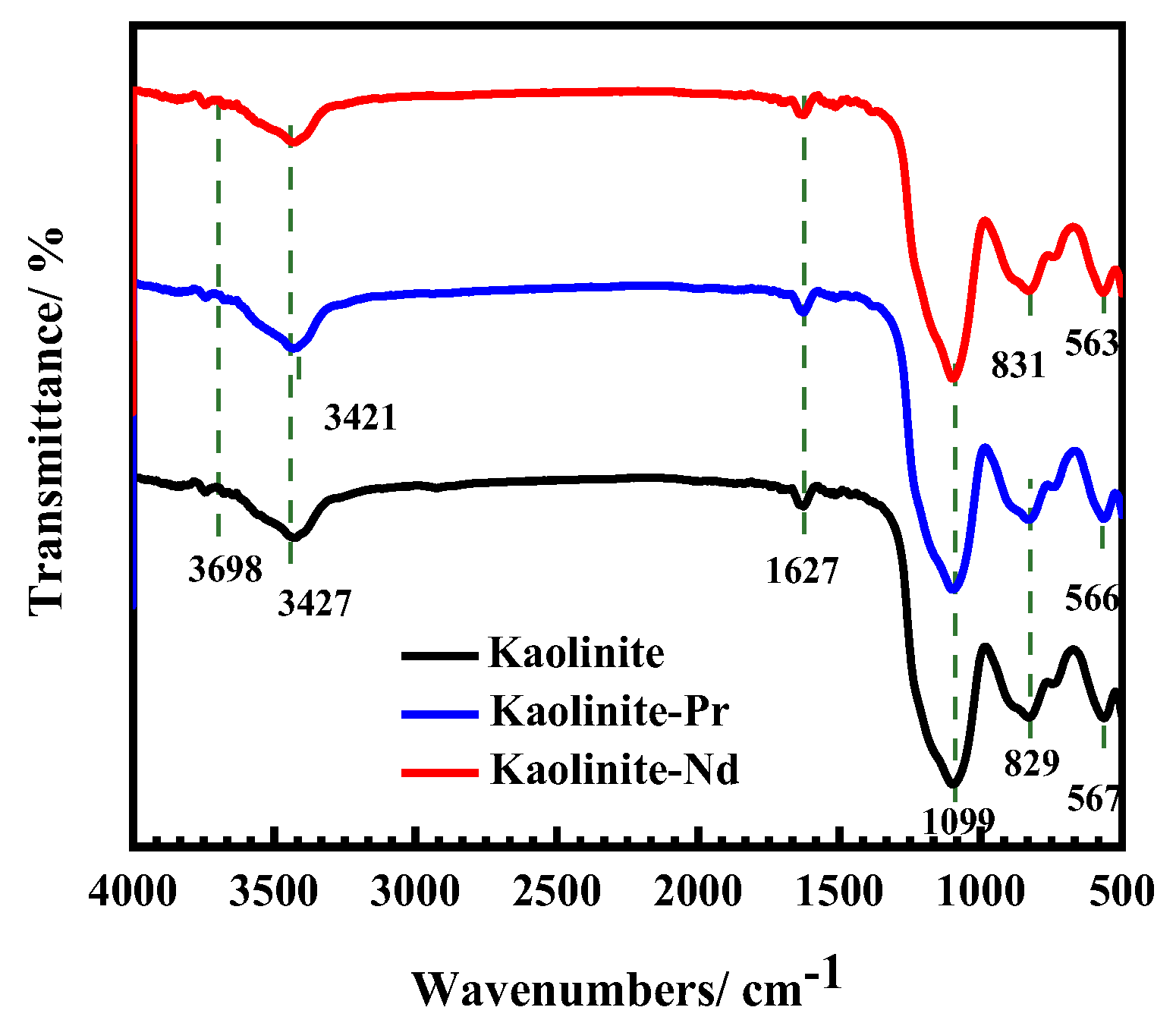
3.5.2. Infrared Spectroscopy Analysis Results of Adsorption Process
3.5.3. Quantum Chemical Analysis Results of Adsorption Process
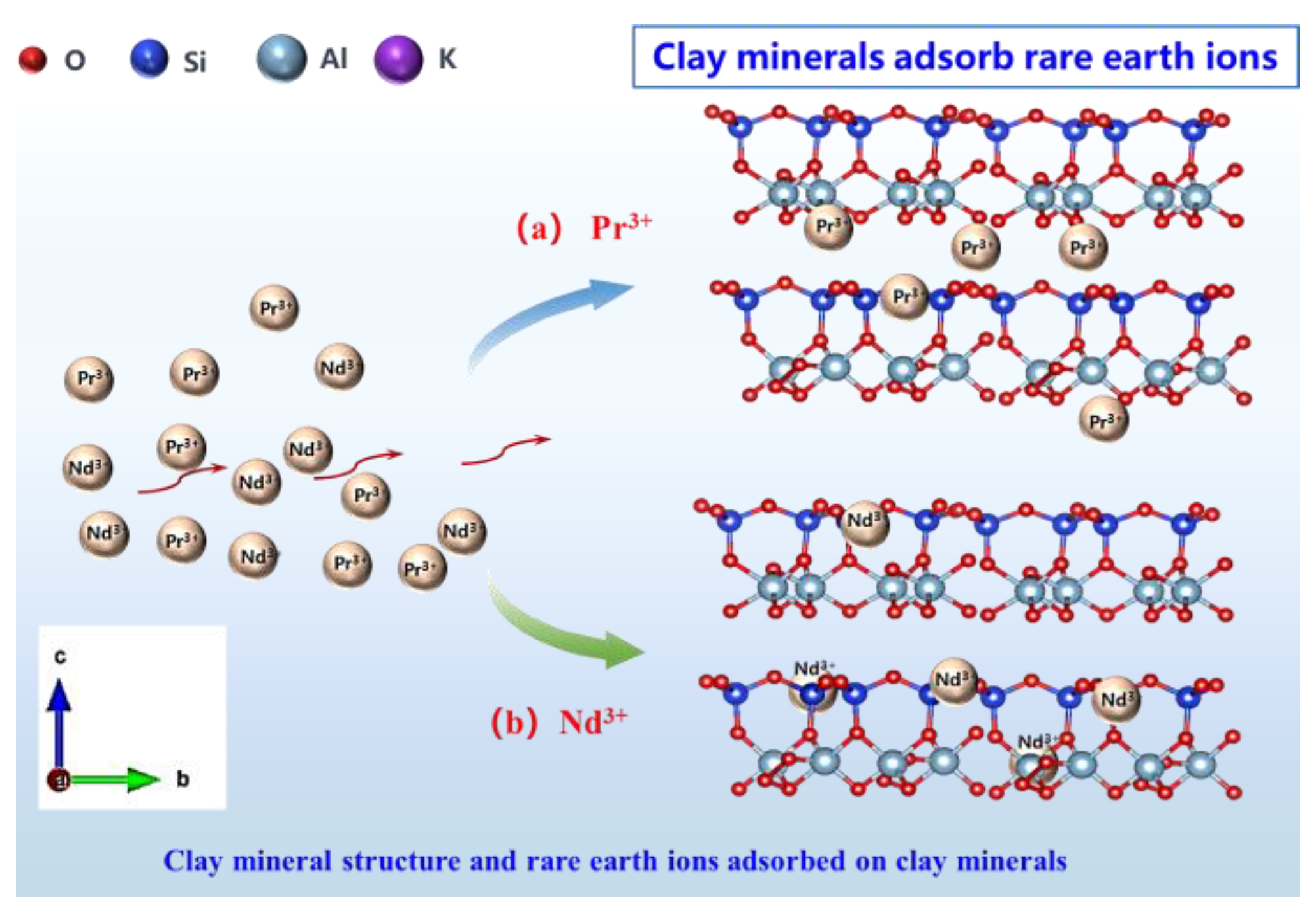
3.5.4. Model of the Microscopic Adsorption Structure of Pr and Nd by Kaolinite and Halloysite
3.6. The Relation Between the Adsorption Recovery and Rare Earths Resources Perspective
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yang XJ, Lin A, Li XL, Wu YD, Zhou WB, Chen ZH. China's ion-adsorption rare earth resources, mining consequences and preservation. Environmental Development. 2013, 8, 131–136. [Google Scholar] [CrossRef]
- Sun ZS, Zhao YM, Yan GH, Yuan HT, Zhang MR, Zhang B. A novel method for low-rank coal drying using steam transient flash evaporation. Fuel. 2023, 354: 129238.
- Balaram, V. Rare earth elements: A review of applications, occurrence, exploration, analysis, recycling, and environmental impact. Geosci. Frontiers. 2019, 10, 1285–1303. [Google Scholar] [CrossRef]
- Gao YQ, Zhang SM, Zhao KY, Wang ZW, Xu SX, Liang ZP, Wu K. Adsorption of La3+ and Ce3+ by poly-γ-glutamic acid crosslinked with polyvinyl alcohol. J. Rare Earths. 2015, 33, 884–891. [Google Scholar] [CrossRef]
- Goodenough KM, Wall F, Merriman D. The rare earth elements: demand, global resources, and challenges for resourcing future generations. Natural Resources Research. 2018, 27, 201–216. [Google Scholar] [CrossRef]
- Izatt RM, Izatt SR, Bruening RL, Izatt NE, Moyer BA. Challenges to achievement of metal sustainability in our high-tech society. Chem. Soc. Reviews. 2014, 43, 2451–2475. [Google Scholar] [CrossRef]
- Lewis LH, Jiménez-Villacorta F. Perspectives on permanent magnetic materials for energy conversion and power generation. Metallurgical and Materials Transactions A. 2013, 44, 2–20. [Google Scholar] [CrossRef]
- Wilburn, DR. Wind energy in the United States and materials required for the land-based wind turbine industry from 2010 through 2030. Washington, DC: US Department of the Interior, US Geological Survey. 2011.
- Xiao YF, Feng ZY, Hu GH, Huang L, Huang XW, Chen YY, Li ML. Leaching and mass transfer characteristics of elements from ion-adsorption type rare earth ore. Rare Metals. 2015, 34, 357–365. [Google Scholar] [CrossRef]
- Huang SX, Feng J, Liu JQ, Yu JX, Chi RA. Adsorption and desorption characteristics of clay minerals on NH4+ in the leaching agent of weathered crust elution-deposited rare earth ore. China Min. Magazine. 2021, 30, 152–158. [Google Scholar]
- Deng ZX, Qin L, Wang GS, Luo SH, Peng CL, Li Q. Metallogenic process of ion adsorption REE ore based on the occurrence regularity of La in kaolin. Ore Geology Reviews. 2019, 112, 103022. [Google Scholar] [CrossRef]
- Jia L, Zhang ZL. Adsorption of La3+, Nd3+ and some heavy metal ions by humic acid in amulti-metal-ion system. J. Chin. Soc. Rare Earths. 2009, 27, 816–821. [Google Scholar]
- Xiao, YF. Study on the green and efficient leaching technology for ion-adsorption type rare earths ore with magnesium salt system. Northeastern University. 2015.
- Jiao YF, He XL, Liao CF, Jiang PG. Studies on the sorption of amino methylene phosphonic acid resin for heavy rare earth metals. Ion Exch. Adsorption. 2009, 25, 306–312. [Google Scholar]
- Yang MJ, Liang XL, Ma LY, Huang J, He HP, Zhu JX. Adsorption of REEs on kaolinite and halloysite: A link to the REE distribution on clays in the weathering crust of granite. Chemical Geology. 2019, 525: 210–217.
- Xue WL, Li FH, Liu Y, Li BY, Huang BB. Mixing soil with bentonite to amend its microstructure and permeability. J. Irrig. Drainage. 2022, 41, 85–92. [Google Scholar]
- Shen W, Wang Z. Research evolvement of treatment technology of Pb2+ in water with clay mineral. Guangzhou Chem. Industry. 2009, 37, 60–62. [Google Scholar]
- Xie, JJ. Structure and physical-chemistry property evolution of heat-treatment palygorskite as well as adsorption for phosphorus. Hefei University of Technology. 2014.
- Tang YN, Cai YT, Yang PJ, Chang KK. Ferric oxide on U(VI) adsorption. Biol. Chem. Engineering. 2016, 2, 23–24. [Google Scholar]
- Coppin F, Berger G, Bauer A, Castet S, Loubet M. Sorption of lanthanides on smectite and kaolinite. Chem. Geology. 2002, 182, 57–68. [Google Scholar] [CrossRef]
- Peixoto AF, Fernandes AC, Pereira C, Pires J, Freire C. Physicochemical characterization of organosilylated halloysite clay nanotubes. Microporous and Mesoporous Materials. 2016, 219, 145–154. [Google Scholar] [CrossRef]
- Li WY, Liu J, Chen H, Deng Y, Zhang B, Wang Z, Zhang X, Hong S. Application of oxalic acid cross-linking activated alumina/chitosan bio composites in defluorination from aqueous solution. Investigation of adsorption mechanism. Chemical engineering journal. 2013, 225, 865–872. [Google Scholar]
- Xu JC, Wu HD, Zhou ZH, Yao PK, Zhang QP. Intercalation-hydrothermal preparation of submicron 13X zeolite with coal-measure kaolin. Bull. Chin. Ceram. Society. 2018, 37, 1188–1194. [Google Scholar]
- Ma WS, Shi JJ, Wang W, Ning P. Surface modification of long-chain alkyl silane on hnts. Silicone Material. 2011, 25, 248–252. [Google Scholar]
- Zhang A, Kang LL, Zhang YM, Ding DQ, Zhang YF. Effect of Kaolinite Particle Size on lts Crystal Structure and Thermal Evolution Behaviors. Bull. Chin. Ceram. Society. 2019, 38, 3964–3971. [Google Scholar]
- Clark SJ, Segall MD, Pickard CJ, Hasnip PJ, Probert MIJ, Refson K, Payne MC. First principles methods using CASTEP. Zeitschrift für kristallographie-crystalline materials. 2005, 220 567–570.
- Li, HT. First-principles study on microstructures and elastic properties of clay mineral. Taiyuan University of Technology. 2016.
- Wang GS, Lai YM, Peng CL. Adsorption of rare earth yttrium and ammonium ions on kaolinite surfaces: a DFT study. Theor. Chem. Accounts. 2018, 137, 1–11. [Google Scholar]
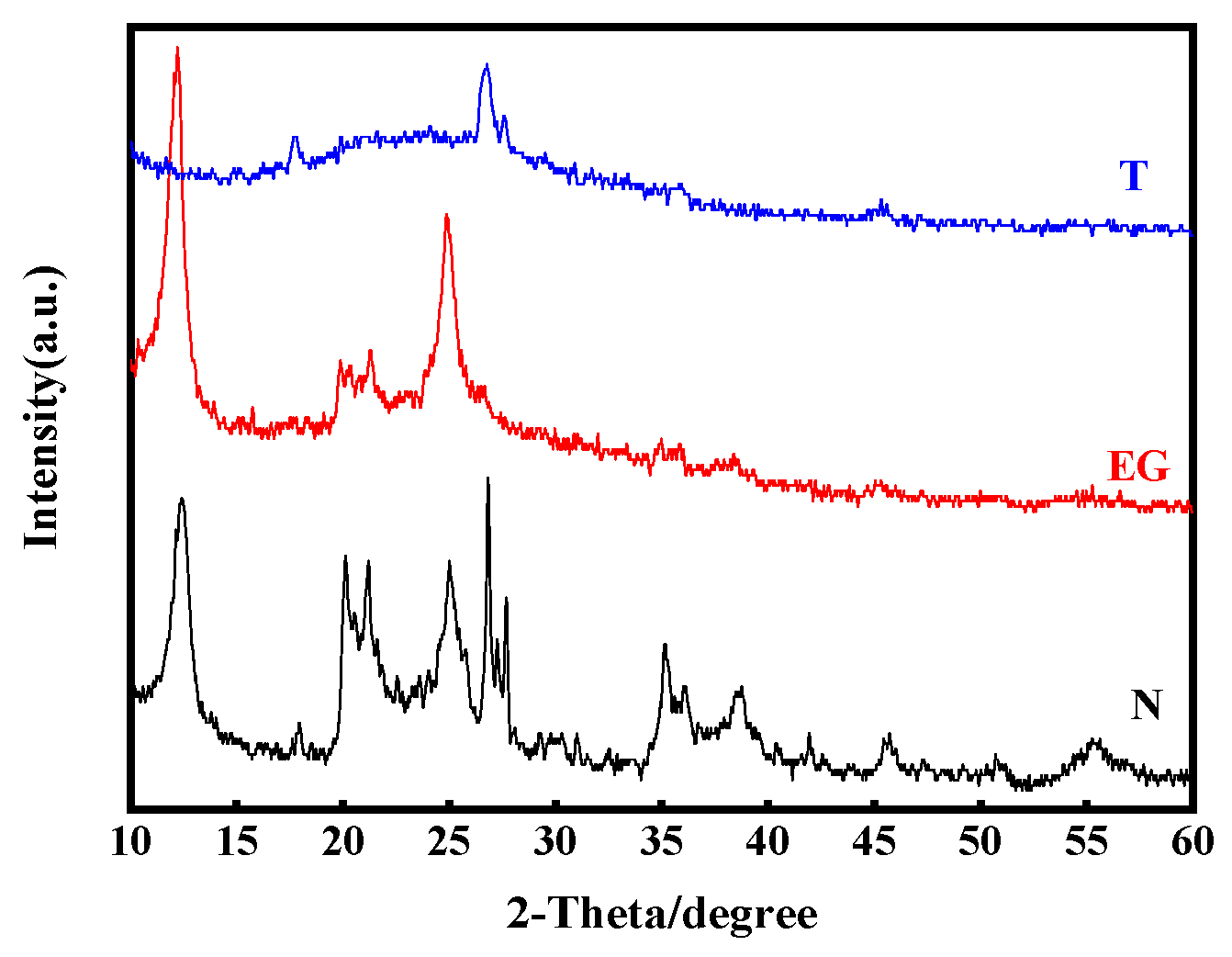
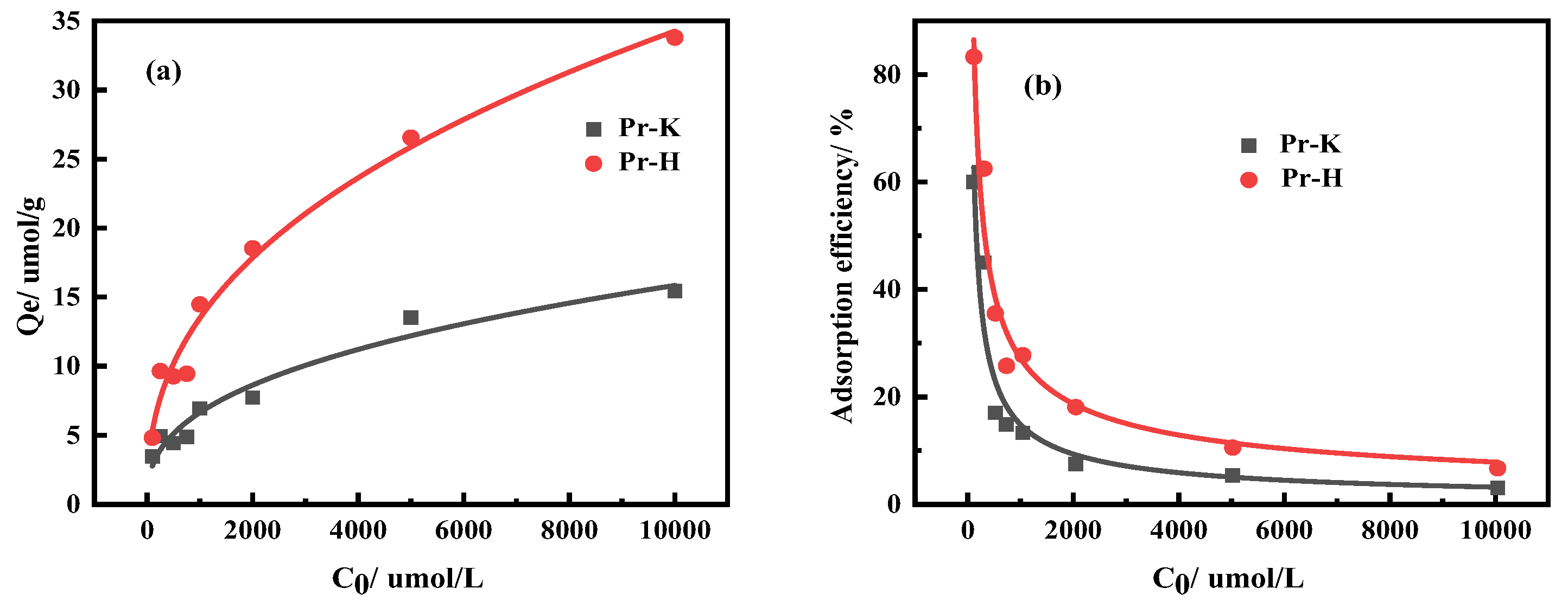
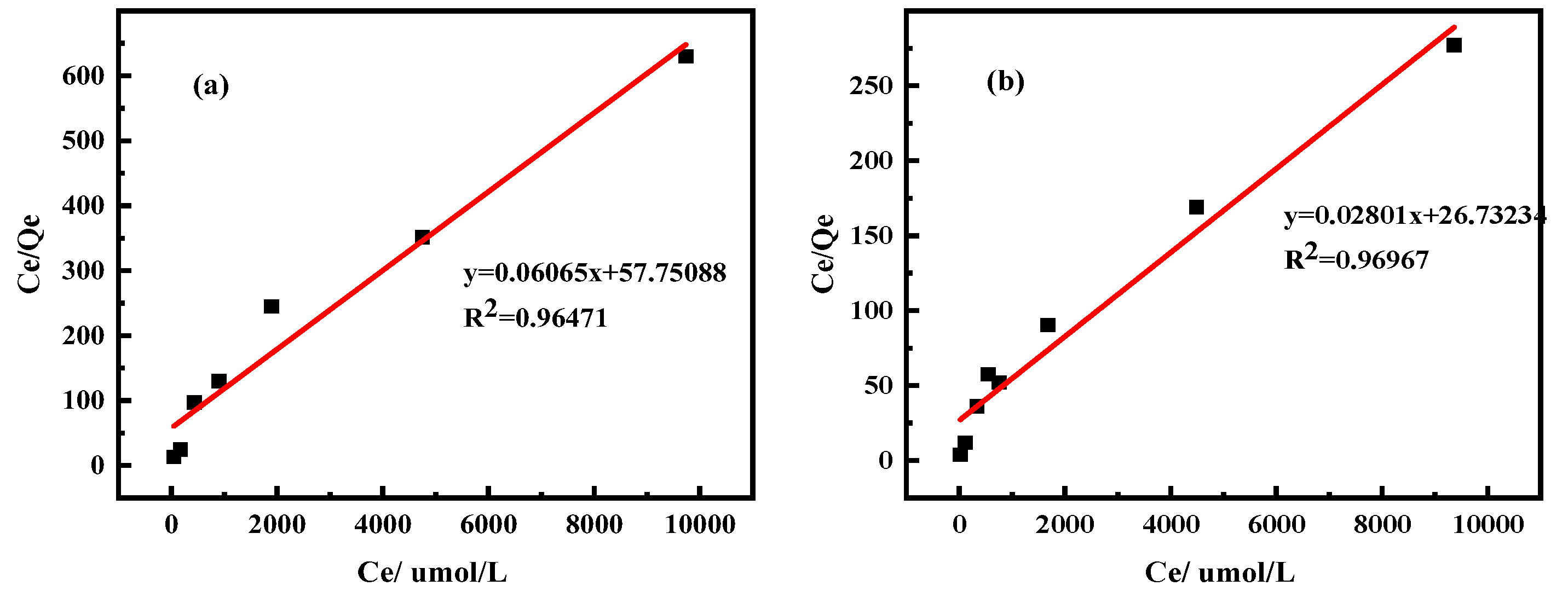
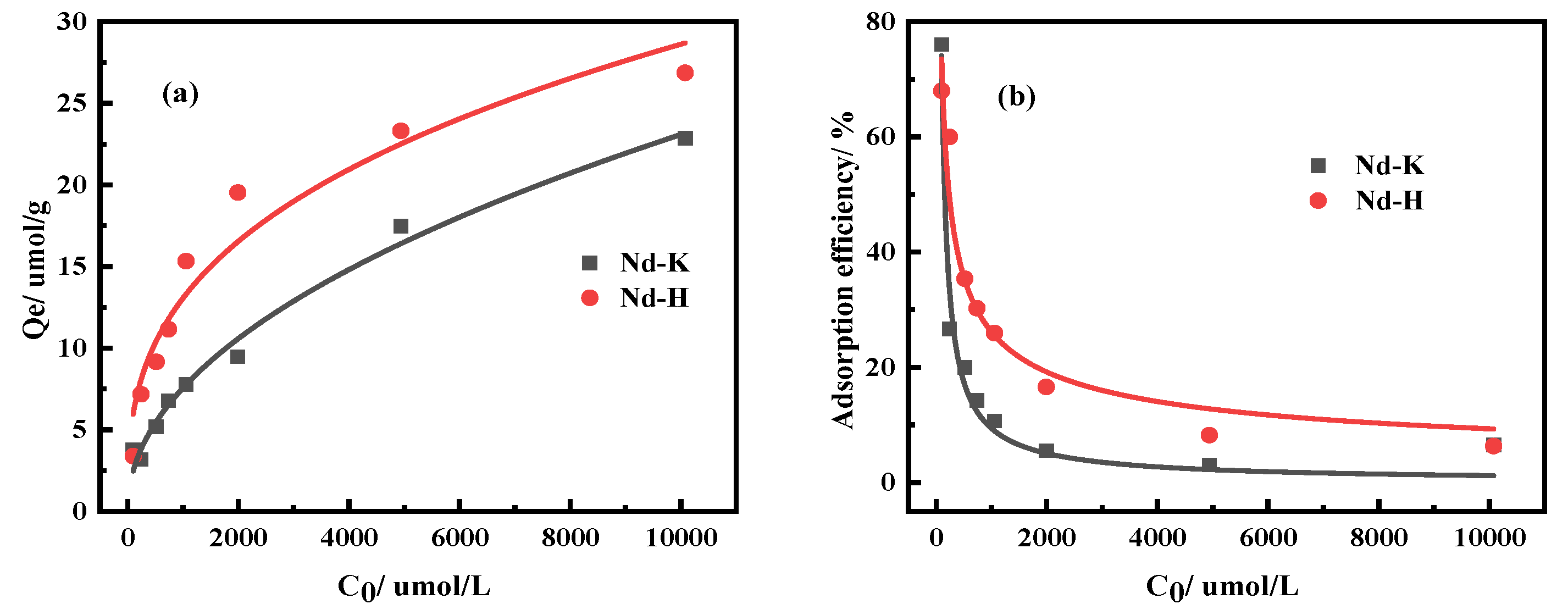


| Ingredient | REO | SiO2 | Al2O3 | MnO | K2O | Na2O |
| Content | 0.12 | 66.23 | 18.14 | 0.13 | 4.21 | 0.57 |
| Ingredient | CaO | TiO2 | P2O5 | Fe2O3 | FeO | Loss |
| Content | 0.04 | 0.05 | 0.09 | 1.60 | 0.6 | 7.1 |
| Specimen | Kaolinite | Halloysite | Illite | Montmorillonite |
|---|---|---|---|---|
| Ledge | 25.06 | 65.78 | 8.13 | 1.03 |
| Equations | Freundlich equation | Langmuir equation | |||||
|---|---|---|---|---|---|---|---|
| In KF | 1/n | R2 | 1/(QmaxKL) | Qm(µmol /g) | R2 | ||
| Clay minerals | Kaolinite | 0.083 | 0.269 | 0.607 | 57.751 | 16.488 | 0.965 |
| Halloysite | 0.590 | 0.311 | 0.937 | 26.732 | 35.702 | 0.970 | |
| Equations | Frendlich equation | Langmuir equation | |||||
|---|---|---|---|---|---|---|---|
| In KF | 1/n | R2 | 1/(QmaxKL) | Qm(µmol /g) | R2 | ||
| Clay minerals | Kaolinite | -0.091 | 0.292 | 0.558 | 64.471 | 23.613 | 0.978 |
| Halloysite | 0.177 | 0.358 | 0.951 | 25.723 | 28.571 | 0.997 | |
| Single atoms | Model 1: kaolinite (001) | |||
|---|---|---|---|---|
| atom | Etot (eV) | adsorbate | Etot (eV) | Eads (eV) |
| Pr | -0.157 | Pr | -737.186 | -13.28 |
| Nd | -0.156 | Nd | -737.295 | -13.39 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




