Submitted:
08 July 2025
Posted:
08 July 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
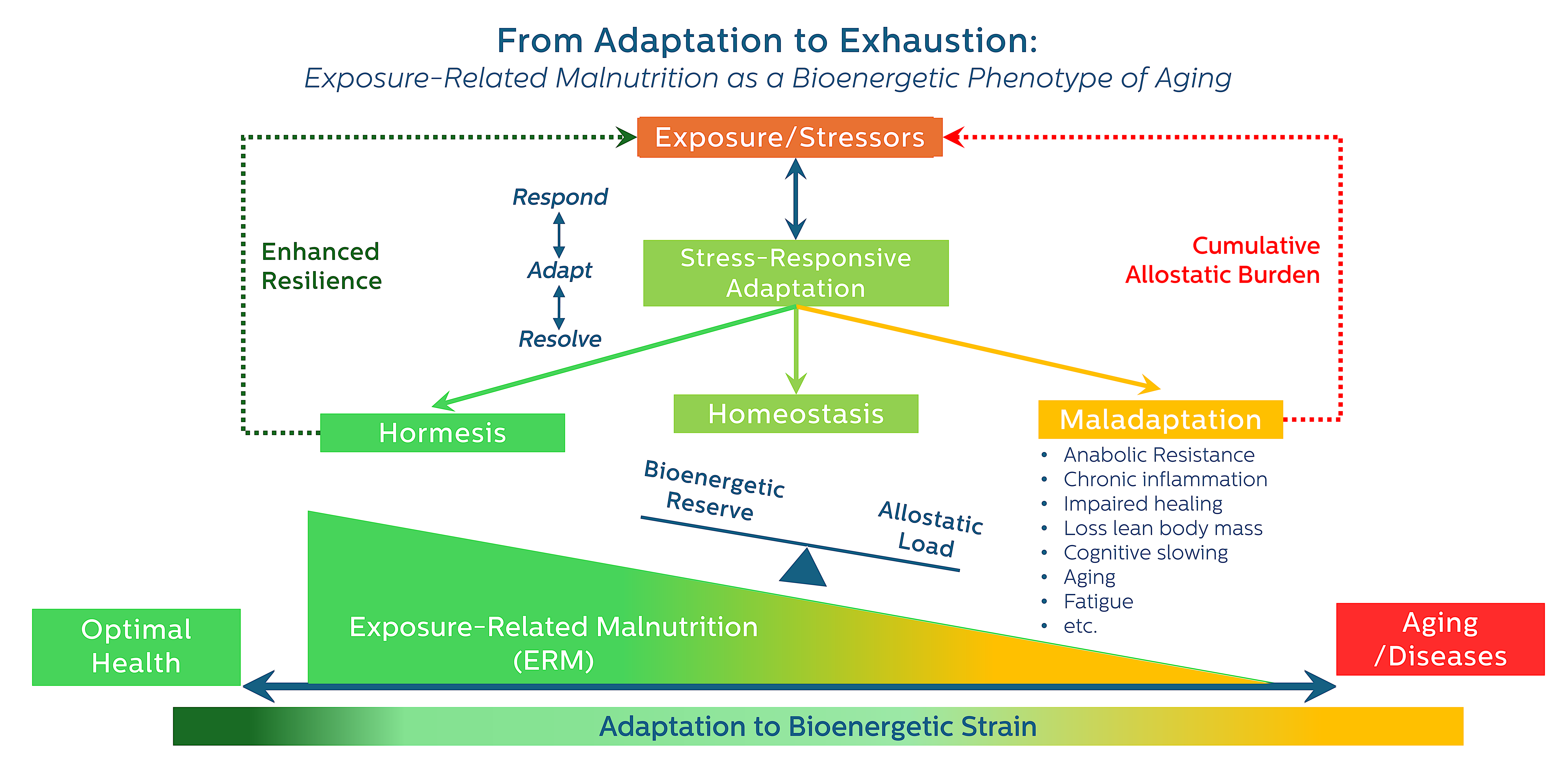
2. Conceptual Model:
“Respond → Adapt → Resolve: A Bioenergetic Trajectory of Stress and Aging”
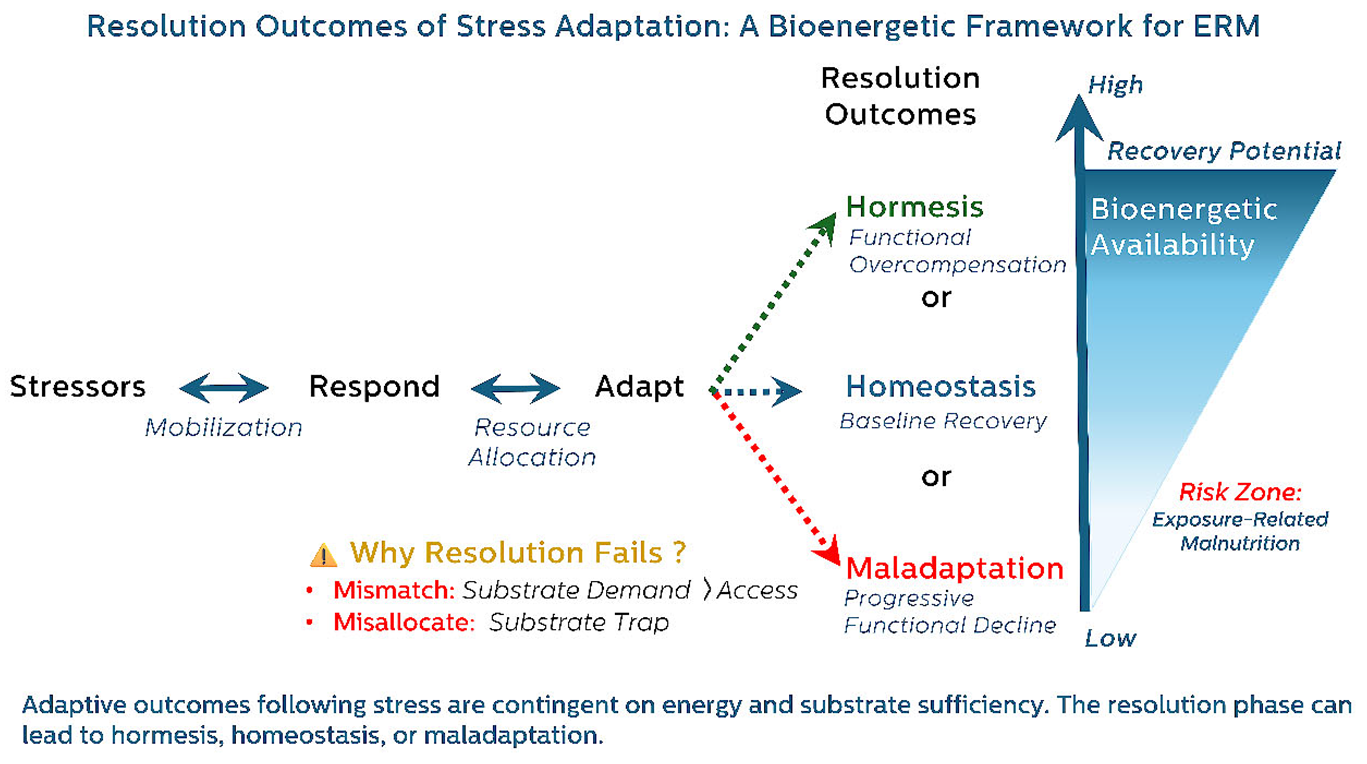
3. Mechanistic Pathways Underlying ERM
3.1. From Central Command to Cellular Collapse: The Bioenergetic Logic of Maladaptive Adaptation
3.2. Neuroendocrine Axis: Central Command of Substrate Allocation
3.3. Immune Reprogramming and Inflammaging: Energy-Intensive Surveillance
3.4. Skeletal Muscle and Anabolic Resistance: The Energetic Reservoir Depleted
3.5. Cellular Integrated Stress Response: Translational Triage Under Strain
3.6. Mitochondrial Stress Response and Mitokines: The Energetic Fulcrum and Feedback Signal
3.7. Closing the Loop: From Peripheral Strain to Central Reprogramming
4. Defining ERM as a Preclinical Aging Phenotype
4.1. ERM vs. Classical Malnutrition Syndromes
4.2. ERM as the Early Metabolic Signature of Resilience Loss in Aging
4.3. Recognizing ERM Phenotypes
4.3.1. Functional Biomarkers: The First Line of ERM Detection
- Handgrip strength and muscle power (dynapenia/powerpenia) have stronger associations with morbidity and mortality than muscle mass alone, and reflect early disruption in anabolic signaling and mitochondrial energy metabolism.
- Gait speed and balance tests capture neuromuscular coordination and are sensitive to cognitive and central nervous system compromise.
- Calf circumference, particularly in the context of preserved BMI, serves as a practical surrogate for declining peripheral muscle mass and functional reserve.
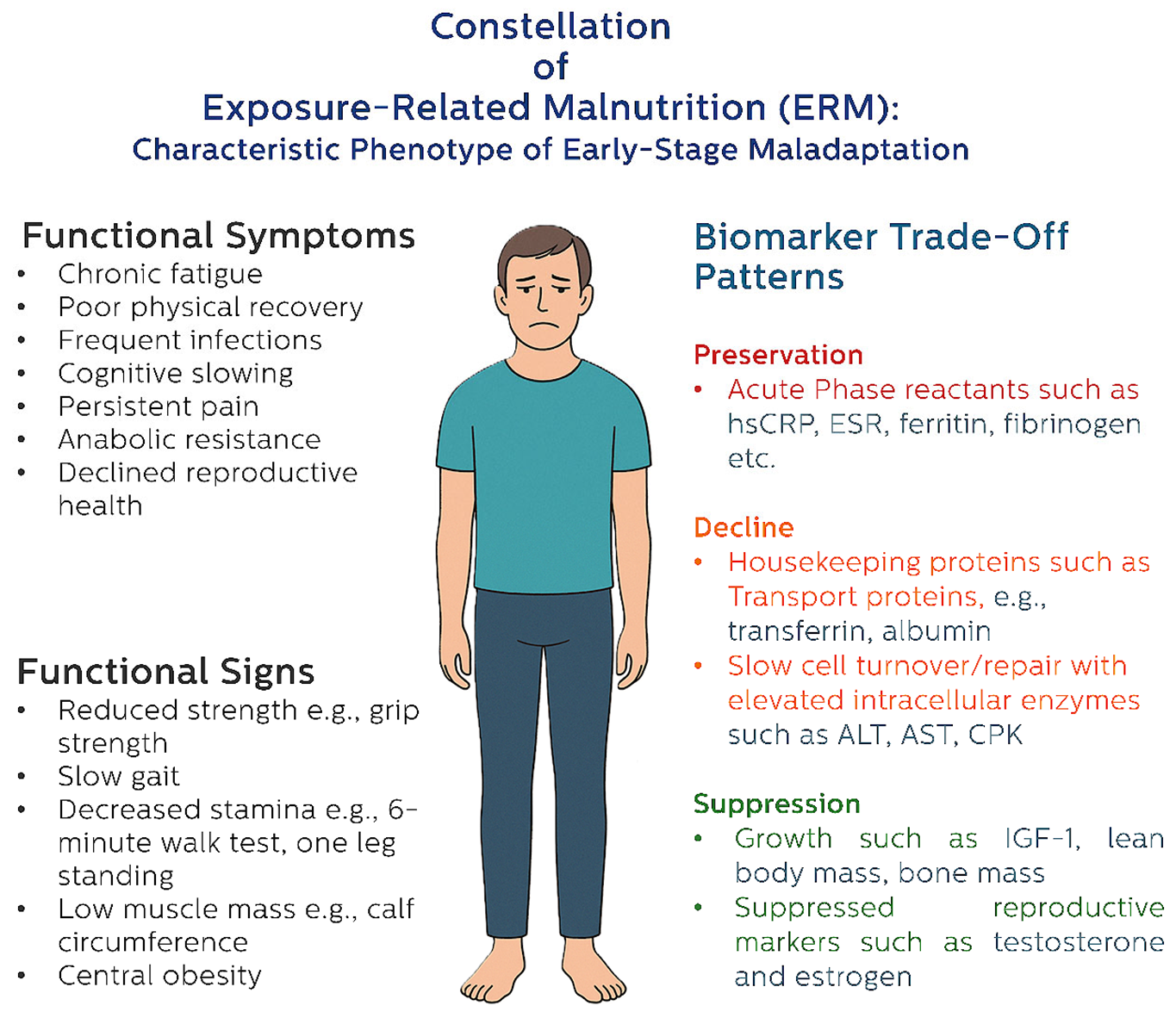
4.3.2. From Structure to Cellular Function: Compositional and Biophysical Markers
- Progressive decline in skeletal muscle mass and bone mineral content signals a shift away from long-term structural investment, consistent with catabolic resource diversion.
- Accumulation of visceral fat, particularly in the presence of stable or rising BMI, reflects a maladaptive redistribution of energy stores—often sustained by hyperinsulinemia and glucose-driven metabolic programming under stress.
- Reduction in phase angle (PhA), a marker of cell membrane integrity and intracellular water balance, reflects impaired cellular vitality and bioenergetic efficiency. Rather than relying on a single cutoff, declining trends in PhA may indicate cumulative stress effects and loss of physiological plasticity. Reduced PhA is also linked to anabolic resistance, sarcopenia, and increased frailty (Akamatsu et al., 2022; Norman et al., 2012).
4.3.3. Patterns Over Points: The ERM Signature
4.3.4. Pattern of Biochemical Trade-Offs: Systemic Signals of Strain
- Preservation or elevation of acute-phase reactants (e.g., CRP, ferritin), coagulation factors, and stress proteins indicate active immune prioritization (Cederholm & Bosaeus, 2024; Sganga et al., 1985).
- Decline in housekeeping proteins such as prealbumin and transferrin marks hepatic reprioritization away from maintenance functions (Evans et al., 2021; Paulussen et al., 2021).
- Suppression of long-term anabolic markers, including IGF-1, sex hormones, and proteins related to muscle, bone, and reproductive function, reflects deeper systemic sacrifice in favor of short-term homeostasis (Bian et al., 2020; Payea et al., 2024; Ryan & Ryznar, 2022).
4.4. ERM and the GLIM Criteria: A Missing Middle
- a “pre-GLIM” phenotype, representing an earlier, subclinical stage of adaptation failure, or
- a parallel subtype of functional malnutrition, primarily driven by maladaptive stress physiology rather than overt intake deficiency.
5. ERM and the Hallmarks of Aging: Reframing Aging as a Failure to Resolve Adaptation
5.1. Mitochondrial Dysfunction → Bioenergetic Reversibility
5.2. Altered Nutrient Sensing → Adaptive Metabolic Flexibility
5.3. Cellular Senescence → Conditional Arrest
5.4. Stem Cell Exhaustion → Resource-Dependent Dormancy
5.5. From Irreversibility to Intervention: ERM as a Critical Inflection Point
6. Translational and Clinical Relevance: Reframing Healthspan Through Adaptive Recovery
6.1. A Window for Preventive Intervention
6.2. Strategies to Restore Metabolic Governance
- Lifestyle-Circadian Synchronization
- Mitochondrial and Nutritional Support for Resilience
- Neuroendocrine and Stress Axis Modulation
6.3. Toward Resilience-Informed Healthspan Strategies
- Monitoring of functional reserve (e.g., grip strength, heart rate recovery, phase angle)
- Recognition of dynamic biomarker constellations rather than reliance on static thresholds
- Restoration of energy availability and systemic plasticity
7. Conclusions
- Systematic reviews should first consolidate existing evidence across the domains of stress physiology, metabolic adaptation, and malnutrition to identify converging biomarker patterns, functional deficits, and compensatory trade-offs consistent with ERM.
- Exploratory studies, including retrospective analyses, biomarker clustering, and cohort profiling, are essential to refine the diagnostic criteria for ERM and to inform the development of a robust staging model that integrates functional symptoms, clinical signs, performance testing, and biomarker patterns.
- With phenotype recognition and staging in place, intervention trials can be designed to target the mechanisms underpinning ERM. These may include strategies to enhance mitochondrial efficiency, restore micronutrient and amino acid availability, synchronize circadian and neuroendocrine rhythms, and promote recovery following sustained stress.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Declaration of generative AI and AI-assisted technologies in the writing process
List of Abbreviations
| Abbreviation | Full Term |
| AMPK | AMP-activated Protein Kinase |
| ATF4 | Activating Transcription Factor 4 |
| ATP | Adenosine Triphosphate |
| BEC | Brain–Body Energy Conservation |
| BIA | Bioelectrical Impedance Analysis |
| BMI | Body Mass Index |
| cGAS–STING | cyclic GMP–AMP synthase–stimulator of interferon genes |
| CED | Chronic Energy Deficiency |
| CHOP | C/EBP Homologous Protein |
| CRP | C-Reactive Protein |
| DHEA | Dehydroepiandrosterone |
| DRM | Disease-Related Malnutrition |
| eIF2α | eukaryotic Initiation Factor 2 Alpha |
| ERM | Exposure-Related Malnutrition |
| FGF21 | Fibroblast Growth Factor 21 |
| GDF15 | Growth Differentiation Factor 15 |
| GLIM | Global Leadership Initiative on Malnutrition |
| HPA | Hypothalamic–Pituitary–Adrenal (axis) |
| IGF-1 | Insulin-like Growth Factor 1 |
| ISR | Integrated Stress Response |
| MBSR | Mindfulness-Based Stress Reduction |
| mTORC1 | mechanistic Target of Rapamycin Complex 1 |
| mt-ISR | Mitochondrial Integrated Stress Response |
| mtDNA | Mitochondrial DNA |
| MPS | Muscle Protein Synthesis |
| NLRP3 | NOD-like receptor family pyrin domain-containing 3 |
| PhA | Phase Angle |
| RED-S | Relative Energy Deficiency in Sport |
| ROS | Reactive Oxygen Species |
| SAM | Sympathetic–Adrenal–Medullary |
| SASP | Senescence-Associated Secretory Phenotype |
| SCENITH | Single-Cell Energetic metabolism by Translation Inhibition |
| scRNA-seq | single-cell RNA sequencing |
| UPR | Unfolded Protein Response |
References
- Akamatsu, Y., Kusakabe, T., Arai, H., Yamamoto, Y., Nakao, K., Ikeue, K.,…Satoh-Asahara, N. (2022). Phase angle from bioelectrical impedance analysis is a useful indicator of muscle quality. J Cachexia Sarcopenia Muscle, 13(1), 180-189. [CrossRef]
- Alack, K., Pilat, C., & Krüger K*Shared, a. (2019). Current Knowledge and New Challenges in Exercise Immunology. Deutsche Zeitschrift für Sportmedizin, Volume 70(No. 10), 250-260. [CrossRef]
- Ames, B. N. (2006). Low micronutrient intake may accelerate the degenerative diseases of aging through allocation of scarce micronutrients by triage. Proc Natl Acad Sci U S A, 103(47), 17589-17594. [CrossRef]
- Arron, H. E., Marsh, B. D., Kell, D. B., Khan, M. A., Jaeger, B. R., & Pretorius, E. (2024). Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: the biology of a neglected disease. Front Immunol, 15, 1386607. [CrossRef]
- Beck, F. K., & Rosenthal, T. C. (2002). Prealbumin: a marker for nutritional evaluation. Am Fam Physician, 65(8), 1575-1578. https://www.aafp.org/afp/2002/0415/p1575.pdf.
- Bian, A., Ma, Y., Zhou, X., Guo, Y., Wang, W., Zhang, Y., & Wang, X. (2020). Association between sarcopenia and levels of growth hormone and insulin-like growth factor-1 in the elderly. BMC Musculoskelet Disord, 21(1), 214. [CrossRef]
- Bobba-Alves, N., Sturm, G., Lin, J., Ware, S. A., Karan, K. R., Monzel, A. S.,…Picard, M. (2023). Cellular allostatic load is linked to increased energy expenditure and accelerated biological aging. Psychoneuroendocrinology, 155, 106322. [CrossRef]
- Brzezniakiewicz-Janus, K., Jarczak, J., Konopko, A., Ratajczak, J., Kucia, M., & Ratajczak, M. Z. (2025). Mitochondria Express Functional Signaling Ligand-Binding Receptors that Regulate their Biological Responses—the Novel Role of Mitochondria as Stress-Response Sentinels. Stem Cell Reviews and Reports. [CrossRef]
- Cabre, H. E., Moore, S. R., Smith-Ryan, A. E., & Hackney, A. C. (2022). Relative Energy Deficiency in Sport (RED-S): Scientific, Clinical, and Practical Implications for the Female Athlete. Dtsch Z Sportmed, 73(7), 225-234. [CrossRef]
- Cahill, G. F., Jr. (2006). Fuel metabolism in starvation. Annu Rev Nutr, 26, 1-22. [CrossRef]
- Calabrese, E. J., Nascarella, M., Pressman, P., Hayes, A. W., Dhawan, G., Kapoor, R.,…Agathokleous, E. (2024). Hormesis determines lifespan. Ageing Res Rev, 94, 102181. [CrossRef]
- Cederholm, T., & Bosaeus, I. (2024). Malnutrition in Adults. New England Journal of Medicine, 391(2), 155-165. [CrossRef]
- Cederholm, T., Jensen, G. L., Correia, M. I. T. D., Gonzalez, M. C., Fukushima, R., Pisprasert, V.,…Compher, C. (2025). The GLIM consensus approach to diagnosis of malnutrition: A 5-year update. Clin Nutr, 49, 11-20. [CrossRef]
- Cheung, T. H., & Rando, T. A. (2013). Molecular regulation of stem cell quiescence. Nat Rev Mol Cell Biol, 14(6), 329-340. [CrossRef]
- Chrousos, G. P. (2009). Stress and disorders of the stress system. Nat Rev Endocrinol, 5(7), 374-381. [CrossRef]
- Costa-Mattioli, M., & Walter, P. (2020). The integrated stress response: From mechanism to disease. Science, 368(6489), eaat5314. [CrossRef]
- Crane, P. A., Wilkinson, G., & Teare, H. (2022). Healthspan versus lifespan: new medicines to close the gap. Nature Aging, 2(11), 984-988. [CrossRef]
- Demirovic, D., & Rattan, S. I. S. (2013). Establishing cellular stress response profiles as biomarkers of homeodynamics, health and hormesis. Experimental gerontology, 48, 94-98.
- Diaz-Meco, M. T., Linares, J. F., & Moscat, J. (2025). Hijacking the powerhouse: Mitochondrial transfer and mitophagy as emerging mechanisms of immune evasion. Molecular Cell, 85(7), 1258-1259. [CrossRef]
- Dong, L.-F., Rohlena, J., Zobalova, R., Nahacka, Z., Rodriguez, A.-M., Berridge, M. V., & Neuzil, J. (2023). Mitochondria on the move: Horizontal mitochondrial transfer in disease and health. Journal of Cell Biology, 222(3). [CrossRef]
- Evans, D. C., Corkins, M. R., Malone, A., Miller, S., Mogensen, K. M., Guenter, P.,…Committee, A. M. (2021). The Use of Visceral Proteins as Nutrition Markers: An ASPEN Position Paper. Nutr Clin Pract, 36(1), 22-28. [CrossRef]
- Ferreira, R. P., & Duarte, J. A. (2023). Protein Turnover in Skeletal Muscle: Looking at Molecular Regulation towards an Active Lifestyle. Int J Sports Med, 44(11), 763-777. [CrossRef]
- Foy, B. H., Petherbridge, R., Roth, M. T., Zhang, C., De Souza, D. C., Mow, C.,…Higgins, J. M. (2024). Haematological setpoints are a stable and patient-specific deep phenotype. Nature. [CrossRef]
- Franceschi, C., Garagnani, P., Parini, P., Giuliani, C., & Santoro, A. (2018). Inflammaging: a new immune–metabolic viewpoint for age-related diseases. Nature Reviews Endocrinology, 14(10), 576-590. [CrossRef]
- Fülöp, B., Borbély, É., & Helyes, Z. (2025). How does chronic psychosocial distress induce pain? Focus on neuroinflammation and neuroplasticity changes. Brain, Behavior, & Immunity—Health, 44, 100964. [CrossRef]
- Fulop, T., Larbi, A., Dupuis, G., Le Page, A., Frost, E. H., Cohen, A. A.,…Franceschi, C. (2018). Immunosenescence and Inflamm-Aging As Two Sides of the Same Coin: Friends or Foes? [Review]. Front Immunol, 8(1960). [CrossRef]
- Furrer, R., & Handschin, C. (2025a). Biomarkers of aging: from molecules and surrogates to physiology and function. Physiol Rev, 105(3), 1609-1694. [CrossRef]
- Furrer, R., & Handschin, C. (2025b). Biomarkers of aging: functional aspects still trump molecular parameters. npj Aging, 11(1), 15. [CrossRef]
- Gulhar, R., Ashraf, M. A., & Jialal, I. (2024). Physiology, Acute Phase Reactants. In StatPearls. StatPearls Publishing.
- Harrell, C. S., Gillespie, C. F., & Neigh, G. N. (2016). Energetic stress: The reciprocal relationship between energy availability and the stress response. Physiol Behav, 166, 43-55. [CrossRef]
- Hetz, C., & Papa, F. R. (2018). The Unfolded Protein Response and Cell Fate Control. Molecular Cell, 69(2), 169-181. [CrossRef]
- Hetz, C., & Saxena, S. (2017). ER stress and the unfolded protein response in neurodegeneration. Nat Rev Neurol, 13(8), 477-491. [CrossRef]
- Ikeue, K., Kato, H., Tanaka, M., Yamakage, H., Kato, S., Iwasa, M.,…Satoh-Asahara, N. (2025). Phase Angle Is a Potential Novel Early Marker for Sarcopenia and Cognitive Impairment in the General Population. J Cachexia Sarcopenia Muscle, 16(3), e13820. [CrossRef]
- Islam, M. N., Das, S. R., Emin, M. T., Wei, M., Sun, L., Westphalen, K.,…Bhattacharya, J. (2012). Mitochondrial transfer from bone-marrow-derived stromal cells to pulmonary alveoli protects against acute lung injury. Nat Med, 18(5), 759-765. [CrossRef]
- Kivimäki, M., Bartolomucci, A., & Kawachi, I. (2023). The multiple roles of life stress in metabolic disorders. Nature Reviews Endocrinology, 19(1), 10-27. [CrossRef]
- Langston, P. K., & Mathis, D. (2024). Immunological regulation of skeletal muscle adaptation to exercise. Cell Metab. [CrossRef]
- Li, Y., Wang, K., Jigeer, G., Jensen, G., Tucker, K. L., Lv, Y.,…Gao, X. (2024). Healthy Lifestyle and the Likelihood of Becoming a Centenarian. JAMA Network Open, 7(6), e2417931-e2417931. [CrossRef]
- Lockhart, S. M., Saudek, V., & O’Rahilly, S. (2020). GDF15: A Hormone Conveying Somatic Distress to the Brain. Endocr Rev, 41(4). [CrossRef]
- Lopez-Otin, C., Blasco, M. A., Partridge, L., Serrano, M., & Kroemer, G. (2013). The hallmarks of aging. Cell, 153(6), 1194-1217. [CrossRef]
- López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M., & Kroemer, G. (2023). Hallmarks of aging: An expanding universe. Cell. [CrossRef]
- López-Otín, C., & Kroemer, G. (2021). Hallmarks of Health. Cell, 184(1), 33-63. [CrossRef]
- López-Otín, C., & Kroemer, G. (2024). The missing hallmark of health: psychosocial adaptation. Cell Stress, 8, 21-50. [CrossRef]
- Martel, J., Ojcius, D. M., & Young, J. D. (2024). Lifestyle interventions to delay senescence. Biomedical Journal, 47(2), 100676. [CrossRef]
- McEwen, B. S. (2007). Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev, 87(3), 873-904. [CrossRef]
- McEwen, B. S., & Wingfield, J. C. (2003). The concept of allostasis in biology and biomedicine. Hormones and Behavior, 43(1), 2-15. [CrossRef]
- McEwen, C. A. (2022). Connecting the biology of stress, allostatic load and epigenetics to social structures and processes. Neurobiol Stress, 17, 100426. [CrossRef]
- Monzel, A. S., Levin, M., & Picard, M. (2023). The energetics of cellular life transitions. Life Metabolism, 3(3). [CrossRef]
- Mountjoy, M., Sundgot-Borgen, J. K., Burke, L. M., Ackerman, K. E., Blauwet, C., Constantini, N.,…Budgett, R. (2018). IOC consensus statement on relative energy deficiency in sport (RED-S): 2018 update. Br J Sports Med, 52(11), 687-697. [CrossRef]
- Muscaritoli, M., Imbimbo, G., Jager-Wittenaar, H., Cederholm, T., Rothenberg, E., di Girolamo, F. G.,…Molfino, A. (2023). Disease-related malnutrition with inflammation and cachexia. Clin Nutr, 42(8), 1475-1479. [CrossRef]
- National Academies of Sciences, E., & Medicine. (2024). Toward a Common Research Agenda in Infection-Associated Chronic Illnesses: Proceedings of a Workshop. The National Academies Press. [CrossRef]
- Ni, H.-M., Williams, J. A., & Ding, W.-X. (2015). Mitochondrial dynamics and mitochondrial quality control. Redox Biology, 4, 6-13. [CrossRef]
- Norman, K., Stobäus, N., Pirlich, M., & Bosy-Westphal, A. (2012). Bioelectrical phase angle and impedance vector analysis – Clinical relevance and applicability of impedance parameters. Clin Nutr, 31(6), 854-861. [CrossRef]
- Olenchock, B. A., Rathmell, J. C., & Vander Heiden, M. G. (2017). Biochemical Underpinnings of Immune Cell Metabolic Phenotypes. Immunity, 46(5), 703-713. [CrossRef]
- Pakos-Zebrucka, K., Koryga, I., Mnich, K., Ljujic, M., Samali, A., & Gorman, A. M. (2016). The integrated stress response. EMBO reports, 17(10), 1374-1395. [CrossRef]
- Paulussen, K. J. M., McKenna, C. F., Beals, J. W., Wilund, K. R., Salvador, A. F., & Burd, N. A. (2021). Anabolic Resistance of Muscle Protein Turnover Comes in Various Shapes and Sizes. Front Nutr, 8, 615849. [CrossRef]
- Payea, M. J., Dar, S. A., Anerillas, C., Martindale, J. L., Belair, C., Munk, R.,…Maragkakis, M. (2024). Senescence suppresses the integrated stress response and activates a stress-remodeled secretory phenotype. Molecular Cell, 84(22), 4454-4469.e4457. [CrossRef]
- Peters, A. (2004). The selfish brain: competition for energy resources. American Journal of Human Biology, 23(1), 29-34. [CrossRef]
- Peters, A., Sprengell, M., & Kubera, B. (2022). The principle of ‘brain energy on demand’ and its predictive power for stress, sleep, stroke, obesity and diabetes. Neuroscience & Biobehavioral Reviews, 141, 104847. [CrossRef]
- Picard, M., McEwen, B., Epel, E., & Sandi, C. (2018). An Energetic View of Stress: Focus on Mitochondria. Front Neuroendocrinol, 49, 72-85. [CrossRef]
- Picard, M., & Shirihai, O. S. (2022). Mitochondrial signal transduction. Cell Metab, 34(11), 1620-1653. [CrossRef]
- Prisabela, M., Nadhiroh, S. R., & Isaura, E. R. (2023). Characteristics of Pregnant Woman with Chronic Energy Deficiency in Puskesmas Gesang, Lumajang on 2020: Descriptive Analysis. Media Gizi Kesmas, 12(2), 643-648. [CrossRef]
- Qi, Y., Rajbanshi, B., Hao, R., Dang, Y., Xu, C., Lu, W.,…Zhang, X. (2025). The dual role of PGAM5 in inflammation. Exp Mol Med, 57(2), 298-311. [CrossRef]
- Rappaport, S. M., Barupal, D. K., Wishart, D., Vineis, P., & Scalbert, A. (2014). The blood exposome and its role in discovering causes of disease. Environ Health Perspect, 122(8), 769-774. [CrossRef]
- Rattan, S. I. S. (2020). Homeostasis, Homeodynamics and Aging. In S. I. S. Rattan (Ed.), Encyclopedia of Biomedical Gerontology (pp. 238-241). Academic Press. [CrossRef]
- Ray, C. S., Singh, B., Jena, I., Behera, S., & Ray, S. (2017). Low Alkaline Phosphatase (ALP) In Adult Population an Indicator of Zinc (Zn) and Magnesium (Mg) Deficiency. Current Research in Nutrition and Food Science Journal, 5, 347-352.
- Ristow, M., & Schmeisser, K. (2014). Mitohormesis: Promoting Health and Lifespan by Increased Levels of Reactive Oxygen Species (ROS). Dose-Response, 12(2), dose-response.13-035.Ristow. [CrossRef]
- Ryall, J. G., Cliff, T., Dalton, S., & Sartorelli, V. (2015). Metabolic Reprogramming of Stem Cell Epigenetics. Cell Stem Cell, 17(6), 651-662. [CrossRef]
- Ryan, M., & Ryznar, R. (2022). The Molecular Basis of Resilience: A Narrative Review [Review]. Frontiers in Psychiatry, 13. [CrossRef]
- Sapolsky, R. M. (2004). Why zebras don’t get ulcers: The acclaimed guide to stress, stress-related diseases, and coping (3rd ed.). Henry Holt.
- Schmauck-Medina, T., Molière, A., Lautrup, S., Zhang, J., Chlopicki, S., Madsen, H. B.,…Fang, E. F. (2022). New hallmarks of ageing: A 2022 Copenhagen ageing meeting summary. Aging (Albany NY). [CrossRef]
- Seeman, T. E., McEwen, B. S., Rowe, J. W., & Singer, B. H. (2001). Allostatic load as a marker of cumulative biological risk: MacArthur studies of successful aging. Proc Natl Acad Sci U S A, 98(8), 4770-4775. [CrossRef]
- Selye, H. (1950). Stress and the General Adaptation Syndrome. British Medical Journal, 1(4667), 1383-1392. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2038162/.
- Sganga, G., Siegel, J. H., Brown, G., Coleman, B., Wiles, C. E., III, Belzberg, H.,…Placko, R. (1985). Reprioritization of Hepatic Plasma Protein Release in Trauma and Sepsis. Archives of Surgery, 120(2), 187-199. [CrossRef]
- Shaulson, E. D., Cohen, A. A., & Picard, M. (2024). The brain–body energy conservation model of aging. Nature Aging, 4(10), 1354-1371. [CrossRef]
- Shirai, T., & Tsushita, K. (2024). Lifestyle Medicine and Japan’s Longevity Miracle. American Journal of Lifestyle Medicine, 18(4), 598-607. [CrossRef]
- Spees, J. L., Olson, S. D., Whitney, M. J., & Prockop, D. J. (2006). Mitochondrial transfer between cells can rescue aerobic respiration. Proc Natl Acad Sci U S A, 103(5), 1283-1288. [CrossRef]
- Srour, R. A., & Keyes, D. (2025). Lifestyle Mindfulness In Clinical Practice. In StatPearls. StatPearls Publishing Copyright © 2025, StatPearls Publishing LLC.
- Tippairote, T., Bjørklund, G., & Yaovapak, A. (2021). The continuum of disrupted metabolic tempo, mitochondrial substrate congestion, and metabolic gridlock toward the development of non-communicable diseases. Critical Reviews in Food Science and Nutrition. [CrossRef]
- Tsigos, C., & Chrousos, G. P. (2002). Hypothalamic–pituitary–adrenal axis, neuroendocrine factors and stress. Journal of Psychosomatic Research, 53(4), 865-871. [CrossRef]
- Vermeulen, R., Schymanski, E. L., Barabási, A. L., & Miller, G. W. (2020). The exposome and health: Where chemistry meets biology. Science, 367(6476), 392-396. [CrossRef]
- Walrand, S., Guillet, C., & Boirie, Y. (2021). Nutrition, Protein Turnover and Muscle Mass. In Sarcopenia (pp. 45-62). [CrossRef]
- Wang, X., & Zhang, G. (2025). The mitochondrial integrated stress response: A novel approach to anti-aging and pro-longevity. Ageing Res Rev, 103, 102603. [CrossRef]
- Wek, R. C. (2018). Role of eIF2α Kinases in Translational Control and Adaptation to Cellular Stress. Cold Spring Harb Perspect Biol, 10(7). [CrossRef]
- WHO. (2025). Noncommunicable diseases. WHO. Retrieved March 25 from https://www.who.int/health-topics/noncommunicable-diseases#tab=tab_1.
- Wiley, C. D., & Campisi, J. (2021). The metabolic roots of senescence: mechanisms and opportunities for intervention. Nature Metabolism, 3(10), 1290-1301. [CrossRef]
- Willmann, K., & Moita, L. F. (2024). Physiologic disruption and metabolic reprogramming in infection and sepsis. Cell Metab. [CrossRef]
- Wu, F., Mu, W.-C., Markov, N. T., Fuentealba, M., Halaweh, H., Senchyna, F.,…Furman, D. (2025). Immunological biomarkers of aging. The Journal of Immunology, 214(5), 889-902. [CrossRef]
- Wu, Z., Qu, J., Zhang, W., & Liu, G.-H. (2024). Stress, epigenetics, and aging: Unraveling the intricate crosstalk. Molecular Cell, 84(1), 34-54. [CrossRef]
- Yiallouris, A., Tsioutis, C., Agapidaki, E., Zafeiri, M., Agouridis, A. P., Ntourakis, D., & Johnson, E. O. (2019). Adrenal Aging and Its Implications on Stress Responsiveness in Humans [Review]. Frontiers in Endocrinology, 10(54). [CrossRef]
- Zhang, B., Chang, J. Y., Lee, M. H., Ju, S.-H., Yi, H.-S., & Shong, M. (2024). Mitochondrial Stress and Mitokines: Therapeutic Perspectives for the Treatment of Metabolic Diseases. Diabetes Metab J, 48(1), 1-18. [CrossRef]
| Domain | GLIM Malnutrition Framework | ERM Malnutrition Framework |
| Focus | Observable malnutrition | Subclinical bioenergetic exhaustion |
| Phenotypic Criteria | Weight loss, low BMI, low muscle mass | Fatigue, immune dysfunction, reduced phase angle, anabolic resistance |
| Etiologic Criteria | Inflammation, reduced intake, disease burden | Chronic adaptation to physiological stress, environmental burden, and cumulative lifestyle exposures |
| Detection Sensitivity | Moderate-to-late-stage malnutrition | High; based on emerging biomarkers and functional pattern recognition |
| Intervention Window | Post-functional decline | Early, targets reversible physiological compromise before functional decline |
| Biomarker Use | Optional; not central to diagnosis | Central to detection; includes acute phase reactants, cellular turnover, anabolic and stress-response markers |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




