Submitted:
09 April 2025
Posted:
10 April 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Classes of RNA Therapeutics
Antisense Oligonucleotides (ASOs)
Small Interfering RNA (siRNA) Therapies
Messenger RNA (mRNA) Therapies
MicroRNA Based Therapies
RNA Editing Technologies
Alternative Approaches to RNA Targeting
Clinical Applications and Recent Advances in RNA Therapeutics
Cardiovascular Diseases
Genetic Disorders
Oncology: RNA-Based Cancer Therapies
Neurological Disorders
Infectious Diseases
Expanding the Scope of RNA Therapeutics
Challenges in RNA Delivery
Prospects and Innovations in RNA Delivery
Conclusions and Outlook
Author Declarations
References
- Adams, D.; Gonzalez-Duarte, A.; O’Riordan, W. D.; Yang, C.-C.; Ueda, M.; Kristen, A. V.; Tournev, I.; Schmidt, H. H.; Coelho, T.; Berk, J. L.; Lin, K.-P.; Vita, G.; Attarian, S.; Planté-Bordeneuve, V.; Mezei, M. M.; Campistol, J. M.; Buades, J.; Brannagan, T. H.; Kim, B. J.; Suhr, O. B. Patisiran, an RNAi Therapeutic, for Hereditary Transthyretin Amyloidosis. New England Journal of Medicine 2018, 379(1), 11–21. [Google Scholar] [CrossRef] [PubMed]
- Alshaer, W.; Zureigat, H.; Al Karaki, A.; Al-Kadash, A.; Gharaibeh, L.; Hatmal, M. M.; Aljabali, A. A. A.; Awidi, A. siRNA: Mechanism of action, challenges, and therapeutic approaches. European Journal of Pharmacology 2021, 905, 174178. [Google Scholar] [CrossRef] [PubMed]
- Antony, J. S.; Birrer, P.; Bohnert, C.; Zimmerli, S.; Hillmann, P.; Schaffhauser, H.; Hoeflich, C.; Hoeflich, A.; Khairallah, R.; Satoh, A. T.; Kappeler, I.; Ferreira, I.; Zuideveld, K. P.; Metzger, F. Local application of engineered insulin-like growth factor I mRNA demonstrates regenerative therapeutic potential in vivo. Molecular Therapy Nucleic Acids 2023, 34. [Google Scholar] [CrossRef] [PubMed]
- Baden, L. R.; Sahly, H. M. E.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S. A.; Rouphael, N.; Creech, C. B.; McGettigan, J.; Khetan, S.; Segall, N.; Solis, J.; Brosz, A.; Fierro, C.; Schwartz, H.; Neuzil, K.; Corey, L.; Zaks, T. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. New England Journal of Medicine 2021, 384(5), 403–416. [Google Scholar] [CrossRef]
- Bartel, D. P. Metazoan MicroRNAs. Cell 2018, 173(1), 20–51. [Google Scholar] [CrossRef]
- Beg, M. S.; Brenner, A. J.; Sachdev, J.; Borad, M.; Kang, Y.-K.; Stoudemire, J.; Smith, S.; Bader, A. G.; Kim, S.; Hong, D. S. Phase I study of MRX34, a liposomal miR-34a mimic, administered twice weekly in patients with advanced solid tumors. Investigational New Drugs 2017, 35(2), 180–188. [Google Scholar] [CrossRef]
- Bennett, C. F.; Kordasiewicz, H. B.; Cleveland, D. W. Antisense Drugs Make Sense for Neurological Diseases. Annual Review of Pharmacology and Toxicology 2021, 61, 831–852. [Google Scholar] [CrossRef]
- Benson, M. D.; Waddington-Cruz, M.; Berk, J. L.; Polydefkis, M.; Dyck, P. J.; Wang, A. K.; Planté-Bordeneuve, V.; Barroso, F. A.; Merlini, G.; Obici, L.; Scheinberg, M.; Brannagan, T. H.; Litchy, W. J.; Whelan, C.; Drachman, B. M.; Adams, D.; Heitner, S. B.; Conceição, I.; Schmidt, H. H.; Coelho, T. Inotersen Treatment for Patients with Hereditary Transthyretin Amyloidosis. New England Journal of Medicine 2018, 379(1), 22–31. [Google Scholar] [CrossRef]
- Bhinder, B.; Gilvary, C.; Madhukar, N. S.; Elemento, O. Artificial Intelligence in Cancer Research and Precision Medicine. Cancer Discovery 2021, 11(4), 900–915. [Google Scholar] [CrossRef]
- Bian, X.; Guo, Q.; Yau, L.-F.; Yang, L.; Wang, X.; Zhao, S.; Wu, S.; Qin, X.; Jiang, Z.-H.; Li, C. Berberine-inspired ionizable lipid for self-structure stabilization and brain targeting delivery of nucleic acid therapeutics. Nature Communications 2025, 16(1), 2368. [Google Scholar] [CrossRef]
- Bloom, K.; van den Berg, F.; Arbuthnot, P. Self-amplifying RNA vaccines for infectious diseases. Gene Therapy 2021, 28(3), 117–129. [Google Scholar] [CrossRef] [PubMed]
- Booth, B. J.; Nourreddine, S.; Katrekar, D.; Savva, Y.; Bose, D.; Long, T. J.; Huss, D. J.; Mali, P. RNA editing: Expanding the potential of RNA therapeutics. Molecular Therapy 2023, 31(6), 1533–1549. [Google Scholar] [CrossRef]
- Bottens, R. A.; Yamada, T. Cell-Penetrating Peptides (CPPs) as Therapeutic and Diagnostic Agents for Cancer. Cancers 2022, 14(22), 5546. [Google Scholar] [CrossRef]
- Bowlby, B. Synthetic riboswitches detect biomarker proteins. BioTechniques. 9 June 2023. Available online: https://www.biotechniques.com/diagnostics-preclinical/synthetic-riboswitches-detect-biomarker-proteins/ (accessed on 19 November 2024).
- Brannagan, T. H.; Berk, J. L.; Gillmore, J. D.; Maurer, M. S.; Waddington-Cruz, M.; Fontana, M.; Masri, A.; Obici, L.; Brambatti, M.; Baker, B. F.; Hannan, L. A.; Buchele, G.; Viney, N. J.; Coelho, T.; Nativi-Nicolau, J. Liver-directed drugs for transthyretin-mediated amyloidosis. Journal of the Peripheral Nervous System 2022, 27(4), 228–237. [Google Scholar] [CrossRef] [PubMed]
- Brasil, S.; Pascoal, C.; Francisco, R.; dos Reis Ferreira, V.; A. Videira, P.; Valadão, G. Artificial Intelligence (AI) in Rare Diseases: Is the Future Brighter? Genes 2019, 10(12), 978. [Google Scholar] [CrossRef]
- Breaker, R. R. Riboswitches and the RNA world. Cold Spring Harbor Perspectives in Biology 2012, 4(2), a003566. [Google Scholar] [CrossRef] [PubMed]
- Calin, G. A.; Croce, C. M. MicroRNA signatures in human cancers. Nature Reviews. Cancer 2006, 6(11), 857–866. [Google Scholar] [CrossRef]
- Cao, Q.; Fang, H.; Tian, H. mRNA vaccines contribute to innate and adaptive immunity to enhance immune response in vivo. Biomaterials 2024, 310, 122628. [Google Scholar] [CrossRef]
- Chatterjee, S.; Kon, E.; Sharma, P.; Peer, D. Endosomal escape: A bottleneck for LNP-mediated therapeutics. Proceedings of the National Academy of Sciences 2024, 121(11), e2307800120. [Google Scholar] [CrossRef]
- Chen, S.; Heendeniya, S. N.; Le, B. T.; Rahimizadeh, K.; Rabiee, N.; Zahra; ul ain, Q.; Veedu, R. N. Splice-Modulating Antisense Oligonucleotides as Therapeutics for Inherited Metabolic Diseases. Biodrugs 2024, 38(2), 177–203. [Google Scholar] [CrossRef]
- Chery, J. RNA therapeutics: RNAi and antisense mechanisms and clinical applications. Postdoc Journal: A Journal of Postdoctoral Research and Postdoctoral Affairs 2016, 4(7), 35–50. [Google Scholar] [CrossRef] [PubMed]
- Childs-Disney, J. L.; Yang, X.; Gibaut, Q. M. R.; Tong, Y.; Batey, R. T.; Disney, M. D. Targeting RNA structures with small molecules. Nature Reviews. Drug Discovery 2022, 21(10), 736–762. [Google Scholar] [CrossRef]
- Choi, G.-W.; Kim, J. H.; Kang, D. W.; Cho, H.-Y. A journey into siRNA therapeutics development: A focus on Pharmacokinetics and Pharmacodynamics. European Journal of Pharmaceutical Sciences 2025, 205, 106981. [Google Scholar] [CrossRef]
- Coelho, T.; Marques, W.; Dasgupta, N. R.; Chao, C.-C.; Parman, Y.; França, M. C.; Guo, Y.-C.; Wixner, J.; Ro, L.-S.; Calandra, C. R.; Kowacs, P. A.; Berk, J. L.; Obici, L.; Barroso, F. A.; Weiler, M.; Conceição, I.; Jung, S. W.; Buchele, G.; Brambatti, M. NEURO-TTRansform Investigators. (2023). Eplontersen for Hereditary Transthyretin Amyloidosis With Polyneuropathy. JAMA 330(15), 1448–1458. [CrossRef]
- Crooke, S. T.; Witztum, J. L.; Bennett, C. F.; Baker, B. F. RNA-Targeted Therapeutics. Cell Metabolism 2018, 27(4), 714–739. [Google Scholar] [CrossRef]
- Erion, D. M.; Liu, L. Y.; Brown, C. R.; Rennard, S.; Farah, H. Editing Approaches to Treat Alpha-1 Antitrypsin Deficiency. CHEST 2025, 167(2), 444–452. [Google Scholar] [CrossRef] [PubMed]
- Fàbrega, C.; Aviñó, A.; Eritja, R. Chemical Modifications in Nucleic Acids for Therapeutic and Diagnostic Applications. Chemical Record (New York, N.Y.) 2022, 22(4), e202100270. [Google Scholar] [CrossRef] [PubMed]
- Finkel, R. S.; Mercuri, E.; Darras, B. T.; Connolly, A. M.; Kuntz, N. L.; Kirschner, J.; Chiriboga, C. A.; Saito, K.; Servais, L.; Tizzano, E.; Topaloglu, H.; Tulinius, M.; Montes, J.; Glanzman, A. M.; Bishop, K.; Zhong, Z. J.; Gheuens, S.; Bennett, C. F.; Schneider, E.; ENDEAR Study Group. Nusinersen versus Sham Control in Infantile-Onset Spinal Muscular Atrophy. The New England Journal of Medicine 2017, 377(18), 1723–1732. [Google Scholar] [CrossRef]
- Foinquinos, A.; Batkai, S.; Genschel, C.; Viereck, J.; Rump, S.; Gyöngyösi, M.; Traxler, D.; Riesenhuber, M.; Spannbauer, A.; Lukovic, D.; Weber, N.; Zlabinger, K.; Hašimbegović, E.; Winkler, J.; Fiedler, J.; Dangwal, S.; Fischer, M.; Roche; de la, J.; Wojciechowski, D.; Thum, T. Preclinical development of a miR-132 inhibitor for heart failure treatment. Nature Communications 2020, 11, 633. [Google Scholar] [CrossRef]
- Frangoul, H.; Altshuler, D.; Cappellini, M. D.; Chen, Y.-S.; Domm, J.; Eustace, B. K.; Foell, J.; Fuente; de la, J.; Grupp, S.; Handgretinger, R.; Ho, T. W.; Kattamis, A.; Kernytsky, A.; Lekstrom-Himes, J.; Li, A. M.; Locatelli, F.; Mapara, M. Y.; Montalembert, M. de; Rondelli, D.; Corbacioglu, S. CRISPR-Cas9 Gene Editing for Sickle Cell Disease and β-Thalassemia. New England Journal of Medicine 2021, 384(3), 252–260. [Google Scholar] [CrossRef]
- News, AP. Gates Foundation funding $40 million effort to help develop mRNA vaccines in Africa in coming years. 2023. Available online: https://apnews.com/article/bill-gates-foundation-africa-vaccines-mrna-institut-pasteur-biovac-fa28c0502925a4152df1709cc8f228fe (accessed on 7 April 2025).
- Geller, D. E.; Crowley, C.; Froehlich, J.; Schwabe, C.; O’Carroll, M. WS10.03 Inhaled LUNAR®-CFTR mRNA (ARCT-032) is safe and well-tolerated: A phase 1 study. Journal of Cystic Fibrosis 2024, 23, S19. [Google Scholar] [CrossRef]
- Goldfarb, D. S.; Lieske, J. C.; Groothoff, J.; Schalk, G.; Russell, K.; Yu, S.; Vrhnjak, B. Nedosiran in primary hyperoxaluria subtype 3: Results from a phase I, single-dose study (PHYOX4). Urolithiasis 2023, 51(1), 80. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Jiang, X.; Gui, S. RNA interference-based nanosystems for inflammatory bowel disease therapy. International Journal of Nanomedicine 2016, 11, 5287–5310. [Google Scholar] [CrossRef]
- Hadjiolov, A. A.; Venkov, P. V.; Tsanev, R. G. Ribonucleic acids fractionation by density-gradient centrifugation and by agar gel electrophoresis: A comparison. Analytical Biochemistry 1966, 17(2), 263–267. [Google Scholar] [CrossRef]
- Han, E. L.; Tang, S.; Kim, D.; Murray, A. M.; Swingle, K. L.; Hamilton, A. G.; Mrksich, K.; Padilla, M. S.; Palanki, R.; Li, J. J.; Mitchell, M. J. Peptide-Functionalized Lipid Nanoparticles for Targeted Systemic mRNA Delivery to the Brain. Nano Letters 2025, 25(2), 800–810. [Google Scholar] [CrossRef] [PubMed]
- Hanson, L. R.; Frey, W. H. Intranasal delivery bypasses the blood-brain barrier to target therapeutic agents to the central nervous system and treat neurodegenerative disease. BMC Neuroscience 2008, 9 Suppl 3, S5. [Google Scholar] [CrossRef]
- Hassett, K. J.; Rajlic, I. L.; Bahl, K.; White, R.; Cowens, K.; Jacquinet, E.; Burke, K. E. mRNA vaccine trafficking and resulting protein expression after intramuscular administration. Molecular Therapy Nucleic Acids 2024, 35(1). [Google Scholar] [CrossRef]
- Heine, A.; Juranek, S.; Brossart, P. Clinical and immunological effects of mRNA vaccines in malignant diseases. Molecular Cancer 2021, 20(1), 52. [Google Scholar] [CrossRef]
- Jimeno, S.; Prados-Carvajal, R.; Fernández-Ávila, M. J.; Silva, S.; Silvestris, D. A.; Endara-Coll, M.; Rodríguez-Real, G.; Domingo-Prim, J.; Mejías-Navarro, F.; Romero-Franco, A.; Jimeno-González, S.; Barroso, S.; Cesarini, V.; Aguilera, A.; Gallo, A.; Visa, N.; Huertas, P. ADAR-mediated RNA editing of DNA:RNA hybrids is required for DNA double strand break repair. Nature Communications 2021, 12(1), 5512. [Google Scholar] [CrossRef]
- Jones, C. H.; Androsavich, J. R.; So, N.; Jenkins, M. P.; MacCormack, D.; Prigodich, A.; Welch, V.; True, J. M.; Dolsten, M. Breaking the mold with RNA—a “RNAissance” of life science. Npj Genomic Medicine 2024, 9(1), 1–14. [Google Scholar] [CrossRef]
- Karikó, K.; Buckstein, M.; Ni, H.; Weissman, D. Suppression of RNA recognition by Toll-like receptors: The impact of nucleoside modification and the evolutionary origin of RNA. Immunity 2005, 23(2), 165–175. [Google Scholar] [CrossRef]
- Katzmann, J. L.; Packard, C. J.; Chapman, M. J.; Katzmann, I.; Laufs, U. Targeting RNA With Antisense Oligonucleotides and Small Interfering RNA in Dyslipidemias: JACC State-of-the-Art Review. Journal of the American College of Cardiology 2020, 76(5), 563–579. [Google Scholar] [CrossRef] [PubMed]
- Lamb, Y. N. BNT162b2 mRNA COVID-19 Vaccine: First Approval. Drugs 2021a, 81(4), 495–501. [Google Scholar] [CrossRef]
- Lamb, Y. N. Inclisiran: First Approval. Drugs 2021b, 81(3), 389–395. [Google Scholar] [CrossRef]
- Leclair, N. K.; Brugiolo, M.; Park, S.; Devoucoux, M.; Urbanski, L.; Angarola, B. L.; Yurieva, M.; Anczuków, O. Antisense oligonucleotide-mediated TRA2β poison exon inclusion induces the expression of a lncRNA with anti-tumor effects. Nature Communications 2025, 16(1), 1670. [Google Scholar] [CrossRef]
- Liao, Z.; Liu, T.; Yao, Z.; Hu, T.; Ji, X.; Yao, B. Harnessing stimuli-responsive biomaterials for advanced biomedical applications. Exploration 2024, 20230133. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, Y.; Zhou, S.; Dain, L.; Mei, L.; Zhu, G. Circular RNA: An emerging frontier in RNA therapeutic targets, RNA therapeutics, and mRNA vaccines. Journal of Controlled Release: Official Journal of the Controlled Release Society 2022, 348, 84–94. [Google Scholar] [CrossRef]
- Lu, Y.; Huang, W.; Li, M.; Zheng, A. Exosome-Based Carrier for RNA Delivery: Progress and Challenges. Pharmaceutics 2023, 15(2), Article 2. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Huang, Z.; Sun, F.; Guo, F.; Wang, Y.; Kao, S.; Yang, G.; Huang, J.; Li, J.; Zhao, S.; He, Y. The clinical effects of inclisiran, a first-in-class LDL-C lowering siRNA therapy, on the LDL-C levels in Chinese patients with hypercholesterolemia. Journal of Clinical Lipidology 2023, 17(3), 392–400. [Google Scholar] [CrossRef]
- Matarazzo, L.; Bettencourt, P. J. G. mRNA vaccines: A new opportunity for malaria, tuberculosis and HIV. Frontiers in Immunology 2023, 14, 1172691. [Google Scholar] [CrossRef]
- McLoughlin, H. S.; Moore, L. R.; Chopra, R.; Komlo, R.; McKenzie, M.; Blumenstein, K. G.; Zhao, H.; Kordasiewicz, H. B.; Shakkottai, V. G.; Paulson, H. L. Oligonucleotide therapy mitigates disease in spinocerebellar ataxia type 3 mice. Annals of Neurology 2018, 84(1), 64–77. [Google Scholar] [CrossRef] [PubMed]
- McNamara, J. O.; Andrechek, E. R.; Wang, Y.; Viles, K. D.; Rempel, R. E.; Gilboa, E.; Sullenger, B. A.; Giangrande, P. H. Cell type-specific delivery of siRNAs with aptamer-siRNA chimeras. Nature Biotechnology 2006, 24(8), 1005–1015. [Google Scholar] [CrossRef] [PubMed]
- Milewska, S.; Sadowska, A.; Stefaniuk, N.; Misztalewska-Turkowicz, I.; Wilczewska, A. Z.; Car, H.; Niemirowicz-Laskowska, K. Tumor-Homing Peptides as Crucial Component of Magnetic-Based Delivery Systems: Recent Developments and Pharmacoeconomical Perspective. International Journal of Molecular Sciences 2024, 25(11), 6219. [Google Scholar] [CrossRef]
- Miller, T. M.; Cudkowicz, M. E.; Genge, A.; Shaw, P. J.; Sobue, G.; Bucelli, R. C.; Chiò, A.; Van Damme, P.; Ludolph, A. C.; Glass, J. D.; Andrews, J. A.; Babu, S.; Benatar, M.; McDermott, C. J.; Cochrane, T.; Chary, S.; Chew, S.; Zhu, H.; Wu, F. VALOR and OLE Working Group. (2022). Trial of Antisense Oligonucleotide Tofersen for SOD1 ALS. The New England Journal of Medicine 387(12), 1099–1110. [CrossRef]
- Neves, K. B.; Rios, F. J.; Sevilla-Montero, J.; Montezano, A. C.; Touyz, R. M. Exosomes and the cardiovascular system: Role in cardiovascular health and disease. The Journal of Physiology 2023, 601(22), 4923–4936. [Google Scholar] [CrossRef]
- Ng, E. W. M.; Shima, D. T.; Calias, P.; Cunningham, E. T.; Guyer, D. R.; Adamis, A. P. Pegaptanib, a targeted anti-VEGF aptamer for ocular vascular disease. Nature Reviews. Drug Discovery 2006, 5(2), 123–132. [Google Scholar] [CrossRef]
- Niazi, S. K. RNA Therapeutics: A Healthcare Paradigm Shift. Biomedicines 2023, 11(5), 1275. [Google Scholar] [CrossRef]
- Nissen, S. E.; Linnebjerg, H.; Shen, X.; Wolski, K.; Ma, X.; Lim, S.; Michael, L. F.; Ruotolo, G.; Gribble, G.; Navar, A. M.; Nicholls, S. J. Lepodisiran, an Extended-Duration Short Interfering RNA Targeting Lipoprotein(a). JAMA 2023, 330(21), 2075–2083. [Google Scholar] [CrossRef] [PubMed]
- Nissen, S. E.; Wang, Q.; Nicholls, S. J.; Navar, A. M.; Ray, K. K.; Schwartz, G. G.; Szarek, M.; Stroes, E. S. G.; Troquay, R.; Dorresteijn, J. A. N.; Fok, H.; Rider, D. A.; Romano, S.; Wolski, K.; Rambaran, C. Zerlasiran—A Small-Interfering RNA Targeting Lipoprotein(a): A Phase 2 Randomized Clinical Trial. JAMA 2024, 332(23), 1992–2002. [Google Scholar] [CrossRef]
- Novartis. Novartis [WWW Document]. 2022. Available online: https://www.novartis.com/home (accessed on 7 April 2025).
- Ouranidis, A.; Davidopoulou, C.; Tashi, R.-K.; Kachrimanis, K. Pharma 4.0 Continuous mRNA Drug Products Manufacturing. Pharmaceutics 2021, 13(9), 1371. [Google Scholar] [CrossRef]
- Paunovska, K.; Loughrey, D.; Dahlman, J. E. Drug delivery systems for RNA therapeutics. Nature Reviews Genetics 2022, 23(5), 265–280. [Google Scholar] [CrossRef] [PubMed]
- PhD, J. D. G. Novo Nordisk and Korro Bio Take RNA Editing to Cardiometabolic Diseases [WWW Document]. Inside Precision Medicine. 20 September 2024. Available online: https://www.insideprecisionmedicine.com/topics/precision-medicine/novo-nordisk-and-korro-bio-take-rna-editing-to-cardiometabolic-diseases/ (accessed on 4 April 2025).
- Philippidis, A. Seven Biopharma Trends to Watch in 2025. GEN Edge 2025, 7(1), 1–9. [Google Scholar] [CrossRef]
- Pirhaghi, M.; Mamashli, F.; Moosavi-Movahedi, F.; Arghavani, P.; Amiri, A.; Davaeil, B.; Mohammad-Zaheri, M.; Mousavi-Jarrahi, Z.; Sharma, D.; Langel, Ü.; Otzen, D. E.; Saboury, A. A. Cell-Penetrating Peptides: Promising Therapeutics and Drug-Delivery Systems for Neurodegenerative Diseases. Molecular Pharmaceutics 2024, 21(5), 2097–2117. [Google Scholar] [CrossRef]
- Planté-Bordeneuve, V.; Perrain, V. Vutrisiran: A new drug in the treatment landscape of hereditary transthyretin amyloid polyneuropathy. Expert Opinion on Drug Discovery 2024, 19(4), 393–402. [Google Scholar] [CrossRef]
- Presnyak, V.; Alhusaini, N.; Chen, Y.-H.; Martin, S.; Morris, N.; Kline, N.; Olson, S.; Weinberg, D.; Baker, K. E.; Graveley, B. R.; Coller, J. Codon optimality is a major determinant of mRNA stability. Cell 2015, 160(6), 1111–1124. [Google Scholar] [CrossRef] [PubMed]
- Rai, M. F.; Pan, H.; Yan, H.; Sandell, L. J.; Pham, C. T. N.; Wickline, S. A. Applications of RNA interference in the treatment of arthritis. Translational Research: The Journal of Laboratory and Clinical Medicine 2019, 214, 1–16. [Google Scholar] [CrossRef]
- Ratni, H.; Ebeling, M.; Baird, J.; Bendels, S.; Bylund, J.; Chen, K. S.; Denk, N.; Feng, Z.; Green, L.; Guerard, M.; Jablonski, P.; Jacobsen, B.; Khwaja, O.; Kletzl, H.; Ko, C.-P.; Kustermann, S.; Marquet, A.; Metzger, F.; Mueller, B.; Mueller, L. Discovery of Risdiplam, a Selective Survival of Motor Neuron-2(SMN2) Gene Splicing Modifier for the Treatment of Spinal Muscular Atrophy(SMA). Journal of Medicinal Chemistry 2018, 61(15), 6501–6517. [Google Scholar] [CrossRef]
- Ravichandiran, V.; Kesharwani, A.; Anupriya; Bhaskaran, M.; Parihar, V. K.; Bakhshi, S.; Velayutham, R.; Kumarasamy, M. Overcoming biological barriers: Precision engineered extracellular vesicles for personalized neuromedicine. Precision Medicine and Engineering 2024, 1(2), 100010. [Google Scholar] [CrossRef]
- Reshetnikov, V.; Terenin, I.; Shepelkova, G.; Yeremeev, V.; Kolmykov, S.; Nagornykh, M.; Kolosova, E.; Sokolova, T.; Zaborova, O.; Kukushkin, I.; Kazakova, A.; Kunyk, D.; Kirshina, A.; Vasileva, O.; Seregina, K.; Pateev, I.; Kolpakov, F.; Ivanov, R. Untranslated Region Sequences and the Efficacy of mRNA Vaccines against Tuberculosis. International Journal of Molecular Sciences 2024, 25(2), 888. [Google Scholar] [CrossRef]
- Rhim, J.; Baek, W.; Seo, Y.; Kim, J. H. From Molecular Mechanisms to Therapeutics: Understanding MicroRNA-21 in Cancer. Cells 2022, 11(18), 2791. [Google Scholar] [CrossRef]
- Rigo, F.; Chun, S. J.; Norris, D. A.; Hung, G.; Lee, S.; Matson, J.; Fey, R. A.; Gaus, H.; Hua, Y.; Grundy, J. S.; Krainer, A. R.; Henry, S. P.; Bennett, C. F. Pharmacology of a central nervous system delivered 2’-O-methoxyethyl-modified survival of motor neuron splicing oligonucleotide in mice and nonhuman primates. The Journal of Pharmacology and Experimental Therapeutics 2014, 350(1), 46–55. [Google Scholar] [CrossRef] [PubMed]
- Roberts, T. C.; Langer, R.; Wood, M. J. A. Advances in oligonucleotide drug delivery. Nature Reviews Drug Discovery 2020, 19(10), 673–694. [Google Scholar] [CrossRef] [PubMed]
- Rowe, S.; Zuckerman, J.; Dorgan, D.; Lascano, J.; McCoy, K.; Jain, M.; Schechter, M.; Lommatzsch, S.; Indihar, V.; Lechtzin, N.; Mcbennett, K.; Callison, C.; Brown, C.; Liou, T.; Macdonald, K.; Nasr, S.; Bodie, S.; Vaughn, M.; Meltzer, E.; Barbier, A. Inhaled mRNA therapy for treatment of cystic fibrosis: Interim results of a randomized, double-blind, placebo-controlled phase 1/2 clinical study. Journal of Cystic Fibrosis: Official Journal of the European Cystic Fibrosis Society 2023, 22. [Google Scholar] [CrossRef]
- Rupaimoole, R.; Slack, F. J. MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases. Nature Reviews Drug Discovery 2017, 16(3), 203–222. [Google Scholar] [CrossRef]
- Silva-Pilipich, N.; Beloki, U.; Salaberry, L.; Smerdou, C. Self-Amplifying RNA: A Second Revolution of mRNA Vaccines against COVID-19. Vaccines 2024, 12(3), 318. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Mehra, A.; Arora, S.; Gugulothu, D.; Vora, L. K.; Prasad, R.; Khatri, D. K. Exosome-mediated delivery and regulation in neurological disease progression. International Journal of Biological Macromolecules 2024, 264, 130728. [Google Scholar] [CrossRef]
- Sousa, C.; Videira, M. Dual Approaches in Oncology: The Promise of siRNA and Chemotherapy Combinations in Cancer Therapies. Onco 2025, 5(1), Article 1. [Google Scholar] [CrossRef]
- Southwell, A. L.; Kordasiewicz, H. B.; Langbehn, D.; Skotte, N. H.; Parsons, M. P.; Villanueva, E. B.; Caron, N. S.; Østergaard, M. E.; Anderson, L. M.; Xie, Y.; Cengio, L. D.; Findlay-Black, H.; Doty, C. N.; Fitsimmons, B.; Swayze, E. E.; Seth, P. P.; Raymond, L. A.; Frank Bennett, C.; Hayden, M. R. Huntingtin suppression restores cognitive function in a mouse model of Huntington’s disease. Science Translational Medicine 2018, 10(461), eaar3959. [Google Scholar] [CrossRef]
- Sparmann, A.; Vogel, J. RNA-based medicine: From molecular mechanisms to therapy. The EMBO Journal 2023, 42(21), e114760. [Google Scholar] [CrossRef]
- Sun, X.; Setrerrahmane, S.; Li, C.; Hu, J.; Xu, H. Nucleic acid drugs: Recent progress and future perspectives. Signal Transduction and Targeted Therapy 2024, 9(1), 1–31. [Google Scholar] [CrossRef]
- Sun, Y.; Cao, Y.; Song, Y.; Li, J.; Hou, Y.; Huang, W.; Xie, G.; Yang, W.; Zhang, R. Improved RNA base editing with guide RNAs mimicking highly edited endogenous ADAR substrates. Nature Biotechnology 2025, 1–13. [Google Scholar] [CrossRef]
- Svitkin, Y. V.; Cheng, Y. M.; Chakraborty, T.; Presnyak, V.; John, M.; Sonenberg, N. N1-methyl-pseudouridine in mRNA enhances translation through eIF2α-dependent and independent mechanisms by increasing ribosome density. Nucleic Acids Research 2017, 45(10), 6023–6036. [Google Scholar] [CrossRef] [PubMed]
- Syed, Y. Y. Nedosiran: First Approval. Drugs 2023, 83(18), 1729–1733. [Google Scholar] [CrossRef] [PubMed]
- Tejedor, S.; Wågberg, M.; Correia, C.; Åvall, K.; Hölttä, M.; Hultin, L.; Lerche, M.; Davies, N.; Bergenhem, N.; Snijder, A.; Marlow, T.; Dönnes, P.; Fritsche-Danielson, R.; Synnergren, J.; Jennbacken, K.; Hansson, K. The Combination of Vascular Endothelial Growth Factor A (VEGF-A) and Fibroblast Growth Factor 1 (FGF1) Modified mRNA Improves Wound Healing in Diabetic Mice: An Ex Vivo and In Vivo Investigation. Cells 2024, 13(5), Article 5. [Google Scholar] [CrossRef] [PubMed]
- Tenforde, M. W.; Olson, S. M.; Self, W. H.; Talbot, H. K.; Lindsell, C. J.; Steingrub, J. S.; Shapiro, N. I.; Ginde, A. A.; Douin, D. J.; Prekker, M. E.; Brown, S. M.; Peltan, I. D.; Gong, M. N.; Mohamed, A.; Khan, A.; Exline, M. C.; Files, D. C.; Gibbs, K. W.; Stubblefield, W. B.; HAIVEN Investigators. Effectiveness of Pfizer-BioNTech and Moderna Vaccines Against COVID-19 Among Hospitalized Adults Aged ≥65 Years—United States, January-March 2021. MMWR. Morbidity and Mortality Weekly Report 2021, 70(18), 674–679. [Google Scholar] [CrossRef]
- Trang, P.; Wiggins, J. F.; Daige, C. L.; Cho, C.; Omotola, M.; Brown, D.; Weidhaas, J. B.; Bader, A. G.; Slack, F. J. Systemic delivery of tumor suppressor microRNA mimics using a neutral lipid emulsion inhibits lung tumors in mice. Molecular Therapy: The Journal of the American Society of Gene Therapy 2011, 19(6), 1116–1122. [Google Scholar] [CrossRef]
- Ulbrich, K.; Hekmatara, T.; Herbert, E.; Kreuter, J. Transferrin-and transferrin-receptor-antibody-modified nanoparticles enable drug delivery across the blood–brain barrier (BBB). European Journal of Pharmaceutics and Biopharmaceutics 2009, 71(2), 251–256. [Google Scholar] [CrossRef]
- Vallina, N. S.; McRae, E. K. S.; Geary, C.; Andersen, E. S. An RNA origami robot that traps and releases a fluorescent aptamer. Science Advances 2024, 10(12), eadk1250. [Google Scholar] [CrossRef] [PubMed]
- van Rooij, E.; Kauppinen, S. Development of microRNA therapeutics is coming of age. EMBO Molecular Medicine 2014, 6(7), 851–864. [Google Scholar] [CrossRef]
- Vavilis, T.; Stamoula, E.; Ainatzoglou, A.; Sachinidis, A.; Lamprinou, M.; Dardalas, I.; Vizirianakis, I. S. mRNA in the Context of Protein Replacement Therapy. Pharmaceutics 2023, 15(1), 166. [Google Scholar] [CrossRef]
- Vickers, T. A.; Crooke, S. T. Antisense oligonucleotides capable of promoting specific target mRNA reduction via competing RNase H1-dependent and independent mechanisms. PloS One 2014, 9(10), e108625. [Google Scholar] [CrossRef] [PubMed]
- Weber, J. S.; Carlino, M. S.; Khattak, A.; Meniawy, T.; Ansstas, G.; Taylor, M. H.; Kim, K. B.; McKean, M.; Long, G. V.; Sullivan, R. J.; Faries, M.; Tran, T. T.; Cowey, C. L.; Pecora, A.; Shaheen, M.; Segar, J.; Medina, T.; Atkinson, V.; Gibney, G. T.; Zaks, T. Individualised neoantigen therapy mRNA-4157 (V940) plus pembrolizumab versus pembrolizumab monotherapy in resected melanoma (KEYNOTE-942): A randomised, phase 2b study. The Lancet 2024, 403(10427), 632–644. [Google Scholar] [CrossRef]
- Wei, P.-S.; Thota, N.; John, G.; Chang, E.; Lee, S.; Wang, Y.; Ma, Z.; Tsai, Y.-H.; Mei, K.-C. Enhancing RNA-lipid nanoparticle delivery: Organ-and cell-specificity and barcoding strategies. Journal of Controlled Release 2024, 375, 366–388. [Google Scholar] [CrossRef]
- Whitley, J.; Zwolinski, C.; Denis, C.; Maughan, M.; Hayles, L.; Clarke, D.; Snare, M.; Liao, H.; Chiou, S.; Marmura, T.; Zoeller, H.; Hudson, B.; Peart, J.; Johnson, M.; Karlsson, A.; Wang, Y.; Nagle, C.; Harris, C.; Tonkin, D.; Johnson, M. R. Development of mRNA manufacturing for vaccines and therapeutics: mRNA platform requirements and development of a scalable production process to support early phase clinical trials. Translational Research 2022, 242, 38–55. [Google Scholar] [CrossRef]
- Win, M. N.; Smolke, C. D. A modular and extensible RNA-based gene-regulatory platform for engineering cellular function. Proceedings of the National Academy of Sciences of the United States of America 2007, 104(36), 14283–14288. [Google Scholar] [CrossRef]
- Xu, Y.; Gong, F.; Golubovic, A.; Strilchuk, A.; Chen, J.; Zhou, M.; Dong, S.; Seto, B.; Li, B. Rational design and modular synthesis of biodegradable ionizable lipids via the Passerini reaction for mRNA delivery. Proceedings of the National Academy of Sciences 2025, 122(5), e2409572122. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Okada, S.; Sakurai, M. Adenosine-to-inosine RNA editing in neurological development and disease. RNA Biology 2021, 18(7), 999–1013. [Google Scholar] [CrossRef]
- Yip, T.; Qi, X.; Yan, H.; Chang, Y. RNA Origami Functions as a Self-Adjuvanted Nanovaccine Platform for Cancer Immunotherapy. ACS Nano 2024, 18(5), 4056–4067. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Rossi, J. Aptamers as targeted therapeutics: Current potential and challenges. Nature Reviews. Drug Discovery 2017, 16(3), 181–202. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, S.-J. Advances in machine-learning approaches to RNA-targeted drug design. Artificial Intelligence Chemistry 2024, 2(1), 100053. [Google Scholar] [CrossRef]
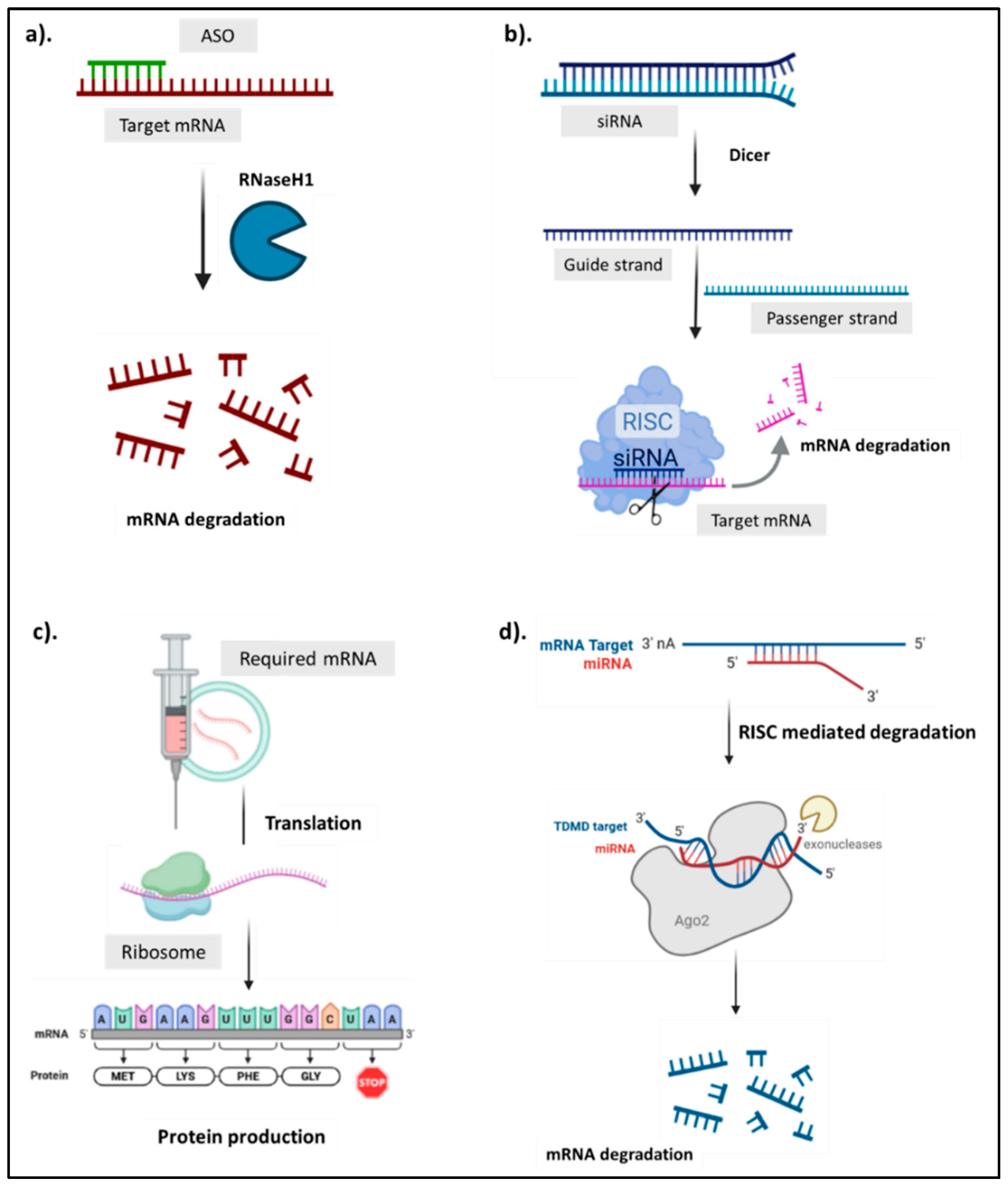
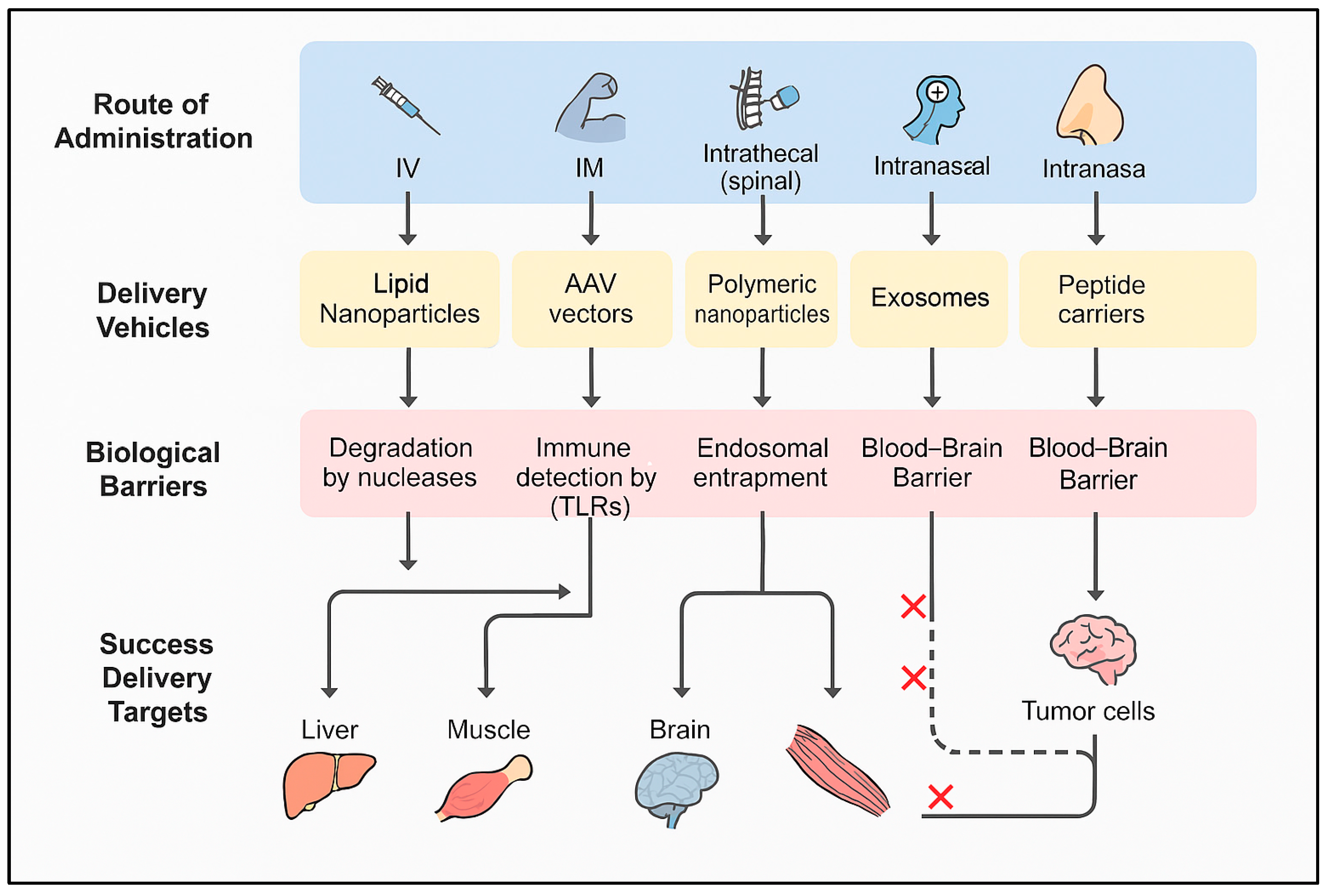
| Type | Size (nt) | Target | Mechanism of Action | Stability | Approved Drugs | References |
|---|---|---|---|---|---|---|
| mRNA | >1,000 | Cytoplasm | Protein translation | Low to moderate | Comirnaty (Pfizer-BioNTech, Spikevax (Moderna) | (Lamb, 2021a) (Baden et al., 2021 |
| siRNA | ~21–23 | mRNA | mRNA cleavage and degradation | Moderate | Onpattro (patisiran), Leqvio | (Adams et al., 2018) (Lamb, 2021b |
| ASO | ~15–25 | pre-mRNA/mRNA | Splicing modulation or translation inhibition | High | Spinraza (nusinersen), Tegsedi | (Finkel et al., 2017) (Benson et al., 2018 |
| miRNA mimic | ~22 | mRNA | Translational repression or silencing | Low to moderate | Experimental (no major approvals yet) | (Rupaimoole & Slack, 2017 |
| CRISPR/Cas9 | sgRNA + Cas9 | DNA (genome) | Site-specific gene editing (DSB + repair) | Variable | In clinical trials | (Frangoul et al., 2021 |
| Delivery System | Mechanism/Target | Advantages | Limitations | Example(s) |
|---|---|---|---|---|
| Lipid Nanoparticles (LNPs) | Endosomal uptake, liver targeting | High efficiency, used in vaccines | Limited tissue targeting, immune activation | Comirnaty, Onpattro |
| GalNAc Conjugates | ASGPR-mediated hepatocyte targeting | Precise liver delivery, low immunogenicity | Limited to liver | Givosiran, Inclisiran |
| Polymeric Nanoparticles | Endosomal escape | Tunable release, biocompatibility | Lower efficiency, potential toxicity | Experimental |
| Exosomes | Natural vesicle-mediated delivery | Low immunogenicity, endogenous cargo | Scalability issues, heterogeneity | Preclinical studies |
| Viral Vectors | Direct genome delivery | Efficient expression, long-term effects | Insertional mutagenesis, immune response | Used in CRISPR trials |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




