Submitted:
08 April 2025
Posted:
08 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Plasmids
2.2. Production of Hybridomas
2.3. Flow Cytometric Analysis
2.4. Determination of Dissociation Constant (KD) by Flow Cytometry
2.5. Immunoblotting
2.6. Immunohistochemical Analysis
3. Results
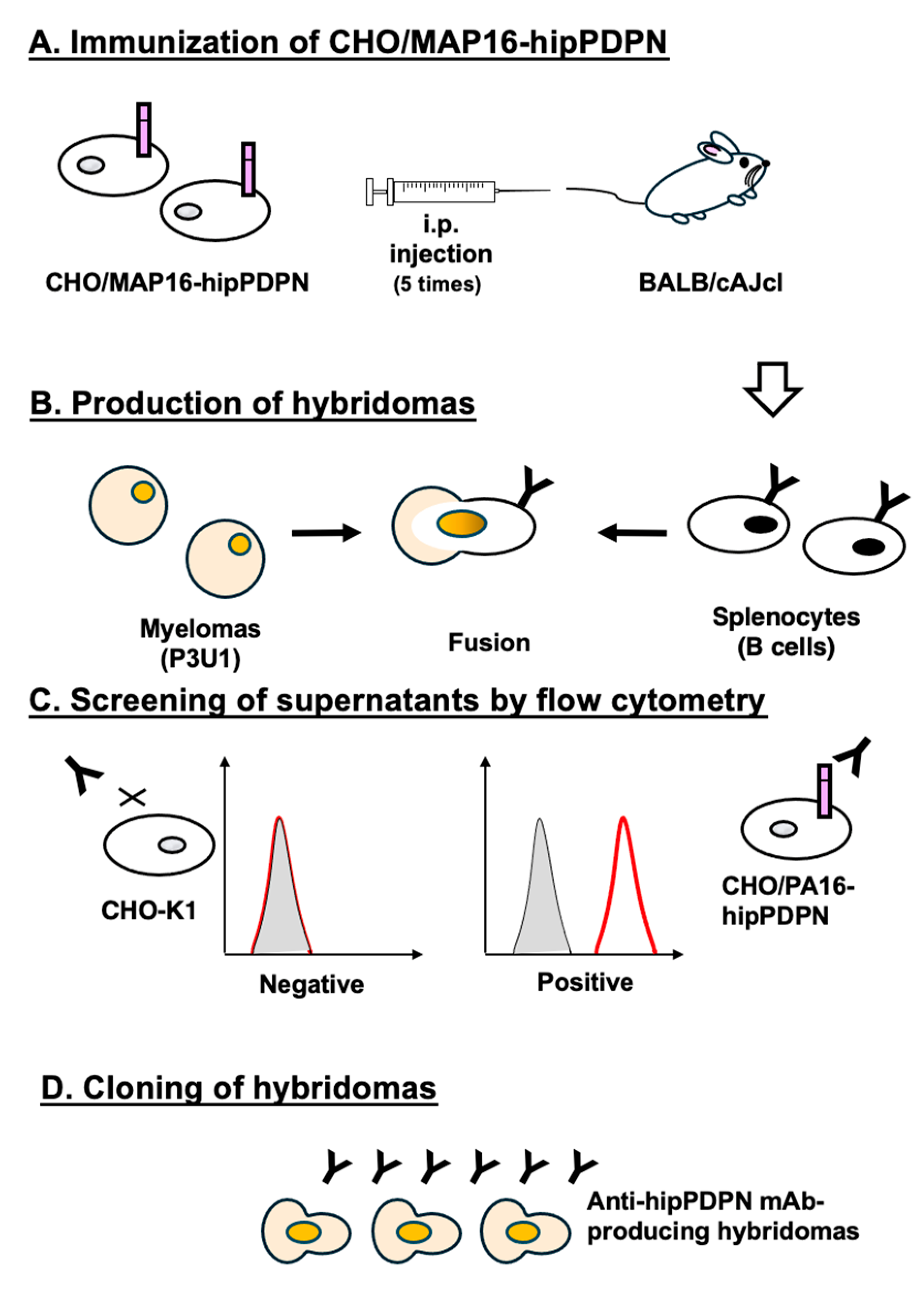
3.1. Development of Anti-hipPDPN mAbs Using the CBIS Method
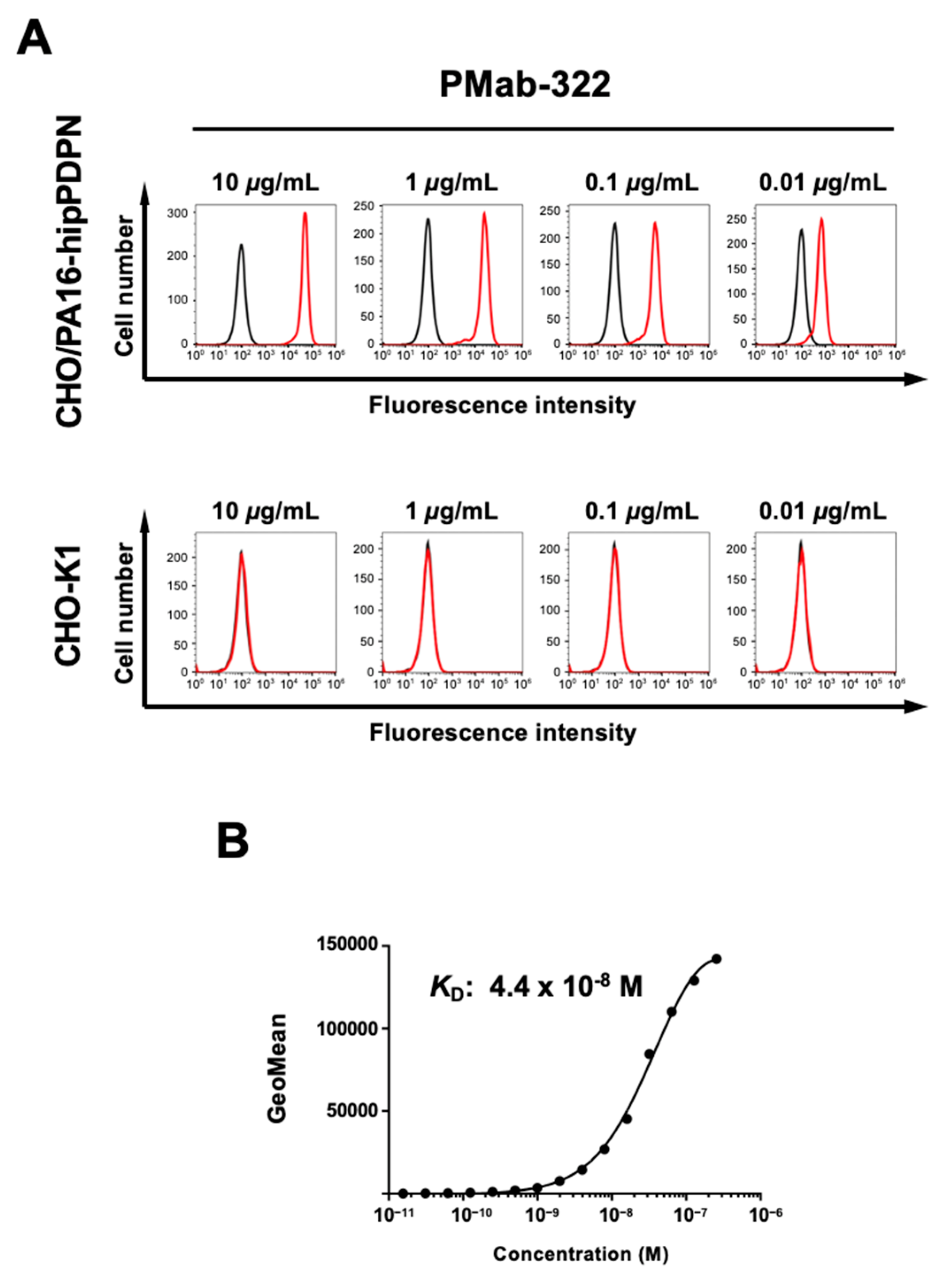
3.2. Flow Cytometry Using PMab-322
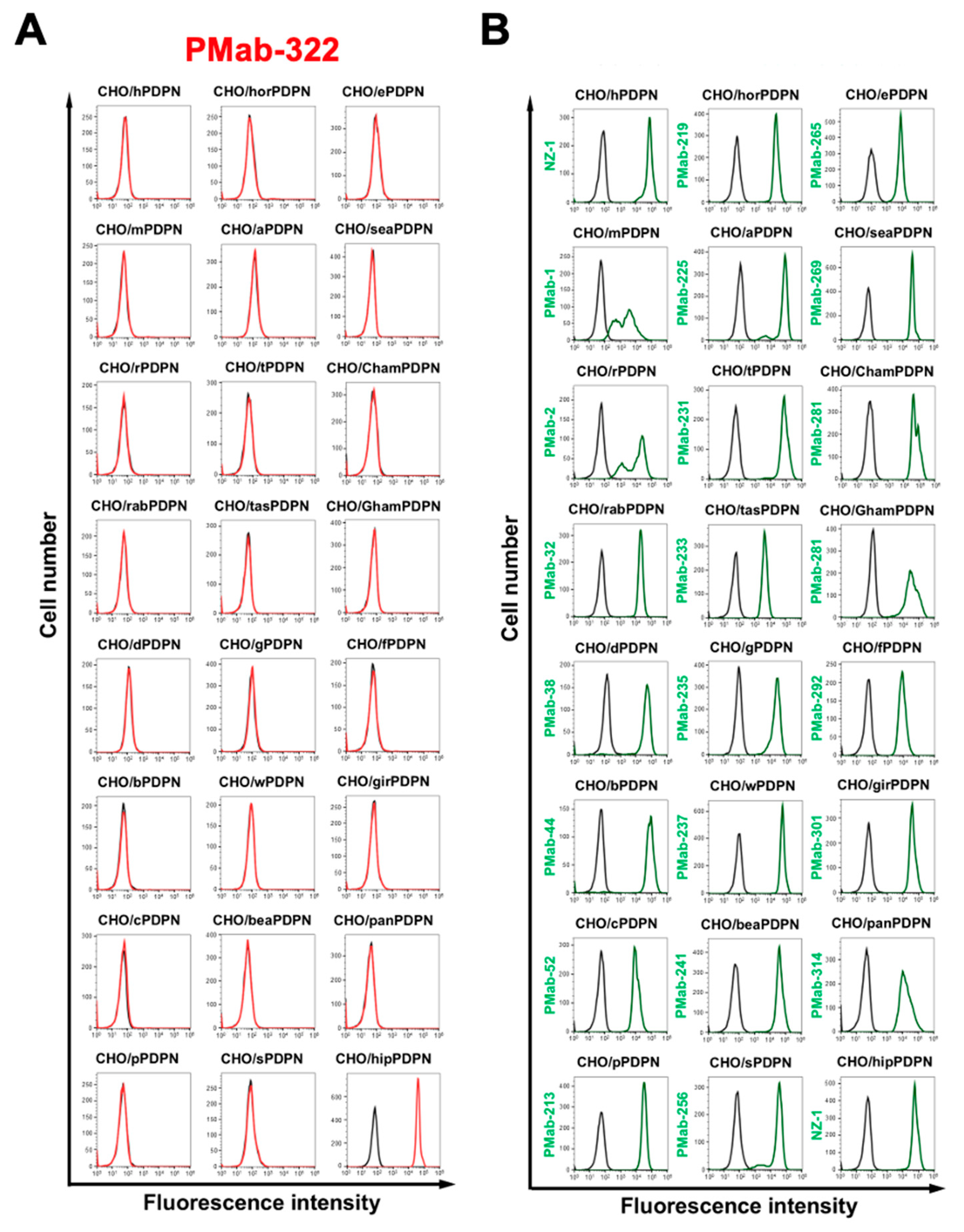
3.3. Specificity of PMab-322 Against 24 Species-PDPN Expressed CHO-K1
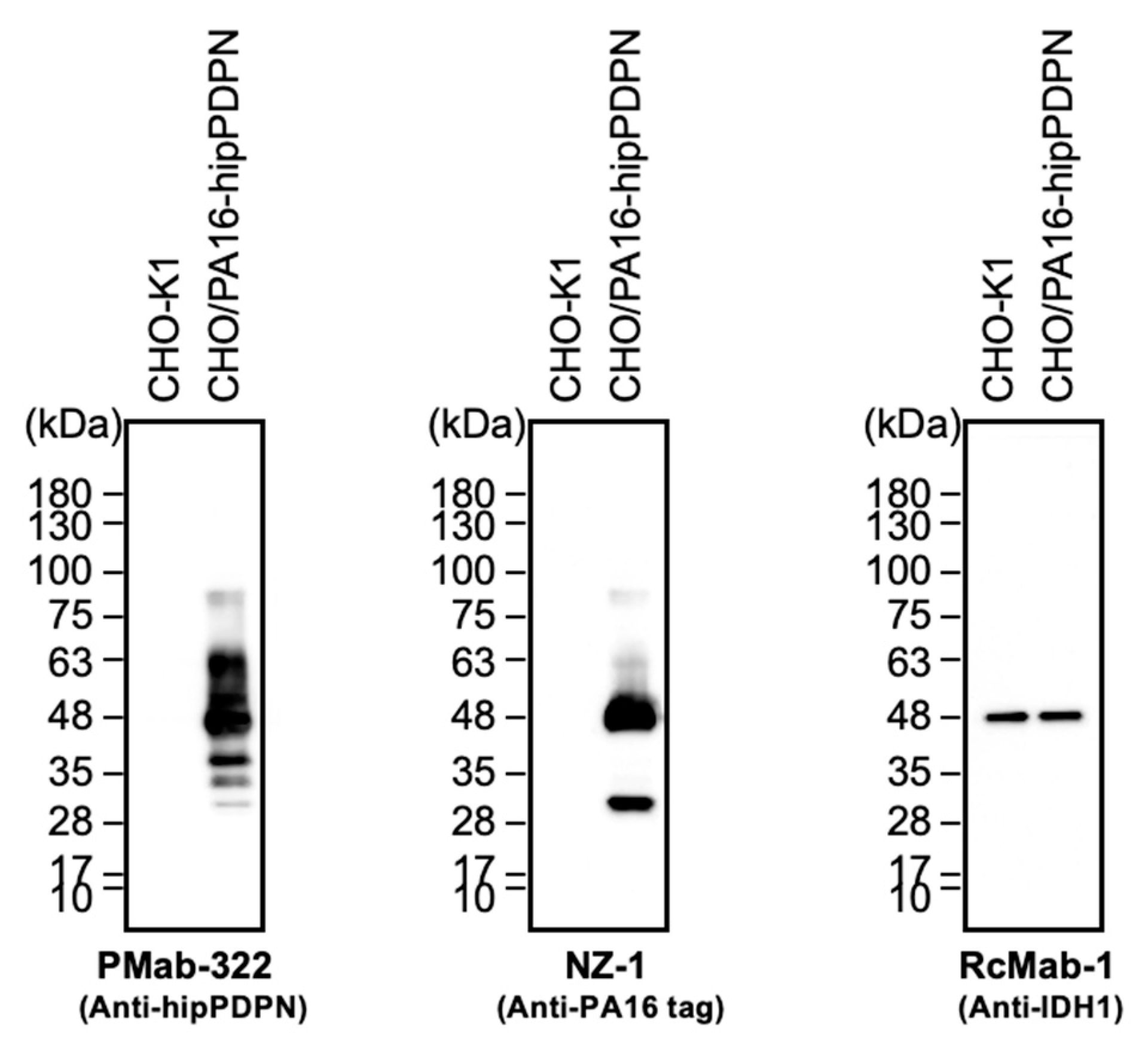
3.4. Immunoblotting Using PMab-322
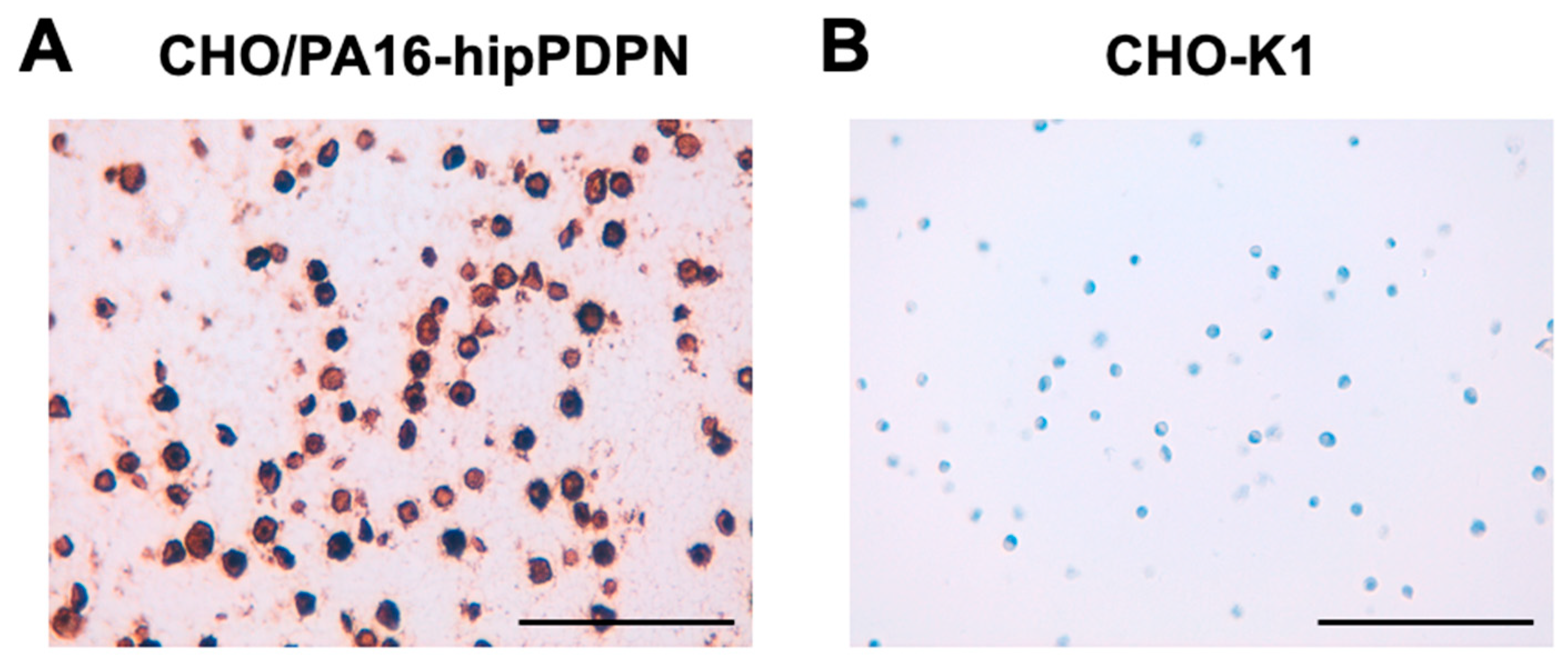
3.5. Immunohistochemistry Using PMab-322
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kato, Y.; Fujita, N.; Kunita, A.; et al. Molecular identification of Aggrus/T1alpha as a platelet aggregation-inducing factor expressed in colorectal tumors. J Biol Chem 2003, 278, 51599–51605. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Kaneko, M.K.; Kato, Y. Roles of Podoplanin in Malignant Progression of Tumor. Cells 2022;11(3).
- Hirakawa, S.; Hong, Y.K.; Harvey, N.; et al. Identification of vascular lineage-specific genes by transcriptional profiling of isolated blood vascular and lymphatic endothelial cells. Am J Pathol 2003, 162, 575–586. [Google Scholar] [CrossRef] [PubMed]
- Petrova, T.V.; Mäkinen, T.; Mäkelä, T.P.; et al. Lymphatic endothelial reprogramming of vascular endothelial cells by the Prox-1 homeobox transcription factor. Embo j 2002, 21, 4593–4599. [Google Scholar] [CrossRef]
- Breiteneder-Geleff, S.; Matsui, K.; Soleiman, A.; et al. Podoplanin, novel 43-kd membrane protein of glomerular epithelial cells, is down-regulated in puromycin nephrosis. Am J Pathol 1997, 151, 1141–1152. [Google Scholar]
- Asai, J. The Role of Podoplanin in Skin Diseases. Int J Mol Sci 2022;23(3).
- Dobbs, L.G.; Williams, M.C.; Gonzalez, R. Monoclonal antibodies specific to apical surfaces of rat alveolar type I cells bind to surfaces of cultured, but not freshly isolated, type II cells. Biochim Biophys Acta 1988, 970, 146–156. [Google Scholar] [CrossRef]
- Rishi, A.K.; Joyce-Brady, M.; Fisher, J.; et al. Cloning, characterization, and development expression of a rat lung alveolar type I cell gene in embryonic endodermal and neural derivatives. Dev Biol 1995, 167, 294–306. [Google Scholar] [CrossRef]
- Williams, M.C.; Cao, Y.; Hinds, A.; Rishi, A.K.; Wetterwald, A. T1 alpha protein is developmentally regulated and expressed by alveolar type I cells, choroid plexus, and ciliary epithelia of adult rats. Am J Respir Cell Mol Biol 1996, 14, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Schacht, V.; Ramirez, M.I.; Hong, Y.K.; et al. T1alpha/podoplanin deficiency disrupts normal lymphatic vasculature formation and causes lymphedema. Embo j 2003, 22, 3546–3556. [Google Scholar] [CrossRef]
- Ramirez, M.I.; Millien, G.; Hinds, A.; et al. T1alpha, a lung type I cell differentiation gene, is required for normal lung cell proliferation and alveolus formation at birth. Dev Biol 2003, 256, 61–72. [Google Scholar] [CrossRef]
- Honma, M.; Minami-Hori, M.; Takahashi, H.; Iizuka, H. Podoplanin expression in wound and hyperproliferative psoriatic epidermis: regulation by TGF-β and STAT-3 activating cytokines, IFN-γ, IL-6, and IL-22. J Dermatol Sci 2012, 65, 134–140. [Google Scholar] [CrossRef]
- Yoon, S.Y.; Dieterich, L.C.; Tacconi, C.; et al. An important role of podoplanin in hair follicle growth. PLoS One 2019, 14, e0219938. [Google Scholar] [CrossRef] [PubMed]
- Boisserie, J.R.; Fisher, R.E.; Lihoreau, F.; Weston, E.M. Evolving between land and water: key questions on the emergence and history of the Hippopotamidae (Hippopotamoidea, Cetancodonta, Cetartiodactyla). Biol Rev Camb Philos Soc 2011, 86, 601–625. [Google Scholar] [CrossRef]
- Gatesy, J.; Geisler, J.H.; Chang, J.; et al. A phylogenetic blueprint for a modern whale. Mol Phylogenet Evol 2013, 66, 479–506. [Google Scholar] [CrossRef]
- Shimamura, M.; Yasue, H.; Ohshima, K.; et al. Molecular evidence from retroposons that whales form a clade within even-toed ungulates. Nature 1997, 388, 666–670. [Google Scholar] [CrossRef] [PubMed]
- Gatesy, J. More DNA support for a Cetacea/Hippopotamidae clade: the blood-clotting protein gene gamma-fibrinogen. Mol Biol Evol 1997, 14, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Gatesy, J.; Hayashi, C.; Cronin, M.A.; Arctander, P. Evidence from milk casein genes that cetaceans are close relatives of hippopotamid artiodactyls. Mol Biol Evol 1996, 13, 954–963. [Google Scholar] [CrossRef]
- Okada, Y.; Suzuki, H.; Kaneko, M.K.; Kato, Y. Development of a Sensitive Anti-Mouse CD39 Monoclonal Antibody (C(39)Mab-1) for Flow Cytometry and Western Blot Analyses. Monoclon Antib Immunodiagn Immunother 2024, 43, 24–31. [Google Scholar] [CrossRef]
- Kudo, Y.; Suzuki, H.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Development of a Novel Anti-CD44 Variant 5 Monoclonal Antibody C(44)Mab-3 for Multiple Applications against Pancreatic Carcinomas. Antibodies (Basel) 2023;12(2).
- Kato, Y.; Kaneko, M.K. A cancer-specific monoclonal antibody recognizes the aberrantly glycosylated podoplanin. Sci Rep 2014, 4, 5924. [Google Scholar] [CrossRef]
- Heydemann, L.; Ciurkiewicz, M.; Störk, T.; et al. Respiratory long COVID in aged hamsters features impaired lung function post-exercise with bronchiolization and fibrosis. Nat Commun 2025, 16, 2080. [Google Scholar] [CrossRef]
- Kaneko, M.K.; Honma, R.; Ogasawara, S.; et al. PMab-38 Recognizes Canine Podoplanin of Squamous Cell Carcinomas. Monoclon Antib Immunodiagn Immunother 2016, 35, 263–266. [Google Scholar] [CrossRef]
- Kato, Y.; Ito, Y.; Ohishi, T.; et al. Antibody-Drug Conjugates Using Mouse-Canine Chimeric Anti-Dog Podoplanin Antibody Exerts Antitumor Activity in a Mouse Xenograft Model. Monoclon Antib Immunodiagn Immunother 2020, 39, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Fujii, Y.; Kaneko, M.; Neyazaki, M.; et al. PA tag: a versatile protein tagging system using a super high affinity antibody against a dodecapeptide derived from human podoplanin. Protein Expr Purif 2014, 95, 240–247. [Google Scholar] [CrossRef]
- Fujii, Y.; Kaneko, M.K.; Kato, Y. MAP Tag: A Novel Tagging System for Protein Purification and Detection. Monoclon Antib Immunodiagn Immunother 2016, 35, 293–299. [Google Scholar] [CrossRef]
- Kato, Y.; Kaneko, M.K.; Kuno, A.; et al. Inhibition of tumor cell-induced platelet aggregation using a novel anti-podoplanin antibody reacting with its platelet-aggregation-stimulating domain. Biochem Biophys Res Commun 2006, 349, 1301–1307. [Google Scholar] [CrossRef] [PubMed]
- Kaji, C.; Tsujimoto, Y.; Kato Kaneko, M.; Kato, Y.; Sawa, Y. Immunohistochemical Examination of Novel Rat Monoclonal Antibodies against Mouse and Human Podoplanin. Acta. Histochem. Cytochem. 2012, 45, 227–237. [Google Scholar] [CrossRef]
- Kato, Y.; Furusawa, Y.; Itai, S.; et al. Establishment of an Anticetacean Podoplanin Monoclonal Antibody PMab-237 for Immunohistochemical Analysis. Monoclon Antib Immunodiagn Immunother 2019, 38, 108–113. [Google Scholar] [CrossRef]
- Satofuka, H.; Suzuki, H.; Tanaka, T.; et al. Development of an anti-human EphA2 monoclonal antibody Ea2Mab-7 for multiple applications. Biochemistry and Biophysics Reports 2025, 42, 101998. [Google Scholar] [CrossRef] [PubMed]
- Ikota, H.; Nobusawa, S.; Arai, H.; et al. Evaluation of IDH1 status in diffusely infiltrating gliomas by immunohistochemistry using anti-mutant and wild type IDH1 antibodies. Brain Tumor Pathol 2015, 32, 237–244. [Google Scholar] [CrossRef]
- Springer, M.S.; Signore, A.V.; Paijmans, J.L.; et al. Interordinal gene capture, the phylogenetic position of Steller’s sea cow based on molecular and morphological data, and the macroevolutionary history of Sirenia. Mol Phylogenet Evol 2015, 91, 178–193. [Google Scholar] [CrossRef]
- O’Leary, M.A.; Gatesy, J. Impact of increased character sampling on the phylogeny of Cetartiodactyla (Mammalia): combined analysis including fossils. Cladistics 2008, 24, 397–442. [Google Scholar] [CrossRef]
- Springer, M.S.; Guerrero-Juarez, C.F.; Huelsmann, M.; et al. Genomic and anatomical comparisons of skin support independent adaptation to life in water by cetaceans and hippos. Curr Biol 2021, 31, 2124–2139e2123. [Google Scholar] [CrossRef] [PubMed]
- Otero-Sabio, C.; Centelleghe, C.; Corain, L.; et al. Microscopic anatomical, immunohistochemical, and morphometric characterization of the terminal airways of the lung in cetaceans. J Morphol 2021, 282, 291–308. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



