1. Introduction
Chemotherapy (CT) with cisplatin (cis-diamminedichloroplatinum II, CDDP) has been used to treat cancer patients since the early 1970s [
1] and emerged as a standard of care for a variety of tumor entities, including leukemia, lymphoma, breast, testicular, ovarian, head and neck cancer, cervical cancer and sarcomas [
2]. Subsequent combination with radiotherapy (RT) as neoadjuvant, primary, or adjuvant concurrent chemoradiotherapy (CRT) has drastically improved clinical outcomes through spatial cooperation, cytotoxic enhancement, biological cooperation, temporary modulation, and normal tissue protection [
3,
4]. Concurrent CRT with CDDP achieves additive or synergistic anti-neoplastic effects for many locally advanced (LA) tumors with good locoregional control and acceptable toxicity profiles [
2].
CDDP and ionizing radiation (IR) both have DNA-damaging effects. IR induces a large variety of genomic lesions such as sublethal base damage and single-strand breaks (SSBs) or fatal DNA double-strand breaks (DSBs). The main target of CDDP is also considered to be nuclear DNA through the induction of inter- and intrastrand crosslinks, although just about 1% of intracellular CDDP reacts with genomic DNA. CDDP-induced DNA adducts can excerpt a radiosensitizing effect by turning IR-induced sublethal DNA damage into irreparable lesions leading to cytogenetic damage, cell cycle arrest, and apoptosis [
5,
6,
7]. This response has also been attributed to CDDP-mediated inhibition of non-homologous end joining (NHEJ) as the predominant mechanism of DSB repair [
8,
9]. Besides, CDDP also has effects on mitochondria, reactive oxygen species, lysosomes, and calcium signaling, which contributes to the synergies with IR for cytotoxic effects [
10]. However, the precise molecular mechanisms of CDDP and their interactions with each other and IR remain largely unclear [
11]. While the combined effect in the tumor is desired, toxicities in normal tissue should be avoided as far as possible, which are generally higher with concomitant CRT than with the respective monotherapies [
12,
13]. This applies not only to normal solid tissue but also to the hematologic system of cancer patients, which is inevitably exposed to the systemic effects of CDDP administered intravenously and IR during external beam RT. Lymphocytes in particular, as the main players in the inflammatory anti-tumor response, are highly susceptible to adverse effects of genotoxic cancer therapies [
14,
15]. Grade III/IV lymphopenia is a common side effect of CRT and a poor prognostic factor for many solid tumor types [
16]. Also, RT exposure of tumor-draining lymph nodes impairs T cell priming and maturation of cytotoxic T cells [
17,
18]. Although these effects of genotoxic therapies on the hematologic system have been long known, they are gaining increasing attention in light of multimodal cancer therapies with immune checkpoint inhibitors (ICI) [
19,
20,
21]. At present, the failure of multimodal therapies with ICI is primarily attributed to the adverse effects of genotoxic therapies on the hematologic system, which is crucial to the patient's inherent anti-tumor immune response [
17,
22]. A key example is LA head and neck tumors, where the still generally poor survival rates in concurrent CRT could not be improved by immuno-oncological strategies with ICI [
23,
24,
25].
The radiation burden on the hematological system of cancer patients can be monitored
in vivo using biodosimetric markers that are primarily based on the detection of IR-induced DNA damage in peripheral blood lymphocytes (PBLs) [
26,
27,
28]. The most common assays are immunodetection of nuclear foci of DNA repair proteins γH2AX or 53BP1 as DSB surrogate markers, chromosomal aberrations, or micronuclei [
29]. The effects of a concomitant CT on these radiobiological endpoints, in particular the role of CDDP as a potential confounder and its impact on the sensitivity of PBL to IR, have not been thoroughly investigated to date. Scarcely available studies indicate an impairment of the IR-induced DSB response, stronger cell cycle arrest, and increased apoptosis in PBLs of cancer patients who received concomitant CRT with CDDP [
30,
31,
32].
To increase the understanding of the effects of a combination of genotoxic therapies on the hematological system, we examined the ex vivo effects of CDDP pretreatments and subsequent IR exposure of isolated PBLs from buffy coats of healthy donors on the induction and repair of DSBs, micronuclei formation, cell cycle, and apoptosis. The data generated provide detailed information on the confounding effect of cytostatic drugs on these endpoints used as predictive biomarkers and for biodosimetry during RT and the adverse effects of corresponding therapeutic combinations on the hematological system of tumor patients.
2. Materials and Methods
Isolation of peripheral blood mononuclear cells
Buffy coats as leukocyte-rich plasma of healthy human donors were kindly provided by the Institute for Transfusion Medicine - Transfusion Centre of the University Medical Center Mainz. Ethical approval was obtained from the Medical Association of Rhineland-Palatinate [no. 2023-17191 and 2024-17401_2], and all research was performed following relevant guidelines and regulations. All donors provided informed consent and research has been performed following the Declaration of Helsinki. 1-2 ml of buffy coat was diluted with 3-4 ml of X-VIVO
TM 15 media (Lonza Group Ltd., Basel, Switzerland), and peripheral blood mononuclear cells (PBMCs) were separated by Histopaque (Histopaque 1077, Sigma-Aldrich) density gradient centrifugation as described previously [
26,
33,
34]. Isolated PBMCs were washed once in 2mM ethylenediaminetetraacetic acid (EDTA, Merck, Germany) / 1x phosphate-buffered saline (PBS, Merck, Germany), resuspended in X-VIVO
TM 15 media, counted, and used for assays.
Irradiation
Cells were exposed to X-rays (140 kV) at room temperature using the D3150 X-ray Therapy System (Gulmay Medical Ltd., Byfleet, UK) at a dose rate of 3.6 Gy/min or were mock-treated (0 Gy), i.e., kept for the same time in the radiation device control room.
γH2AX and 53BP1 foci quantification
1-2 x 10
6 PBMCs were diluted in 5 ml of X-VIVO
TM 15 media. CDDP (Accord Healthcare GmbH, München, Germany) was added at concentrations of 0, 1, 5, 10, and 20 µg/ml, and cells were incubated in a humidified incubator at 5% CO
2 and 37°C. After 1h or 24h, the medium was replaced with CDDP-free X-VIVO
TM 15 medium, and the cells were irradiated with 0.5 Gy or 2 Gy X-rays. The samples irradiated with 0.5 Gy or 2 Gy were processed after 0.5h or 24h, respectively. A sham-irradiated sample (0 Gy) was included in each case. Samples with irradiation only were treated accordingly without CDDP. Fixation of PBLs, γH2AX, and 53BP1 immunostaining, fluorescence microscopy, image capturing, and scoring of foci were performed as described previously [
26,
33,
34,
35]. Three independent repeat experiments were carried out with individual samples for all experimental conditions. For each data point, at least 100 sham-irradiated cells, 50 cells at 0.5h post-irradiation, and 100 cells at 24h post-irradiation were analyzed. To determine the number of radiation-induced foci (RIF), the corresponding basal values were subtracted from the total yield.
Cytokinesis block micronucleus (CBMN) Assay
3 x 106 cells were diluted in 5 ml of X-VIVOTM 15 media containing 20% heat-inactivated fetal calf serum (FCS, Biochrom, Berlin, Germany) and 1% phytohemagglutinin (PHA, Thermo Fisher Scientific, Karlsruhe, Germany) to simulate proliferation. CDDP was added at concentrations of 0, 1, 2.5, 5, 7.5, 10, and 20 µg/ml, and cells were incubated in a humidified incubator at 5% CO2 and 37° C. After 4h or 24h, the medium was replaced with CDDP-free X-VIVOTM 15 media containing 20% heat-inactivated FCS, 1% PHA, and 6 µg/mL cytochalasin B (Biomol, Hamburg, Germany) to abrogate cytokinesis and accumulate binucleated cells. Cells were irradiated with 2 Gy X-rays and incubated in a humidified incubator at 5% CO2 and 37° C up to 72h after the start of cultivation. Samples with irradiation only were treated accordingly without CDDP. Cells were spun down, treated for 10 minutes with 0.075 M potassium chloride solution (Merck, Darmstadt, Germany) for hypotonic shock, and fixed twice with Carnoy’s solution. Cells were dropped on slides and mounted with a HOECHST33258 antifade mountant. Image acquisition by fluorescence microscopy, and automated scoring of micronuclei (MN) in binucleated (BN) cells was performed using the Metafer platform and the MNScore software (Metasystems, Altlussheim, Germany). The binucleation index was calculated as the ratio of the number of binucleated cells/ total number of cells (mononucleated + binucleated cells). On average, 38,747 ± 10,260 total cells and 3,854 ± 1,426 BN cells were analyzed per sample to evaluate MN and the binucleation index. To determine the number of radiation-induced MN, the corresponding basal values were subtracted. Three independent repeat experiments were carried out with single samples for all experimental conditions.
Apoptosis (AnnexinV-FITC/PI Staining)
For apoptosis measurement, 1 x 106 PBMCs were diluted in 5 ml of X-VIVOTM 15 medium. CDDP was added at concentrations of 0, 1, 5, 10, and 20 µg/ml, and cells were incubated in a humidified incubator at 5% CO2 and 37° C. After 24h, the medium was replaced with CDDP-free X-VIVOTM 15 medium, and the cells were sham-irradiated (0 Gy) or exposed to X-rays. The cells were incubated in a humidified incubator at 5% CO2 and 37° C for another 24h. Samples with irradiation only were treated accordingly without CDDP. The fractions of viable, early apoptotic, late apoptotic, and necrotic cells were assessed using an AnnexinV-FITC/PI detection kit (Miltenyi Biotec, Bergisch Gladbach, Germany) according to the manufacturer’s instructions. At least 10,000 cells per sample were measured in a BD FACSCanto II flow cytometer (BD Biosciences, Billerica, USA). The data were analyzed using the Flowing Software (flowingsoftware.btk.fi). At least three independent experiments with biological duplicates or triplicates were performed for all experimental conditions.
Cell cycle measurement
To investigate the effects of CDDP on the cell cycle of PBLs, 1 x 106 cells were diluted in 5 ml of X-VIVOTM 15 media containing 20% heat-inactivated FCS and 1% PHA to stimulate proliferation. At the same time, CDDP treatment was started with concentrations of 0, 1, 5, 10, and 20 µg/ml. After 24h of incubation in a humidified incubator at 5% CO2 and 37° C, the medium was replaced with CDDP-free X-VIVOTM 15 media containing 20% heat-inactivated FCS and 1% PHA. In a separate experimental setting, an additional 100 ng/ml of nocodazole (Thermo Scientific Chemicals, Karlsruhe, Germany) was added to accumulate mitotic cells. After a further 48h of incubation, the cells were washed once in 2mM EDTA/ 1x PBS, fixed in 70% Ethanol at -20° C, treated with RNaseA (Merck, Darmstadt, Germany), and stained with propidium iodide (PI, Merck, Darmstadt, Germany). At least 10,000 cells per sample were analyzed in a BD FACSCanto II flow cytometer (BD Biosciences, Billerica, USA). Quantification of the fractions of cells in G1, S, and G2/M was performed using the Flowing Software (flowingsoftware.btk.fi). At least three independent experiments with biological triplicates were performed for all experimental conditions.
Data and statistical analysis
Summarized average yields and data are provided as the mean ± standard deviation unless stated otherwise. Data handling, plotting, and statistics were done using SigmaPlot Version 14 (Systat Software, USA). The relationship between two variables was analyzed using the Pearson test. For comparison of the means of two or more groups, the Student’s t-test or the one-way analysis of variance (ANOVA) was used, respectively. To test for the heterogeneity of the slopes of linear regressions the analysis of covariance (ANCOVA) was applied. All levels of significance were set to α = 0.05.
3. Results
3.1. DSB Repair foci
We first examined the impact of CDDP pretreatment on IR-induced DSBs and their repair in unstimulated PBLs in G1 using the γH2AX/53BP1 foci assay. Exemplary fluorescence microscopy images of γH2AX/53BP1 foci in PBLs are shown in
Figure 1A for different treatments. The initial γH2AX/53BP1 RIF were analyzed 0.5h post-irradiation with 0.5 Gy, while residual RIF were assessed 24h after 2 Gy, following either a 1h or 24h CDDP pretreatment (
Figure 1B-E). Different IR doses ensured countable foci numbers in PBLs at different examination times [
26].
Pulse treatment of PBLs with increasing concentrations of CDDP alone for 1h did not alter γH2AX/53BP1 foci levels compared to mock-treated cells, with a total average of 0.19 ± 0.02 foci per PBL (
Figure 1B). 0.5h after 0.5 Gy X-rays, a comparable average of 4.64 ± 0.19 γH2AX/53BP1 total foci per PBL was observed, regardless of CDDP concentration (
Figure 1B). Similarly, the levels of initial RIF with an average of 4.48 ± 0.20 per cell were also in this range and were comparable between all concentrations of CDDP pulse treatment (
Figure 1B). Also, a 24h incubation of PBLs after a 1h pulse treatment and 2 Gy X-rays showed no impact on the average basal foci level of 0.17 ± 0.05 per cell following CDDP treatment alone, nor on the average total residual foci and RIF after irradiation of 2.01 ± 0.29 and 1.92 ± 0.11 per cell, respectively (
Figure 1C). These basal foci and RIF values in PBLs closely match our extensive previous γH2AX foci data sets from 14 healthy donors and more than 150 patients with benign or malignant pathologies for similar doses and time points [
26,
33,
34]. Thus, short-term pulse treatment with CDDP did not impact initial and residual levels of basal or IR-induced γH2AX/53BP1 foci in PBLs, also at a higher IR dose of 1 Gy (
Supplementary Figure S1).
Given this finding and our interest in prolonged CDDP exposure effects on PBLs, we switched to a continuous 24h CDDP pre-treatment before irradiation. 24h CDDP pretreatment alone caused a concentration-dependent increase in γH2AX/53BP1 foci, peaking at 3.21 ± 1.45 foci per PBL at 20 µg/ml CDDP (
Figure 1D). 0.5h after 0.5 Gy exposures, a comparable 5.4 ± 0.37 total foci per PBL were observed across all CDDP concentrations (
Figure 1D). The increase in CDDP-induced basal foci led to a non-significant decrease in RIF per PBL, depending on the CDDP concentration (
Figure 1D). Prolonged incubation to assess residual RIF 24h after 2 Gy exposures led to a significant increase in mean CDDP-induced foci from 5 µg/ml, saturating at 2.46 ± 0.09 foci per PBL at higher concentrations (
Figure 1E). 24h post-2 Gy exposure, a comparable average of 2.42 ± 0.10 total foci per PBL was observed across all CDDP concentrations (
Figure 1E). The increasing background yield of CDDP-induced foci caused a significant decrease with partially negative residual RIF per PBL (
Figure 1E). Notably, a 24h CDDP treatment and in particular the combination with IR led to an increase in cells with pan nuclear γH2AX signals, a strong indicator of DNA fragmentation during apoptosis (
Supplementary Figure S2).
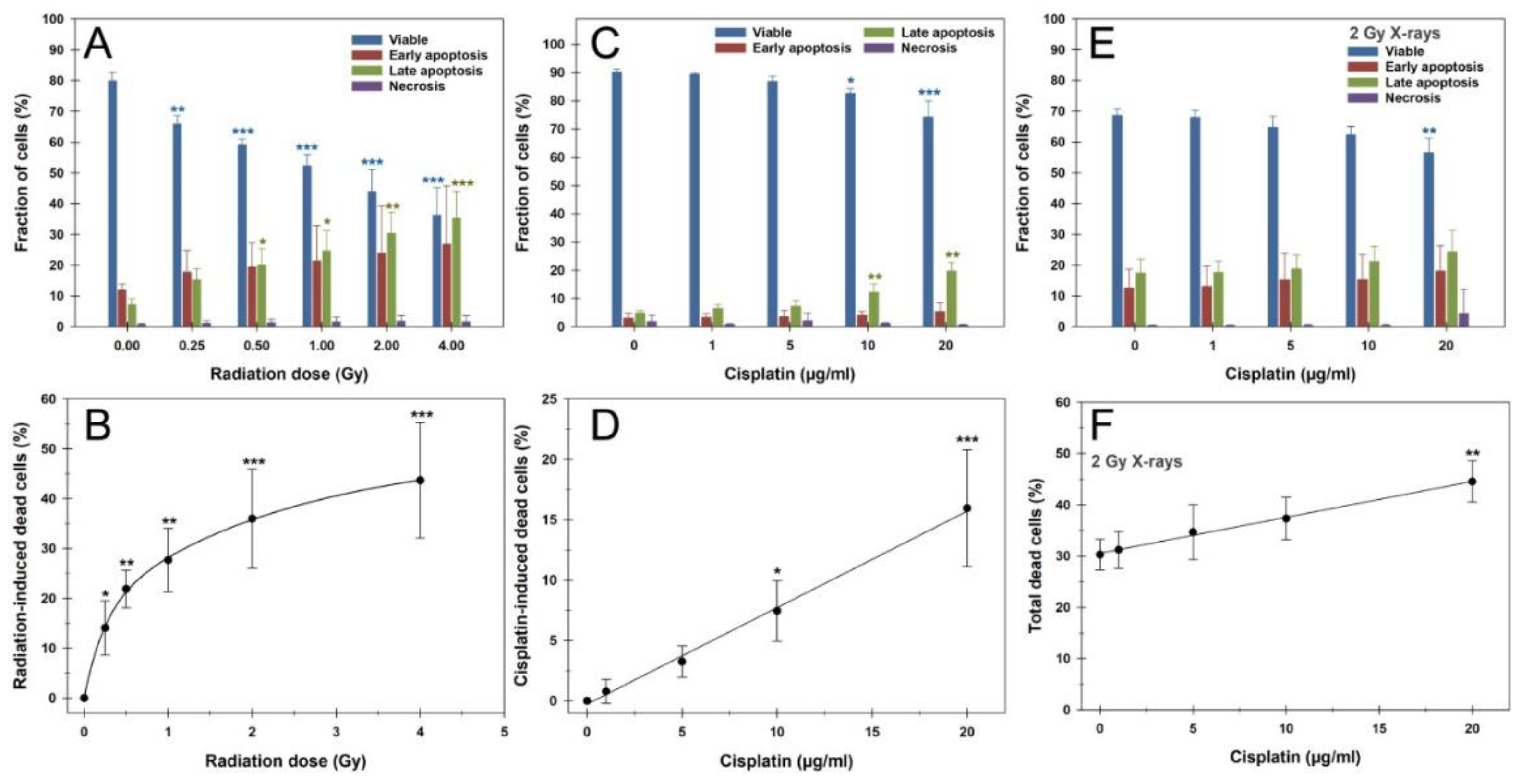
3.2. Apoptosis
Based on these observations, we conducted cell death measurements in G1 PBLs 24h after treatments using the AnnexinV/PI assay, as shown in
Figure 2. 24h after irradiation with X-rays of up to 4 Gy, a significant dose-dependent increase in the proportion of dead cells comprising early and late apoptosis plus necrosis was observed from the lowest dose of 0.25 Gy (
Figure 2A and B). Early and late apoptosis occurred in equal proportions, while necrosis was generally negligible for all treatments. Following a steep increment up to 1 Gy, the fraction of dead cells leveled off and was best fitted with the four-parameter exponential rise to maximum function Y = 17.2 * (1 - exp(-4.2 * X)) + 32.4 * (1 - exp(-0.43 * X)) (R = 1.0).
A 24h treatment with CDDP, followed by 24h incubation after CDDP withdrawal, caused a concentration-dependent linear increase in dead cells following Y = 0.78 * X (R = 1.0), with significant proportions from a concentration of 10 µg/ml (
Figure 2B and C). Here, late apoptosis dominated in particular. After 2 Gy irradiation of PBLs pre-treated with CDDP for 24h followed by 24h incubation without CDDP, a CDDP-dependent linear increase in dead cells was again observed (
Figure 2E and F). A significant difference from irradiation alone was only present at the highest CDDP concentration of 20 µg/ml. The linear increase in total dead cells followed the linear function Y = 30.6 + 0.70 * X (R = 1.0). Since this slope was only slightly and nonsignificantly (p = 0.86) lower than with CDDP alone, this indicates an additive effect of CDDP with IR.
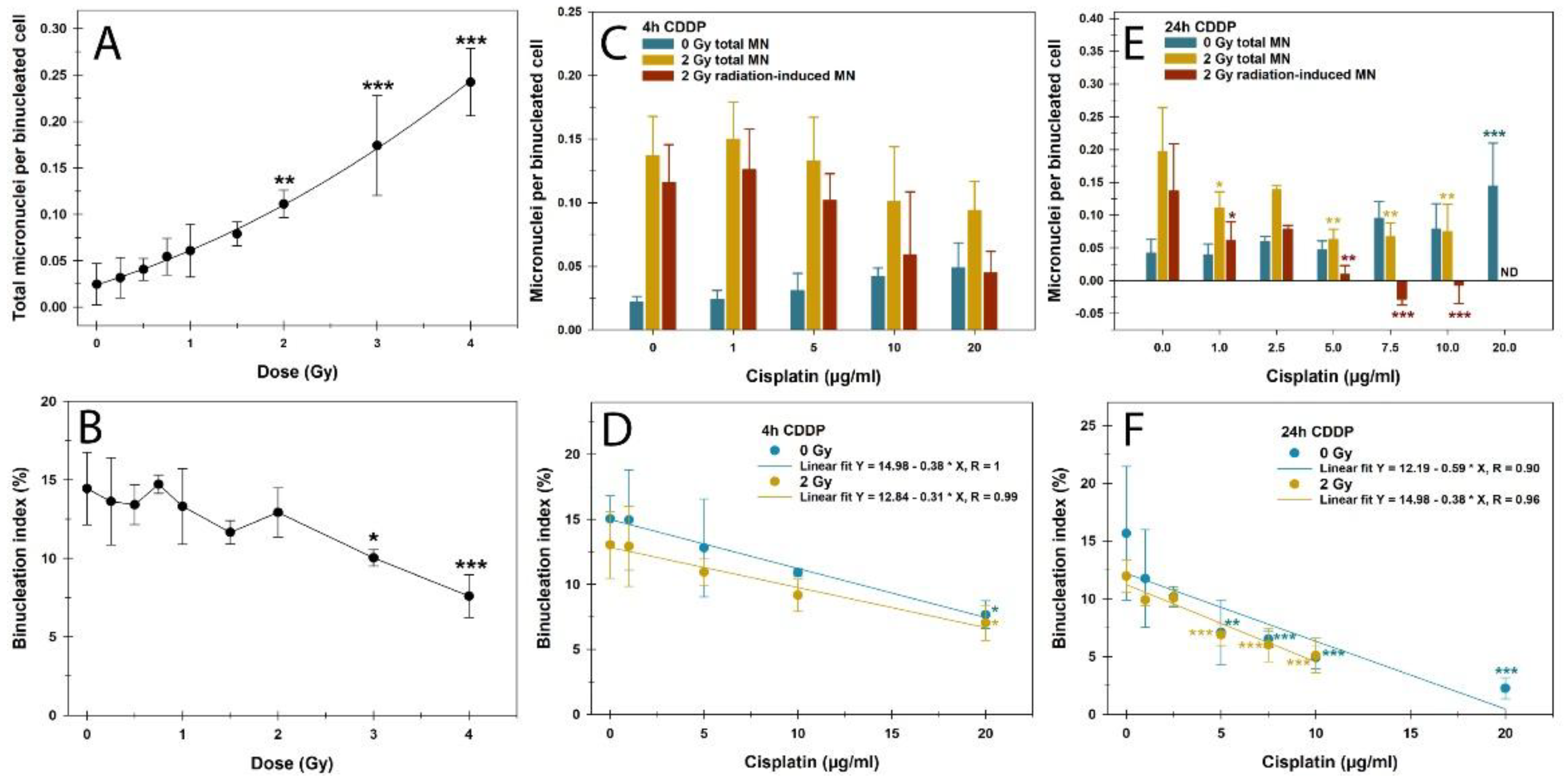
3.3. CBMN Assay and Cell Cycle
Our next step was to investigate the influence of CDDP on the detection of IR-induced DNA damage in the proliferation-dependent CBMN assay. Irradiation with X-ray doses of up to 4 Gy led to an increase of MN in BN PBLs with the linear quadratic function Y = 0.024 + 0.031 * X + 0.006 * X
2 (R = 1.0) (
Figure 3A). The binucleation index, measuring proliferation activity and cell cycle arrest from genotoxic influences, remained stable up to 2 Gy and showed a significant reduction only from 3 Gy onwards. (
Figure 3B). Therefore, a 2 Gy dose was chosen for the combined treatment regimes.
CDDP pretreatment for this assay was based on DNA damage results from the γH2AX/53BP1 foci assay over 4h and 24h, followed by incubation in a CDDP-free medium for up to 72h. For both pretreatment regimens, MN increased with CDDP concentration, more pronounced after the 24h pretreatment (
Figure 3C, E). Similarly, CDDP pretreatment reduced the binucleation index in a largely linear concentration- and time-dependent manner, with the strongest effect in the 24h pretreatment regime (
Figure 3D, F). However, no significant difference was observed between the linear slopes of sham-irradiated and irradiated samples of the respective CDDP pretreatments as provided in
Figure 3D and F (p > 0.05). After 24h pretreatment with CDDP concentrations ≥ 10 µg/ml, binucleation index and proliferation activity were very low and ≤ 5%, as confirmed by cell cycle measurements (
Figure 4). Therefore, a low concentration range of ≤ 10 µg/ml was selected for 24h CDDP pretreatment followed by 2 Gy X-rays to score MN in BN cells (
Figure 3C, E). The 4h CDDP pretreatment reduced the average total MN per BN cell after 2 Gy X-rays at CDDP concentrations from 10 µg/ml, though not significantly (
Figure 3C). In contrast, the 24h CDDP pretreatment significantly decreased MN frequencies in BN cells already at 1 µg/ml CDDP (
Figure 3E). As for the DSB foci assay, this resulted in partially negative values for IR-induced MN from 7.5 µg/ml CDDP. These findings highlight a strong confounding effect of prolonged CDDP exposure on this proliferation-dependent assay for detecting IR-induced DNA damage, even at low concentrations.
4. Discussion
This study examined the effects of CDDP, X-rays, and their combination on PBLs from healthy donors under controlled ex vivo conditions, focusing on DNA damage, proliferation, and cell death to better understand their impact on biodosimetry and adverse hematologic treatment effects during CRT. Both treatments alone induced DSBs, micronuclei, and cell death. Combined treatment additively increased cell death, strongly inhibited proliferation and reduced IR-associated DNA damage markers due to cell depletion and suppressed cell division. These findings highlight the adverse hematologic effects of CDDP and its confounding impact on biodosimetry during CRT.
The cytotoxic and antiproliferative actions of CDDP underlie both its therapeutic and side effects [
11]. Systemic CDDP administration impacts the hematological system also for solid tumors, and its frequent combination with external beam RT can cause excessive damage to the highly radiosensitive blood system [
16]. A key example is platinum-based CRT for head and neck tumor patients, where lymphopenia is a negative prognostic factor, alongside other factors, e.g. nutrition [
36]. Rajkumar et al. [
37] measured the concentration of CDDP in the plasma of head and neck cancer patients receiving intravenous infusion of 40 mg/m
2 for 1h or 100 mg/m
2 for 3h up to 5h or 24h, respectively. Although free CDDP concentrations dropped significantly from peak values of ~6 µg/ml (1h regime) and ~11 µg/ml (3h regime) 1h after CDDP discontinuation, plasma levels around 2 µg/ml remained detectable at 5h and 24h post-treatment, respectively. Elevated serum levels of cytostatic drugs including CDDP can even persist for decades after treatment and are linked to adverse long-term effects [
38,
39]. In this context, however, no studies exist on combined effects with civilian radiation exposures from natural background radiation or planned medical exposures. Based on Rajkumar et al. [
37], we used 1–20 µg/ml CDDP in pulse or continuous pretreatment scenarios to study its sole and combined effects with IR on PBLs under well-defined
ex vivo conditions.
We first examined CDDP's impact on the induction and repair of IR-induced DSBs, the most prompt and severe IR damage, whose repair is crucial for cell survival. A 1h CDDP pulse had no significant impact on colocalizing γH2AX/53BP1 foci frequencies used as DSB surrogate markers in PBLs. Only a 24h CDDP treatment increased foci rates in a concentration-dependent manner, significantly after an additional 24h incubation after CDDP withdrawal. Since similar initial or residual total foci counts were observed post-irradiation, residual RIF frequencies were significantly reduced 24h after 2 Gy irradiation after subtracting CDDP-induced baseline values from low CDDP concentration onwards. Thus, even low-dose continuous CDDP exposure can significantly impact the quantitation of IR-induced DSB surrogate markers in PBLs for biodosimetry and as predictive biomarkers in a clinical context. Whether CDDP affects the formation of IR-induced DSB repair foci in PBLs has only been studied
in vivo and
ex vivo by Sak et al. [
31], including γH2AX as a general DSB marker and Rad51 as a marker of resection-dependent DSB repair by homologous recombination. Their studies were carried out on blood samples from 28 cancer patients with intrathoracic, pelvic, or head and neck tumors who received CDDP concurrent with RT. When patients' blood samples were irradiated
ex vivo on different days post
in vivo CDDP infusion, mean γH2AX foci frequencies in PBLs dropped from about 10 to 6 per Gy between day 0 and day 4, before returning to pre-treatment levels. Similarly, for the
in vivo response 0.5h after RT, with CDDP given 1–1.5h before RT, a significant decrease in relative γH2AX foci rates in PBLs by 28% to 46% was observed between days 0 and 4 after RT. The authors also examined the sole
ex vivo effect of CDDP on γH2AX foci in untreated patient samples before therapy. PBLs treated
ex vivo for 1h with up to 50 µg/ml CDDP showed no altered foci values, aligning with our data. However, 0.5h after 1 Gy γ-irradiation, a concentration-dependent decrease in mean foci rates was observed, starting at 1 µg/ml CDDP. This observation is in stark contrast with our results on isolated PBLs from healthy donors, which showed no effect of a 1h pulse pretreatment with CDDP concentrations up to 20 µg/ml on the initial RIF rates. The reason for this inconsistency between studies remains unraveled, with the only difference being between cancer patients and healthy donors.
A reduced formation of IR-induced DSB repair foci has been generally linked to the impairment of repair mechanisms by CDDP adducts on DSBs, which prevent binding and decrease the activity of the KU70/80 heterodimer of the DNA protein kinase (DNA-PK) [
8,
9,
40,
41,
42]. Abrogation of KU70/80 DSB end-binding and activity, the initiation step of NHEJ as the primary DSB repair pathway, increases lesion complexity and lethality, enhancing the combinatorial effect of CDDP and RT in tumor cell inactivation [
8,
42]. A major target of DNA-PK is the histone variant H2AX, which is redundantly phosphorylated at Ser139 by Ataxia Telangiectasia Mutated (ATM) and DNA-PK catalytic subunit (DNA-PKcs) immediately after DSB induction [
43]. Thus, despite potentially impaired DNA-PKcs activity in the presence of CDDP, H2AX should still be fully phosphorylated by ATM and form γH2AX foci. Additionally, using 53BP1 as another DSB marker confirmed our γH2AX observations. Despite lower RIF formation, Sak et al. [
31] found no impairment in NHEJ-mediated repair of IR-induced DSBs by CDDP in PBLs. Overall, a mechanism for the potential CDDP-related reduction of early RIF remains unraveled. Also, our data show no effect of CDDP on initial RIF levels shortly after irradiation and do not support these previous observations. However, since we withdrew CDDP before irradiation, CDDP adduct formation may have shifted toward Ku70/80 binding and activity at DSBs, potentially influencing RIF formation. The effect of CDDP on the repair of IR-induced DSBs may also depend on the concentration and timing of the treatments [
44].
We attribute the significant impact of 24h CDDP pretreatment on residual RIF levels 24h post-irradiation to increased cell death rates. PBLs with severe pre-damage from continuous CDDP treatment likely fail to survive additional IR-induced damage, leading to apoptosis. This is supported by higher rates of apoptotic PBLs with pan-nuclear γH2AX signals that do not allow cumulative CDDP- plus IR-induced foci quantification. Our Annexin V/PI apoptosis measurements confirmed the mostly additive effect of CDDP pretreatment followed by IR on PBL cell death.
As a second endpoint of DNA damage in PBLs, we used the proliferation-dependent CBMN assay, another routine biodosimeter after radiation accidents, occupational radiation exposures, and RT in tumor patients [
45,
46]. Expectedly, CDDP and IR alone induced cytogenetic damage detected as MN in BN cells. Compared to IR, CDDP pretreatment for 4h and 24h followed by 2 Gy X-rays had a mild or a highly significant reducing impact on MN frequencies, respectively. In general, decreased MN yields correlated with a significant BN index decrement due to CDDP pretreatment. These observations are also clinically relevant for multimodal tumor therapies, particularly for the crucial antineoplastic activity of proliferative CD8+ T cells [
47].
Dolling et al. [
44] already demonstrated a significant BN index reduction in normal human fibroblasts after a 1 µg/ml CDDP pulse for 0.5h, but no enhancement of IR-induced effects with 4 Gy X-ray irradiation 24h later. While CDDP treatment just before irradiation slightly increased BN cells with MN, continuous 24h treatment at 1 µg/ml and lower reversed this effect. These results were interpreted more as CDDP-mediated stimulation of an adaptive response promoting repair of subsequent IR-induced DNA damage rather than an effect on the cell cycle. Also, Azab et al. [
48] examined CDDP's cytogenetic effects, finding increased chromosomal aberrations and a significant mitotic index drop, but no change in the general proliferation index at 0.4 µg/ml CDDP in cultured PBLs. Along with our similar BN index results for higher CDDP concentrations, this was confirmed by our cell cycle measurements with or without nocodazole as a spindle inhibitor. Without nocodazole, only a high concentration of 20 µg/ml CDDP significantly affected cell cycle distribution. With nocodazole, G2/M cell proportions decreased concentration-dependently, while S-phase proportions increased, suggesting replicative stress induced by CDDP adducts. Even though oncology therapies combine CDDP and spindle inhibitors with a significant benefit for overall survival, e.g. in advanced or metastatic non-small cell lung cancer as CDDP plus vinorelbine, a combinatorial antineoplastic effect on the cell cycle has not yet been investigated [
49].
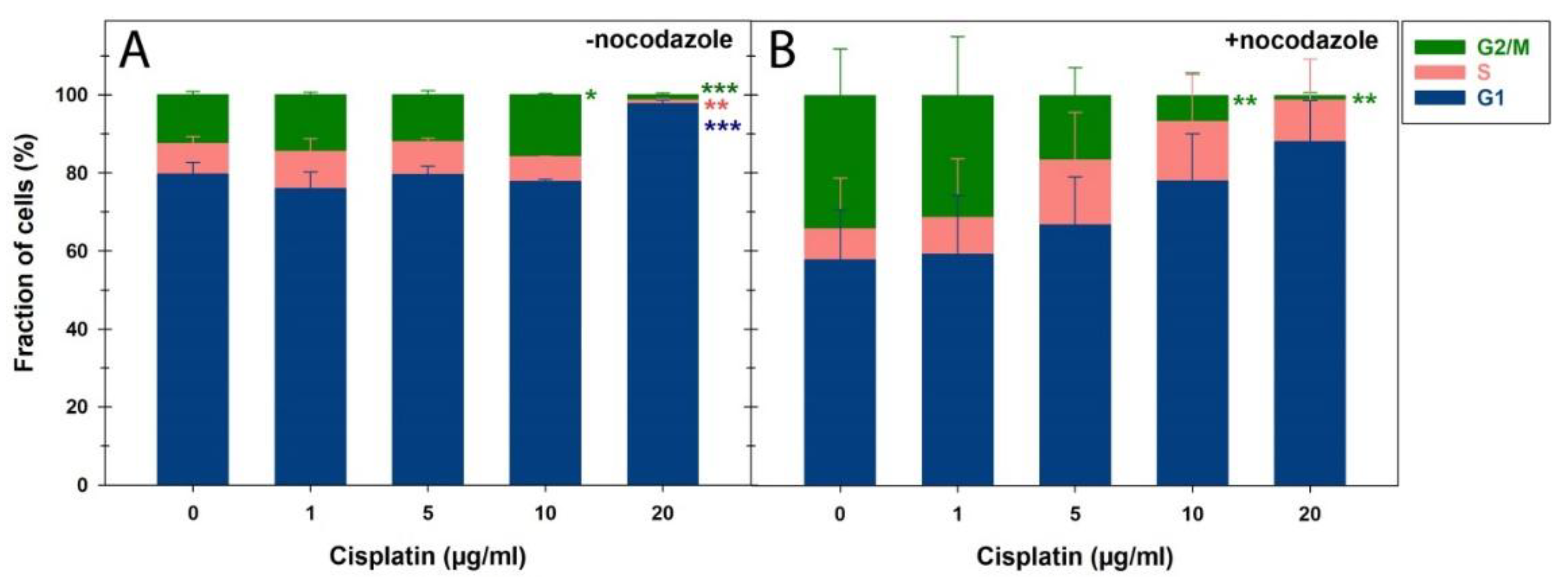
Wegierek-Ciuk et al. [
32] studied CDDP's impact on IR-induced MN formation, BN index, and apoptosis based on morphological features in PBLs from gynecologic cancer patients undergoing definitive RT or CDDP-based CRT. Similar to the experimental settings of Sak et al. [
31],
ex vivo γ-irradiation of blood samples from CDDP-based CRT patients with 2 Gy led to fewer MN, higher apoptosis rates, and a lower BN index in PBLs compared to definitive RT.
In vivo, despite higher tumor doses, larger target volumes, and genotoxic CDDP, CRT patients did not show higher MN rates during treatment than RT-only patients, again due to increased apoptosis and a lower BN index. The authors attributed these observations of CDDP's sensitizing effect on radiation cytotoxicity, promoting an ‘overkill effect’ by eliminating severely damaged cells, aligning with our findings. However, the issue with
in vivo or mixed
in vivo-
ex vivo studies is the complexity and imprecision of intrinsic patient parameters, therapeutic exposures to CDDP and RT, and other cytostatic agents or confounders. Thus, we first chose a well-defined
ex vivo approach with isolated PBLs from healthy donors to guide future
in vivo studies, which are also currently underway in our laboratory.
5. Conclusions
Our analysis of CDDP alone or with IR on isolated PBLs under controlled ex vivo conditions showed significant effects on radiobiological and biodosimetric DNA damage and repair markers. Apoptosis induction and reduced division activity, even at low CDDP concentrations, must be considered in clinical radiation studies to prevent misinterpretation. Moreover, persistently low CDDP blood levels may have deleterious and immunosuppressive long-term effects, especially with additional radiation exposure.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org, Figure S1: Quantification of colocalized γH2AX and 53BP1 (γH2AX/53BP1) foci in peripheral blood lymphocytes (PBLs) after cisplatin (CDDP), 1 Gy X-rays, or their combination, with a 1h CDDP pretreatment before irradiation. Figure S2: Immunofluorescence microscopy overview shows γH2AX signals (green) in counterstained lymphocyte nuclei (blue) 24 hours post-irradiation (2 Gy X-rays) after 24-hour pretreatment with increasing CDDP concentrations.
Author Contributions
Conceptualization: S.Z., J.K., S.D., H.S. Data curation: S.Z., A.B., B.A. Formal analysis: S.Z. Funding acquisition: S.Z, H.S. Investigation: S.Z, A.B, B.A. Methodology: S.Z, J.K, S.D, H.S. Project administration: S.Z. Resources: S.Z, H.S. Software Supervision: S.Z. Validation: S.Z., J.K., S.D., H.S. Visualization: S.Z. Writing – original draft: S.Z. Writing – review & editing: S.Z., J.K., S.D., H.S., A.B., B.A.
Funding
This study was supported by the German Federal Ministry of Education and Research, grant number 02NUK084A.
Institutional Review Board Statement
Studies with human blood or blood components were approved by the Ethics Committee of the Medical Association of Rhineland-Palatinate [no. 2023-17191 and 2024-17401_2].
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Acknowledgments
The authors thank Giusy Carlino for excellent technical support, all donors for providing blood samples, and the Institute of Transfusion Medicine - Transfusion Center, Johannes Gutenberg University Medical Center, Mainz, Germany, for buffy coat production and supply.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Abbreviations
The following abbreviations are used in this manuscript:
IR Ionizing radiation
PBMC Peripheral blood mononuclear cells
PBL Peripheral blood lymphocyte
PI Propidium iodide
CDDP Cis-diamminedichloroplatinum II
CT Chemotherapy
RT Radiotherapy
CRT Chemoradiotherapy
LA Locally advanced
SSB Single-strand break
DSB Double-strand break
NHEJ Non-homologous end joining
ICI Immune checkpoint inhibitors
EDTA Ethylenediaminetetraacetic acid
PBS Phosphate-buffered saline
FCS Fetal calf serum
RIF Radiation-induced foci
BN Binucleated
MN Micronuclei
ND Not determined
References
- Makovec, T. Cisplatin and beyond: molecular mechanisms of action and drug resistance development in cancer chemotherapy. Radiol Oncol 2019, 53, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.; Kumar, S.; Tchounwou, P.B. Cisplatin-Based Chemotherapy of Human Cancers. J Cancer Sci Ther 2019, 11. [Google Scholar]
- Steel, G.G.; Peckham, M.J. Exploitable mechanisms in combined radiotherapy-chemotherapy: the concept of additivity. Int J Radiat Oncol Biol Phys 1979, 5, 85–91. [Google Scholar] [CrossRef]
- Seiwert, T.Y.; Salama, J.K.; Vokes, E.E. The concurrent chemoradiation paradigm--general principles. Nat Clin Pract Oncol 2007, 4, 86–100. [Google Scholar] [CrossRef]
- Yang, L.X.; Douple, E.B.; O'Hara, J.A.; Wang, H.J. Production of DNA double-strand breaks by interactions between carboplatin and radiation: a potential mechanism for radiopotentiation. Radiat Res 1995, 143, 309–315. [Google Scholar] [CrossRef]
- Sorenson, C.M.; Eastman, A. Mechanism of cis-diamminedichloroplatinum(II)-induced cytotoxicity: role of G2 arrest and DNA double-strand breaks. Cancer Res 1988, 48, 4484–4488. [Google Scholar]
- Shinkai, T.; Saijo, N.; Eguchi, K.; Sasaki, Y.; Tamura, T.; Sakurai, M.; Suga, J.; Nakano, H.; Nakagawa, K.; Hong, W.S.; et al. Cytogenetic effect of carboplatin on human lymphocytes. Cancer Chemother Pharmacol 1988, 21, 203–207. [Google Scholar] [CrossRef]
- Boeckman, H.J.; Trego, K.S.; Turchi, J.J. Cisplatin sensitizes cancer cells to ionizing radiation via inhibition of nonhomologous end joining. Molecular cancer research : MCR 2005, 3, 277–285. [Google Scholar] [CrossRef]
- Sears, C.R.; Turchi, J.J. Complex cisplatin-double strand break (DSB) lesions directly impair cellular non-homologous end-joining (NHEJ) independent of downstream damage response (DDR) pathways. J Biol Chem 2012, 287, 24263–24272. [Google Scholar] [CrossRef]
- Tchounwou, P.B.; Dasari, S.; Noubissi, F.K.; Ray, P.; Kumar, S. Advances in Our Understanding of the Molecular Mechanisms of Action of Cisplatin in Cancer Therapy. J Exp Pharmacol 2021, 13, 303–328. [Google Scholar] [CrossRef]
- Nagasawa, S.; Takahashi, J.; Suzuki, G.; Hideya, Y.; Yamada, K. Why Concurrent CDDP and Radiotherapy Has Synergistic Antitumor Effects: A Review of In Vitro Experimental and Clinical-Based Studies. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Kirwan, J.M.; Symonds, P.; Green, J.A.; Tierney, J.; Collingwood, M.; Williams, C.J. A systematic review of acute and late toxicity of concomitant chemoradiation for cervical cancer. Radiotherapy and oncology : journal of the European Society for Therapeutic Radiology and Oncology 2003, 68, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Furuse, K.; Fukuoka, M.; Kawahara, M.; Nishikawa, H.; Takada, Y.; Kudoh, S.; Katagami, N.; Ariyoshi, Y. Phase III study of concurrent versus sequential thoracic radiotherapy in combination with mitomycin, vindesine, and cisplatin in unresectable stage III non-small-cell lung cancer. J Clin Oncol 1999, 17, 2692–2699. [Google Scholar] [CrossRef] [PubMed]
- Trowell, O.A. The sensitivity of lymphocytes to ionising radiation. J Pathol Bacteriol 1952, 64, 687–704. [Google Scholar] [CrossRef]
- Heylmann, D.; Ponath, V.; Kindler, T.; Kaina, B. Comparison of DNA repair and radiosensitivity of different blood cell populations. Scientific reports 2021, 11, 2478. [Google Scholar] [CrossRef]
- Grossman, S.A.; Ellsworth, S.; Campian, J.; Wild, A.T.; Herman, J.M.; Laheru, D.; Brock, M.; Balmanoukian, A.; Ye, X. Survival in Patients With Severe Lymphopenia Following Treatment With Radiation and Chemotherapy for Newly Diagnosed Solid Tumors. J Natl Compr Canc Netw 2015, 13, 1225–1231. [Google Scholar] [CrossRef]
- Darragh, L.B.; Gadwa, J.; Pham, T.T.; Van Court, B.; Neupert, B.; Olimpo, N.A.; Nguyen, K.; Nguyen, D.; Knitz, M.W.; Hoen, M.; et al. Elective nodal irradiation mitigates local and systemic immunity generated by combination radiation and immunotherapy in head and neck tumors. Nature communications 2022, 13, 7015. [Google Scholar] [CrossRef]
- Telarovic, I.; Yong, C.S.M.; Kurz, L.; Vetrugno, I.; Reichl, S.; Fernandez, A.S.; Cheng, H.W.; Winkler, R.; Guckenberger, M.; Kipar, A.; et al. Delayed tumor-draining lymph node irradiation preserves the efficacy of combined radiotherapy and immune checkpoint blockade in models of metastatic disease. Nat Commun 2024, 15, 5500. [Google Scholar] [CrossRef]
- Galluzzi, L.; Aryankalayil, M.J.; Coleman, C.N.; Formenti, S.C. Emerging evidence for adapting radiotherapy to immunotherapy. Nat Rev Clin Oncol 2023. [Google Scholar] [CrossRef]
- Saddawi-Konefka, R.; O'Farrell, A.; Faraji, F.; Clubb, L.; Allevato, M.M.; Jensen, S.M.; Yung, B.S.; Wang, Z.; Wu, V.H.; Anang, N.A.; et al. Lymphatic-preserving treatment sequencing with immune checkpoint inhibition unleashes cDC1-dependent antitumor immunity in HNSCC. Nat Commun 2022, 13, 4298. [Google Scholar] [CrossRef]
- Rosenberg, A.J.; Juloori, A.; Jelinek, M.J.; Agrawal, N.; Cursio, J.F.; Cipriani, N.; Lingen, M.W.; Izumchenko, E.; Katipally, R.; Chin, J.; et al. Neoadjuvant Nivolumab Plus Chemotherapy Followed by Response-Stratified Chemoradiation Therapy in HPV-Negative Head and Neck Cancer: The DEPEND Phase 2 Nonrandomized Clinical Trial. JAMA Oncol 2025. [Google Scholar] [CrossRef]
- Marciscano, A.E.; Ghasemzadeh, A.; Nirschl, T.R.; Theodros, D.; Kochel, C.M.; Francica, B.J.; Muroyama, Y.; Anders, R.A.; Sharabi, A.B.; Velarde, E.; et al. Elective Nodal Irradiation Attenuates the Combinatorial Efficacy of Stereotactic Radiation Therapy and Immunotherapy. Clinical cancer research : an official journal of the American Association for Cancer Research 2018, 24, 5058–5071. [Google Scholar] [CrossRef]
- Lee, N.Y.; Ferris, R.L.; Psyrri, A.; Haddad, R.I.; Tahara, M.; Bourhis, J.; Harrington, K.; Chang, P.M.; Lin, J.C.; Razaq, M.A.; et al. Avelumab plus standard-of-care chemoradiotherapy versus chemoradiotherapy alone in patients with locally advanced squamous cell carcinoma of the head and neck: a randomised, double-blind, placebo-controlled, multicentre, phase 3 trial. Lancet Oncol 2021, 22, 450–462. [Google Scholar] [CrossRef]
- Machiels, J.P.; Tao, Y.; Licitra, L.; Burtness, B.; Tahara, M.; Rischin, D.; Alves, G.; Lima, I.P.F.; Hughes, B.G.M.; Pointreau, Y.; et al. Pembrolizumab plus concurrent chemoradiotherapy versus placebo plus concurrent chemoradiotherapy in patients with locally advanced squamous cell carcinoma of the head and neck (KEYNOTE-412): a randomised, double-blind, phase 3 trial. Lancet Oncol 2024, 25, 572–587. [Google Scholar] [CrossRef]
- Haddad, R.; Fayette, J.; Teixeira, M.; Prabhash, K.; Mesia, R.; Kawecki, A.; Dechaphunkul, A.; Dinis, J.; Guo, Y.; Masuda, M.; et al. Atezolizumab in High-Risk Locally Advanced Squamous Cell Carcinoma of the Head and Neck: A Randomized Clinical Trial. Jama 2025. [Google Scholar] [CrossRef]
- Zahnreich, S.; Ebersberger, A.; Kaina, B.; Schmidberger, H. Biodosimetry Based on gamma-H2AX Quantification and Cytogenetics after Partial- and Total-Body Irradiation during Fractionated Radiotherapy. Radiat Res 2015, 183, 432–446. [Google Scholar] [CrossRef]
- Durante, M.; Yamada, S.; Ando, K.; Furusawa, Y.; Kawata, T.; Majima, H.; Nakano, T.; Tsujii, H. Measurements of the equivalent whole-body dose during radiation therapy by cytogenetic methods. Phys Med Biol 1999, 44, 1289–1298. [Google Scholar] [CrossRef]
- Sak, A.; Grehl, S.; Erichsen, P.; Engelhard, M.; Grannass, A.; Levegrun, S.; Pottgen, C.; Groneberg, M.; Stuschke, M. gamma-H2AX foci formation in peripheral blood lymphocytes of tumor patients after local radiotherapy to different sites of the body: dependence on the dose-distribution, irradiated site and time from start of treatment. Int J Radiat Biol 2007, 83, 639–652. [Google Scholar] [CrossRef]
- Rothkamm, K.; Beinke, C.; Romm, H.; Badie, C.; Balagurunathan, Y.; Barnard, S.; Bernard, N.; Boulay-Greene, H.; Brengues, M.; De Amicis, A.; et al. Comparison of established and emerging biodosimetry assays. Radiation research 2013, 180, 111–119. [Google Scholar] [CrossRef]
- Fleckenstein, J.; Kühne, M.; Seegmüller, K.; Derschang, S.; Melchior, P.; Gräber, S.; Fricke, A.; Rübe, C.E.; Rübe, C. The impact of individual in vivo repair of DNA double-strand breaks on oral mucositis in adjuvant radiotherapy of head-and-neck cancer. Int J Radiat Oncol Biol Phys 2011, 81, 1465–1472. [Google Scholar] [CrossRef]
- Sak, A.; Grehl, S.; Engelhard, M.; Wierlemann, A.; Kaelberlah, H.P.; Erichsen, P.; Pöttgen, C.; Groneberg, M.; Stuschke, M. Long-term in vivo effects of cisplatin on gamma-H2AX foci signaling in peripheral lymphocytes of tumor patients after irradiation. Clin Cancer Res 2009, 15, 2927–2934. [Google Scholar] [CrossRef] [PubMed]
- Węgierek-Ciuk, A.; Lankoff, A.; Lisowska, H.; Kędzierawski, P.; Akuwudike, P.; Lundholm, L.; Wojcik, A. Cisplatin Reduces the Frequencies of Radiotherapy-Induced Micronuclei in Peripheral Blood Lymphocytes of Patients with Gynaecological Cancer: Possible Implications for the Risk of Second Malignant Neoplasms. Cells 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Zahnreich, S.; Ebersberger, A.; Karle, H.; Kaina, B.; Schmidberger, H. Quantification of Radiation Biomarkers in Leukocytes of Breast Cancer Patients Treated with Different Modalities of 3D-CRT or IMRT. Radiat Res 2016, 186, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Zahnreich, S.; Rösler, H.P.; Schwanbeck, C.; Karle, H.; Schmidberger, H. Radiation-induced DNA double-strand breaks in peripheral leukocytes and therapeutic response of heel spur patients treated by orthovoltage X-rays or a linear accelerator. Strahlentherapie und Onkologie : Organ der Deutschen Rontgengesellschaft... [et al]. [CrossRef]
- Zahnreich, S.; Weber, B.; Rösch, G.; Schindler, D.; Schmidberger, H. Compromised repair of radiation-induced DNA double-strand breaks in Fanconi anemia fibroblasts in G2. DNA repair 2020, 96, 102992. [Google Scholar] [CrossRef]
- Campian, J.L.; Sarai, G.; Ye, X.; Marur, S.; Grossman, S.A. Association between severe treatment-related lymphopenia and progression-free survival in patients with newly diagnosed squamous cell head and neck cancer. Head Neck 2014, 36, 1747–1753. [Google Scholar] [CrossRef]
- Rajkumar, P.; Mathew, B.S.; Das, S.; Isaiah, R.; John, S.; Prabha, R.; Fleming, D.H. Cisplatin Concentrations in Long and Short Duration Infusion: Implications for the Optimal Time of Radiation Delivery. J Clin Diagn Res 2016, 10, Xc01–xc04. [Google Scholar] [CrossRef]
- Gietema, J.A.; Meinardi, M.T.; Messerschmidt, J.; Gelevert, T.; Alt, F.; Uges, D.R.; Sleijfer, D.T. Circulating plasma platinum more than 10 years after cisplatin treatment for testicular cancer. Lancet 2000, 355, 1075–1076. [Google Scholar] [CrossRef]
- Trendowski, M.R.; El-Charif, O.; Ratain, M.J.; Monahan, P.; Mu, Z.; Wheeler, H.E.; Dinh, P.C., Jr.; Feldman, D.R.; Ardeshir-Rouhani-Fard, S.; Hamilton, R.J.; et al. Clinical and Genome-Wide Analysis of Serum Platinum Levels after Cisplatin-Based Chemotherapy. Clin Cancer Res 2019, 25, 5913–5924. [Google Scholar] [CrossRef]
- Turchi, J.J.; Henkels, K.M.; Zhou, Y. Cisplatin-DNA adducts inhibit translocation of the Ku subunits of DNA-PK. Nucleic Acids Res 2000, 28, 4634–4641. [Google Scholar] [CrossRef]
- Diggle, C.P.; Bentley, J.; Knowles, M.A.; Kiltie, A.E. Inhibition of double-strand break non-homologous end-joining by cisplatin adducts in human cell extracts. Nucleic Acids Res 2005, 33, 2531–2539. [Google Scholar] [CrossRef]
- Wilson, G.D.; Bentzen, S.M.; Harari, P.M. Biologic basis for combining drugs with radiation. Semin Radiat Oncol 2006, 16, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Stiff, T.; O'Driscoll, M.; Rief, N.; Iwabuchi, K.; Löbrich, M.; Jeggo, P.A. ATM and DNA-PK function redundantly to phosphorylate H2AX after exposure to ionizing radiation. Cancer Res 2004, 64, 2390–2396. [Google Scholar] [CrossRef] [PubMed]
- Dolling, J.A.; Boreham, D.R.; Brown, D.L.; Mitchel, R.E.; Raaphorst, G.P. Modulation of radiation-induced strand break repair by cisplatin in mammalian cells. Int J Radiat Biol 1998, 74, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Sommer, S.; Buraczewska, I.; Kruszewski, M. Micronucleus Assay: The State of Art, and Future Directions. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Fenech, M. Cytokinesis-block micronucleus cytome assay. Nat Protoc 2007, 2, 1084–1104. [Google Scholar] [CrossRef]
- Monjazeb, A.M.; Giobbie-Hurder, A.; Lako, A.; Thrash, E.M.; Brennick, R.C.; Kao, K.Z.; Manuszak, C.; Gentzler, R.D.; Tesfaye, A.; Jabbour, S.K.; et al. A Randomized Trial of Combined PD-L1 and CTLA-4 Inhibition with Targeted Low-Dose or Hypofractionated Radiation for Patients with Metastatic Colorectal Cancer. Clin Cancer Res 2021, 27, 2470–2480. [Google Scholar] [CrossRef]
- Azab, B.; Alassaf, A.; Abu-Humdan, A.; Dardas, Z.; Almousa, H.; Alsalem, M.; Khabour, O.; Hammad, H.; Saleh, T.; Awidi, A. Genotoxicity of cisplatin and carboplatin in cultured human lymphocytes: a comparative study. Interdiscip Toxicol 2019, 12, 93–97. [Google Scholar] [CrossRef]
- Nobili, S.; Lavacchi, D.; Perrone, G.; Vicini, G.; Tassi, R.; Landini, I.; Grosso, A.; Roviello, G.; Mazzanti, R.; Santomaggio, C.; et al. Vinorelbine in Non-Small Cell Lung Cancer: Real-World Data From a Single-Institution Experience. Oncol Res 2020, 28, 237–248. [Google Scholar] [CrossRef]
Figure 1.
Quantification of colocalized γH2AX and 53BP1 (γH2AX/53BP1) foci in peripheral blood lymphocytes (PBL) after cisplatin (CDDP), X-rays, or their combination, with a CDDP pretreatment before irradiation. (A) Representative fluorescence microscopic images of γH2AX/53BP1 foci in PBLs at 0.5h and 24h post-irradiation, without or with 24h 10 µg/ml CDDP pretreatment. Quantification of γH2AX/53BP1 foci in PBLs with (B, C) a 1h pulse or (D, E) 24h continuous CDDP pretreatment (B, D) 0.5h or (C, D) 24h after X-ray exposure with 0.5 Gy or 2 Gy, respectively. Foci yields are presented as basal yields for CDDP treatment only (0 Gy total foci), total foci after CDDP followed by X-rays, and radiation-induced foci (total foci - 0 Gy total foci). Data are presented as means and standard deviations from three independent experiments with single samples. Statistics were performed by one-way ANOVA comparison to the mock-treated control (0 µg/ml CDDP, 0 Gy) for 0 Gy total foci and to 0 µg/ml CDDP total foci for radiation-induced foci (* p < 0.05; ** p < 0.01; *** p < 0.001).
Figure 1.
Quantification of colocalized γH2AX and 53BP1 (γH2AX/53BP1) foci in peripheral blood lymphocytes (PBL) after cisplatin (CDDP), X-rays, or their combination, with a CDDP pretreatment before irradiation. (A) Representative fluorescence microscopic images of γH2AX/53BP1 foci in PBLs at 0.5h and 24h post-irradiation, without or with 24h 10 µg/ml CDDP pretreatment. Quantification of γH2AX/53BP1 foci in PBLs with (B, C) a 1h pulse or (D, E) 24h continuous CDDP pretreatment (B, D) 0.5h or (C, D) 24h after X-ray exposure with 0.5 Gy or 2 Gy, respectively. Foci yields are presented as basal yields for CDDP treatment only (0 Gy total foci), total foci after CDDP followed by X-rays, and radiation-induced foci (total foci - 0 Gy total foci). Data are presented as means and standard deviations from three independent experiments with single samples. Statistics were performed by one-way ANOVA comparison to the mock-treated control (0 µg/ml CDDP, 0 Gy) for 0 Gy total foci and to 0 µg/ml CDDP total foci for radiation-induced foci (* p < 0.05; ** p < 0.01; *** p < 0.001).
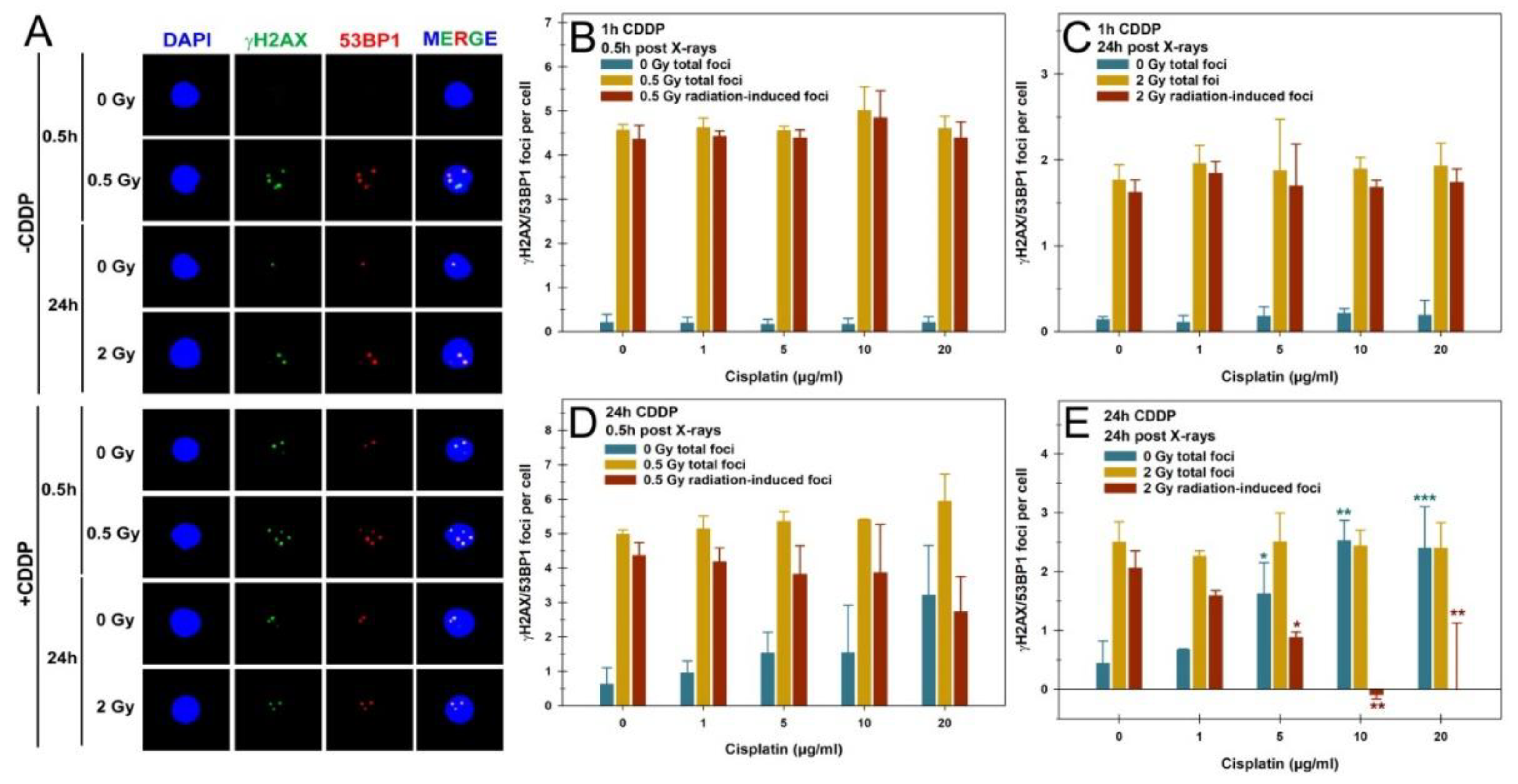
Figure 2.
Apoptosis measurement by AnnexinV-FITC/PI staining and flow cytometry of peripheral blood lymphocytes after (A, B) X-rays, (C, D) cisplatin, or (E, F) their combination, as a 24h continuous CDDP pretreatment before 2 Gy X-rays. (A, C, E) The upper panel shows the fractions of viable, early apoptotic, late apoptotic, and necrotic cells. (B, D, F) The lower panel presents radiation- or cisplatin-induced cell death, summing early apoptotic, late apoptotic, and necrotic cells, based on radiation dose or cisplatin concentration. The data are presented as mean values and standard deviations of three independent experiments with biological duplicates or triplicates. Statistics were performed by one-way ANOVA comparison to the (A-D) mock-treated control (0 Gy, 0 µg/ml CDDP) or (E, F) 2 Gy X-irradiated samples without CDDP pretreatment (* p < 0.05; ** p < 0.01; *** p < 0.001).
Figure 2.
Apoptosis measurement by AnnexinV-FITC/PI staining and flow cytometry of peripheral blood lymphocytes after (A, B) X-rays, (C, D) cisplatin, or (E, F) their combination, as a 24h continuous CDDP pretreatment before 2 Gy X-rays. (A, C, E) The upper panel shows the fractions of viable, early apoptotic, late apoptotic, and necrotic cells. (B, D, F) The lower panel presents radiation- or cisplatin-induced cell death, summing early apoptotic, late apoptotic, and necrotic cells, based on radiation dose or cisplatin concentration. The data are presented as mean values and standard deviations of three independent experiments with biological duplicates or triplicates. Statistics were performed by one-way ANOVA comparison to the (A-D) mock-treated control (0 Gy, 0 µg/ml CDDP) or (E, F) 2 Gy X-irradiated samples without CDDP pretreatment (* p < 0.05; ** p < 0.01; *** p < 0.001).
Figure 3.
Cytokinesis-block micronucleus assay data showing (A, C, E) micronuclei (MN) frequencies in binucleated (BN) cells and (B, D, F) the binucleation index in peripheral blood lymphocytes after (A, B) X-rays, or (C-F) cisplatin (CDDP), and their combination with a (C, D) 4h or (E, F) 24h CDDP pretreatment before 2 Gy X-ray exposure. The data are presented as mean values and standard deviations of three independent experiments with single samples. Statistics were performed by one-way ANOVA comparison to the mock-treated control (0 µg/ml CDDP, 0 Gy) (* p < 0.05; ** p < 0.01; *** p < 0.001). ND, not determined.
Figure 3.
Cytokinesis-block micronucleus assay data showing (A, C, E) micronuclei (MN) frequencies in binucleated (BN) cells and (B, D, F) the binucleation index in peripheral blood lymphocytes after (A, B) X-rays, or (C-F) cisplatin (CDDP), and their combination with a (C, D) 4h or (E, F) 24h CDDP pretreatment before 2 Gy X-ray exposure. The data are presented as mean values and standard deviations of three independent experiments with single samples. Statistics were performed by one-way ANOVA comparison to the mock-treated control (0 µg/ml CDDP, 0 Gy) (* p < 0.05; ** p < 0.01; *** p < 0.001). ND, not determined.
Figure 4.
Cell cycle distribution of peripheral blood lymphocytes by flow cytometry after cisplatin (CDDP) treatment for 24h in the (A) absence or (B) presence of nocodazole. The data are presented as mean values and standard deviations of three independent experiments with biological triplicates. Statistics were performed by one-way ANOVA comparison to mock-treated cells (0 µg/ml CDDP) (* p < 0.05; ** p < 0.01; *** p < 0.001).
Figure 4.
Cell cycle distribution of peripheral blood lymphocytes by flow cytometry after cisplatin (CDDP) treatment for 24h in the (A) absence or (B) presence of nocodazole. The data are presented as mean values and standard deviations of three independent experiments with biological triplicates. Statistics were performed by one-way ANOVA comparison to mock-treated cells (0 µg/ml CDDP) (* p < 0.05; ** p < 0.01; *** p < 0.001).
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).