Submitted:
27 March 2025
Posted:
31 March 2025
You are already at the latest version
Abstract
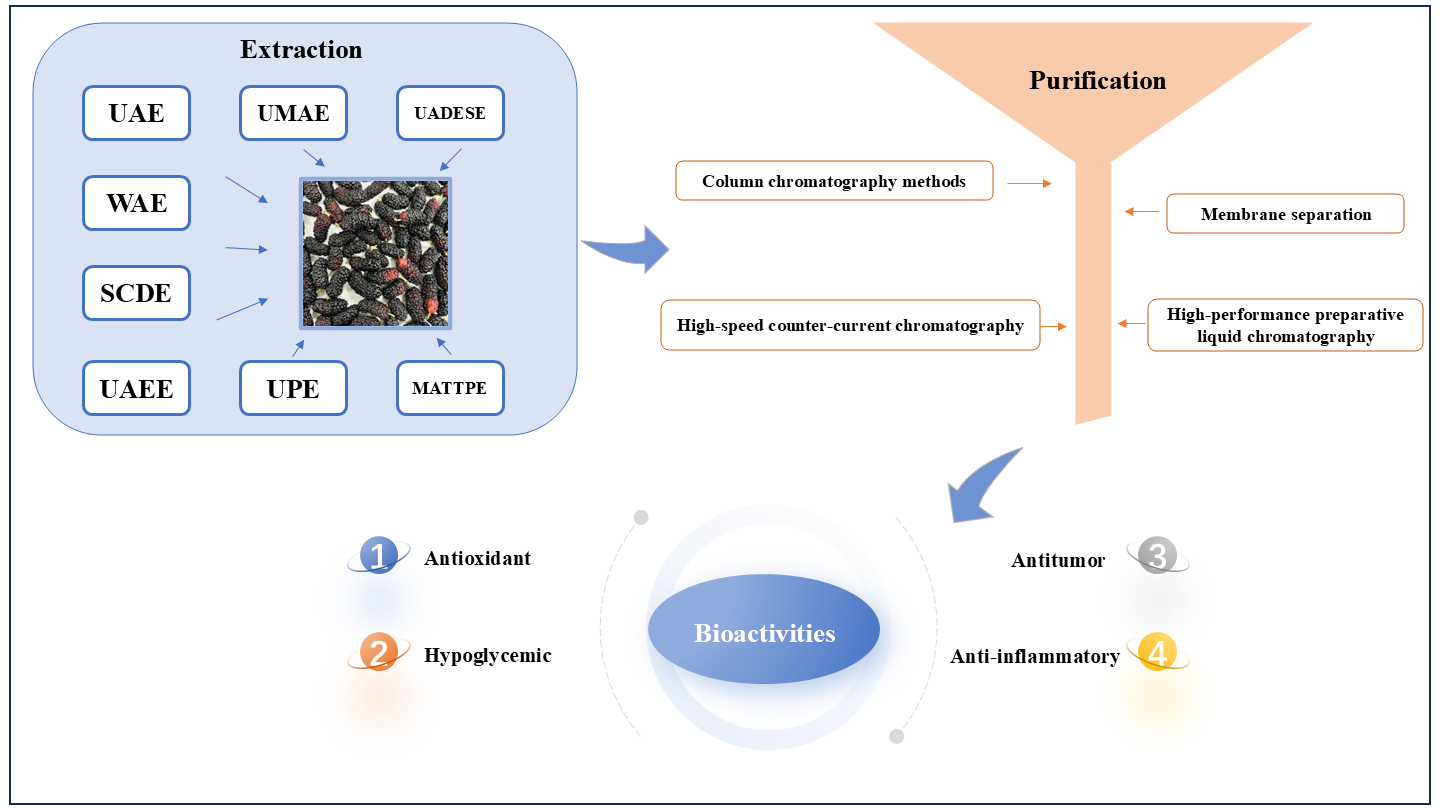
Keywords:
1. Introduction
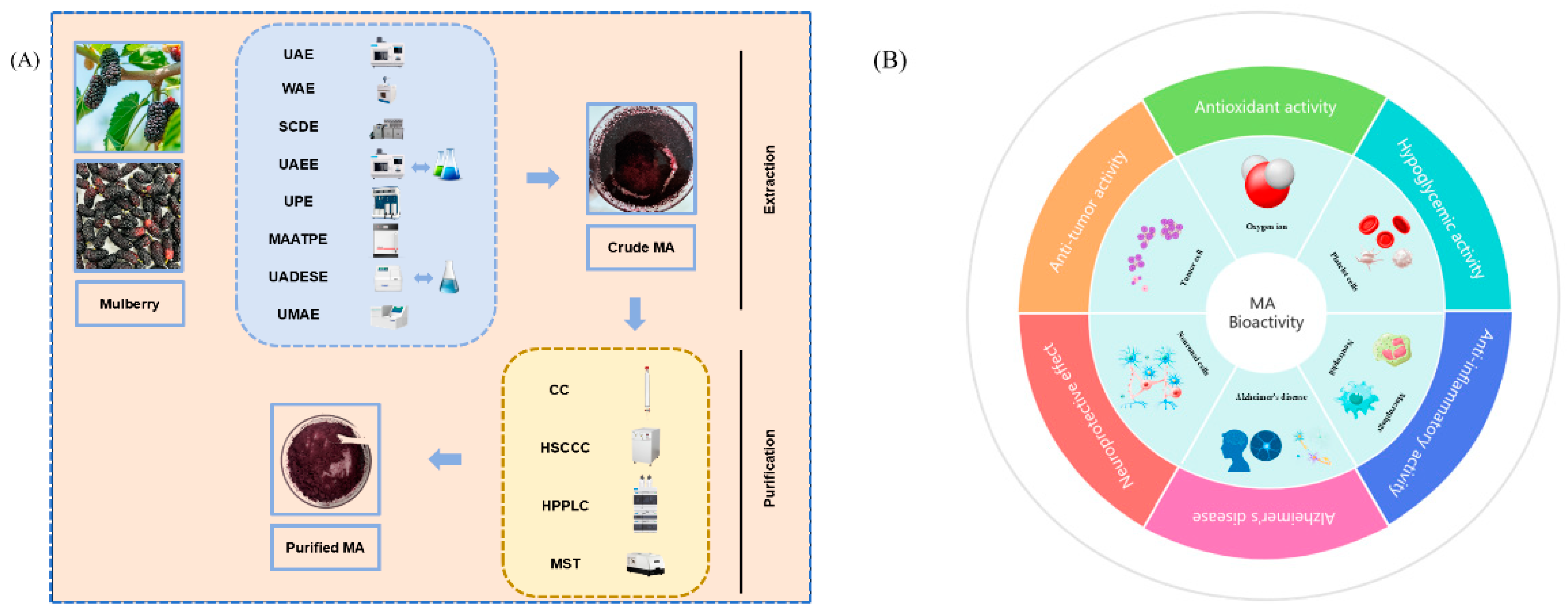
2. Extraction Methods for Mulberry Anthocyanins
2.1. Solvent Extraction Method (SEM)
2.2. Ultrasound-Assisted Extraction
2.3. Microwave-Assisted Extraction
2.4. Ultrahigh-Pressure-Assisted Extraction
2.5. Combined Extraction Methods
3. Purification of Anthocyanins from Mulberries
3.1. Column Chromatography Methods(CC)
3.2. High-Speed Counter-Current Chromatography (HSCCC)
3.3. High-Performance Preparative Liquid Chromatography (HPPLC)
3.4. Membrane Separation Method (MBS)
4. Bioactivities of Mulberry Anthocyanins
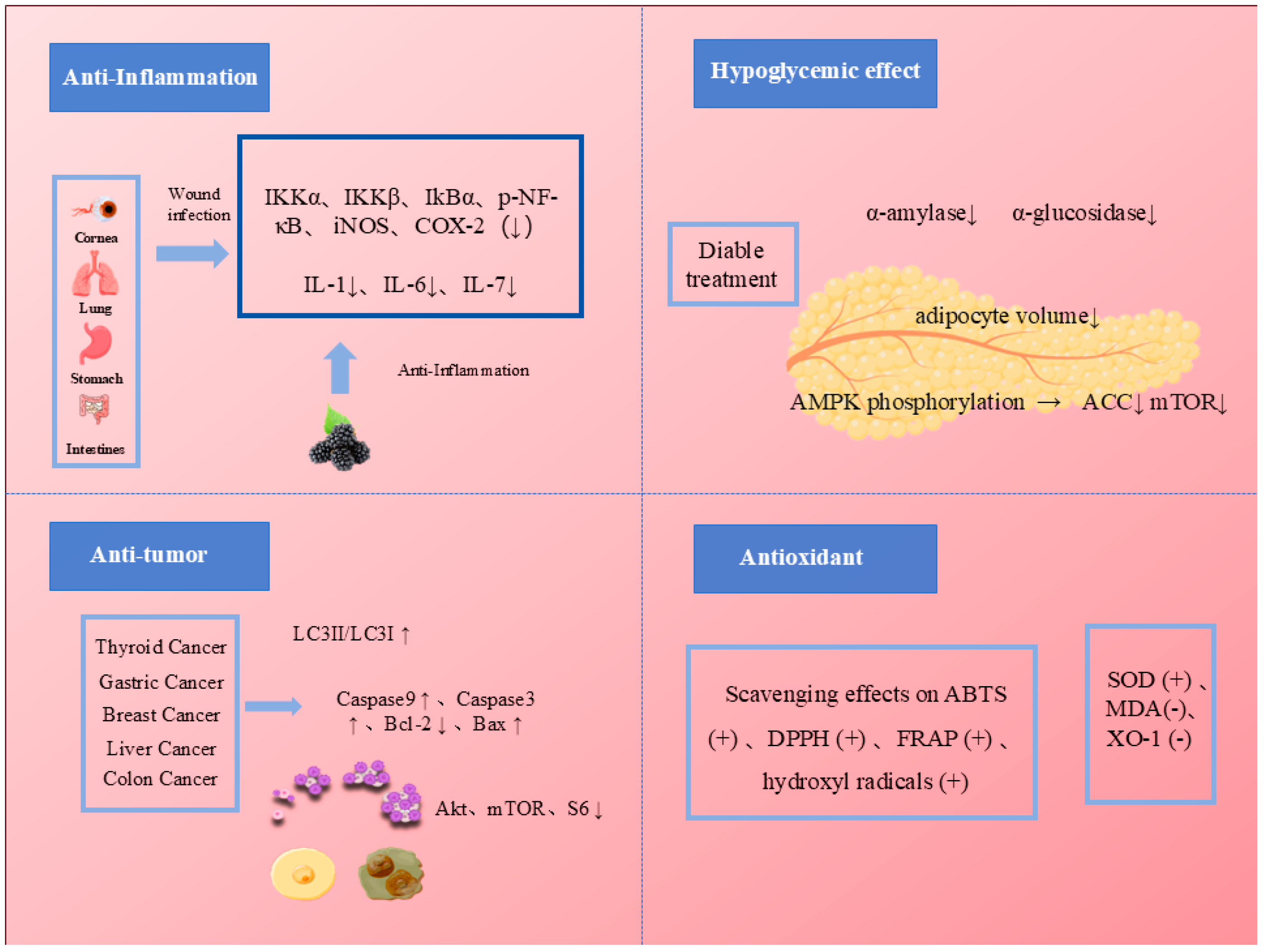
4.1. Antioxidant Activity of Mulberry Anthocyanins
4.2. Hypoglycemic Ability of Mulberry Anthocyanins
4.3. Antitumor Effects of Mulberry Anthocyanins
4.4. Anti-Inflammatory Activity of Mulberry Anthocyanins
4.5. Other Bioactivities of Mulberry Anthocyanins
5. Concluding Remarks and Prospects
Institutional Review Board Statement
Acknowledgements
Conflicts of Interest
References
- Wen, P.; Hu, T.G.; Linhardt, R.J.; Liao, S.T.; Wu, H.; Zou, Y.X. Mulberry: A review of bioactive compounds and advanced processing technology. Trends Food Sci. Technol. 2019, 83, 138–158. [Google Scholar] [CrossRef]
- Zhou, Y.C.; Zhang, R.J.; Wang, J.Y.; Tong, Y.C.; Zhang, J.; Li, Z.Z.; Zhang, H.S.; Abbas, Z.; Si, D.Y.; Wei, X.B. Isolation, Characterization, and Functional Properties of Antioxidant Peptides from Mulberry Leaf Enzymatic Hydrolysates. Antioxidants 2024, 13, 16. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.H.; Liu, F.; Xiong, L. Medicinal parts of mulberry (leaf, twig, root bark, and fruit) and compounds thereof are excellent traditional Chinese medicines and foods for diabetes mellitus. J. Funct. Food. 2023, 106, 21. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kwon, J.W. Anti-inflammatory effect of mulberry anthocyanins on experimental dry eye. Acta Ophthalmol. 2024, 102, 1. [Google Scholar] [CrossRef]
- Liu, X.; Cui, S.Y.; Dan, C.Y.; Li, W.L.; Xie, H.Q.; Li, C.H.; Shi, L.E. Phellinus baumii Polyphenol: A Potential Therapeutic Candidate against Lung Cancer Cells. Int. J. Mol. Sci. 2022, 23, 22. [Google Scholar] [CrossRef]
- Bai, X.; Zhao, X.Y.; Liu, K.F.; Yang, X.T.; He, Q.Z.; Gao, Y.L.; Li, W.N.; Han, W.W. Mulberry Leaf Compounds and Gut Microbiota in Alzheimer’s Disease and Diabetes: A Study Using Network Pharmacology, Molecular Dynamics Simulation, and Cellular Assays. Int. J. Mol. Sci. 2024, 25, 27. [Google Scholar] [CrossRef]
- Mo, R.L.; Zhang, N.; Li, J.X.; Jin, Q.; Zhu, Z.X.; Dong, Z.X.; Li, Y.; Zhang, C.; Yu, C. Transcriptomic Analysis Provides Insights into Anthocyanin Accumulation in Mulberry Fruits. Horticulturae 2022, 8, 13. [Google Scholar] [CrossRef]
- Tan, J.Q.; Han, Y.M.; Han, B.; Qi, X.M.; Cai, X.; Ge, S.Q.; Xue, H.K. Extraction and purification of anthocyanins: A review. J. Agric. Food Res. 2022, 8, 7. [Google Scholar] [CrossRef]
- Dong, Y.H.; Zhong, W.T.; Yang, C.M.; Zhang, Y.Z.; Yang, D.S. Study on anthocyanins from Lycium ruthenicum Murr via ultrasonic microwave synergistic extraction and its antioxidant properties. Front. Sustain. Food Syst. 2022, 6, 16. [Google Scholar] [CrossRef]
- Xie, J.; Xu, Y.; Shishir, M.R.I.; Zheng, X.; Chen, W. Green extraction of mulberry anthocyanin with improved stability using β-cyclodextrin. Journal of the Science of Food and Agriculture 2018, 99, 2494–2503. [Google Scholar] [CrossRef]
- Chen, C.; Mokhtar, R.A.M.; Sani, M.S.A.; Noor, N. The Effect of Maturity and Extraction Solvents on Bioactive Compounds and Antioxidant Activity of Mulberry (Morus alba) Fruits and Leaves. Molecules 2022, 27, 20. [Google Scholar] [CrossRef] [PubMed]
- Suriyaprom, S.; Kaewkod, T.; Promputtha, I.; Desvaux, M.; Tragoolpua, Y.J.P. Evaluation of antioxidant and antibacterial activities of white mulberry (Morus alba L.) fruit extracts. 2021, 10, 2736. [Google Scholar] [CrossRef] [PubMed]
- Budiman, A.; Praditasari, A.; Rahayu, D.; Aulifa, D.L.J.J.o.P.; Sciences, B. Formulation of antioxidant gel from black mulberry fruit extract (Morus nigra L.). 2019, 11, 216–222. [Google Scholar] [CrossRef]
- Saensouk, S.; Senavongse, R.; Papayrata, C.; Chumroenphat, T.J.H. Evaluation of color, phytochemical compounds and antioxidant activities of mulberry fruit (Morus alba L.) during ripening. Horticulturae 2022, 8, 1146. [Google Scholar] [CrossRef]
- Zhang, B.; Jiang, X.; Huang, G.; Xin, X.; Attaribo, T.; Zhang, Y.; Gui, Z.J.S. Preparation and characterization of methylated anthocyanins from mulberry fruit with iodomethane as a donor. Anthocyanins 2019, 59, 106–116. [Google Scholar]
- Fang, J.L.; Jia, S.S.; Lin, Y.; Yuan, K.; Jin, S.H. Extraction, Purification, Content Analysis and Hypoglycemic Effect of Mulberry marc Anthocyanin. Pharmacognosy Magazine 2020, 16, 68–75. [Google Scholar] [CrossRef]
- Rocha, R.; Pinela, J.; Abreu, R.M.V.; Añibarro-Ortega, M.; Pires, T.; Saldanha, A.L.; Alves, M.J.; Nogueira, A.; Ferreira, I.; Barros, L. Extraction of Anthocyanins from Red Raspberry for Natural Food Colorants Development: Processes Optimization and In Vitro Bioactivity. Processes 2020, 8, 19. [Google Scholar] [CrossRef]
- Chen, Z.L.; Ma, J.; Li, P.; Wen, B.; Wang, Y.; Ma, Y.H.; Huang, W.Y. Preparation of hypoglycemic anthocyanins from mulberry (Fructus mori) fruits by ultrahigh pressure extraction. Innovative Food Science & Emerging Technologies 2023, 84, 11. [Google Scholar] [CrossRef]
- Zannou, O.; Oussou, K.F.; Chabi, I.B.; Awad, N.M.; Aïssi, M.V.; Goksen, G.; Mortas, M.; Oz, F.; Proestos, C.; Kayodé, A.P.J.N. Nanoencapsulation of Cyanidin 3-O-Glucoside: Purpose, technique, bioavailability, and stability. 2023, 13, 617. [CrossRef]
- Li, Y.; Tao, F.; Wang, Y.; Cui, K.; Cao, J.; Cui, C.; Nan, L.; Li, Y.; Yang, J.; Wang, Z. Process optimization for enzymatic assisted extraction of anthocyanins from the mulberry wine residue. IOP Conference Series: Earth and Environmental Science 2020, 559. [CrossRef]
- Zhang, L.; Fan, G.; Khan, M.A.; Yan, Z.; Beta, T. Ultrasonic-assisted enzymatic extraction and identification of anthocyanin components from mulberry wine residues. Food Chem 2020, 323, 126714. [Google Scholar] [CrossRef]
- Bi, Y.; Chi, X.; Zhang, R.; Lu, Y.; Wang, Z.; Dong, Q.; Ding, C.; Yang, R.; Jiang, L. Highly efficient extraction of mulberry anthocyanins in deep eutectic solvents: Insights of degradation kinetics and stability evaluation. Innovative Food Science & Emerging Technologies 2020, 66. [CrossRef]
- Guo, N.; Ping, K.; Jiang, Y.W.; Wang, L.T.; Niu, L.J.; Liu, Z.M.; Fu, Y.J. Natural deep eutectic solvents couple with integrative extraction technique as an effective approach for mulberry anthocyanin extraction. Food Chem 2019, 296, 78–85. [Google Scholar] [CrossRef]
- Wang, D.Q.; Cui, H.P.; Zong, K.L.; Hu, H.C.; Yang, J.T. Extraction of functional natural products employing microwave-assisted aqueous two-phase system: application to anthocyanins extraction from mulberry fruits. Prep. Biochem. Biotechnol. 2024, 54, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Yan, X.; Li, X.; Liu, S.; Liu, C.J.C.C. Optimization of ultrasonic-microwave assisted extraction of anthocyanins from mulberry and theirs antioxidant activities. 2020, 45, 172–178. [CrossRef]
- Johnson, J.; Collins, T.; Walsh, K.; Naiker, M. Solvent extractions and spectrophotometric protocols for measuring the total anthocyanin, phenols and antioxidant content in plums. Chem. Pap. 2020, 74, 4481–4492. [Google Scholar] [CrossRef]
- Muangrat, R.; Williams, P.T.; Saengcharoenrat, P. Subcritical solvent extraction of total anthocyanins from dried purple waxy corn: Influence of process conditions. J. Food Process Preserv. 2017, 41, 8. [Google Scholar] [CrossRef]
- Budiman, A.; Praditasari, A.; Rahayu, D.; Aulifa, D.L. Formulation of Antioxidant Gel from Black Mulberry Fruit Extract (Morus nigra L.). J Pharm Bioallied Sci 2019, 11, 216–222. [Google Scholar] [CrossRef]
- Yusoff, I.M.; Taher, Z.M.; Rahmat, Z.; Chua, L.S. A review of ultrasound-assisted extraction for plant bioactive compounds: Phenolics, flavonoids, thymols, saponins and proteins. Food Res. Int. 2022, 151, 17. [Google Scholar] [CrossRef]
- López-Salazar, H.; Camacho-Díaz, B.H.; Ocampo, M.L.A.; Jiménez-Aparicio, A.R. Microwave-assisted Extraction of Functional Compounds from Plants: A Review. BioResources 2023, 18, 6614–6638. [Google Scholar] [CrossRef]
- Liu, W.; Yang, C.; Zhou, C.; Wen, Z.; Dong, X. An improved microwave-assisted extraction of anthocyanins from purple sweet potato in favor of subsequent comprehensive utilization of pomace. Food and Bioproducts Processing 2019, 115, 1–9. [Google Scholar] [CrossRef]
- Chen, R.; Meng, F.; Zhang, S.; Liu, Z. Effects of ultrahigh pressure extraction conditions on yields and antioxidant activity of ginsenoside from ginseng. Separation and Purification Technology 2009, 66, 340–346. [Google Scholar] [CrossRef]
- Xi, J. Effect of High Pressure Processing on the Extraction of Lycopene in Tomato Paste Waste. Chemical Engineering & Technology 2006, 29, 736–739. [Google Scholar] [CrossRef]
- Khan, S.A.; Aslam, R.; Makroo, H.A. High pressure extraction and its application in the extraction of bio-active compounds: A review. Journal of Food Process Engineering 2018, 42. [Google Scholar] [CrossRef]
- Lee, H.S.; Lee, H.J.; Yu, H.J.; Ju do, W.; Kim, Y.; Kim, C.T.; Kim, C.J.; Cho, Y.J.; Kim, N.; Choi, S.Y.; et al. A comparison between high hydrostatic pressure extraction and heat extraction of ginsenosides from ginseng (Panax ginseng CA Meyer). J Sci Food Agric 2011, 91, 1466–1473. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Deng, Y.; Yang, Y.; Dong, H.; Li, L.; Chen, G. Comparison of different drying pretreatment combined with ultrasonic-assisted enzymolysis extraction of anthocyanins from Lycium ruthenicum Murr. Ultrasonics Sonochemistry 2024, 107, 106933. [Google Scholar] [CrossRef] [PubMed]
- Umego, E.C.; He, R.; Ren, W.; Xu, H.; Ma, H. Ultrasonic-assisted enzymolysis: Principle and applications. Process Biochemistry 2021, 100, 59–68. [Google Scholar] [CrossRef]
- Zainal-Abidin, M.H.; Hayyan, M.; Hayyan, A.; Jayakumar, N.S. New horizons in the extraction of bioactive compounds using deep eutectic solvents: A review. Analytica Chimica Acta 2017, 979, 1–23. [Google Scholar] [CrossRef]
- Bi, Y.; Chi, X.; Zhang, R.; Lu, Y.; Wang, Z.; Dong, Q.; Ding, C.; Yang, R.; Jiang, L. Highly efficient extraction of mulberry anthocyanins in deep eutectic solvents: Insights of degradation kinetics and stability evaluation. Innovative Food Science & Emerging Technologies 2020, 66, 102512. [Google Scholar] [CrossRef]
- Lin, S.X.; Meng, X.J.; Tan, C.; Tong, Y.Q.; Wan, M.Z.; Wang, M.Y.; Zhao, Y.; Deng, H.T.; Kong, Y.W.; Ma, Y. Composition and antioxidant activity of anthocyanins from Aronia melanocarpa extracted using an ultrasonic-microwave-assisted natural deep eutectic solvent extraction method. Ultrasonics Sonochemistry 2022, 89, 15. [Google Scholar] [CrossRef]
- Yang, D.; Li, M.M.; Wang, W.J.; Zheng, G.D.; Yin, Z.P.; Chen, J.G.; Zhang, Q.F. Separation and purification of anthocyanins from Roselle by macroporous resins. LWT-Food Sci. Technol. 2022, 161, 8. [Google Scholar] [CrossRef]
- Wang, X.Y.; Su, J.Q.; Chu, X.L.; Zhang, X.Y.; Kan, Q.B.; Liu, R.X.; Fu, X. Adsorption and Desorption Characteristics of Total Flavonoids from Acanthopanax senticosus on Macroporous Adsorption Resins. Molecules 2021, 26, 19. [Google Scholar] [CrossRef]
- Yang, L.F.; Nie, W.; Cui, Y.P.; Yue, F.L.; Fan, X.T.; Sun, R.Y. Purification with macroporous resin and antioxidant activity of polyphenols from sweet potato leaves. Chem. Pap. 2024, 78, 181–188. [Google Scholar] [CrossRef]
- Huang, H.; Xie, C.; Wu, C.; Li, T.; Li, X.; Zhou, D.; Yang, X.; Fan, G. Purification of mulberry anthocyanins by macroporous resin and its protective effect against H2O2-induced oxidative stress in vascular endothelial cells. Food Bioscience 2024. [CrossRef]
- Li, Y.T.; Huang, T.; Wang, J.Z.; Yan, C.H.; Gong, L.C.; Wu, F.A.; Wang, J. Efficient acquisition of high-purity cyanidin-3-O-glucoside from mulberry fruits: An integrated process of ATPS whole-cell transformation and semi-preparative HPLC purification. Food Chem. 2023, 404, 11. [Google Scholar] [CrossRef]
- Liao, Z.; Zhang, X.; Chen, X.; Battino, M.; Giampieri, F.; Bai, W.; Tian, L. Recovery of value-added anthocyanins from mulberry by a cation exchange chromatography. Curr Res Food Sci 2022, 5, 1445–1451. [Google Scholar] [CrossRef] [PubMed]
- Spórna-Kucab, A.; Jerz, G.; Kumorkiewicz-Jamro, A.; Tekieli, A.; Wybraniec, S. High-speed countercurrent chromatography for isolation and enrichment of betacyanins from fresh and dried leaves of Atriplex hortensis L. var. “Rubra”. J. Sep. Sci. 2021, 44, 4222–4236. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Yang, X.; Wang, N.N.; Li, H.M.; Zhao, J.Y.; Li, Y.L. Separation of six compounds including two n-butyrophenone isomers and two stibene isomers from Rheum tanguticum Maxim by recycling high speed counter-current chromatography and preparative high-performance liquid chromatography. J. Sep. Sci. 2018, 41, 10. [Google Scholar] [CrossRef]
- Chen, Y.; Du, F.; Wang, W.; Li, Q.; Zheng, D.H.; Zhang, W.J.; Zhao, T.; Mao, G.H.; Feng, W.W.; Wu, X.Y.; et al. Large-scale isolation of high-purity anthocyanin monomers from mulberry fruits by combined chromatographic techniques. J. Sep. Sci. 2017, 40, 3506–3512. [Google Scholar] [CrossRef]
- Zou, H.; Ma, Y.; Xu, Z.Z.; Liao, X.J.; Chen, A.L.; Yang, S.M. Isolation of strawberry anthocyanins using high-speed counter-current chromatography and the copigmentation with catechin or epicatechin by high pressure processing. Food Chem. 2018, 247, 81–88. [Google Scholar] [CrossRef]
- Nie, J.; Chen, D.; Ye, J.; Lu, Y.; Dai, Z. Preparative separation of three terpenoids from edible brown algae Sargassum fusiforme by high-speed countercurrent chromatography combined with preparative high-performance liquid chromatography. Algal Research 2021, 59, 102449. [Google Scholar] [CrossRef]
- Li, S.; Zhang, H.; Huai, J.; Wang, H.; Li, S.; Zhuang, L.; Zhang, J. An online preparative high-performance liquid chromatography system with enrichment and purification modes for the efficient and systematic separation of Panax notoginseng saponins. Journal of Chromatography A 2023, 1709, 464378. [Google Scholar] [CrossRef]
- Cissé, M.; Vaillant, F.; Pallet, D.; Dornier, M. Selecting ultrafiltration and nanofiltration membranes to concentrate anthocyanins from roselle extract (Hibiscus sabdariffa L). Food Res. Int. 2011, 44, 2607–2614. [Google Scholar] [CrossRef]
- Martín, J.; Díaz-Montaña, E.J.; Asuero, A.G. Recovery of Anthocyanins Using Membrane Technologies: A Review. Crit. Rev. Anal. Chem. 2018, 48, 143–175. [Google Scholar] [CrossRef]
- Ockermann, P.; Lizio, R.; Hansmann, J. Healthberry 865® and a Subset of Its Single Anthocyanins Attenuate Oxidative Stress in Human Endothelial In Vitro Models. Nutrients 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, L.; Chai, T.; Xu, H.; Du, H.-y.; Jiang, Y. Mulberry leaf multi-components exert hypoglycemic effects through regulation of the PI-3K/Akt insulin signaling pathway in type 2 diabetic rats. Journal of Ethnopharmacology 2024, 319, 117307. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Qi, X.M.; Liu, Y.; Guo, J.; Zhu, R.Y.; Chen, W.; Zheng, X.D.; Yu, T. Dietary supplementation with purified mulberry (Morus australis Poir) anthocyanins suppresses body weight gain in high-fat diet fed C57BL/6 mice. Food Chem. 2013, 141, 482–487. [Google Scholar] [CrossRef]
- Yan, F.J.; Zheng, X.D. Anthocyanin-rich mulberry fruit improves insulin resistance and protects hepatocytes against oxidative stress during hyperglycemia by regulating AMPK/ACC/mTOR pathway. J. Funct. Food. 2017, 30, 270–281. [Google Scholar] [CrossRef]
- Yan, F.J.; Dai, G.H.; Zheng, X.D. Mulberry anthocyanin extract ameliorates insulin resistance by regulating PI3K/AKT pathway in HepG2 cells and db/db mice. J. Nutr. Biochem. 2016, 36, 68–80. [Google Scholar] [CrossRef]
- Long, H.L.; Zhang, F.F.; Wang, H.L.; Yang, W.S.; Hou, H.T.; Yu, J.K.; Liu, B. Mulberry anthocyanins improves thyroid cancer progression mainly by inducing apoptosis and autophagy cell death. Kaohsiung J. Med. Sci. 2018, 34, 255–262. [Google Scholar] [CrossRef]
- Huang, H.P.; Chang, Y.C.; Wu, C.H.; Hung, C.N.; Wang, C.J. Anthocyanin-rich Mulberry extract inhibit the gastric cancer cell growth in vitro and xenograft mice by inducing signals of p38/p53 and c-jun. Food Chem. 2011, 129, 1703–1709. [Google Scholar] [CrossRef]
- Cho, E.; Chung, E.Y.; Jang, H.-Y.; Hong, O.-Y.; Chae, H.S.; Jeong, Y.-J.; Kim, S.-Y.; Kim, B.-S.; Yoo, D.J.; Kim, J.-S.J.A.-C.A.i.M.C. Anti-cancer effect of cyanidin-3-glucoside from mulberry via caspase-3 cleavage and DNA fragmentation in vitro and in vivo. 2017, 17, 1519–1525. [CrossRef]
- Mo, J.L.; Ni, J.D.; Zhang, M.; Xu, Y.; Li, Y.T.; Karim, N.; Chen, W. Mulberry Anthocyanins Ameliorate DSS-Induced Ulcerative Colitis by Improving Intestinal Barrier Function and Modulating Gut Microbiota. Antioxidants 2022, 11, 17. [Google Scholar] [CrossRef]
- Jung, S.; Lee, M.S.; Chang, E.; Kim, C.T.; Kim, Y. Mulberry (Morus alba L.) Fruit Extract Ameliorates Inflammation via Regulating MicroRNA-21/132/143 Expression and Increases the Skeletal Muscle Mitochondrial Content and AMPK/SIRT Activities. Antioxidants 2021, 10, 17. [Google Scholar] [CrossRef]
- Lee, D.; Lee, S.R.; Kang, K.S.; Kim, K.H. Bioactive Phytochemicals from Mulberry: Potential Anti-Inflammatory Effects in Lipopolysaccharide-Stimulated RAW 264.7 Macrophages. Int. J. Mol. Sci. 2021, 22, 13. [Google Scholar] [CrossRef]
- Khalifa, I.; Xia, D.; Dutta, K.; Peng, J.M.; Jia, Y.Y.; Li, C.M. Mulberry anthocyanins exert anti-AGEs effects by selectively trapping glyoxal and structural-dependently blocking the lysyl residues of β-lactoglobulins. Bioorganic Chem. 2020, 96, 11. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.J.; Chen, X.A.; Zheng, X.D. Protective effect of mulberry fruit anthocyanin on human hepatocyte cells (LO2) and Caenorhabditis elegans under hyperglycemic conditions. Food Res. Int. 2017, 102, 213–224. [Google Scholar] [CrossRef] [PubMed]
- You, Y.L.; Liang, C.; Han, X.; Guo, J.L.; Ren, C.L.; Liu, G.J.; Huang, W.D.; Zhan, J.C. Mulberry anthocyanins, cyanidin 3-glucoside and cyanidin 3-rutinoside, increase the quantity of mitochondria during brown adipogenesis. J. Funct. Food. 2017, 36, 348–356. [Google Scholar] [CrossRef]
- Shih, P.H.; Chan, Y.C.; Liao, J.W.; Wang, M.F.; Yen, G.C. Antioxidant and cognitive promotion effects of anthocyanin-rich mulberry (Morus atropurpurea L.) on senescence-accelerated mice and prevention of Alzheimer’s disease. J. Nutr. Biochem. 2010, 21, 598–605. [Google Scholar] [CrossRef]
| Source | Extraction methods | Extraction solvent | Extractioncondition | Yield | Reference |
|---|---|---|---|---|---|
| 4 varieties of mulberry | SEM | β-CD, Hβ-CD | Temperature for extraction was set at 20 °C, duration was about 45 minutes, and β-CD concentration was 45 g/L. | 1.2 g/kg | [10] |
| mulberry | SEM | methanol, ethanol, acetone | the optimal condition is 60% ethanol extraction | Not reported | [11] |
| White mulberry | SEM | ethanol | 95% ethanol, extraction temperature 45 °C, time 3 hours | Not reported | [12] |
| mulberry | SEM | ethanol | 96% ethanol, extraction temperature: 30 °C | Not reported | [13] |
| mulberry | UAE | methanol | 70% methanol, 1:55 ratio, extraction time 60 min | 6.479% | [14] |
| mulberry | UAE | methanol oxidation | Ultrasound-assisted extraction with a mixture of methanol and 1.0 N HCl in an 85:15 ratio. | Not reported | [15] |
| mulberry | UAE | ethanol | 50% ethanol (pH 2.0 ± 0.1), ultrasonic extraction three times 30 °C extraction temperature: |
Not reported | [16] |
| Red raspberry | UAE | ethanol-citric acid | ultrasonic power: 466 W, time 16 min, material-liquid ratio is 4:1 | 9.0 ± 0.8 mg/g | [17] |
| mulberry | UPE | ethanol | extraction pressure is 429.52 MPa, and the material-to-liquid ratio is 12.371:1. | 1.97 ± 0.01 mg/g | [18] |
| mulberry | MAE | Methanol oxidation | 59.6% methanol, 425W microwave power, 25 (v/w) material-to-liquid ratio, extraction time 132 s | 54.72 mg/g | [19] |
| mulberry wine residue | EAE | ethanol | Solid-to-liquid ratio 1:20, extract 58 min at pH 5.9 and 45 °C. | 6.040 mg/g | [20] |
| mulberry wine residue | UAEE | ethanol oxidation | extraction temperature 52 °C, 0.22% pectinase, 315 W ultrasonic power, 94 min | 6.03 mg/g | [21] |
| mulberry | UADESE | ChCl/HL (1:2)-water (80/20v/v) | temperature 57.24 °C, extraction time 31.54 min, material-to-liquid ratio 10.76:1. | 6.84± 0.21 mg/g | [22] |
| mulberry | NADES | the molar ratio of chlorine, citric acid, and glucose is 1:1:1 (which includes 30% distilled water) | The extraction process lasts for 30 minutes, with a solid-to-liquid ratio of 22 mL per gram, under a negative pressure of -0.08 MPa. | 6.05 mg/g | [23] |
| mulberry | MAATPE | ethanol/ammonium sulfate | 39% ethanol (w/w), 13% ammonium sulfate (w/w), material-to-liquid ratio 45:1, microwave power 480W, microwave time 3 min | Not reported | [24] |
| mulberry | UMAE | ethanol | Microwave power is configured to 150W, ultrasonic power to 360W, extraction time to 2 minutes, with a 1:70 ratio of material to solvent. | 13.57±1.30 mg/g | [25] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




