Submitted:
27 March 2025
Posted:
27 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Catalyst Preparation
2.2. HDN Activity Evaluation Device
2.3. Reaction Materials and Conditions
2.4. Catalyst Characterization
2.4.1. X-Ray Diffractometer
2.4.2. Nitrogen Adsorption and Desorption Apparatus
2.5. Hydrogenation Product Analysis
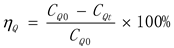
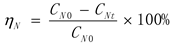
3. Result
3.1. Carrier and Catalyst XRD Analysis
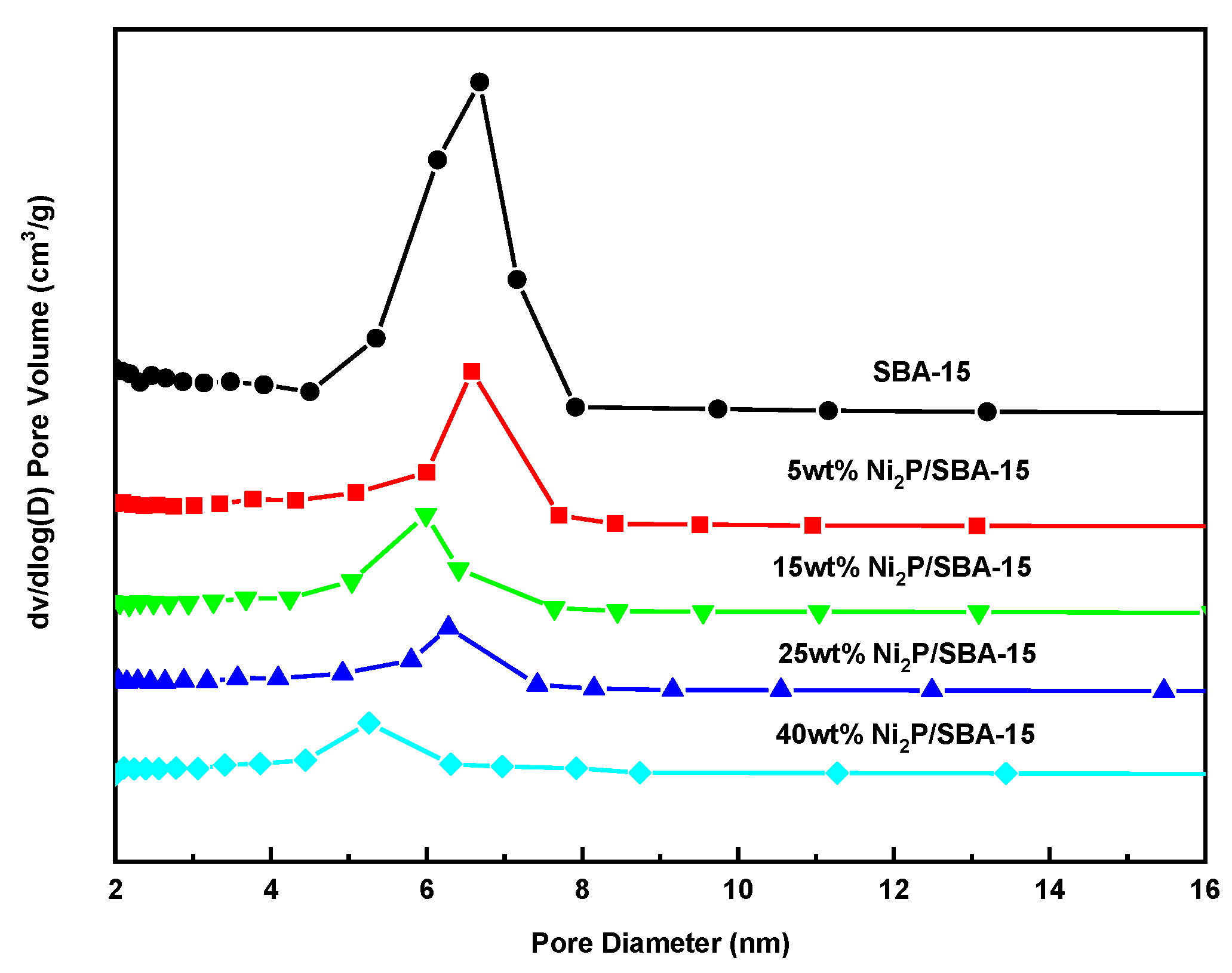
3.2. Analysis of Carrier and Catalyst Pore Structure
3.3. Study of the Denitrogenation Reaction of Quinoline
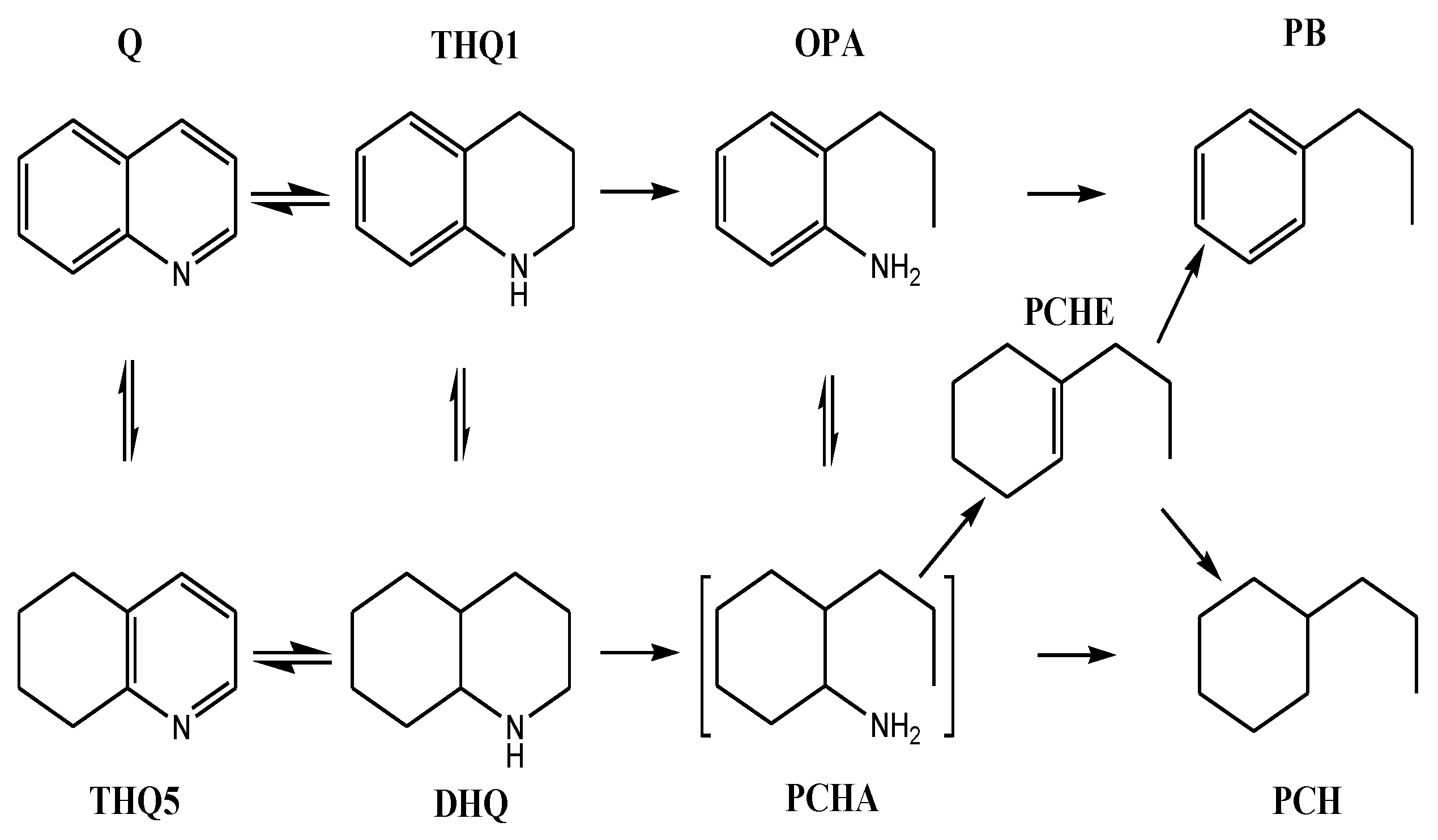
3.3.1. Hydrogenation Pathway of Quinoline
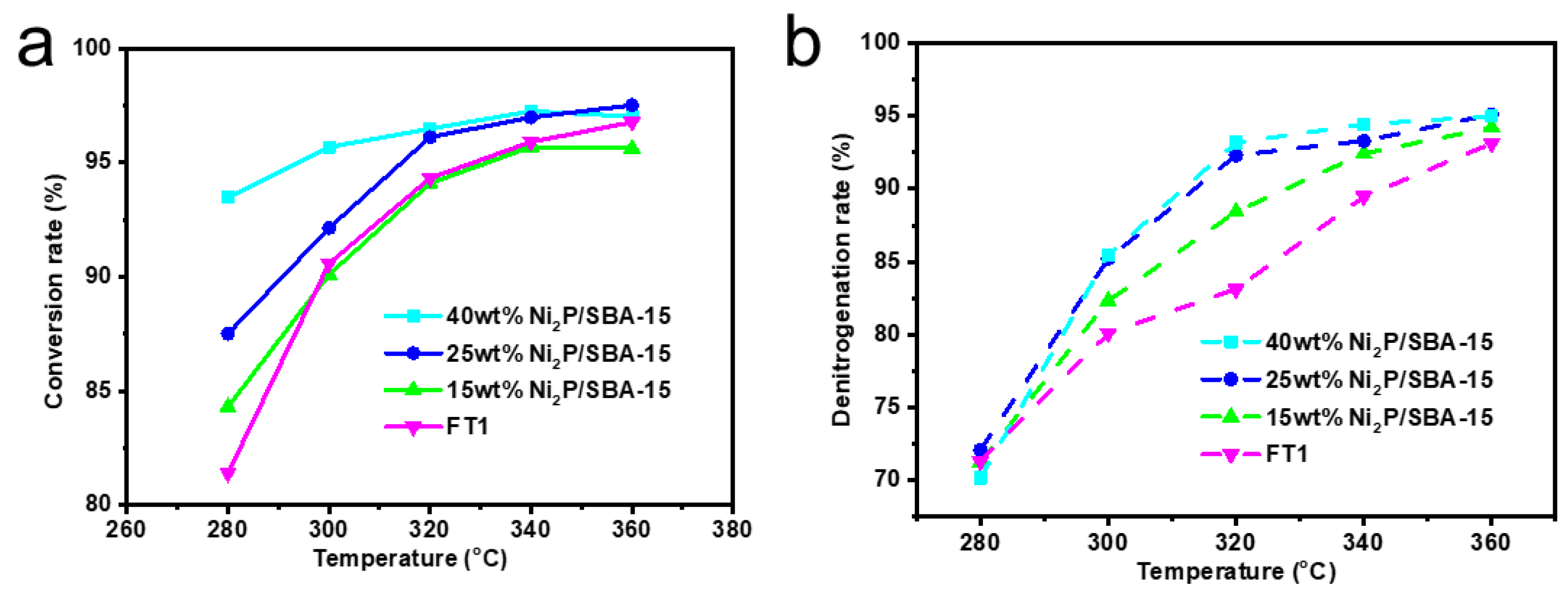
3.3.2. Effect of Different Temperatures on Conversion and Denitrogenation Rates
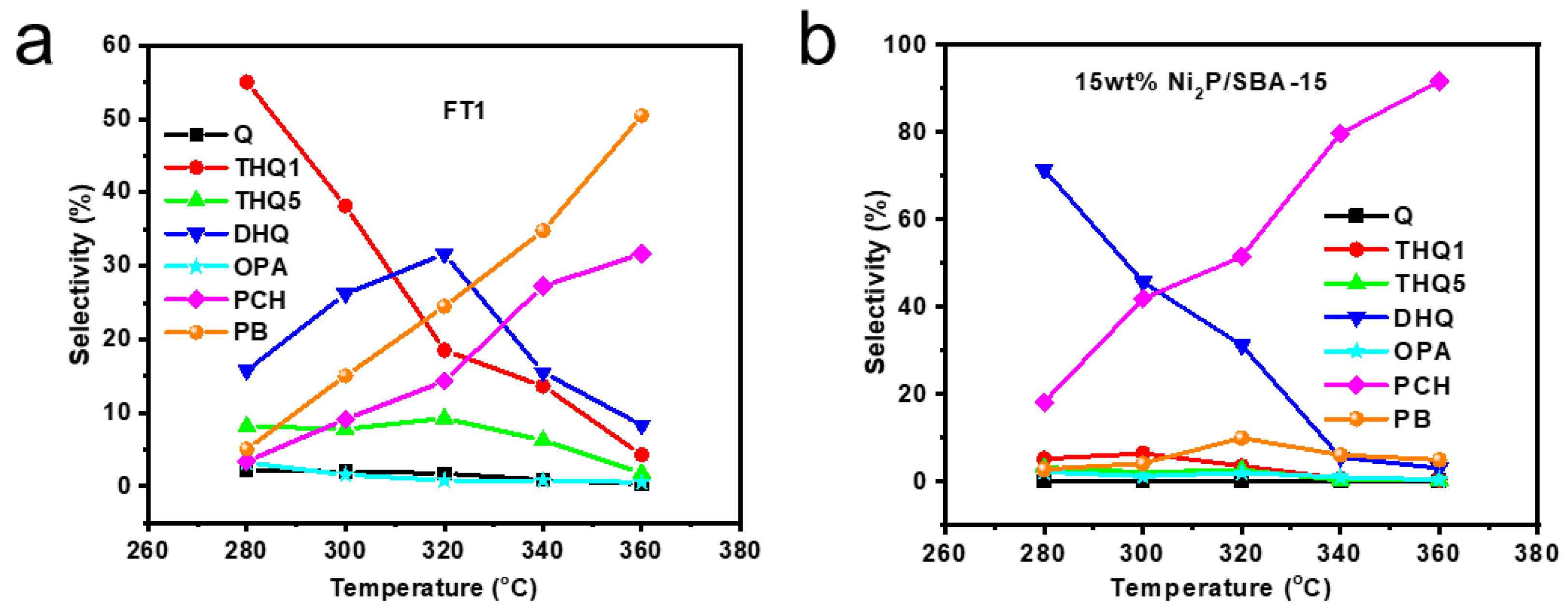
3.3.3. Selectivity Study of Quinoline Hydrogenation Products
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Murti S D S, Sakanishi K, Okuma O. Detailed characterization of heteroatom-containing molecules in light distillates derived from Tanito Harum coal and its hydrotreated oil. Fuel 2002, 81, 2241–2248. [Google Scholar] [CrossRef]
- Murti S D S, Choi K H, Sakanishi K, Okuma O, Korai Y, Mochida I. Analysis and removal of heteroatom containing species in coal liquid distillate over NiMo catalysts. Fuel 2005, 84, 135–142. [Google Scholar] [CrossRef]
- Wei Y, Liu D S, Yang P, et al. Removal of Basic Nitrogen of Diesel Oil by Chemical Method. Journal of Fushun Petroleum Institute 2002, 22, 1–4. [Google Scholar]
- Xiang C, Chai Y, Liu Y; et al. Mutual Influence of Hydrodesulfurization of Dibenzothiophene and Hydrodenitrogenation of Quinoline over NiMoS/γ-Al2O3 Catalyst. Chinese Journal of Catalysis 2008, 24, 151–157. [Google Scholar]
- Qing-Jie L I, Zhao Z F, Feng-Yan L I; et al. Study on the preparation of γ-alumina-supported molybdenum phosphide hydrofining catalysts. Journal of Fuel Chemistry and Technology 2006, 34, 126–128. [Google Scholar]
- Oyama S, T. Novel catalysts for advanced hydroprocessing: Transition metal phosphides. J. Catal. 2003, 216, 343–352. [Google Scholar] [CrossRef]
- Sun F, X. Advance in the Research on Hydrodesulfurization and Hydrodenitrogenation on Transition Metal Phosphides[J]. Acta Petrolei Sinica 2005, 21, 1–11. [Google Scholar]
- Liu L H, Liu S Q, Chai Y M, et al. Preparation mechanism and hydrodenitrogenation performance of nickel phosphide catalyst. Journal of Fuel Chemistry and Technology 2013, 41, 335–340. [Google Scholar]
- Robinson W R A M, Van Gestel J N M, Koranyi T I. Phosphorus promotion of Ni (Co)-containing Mo-free catalysts in quinoline hydrodenitrogenation. J. Catal. 1996, 161, 539–550. [Google Scholar] [CrossRef]
- Li W, Dhandapani B, Oyama S T. Molybdenum phosphide: A novel catalyst for Hydrodenitrogenation. Chem. Lett. 1998, 27, 207–208. [Google Scholar] [CrossRef]
- Stinner C, Prins R, Weber T. Formation, structure, and HDN activity of unsupported molybdenum phosphides. J. Catal. 2000, 191, 438–444. [Google Scholar] [CrossRef]
- Zhai Q, Cai J, Hui Y U; et al. SYNTHESIS OF SBA-15 MOLECULAR SIEVE[J]. Journal of the Chinese Ceramic Society 2006, 34, 385–388. [Google Scholar]
- Zheng X C, Yuan C Y, Zhao W P; et al. Synthesis and Characterization of Mesoporous Molecular Sieves SBA-15[J]. Journal of Zhengzhou University 2008, 40, 101–106. [Google Scholar]
- Oyama S T, Wang X, Lee Y-K, Bando K, Requejo F G. Effect of phosphorus content in nickel phosphide catalysts studied by XAFS and other techniques. J. Catal. 2002, 210, 207–217. [Google Scholar] [CrossRef]
- Korányi T I, Vít Z, Poduval D G. SBA-15-supported nickel phosphide hydrotreating catalysts. J. Catal. 2008, 253, 119–131. [Google Scholar] [CrossRef]
- Massoth F E, Kim S C. Kinetics of the HDN of quinoline under vapor-phase conditions. Industrial & engineering chemistry research 2003, 42, 1011–1022. [Google Scholar]
- Satterfield C N, Cocchetto J F. Reaction network and kinetics of the vapor-phase catalytic hydrodenitrogenation of quinoline. Industrial & Engineering Chemistry Process Design and Development 1981, 20, 53–62. [Google Scholar]
- Perot G, Brunet S, Canaff C; et al. Transformation of Quinolines and Anilines Over NiMo-Al2O3 Catalysts. Bulletin des Sociétés Chimiques Belges 1987, 96, 865–870. [Google Scholar] [CrossRef]
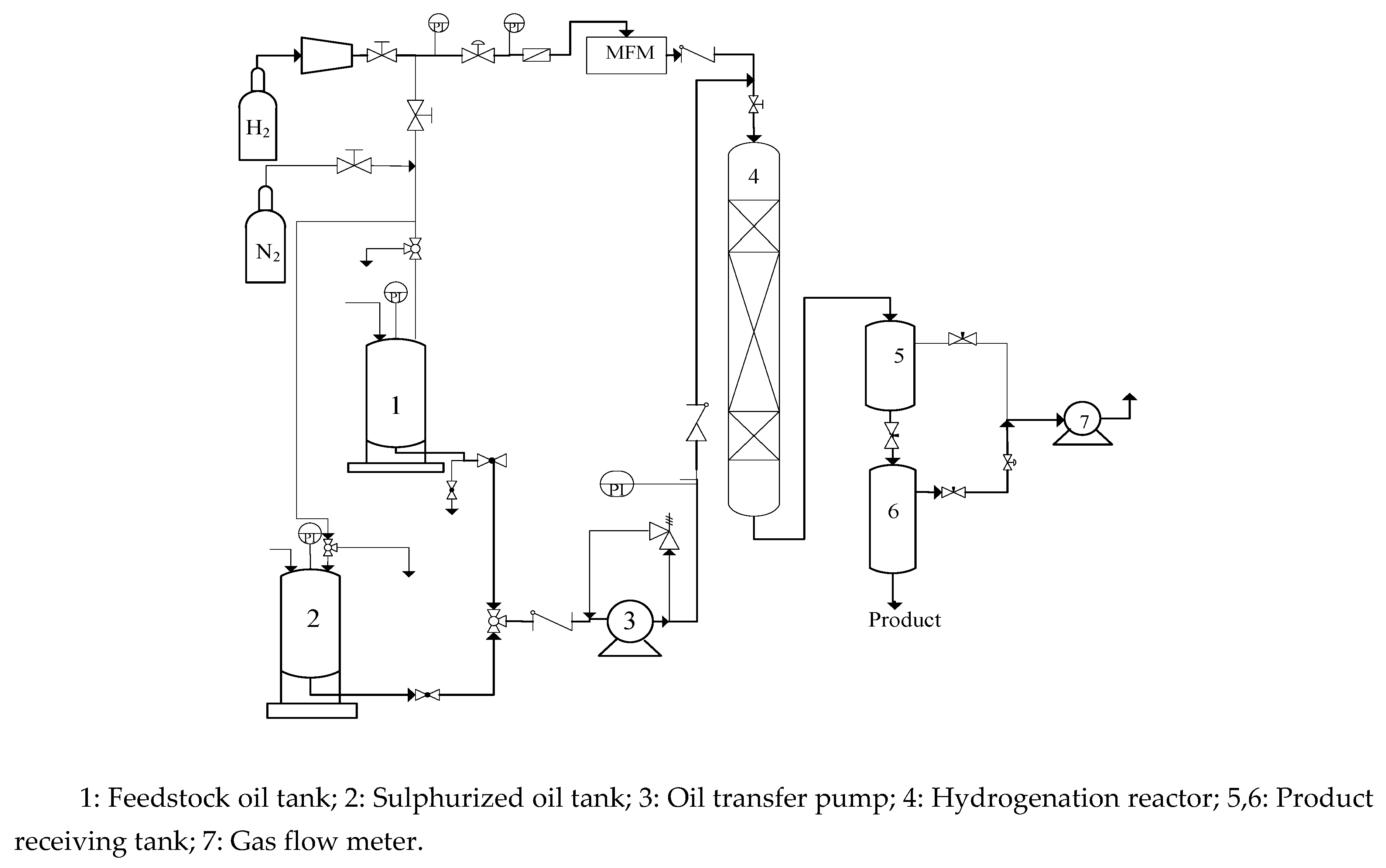
| pore volume (mL/g) |
Specific surface (m2/g) | Average pore size (nm) |
intensity (N/cm) |
Effective Component/(wt/%) | |||
|---|---|---|---|---|---|---|---|
| WO3 | MoO3 | NiO | P | ||||
| ≮0.25 | ≮150 | 8.13 | ≮182 | ≮24.0 | ≮2.4 | ≮2.5 | 0.3-1.7 |
| Catalyst | Ni (wt%) | Specific surface (m2 g-1) |
pore volume (cm3 g-1) |
pore size (nm) |
|---|---|---|---|---|
| SBA-15 | — | 715 | 1.16 | 6.8 |
| Ni2P/SBA-15 | 5 | 431 | 0.65 | 6.3 |
| 15 | 342 | 0.49 | 5.9 | |
| 25 | 255 | 0.40 | 5.4 | |
| 40 | 167 | 0.33 | 4.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




