1. Introduction
The microcrystalline graphite ore is located in the Canindé region, Ceará, in northeastern Brazil. Geologically, it is embedded in schists and paragneisses of the Central Domain of the northern subprovince of the Borborema Province [
1]. Carbon [
2], an abundant element in the biosphere, has been known since ancient times. In its native form, carbon mainly occurs as the diamond and graphite phases. Despite having identical chemical compositions (theoretically pure carbon), these minerals exhibit completely different physical, structural, and crystallochemical properties due to the distinct pressure and temperature conditions under which they crystallize. Graphite is stable at petrologically lower temperature and pressure conditions compared to diamond. Consequently, graphite has a less dense crystalline structure than diamond.
Natural graphite deposits are rarely found in a pure state [
3]; they usually contain impurities such as silicates, sulfides, iron oxides or other substances, making purification necessary for high-performance applications [
4]. Graphite is an industrially significant mineral due to its chemical composition and unique structure. Its chemical formula is represented by pure carbon (C), with a crystalline structure composed of layers of carbon atoms arranged in a hexagonal network [
5]. These layers are held together by Van der Waals forces, allowing them to slide over each other, which gives graphite properties such as solid lubrication and high thermal and electrical resistance. Beyond its importance as a raw material for various industries, natural graphite is a strategic resource for producing advanced materials. With the growing advancements in nanotechnology and electronics, the demand for high-purity graphite has increased significantly. Obtaining a high-quality graphite concentrate is essential for manufacturing high-performance products, including lithium-ion batteries, supercapacitors, and conductive materials [
6].
Various methods are employed for graphite purification, including thermal, chemical, and physical processes [
7]. The thermal method involves heating at high temperatures to volatilize impurities but requires high energy consumption. Chemical methods use strong acids and bases to dissolve impurities, which can generate harmful environmental waste. Physical beneficiation includes techniques such as gravity and electrostatic separation, which may have limited efficiency in removing finely dispersed impurities. In this context, flotation emerges as a highly effective method, as it allows the selective separation of graphite from impurities. The graphite has good natural floatability and hydrophobicity. Therefore, most graphite processing plants use the flotation method to purify graphite ore, that is, adding a series of flotation reagents to enrich the graphite ore in the gas-liquid interface to achieve separation from impurity minerals. Graphite flotation method can make the grade of graphite carbon reach 75-90%, or even about 95% [
8].
Graphite has widely diversified technological applications due to its structural and electronic properties. Its transformation into nanomaterials, such as graphene, further expands its potential applications in fields such as advanced electronics, biomedical devices, sensors, and energy storage. Graphene is obtained by exfoliating graphite layers [
9], resulting in atomic-thin sheets with exceptional properties such as high mechanical strength, excellent electrical and thermal conductivity, and a large surface area. This transformation process can occur through mechanical, chemical, or electrochemical methods, with material stability being a critical factor for its applicability. Graphene nanoplates, which are intermediates between graphite and monolayer graphene, represent a promising alternative due to their ease of production and superior stability in various applications [
10].
Among the methods used for graphene production,
acid leaching stands out, as it selectively removes impurities and destabilizes graphite layers. This process uses acidic solutions to oxidize and separate individual graphene layers, resulting in materials with high purity and controlled structural properties. Acid leaching offers significant advantages, such better control over graphene morphology and its properties. Additionally, this technique enables the production of graphene nanoplates with a high degree of crystallinity, enhancing their electrical and mechanical properties, making them ideal for applications in electronic devices and structural composites [
10].
The exploration and beneficiation of graphite in the Canindé region, Ceará, have great potential to generate high-value-added products and boost local socioeconomic development. The production of graphene nanoplates from graphite extracted in the region can foster new industries focused on advanced technologies, such as high-capacity batteries, flexible electronic devices, and multifunctional coatings. Furthermore, developing a local production chain based on graphite purification and nanomaterial production could attract investments and create skilled jobs, strengthening the regional economy and promoting sustainable growth [
11].
Graphene-like products obtained by exfoliating this graphite have a wide range of applications and can be used in
composite materials (incorporating graphene into polymers [
12] and other materials significantly improves mechanical properties such as strength and hardness, making it useful for applications requiring lightweight and durable materials [
13]) and
special coatings [
14] (corrosion protection [
15], flexible displays and sensors, antistatic and antimicrobial coatings, etc. [
16]). Due to the challenges of stabilizing graphene, graphite nanoplatelets have been considered a substitute for graphene in various applications. These nanoplatelets may occur naturally, associated with geological shear zones [
4], or be obtained through simple beneficiation processes.
This study aims to develop and optimize methods for purifying and preparing graphene nanoplatelets from natural graphite from Canindé. For this purpose, flotation, leaching, and chemical treatment processes will be investigated to obtain materials with enhanced properties for use in strategic sectors. The results of this study are expected to contribute to advancing knowledge on graphite beneficiation and its applications in nanomaterials, opening new technological and industrial opportunities for the region. Additionally, the economic and environmental feasibility of the employed processes will be evaluated to ensure that the conversion of graphite into graphene nanoplatelets occurs sustainably and efficiently.
2. Experimental
2.1. Experimental Methods
The sample preparation was carried out at Lagetec, Department of Geology, Federal University of Ceará (UFC), while the flotation study was conducted at the Laboratory for the Development of Ceramic Materials, Department of Metallurgical and Materials Engineering, UFC. The graphite purification and nanomaterial formation experiments were performed at the Ceará Center for Technology and Industrial Quality (NUTEC). Chemical analyses by X-ray fluorescence and X-ray diffraction were conducted at the X-ray Laboratory, Department of Physics, UFC. Raman spectroscopy and AFM measurements were performed at the Analytical Center and the Structural Crystallography Laboratory, both within the Department of Physics at UFC. Transmission Electron Microscopy (TEM) analyses were conducted at the National Nanotechnology Laboratory (LNNano/CNPEM).
2.1.1. Flotation Experiment
Flotation tests were conducted using a sample of microcrystalline graphite ore. The ore needs to be ground to meet the feed requirements before flotation and was reduced to below 3.35 mm. The focus of the present study was to investigate parameters such as grinding time and reagent dosage in order to achieve higher graphite recovery through flotation. For natural graphite, a process flow based on staged grinding and flotation is generally used. Laboratory-scale rougher flotation tests were performed to study the grinding time and the dosage of reagents, kerosene (collector) and Flotanol D-25 (frother) [
7,
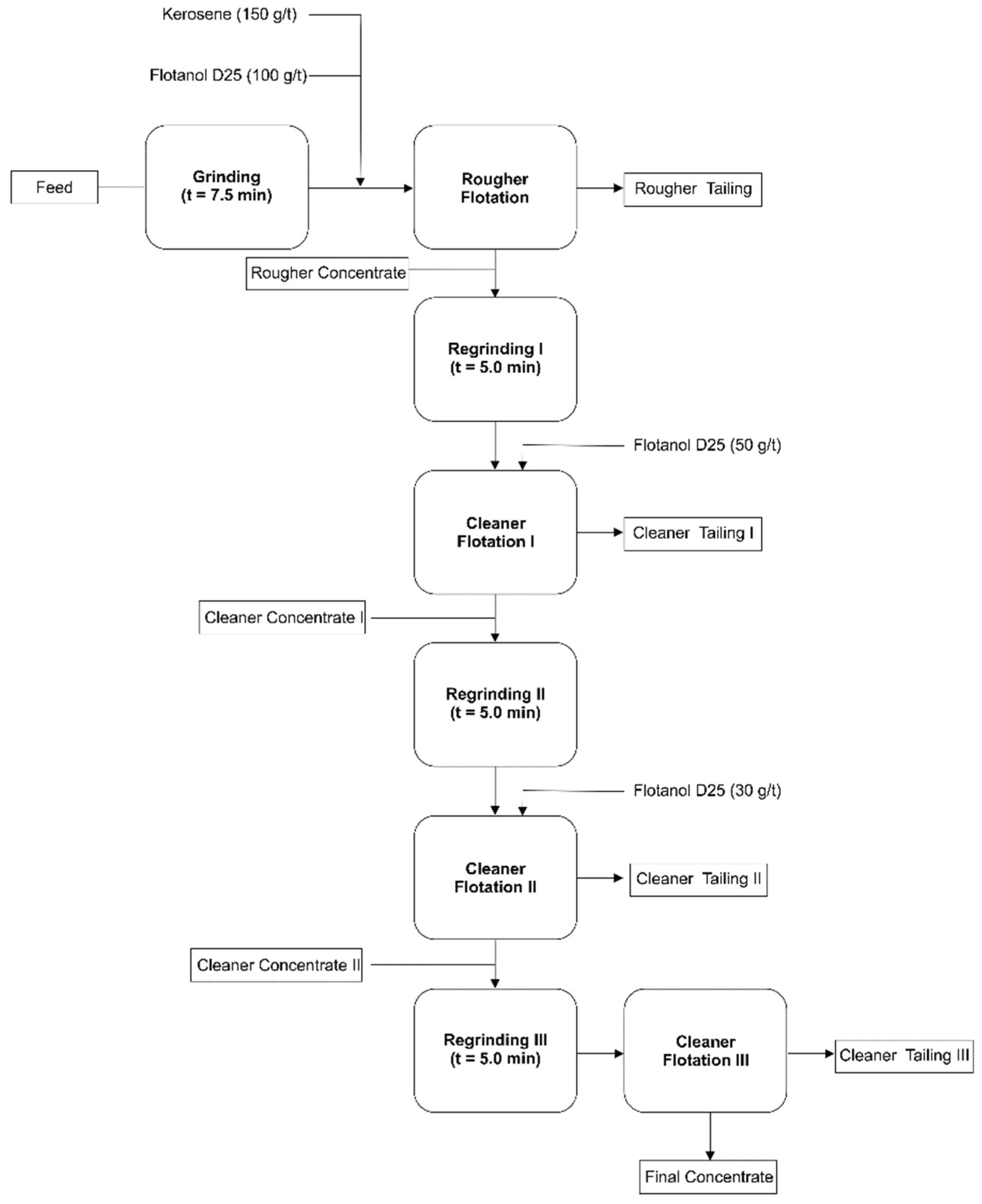
8]. Rougher grinding time tests were carried out in a 12” × 8” mill using a grinding charge composed of bars with varying diameters. Tests were conducted using different grinding times of 2.5, 5, 7.5, and 10 minutes, while maintaining constant dosages of 200 g/t of collector and 75 g/t of frother. It was observed that the highest graphite recovery was achieved at a grinding time of 7.5 minutes, as the goal of this stage was to maximize graphite recovery during the rougher flotation. Therefore, this grinding time was adopted for the subsequent reagent dosage tests (
Figure 1).
The grinding was performed under wet conditions, with a solids content of 65%. That is, approximately 550 mL of water was added for each 1 kg sample. In this case, the pH considered was the natural pH of water (around 7), since graphite recovery is not significantly affected by pH variations above 4. The decrease in recovery at pH values below 4 is associated with a reduction in the contact angle [
17]. The air flow rate in the flotation cell during the tests were approximately 3.0 L/min. The frother Flotanol D-25 was supplied by Clariant. The study demonstrated that it was possible to obtain a rougher concentrate with a graphite carbon content of 23.4% and a recovery of 86.4% using a grinding time of 7.5 minutes and reagent dosages of 150 g/t of kerosene and 100 g/t of Flotanol D-25. Under these conditions, to further concentrate the graphite, a flotation circuit was carried out, consisting of a rougher stage followed by three successive stages of regrinding and cleaner flotation. During the selection process, the target minerals and impurities are gradually dissociated through multiple grinding stages, and the optimal number of grinding and flotation steps is experimentally studied and determined.
2.1.2. Chemical Purification in Alkaline Environment (C-NaOH Treatment)
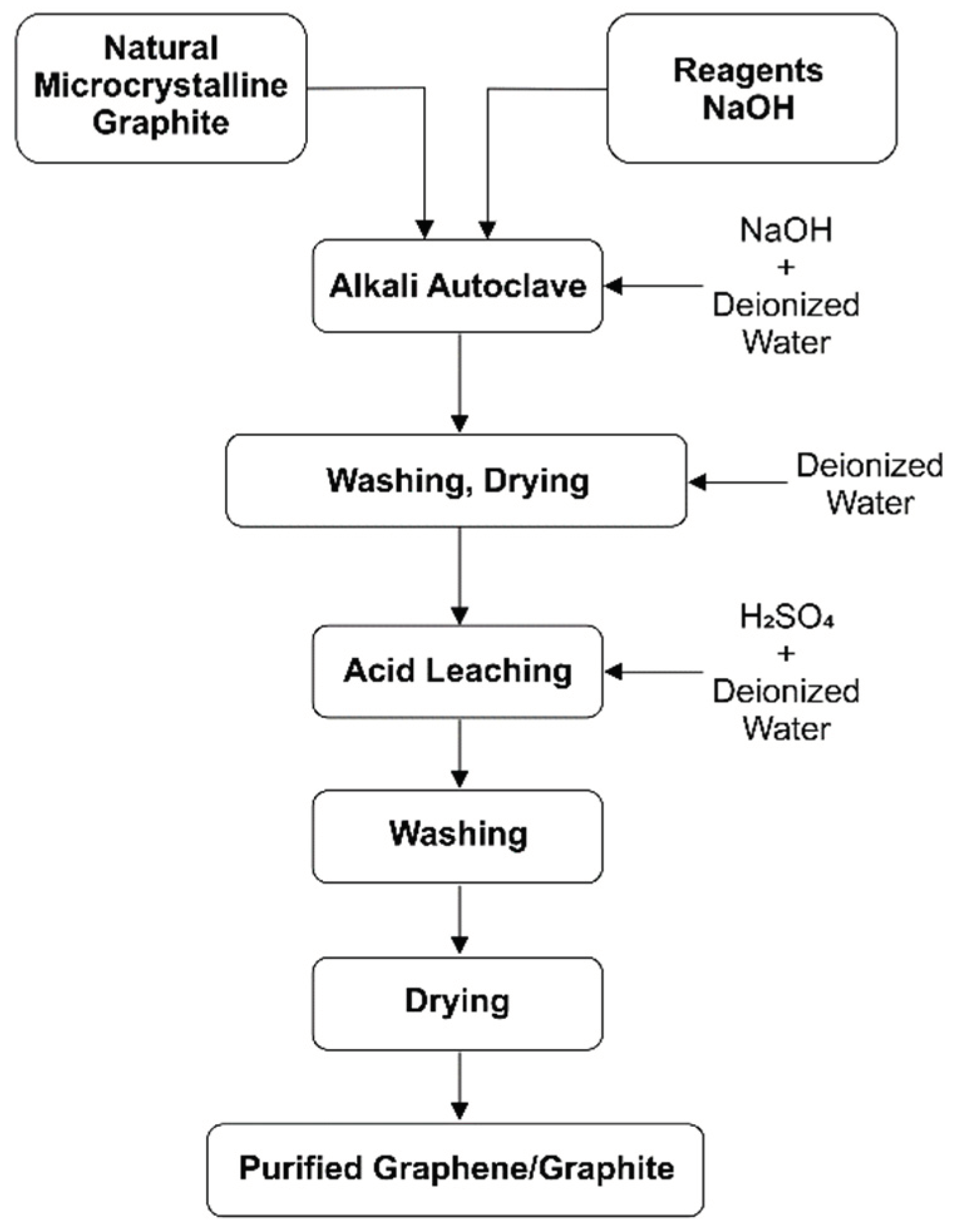
The purification of graphite was carried out through an alkaline treatment [
18] in a hydrothermal reactor [
19]. Initially, a 1M sodium hydroxide solution was prepared using 61 mL of deionized water. Sodium hydroxide – puriss. p.a., ACS reagent, Ph. Eur. grade, K ≤0.02%, ≥98%, pellets. Then, 4.00 g of graphite (C-bulk) was dispersed in the solution, forming a dense and homogeneous suspension. The suspension was transferred to a Teflon vessel, which was properly sealed and placed inside a hydrothermal reactor. The system was then subjected to a thermal treatment in a muffle furnace at 200 °C for 6 hours. After this period, the material was cooled to room temperature and subjected to a neutralization process. The pH adjustment was performed until reaching pH 7 through successive washings with deionized water. The material was centrifuged and washed to ensure the complete removal of alkaline residues [
19]. Finally, the sample was subjected to a drying process in an oven at 70 °C for 24 hours, ensuring the removal of residual moisture before subsequent processing and characterization steps (
Figure 2).
2.1.3. Leaching in Acidic Environment (C-NaOH+H2SO4 Treatment)
The acid leaching of the previously purified material was carried out using sulfuric acid (H₂SO₄) in an aqueous medium under magnetic stirring. For this purpose, a 1.0 M H₂SO₄ solution was prepared by diluting the required amount of analytical-grade sulfuric acid (98% purity, 18.4 M) in 25 mL of deionized water. Sulfuric acid – for inorganic trace analysis, 93–98%, density: 1.840 g/mL at 25 °C, Merck. The powder obtained from the hydrothermal purification step (C-NaOH treatment) was then added to this solution. The mixture was kept under constant magnetic stirring in a heated water bath at 90 °C for 120 minutes. After the reaction time, the material was centrifuged and washed until reaching pH 7, then dried in an oven at 70 °C until complete moisture removal [
18]. The final product was labeled as “C-NaOH+H₂SO₄ treatment” (
Figure 2).
2.2. Characterizations
To determine the crystalline structure of the solids, X-ray diffraction (XRD) measurements were performed using a Panalytical® Xpertpro MPD diffractometer equipped with a Co-Kα radiation tube, operating at 40 kV, 20 mA, and a step size of 2° per minute. The analyzed samples were randomly oriented using the powder method. The 2θ angle range was from 0 to 100°. The interplanar spacing (d) was used to interpret the mineral peaks.
The X-ray fluorescence (XRF) method was used for the chemical analysis of the mineral in powder form, allowing qualitative and semi-quantitative determination of the elements. XRF measurements were collected using a sequential wavelength-dispersive X-ray spectrometer (WDX) RIGAKU ZSX Mini II, operating at 40 kV and 1.2 mA, with a Pd (palladium) tube capable of performing semi-quantitative analysis of elements ranging from fluorine to uranium.
Fourier-transform infrared spectroscopy (FTIR) measurements were conducted using a SHIMADZU IRXross instrument, operating in the frequency range of 4000 to 400 cm⁻¹, with 64 scans. Absorbance was used as the analytical method, and potassium bromide (KBr) pellets served as the sample support [
20].
To identify the physicochemical structure of the analyzed materials [
21,
22], Raman spectroscopy measurements were performed using an Alpha 300 spectrometer from Witec. A 532 nm excitation laser with a power of 300 microwatts was focused on the sample through a 10× magnification objective lens. Low excitation power was used to prevent sample degradation. The Raman spectra of all three samples exhibited the characteristic peaks of carbon-based materials (D, G, and 2D bands). The Raman spectra were analyzed using the Fityk software (version 1.3.1), where baseline subtraction and spectral deconvolution into Lorentzian components were performed. No significant Raman peak shifts were observed among the different samples. For comparison purposes, all spectra were normalized relative to the G band.
Atomic Force Microscopy (AFM) analyses were also conducted using an Asylum MFP-3D system, operating in intermittent contact mode with silicon tips (NCHR-W, manufactured by NanoWorld, characterized by a force constant of 42 N/m and a resonance frequency of 320 kHz). A scanning area of 10 µm × 10 µm was analyzed for each sample. All samples were dispersed in ethanol using magnetic stirring at room temperature for 15 minutes and subsequently deposited onto a silicon substrate suitable for nanomaterial characterization via atomic force microscopy.
Transmission Electron Microscopy (TEM) analyses were also conducted using a TEM-FEG (JEOL JEM-2100F) operated at 200 kV. A fraction of the sample (~2 mg) was dispersed using an ultrasonic bath for 2 minutes and subsequently deposited onto an Ultrathin Carbon Film on Lacey Carbon Support Film, 400 mesh (TEM grid).
3. Results and Discussion
3.1. Flotation
Graphite recovery was determined based on the mass balance of the process and the graphite carbon content of the concentrates and tailings, calculated by the loss on ignition (LOI) method. First, crucibles were weighed on an analytical balance, and their masses were recorded. Then, using a spatula, a small amount of each sample was added to the respective crucibles (2 g for flotation test concentrates and 3 to 4 g for tailings), and the combined mass of the crucible and wet sample was recorded. After the initial weighing, the crucibles containing the samples were placed in a furnace at 600 °C for 30 minutes to calcine the volatile matter (organic material). After this step, the crucibles were removed from the furnace and weighed again. The samples were then returned to the furnace for 90 minutes at 960 °C to burn off the graphitic carbon. Finally, the crucibles containing the dry, carbon-free residue were weighed, and the graphitic carbon content of the samples was calculated.
The microcrystalline graphite ore sample exhibited a graphite carbon content of 6.85%, indicating a significant number of impurities associated with and mixed into the natural graphite. The flotation study results demonstrated that a graphite concentrate with a graphite carbon content of 76.6% and a recovery rate of 64.0% was obtained in the laboratory, increasing the ore's purity by removing impurities. However, these results also highlight the need for further studies to improve recovery and achieve a concentrate with a higher graphite carbon content. Given the necessity for more efficient purification, an autoclave purification method was applied in a basic medium, followed by acid leaching.
3.2. XRD Analysis
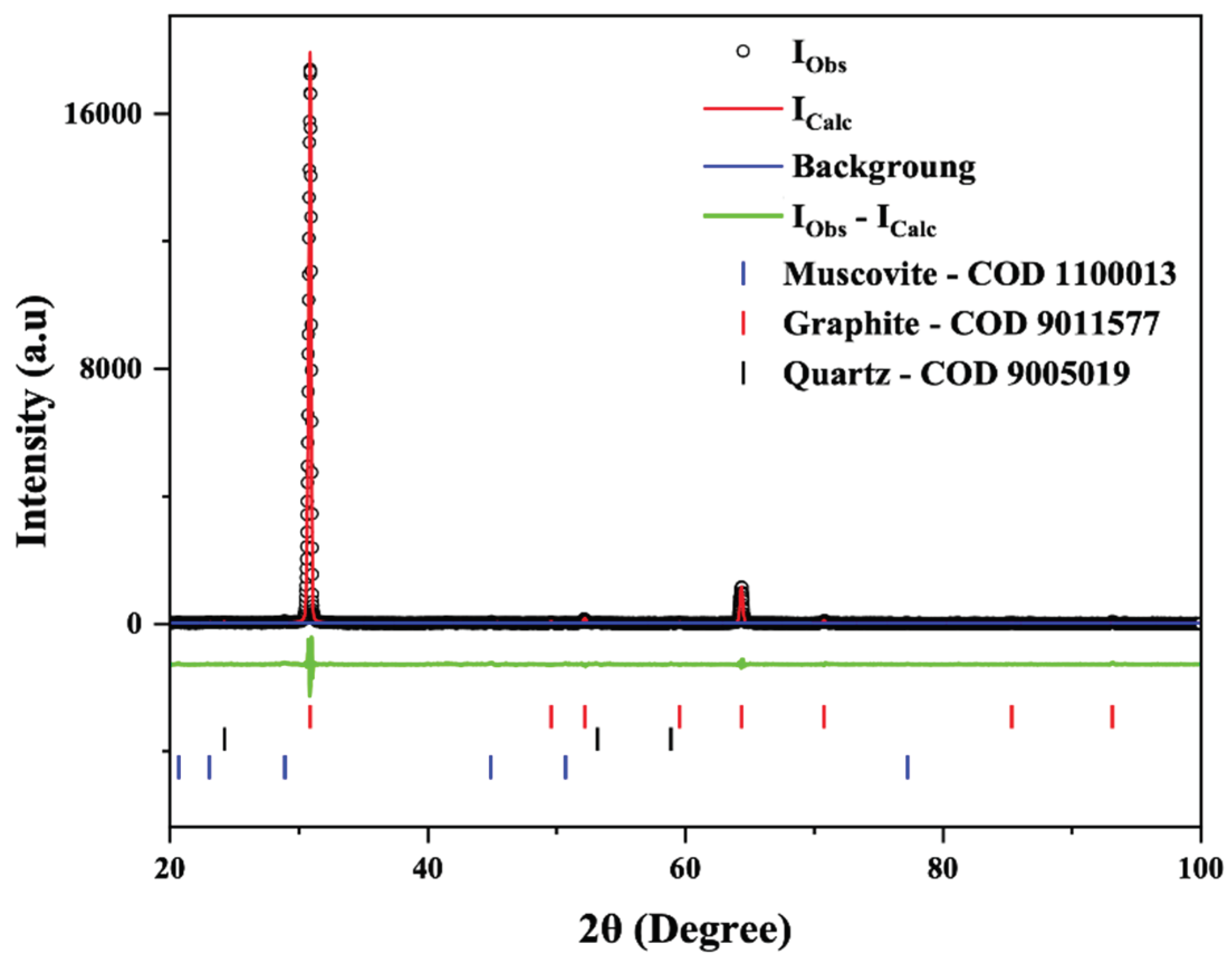
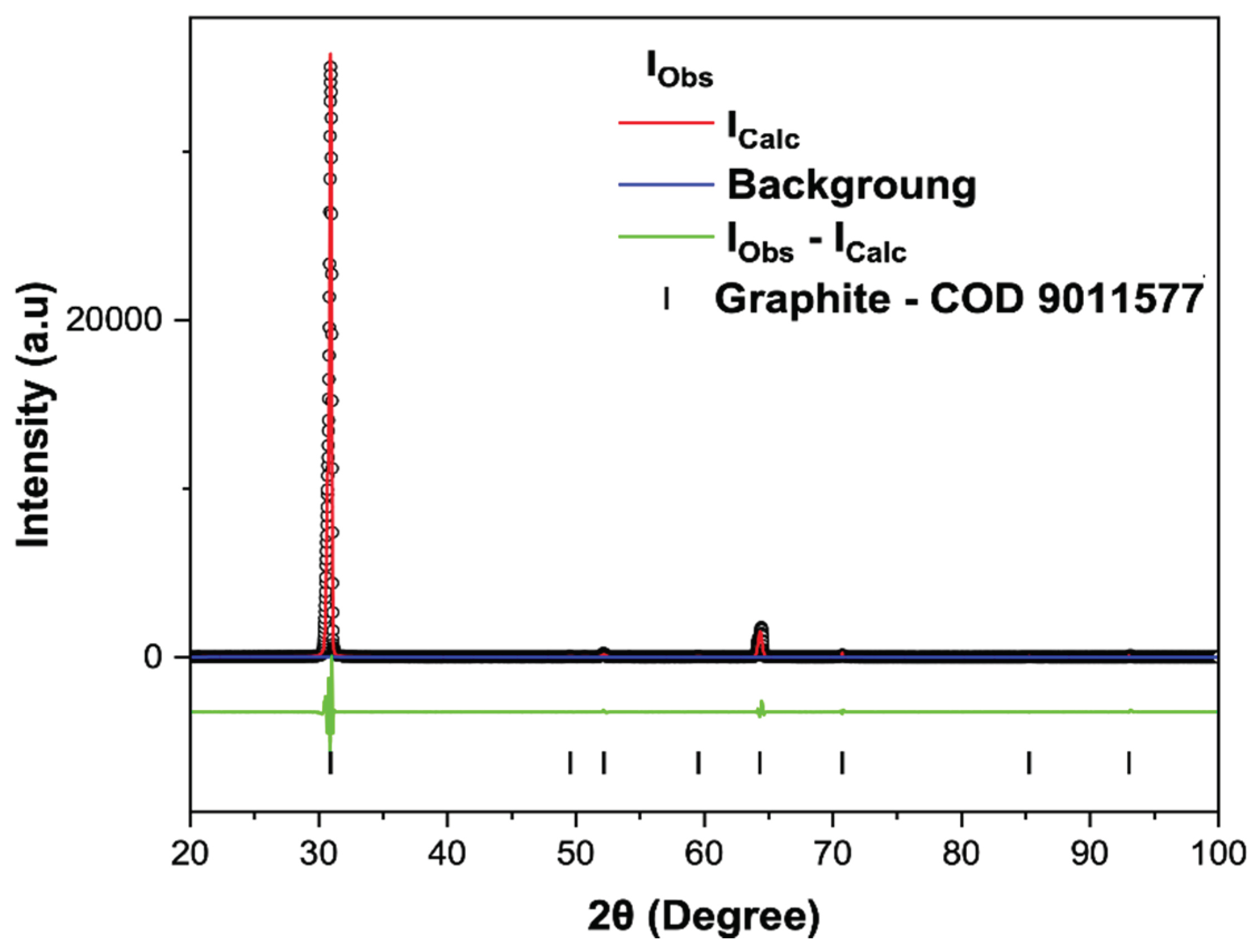
For the X-ray diffraction (XRD) analyses, the powders obtained from the following stages were used: C-Bulk (concentrate - after flotation) and C-NaOH+H₂SO₄ treatment (final graphene-like obtained). According to the results, graphite is responsible for the highest diffraction peak intensity, with a peak characteristic of graphite (002). Depending on the formation environment, graphite associations may involve muscovite, which was well-matched to most peaks, though with low intensity. The quartz mineral phase is also present. This association of the three minerals is common in deposits, especially when the host rock is a graphitic schist. In all samples, the graphite peak has a 2θ value at 31.5° and an average d₀₀₂ spacing of 3.35 Å [
23,
24]. Additionally, muscovite and quartz were interpreted in the same peak, coexisting with the graphite peak in the C-Bulk sample and result of the calculated pattern by the Rietveld Method using Crystallographic Information File (
Figure 3). Secondary peaks at 1.41 Å, in smaller proportions, were also identified, reinforcing the mineral interpretation for graphite. The XRD refinement pattern for C-Bulk sample (concentrate - after flotation) consists of graphite with space group P63/mmc (71.4%), quartz with space group P3121 (28.5%) and muscovite with space group C12/c1 (0.1%). The C-NaOH+H₂SO₄ treatment sample showed an almost singular graphite peak with space group P63/mmc (100%) and result of the calculated pattern obtained with Rietveld refinement using Crystallographic Information File (
Figure 4), indicating a well-structured arrangement in the analyzed sample. The lattice parameter and crystallite size from the Rietveld refinements are summarized in in
Table 1.
The crystalline sizes were estimated by the Scherrer equation (1), in which: β represents the full width at half maximum (FWHM) in radians, adjusted for the instrumental line broadening, while θ denotes the Bragg angle, K is a dimensional shape factor, and λ represents the radiation wavelength.
3.3. XRF Analysis
For the chemical analyses of major and minor elements in the whole-rock samples using the X-ray fluorescence (XRF) method, the powders obtained from the following samples were used: graphite ore (before flotation), C-Bulk (concentrate - after flotation), C-NaOH treatment (treatment in a basic medium), and C-NaOH+H₂SO₄ treatment (graphene-like material).
The chemical composition
of major and minor elements in the whole-rock of the graphite ore sample shows SiO₂ = 42.43%, Al₂O₃ = 18.88%, CaO = 2.19%, MgO = 1.06%, K₂O = 7.55%, TiO₂ = 2.69%, MnO = 2.69%, Fe₂O₃ = 23.81%, SO₃ = 0.34%, and Cl = 0.62%. The C-Bulk sample contains SiO₂ = 31.02%, Al₂O₃ = 15.65%, CaO = 2.44%, K₂O = 6.32%, TiO₂ = 2.90%, MnO = 0.54%, Fe₂O₃ = 36.16%, SO₃ = 0.37%, Cl = 0.75%, P₂O₅ = 3.67%, and Co₂O₃ = 0.18%. The C-NaOH treatment sample presents SiO₂ = 29.75%, Al₂O₃ = 12.88%, CaO = 2.61%, MgO = 1.06%, K₂O = 5.13%, TiO₂ = 3.52%, MnO = 0.34%, Fe₂O₃ = 39.78%, P₂O₅ = 3.73%, and Rh₂O₃ = 1.22%. The C-NaOH+H₂SO₄ treatment sample exhibits SiO₂ = 75.65%, Al₂O₃ = 3.63%, CaO = 4.70%, K₂O = 3.71%, Fe₂O₃ = 0.86%, and P₂O₅ = 11.45%. These values are presented in
Table 2.
The chemical composition of major and minor elements in the whole-rock of the sample before flotation (raw graphite) corresponds to the mineralogy of microcrystalline graphite ore, which, in addition to graphite, also comprises muscovite and quartz. The high Fe₂O₃ content (23.81%) suggests a composition similar to ferrimuscovite, a variety of muscovite that can occur in high-pressure metamorphic rocks or hydrothermal deposits. The values of SiO₂ (31.02%), Al₂O₃ (15.65%), K₂O (6.32%), and MnO (0.54%) in the sample after flotation (C-Bulk) decreased, while the MgO content dropped to 0.00%. In contrast, CaO increased to 2.44%, TiO₂ to 2.90%, Fe₂O₃ to 36.16%, and Cl to 0.75%. After flotation, the composition also showed the presence of SO₃ (0.37%), P₂O₅ (3.67%), and Co₂O₃ (0.18%). These chemical analysis results for C-Bulk indicate that the flotation of microcrystalline graphite ore is essential for graphite concentration but still requires a more efficient purification process to improve the graphite carbon content.
The chemical composition of the C-NaOH treatment sample (basic medium treatment) compared to C-Bulk shows slightly lower values for SiO₂ (29.75%), Al₂O₃ (12.88%), K₂O (5.13%), and MnO (0.34%). However, the MgO content increased to 1.06%, while CaO (2.61%), TiO₂ (3.52%), Fe₂O₃ (39.78%), and P₂O₅ (3.73%) increased, with the appearance of Rh₂O₃ (1.22%) in the composition.
The C-NaOH+H₂SO₄ treatment sample presents the highest purity composition among all processing stages, showing a significant reduction in Al₂O₃ (3.63%), K₂O (3.71%), and Fe₂O₃ (0.86%), with the complete removal of MgO, TiO₂, and MnO. Additionally, CaO increased to 4.70%, P₂O₅ to 11.45%, and SiO₂ rose significantly to 75.43%.
The chemical composition of the C-NaOH+H₂SO₄ treatment sample demonstrates a strong reduction of impurities such as ferrimuscovite, through the reduction and elimination of elements that compose this mica. Since muscovite is the main impurity in microcrystalline graphite ore, the chemical data obtained by XRF and presented in this study indicate a purer natural microcrystalline graphite concentrate.
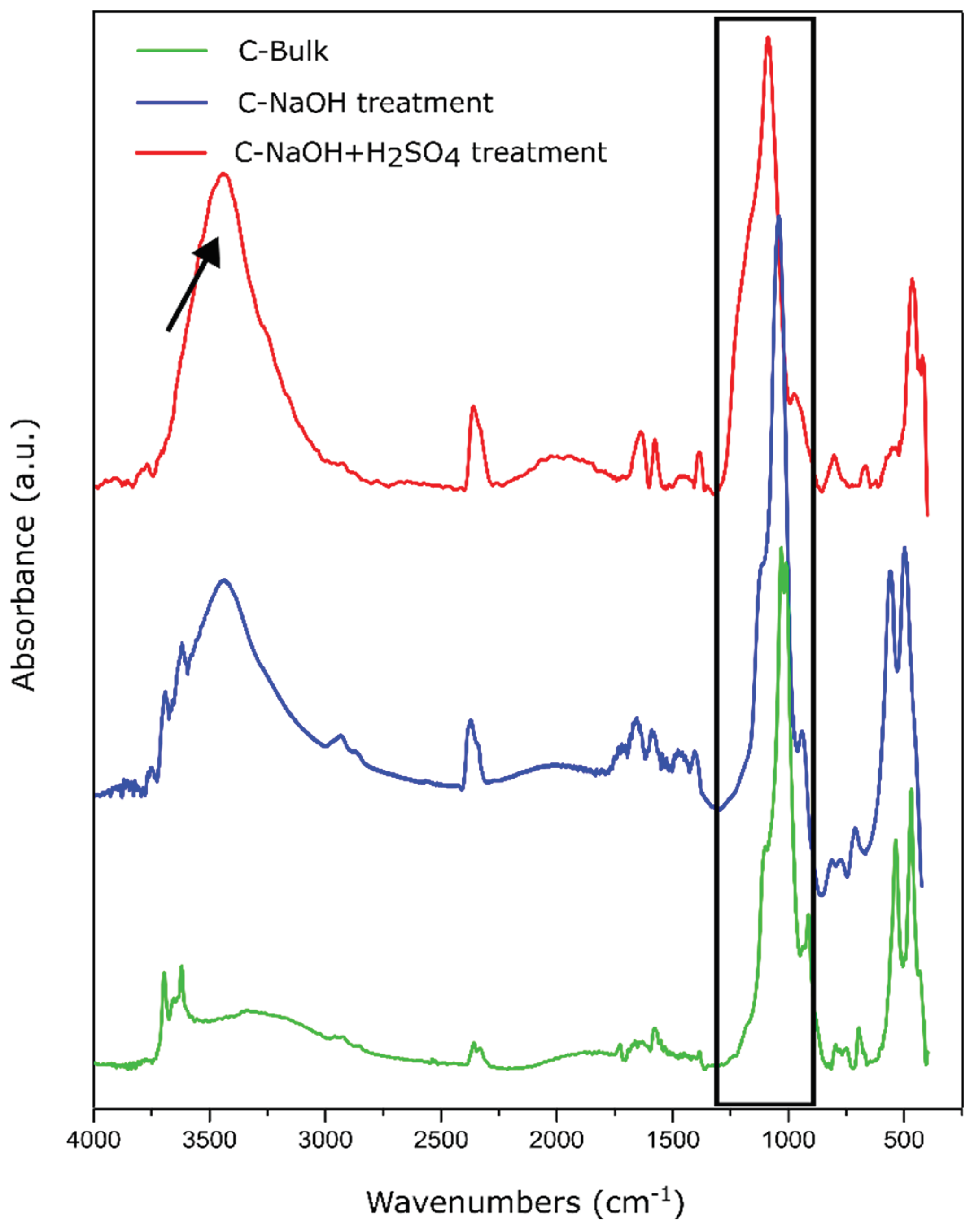
3.4. FTIR Analysis
Fourier-transform infrared spectroscopy (FTIR) was used to investigate the presence of functional groups throughout the purification and delamination stages of natural graphite. In the raw sample (C-Bulk), a low-intensity spectrum was observed, with prominent bands in the 1000–1100 cm⁻¹ region, associated with Si–O–Si and Al–O–Si bonds from phyllosilicates such as muscovite, commonly found in natural mineral-derived graphite. The absence of significant bands in the O–H stretching (~3400 cm⁻¹) and C=O (~1720 cm⁻¹) regions suggests that the carbonaceous structure is free from oxygen-containing functional groups, supporting the bulk nature of the starting graphite (
Figure 5).
After treatment in a basic medium (C-NaOH), an intensified band appears at ~3430 cm⁻¹, characteristic of the stretching of free hydroxyl (–OH) groups, which may result from surface hydration. Additionally, a moderate band is observed at ~1620 cm⁻¹, attributed to the bending vibration of adsorbed water or to the C=C stretching of the sp² aromatic framework, indicating structural reorganization and possible layer opening [
25,
26].
The samples treated with H₂SO₄ exhibited a pronounced increase in the intensities of these bands, especially at ~3430 cm⁻¹, indicating a higher density of hydroxyl groups and enhanced surface water adsorption; at ~1720 cm⁻¹, associated with the C=O stretching vibration of carboxylic groups (–COOH), commonly found at the edges of nanoplatelets after mild functionalization; and at ~1030 cm⁻¹, which remained present, suggesting residual C–O functionalities, possibly originating from esters or sulfate remnants.
The simultaneous presence of bands attributed to C=O, C–OH, and C=C indicates that the material did not undergo severe oxidation, with the observed spectral behavior being consistent with that of mildly functionalized graphene, especially in few-layer samples where the surface remains partially passivated and the sp² structure is preserved.
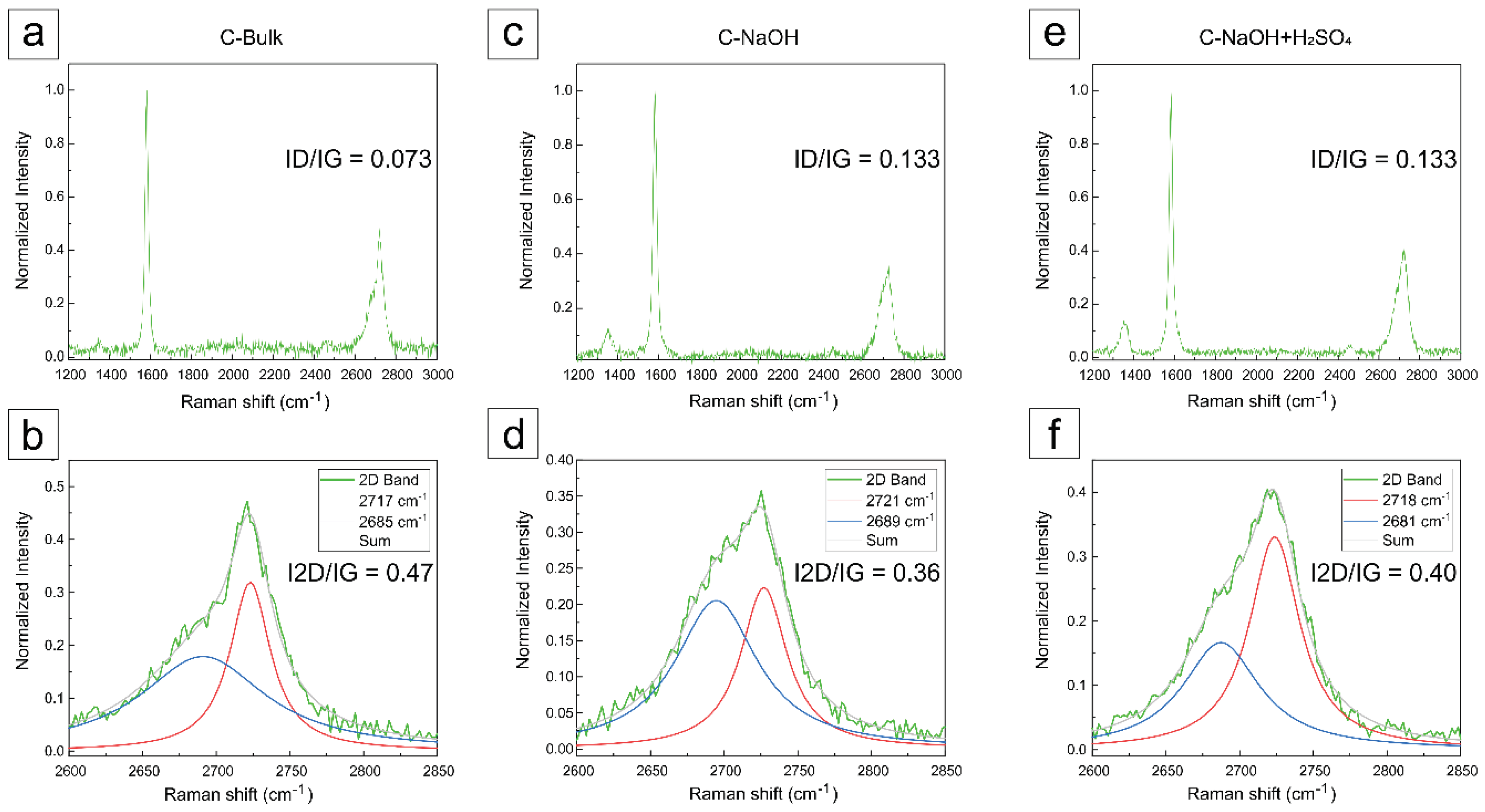
3.5. Raman Analysis
The Raman spectra were normalized for all samples. The intensity ratios between the D and G bands are indicated in the graphs, and the main features in the Raman spectra of graphite and graphene are represented by the D, G, and 2D bands. The main bands observed in the Raman spectra of graphitic materials include: the G band, associated with the E₂g vibrational mode of the hexagonal graphite lattice, indicative of ordered sp² carbon; the D band, attributed to structural defects and disorder in the lattice, including edges, vacancies, and functional groups; and the 2D (or G') band, which is a second-order overtone of the D band and is essential for distinguishing single-layer graphene from few-layer graphene and graphite, based on its shape and relative intensity with respect to the G band (
Figure 6).
A typical Raman spectrum of graphene-like materials is primarily characterized by G, D and 2D bands. The G band arises from the E
2g phonon of the hexagonal lattice and is mainly related to sp
2 coordination. The D band originates from a double resonance process involving lattice defects, such as vacancies or structural disorders. The last 2D band results from a higher order process involving two phonon scattering process. Notably, the intensity ratio of the 2D to G band (I2D/IG) ca be understood as a fingerprint for distinguishing between monolayer graphene, multilayer graphene and graphite [
21,
27]. In contrast, the intensity ratio of the D to G band (ID/IG) offers valuable insights into the atomic structure, including the degree of oxidation and presence of structural defects [
30,
35]. The corresponding normalized Raman spectra of all samples and the D/G and 2D/G bands are indicated in
Figure 6(a)-(f).
For the analysis of the Raman spectra of the produced samples, graphical deconvolution was performed using Lorentzian convolution components with the Fityk 1.3.1 software, after manual baseline subtraction, as shown in
Figure 6 and
Figure 7.
In the graphite concentrate (C-Bulk), as illustrated in
Figure 6a, the G band associated with graphite appears at approximately 1579 cm⁻¹, while the 2D band is observed around 2720 cm⁻¹, exhibiting a characteristic shoulder typical of graphite [
21,
28,
29,
30,
31,
32,
33,
34]. After the deconvolution of the 2D band (
Figure 6b), two components were revealed at 2685 cm⁻¹ and 2717 cm⁻¹, which are characteristic of multilayer graphite [
31,
32,
33,
34]. The ID/IG ratio of the sample (C-bulk) was 0.073, reflecting a low density of apparent defects, although partially masked by mineral impurities present in the matrix. [
36].
The purification process in basic medium revealed significant alterations in the carbonaceous structure throughout the treatment. The sample treated in basic medium (
Figure 6c) exhibited similar bands at 2689 cm⁻¹ and 2721 cm⁻¹ (
Figure 6d), still displaying a broad and asymmetric profile, which suggests a partially exfoliated structure. An increase in the ID/IG ratio to 0.133 was observed, indicating the introduction of structural defects, possibly related to partial delamination and functionalization induced by sodium hydroxide at high temperature, as reported in the literature [
37,
38].
In the final sample, subjected to acid treatment, an intensified and more defined D band at ~1336 cm⁻¹ was observed compared to the previous samples (
Figure 6e), indicating a higher density of defects or edges in the material. Additionally, bands at 2681 cm⁻¹ and 2718 cm⁻¹ were observed (
Figure 6f), with a very subtle shoulder detected only after spectral deconvolution. The profile is characteristic of stacked structures, supporting the hypothesis of graphene nanoplatelet formation [
36,
37].
After the acid purification step, the ID/IG ratio remained at 0.133, suggesting that no additional defects were introduced, but rather a reorganization of the layered structure occurred. These findings indicate the formation of few-layer graphene nanoplatelets as a direct result of the acid purification process [
39,
40].
According to these measurements, the distinction between graphene nanoplatelets and graphene oxide (GO) can be assessed by analyzing the ID/IG ratio, which typically ranges from 0.8 to 1.2 in GO due to its high density of defects and disruptive oxygen-containing groups in the sp² structure. In contrast, the sample produced after acid purification exhibited an ID/IG ratio of only 0.133, indicating few structural defects. Moreover, the 2D band profile was more asymmetric and consistent with few-layer graphene. In GO, this band tends to be significantly broadened or even absent due to the breakdown of the conjugated bonding network [
37,
38].
The absence of excessive broadening in the G and 2D bands, as well as the I₂D/IG ratio below 1.5, rules out the presence of pure single-layer graphene. However, the shape of the 2D band (Figure 6) resembles that of graphene with three to five layers [
21,
27], this confirms the formation of few-layer, non-oxidized nanoplatelets - i.e., graphene nanoplatelets (after C-NaOH + H₂SO₄ treatment). Moreover, the absence of significant broadening in the 2D band indicates that the material did not undergo severe oxidation, which would typically be evidenced by suppression of the 2D band and enhancement of the D band, as commonly observed in graphene oxides [
36]. The I₂D/IG values obtained for the three samples indicate that the analyzed material consists of multilayer graphite nanoplatelets (C-bulk) and few-layer graphene nanoplatelets (after C-NaOH + H₂SO₄ treatment), both with more than one layer. These differ from monolayer graphene (I₂D/IG > 2) and graphene oxide, which typically exhibits a significantly suppressed 2D band due to the disruption of the π-conjugated network. The variation in I₂D/IG values following chemical treatments reflects the structural and functional changes induced, consistent with those expected from graphene functionalization and reduction processes [
39,
40].
The acid treatment resulted in an I₂D/IG value of 0.40, slightly higher than that of the base-treated sample (I₂D/IG = 0.36). This suggests that the acid treatment may have promoted a mild reduction of previously introduced oxygen-containing groups, partially restoring the conjugated structure of graphene and consequently increasing the relative intensity of the 2D band. However, the value still indicates the presence of a few layers and possible residual defects [
36].
The I₂D/IG ratio of the samples indicates that they are graphene/graphite nanoplatelets with multiple layers and is not the most reliable parameter for determining the exact number of layers. However, the shape of the 2D bands allows for the differentiation of graphene with fewer than five layers [
21]. To elucidate the dimensional characteristics of the analyzed samples, additional characterization techniques such as AFM and TEM were required.
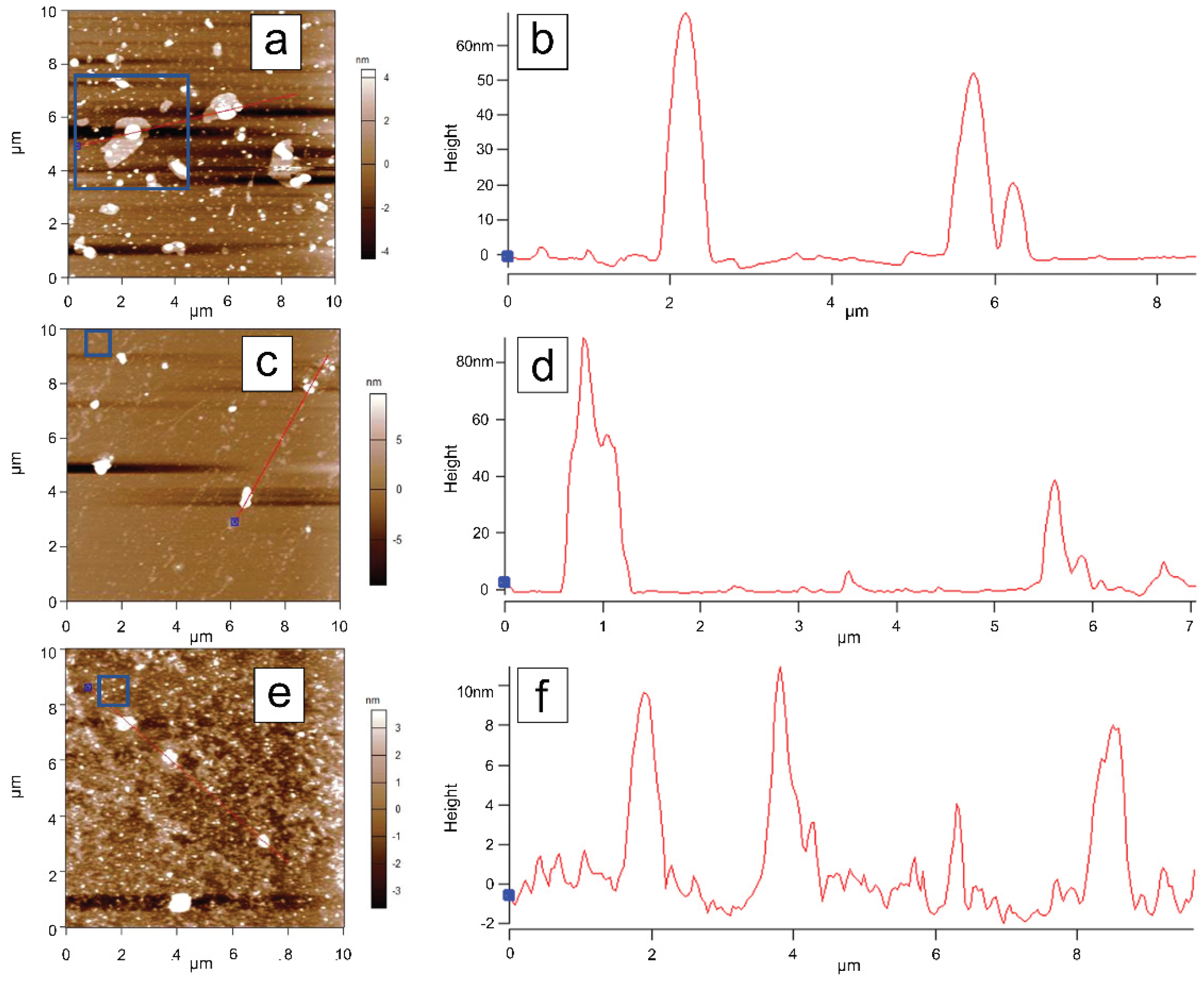
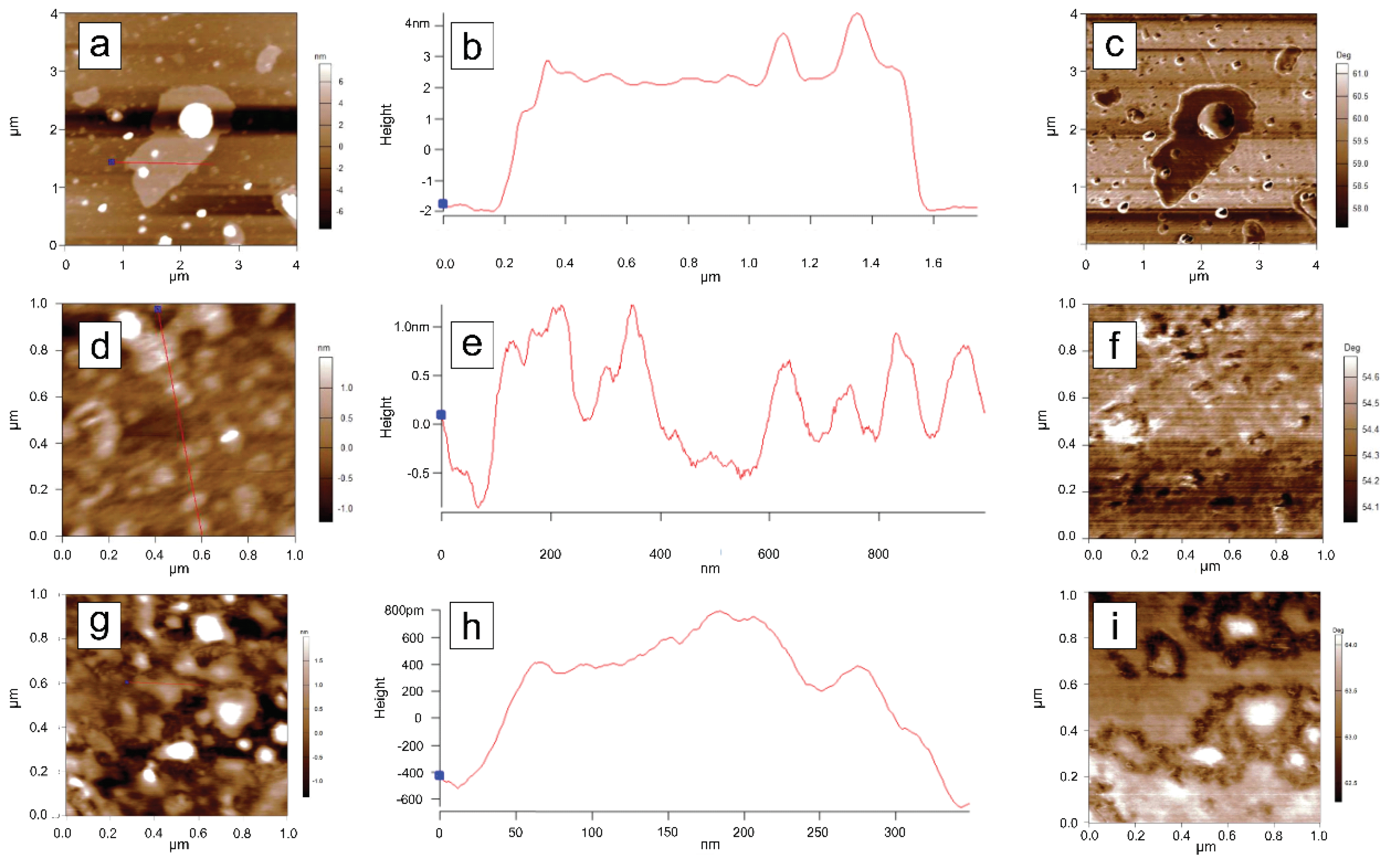
3.6. Atomic Force Microscopy (AFM) Analysis
To investigate the topographical features of the samples, atomic force microscopy (AFM) measurements were performed on samples dispersed by magnetic stirring in water and deposited onto a silicon substrate. Initially, 10 µm × 10 µm areas were scanned for each sample. The C-Bulk sample (
Figure 8a) is characterized by the presence of numerous particles with sizes on the order of tens of nanometers, as well as lamellar structures (
Figure 8b), which are indicative of the early stages of nanoparticle formation.
In the AFM scan of the C-NaOH sample (
Figure 8c), structures with sizes on the order of several tens of nanometers were also identified, although in smaller quantities compared to the C-Bulk sample (
Figure 8d). The large lamellar structures observed in the C-Bulk sample appear to have been fragmented during this purification step, resulting in even smaller structures.
The AFM scan of the C-NaOH+H₂SO₄ sample (
Figure 8e) indicates greater uniformity in particle size, with the largest structures measuring approximately 10 nm (
Figure 8f), representing a significant reduction compared to the previous samples. To better highlight the topographical features of the smaller structures, additional scans were performed in the areas outlined by blue rectangles in each measurement.
For each sample, a high-resolution scan was performed in a reduced area. In the C-Bulk sample (
Figure 9a), a large lamellar structure is observed, with a thickness of approximately 4.0 nm (
Figure 9b–c), consistent with a graphene-like structure that may have begun to form at this stage, although still in small quantity and within a heterogeneous sample. In the C-NaOH sample (
Figure 9d), irregularly sized structures are observed, with heights on the order of 1.5 nm (
Figure 9e–f). In the C-NaOH+H₂SO₄ sample, structures are observed with heights (
Figure 9g) around 1.2 nm (
Figure 9h–i).
The phase images corresponding to each topographic image in
Figure 9 are shown alongside their respective profile sections in
Figure 9c,f,i. Since contrast variations in AFM phase images of graphene samples indicate the presence of different numbers of stacked layers [
41], it can be concluded that C-NaOH+H₂SO₄ sample is characterized by the stacking of multiple graphene layers, which vary heterogeneously across the analyzed area.
AFM analyses reveal that the purification process resulted in a low-dimensional, heterogeneous sample, characteristic of graphene nanoplatelets, in agreement with the other structural characterizations [
36,
40].
3.7. Transmission Electron Microscopy (TEM) Analysis
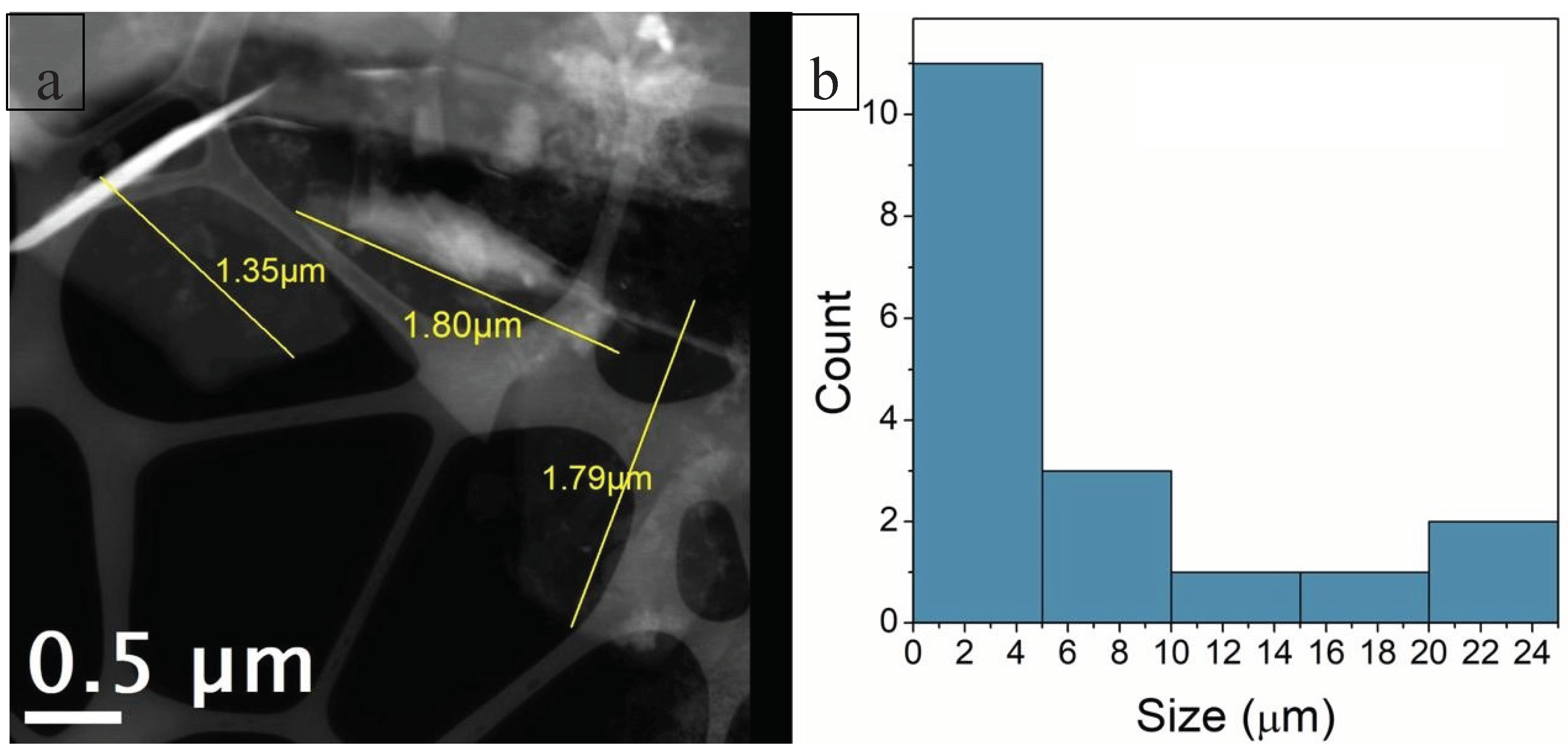
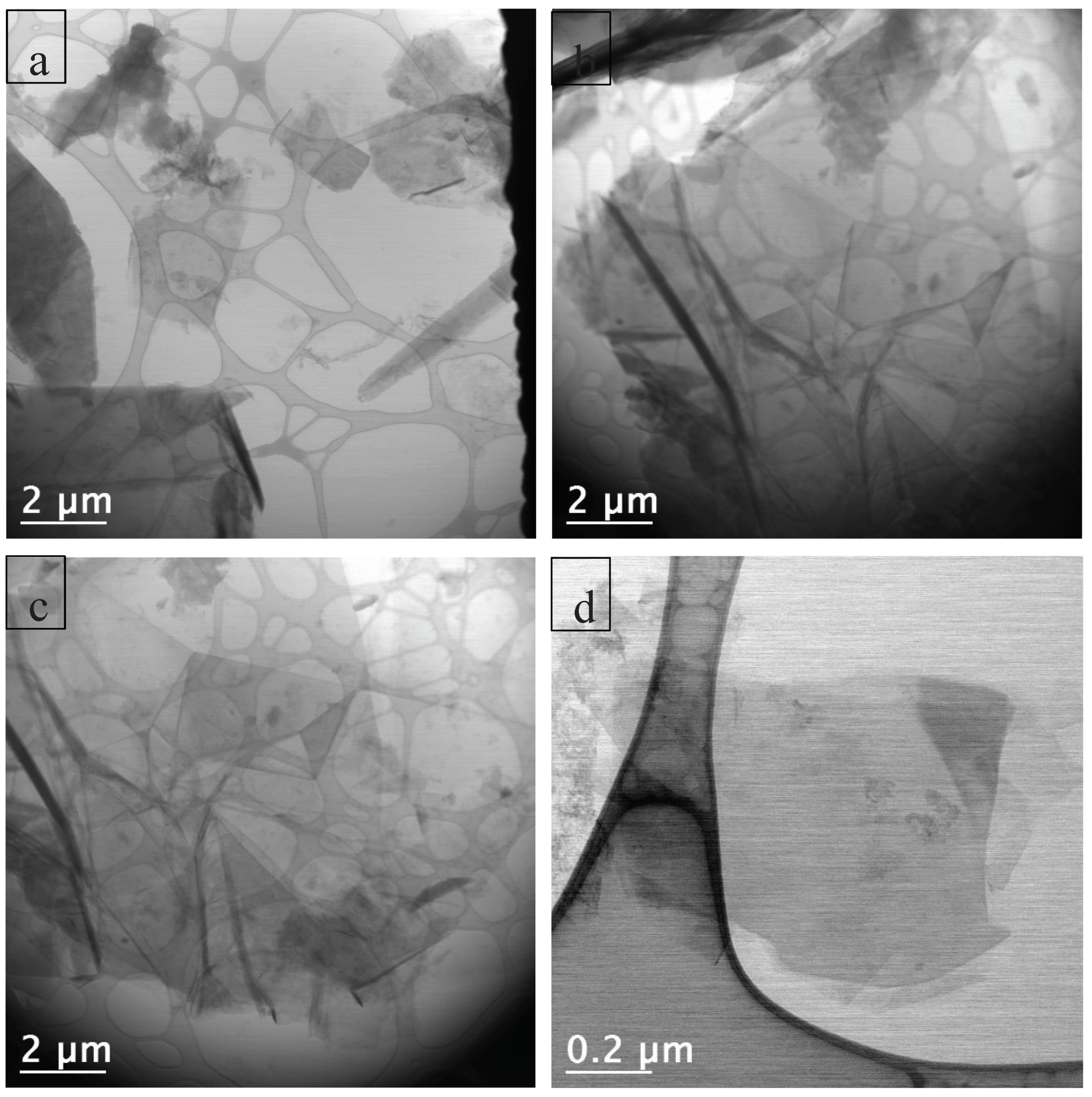
The use of scanning mode measurements (STEM – Scanning Transmission Electron Microscopy) allowed for the measurement of thin sheet dimensions and the determination of their size distribution (
Figure 10). All regions were analyzed using both BF (Bright Field) and HAADF (High Angle Annular Dark Field) detectors simultaneously. Low-magnification TEM images (10,000× and 15,000×) revealed that the C-NaOH+H₂SO₄ sample is composed of thin sheets of varying sizes (graphene and micrographite nanoplatelets). Using only manual agitation, few edge regions were suitable for analysis, and in these regions, the presence of thin sheets was observed, suggesting that the sample is composed of thin layers (graphene and micrographite nanoplatelets). The use of an ultrasonic bath for 2 minutes promoted the separation of these layers, resulting in the deposition of several thin sheets (graphene nanoplatelets) of various sizes on the grid. It is important to note that, due to the sheets being rolled, torn, and/or overlapping, the average size obtained represents only an estimate. Size distribution analysis was performed using Gatan Digital Micrograph software, and the particle size distribution histogram was prepared using Origin 8.1. The graphene nanoplates (<5 nm) are nearly transparent and with some visible wrinkles, suggesting that the samples are mainly composed of few-layer graphene (
Figure 11). For the graphene nanoplates (>5 nm), they are less transparent due to the higher thickness (
Figure 12). The micrographite has the similar grain size of ~1 μm with graphene nanoplates, but its thickness is nearly the same as the bulk graphite.
4. Conclusions
The microcrystalline graphite ore sample from Canindé, CE, was subjected to flotation tests combined with chemical purification in a basic medium followed by acid leaching. After this process, the sample exhibited high purity (99% graphite) and strong potential for graphene nanoplates and graphene-like materials production, displaying a well-defined crystalline structure and thin particles. This combination of processes disrupted the regular stacking of graphite layers, leading to the formation of graphene nanoplatelets, as evidenced by change in the Raman spectrum, FTIR, and AFM between C-Bulk sample and the C-NaOH+H₂SO₄-treated sample, AFM and TEM scan measurements showing particle sizes below 20 nm, consistent with graphene and micrographite nanoplatelets. The chemical composition of the C-NaOH+H₂SO₄ treatment sample indicates a significant reduction in impurities such as ferrimuscovite due to the removal of elements that constitute this mica. The C-Bulk sample (graphite concentrate) contains 76.6% graphitic carbon, which increases to over 99% after leaching, suggesting highly efficient purification. Acid leaching presents significant advantages, such as lower environmental impact and better control over the morphology and properties of the obtained graphene compared to traditional chemical methods that use HF. Therefore, the data obtained from XRD patterns, Raman spectroscopy, FTIR, AFM, and TEM collectively indicate that the combined purification process (basic + acidic) not only enabled the removal of mineral and metallic impurities but also promoted the reorganization and exfoliation of graphite into thinner layers. This approach allowed to produce graphene nanoplatelets without the need for sonication or harsh oxidizing agents, representing a promising and low-cost strategy for the preparation of graphene derivatives from natural sources.
Author Contributions
Conceptualization, L.S.; methodology, L.S., R.S., J.R., A.A., B.A., F.V., T.M. and J.F.; software, L.S., J.R., B.A., F.V. and T.M.; validation, L.S., J.R., A.G. and R.S.; formal analysis, L.S., J.R., A.G. and R.S; investigation, L.S., J.R. and R.S.; data curation, L.S. and J.R.; writing—original draft preparation, L.S.; writing—review and editing, L.S., J.R., A.G.; visualization, L.S., J.R., F.V. and T.M.; supervision, L.S.; project administration, L.S.; funding acquisition, L.S. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Company BrEEX (No. 20002023).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are contained within the article.
Acknowledgments
We thank the Company BREEX for providing samples and logistical support for the research; To the Federal University of Ceará - UFC and the Center for Industrial Quality and Technology of Ceará - NUTEC, which provided the necessary facilities for the completion of this work; To the Mineral Nanotechnology Research Group – UFC/NUTEC/UFSM; To the Materials Research Group - NUTEC, Advanced Materials Chemistry Group - GQMAT/UFC, Analytical Center-UFC, Structural Crystallography Laboratory – UFC, X-ray Laboratory – UFC and Ceramic Materials Laboratory - UFC for the physical structure and discussion of the results; The Ceará Foundation for Support of Scientific and Technological Development (FUNCAP) for technical scientific support.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Van Schmus, W.R.; Oliveira, E.P.; Silva Filho, A.F.; Toteu, S.F. Proterozoic links between the Borborema Province, NE Brazil, and the Central African Fold Belt. Geological Society, London, Special Publications 2011, 294, 69–99. [Google Scholar] [CrossRef]
- Hodkiewicz, J. Characterizing carbon materials with Raman spectroscopy. Thermo Scientific Application Note 2010, 51946. [Google Scholar]
- Belém, J. Caracterização mineralógica, física e termobarométrica de minérios de grafita da província grafítica Bahia-Minas. Master's Thesis, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil, 2006. [Google Scholar]
- Peng, W.; Li, H.; Hu, Y.; Liu, Y.; Song, S. Does silicate mineral impurities in natural graphite affect the characteristics of synthesized graphene? Mater. Res. Bull. 2016, 74, 333–339. [Google Scholar] [CrossRef]
- Nobre, A.G.; Martínez, J.A.E.; Terence, M.C.; Florêncio, O. The action of shear zones in the natural availability of graphite nanoplatelets: the example of the metadolomites of the Itaiacoca Group and the mica schist of the Dom Silverio Group. Brazilian Journal of Animal and Environmental Research 2020, 3, 3108–3118. [Google Scholar] [CrossRef]
- Nobre, A.G.; de Andrade, F.R.D.; Salazar-Naranjo, A.F.; Rigue, J.N.; da Silva, R.B.; Vlach, S.R.F.; et al. Electrical resistance evolution of graphite and talc geological heterostructures under progressive metamorphism. C 2023, 9, 75. [Google Scholar] [CrossRef]
- Chehreh Chelgani, S.; Rudolph, M.; Kratzsch, R.; Sandmann, D.; Gutzmer, J. A review of graphite beneficiation techniques. Miner. Process. Extr. Metall. Rev. 2016, 37, 58–68. [Google Scholar] [CrossRef]
- Vasumathi, N.; Sarjekar, A.; Chandrayan, H.; Chennakesavulu, K.; Reddy, G.R.; Vijaya Kumar, T.V.; et al. A mini review on flotation techniques and reagents used in graphite beneficiation. Int. J. Chem. Eng. 2023, 1007689, 1–15. [Google Scholar] [CrossRef]
- Ithnin, N.F.B.M.; Liu, W.W. Exfoliation of Graphite into Graphene Oxide and Reduction by Plant Extract to Synthesize Graphene. International Journal of Nanoelectronics and Materials 2024, 17. [Google Scholar]
- Nobre, A.G.; da Silva, L.P.N.; de Andrade, F.R.D. Graphene geology and the fourth industrial revolution. In Proceedings of the 7th Brazilian Technology Symposium (BTSym'21); Iano, Y.; Saotome, O.; Kemper Vásquez, G.L.; Cotrim Pezzuto, C.; Arthur, R.; Gomes de Oliveira, G., Eds.; Smart Innovation, Systems and Technologies; Springer: Cham, Switzerland, 2023; Vol. 207.
- Nobre, A.G.; Martínez, J.A.E.; Florêncio, O. Mineral nanotechnology in circular economy. In Proceedings of the 6th Brazilian Technology Symposium (BTSym'20); Iano, Y.; Saotome, O.; Kemper, G.; Mendes de Seixas, A.C.; Gomes de Oliveira, G., Eds.; Smart Innovation, Systems and Technologies; Springer: Cham, Switzerland, 2021; Vol. 233.
- Potts, J.R.; Dreyer, D.R.; Bielawski, C.W.; Ruoff, R.S. Graphene-based polymer nanocomposites. Polymer 2011, 52, 5–25. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, Y.; Wang, M.; et al. A powder-metallurgy-based strategy toward three-dimensional graphene-like network for reinforcing copper matrix composites. Nat. Commun. 2020, 11, 2775. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Wang, C.; Zeng, M.; Fu, L. Graphene: an outstanding multifunctional coating for conventional materials. Small 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Kulyk, B.; Freitas, M.A.; Santos, N.F.; Mohseni, F.; Carvalho, A.F.; Yasakau, K.; et al. A critical review on the production and application of graphene and graphene-based materials in anti-corrosion coatings. Crit. Rev. Solid State Mater. Sci. 2021, 47, 309–355. [Google Scholar] [CrossRef]
- Belov, D.V.; Belyaev, S.N.; Radishchev, D.B.; et al. Synthesis of coatings based on graphene-like materials and study of their physicochemical properties. Inorg. Mater. Appl. Res. 2024, 15, 506–522. [Google Scholar] [CrossRef]
- Wakamatsu, T.; Numata, Y. Flotation of graphite. Minerals Engineering 1991, 4, 975–982. [Google Scholar] [CrossRef]
- Wang, H.; Feng, Q.; Liu, K.; Zuo, K.; Tang, X. A novel technique for microcrystalline graphite beneficiation based on alkali-acid leaching process. Sep. Sci. Technol. 2017, 53, 982–989. [Google Scholar] [CrossRef]
- Xiyue, Z.; Hongjuan, S.; Tongjiang, P.; Li, Z.; Bo, L. Purification mechanism of microcrystalline graphite and dissolution of non-carbon impurity during alkali autoclave-acid leaching. Phys. Chem. Miner. 2024. [Google Scholar] [CrossRef]
- Sala, O. Fundamentos da espectroscopia Raman e no infravermelho, 2nd ed.; UNESP: São Paulo, Brazil, 2008. [Google Scholar]
- Ferrari, A.C. Raman spectroscopy of graphene and graphite: Disorder, electron–phonon coupling, doping and nonadiabatic effects
. Solid State Commun. 2007, 143, 47–57. [Google Scholar] [CrossRef]
- Cuesta-López, S.; Fernández, M.L.; Menendez-Aguado, J.M.; Ricci, C. Raman Spectroscopy as a tool for identification and valorisation assessment of critical raw materials: Graphite case. In Proceedings of the Mineral Exploration Symposium; 2020. [Google Scholar]
- Shokry, S.A.; Elmorsi, A.K.; Elsrogy, H. Preparation and Characterization of Chemically Converted Graphene from Natural Graphite. Egypt. J. Petrol. 2024, 33. [Google Scholar] [CrossRef]
- Xie, X.; Zhang, J.; Chen, Y.; Wang, C. A method for the preparation of graphene from spent graphite of retired lithium-ion batteries. J. Power Sources 2024, 594. [Google Scholar] [CrossRef]
- Hontoria-Lucas C, López-Peinado AJ, López-González JD, Rojas-Cervantes ML, Martín-Aranda RM. Study of oxygen-containing groups in a series of graphite oxides: Physical and chemical characterization. Carbon, 1995, 33, 1585–1592.
- Dreyer DR, Park S, Bielawski CW, Ruoff RS. The chemistry of graphene oxide. Chem. Soc. Rev., 2010, 39, 228–240.
- Zólyomi V, Koltai J, Kürti J. Resonance Raman spectroscopy of graphite and graphene. Phys. Status Solidi B, 2011, 248, 2435–2444.
- Cançado, L.G.; Monken, V.P.; Campos, J.L.E.; Santos, J.C.C.; Backes, C.; Chacham, H.; et al. Science and Metrology of defects in graphene using Raman Spectroscopy. Carbon 2024, 220. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Meyer, J.C.; Scardaci, V.; Casiraghi, C.; Lazzeri, M.; Mauri, F.; et al. Raman spectrum of graphene and graphene layers. Phys. Rev. Lett. 2006, 97, 187401. [Google Scholar] [CrossRef]
- Lu, S.; Yao, M.; Yang, X.; Li, Q.; Xiao, J.; Yao, Z.; et al. High-pressure transformation of graphene nanoplates: A Raman study. Chem. Phys. Lett. 2013, 585, 101–106. [Google Scholar] [CrossRef]
- Panyathip, R.; Sucharitakul, S.; Phaduangdhitidhada, S.; Ngamjarurojana, A.; Kumnorkaew, P.; Choopun, S. Surface Enhanced Raman Scattering in Graphene Quantum Dots Grown via Electrochemical Process. Molecules 2021, 26. [Google Scholar] [CrossRef]
- Pimenta, M.A.; Dresselhaus, G.; Dresselhaus, M.S.; Cançado, L.G.; Jorio, A.; Saito, R. Studying disorder in graphite-based systems by Raman spectroscopy. Phys. Chem. Chem. Phys. 2007, 9, 1276–1290. [Google Scholar] [CrossRef]
- Sparavigna, A.C. Graphene, Graphene Oxide and Carbon Nanotubes in Raman Spectroscopy. Int. J. Sci. 2024, 13. [Google Scholar] [CrossRef]
- Wu, W.; Ranasinghe, J.C.; Chatterjee, A.; Huang, S. Recent advances on Raman spectroscopy of graphene: towards biosensing applications. Mater. Chem. Phys. 2024, 318. [Google Scholar] [CrossRef]
- Ahmad, I.; McCarthy, J.E.; Baranov, A.; Gun’ko, Y.K. Development of Graphene Nano-Platelet Based Counter Electrodes for Solar Cells. Materials 2015, 8, 5953–5973. [Google Scholar] [CrossRef]
- Bokobza L, Bruneel JL, Couzi M. Raman spectra of carbon-based materials (from graphite to carbon black) and of some silicone composites. C, 2015, 1, 77–94.
- Ramirez C, Osendi MI. Characterization of graphene nanoplatelets-Si3N4 composites by Raman spectroscopy. J. Eur. Ceram. Soc., 2013, 33, 471–477.
- Ojrzyńska M, Jamroz J, Maciałowicz M, Wilczyński K, Daniszewska A, Antonowicz J, Zdrojek M. Quality and thickness control of graphene nanoplatelets during large-scale production using liquid phase exfoliation. Mater. Today Commun., 2025, 42, 111543.
- Muzyka R, Drewniak S, Pustelny T, Chrubasik M, Gryglewicz G. Characterization of graphite oxide and reduced graphene oxide obtained from different graphite precursors and oxidized by different methods using Raman spectroscopy. Materials, 2018, 11, 1050.
- Patel J, Kiani A. Comparative study of tribological behaviours of different base greases enhanced by graphene nano platelets. Ind. Lubr. Tribol., 2019, 71, 1054–1061.
- Lavini F, Cellini F, Rejhon M, Kunc J, Berger C, Heer W, Riedo E. Atomic force microscopy phase imaging of epitaxial graphene films. J. Phys. Mater., 2020, 3, 045001.
Figure 1.
Flowsheet for graphite flotation (Final test).
Figure 1.
Flowsheet for graphite flotation (Final test).
Figure 2.
Flowchart of the integrated alkali-acid treatments for graphite purification.
Figure 2.
Flowchart of the integrated alkali-acid treatments for graphite purification.
Figure 3.
Rietveld refinement of C-Bulk sample. X-ray diffraction patterns (black circles) together with the result of the calculated pattern obtained with Rietveld refinement (red line). The green line is the difference between the experimental and calculated intensities.
Figure 3.
Rietveld refinement of C-Bulk sample. X-ray diffraction patterns (black circles) together with the result of the calculated pattern obtained with Rietveld refinement (red line). The green line is the difference between the experimental and calculated intensities.
Figure 4.
Rietveld refinement of C-NaOH+H2SO4 treatment sample. X-ray diffraction patterns (black circles) together with the result of the calculated pattern obtained with Rietveld refinement (red line). The green line is the difference between the experimental and calculated intensities.
Figure 4.
Rietveld refinement of C-NaOH+H2SO4 treatment sample. X-ray diffraction patterns (black circles) together with the result of the calculated pattern obtained with Rietveld refinement (red line). The green line is the difference between the experimental and calculated intensities.
Figure 5.
FTIR spectrum of the C-Bulk, C-NaOH treatment and C-NaOH+H2SO4 treatment samples.
Figure 5.
FTIR spectrum of the C-Bulk, C-NaOH treatment and C-NaOH+H2SO4 treatment samples.
Figure 6.
Raman spectra with the deconvolution of 2D peaks (two spectral components). Raman spectra (a) C-Bulk, (c) C-NaOH and (e) C-NaOH+H2SO4. Deconvolution of 2D peaks (b) C-Bulk, (d) C-NaOH and (f) C-NaOH+H2SO4.
Figure 6.
Raman spectra with the deconvolution of 2D peaks (two spectral components). Raman spectra (a) C-Bulk, (c) C-NaOH and (e) C-NaOH+H2SO4. Deconvolution of 2D peaks (b) C-Bulk, (d) C-NaOH and (f) C-NaOH+H2SO4.
Figure 7.
Raman spectra with the deconvolution of 2D peaks (a spectral component). Raman spectra (a) C-Bulk, (c) C-NaOH and (e) C-NaOH+H2SO4. Deconvolution of 2D peaks (b) C-Bulk, (d) C-NaOH and (f) C-NaOH+H2SO4.
Figure 7.
Raman spectra with the deconvolution of 2D peaks (a spectral component). Raman spectra (a) C-Bulk, (c) C-NaOH and (e) C-NaOH+H2SO4. Deconvolution of 2D peaks (b) C-Bulk, (d) C-NaOH and (f) C-NaOH+H2SO4.
Figure 8.
AFM topographic images of dispersions from the three samples. A gradual reduction in the maximum particle size within the scanned regions can be observed. Panels (a), (c), and (e) show the AFM scans of the C-Bulk, C-NaOH, and C-NaOH+H₂SO₄ samples, respectively. Panels (b), (d), and (f) present the cross-sectional profiles highlighted in the corresponding AFM scans (a), (c), and (e), respectively.
Figure 8.
AFM topographic images of dispersions from the three samples. A gradual reduction in the maximum particle size within the scanned regions can be observed. Panels (a), (c), and (e) show the AFM scans of the C-Bulk, C-NaOH, and C-NaOH+H₂SO₄ samples, respectively. Panels (b), (d), and (f) present the cross-sectional profiles highlighted in the corresponding AFM scans (a), (c), and (e), respectively.
Figure 9.
a), (d), and (g) show the AFM scans of the highlighted in
Figure 8. C-Bulk, C-NaOH, and C-NaOH+H₂SO₄ samples, respectively. The cross-sectional profiles extracted from each scan are shown adjacent to the corresponding topographic images, along with the respective phase images, where increased contrast can be observed in the analyzed regions.
Figure 9.
a), (d), and (g) show the AFM scans of the highlighted in
Figure 8. C-Bulk, C-NaOH, and C-NaOH+H₂SO₄ samples, respectively. The cross-sectional profiles extracted from each scan are shown adjacent to the corresponding topographic images, along with the respective phase images, where increased contrast can be observed in the analyzed regions.
Figure 10.
C-NaOH+H₂SO₄ sample (graphene and micrographite nanoplatelets): (a) HAADF image illustrating the measurement of the thin sheet dimensions. (b) Size distribution histogram of the thin sheets. This histogram includes only the regions where sheet edges were clearly visible for measurement. Due to folding, tearing, and/or overlapping of the sheets, these measurements represent only an estimate.
Figure 10.
C-NaOH+H₂SO₄ sample (graphene and micrographite nanoplatelets): (a) HAADF image illustrating the measurement of the thin sheet dimensions. (b) Size distribution histogram of the thin sheets. This histogram includes only the regions where sheet edges were clearly visible for measurement. Due to folding, tearing, and/or overlapping of the sheets, these measurements represent only an estimate.
Figure 11.
C-NaOH+H₂SO₄ sample (graphene and micrographite nanoplatelets) - TEM images of (a), (b), (c) and (d): The graphene nanoplatelets (<5 nm) are nearly transparent, suggesting few-layer graphene. The graphene nanoplatelets (>5 nm) and micrographite are less transparent due to the higher thickness.
Figure 11.
C-NaOH+H₂SO₄ sample (graphene and micrographite nanoplatelets) - TEM images of (a), (b), (c) and (d): The graphene nanoplatelets (<5 nm) are nearly transparent, suggesting few-layer graphene. The graphene nanoplatelets (>5 nm) and micrographite are less transparent due to the higher thickness.
Figure 12.
(a) BF image of a region of the grid containing the sample, where structures of different sizes can be identified. A large thin sheet is visible at the center of the image. (b) Folded graphene nanoplatelets.
Figure 12.
(a) BF image of a region of the grid containing the sample, where structures of different sizes can be identified. A large thin sheet is visible at the center of the image. (b) Folded graphene nanoplatelets.
Table 1.
Refinement parameters (Rietveld method) including the lattice parameters, phase, crystallite size calculated by the Scherrer equation for the C-Bulk and C-NaOH+H2SO4 treatment samples.
Table 1.
Refinement parameters (Rietveld method) including the lattice parameters, phase, crystallite size calculated by the Scherrer equation for the C-Bulk and C-NaOH+H2SO4 treatment samples.
| C-Bulk |
Chemical formula |
Space Group |
Weight Fraction (%) |
Lattice Parameters (Å, °) |
Cell Volume (ų) |
Rwp (%) |
GOF (χ²) |
Crystallite Size (nm) |
| Muscovite |
KAl₂(AlSi₃O₁₀)(OH)₂ |
C 1 2/c 1 |
0.1 |
a = 13.20
b = 4.84
c = 14.66
β = 68.89° |
875.86 |
16.9 |
1.53 |
19.01 |
| Graphite |
C |
P 63/m m c |
71.4 |
a = 2.463;
c = 6.719 |
35.330 |
83.04 |
| Quartz |
SiO₂ |
P 31 2 1 |
28.5 |
a = 4.928;
c = 5.519 |
116.125 |
42.30 |
| C-NaOH+H2SO4 treatment |
Chemical formula |
Space Group |
Weight Fraction (%) |
Lattice Parameters (Å, °) |
Cell Volume (ų) |
Rwp (%) |
GOF (χ²) |
Crystallite Size (nm) |
| Graphite |
C |
P 63/m m c |
100.00 |
a = 2.53777
c = 6.77568 |
37.791 |
15.02 |
1.532 |
130 |
Table 2.
Chemical composition of the samples: graphite ore - Stage 1, C-Bulk – Stage 2, C-NaOH treatment - Stage 3, and C-NaOH+H2SO4 treatment - Stage 4.
Table 2.
Chemical composition of the samples: graphite ore - Stage 1, C-Bulk – Stage 2, C-NaOH treatment - Stage 3, and C-NaOH+H2SO4 treatment - Stage 4.
Analyses
Method
Unit
Detection Limit |
SiO2
XRF
%
0.01 |
Al2O3
XRF
%
0.01 |
CaO
XRF
%
0.01 |
MgO
XRF
%
0.01 |
K2O
XRF
%
0.01 |
TiO2
XRF
%
0.01 |
MnO
XRF
%
0.01 |
Fe2O3
XRF
%
0.01 |
SO3
XRF
%
0.01 |
Cl
XRF
%
0.01 |
P2O5
XRF
%
0.01 |
Co2O3
XRF
%
0.01 |
Rh2O3
XRF
%
0.01 |
| Stage 1 |
42.43 |
18.88 |
2.19 |
1.06 |
7.55 |
2.69 |
2.69 |
23.81 |
0.34 |
0.62 |
0.00 |
0.00 |
0.00 |
| Stage 2 |
31.02 |
15.65 |
2.44 |
0.00 |
6.32 |
2.90 |
0.54 |
36.16 |
0.37 |
0.75 |
3.67 |
0.18 |
0.00 |
| Stage 3 |
29.75 |
12.88 |
2.61 |
1.06 |
5.13 |
3.52 |
0.34 |
39.78 |
0.00 |
0.00 |
3.73 |
0.00 |
1.22 |
| Stage 4 |
75.65 |
3.63 |
4.70 |
0.00 |
3.71 |
0.00 |
0.00 |
0.86 |
0.00 |
0.00 |
11.45 |
0.00 |
0.00 |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).