Submitted:
21 March 2025
Posted:
25 March 2025
You are already at the latest version
Abstract
Keywords:
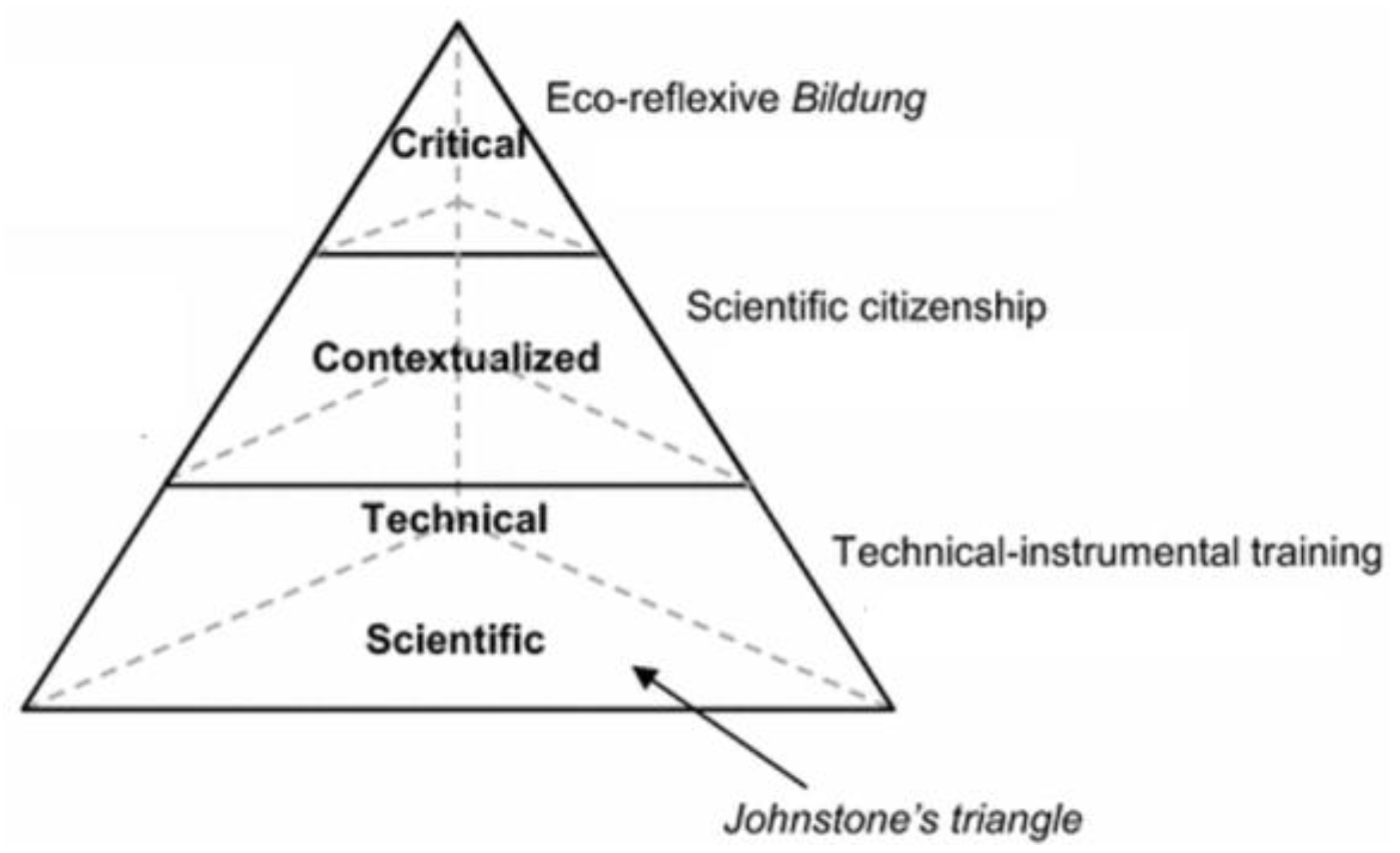
1. Introduction
2. Macroscopic Level
2.1. Physical and Chemical Properties
2.2. Some Typical Reactions and Analytical Methods
2.3. Methods of Preparation
2.4. Some Historical Notes
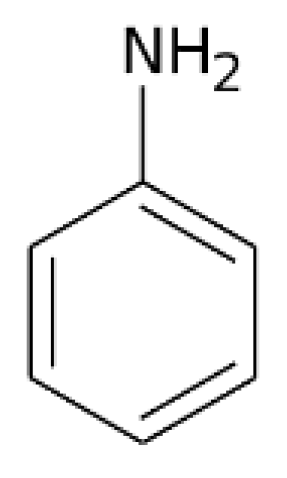
3. Symbolic Level
3.1. Formula, Basic Character, Diagrams
3.2. Reactions
- (1)
- HNO2 + H+ + e- → NO + H2O ΔrG°1 = - F∙E°1
- (2)
-
NO3- + 3 H+ + 2 e- → HNO2 + H2O ΔrG°2 = - 2F∙E°2Linear combination 2(1) – (2):
- (3)
- 3 HNO2(aq) → H+(aq) + NO3-(aq) + 2 NO(g) + H2O(l) ΔrG°3 = - R∙T∙lnK°
3.3. Synthesis
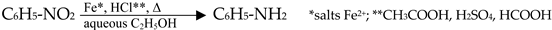
3.4. The Mauveine
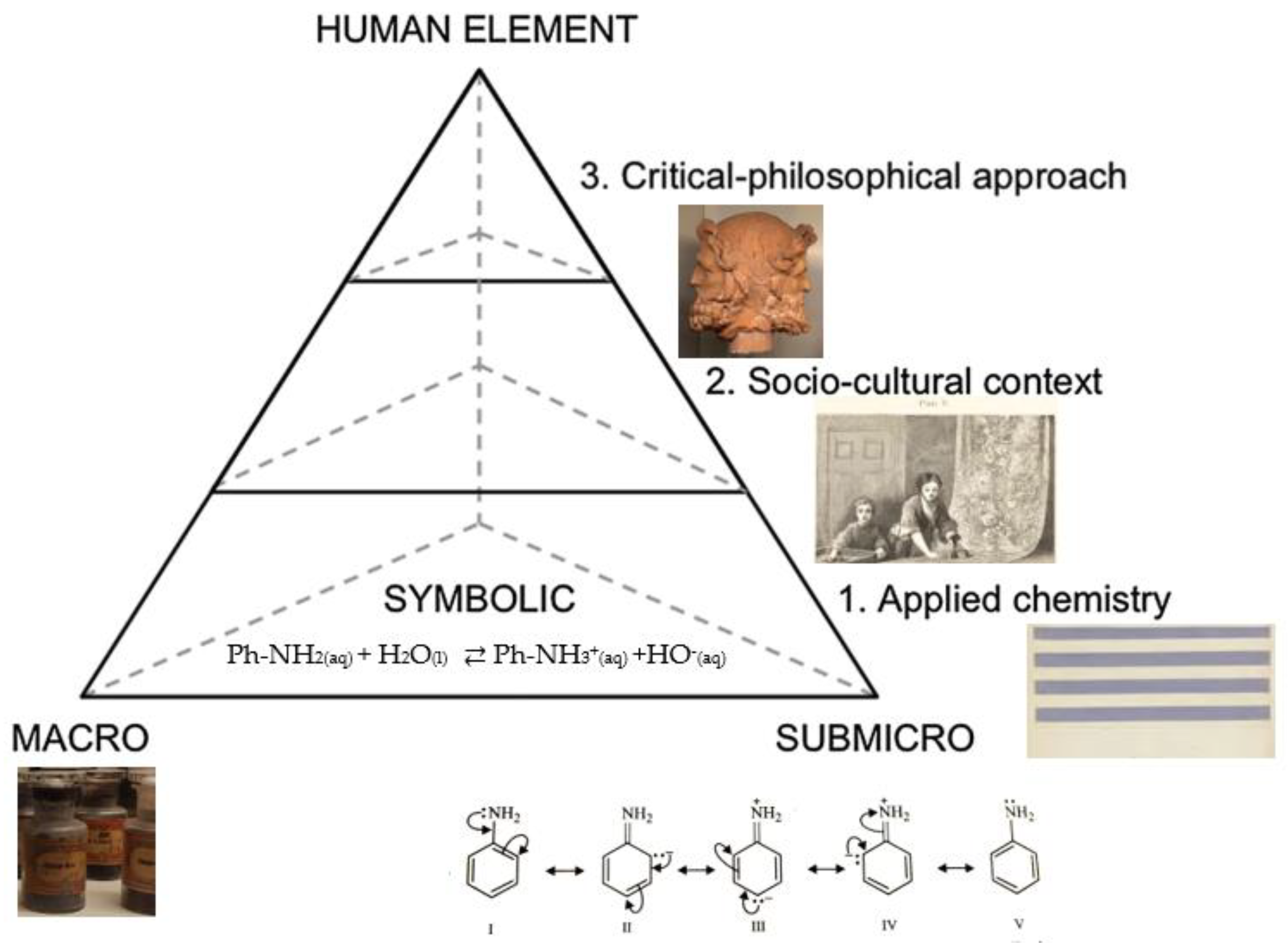
4. Sub-Microscopic Level
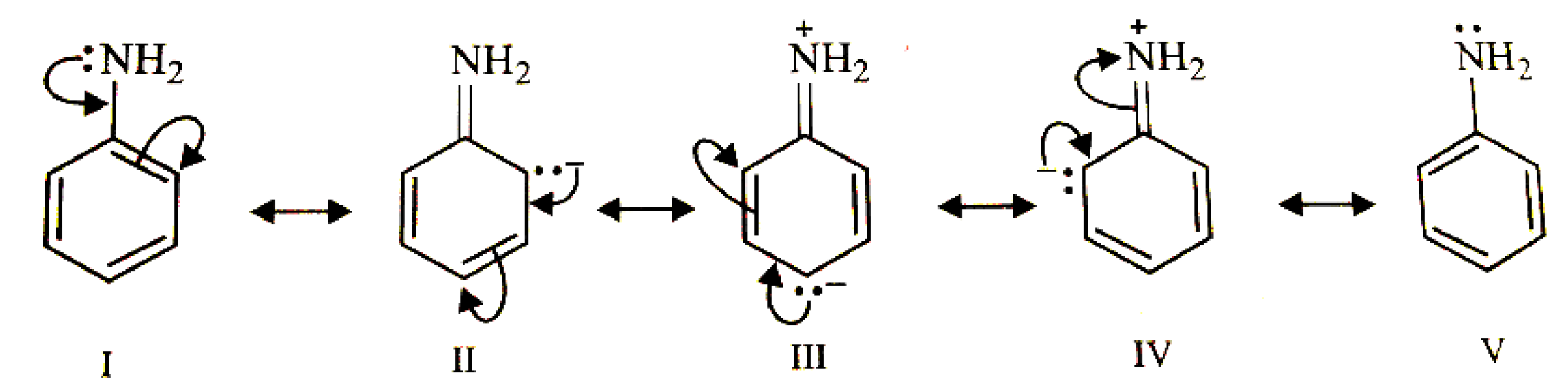
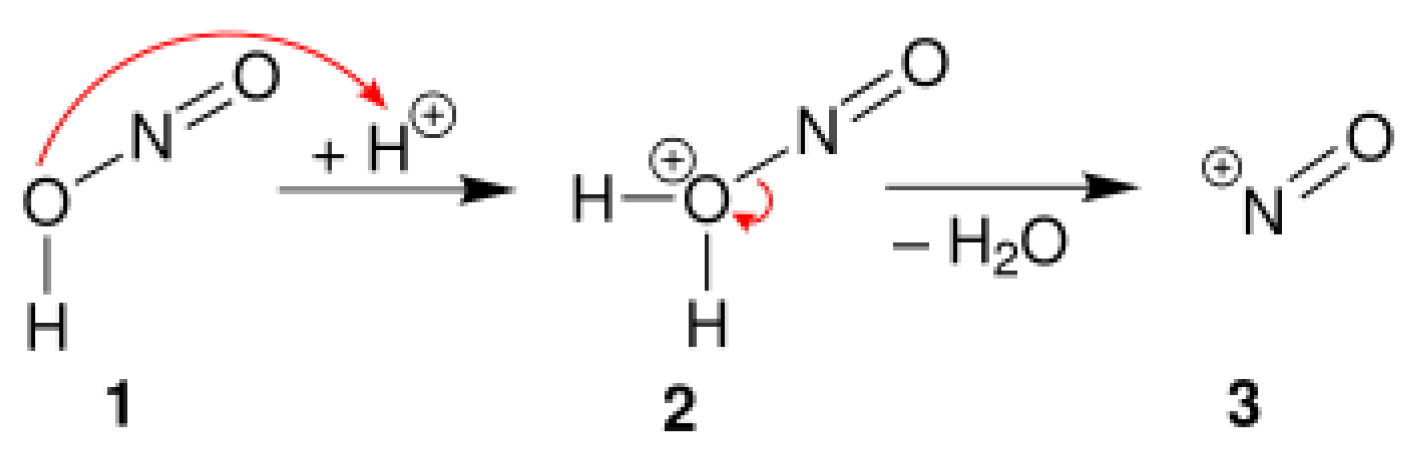
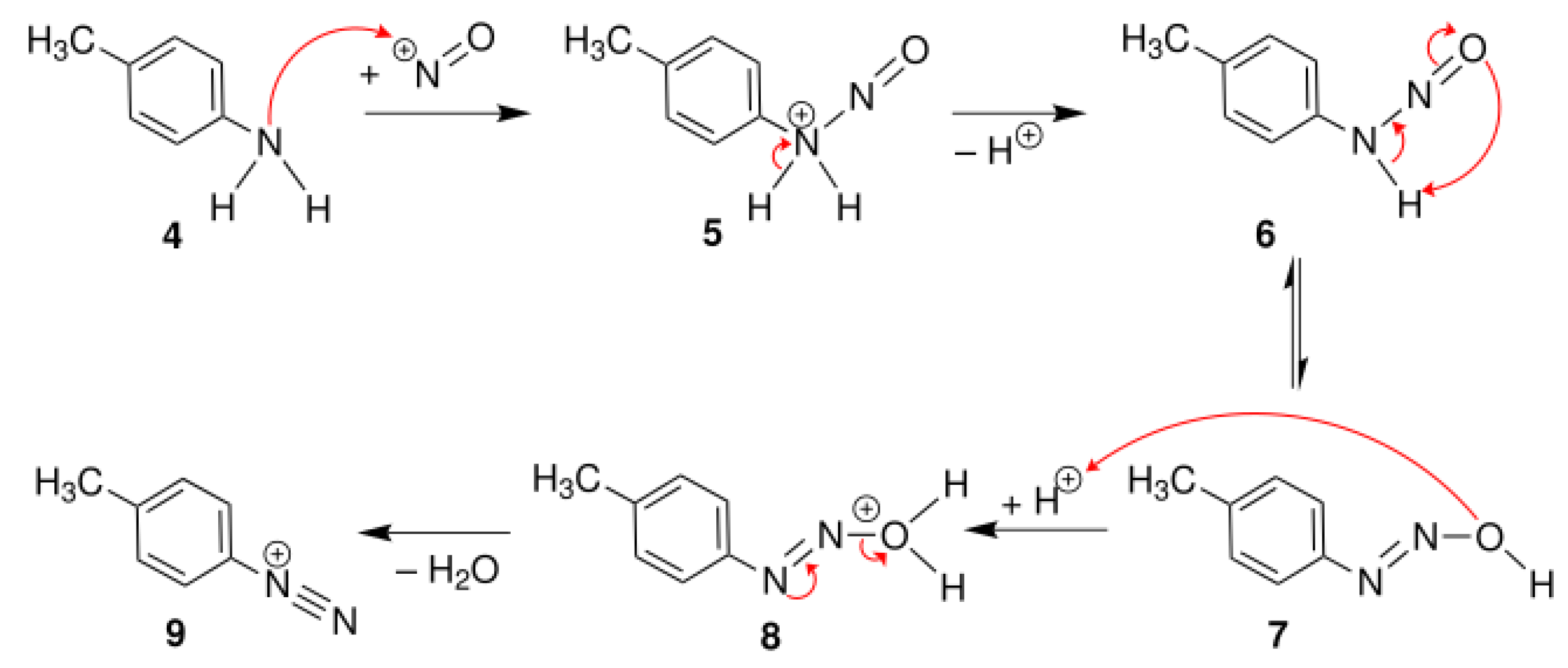
4.1. Diazotization Reaction
4.2. Preparation of Ammonium Salts
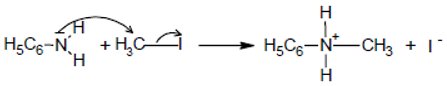
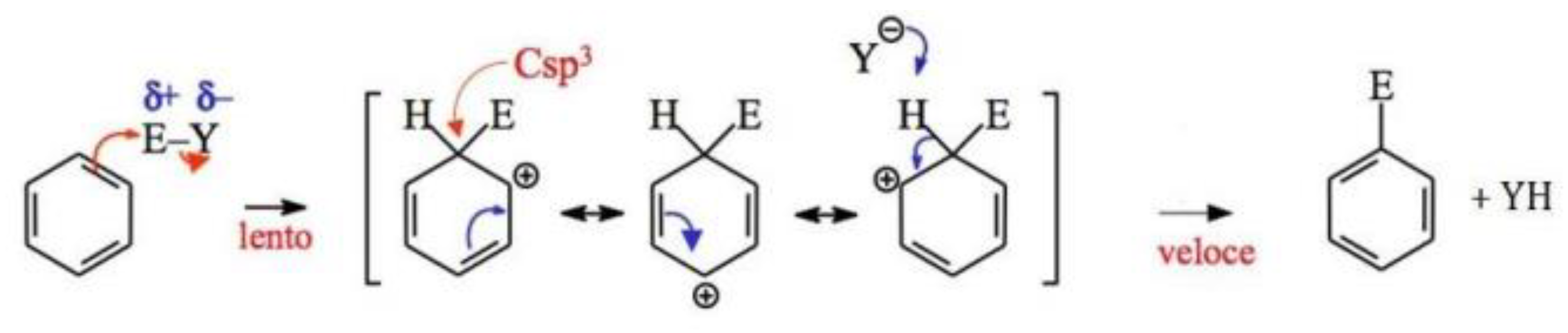
4.3. Electrophilic Aromatic Substitutions
5. Applied Chemistry
6. Socio-Cultural Context
7. Critical-Philosophical Approach
8. Conclusions
Funding
Conflicts of Interest
| 1 | The tables referred to are found on an unnumbered page, between p. 292 and p. 293. |
References
- Scerri, E.R.; McIntyre, L. The Case for the Philosophy of Chemistry. Synthese 1997, 111, 213–232. [Google Scholar] [CrossRef]
- Johnstone, A.H. Thinking about Thinking. International Newsletter of Chemical Education 1991, 36, 7–10. [Google Scholar]
- Mahaffy, P. The Future Shape of Chemistry Education. Chem. Educ. Res. Pract 2004, 5, 229–245. [Google Scholar] [CrossRef]
- Sjöström, J. Towards Bildung-Oriented Chemistry Education. Sci. Educ. 2013, 22, 1873–1890. [Google Scholar] [CrossRef]
- Hagan, E.; Poulin, J. Statistics of the Early Synthetic Dye Industry. Herit. Sci. 2021, 9, 33. [Google Scholar] [CrossRef]
- Sjöström, J. The Discourse of Chemistry (and Beyond). HYLE 2007, 2, 83–97., J. The Discourse of Chemistry (and Beyond). Hyle 2007, 2, 83–97. [Google Scholar]
- Sjöström, J.; Eilks, I. Correction to: The Bildung Theory—From von Humboldt to Klafki and Beyond. In Science Education in Theory and Practice: An Introductory Guide to Learning Theory; Akpan, B., Kennedy, T.J., Eds.; Springer International Publishing: Cham, 2020; pp. C1–C1. [Google Scholar] [CrossRef]
- Schummer, J. Why Chemists Need Philosophy, History, and Ethics. Substantia 2, 5–6. [CrossRef]
- National Center for Biotechnology Information. PubChem Compound Summary for CID 6115, Aniline, 2025. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Aniline (accessed on 13 March 2025).
- Griswold, J.; Andres, D.; Arnett, E.F.; Garland, F.M. Liquid–Vapor Equilibrium of Aniline–Water. Ind. Eng. Chem. 1940, 32, 878–880. [Google Scholar] [CrossRef]
- Ahmad, N. The Oxidation of Aromatic Amines by Sodium Hypochlorite, University of Surrey, Guildford, 1970.
- Friedemann, T.E.; Keegan, P.K.; Witt, N.F. Determination of Furan Aldehydes. Reaction with Aniline in Acetic and Hydrochloric Acid Solutions. Anal. Biochem. 1964, 8, 300–311. [Google Scholar] [CrossRef]
- Dichromate Test for Aniline | Acidified Potassium Dichromate Reaction With Aniline; BMH Learning. Available online: https://www.youtube.com/watch?v=X181RA8G4sM (accessed on 13 March 2025).
- Meyer, A.S.; Boyd, C.M. Determination of Water by Titration with Coulometrically Generated Karl Fischer Reagent. Anal. Chem. 1959, 31, 215–219. [Google Scholar] [CrossRef]
- Yates, E.; Yates, A. Johann Peter Griess FRS (1829–88): Victorian Brewer and Synthetic Dye Chemist. Notes Rec. 2016, 70, 65–81. [Google Scholar] [CrossRef]
- Wisniak, J. Pierre Jacques Antoine Béchamp. Contributions to Chemistry. Rev. CENIC Cienc. Quím. 2020, 51, 114–125. [Google Scholar]
- Clark, J. Preparation of Phenylamine Compounds. Available online: https://chem.libretexts.org/@go/page/3993?pdf (accessed on 14 March 2025).
- Anilina. Chimica; Le garzantine; Garzanti: Milano, 2002; p. 77. [Google Scholar]
- Farmer, S.; Kennepohl, D.; Kabrhel, J.; Roberts, J.; Caserio, M.C. Benzyne. Available online: https://chem.libretexts.org/@go/page/31580?pdf (accessed on 14 March 2025).
- Maar, J.H. Friedlieb Ferdinand Runge (1794-1867) – An Unusual Chemist. Substantia 2025, 9, 73–87. [Google Scholar] [CrossRef]
- Califano, S. Storia Della Chimica. Vol. II. Dalla Chimica Fisica Alle Molecole Della Vita, Digital edition.; Bollati Boringhieri: Torino.
- Plater, M.J.; Raab, A. Who Made Mauveine First: Runge, Fritsche, Beissenhirtz or Perkin? Journal of Chemical Research 2016, 40, 758–762. [Google Scholar] [CrossRef]
- Crookes, W. A Practical Handbook of Dyeing and Calico-Printing; Longmans, Green and Co.: London (UK), 1874. [Google Scholar]
- Iuliano, A. The dye that revolutionised chemistry: Perkin and the discovery of mauveine. Research for Cultural Heritage. Available online: https://researcheritage-eng.blogspot.com/2019/07/Perkin-and-the-discovery-of-mauveine.html (accessed on 14 March 2025).
- Garfield, S. Il Malva Di Perkin. Storia Del Colore Che Ha Cambiato Il Mondo; Garzanti: Milano, 2002. [Google Scholar]
- Sella, A. Karl Fischer’s Titrator. Chemistry World. December 3, 2012. Available online: https://www.chemistryworld.com/opinion/karl-fischers-titrator/5695.article (accessed on 14 March 2025).
- Excess Aniline Undergoes Alkylation with Methyl Iodide to Yield Which of the Following?; doubtbut by Allen. Available online: https://www.doubtnut.com/qna/256666655 (accessed on 13 March 2025).
- D’Auria, M. L’evoluzione Del Pensiero Nello Studio Delle Reazioni Di Sostituzione Elettrofila Aromatica. Thought Evolution in the Study of Electrophilic Aromatic Substitution Reactions Proposed Different. Chim. Sc. 2002, No. 2, 53–56. [Google Scholar]
- Emanuele, L.; D’Auria, M. The Use of Heterocyclic Azo Dyes on Different Textile Materials: A Review. Organics 2024, 5, 277–289. [Google Scholar] [CrossRef]
- Chauhan, N.P.S.; Mozafari, M. Chapter 1 - Polyaniline: An Introduction and Overview. In Fundamentals and Emerging Applications of Polyaniline; Mozafari, M., Chauhan, N.P.S., Eds.; Elsevier, 2019; pp 1–15. [CrossRef]
- Abu-Thabit, N.Y. Chemical Oxidative Polymerization of Polyaniline: A Practical Approach for Preparation of Smart Conductive Textiles. J. Chem. Educ. 2016, 93, 1606–1611. [Google Scholar] [CrossRef]
- Martinsen, H.E.H. Fashionable Chemistry: The History of Printing Cotton in France in the Second Half of the Eighteenth and First Decades of the Nineteenth Century, University of Toronto, Toronto (CA), 2015.
- Gillispie, C.C. The Natural History of Industry. Isis 1957, 48, 398–407. [Google Scholar] [CrossRef]
- Celestino, T. A Practical Handbook of Dyeing and Calico-Printing: Il Secolo Del Colore Si Mostra al Mondo. Rend Accad Naz XL 2024, V (1), 1–7. [Google Scholar]
- Crookes, W. A Recent Triumph of Synthetical Chemistry. Quartely Journal of Science, 1869; 360–362. [Google Scholar]
- Tamburini, D.; Sabatini, F.; Berbers, S.; van Bommel, M.R.; Degano, I. An Introduction and Recent Advances in the Analytical Study of Early Synthetic Dyes and Organic Pigments in Cultural Heritage. Heritage 2024, 7, 1969–2010. [Google Scholar] [CrossRef]
- Romagnoli, A. William Perkin e Il Colora Malva: La Prima Tinta Sintetica. Available online: https://www.missdarcy.it/william-perkin-e-il-color-malva-la-prima-tinta-sintetica/ (accessed on 14 March 2025).
- Beer, T. The Mauve Decade: American Life at the End of the Nineteenth Century; Alfred A. Knopf: New York, 1926. [Google Scholar]
- Daniel, G. Decorative Arts. In Art nouveau: art and design at the turn of the century; Selz, P., Constantine, M., Eds.; The Museum of Modern Art: New York, 1959; pp. 86–121. [Google Scholar]
- Dickens, C.J.H. Oliver Twist; Richard Bentley, 1838.
- Di Martiis, M.S. Lavoro e Salute in Europa Prima Della Rivoluzione Industriale. RIMP 2010, No. 1, 119–162. [Google Scholar]
- Franchini, E. Manifesti e Fogli Volanti Del Movimento Operaio Del Primo Novecento, 2013. Available online: https://filstoria.hypotheses.org/9924 (accessed on 14 March 2025).
- Polanská, K. The Legacy of Emmeline Pankhurst in the British Society, Univerzita Palackého v Olomouci, Olomouc, 2016.
- Hoffmann, R. The Same and Not the Same; Columbia University Press: New York, 1995. [Google Scholar]
- Celestino, T. High School Sustainable and Green Chemistry: Historical–Epistemological and Pedagogical Considerations. Sustainable Chemistry 2023, 4, 304–320. [Google Scholar] [CrossRef]
- Ethics of Chemistry From Poison Gas to Climate Engineering, Digital Edition. ; Schummer, J., Børsen, T., Eds.; World Scientific Publishing: Singapore, Hackensack, London, 2021. [Google Scholar]
- Riesmeier, M. Can Chemical Substances Be Natural? Ambix 2025, 72, 58–76. [Google Scholar] [CrossRef] [PubMed]

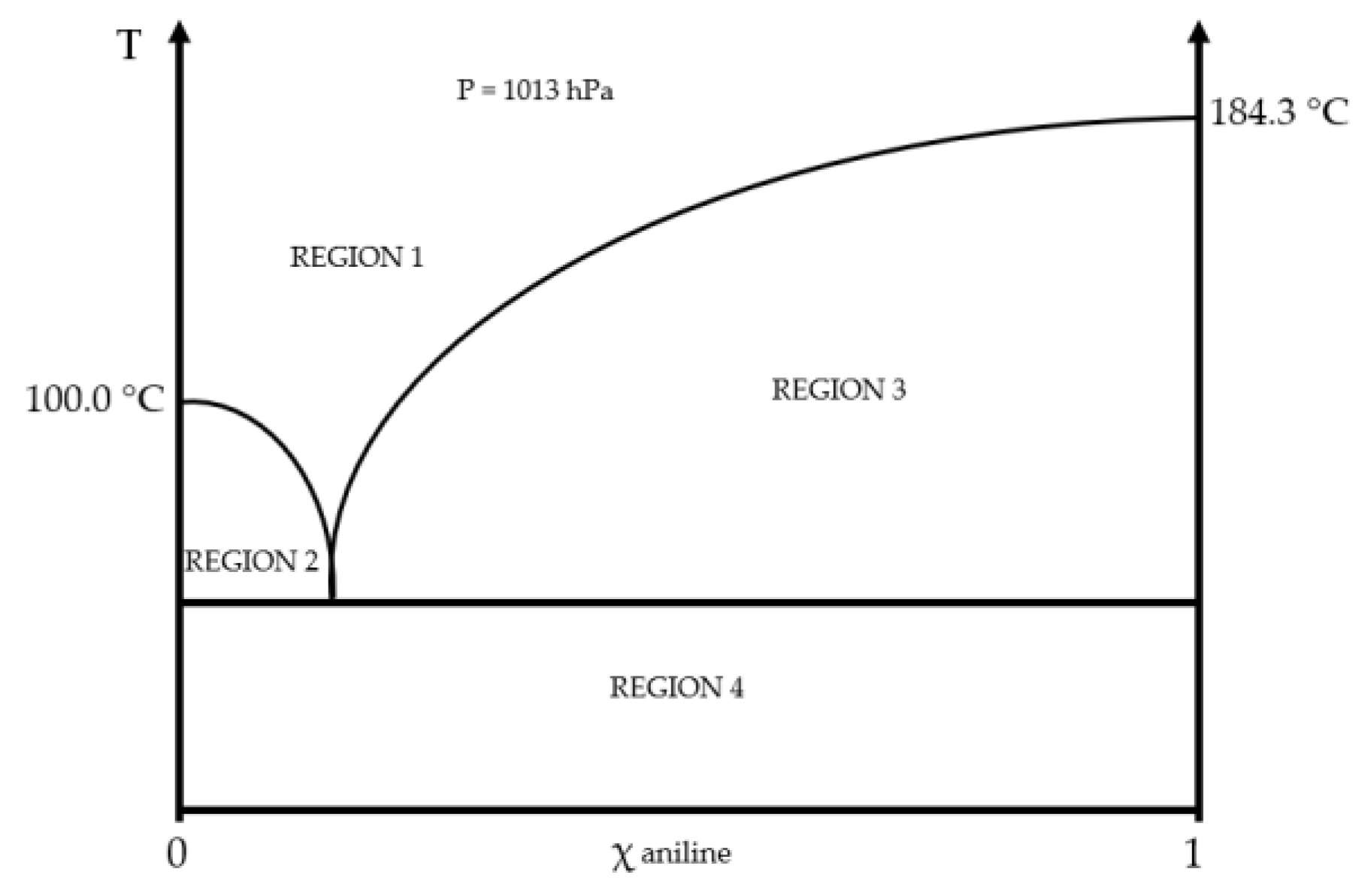
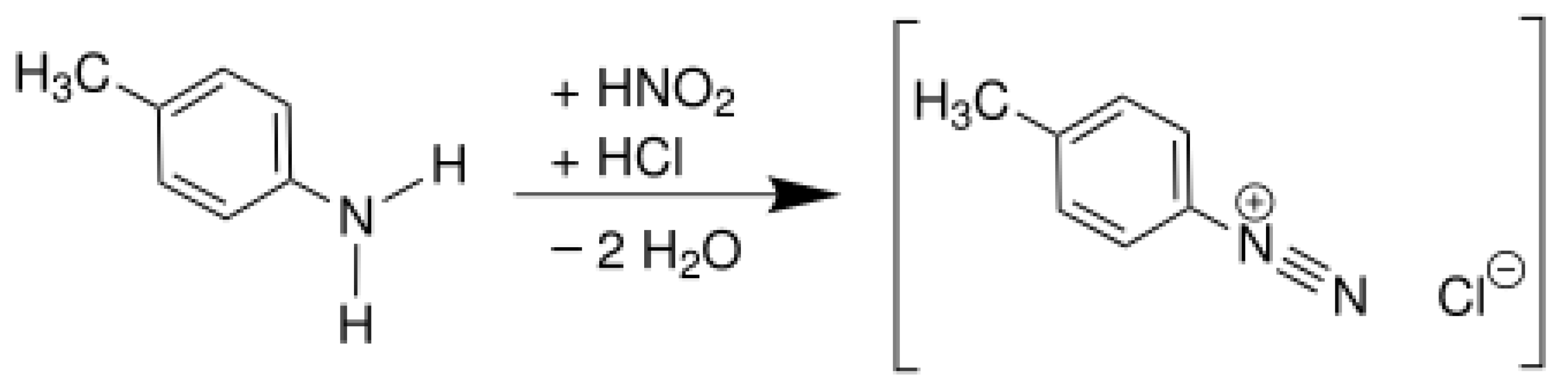
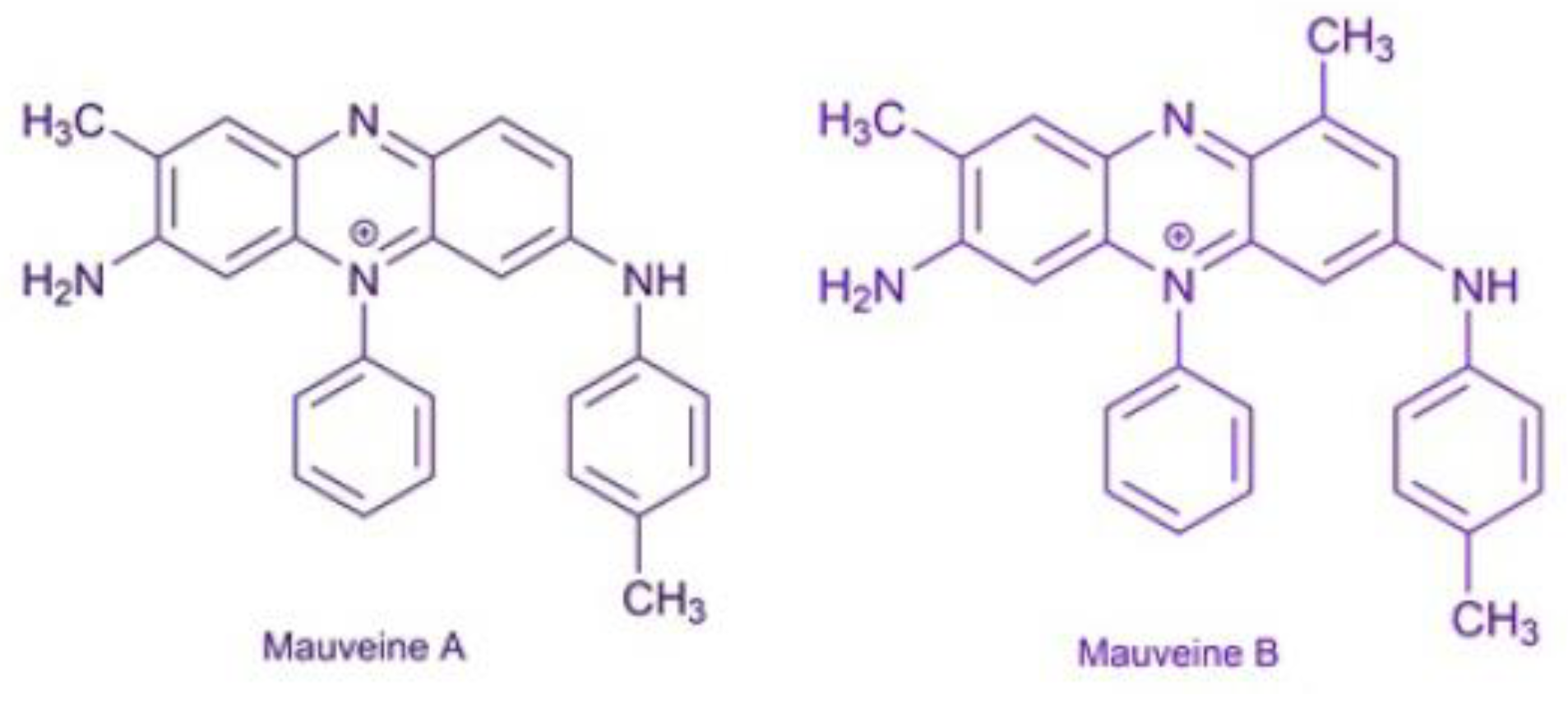
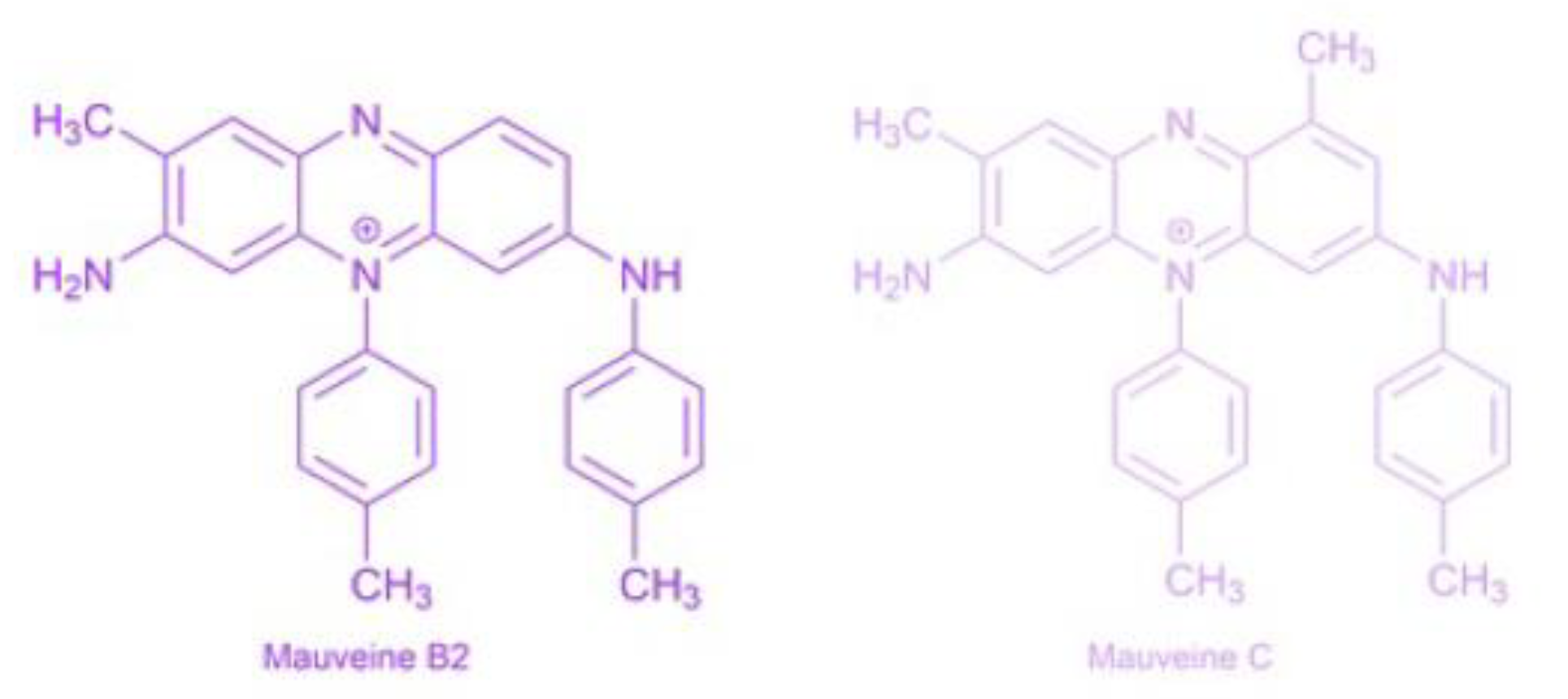
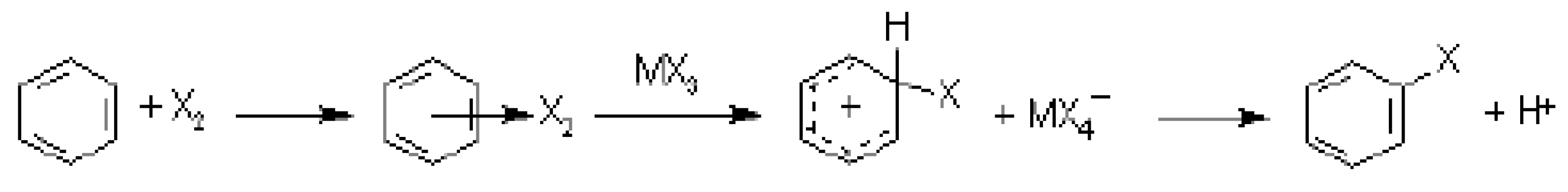



| Properties | Values |
|---|---|
| Molar mass | 93.14 g/mol |
| d (25 °C) | 1.022 g/cm3 |
| Teb (1013 hPa) | 184,3 °C |
| Tfus (1013 hPa) | - 6.2 °C |
| Solubility in water (25 °C) | 36 g/l |
| pKa (25 °C) | 4.6 |
| pKb (25 °C) | 9.4 |
| Flame point | 76 °C |
| Explosion limits | 1.2 - 11% vol. |
| Autoignition temperature | 540 °C |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




