Submitted:
18 March 2025
Posted:
19 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Identification of the MdStb6 Gene Family and Analysis of the Physicochemical Properties of Encoded Proteins
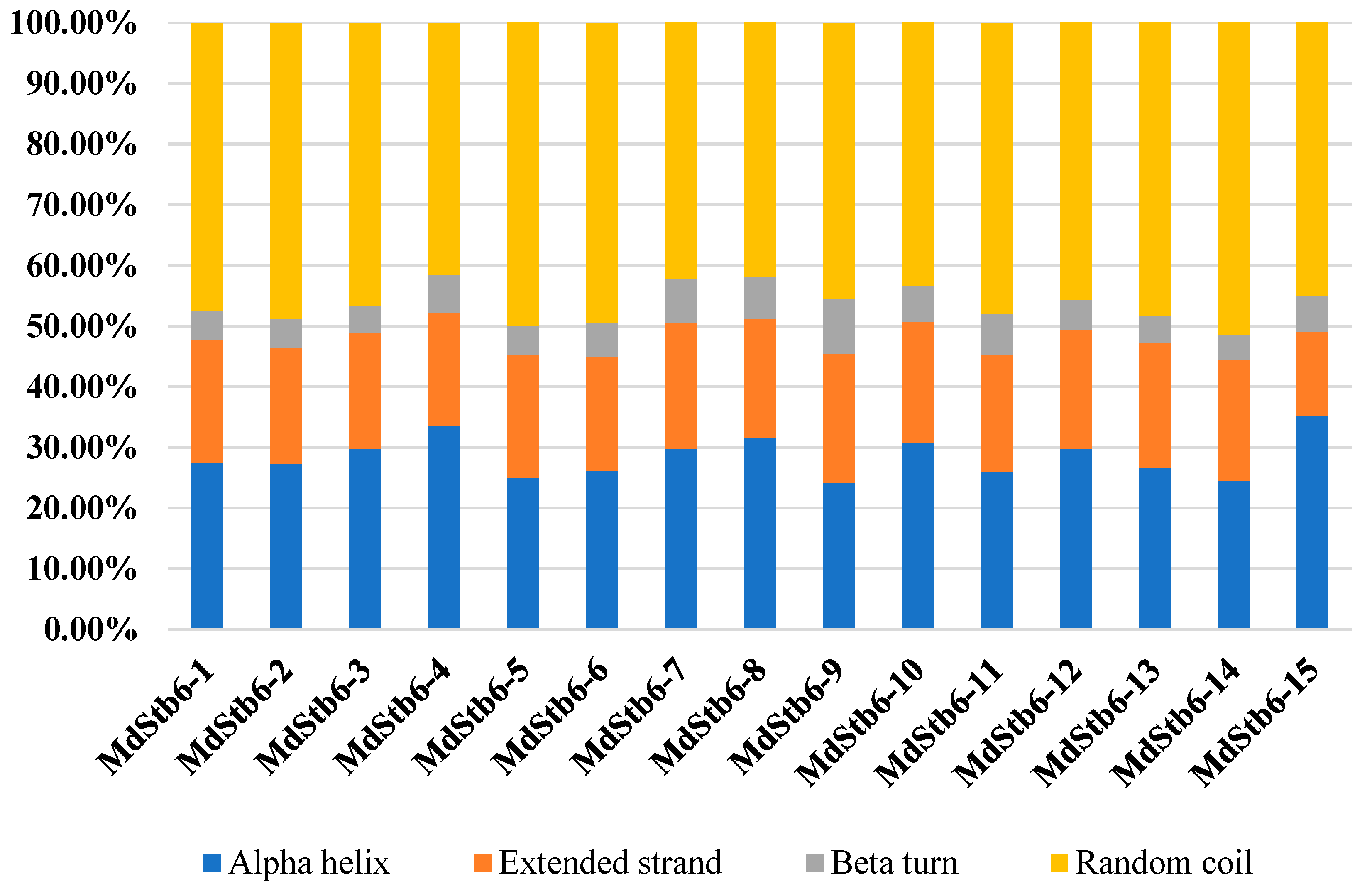
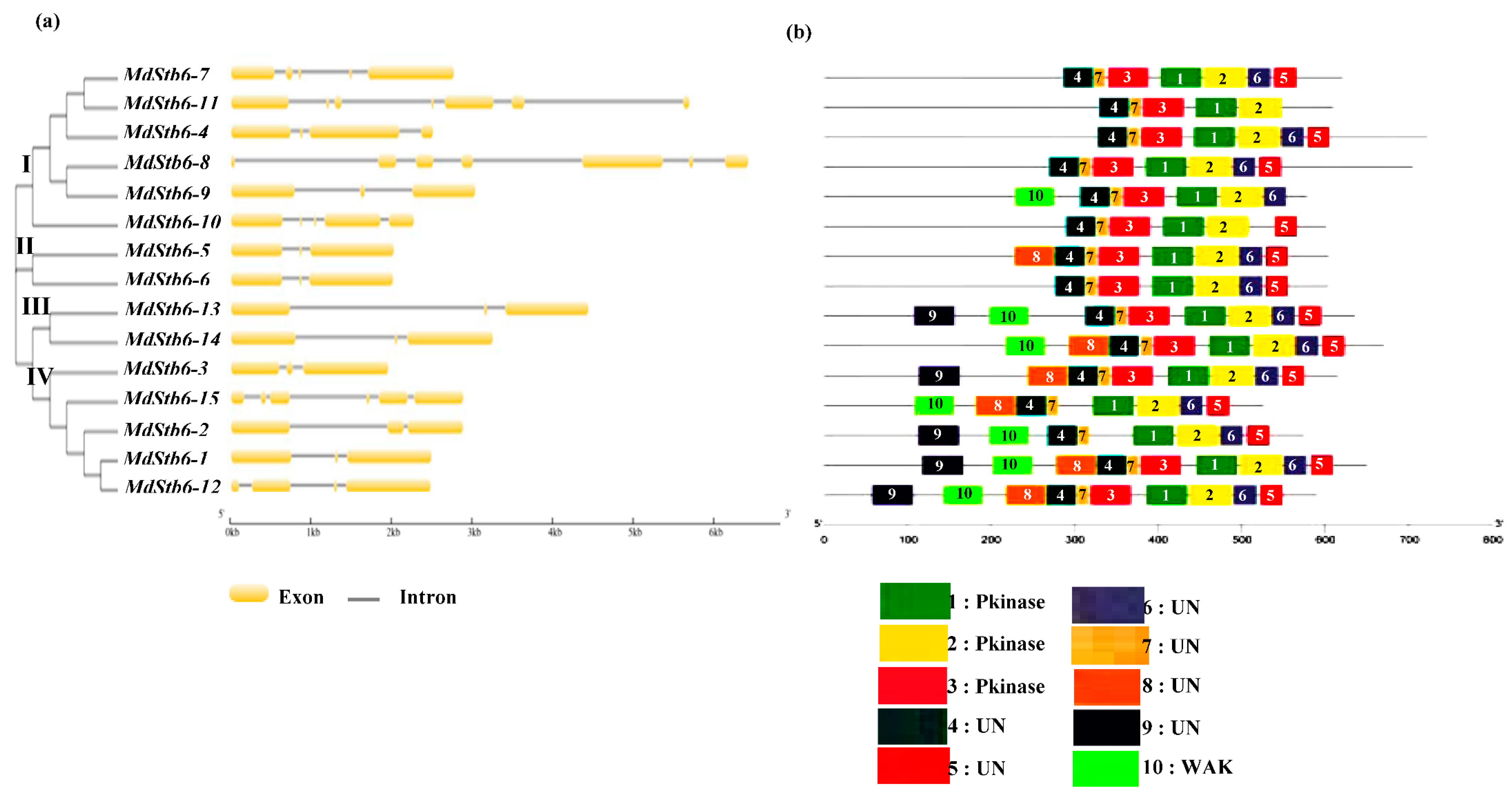
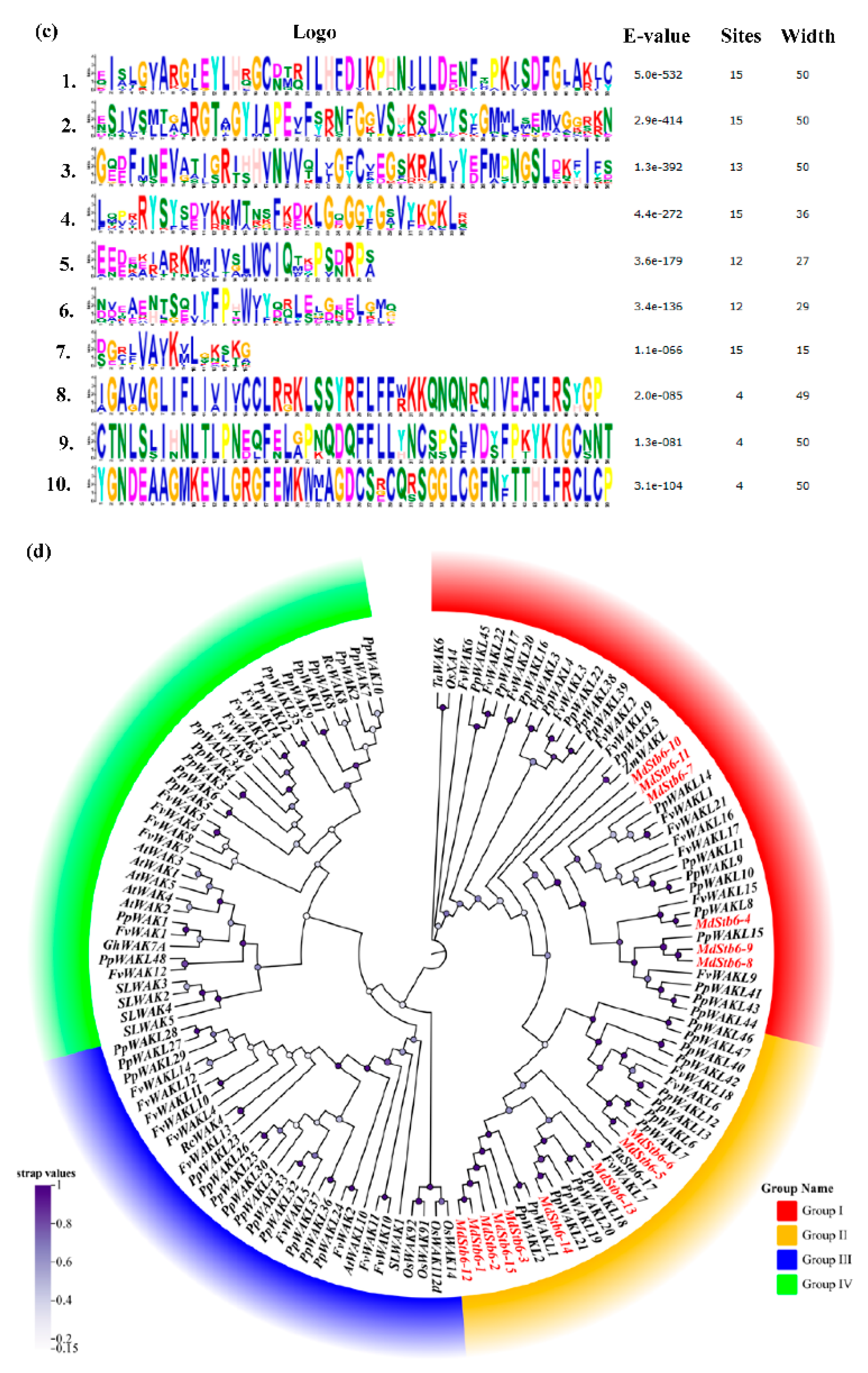
2.2. Gene Structure, Motif Composition and Phylogenetic Analysis of MdStb6 Genes
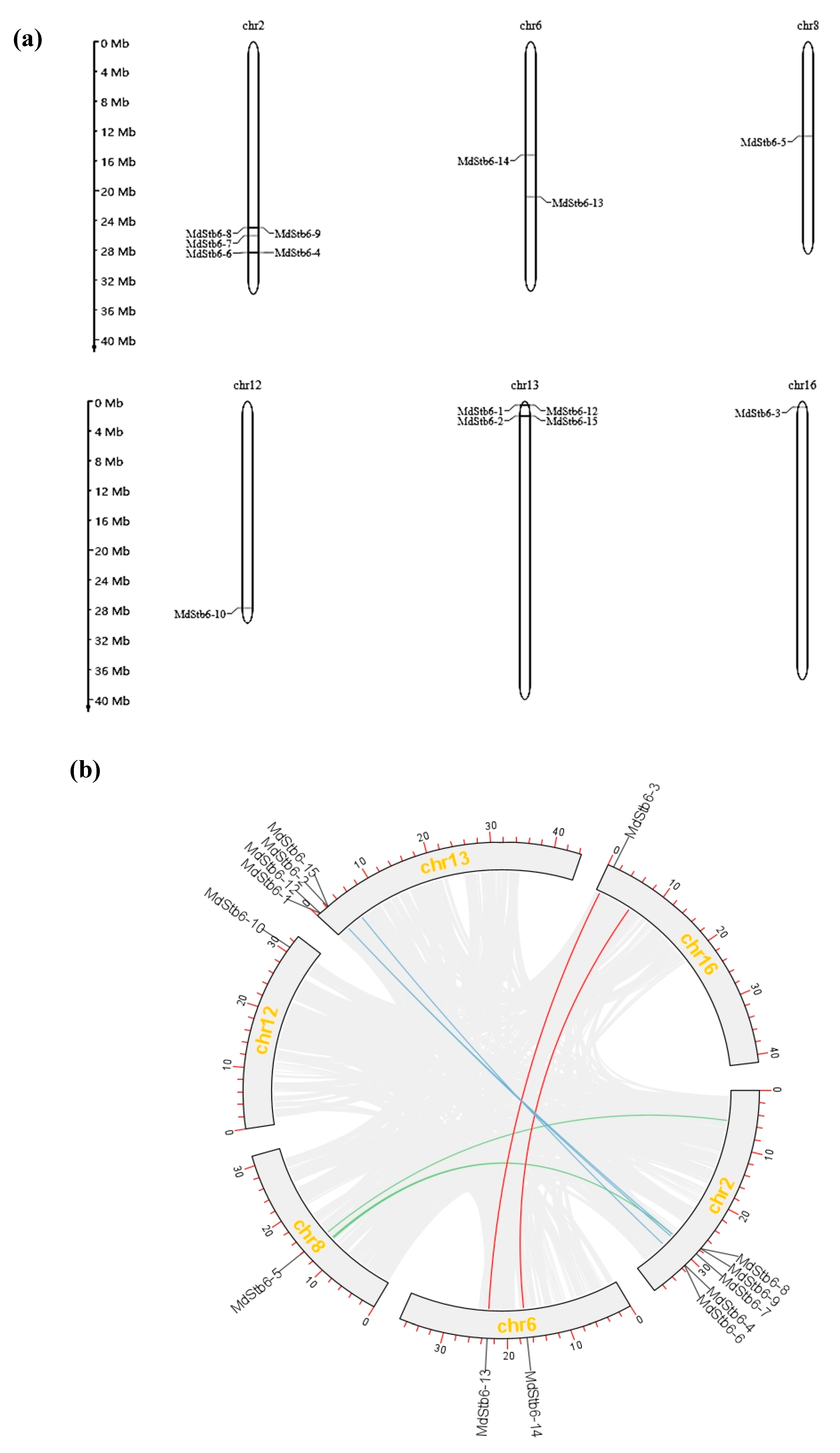
3.3. Chromosomal Locations and Synteny Analysis of MdStb6 Homolog Genes
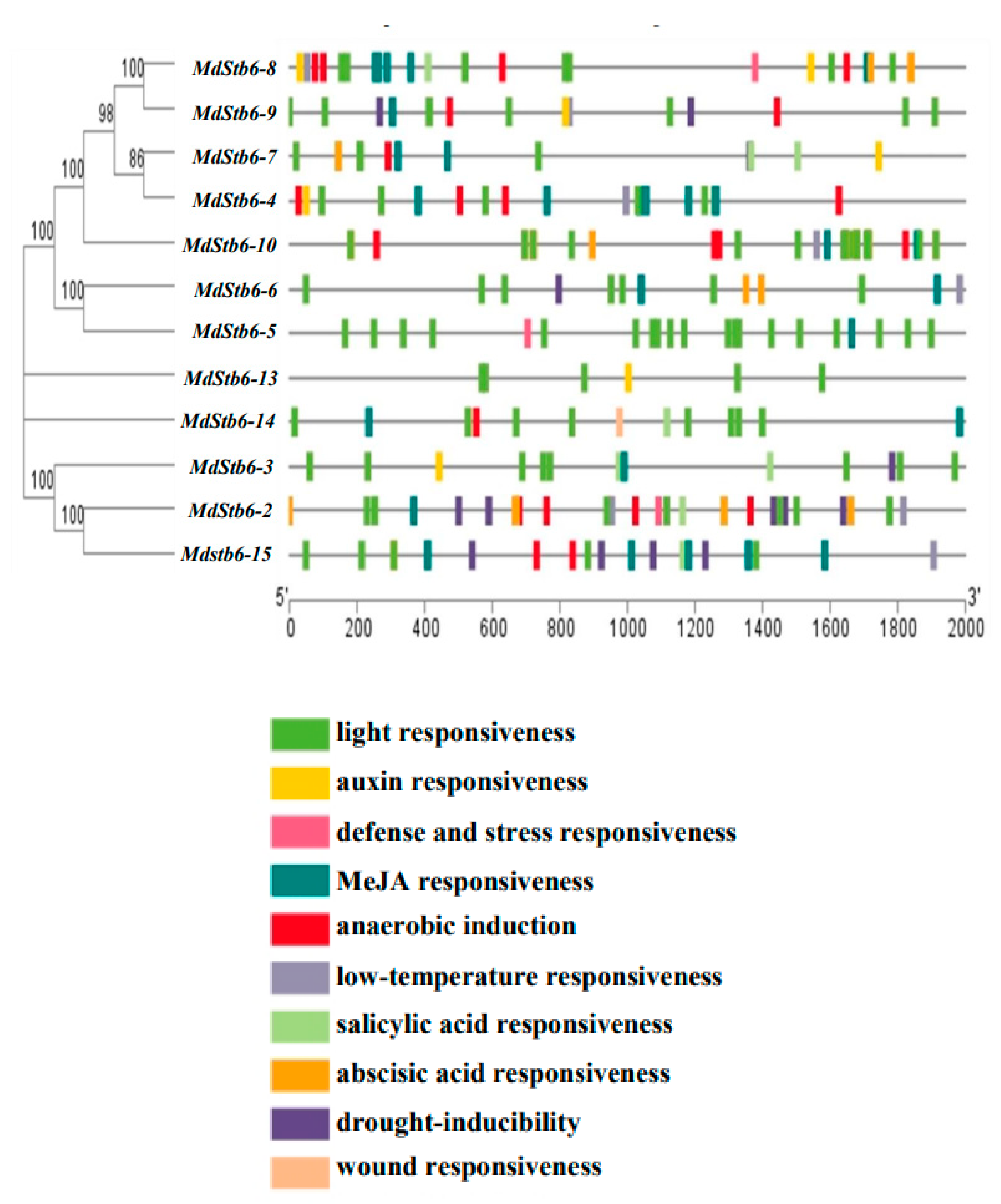
3.4. Cis-Elements in the Promoters of MdStb6 Homologs
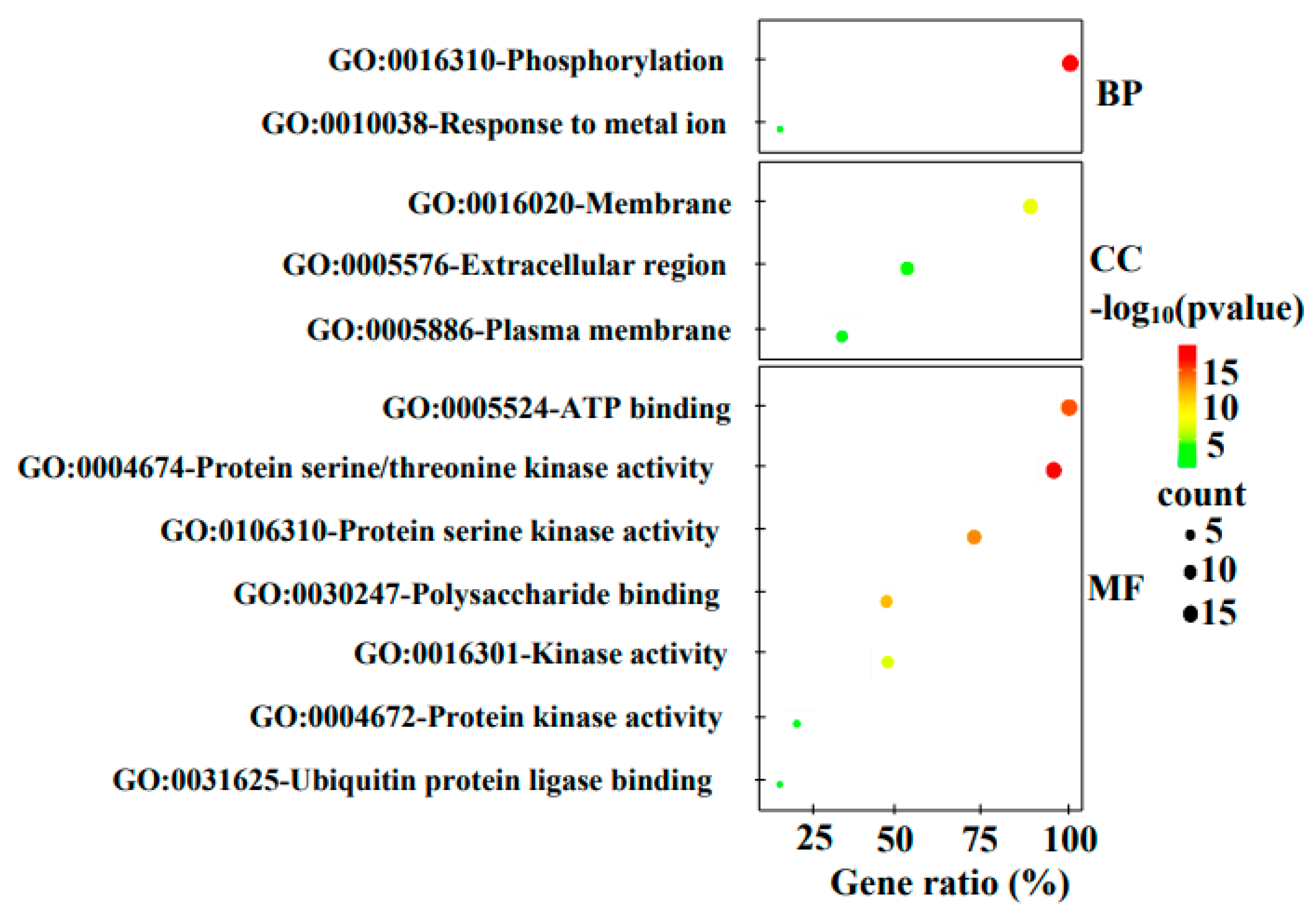
3.5. Gene Ontology (GO) Annotation
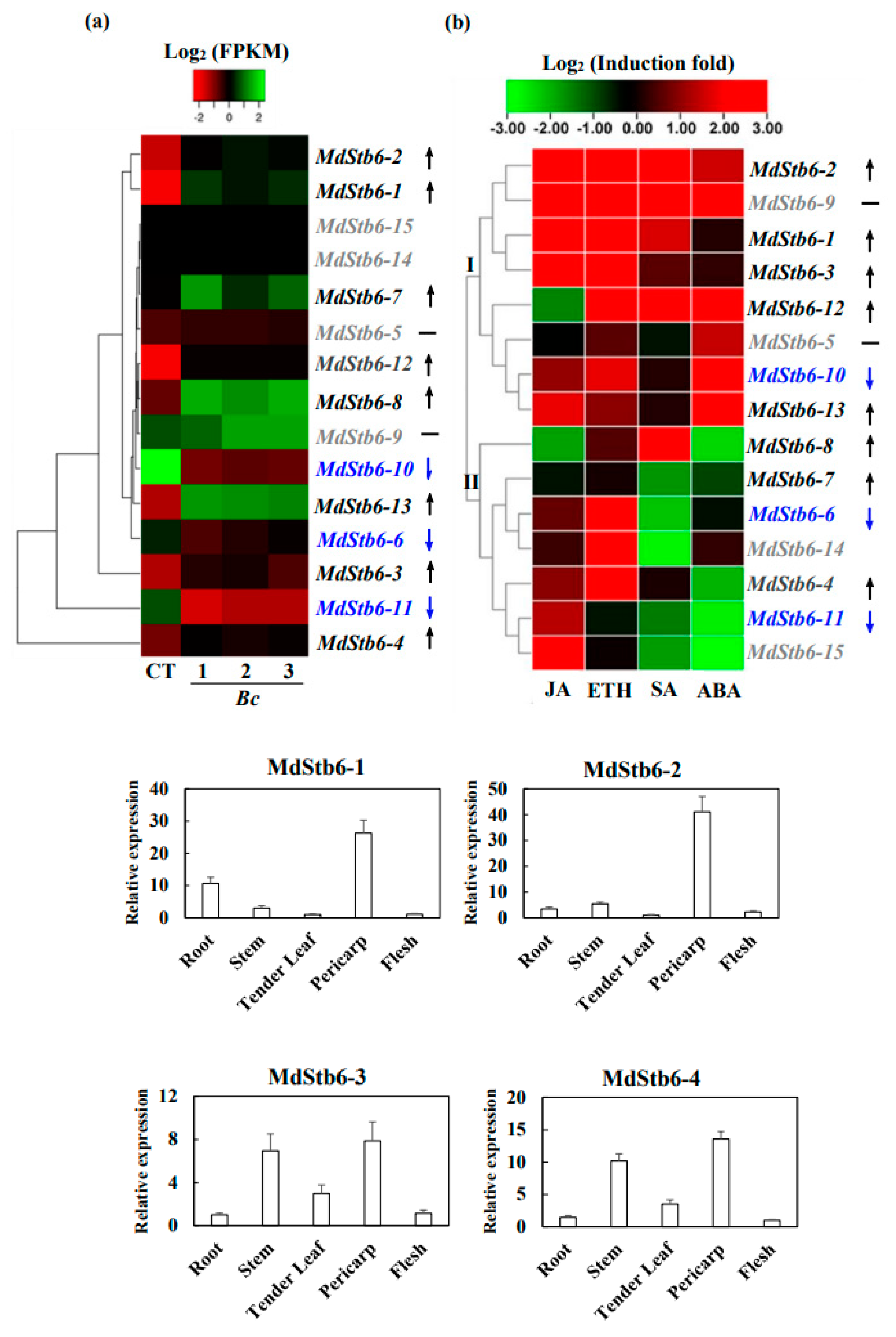
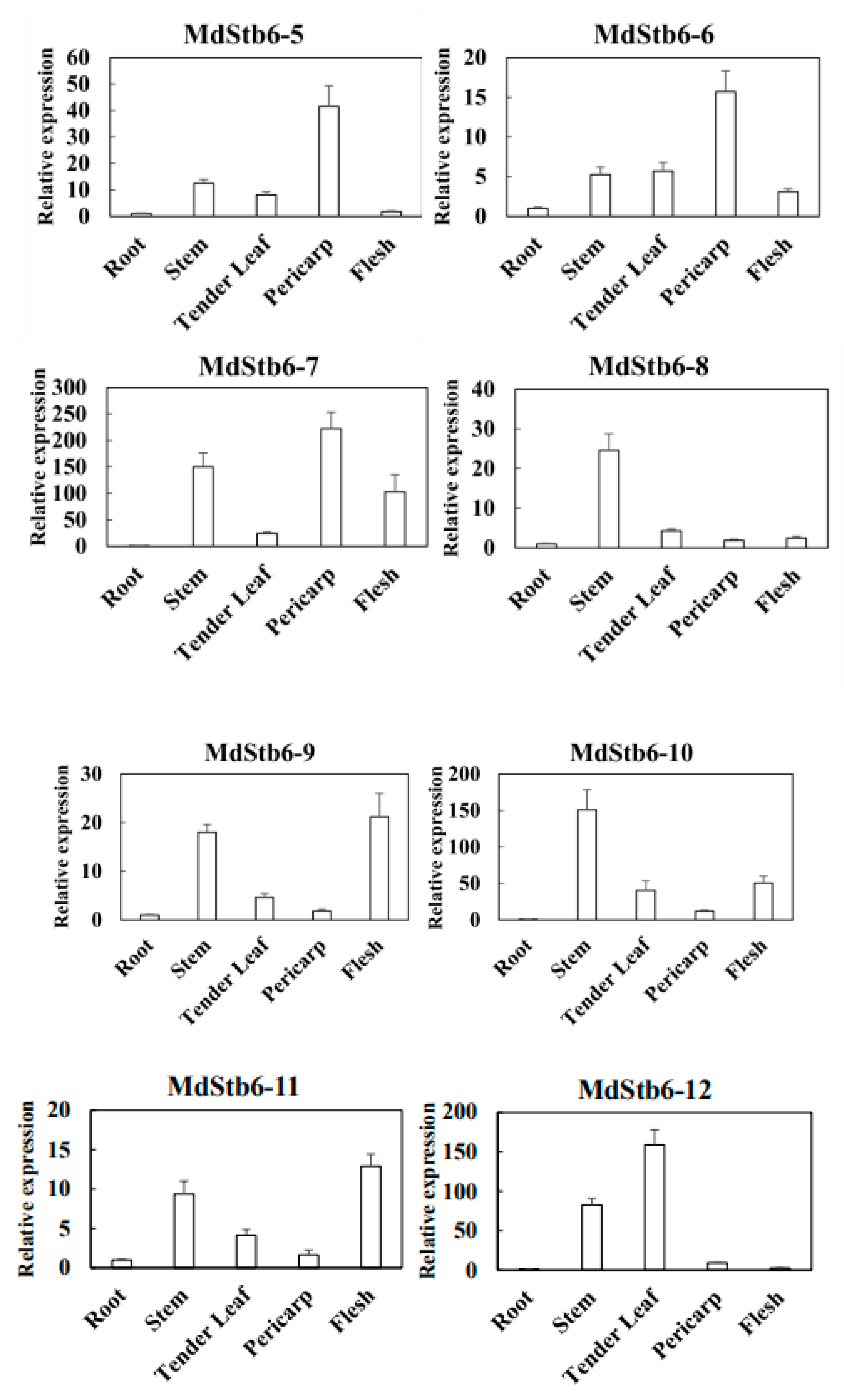
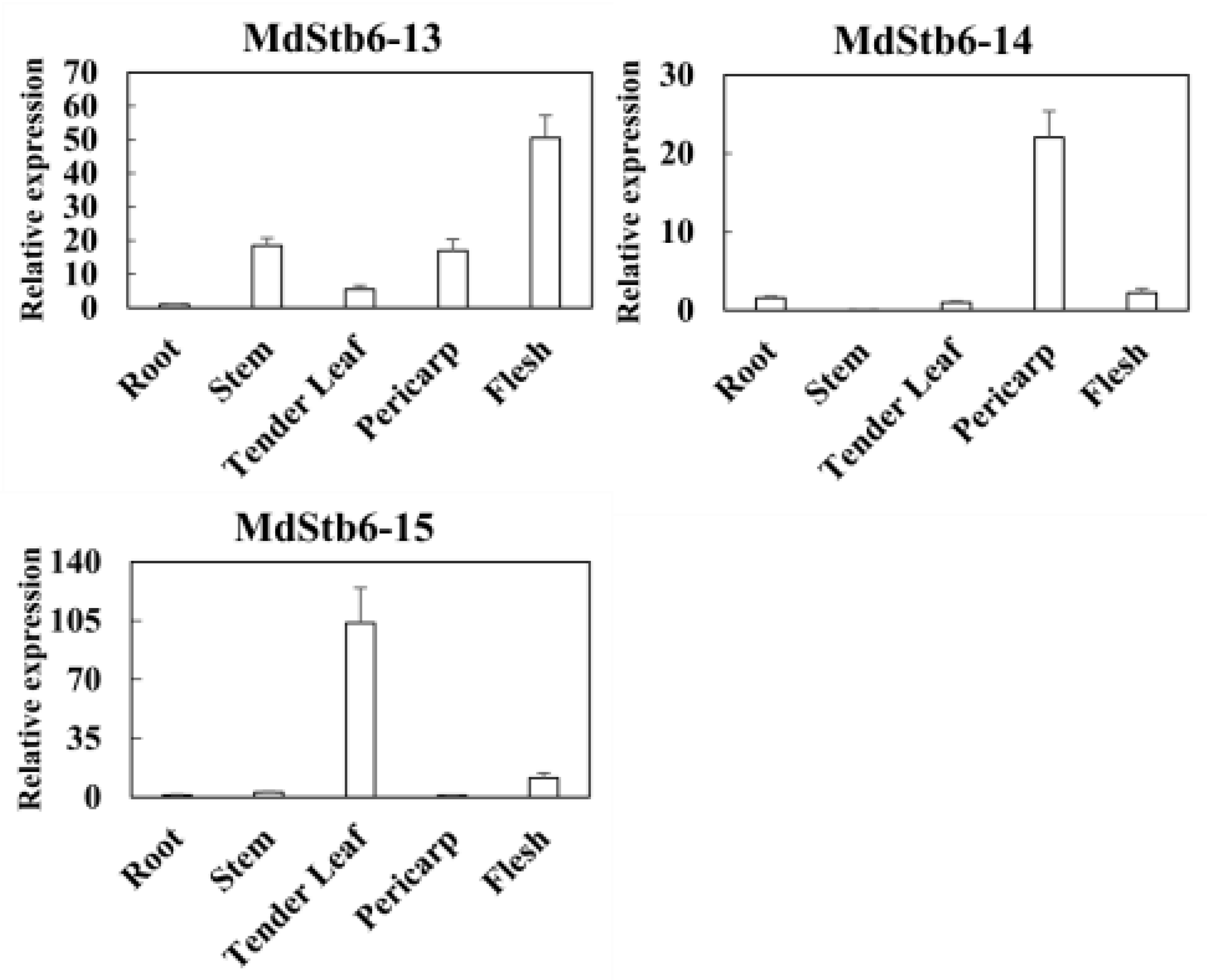
3.6. Response of MdStb6 Homologs to B. cinerea Infection and Defense-Related Plant Hormones and Their Expression Patterns in Different Tissues
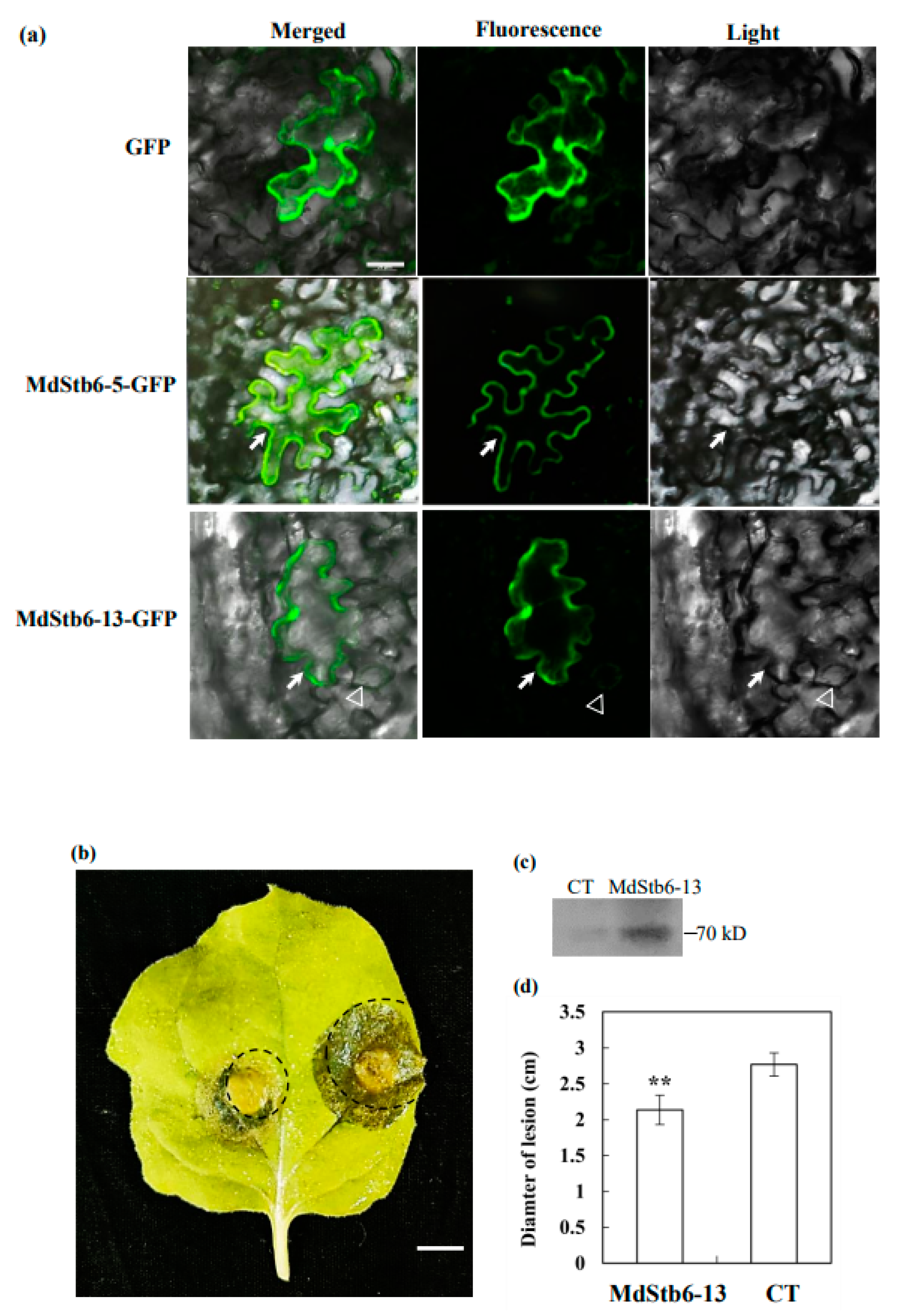
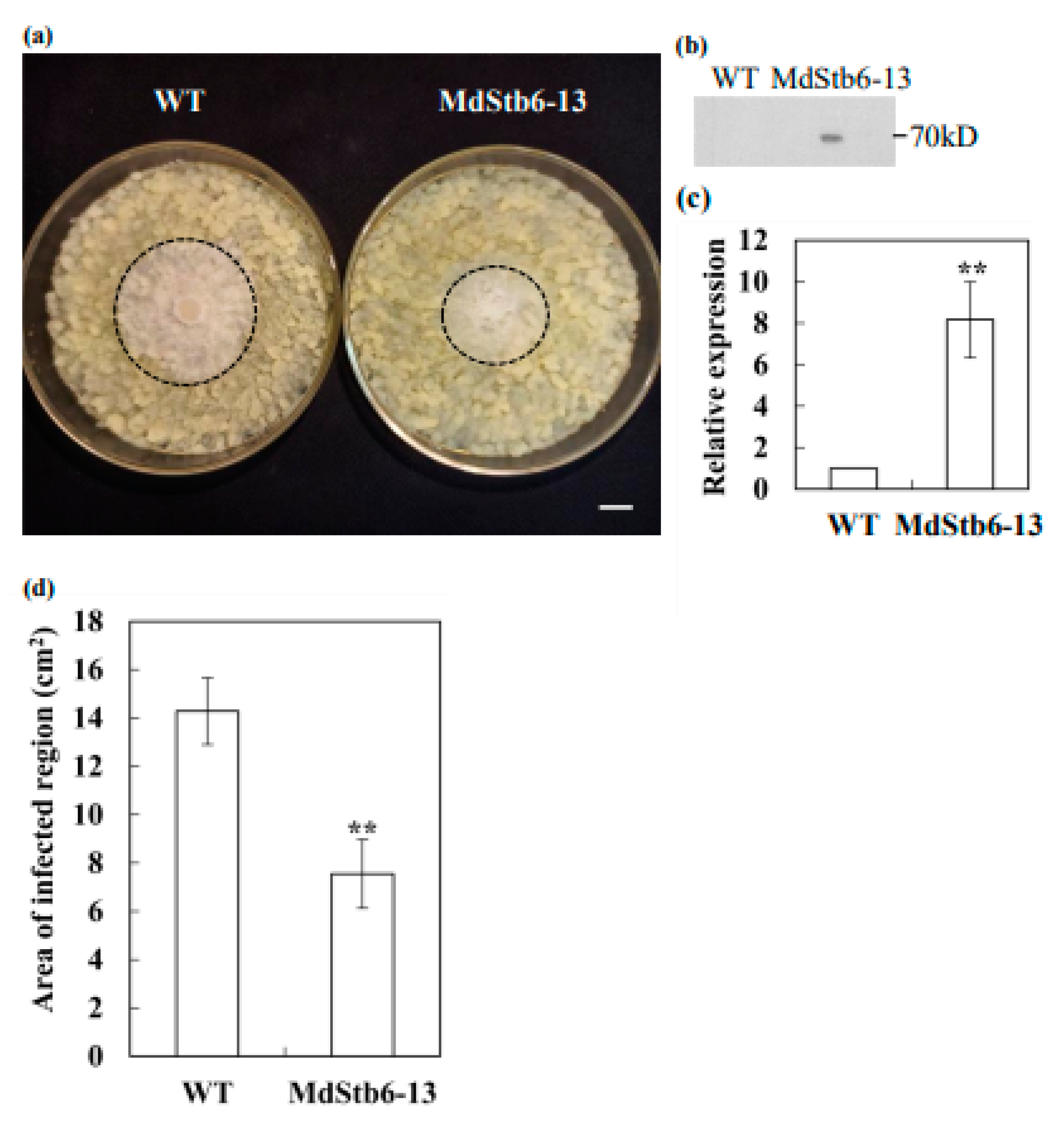
3.7. MdStb6-13confers Resistance to B. cinerea Infection
3. Discussion
4. Materials and Methods
4.1. Identification of the MdStb6 Gene Family and Analysis of Its Physicochemical Properties
4.2. Analysis of MdStb6 Promoter Cis-Acting Elements
4.3. Gene Structure, Motif Composition and Phylogenetic Analysis of MdStb6 Genes
4.4. Chromosomal Locations and Synteny Analysis of MdStb6 Genes
4.5. Gene Ontology (GO) Annotation
4.6. Subcellular Localization
4.7. RNA-seq Analysis of Stb6 Genes in Apple Calli Cells Under B. cinerea Infection
4.8. Plant Hormone Responses Assay and Organ Specific Expression Detection
4.9. Gene Functions Characterization
4.10. Statistic Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| WAK | Wall-associated receptor-kinase |
| WAKL | WAK-like |
| GUB | galacturonan-binding |
| JA | Jasmonic acid |
| MeJA | Methyl jasmonate |
| ETH | Ethylene |
| SA | Salicylic acid |
| ABA | Abscisic acid |
References
- 7 Reasons Why Apples Are Good. Available online: https://health.clevelandclinic.org/benefits-of-apples (accessed on 7 August 2023).
- Abdelhalim, A.; Mazrou, Y.S.A.; Shahin, N.; El-kot, G.A.; Elzaawely, A.A.; Maswada, H.F.; Makhlouf, A.H.; Nehela, Y. Enhancing the storage longevity of apples: The potential of Bacillus subtilis and Streptomyces endus as preventative bioagents against post-harvest gray mold disease, caused by Botrytis cinerea. Plants 2024, 13, 1844. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Huang, Y.; Lian, S.; Saleem, M.; Li, B.; Wang, C. Improving the biocontrol efficacy of Meyerozyma guilliermondii Y-1 with melatonin against postharvest gray mold in apple fruit. Postharvest Biol Tec. 2021, 171, 11351. [Google Scholar] [CrossRef]
- Yuan, Y.; Cao, S.; Sun, J.; Hou, J.; Zhang, M.; Qin, Q.; Li, G. Sterol regulatory element-binding protein Sre1 mediates the development and pathogenicity of the grey mould fungus Botrytis cinerea. Int. J. Mol. Sci. 2025, 26, 1365. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Yang, Z.; Song, W.; Zhao, H.; Ye, Q.; Xu, H.; Hu, B.; Shen, D.; Dou, D. Biofumigation by mustard plants as an application for controlling postharvest gray mold in apple fruits. Agronomy 2023, 13, 1490. [Google Scholar] [CrossRef]
- Singh, R.; Caseys, C.; Kliebenstein, D.J. Genetic and molecular landscapes of the generalist phytopathogen Botrytis cinerea. Mol Plant Pathol 2024, 25, e13404. [Google Scholar] [CrossRef]
- Thomma, B.P.H.J.; Eggermont, K.; Penninckx, I.A.M.A.; Mauch-Mani, B.; Vogelsang, R.; Cammue, P.A.; Broekaert, W.F. Separate jasmonate-dependent and salicylate-dependent defense-response pathways in Arabidopsis are essential for resistance to distinct microbial pathogens. Proc Natl Acad Sci USA 1998, 95, 15107–15111. [Google Scholar] [CrossRef]
- Thomma, B.P.H.J.; Eggermont, K.; Tierens, K.F.M.J.; Broekaert, W.F. Requirement of functional ethylene-insensitive 2 gene for efficient resistance of Arabidopsis to infection by Botrytis cinerea. Plant Physiol 1999, 121, 1093–1101. [Google Scholar] [CrossRef]
- Lloyd, A.J.; Allwood, J.W.; Winder, C.L.; Dunn, W.B.; Heald, J.K.; Cristescu, S.M.; Sivakumaran, A.; Harren, F.J.M.; Mulema, J.; Denby, K.; et al. Metabolomic approaches reveal that cell wall modifications play a major role in ethylene-mediated resistance against Botrytis cinerea. Plant J 2011, 67, 852–868. [Google Scholar] [CrossRef]
- Zhao, Y.; Wei, T.; Yin, K.Q.; Chen, Z.; Gu, H.; Qu, L.J.; Qin, G. Arabidopsis RAP2.2 plays an important role in plant resistance to Botrytis cinerea and ethylene responses. New Phytol 2012, 195, 450–460. [Google Scholar] [CrossRef]
- Zhou, J.; Mu, Q.; Wang, X.; Zhang, J.; Yu, H.; Huang, T.; He, Y.; Dai, S.; Meng, X. Multilayered synergistic regulation of phytoalexin biosynthesis by ethylene, jasmonate, and MAPK signaling pathways in Arabidopsis. Plant Cell 2022, 34, 3066–3087. [Google Scholar] [CrossRef]
- Zhang, M.; Li, W.; Zhang, T.; Liu, Y.; Liu, L. Botrytis cinerea-induced F-box protein 1 enhances disease resistance by inhibiting JAO/JOX-mediated jasmonic acid catabolism in Arabidopsis. Mol Plant 2024, 17, 297–311. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, S.; Plotnikova, J.M.; Lorenzo, G.D.; Ausubel, F.M. Arabidopsis local resistance to Botrytis cinerea involves salicylic acid and camalexin and requires EDS4 and PAD2,but not SID2, EDS5 or PAD4. Plant J 2003, 35, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Veronese, P.; Nakagami, H.; Bluhm, B.; Abuqamar, S.; Chen, X.; Salmeron, J.; Dietrich, R.A.; Hirt, H.; Mengiste, T. The membrane-anchored BOTRYTIS-INDUCED KINASE1 plays distinct roles in Arabidopsis resistance to necrotrophic and biotrophic pathogens. Plant Cell 2006, 18, 257–273. [Google Scholar] [CrossRef]
- Ziemann, S.; van der Linde, K.; Lahrmann, U.; Acar, B.; Kaschani, F.; Colby, T.; Kaiser, M.; Ding, Y.; Schmelz, E.; Huffaker, A.; et al. An apoplastic peptide activates salicylic acid signalling in maize. Nat Plants 2018, 4, 172–180. [Google Scholar] [CrossRef]
- Zhang, W.; Fraiture, M.; Kolb, D.; Löffelhardt, B.; Desaki, Y.; Boutrot, F.F.G.; Tör, M.; Zipfel, C.; Gust, A.A.; Brunner, F. Arabidopsis RECEPTOR-LIKE PROTEIN30 and receptor-like kinase SUPPRESSOR OF BIR1-1/EVERSHED mediate innate immunity to necrotrophic fungi. Plant Cell 2013, 25, 4227–4241. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Kars, I.; Essenstam, B.; Liebrand, T.W.H.; Wagemakers, L.; Elberse, J.; Tagkalaki, P.; Tjoitang, D.; van den Ackerveken, G.; van Kan, J.A.L. Fungal endopolygalacturonases are recognized as microbe-associated molecular patterns by the Arabidopsis receptor-like protein RESPONSIVENESSTO BOTRYTIS POLYGALACTURONASES1. Plant Physiol 2014, 164, 352–364. [Google Scholar] [CrossRef]
- Ono, E.; Mise, K.; Takano, Y. RLP23 is required for Arabidopsis immunity against the grey mould pathogen Botrytis cinerea. Sci Rep 2020, 10, 13798. [Google Scholar] [CrossRef]
- Kemmerling, B.; Schwedt, A.; Rodriguez, P.; Mazzotta, S.; Frank, M.; Qamar, S.A.; Mengiste, T.; Betsuyaku, S.; Parker, J.E.; Müssig, C.; et al. The BRI1-associated kinase 1, BAK1, has a brassinolide-independent role in plant cell-death control. Curr Biol 2007, 17, 1116–1122. [Google Scholar] [CrossRef]
- Xu, S.; Liao, C.J.; Jaiswal, N.; Lee, S.; Yun, D.J.; Lee, S.Y.; Garvey, M.; Kaplan, I.; Mengistea, T. Tomato PEPR1 ORTHOLOG RECEPTOR-LIKE KINASE1 regulates responses to systemin, necrotrophic fungi, and insect herbivory. Plant Cell 2018, 30, 2214–2229. [Google Scholar] [CrossRef]
- Liu, C.; Yu, H.; Voxeur, A.; Rao, X.; Rixon, R.A. FERONIA and wall-associated kinases coordinate defense induced by lignin modification in plant cell walls. Sci Adv 2023, 9, eadf7714. [Google Scholar] [CrossRef]
- Held, J.B.; Rowles, T.; Schulz, W.; McNellis, T.M. Arabidopsis Wall-Associated Kinase 3 is required for harpin-activated immune responses. New Phytol 2024, 242, 853–857. [Google Scholar] [CrossRef] [PubMed]
- Zhong, T.; Zhu, M.; Zhang, Q.; Zhang, Y.; Deng, S.; Guo, C.; Xu, L.; Liu, T.; Li, Y.; Fan, X.; et al. The ZmWAKL-ZmWIK-ZmBLK1-ZmRBH4 module provides quantitative resistance to gray leaf spot in maize. Nat Genet 2024, 56, 315–326. [Google Scholar] [CrossRef]
- Wang, P.; Zhou, L.; Jamieson, P.; Zhang, L.; Zhao, Z.; Babilonia, K.; Shao, W.; Wu, L.; Mustafa, R.; Amin, I.; et al. The cotton wall-associated kinase GhWAK7A mediates responses to fungal wilt pathogens by complexing with the chitin sensory receptors. Plant Cell 2020, 32, 3978–4001. [Google Scholar] [CrossRef]
- Ferrari, S.; Savatin, D.V.; Sicilia, F.; Gramegna, G.; Cervone, F.; De Lorenzo, G. Oligogalacturonides: Plant damage-associated molecular patterns and regulators of growth and development. Front Plant Sci 2013, 4, 49. [Google Scholar] [CrossRef] [PubMed]
- Herold, L.; Ordon, J.; Hua, C.; Kohorn, B.D.; Nürnberger, T.; De Falco, T.A.; Zipfel, C. Arabidopsis WALL-ASSOCIATED KINASES are not required for oligogalacturonide-induced signaling and immunity. Plant Cell 2024, 37, koae317. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, Y.; Chen, M.; Da, L.; Su, Z.; Zhang, Z.; Liu, X. Comparative genomics analysis of WAK/WAKL family in Rosaceae identify candidate WAKs involved in the resistance to Botrytis cinerea. BMC Genomics 2023, 24, 337. [Google Scholar] [CrossRef] [PubMed]
- Zuo, C.; Liu, Y.; Guo, Z.; Mao, J.; Chu, M.; Chen, B. Genome-wide annotation and expression responses to biotic stresses of the WALL-ASSOCIATED KINASE -RECEPTOR-LIKE KINASE (WAK-RLK) gene family in apple (Malus domestica). Eur J Plant Pathol 2019, 153, 771–785. [Google Scholar] [CrossRef]
- Zhong, Z.; Marcel, T.C.; Hartmann, F.E.; Ma, X.; Plissonneau, C.; Zala, M.; Ducasse, A.; Confais, J.; Compain, J.; Lapalu, N.; et al. A small secreted protein in Zymoseptoria tritici is responsible for avirulence on wheat cultivars carrying the Stb6 resistance gene. New Phytol 2017, 214, 619–631. [Google Scholar] [CrossRef]
- Saintenac, C.; Lee, W.S.; Cambon, F.; Rudd, J.J.; King, R.C.; Marande, W.; Powers, S.J.; Bergès, H.; Phillips, A.L.; Uauy, C.; et al. Wheat receptor-kinase-like protein Stb6 controls gene-for-gene resistance to fungal pathogen Zymoseptoria tritici. Nat Genet 2018, 50, 368–374. [Google Scholar] [CrossRef]
- Hu, K.; Cao, J.; Zhang, J.; Xia, F.; Ke, Y.; Zhang, H.; Xie, W.; Liu, H.; Cui, Y.; Cao, Y.; et al. Improvement of multiple agronomic traits by a disease resistance gene via cell wall reinforcement. Nat Plants 2014, 3, 17009. [Google Scholar] [CrossRef]
- Ghiasi Noei, F.; Imami, M.; Didaran, F.; Ghanbari, M.A.; Zamani, E.; Ebrahimi, A.; Aliniaeifard, S.; Farzaneh, M.; Javan-Nikkhah, M.; Feechan, A.; et al. Stb6 mediates stomatal immunity, photosynthetic functionality, and the antioxidant system during the Zymoseptoria tritici-wheat interaction. Front. Plant Sci 2022, 13, 1004691. [Google Scholar] [CrossRef]
- Alassimone, J.; Praz, C.; Lorrain, C.; De Francesco, A.; Carrasco-López, C.; Faino, L.; Shen, Z.; Meile, L.; Sánchez-Vallet, A. The Zymoseptoria tritici avirulence factor AvrStb6 accumulates in hyphae close to stomata and triggers a wheat defense response hindering fungal penetration. Mol Plant Microbe Interact 2024, 37, 432–444. [Google Scholar] [CrossRef] [PubMed]
- Hsu, P.K.; Dubeaux, G.; Takahashi, Y.; Schroeder, J.I. Signaling mechanisms in abscisic acid-mediated stomatal closure. Plant J 2021, 105, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, F.; Yoshida, R.; Ichimura, K.; Mizoguchi, T.; Seo, S.; Yonezawa, M.; Maruyama, K.; Yamaguchi-Shinozaki, K.; Shinozaki, K. The mitogen-activated protein kinase cascade MKK3-MPK6 is an important part of the jasmonate signal transduction pathway in Arabidopsis. Plant Cell 2007, 19, 805–818. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wu, Y.; Yang, F.; Zhang, Y.; Chen, S.; Xie, Q.; Tian, X.; Zhou, J.M. BIK1 interacts with PEPRs to mediate ethylene-induced immunity. Proc Natl Acad Sci USA 2013, 110, 6205–6210. [Google Scholar] [CrossRef]
- Lal, N.K.; Nagalakshmi, U.; Hurlburt, N.K.; Flores, R.; Bak, A.; Sone, P.; Ma, X.; Song, G.; Walley, J.; Shan, L.; et al. The receptor-like cytoplasmic kinase BIK1localizes to the nucleus and regulates defense hormone expression during plant innate immunity. Cell Host Microbe 2018, 23, 485–497. [Google Scholar] [CrossRef]
- Wang, R.; He, F.; Ning, Y.; Wang, G.L. Fine-tuning of RBOH-mediated ROS signaling in plant immunity. Trends Plant Sci 2020, 25, 1060–1062. [Google Scholar] [CrossRef]
- Zhang, W. The regulation of receptor-like cytoplasmic kinase MdBIK1 in resistance to Botrytis cinerea and Botryosphaeria dothidea on apple. Master Thesis, Shandong Agricultural University, Tai’an, China, 2018. [Google Scholar]
- Llorente, F.; Muskett, P.; Sánchez-Vallet, A.; López, G.; Ramos, B.; Sánchez-Rodríguez, C.; Jordá, L.; Parker, J.; Molina, A. Repression of the auxin responses pathway increases Arabidopsis susceptibility to necrotrophic fungi. Mol Plant 2008, 1, 496–509. [Google Scholar] [CrossRef]
- Zhang, H.; Hu, Z.; Lei, C.; Zheng, C.; Wang, J.; Shao, S.; Li, X.; Xia, X.; Cai, X.; Zhou, J.; et al. A plant phytosulfokine peptide initiates auxin-dependent immunity through cytosolic Ca2+ signaling in tomato. Plant Cell 2018, 30, 652–667. [Google Scholar] [CrossRef]
- Ghelli, R.; Brunetti, P.; Napoli, N.; De Paolis, A.; Cecchetti, V.; Tsuge, T.; Serino, G.; Matsui, M.; Mele, G.; Rinaldi, G.; et al. A newly identified flower-specific splice variant of AUXIN RESPONSE FACTOR8 regulates stamen elongation and endothecium lignification in Arabidopsis. Plant Cell 2018, 30, 620–637. [Google Scholar] [CrossRef]
- Wang, W.; Li, Y.; Cai, C.; Zhu, Q. Auxin response factors fine-tune lignin biosynthesis in response to mechanical bending in bamboo. New Phytol 2024, 241, 1161–1176. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Dong, C.; Wu, Y.; Fu, S.; Tauqeer, A.; Gu, X.; Li, Q.; Niu, X.; Liu, P.; Zhang, X.; et al. The JA-to-ABA signaling relay promotes lignin deposition for wound healing in Arabidopsis. Mol Plant 2024, 17, 1594–1605. [Google Scholar] [CrossRef]
- Cantu, D.; Vicente, A.R.; Greve, L.C.; Dewey, F.M.; Bennett, A.B.; Labavitch, J.M.; Powell, A.L.T. The intersection between cell wall disassembly, ripening, and fruit susceptibility to Botrytis cinerea. Proc Natl Acad Sci USA 2008, 105, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.J.; Van Den Abeele, C.; Ortega-Salazar, I.; Papin, V.; Adaskaveg, J.A.; Wang, D.; Casteel, C.L.; Seymour, G.B.; Blanco-Ulate, B. Host susceptibility factors render ripe tomato fruit vulnerable to fungal disease despite active immune responses. J Exp Bot 2021, 72, 2696–2709. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.J.; Adaskaveg, J.A.; Mesquida-Pesci, S.D.; Ortega-Salazar, I.B.; Pattathil, S.; Zhang, L.; Hahn, M.G.; van Kan, J.A.L.; Cantu, D.; Powell, A.L.T.; et al. Botrytis cinerea infection accelerates ripening and cell wall disassembly to promote disease in tomato fruit. Plant Physiol 2023, 191, 575–590. [Google Scholar] [CrossRef]
- Qi, L.; Yan, J.; Li, Y.; Jiang, H.; Sun, J.; Chen, Q.; Li, H.; Chu, J.; Yan, C.; Sun, X.; et al. Arabidosis thaliana plants differentially modulate auxin biosynthesis and transport during defense responses to the necrotrophic pathogen Alternaria brassicicola. New Phytol 2012, 195, 872–882. [Google Scholar] [CrossRef]
- Yang, W.; Zhai, H.; Wu, F.; Deng, L.; Chao, Y.; Meng, X.; Chen, Q.; Liu, H.; Bie, X.; Sun, C.; et al. Peptide REF1 is a local wound signal promoting plant regeneration. Cell 2024, 187, 3024–3038. [Google Scholar] [CrossRef]
- Xin, L.; Zhang, R.; Wang, X.; Liu, X.; Wang, Y.; Qi, P.; Wang, L.; Wu, S.; Chen, X. Extracellular and intracellular infection of Botryosphaeria dothidea and resistance mechanism in apple cells. Hortic Plant J 2023, 28, 209–223. [Google Scholar] [CrossRef]
- Zhang, Z.; Feechan, A.; Pedersen, C.; Newman, M.A.; Qiu, J.L.; Olesen, K.L.; Thordal-Christensen, H. A SNARE-protein has opposing functions in penetration resistance and defence signalling pathways. Plant J 2007, 49, 302–312. [Google Scholar] [CrossRef]
- Hernández-Blanco, C.; Feng, D.X.; Hu, J.; Sánchez-Vallet, A.; Deslandes, L.; Llorente, F.; Berrocal-Lobo, M.; Keller, H.; Barlet, X.; Sánchez-Rodríguez, C.; et al. Impairment of cellulose synthases required for Arabidopsis secondary cell wall formation enhances disease resistance. Plant Cell 2007, 19, 890–903. [Google Scholar] [CrossRef]
- Chassot, C.; Nawrath, C.; Métraux, J.P. Cuticular defects lead to full immunity to a major plant pathogen. Plant J 2007, 49, 972–980. [Google Scholar] [CrossRef] [PubMed]
- Lipka, V.; Dittgen, J.; Bednarek, P.; Bhat, R.; Wiermer, M.; Stein, M.; Landtag, J.; Brandt, W.; Rosahl, S.; Scheel, D.; et al. Pre- and postinvasion defenses both contribute to nonhost resistance in Arabidopsis. Science 2005, 310, 1180–1183. [Google Scholar] [CrossRef]
- Duan, S.; Eom, S.H. Regulation of anthocyanin and lignin contents in postharvest ‘Fuji’ apple irradiated with UV-B. Sci Hortic 2023, 322, 112428. [Google Scholar] [CrossRef]
- Xu, S.; Sun, M.; Yao, J.L.; Liu, X.; Xue, Y.; Yang, G.; Zhu, R.; Jiang, W.; Wang, R.; Xue, C.; et al. Auxin inhibits lignin and cellulose biosynthesis in stone cells of pear fruit via the PbrARF13-PbrNSC-PbrMYB132 transcriptional regulatory cascade. Plant Biotechnol J 2023, 21, 1408–1425. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Wan, S.; Huang, Y.; Li, X.; Jiao, T.; Zhang, Z.; Ma, B.; Zhu, L.; Ma, F.; Li, M. The transcription factor MdBPC2 alters apple growth and promotes dwarfing by regulating auxin biosynthesis. Plant Cell 2024, 36, 585–604. [Google Scholar] [CrossRef]
- Mitchell, A.L.; Attwood, T.K.; Babbitt, P.C.; Blum, M.; Bork, P.; Bridge, A.; Brown, S.D.; Chang, H.Y.; El-Gebali, S.; Fraser, M.I.; et al. InterPro in 2019: Improving coverage, classification and access to protein sequence annotations. Nucleic Acids Res 2019, 47, D351–D360. [Google Scholar] [CrossRef]
- Chou, K.C.; Shen, H.B. Plant-mPLoc: A top-down strategy to augment the power for predicting plant protein subcellular localization. PLoS ONE 2010, 5, e11335. [Google Scholar] [CrossRef]
- Chen, C.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.; Xia, R. TBtools: An integrative toolkit developed for interactive analyses of big biological data. Mol Plant 2020, 13, 1194–1202. [Google Scholar] [CrossRef]
- Hu, B.; Jin, J.; Guo, A.Y.; Zhang, H.; Luo, J.; Gao, G. GSDS 2.0: An upgraded gene feature visualization server. Bioinformatics 2014, 31, 1296–1297. [Google Scholar] [CrossRef]
- Xie, J.; Chen, Y.; Cai, G.; Cai, R.; Hu, Z.; Wang, H. Tree visualization by one table (tvBOT): A web application for visualizing, modifying and annotating phylogenetic trees. Nucleic Acids Res 2023, 51, W587–W592. [Google Scholar] [CrossRef]
- Tang, H.; Bowers, J.E.; Wang, X.; Ming, R.; Alam, M.; Paterson, A.H. Synteny and collinearity in plant genomes. Science 2008, 320, 486–488. [Google Scholar] [CrossRef] [PubMed]
- Conesa, A.; Götz, S. Blast2GO: A comprehensive suite for functional analysis in plant genomics. Int J Plant Genomics 2008, 2008, 619832. [Google Scholar] [CrossRef] [PubMed]
- Popescu, S.C.; Popescu, G.V.; Bachan, S.; Zhang, Z.; Gerstein, M.; Snyder, M.; Dinesh-Kumar, S. MAPK target networks in Arabidopsis thaliana revealed using functional protein microarrays. Genes Dev 2009, 23, 80–92. [Google Scholar] [CrossRef] [PubMed]
| Gene name | Locus name | Identity with Stb6 | Amino acids | Molecular weight (kD) |
pI | Instability index | GUB-WAK domain (start-end) | WAK domain (start-end) | Subcellular localization |
|---|---|---|---|---|---|---|---|---|---|
| MdStb6-1 | MDP0000304127 | 35.01% | 650 | 72.7 | 6.06 | 36.00 | 34-105 | 203-252 | Cytoplasm, Nucleus |
| MdStb6-2 | MDP0000635134 | 33.23% | 574 | 63.9 | 5.21 | 39.42 | 36-100 | 180-247 | Cytoplasm, Nucleus |
| MdStb6-3 | MDP0000198217 | 35.88% | 616 | 67.9 | 6.41 | 38.56 | 32-101 | - | Cytoplasm, Nucleus |
| MdStb6-4 | MDP0000319460 | 26.50% | 723 | 82.0 | 6.76 | 43.04 | 37-100 | - | Nucleus |
| MdStb6-5 | MDP0000148991 | 31.48% | 604 | 68.8 | 8.51 | 39.56 | 24-90 | - | Cell membrane, Cytoplasm |
| MdStb6-6 | MDP0000258582 | 31.54% | 604 | 68.5 | 8.65 | 37.97 | 25-91 | - | Cell membrane, Cytoplasm |
| MdStb6-7 | MDP0000198624 | 30.35% | 621 | 69.7 | 8.35 | 44.2 | 51-114 | - | Cell membrane, Cytoplasm |
| MdStb6-8 | MDP0000277334 | 24.90% | 705 | 80.4 | 6.90 | 55.91 | 102-163 | - | Nucleus |
| MdStb6-9 | MDP0000282292 | 26.53% | 579 | 64.8 | 6.88 | 41.86 | 36-97 | - | Cell membrane, Cytoplasm, Nucleus |
| MdStb6-10 | MDP0000272246 | 27.42% | 602 | 67.8 | 6.08 | 41.7 | 29-94 | - | Cytoplasm, Nucleus |
| MdStb6-11 | MDP0000169760 | 23.12% | 610 | 68.9 | 8.44 | 39.39 | 39-102 | - | Cell membrane, Chloroplast, Cytoplasm, Nucleus |
| MdStb6-12 | MDP0000193657 | 35.47% | 590 | 65.8 | 6.88 | 34.13 | - | 143-192 | Cell membrane, Cytoplasm, Nucleus |
| MdStb6-13 | MDP0000152330 | 34.89% | 636 | 71.6 | 6.09 | 38.26 | 36-102 | 169-248 | Nucleus |
| MdStb6-14 | MDP0000147988 | 34.93% | 670 | 74.2 | 5.91 | 38.95 | 37-103 | 196-268 | Nucleus |
| MdStb6-15 | MDP0000190181 | 34.26% | 526 | 58.5 | 8.01 | 36.45 | - | 92-158 | Nucleus |
| Gene ontology analysis | Numbers of MdStb6 homolog genes |
| Biological process | |
| Phosphorylation | 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15 |
| Response to metal ion | 8, 12 |
| Cellular compartment | |
| Membrane | 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 |
| Extracellular region | 1, 3, 4, 6, 9, 10, 11, 13 |
| Plasma membrane | 4, 11, 13, 15 |
| Molecular function | |
| ATP binding | 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15 |
| Protein serine/threonine kinase activity | 1, 2, 3, 4, 5, 7, 8, 9, 10, 11, 12, 13, 14, 15 |
| Protein serine kinase activity | 1, 2, 3, 4, 5, 6, 9, 10, 11, 13, 15 |
| Polysaccharide binding | 1, 2, 3, 4, 5, 9, 10 |
| Kinase activity | 2,3, 6, 7,9, 12, 15 |
| Protein kinase activity | 6, 9, 11 |
| Ubiquitin protein ligase binding | 11, 13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




