Discussion
There is no doubt that significant advances have been made in cancer therapy over the past decades. However, prognoses show that current treatments have not led—and are unlikely to lead—to a true breakthrough in oncology.7 The greatest paradox of the prevailing approach is that, despite recognizing the immense complexity of genetic and biochemical processes of the cells, we seek solutions by targeting specific genetic mutations or using single molecules to selectively inhibit or influence isolated processes or, more precisely, the functional proteins responsible for them.
More than forty years ago Albert Szent-Györgyi raised the question of whether harmonic regulation of the rapid and complex biochemical and genetic processes of a cell is (or can be) realized at the molecular level. He assumed that large protein molecules (the typical main targets of drug development) were incapable of regulating these processes, and stated that a sub-molecular regulatory system, based on electrons—very lightweight and mobile elementary particles -, fulfills this role.28 The data obtained with DDW7-10 confirmed that sub-molecular changes, namely, increase or decrease of D/H ratio, significantly impact the processes within the cells.
This cellular effect of altering the D/H ratio by deuterium depletion resulted at a systemic level in a severalfold increase in the median survival time (MST) of cancer patients consuming DDW in the studies referred to above19-22 compared to historical controls. These findings underscore the importance of identifying key parameters for fully harnessing the potential of deuterium depletion when integrating it into conventional cancer therapy.
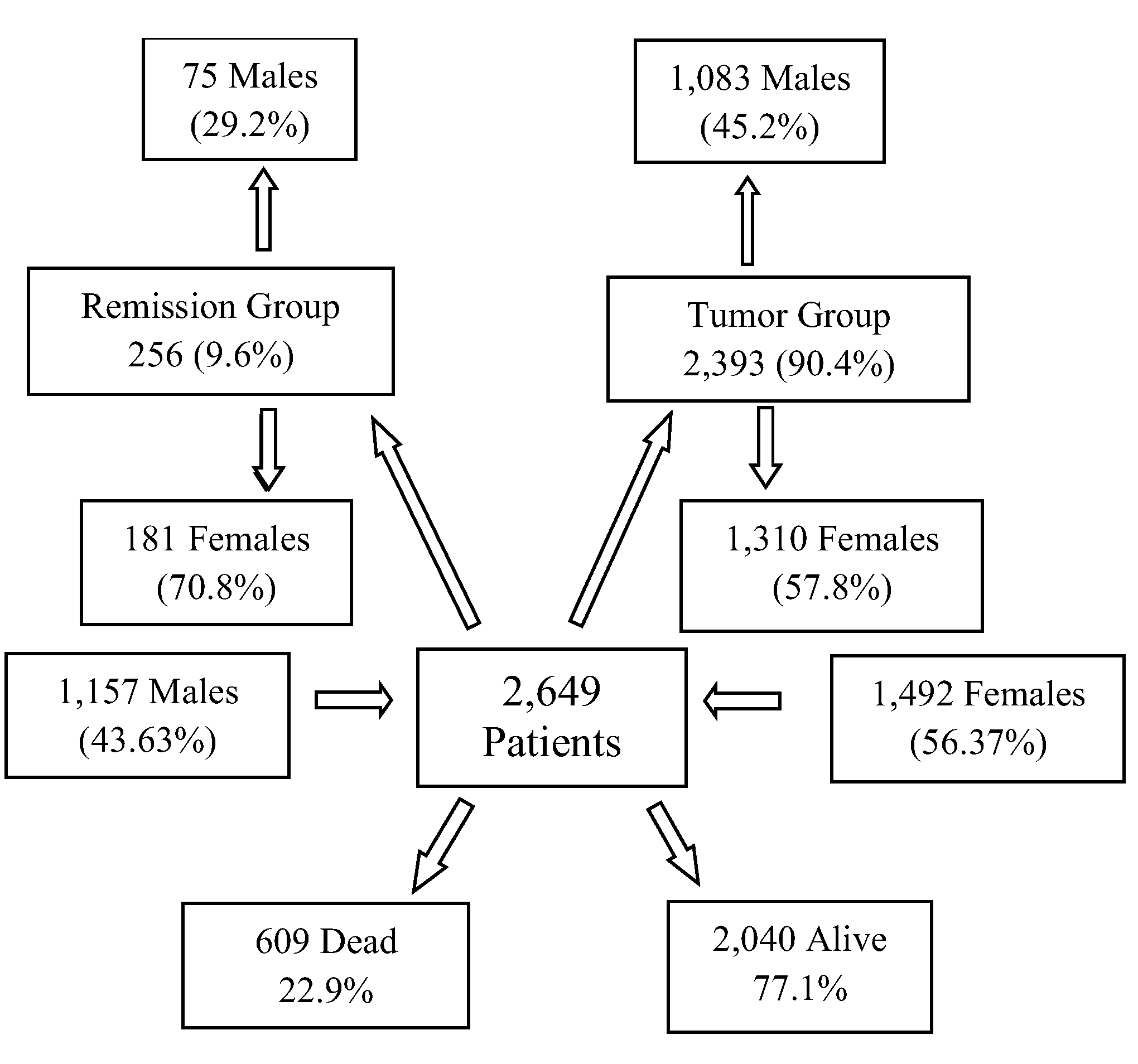
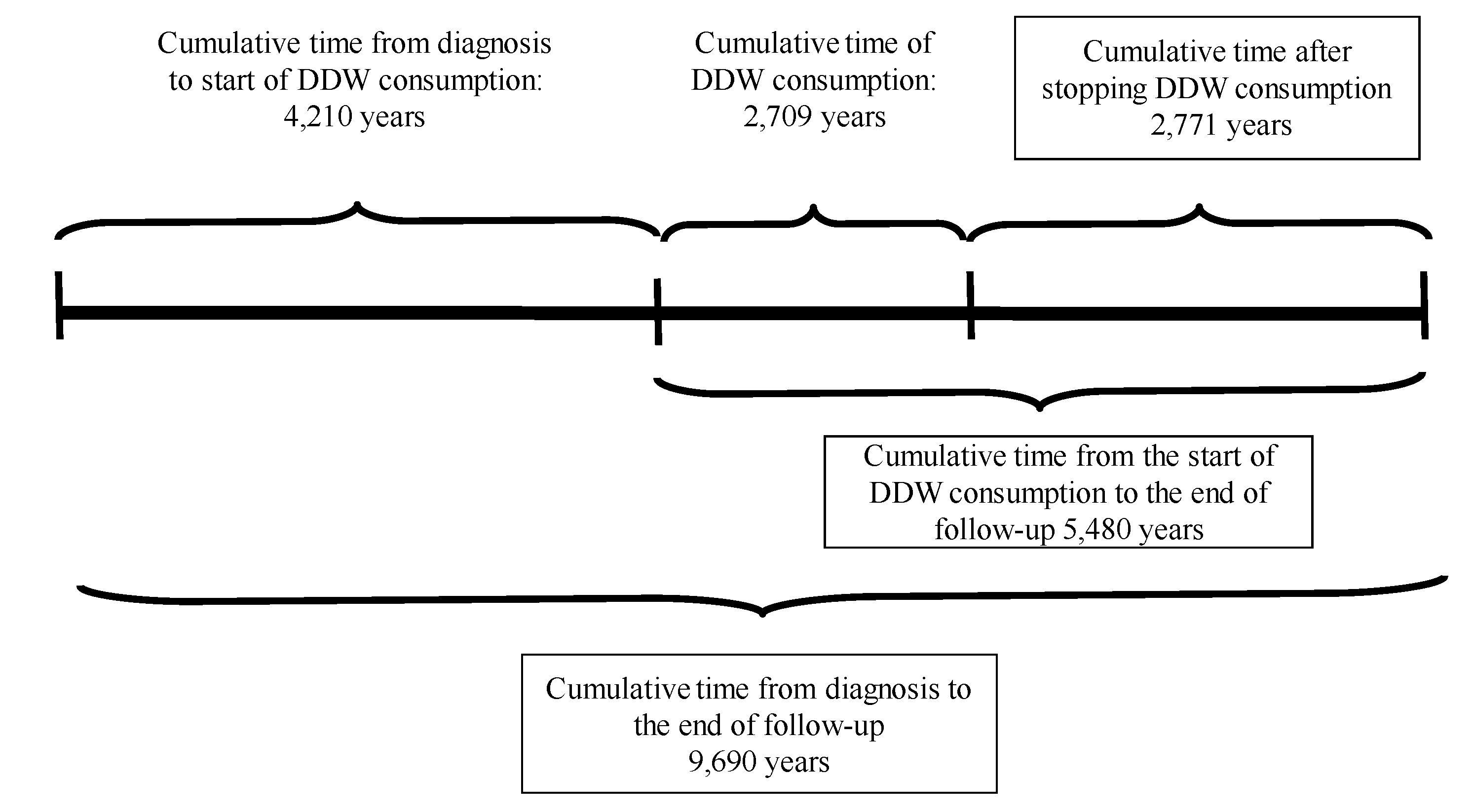
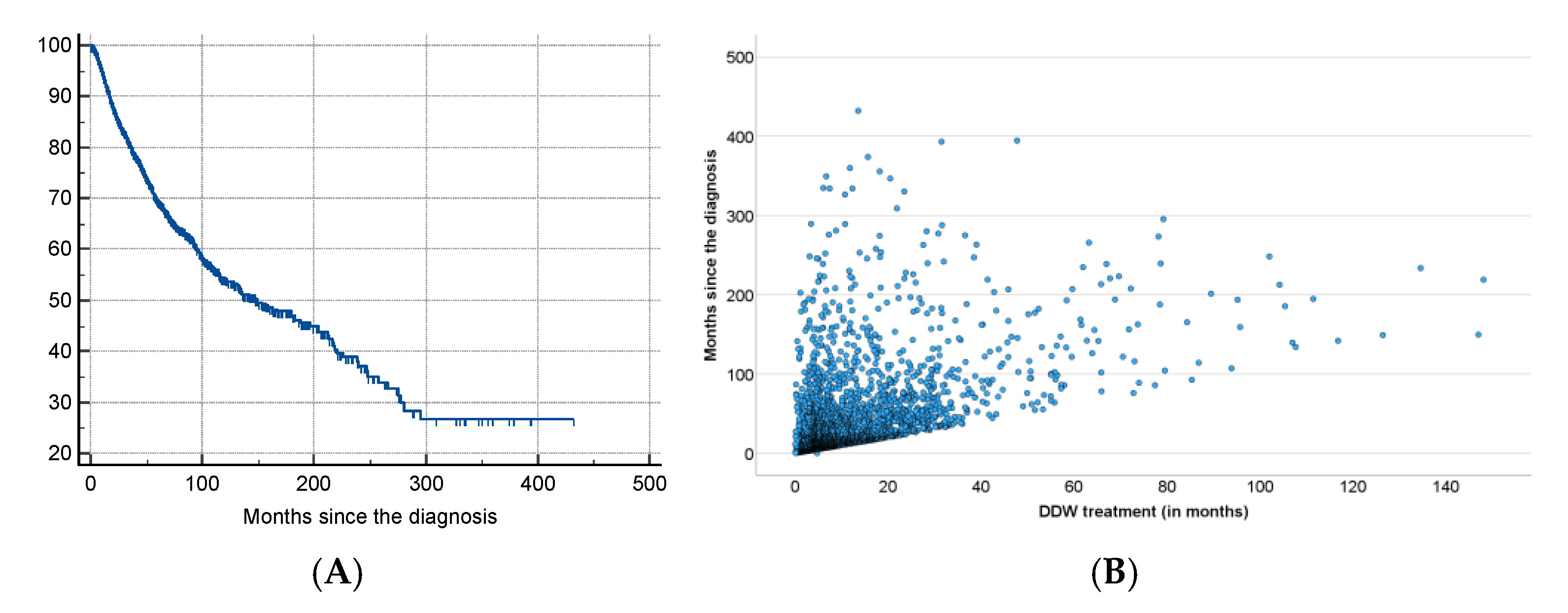
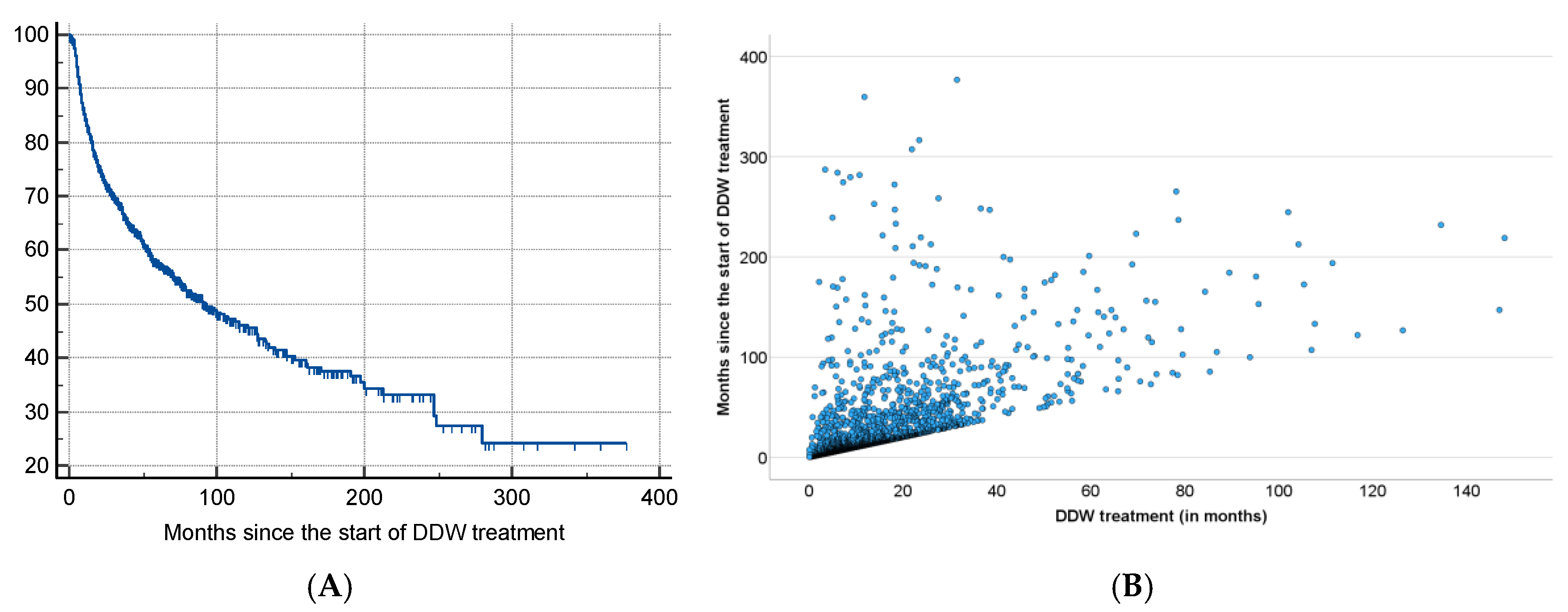
The results of the present study, based on real-world data of 2,649 patients consuming DDW, showed the possible significant impact of integrating deuterium depletion into conventional oncotherapy, and highlighted both its potentials and limitations. The MST for the patients involved increased to 12.4 years from the time of diagnosis and to 7.6 years from the start of DDW consumption, representing a five- and three-fold increase compared to the 2.4-year MST of the overall Hungarian cancer population. Among the patients with detectable tumors (TG group) MST was 5.8 years from the initiation of DDW consumption. In the RG group (patients in remission when starting deuterium depletion) MST from the start of DDW consumption was 23.2 years.
To find the optimal conditions for integrating DDW into conventional therapy, subgroups within the 2,649 cases were defined based on time parameters (see
Tables 4, 5, 6 and 8), and their MST was analyzed. It was concluded that, to maximize the efficacy of DDW, a life expectancy of not less than 3-4 months at the start of DDW consumption is necessary, the length of DDW consumption should be longer than 90-120 days, and DDW consumption should start not later than 9 months after diagnosis. The evidence for these requirements, MST data for all the subgroups calculated from the start of DDW consumption, are summarized in
Table 9.
The data showed that excluding patients with a short application period (who died within 90–120 days after start of DDW or who consumed DDW for less than 90–120 days) increased the MST from 5.8 years to 6.1–6.5 years. When both criteria were applied, MST for patients who lived longer than 90–120 days and consumed DDW for more than 90–120 days went up to 8.0–8.7 years.
However, the most critical factor, highlighted by the numbers in
Table 9, was the time gap between diagnosis and initiation of DDW consumption. When DDW consumption started within 9 months after diagnosis, the MST rose to 11.3 years (95% CI: 8.4–14.1), but it dropped to 9.6 years (95% CI: 7.2-11.8) starting over a year after diagnosis. When the calculation was restricted to patients who lived longer than 120 days after starting DDW and consumed it for more than 120 days, only a minimal additional increase in MST (to 11.6 years) was observed.
This population, who began to use DDW within 9 months after diagnosis, achieved a nearly 5-fold increase in MST compared to the average Hungarian cancer population, emphasizing the importance of timely and sustained DDW consumption in improving survival outcomes.
With an MST of 11.6 years, Hungary’s annual cancer death toll could drop from 33,000 to approximately 5,000–7,000, potentially saving 26,000–28,000 lives each year. In Europe, the cancer death toll could be reduced by 825,000 to 880,000 lives through the integration of deuterium depletion into conventional therapy. This represents a dramatic improvement for the cancer population, underscoring the potential life-saving impact of integrating DDW consumption into therapy.
Deuterium depletion constitutes a paradigm-shifting approach by abandoning the focus on single targets within the cell regulation. Gene expression studies indeed demonstrated that altering deuterium concentration had a profound impact on entire cells or organisms.
In humans, 200–300 genes regulate the cell cycle directly, while many additional genes indirectly engage in related signaling pathways and repair mechanisms. Two previous studies[
25,
30] demonstrated that the expression of specific genes associated with tumor development, such as c-Myc and K-Ras, H-Ras, Bcl2, p53, was inhibited in carcinogen-treated mice consuming DDW. This suggests a potential role of intracellular deuterium level in modulating gene expression linked to tumorigenesis, offering promising insights for future research and therapeutic applications.
An elevated D/H ratio seems thus essential for triggering the expression of specific genes associated with tumor development. A recent study[
31] utilizing nanostring technology investigated the expression of 236 cancer-related genes and 536 kinase genes in deuterium-depleted (40 and 80 ppm D), deuterium-enriched (300 ppm), and regular (150 ppm) media. From the total, 124 cancer-related genes and 135 kinase genes (those with expression changes exceeding 30% and a copy number above 30) were evaluated.
Only seven genes exhibited altered expressions (one upregulated and six downregulated) in deuterium-depleted media, but 97.3% of the evaluated genes were upregulated on deuterium enrichment. These findings align with the above-mentioned
in vivo mouse studies, demonstrating that DDW keeps the D concentration at a low level and inhibits its rise to the threshold necessary to trigger the expression of genes induced by carcinogens. Research confirmed that consuming DDW prevents the increase in the D/H ratio, thereby blocking the activation of the entire set of genes involved in cell cycle regulation. In harmony with these results, a cell cycle analysis revealed that DDW caused cell cycle arrest in the G1/S transition, reduced the number of cells in the S phase, and significantly increased the population of cells in the G1 phase.[
32]
DDW reduces the body’s deuterium concentration[
33] effectively mimicking the role of mitochondria in producing deuterium-depleted metabolic water. Studies suggest that lowering deuterium levels impacts cellular metabolism and generates free radicals[
29], demanding rapid and efficient adaptive responses. Healthy cells with fully functional mitochondria and metabolism can successfully manage this metabolic challenge. In contrast, cancer cells, which typically lack such mitochondrial functionality, fail to adapt, leading to apoptosis, necrosis, and observable tumor regression.[
9,
10]
Safety is a primary concern in the case of any novel therapeutic method to be applied in humans. As for deuterium depletion, the consumption of DDW with 25–125 ppm deuterium content has proven to be safe, with no unexpected toxic or harmful effects observed.[
19,
26,
27] In contrast, some beneficial effects beyond tumor growth inhibition were observed. In a Phase 2 clinical trial involving 30 patients with pre or manifest diabetes, significant increase in blood cell counts within the normal range and a reduction in fasting blood glucose levels were observed.[
26] These findings were consistent with observations in cancer patients undergoing chemotherapy and consuming DDW, where significant blood count deterioration was either absent or delayed. A general improvement in the patients’ physical strength and well-being, associated with DDW consumption, was also repeatedly found. The underlying mechanism is likely the one described in a test with top athletes, where a 44-day regimen of 105 ppm DDW consumption resulted in a delayed increase in lactic acid levels and a reduced anion gap, indicating more efficient mitochondrial function.[
27]
On the other hand, tumor necrosis caused by deuterium depletion was not without some kind of side effects. The most characteristic changes were weakness, drowsiness, increased temperature, fever spikes, intermittently increasing pain, swelling, and softening of the tumor-affected area, minor bleeding in the bladder, stomach, or rectum, brick dust urine, and transient coughing in lung cancer.24 All these side effects were tolerable by the patients and were transient.
In several cases, patients involved in the present study used various supplementary or alternative treatments in parallel with conventional oncotherapy and DDW consumption, and the data suggested a possible negative interaction between deuterium depletion and certain supplementary methods. After a thorough evaluation, several factors were identified that significantly diminished the effectiveness of DDW or rendered it entirely ineffective. These included high doses of antioxidants, such as vitamins A, C, E, and selenium; consumption of Coenzyme Q10 supplements; iron supplementation; intense and prolonged physical exertion; as well as the use of hot tubs and saunas. Understanding these influences can provide critical guidance for optimizing the efficacy of DDW application and highlights the importance of managing patient lifestyles and supplementary treatments during deuterium depletion.
The groundbreaking findings in this study underscore the pivotal role of the D/H ratio in regulating and coordinating millions of molecular processes, including biochemical and genetic functions involved in carcinogenesis. At the eukaryotic level, a sub-molecular regulatory system (SMRS) governs the intricate complexity of life. Utilization of the capacities of DDW enables an intervention in this fundamental regulatory system, opening new avenues for therapeutic applications and a deeper understanding of cellular processes.
Supported by HYD LLC for Cancer Research and Drug Development, Budapest, Hungary
We sincerely thank the patients consuming DDW and their families for voluntarily sharing their experiences and outcomes before, during, and after DDW consumption. Their insights, along with the results of follow-up oncology examinations confirming the effectiveness of conventional treatments, are invaluable.