Submitted:
03 March 2025
Posted:
04 March 2025
You are already at the latest version
Abstract
Grafting is widely used as a breeding method to enhance productivity and resilience. However, the mechanisms of graft healing remain poorly understood. In this study, we performed Malus domestica (‘Hanfu’) homograft and observed morphological and anatomical changes during the healing process in the graft junction within 40 days after grafting (DAG) with focus on vascular connection between the stock and the scion traced with acid fuchsin dye. The observation showed different phases in the healing process. Phase I, from 0 to 20 DAG, involved formation and proliferation of callus, while phase II, from 20 to 40 DAG, involved reestablishment of vascular connection between the stock and the scion. In order to obtain an in-depth understanding of graft healing processes, transcriptomic and metabolomic changes were analyzed in the stock and the scion tissues at the graft junction in different phases. The transcriptomic data showed different gene expression profiles in the scion and stock in phase I, while gene expression profiles were similar between the scion and the stock during phase II, when differentially expressed genes (DGEs) (20 DAG vs 40 DAG) were highly enriched in pathway of sugar metabolism. In agreement with the transcriptomic result, there was significant difference in metabolite profile between the stock and the scion at 20 DAG, while their metabolomic difference reduced at 40 DAG. Metabolomic analysis highlighted increase in many sugars such as sucrose, glucose, raffinose and melibiose before vascular reconnection (phase I) while decrease in these sugars after vascular reconnection (phase II). Exogenous application of glucose, sucrose, raffinose and melibiose promoted vascular reconnection. The results suggest a shift from asymmetry to relative symmetry in gene expression and metabolism between the stock and the scion during the process of graft junction formation, which involves changes in sugar metabolism. Sugar accumulation is important for the healing process between the stock and the scions.
Keywords:
1. Introduction
2. Results
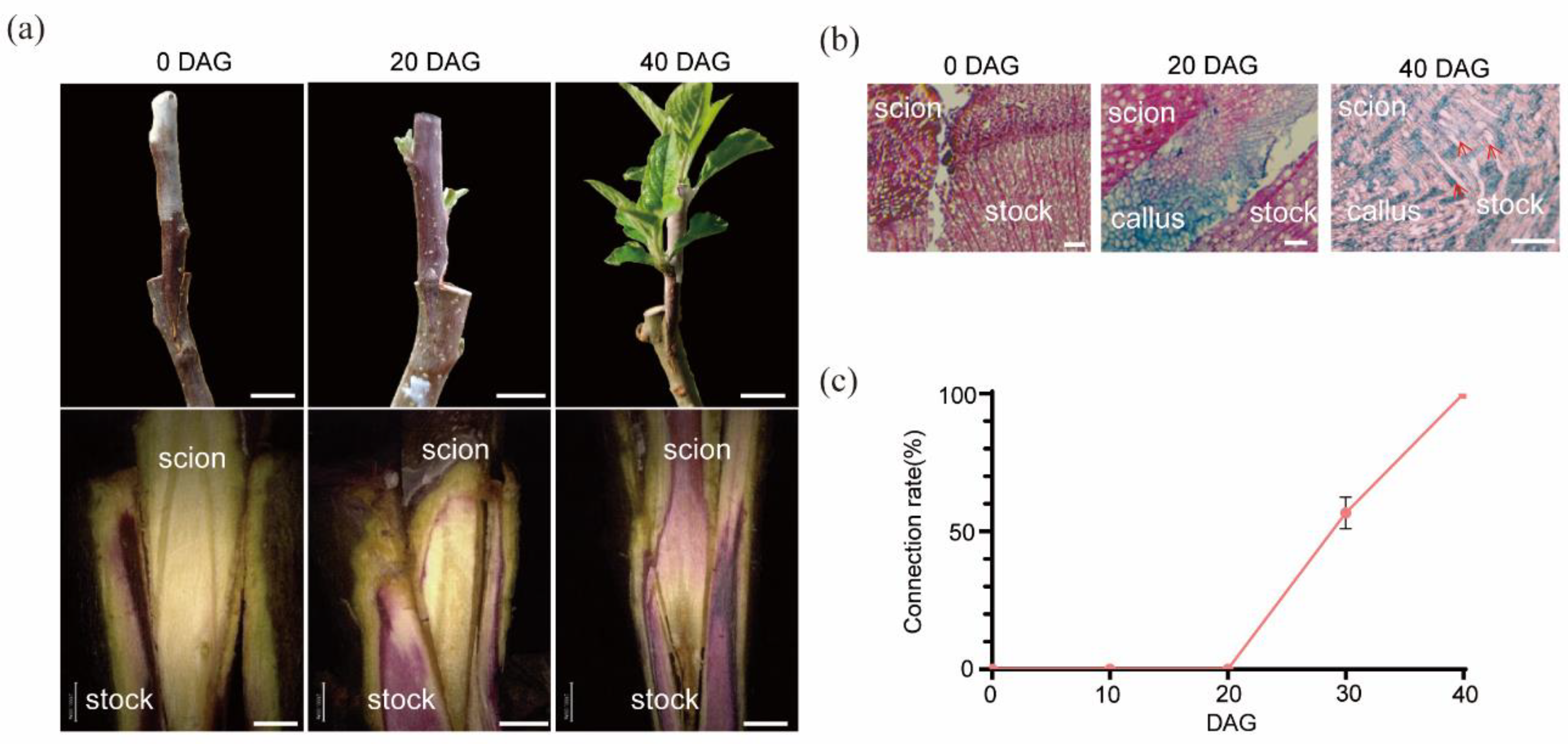
The Process of the Graft Junction Formation
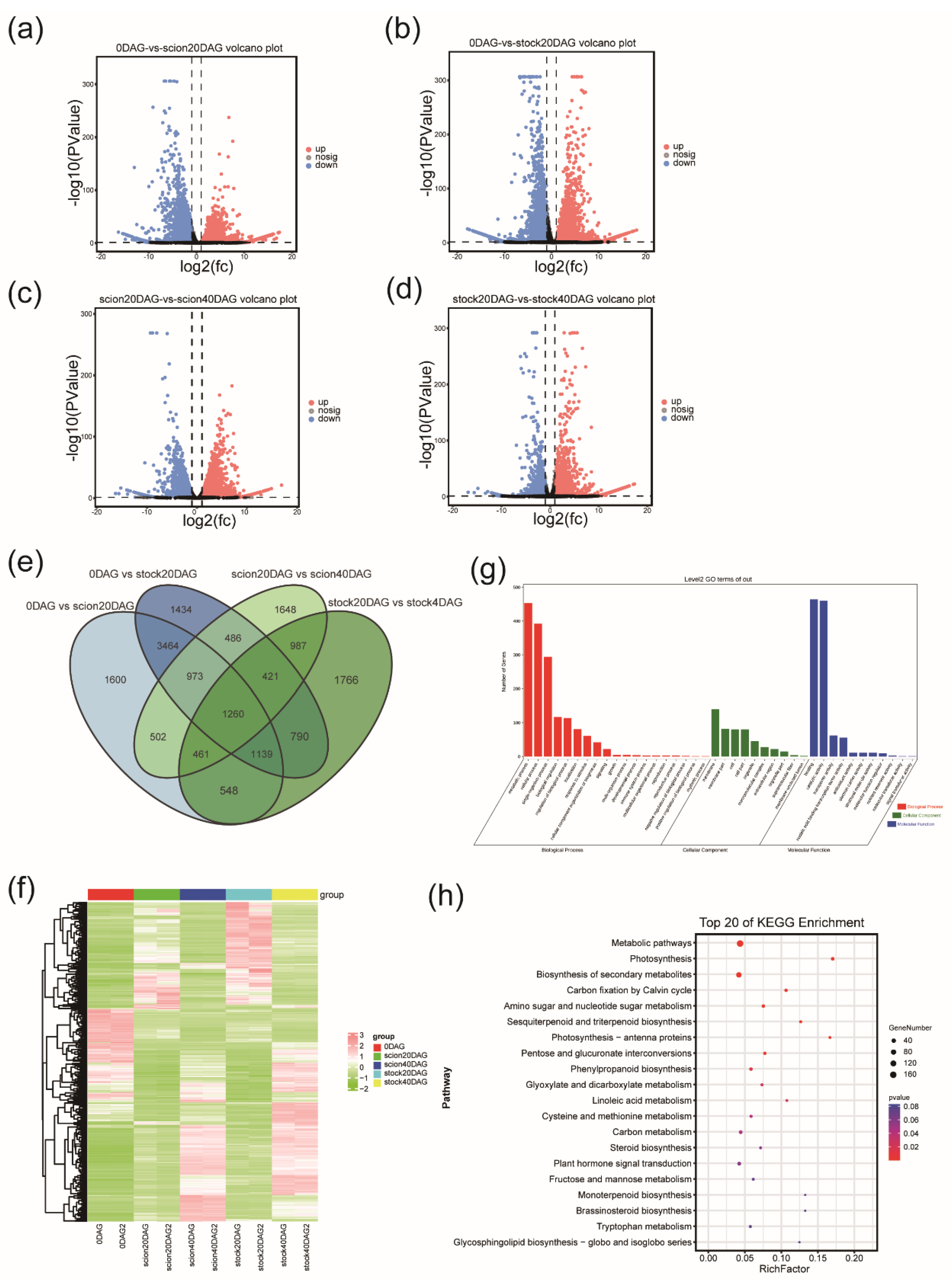
RNA Sequencing and Functional Annotation of DEGs
Weighted Gene Co-Expression Network Analysis (WGCNA)
Pairwise Transcriptomic Comparison Between Stages of Graft Junction Formation
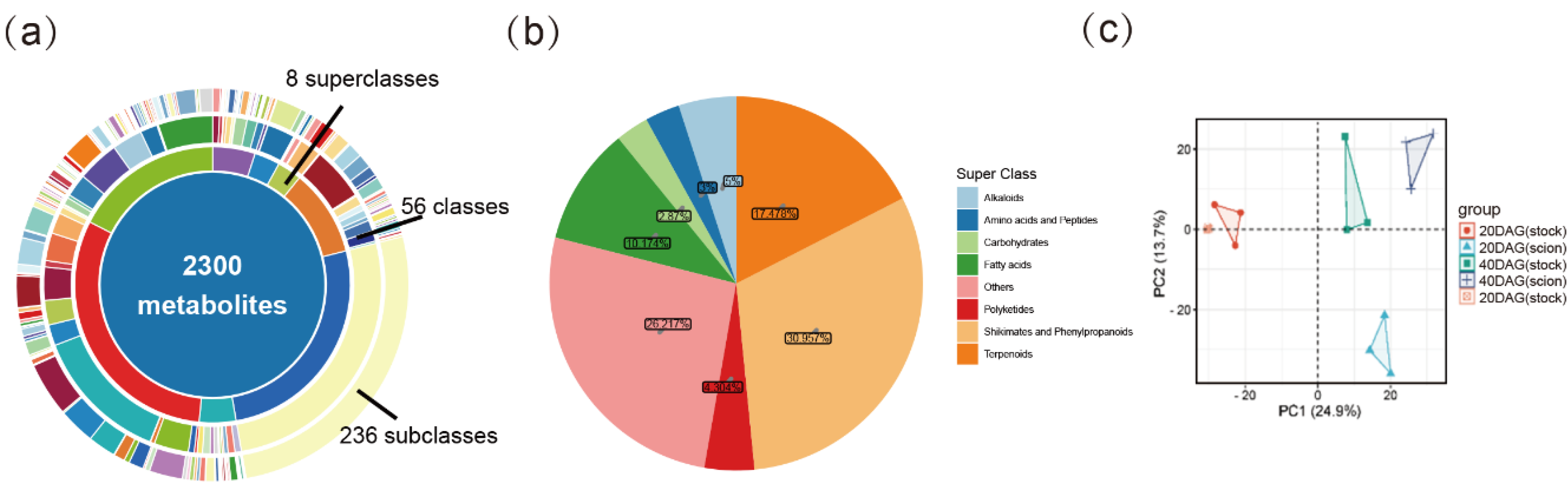
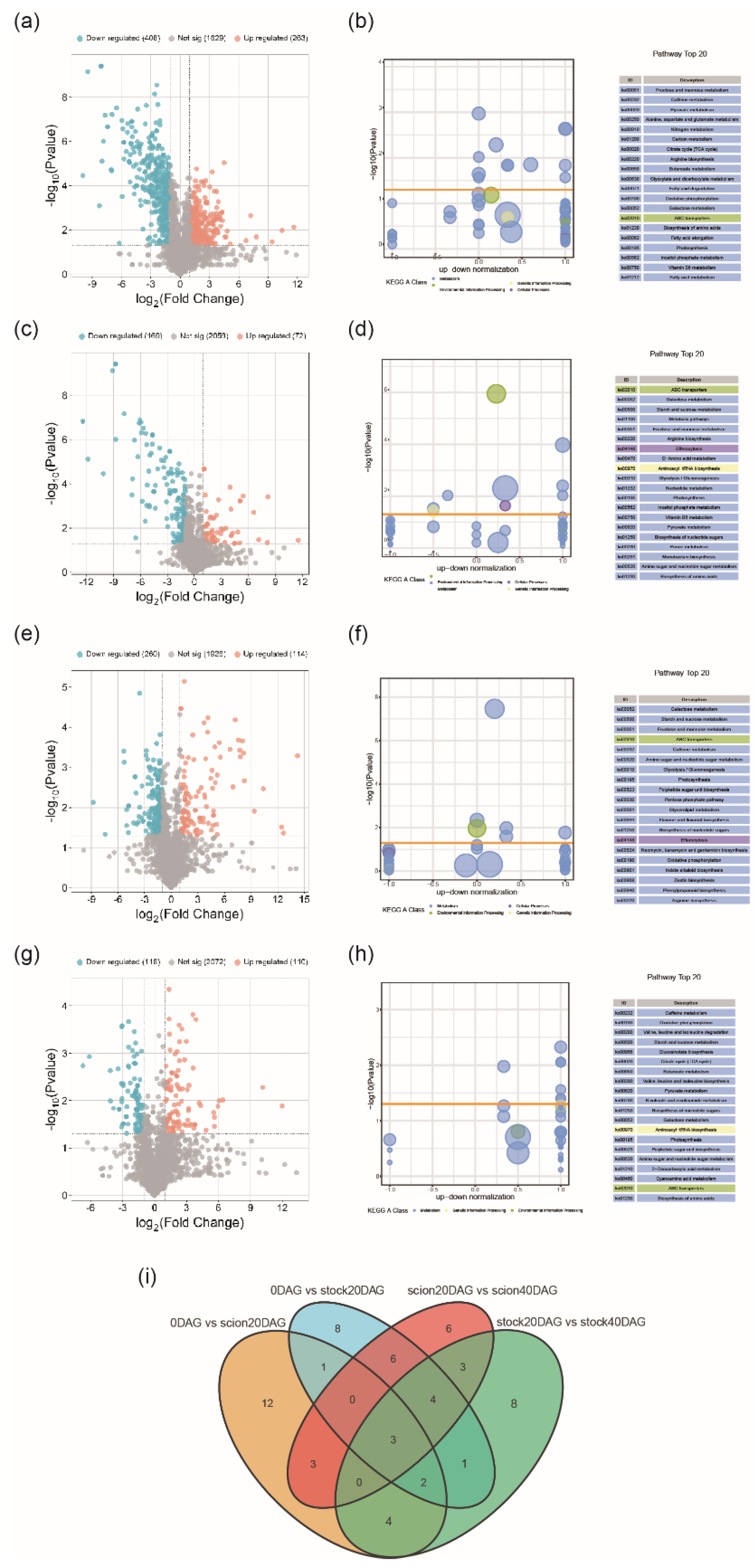
Changes in Metabolite Profiling During Graft Junction Formation
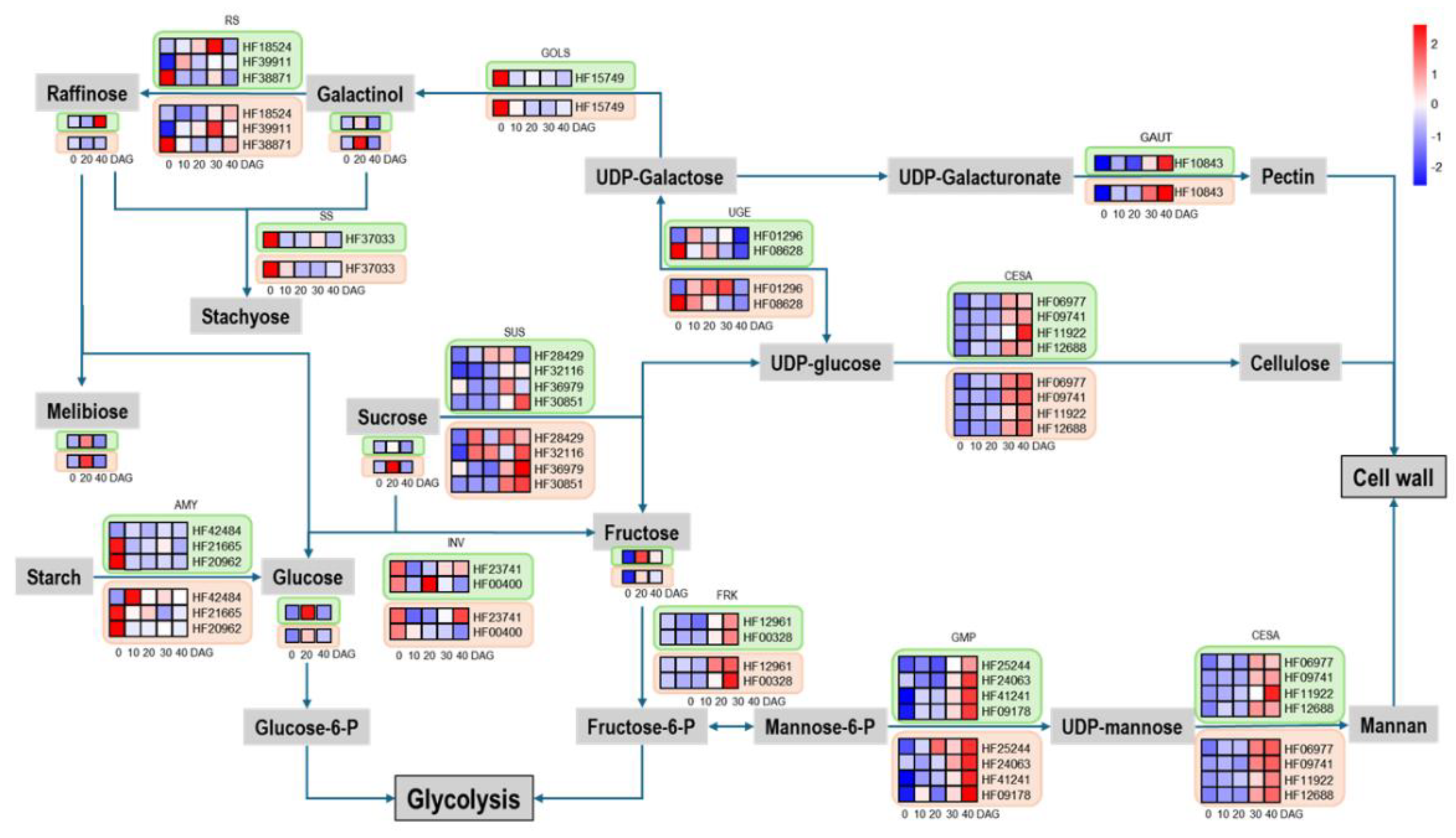
Sugar Metabolism Shift During Graft Junction Formation
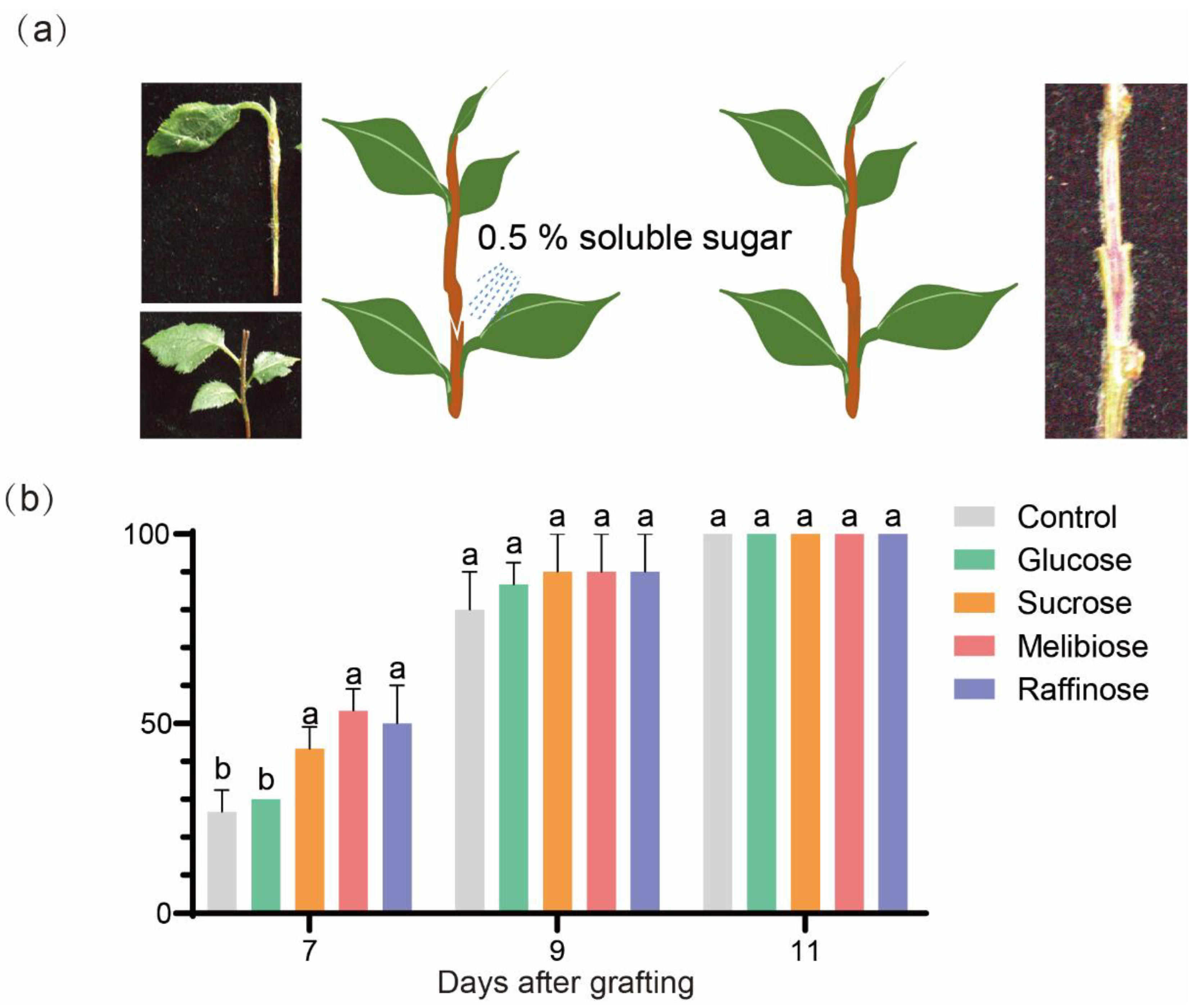
Effects of Sugar Treatments on Graft Junction Formation
3. Discussion
Graft Junction Formation Occurs in Phases
Scion vs Stock Asymmetry Response to Grafting is more Pronounced in Phase I
Graft Junction Formation Involves Shift in Sugar Metabolism and Is Dependent on Sugar Supply
Exogenous Sugars Promoted Graft Junction Formation
4. Materials and Methods
Plant Materials and Grafting Methods
Anatomical Observation for Timeline of Graft Junction Formation
Transcriptome Sequencing and Analyses
Metabolomic Analysis
RT–qPCR Validation
Exogenous Soluble Sugars Treatments on Grafted Plantlets
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huang, Y., Kong, Q. S., Chen, F. & Bie, Z. L. (2014) The history, current status and future prospects of vegetable grafting in China. I Int. Symp . Veg. Grafting 1086, 31–39. [CrossRef]
- Mudge, K., Janick, J., Scofield, S., Goldschmidt, E. E. (2009). A history of grafting. Horticultural Reviews, 35. 35, 437–493. [CrossRef]
- Tedesco, S., Pina, A., Fevereiro, P., Kragler, F. (2020). A phenotypic search on graft compatibility in grapevine. Agronomy, 10(5), 706. [CrossRef]
- Goldschmidt, E. E. (2014). Plant grafting: New mechanisms, evolutionary implications. Frontiers in Plant Science, 5(727), 727. [CrossRef]
- Gur A, Samish RM, Lifshitz E. (1968). Role of cyanogenic glycoside of quince in incompatibility between pear cultivars and quince rootstocks. Horticultural Research 8, 113–134.
- Kollmann R, Yang S, Glockmann C. (1985). Studies on graft unions. II. Continuous and half plasmodesmata in different regions of the graft interface. Protoplasma 126, 19–29.
- Kurotani KI, Notaguchi M. 2021. Cell-to-cell connection in plant grafting—Molecular insights into symplasmic reconstruction. Plant and Cell Physiology 62, 1362–1371. [CrossRef]
- Jeffree CE, Yeoman MM. (1983). Development of intercellular connections between opposing cells in a graft union. New Phytologist 93, 491–509. [CrossRef]
- Flaishman MA, Loginovsky K, Golobowich S, Lev-Yadun S. (2008). Arabidopsis thaliana as a model system for graft union development in homografts and heterografts. Journal of Plant Growth Regulation 27, 231–239. [CrossRef]
- Cookson SJ; et al. (2013) Graft union formation in grapevine induces transcriptional changes related to cell wall modification, wounding, hormone signalling, and secondary metabolism. J Exp Bot 64:2997–3008. [CrossRef]
- Chen, Z., Zhao, J. , Hu, F. , Qin, Y. , Wang, X., Hu, G.(2017). Transcriptome changes between compatible and incompatible graft combination of litchi chinensis by digital gene expression profile. Scientific Reports, 7(1), 3954. [CrossRef]
- Zheng BS; et al. (2010) cDNA-AFLP analysis of gene expression in hickory (Carya cathayensis) during graft process. Tree Physiol 30:297–303. [CrossRef]
- Thomas H, Van den Broeck L, Spurney R, Sozzani R, Frank M. (2021). Gene regulatory networks for compatible versus incompatible grafts identify a role for SlWOX4 during junction formation. The Plant Cell 34, 535–556. [CrossRef]
- Mazur E, Benková E, Friml J. (2016). Vasc cambium regeneration and vessel formation in wounded inflorescence stems of Arabidopsis. Scientific Reports 6, 33754. [CrossRef]
- Yin H, Yan B, Sun J, Jia P , Zhang Z, Yan X, Chai J, Ren Z, Zheng G, Liu H. (2012). Graft-union development: A delicate process that involves cell–cell communication between scion and stock for local auxin accumulation. Journal of Experimental Botany 63, 4219–4232. [CrossRef]
- Notaguchi, M., Kurotani, K. I., Sato, Y., Tabata, R., Kawakatsu, Y., Okayasu,K.; et al. (2020). Cell-cell adhesion in plant grafting is facilitated by β-1,4-glucanases. Science 369, 698–702. [CrossRef]
- Wang, Y. & Kollmann, R. (1996). Vascular differentiation in the graft union of in-vitro grafts with different compatibility.—Structural and functional aspects. J. Plant Physiol. 147, 521–533. [CrossRef]
- Aloni, R. (1980). Role of auxin and sucrose in the differentiation of sieve and tracheary elements in plant tissue cultures. Planta 150, 255–263. [CrossRef]
- Marsch-Martinez, N., Franken, J., Gonzalez-Aguilera, K.L., Folter, S., Angenent, G.C., Alvarez-Buylla, E.R. (2013). An efficient flat-surface collar-free grafting method for Arabidopsis thaliana seedlings. Plant Methods 9, 14. [CrossRef]
- Zhang L, Hu J, Han X, Li J, Gao Y, Richards C M, Zhang C, Tian Y, Liu G, Gu H; et al. (2019). A high-quality apple genome assembly reveals the association of a retrotransposon and red fruit colour. Nat Commun 10, 1494. [CrossRef]
- Cookson, S., J., Clemente, Moreno, & M.; et al. (2014). Heterografting with nonself rootstocks induces genes involved in stress responses at the graft interface when compared with autografted controls. Journal of Experimental Botany. 65, 2473–2481. [CrossRef]
- Pina, A., and Errea, P. (2005). A review of new advances in mechanism of graft compatibility–incompatibility. Sci. Hortic. 106, 1–11. [CrossRef]
- Pina A, Errea P, Martens HJ. (2012). Graft union formation and cell-to-cell communication via plasmodesmata in compatible and incompatible stem unions of Prunus spp. Scientia Horticulturae 143, 144–150. [CrossRef]
- Tiedemann R. (1989). Graft union development and symplastic phloem contact in the heterograft Cucumis sativus on Cucurbita ficifolia. Journal of Plant Physiology, 134(4), 427-440. [CrossRef]
- Melnyk CW, Schuster C, Leyser O, Meyerowitz E. (2015). A developmental framework for graft formation and vascular reconnection in Arabidopsis thaliana. Curr Biol, 25 (10): 1306–1318. [CrossRef]
- Melnyk CW, Gabel A, Hardcastle TJ, Robinson S, Miyashima S, Grosse I, Meyerowitz EM. (2018). Transcriptome dynamics at Arabidopsis graft junctions reveal an intertissue recognition mechanism that activates vascular regeneration. Proceedings of the National Academy of Sciences, USA 115, E2447–E2456. [CrossRef]
- Harrison, M. A. & Pickard, B. G. (1989). Auxin asymmetry during gravitropism by tomato hypocotyls. Plant Physiology, 89(2), 652-657. [CrossRef]
- Asahina M, Azuma K, Pitaksaringkarn W, Yamazaki T, Mitsuda N, Ohme-Takagi M, Yamaguchi S, Kamiya Y, Okada K, Nishimura T, Koshiba T, Yokota T, Kamada H, Satoh S. (2011). Spatially selective hormonal control of RAP2.6L and ANAC071 transcription factors involved in tissue reunion in Arabidopsis. PNAS, 108 (38) 16128-16132. [CrossRef]
- Zhu TF, Wang Y, Su XL, Li X, Wang LC. (2022). Physiological characters in the stock and the scion during graft healing and their relation to graft compatibility in grapefruit. Jiangsu Agricultural Science, 50 (13) : 141-147. [CrossRef]
- Wang L, Ruan YL (2013) . Regulation of cell division and expansion by sugar and auxin signaling. Front Plant Sci, 4: 163. [CrossRef]
- Lopes M A, Larkins B A. (1993). Endosperm origin, development, and function. The Plant Cell, 5(10): 1383-1399. [CrossRef]
- Liu C, Hong J, Xia G, Huang Ji. (2009). Cytological observation on healing responses in grafting of Carya cathayensis. Scientia Silvae Sinicae, 45(6): 34-38.
- Miao L, Li Q, Sun TS, Chai S, Wang CL, Bai LQ, Sun MT, Li YS, Qin X, Zhang ZH, Yu XC. (2021). Sugars promote graft union development in the heterograft of cucumber onto pumpkin. Hortic Res 8, 146, 1-17. [CrossRef]
- Hoermayer L, Montesinos JC, Marhava P, Benková E, Yoshida S, Friml J. (2020). Wounding-induced changes in cellular pressure and localized auxin signalling spatially coordinate restorative divisions in roots. Proc Natl Acad Sci USA. Jun 30;117(26):15322-15331. [CrossRef]
- Cohen, R., Karni, L., Aktas, H. & Edelstein, M. (2010). Hormonal signaling in rootstock-scion interactions. Sci. Hortic. 127, 119–126. [CrossRef]
- Button J. (1978). The Effects of some Carbohydrates on the Growth and Organization of Citrus Ovular Callus. Zeitschrift für Pflanzenphysiologie, 88(1): 61-68. [CrossRef]
- Rasool A, Mansoor S, Bhat K M, Hassan G I, Baba T R, Alyemeni M N, Alsahli A A, El-Serehy H A, Paray B A, Ahmad P. (2020). Mechanisms Underlying Graft Union Formation and Rootstock Scion Interaction in Horticultural Plants. FRONTIERS IN PLANT SCIENCE, 11. [CrossRef]
- Li F, Wang XF. (2008). Advances in the Metabolism and Regulatory Key Enzymes of Raffinose Series Oligosaccharides in Plants. Acta Botanica Boreali-Occidentalia Sinica, 28(4): 8. [CrossRef]
- Liao, Y.; Smyth, G.K.; Shi, W. (2014). featureCounts: An efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics, 30, 923–930. [CrossRef]
- Varet, H.; Brillet-Guéguen, L.; Coppée, J.-Y.; Dillies, M.-A. (2016) SARTools: A DESeq2-and EdgeR-based R pipeline for comprehensive differential analysis of RNA-Seq data. PLoS ONE, 11, e0157022. [CrossRef]
- Westerhuis, J.A.; van Velzen, E.J.; Hoefsloot, H.C.; Smilde, A.K.(2010). Multivariate paired data analysis: Multilevel PLSDA versus OPLSDA. Metabolomics, 6, 119–128. [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



