Submitted:
01 March 2025
Posted:
05 March 2025
You are already at the latest version
Abstract
The growing need to reduce methane emissions from ruminants while enhancing feed utilization has driven the development of innovative in vitro measurement techniques. This review examines the Gas Endeavour (GE) system, an automated volumetric apparatus that quantifies both total gas and methane production in real time during rumen fermentation. Utilizing principles of liquid displacement and buoyancy, the GE system integrates a thermostatically controlled water bath, specialized gas flow cells, and an alkaline CO₂ absorption unit to deliver precise kinetic data on fermentation. Compared to conventional methods—which often rely on manual measurements and post-incubation gas chromatography—the GE system provides continuous monitoring and immediate data acquisition, reducing labor and potential errors. The review discusses the system’s design, operational challenges such as controlling headspace pressure and ensuring consistent inoculum preparation, and its applications in both animal nutrition and biomethane potential assessments. The findings suggest that, with further standardization and protocol refinement, the GE system could significantly advance research aimed at optimizing feed digestibility and mitigating methane emissions in ruminant production systems.
Keywords:
1. Introduction
2. In Vitro Gas Production Techniques (IVGPT)
2.1. Siringe Method (Hohenheim Gas Test)
2.2. In vitro Methane Measuring Techniques
2.5. Limitations of IVGPT
| Reference | Device | Water-bath / air incubator | Devise volume, mL |
Inoculum / medium ratio, mL/mL |
Buffer reference |
Duration of incubation, h | Dietary substrate, buffered medium ratio mg/mL |
Gas venting, and collection | Pressure control | Gas measurement, and analysis |
| Menke et al. [19] | Syringes | Water rotor-bath | 30 | 1:2 | [19] | 24 | 6.67 | Manual, Endpoint sampling |
Yes, moveable glass piston | Manual, no analysis |
| Theodorou et al. [24] | Bottles | Air incubator |
60 | 1:9 | [24] | 24 – 72 | 2-20 | Manual, Endpoint sampling |
No, pressure increase | Manual, no analysis |
| Mauricio et al. [38] | Bottles | Air incubator |
100 | 1:9 | [24] | n.s. | 10.0 | Manual, Endpoint sampling |
No, pressure increase | Manual, no analysis |
| Pell and Schofield [23] | Bottles stirred |
Air incubator |
10 | 1:4 | [43] | n.s. | 10.0 | Manual Endpoint sampling |
No, pressure increase | Manual, CH4 by GC |
| Cone et al. [28] | Bottle shaked |
Water bath |
100 | 1:2 | [44] | 48 | 6.67 | Automated Fixed pressure |
Yes | Manual, |
| Davies et al. [45] | Bottles | Air incubator |
100 | 1:9 | [24] | n.s. | 10.0 | Automated Fixed pressure |
Yes | n.s., n.s. |
| Cornou et al. [35] | Bottles | Air incubator |
60 | 1:2 | [44] | 72 | 8.33 | Automated Fixed pressure |
Yes | Manual, no analysis |
| Muetzel et al. [36] | Bottles | Air incubator |
60 | 1:4 | [46,47] | 48 | 10.0 | Automated Automated |
Yes | Automated, CH4 by GC |
| Pellikaan et al. [18] | Bottles shaked |
Water bath |
60 | 1:2 | [44] | 72 | 8.33 | Automated Vented pressure?? |
Yes | Manual, CH4 by GC |
| Tagliapietra et al. [20] | Bottles | Air incubator |
75 | 1:2 | [44] | 144 | 6.67 | Automated Automated |
Yes | Manual, CH4 by GC |
| Braidot et al. [37] | Bottles stirred | Water bath |
500 | 1:2 | [44] | 48 | 6.67 | Automated Automated |
No, Volume base | Automated, CH4 - infrared |
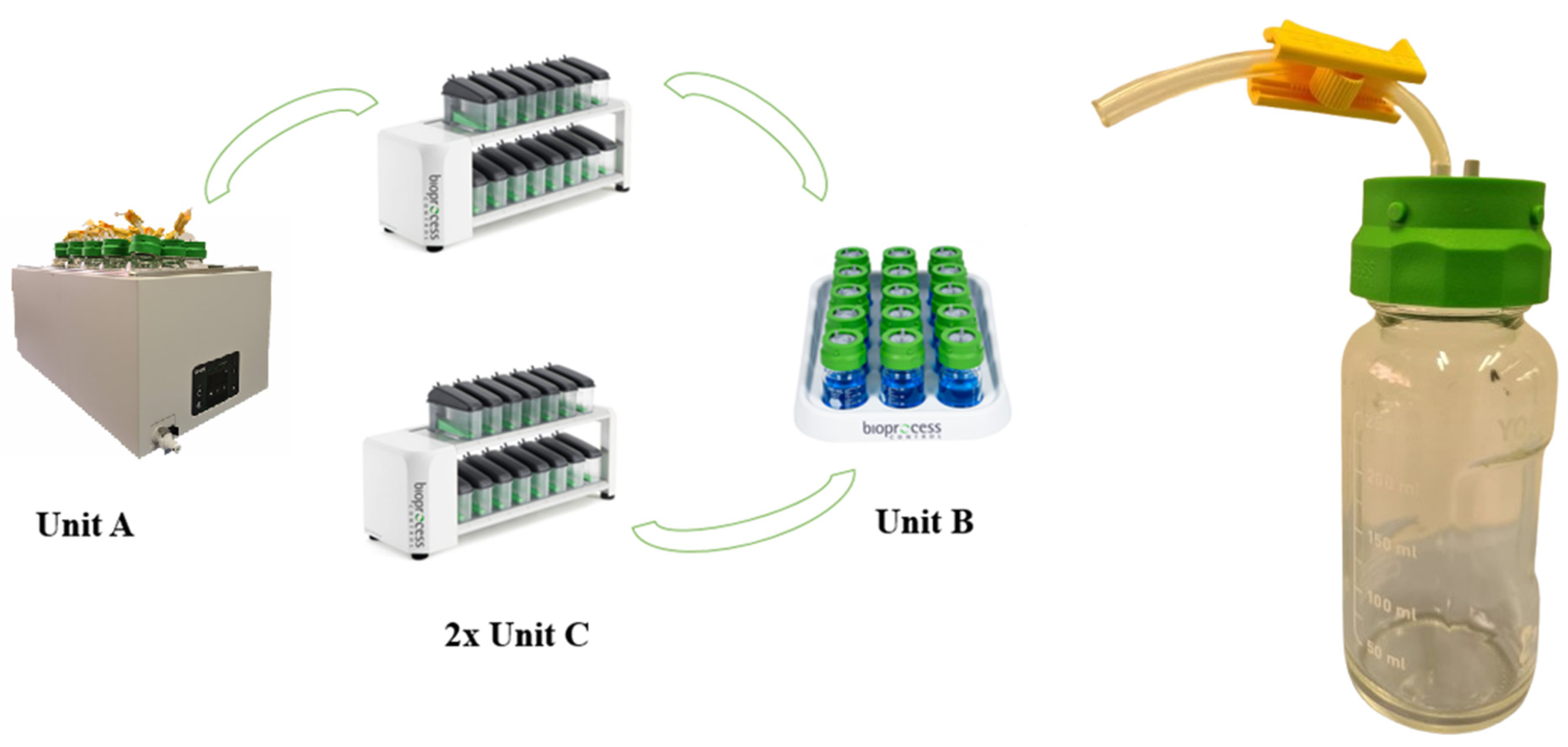
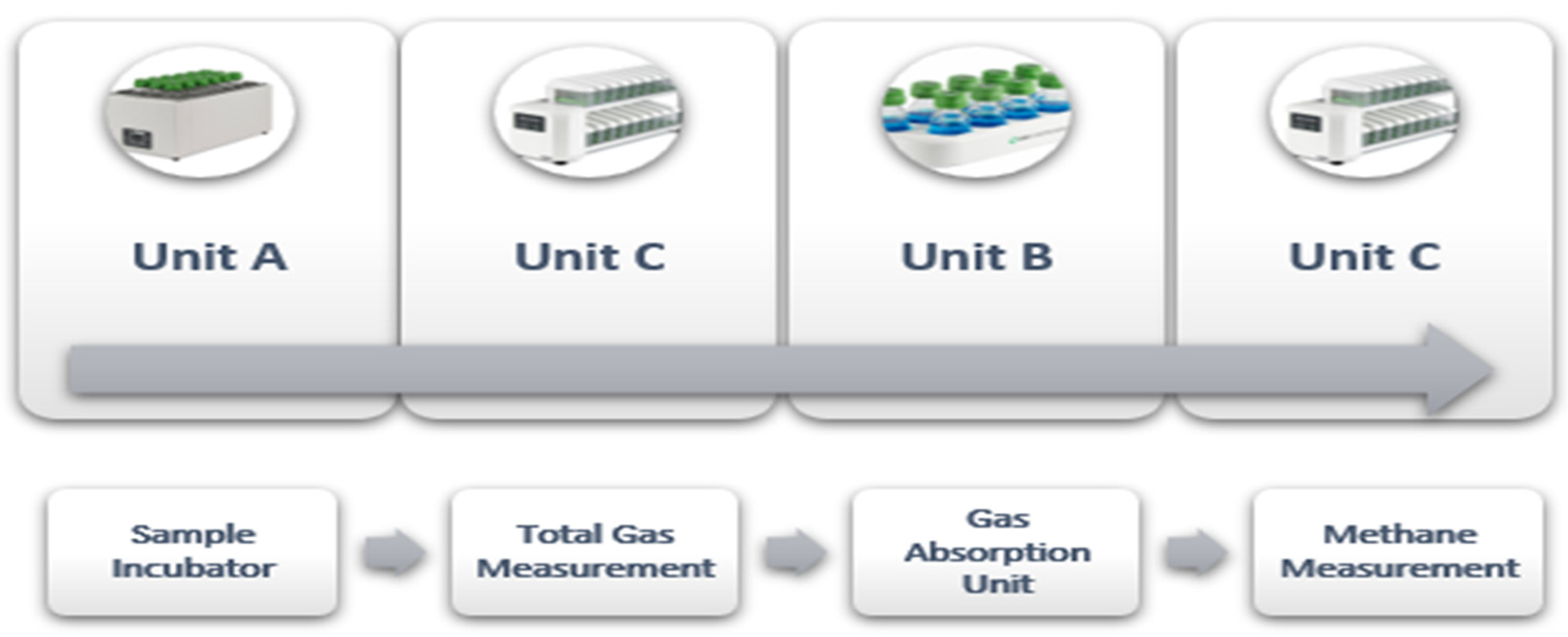
3. The Gas Endeavour System

3.1. Parts and Functionality
3.2. Area of Application of the Gas Endeavour System
3.2.1. Biomethane Potential
3.2.2. Animal Nutrition
3.3. Challenges and Considerations in Practical Use of GE
3.3.1. Buffer, Rumen Inoculum and Feed Substrate Ratio
3.3.2. Bubbling of CO₂
3.3.3. Effect of Shaking on Gas Production
3.3.4. Effect of Headspace Pressure on Gas Production
3.3.5. Effect of Headspace Pressure on Methane Production
3.3.6. Normalization of Gas and Methane Measurement for Temperature, Pressure and Vapor Interference
3.4. Gas Endevour Properties and Characteristics: Strengths and Weaknesses
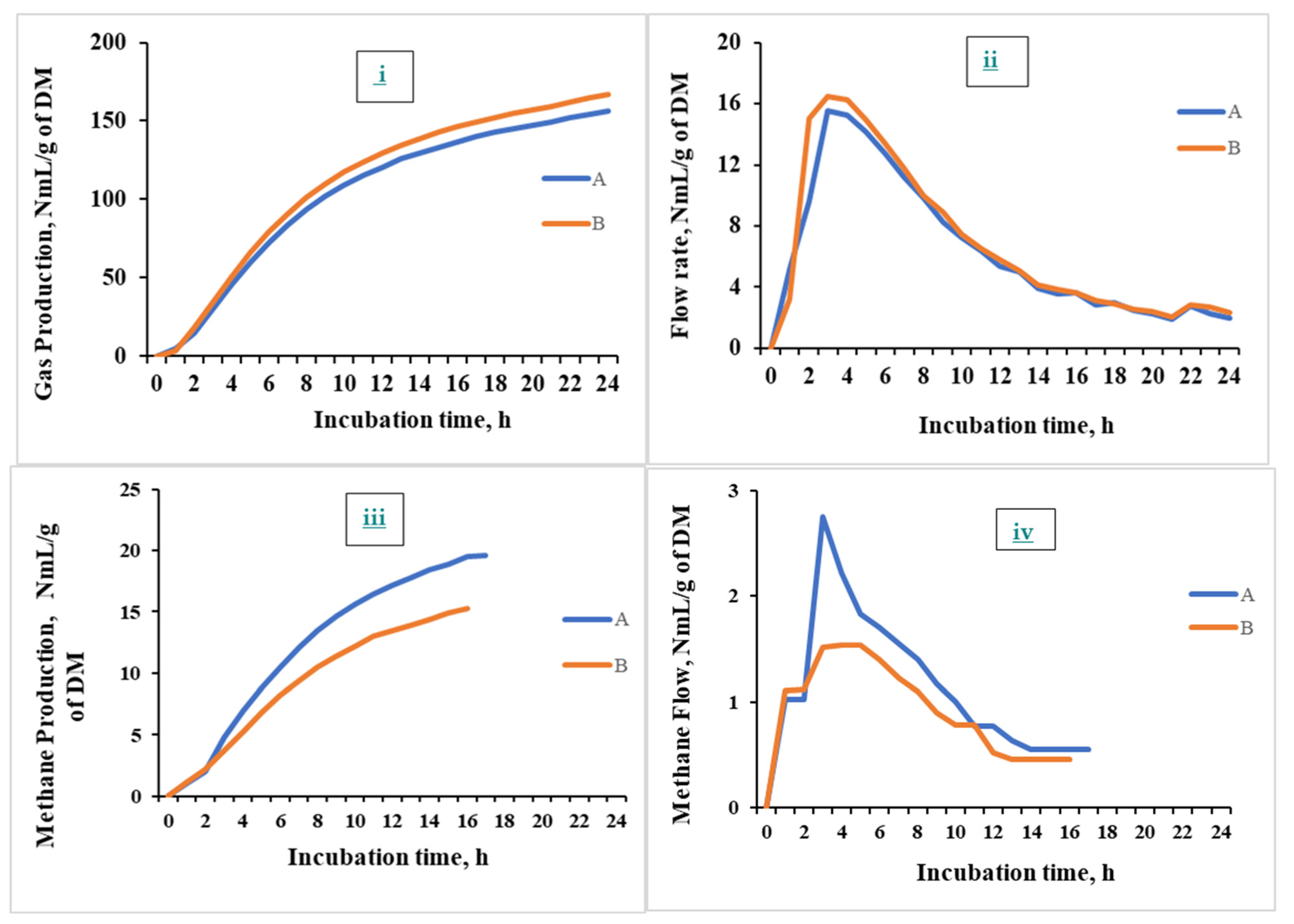
3.4.1. Gas and Methane Measurement
3.4.2. Gas and CH₄ Flow Rate and Kinetics
3.4.3. Real-Time Monitoring
3.4.5. Limitations
4. Potential Future Applications
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Food; Agriculture Organization of the United, N. FAOSTAT statistical database, FAO, 2024: 2024.
- Tubiello, F.N.; Salvatore, M.; Rossi, S.; Ferrara, A.; Fitton, N.; Smith, P. The FAOSTAT database of greenhouse gas emissions from agriculture. Environmental Research Letters 2013, 8, 015009. [Google Scholar] [CrossRef]
- Garnsworthy, P.C.; Craigon, J.; Hernandez-Medrano, J.H.; Saunders, N. On-farm methane measurements during milking correlate with total methane production by individual dairy cows. Journal of Dairy Science 2012, 95, 3166–3180. [Google Scholar] [CrossRef] [PubMed]
- Garnsworthy, P.C.; Craigon, J.; Hernandez-Medrano, J.H.; Saunders, N. Variation among individual dairy cows in methane measurements made on farm during milking. Journal of Dairy Science 2012, 95, 3181–3189. [Google Scholar] [CrossRef]
- Waghorn, G. Technical Manual on Respiration Chamber Designs. 2014.
- Storm, I.M.; Hellwing, A.L.F.; Nielsen, N.I.; Madsen, J. Methods for measuring and estimating methane emission from ruminants. Animals 2012, 2, 160–183. [Google Scholar] [CrossRef]
- Sejian, V.; Lal, R.; Lakritz, J.; Ezeji, T. Measurement and prediction of enteric methane emission. International journal of biometeorology 2011, 55, 1–16. [Google Scholar] [CrossRef]
- Johnson, K.; Huyler, M.; Westberg, H.; Lamb, B.; Zimmerman, P. Measurement of methane emissions from ruminant livestock using a sulfur hexafluoride tracer technique. Environmental science & technology 1994, 28, 359–362. [Google Scholar]
- Lassey, K.R. On the importance of background sampling in applications of the SF6 tracer technique to determine ruminant methane emissions. Animal feed science and technology 2013, 180, 115–120. [Google Scholar] [CrossRef]
- Zhao, Y.; Nan, X.; Yang, L.; Zheng, S.; Jiang, L.; Xiong, B. A review of enteric methane emission measurement techniques in ruminants. Animals 2020, 10, 1004. [Google Scholar] [CrossRef]
- Zimmerman, P.R.Z.S. Method and system for monitoring and reducing ruminant methane production. US830 7785B2, 2012.
- Place, S.E.; Pan, Y.; Zhao, Y.; Mitloehner, F.M. Construction and Operation of a Ventilated Hood System for Measuring Greenhouse Gas and Volatile Organic Compound Emissions from Cattle. Animals 2011, 1, 433–446. [Google Scholar] [CrossRef]
- Silveira, S.R.; Terry, S.A.; Biffin, T.E.; Maurício, R.M.; Pereira, L.G.R.; Ferreira, A.L.; Ribeiro, R.S.; Sacramento, J.P.; Tomich, T.R.; Machado, F.S. Replacement of soybean meal with soybean cake reduces methane emissions in dairy cows and an assessment of a face-mask technique for methane measurement. Frontiers in Veterinary Science 2019, 6, 295. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.K.; Hegarty, R.S.; Cowie, A. Meta-analysis quantifying the potential of dietary additives and rumen modifiers for methane mitigation in ruminant production systems. Animal Nutrition 2021, 7, 1219–1230. [Google Scholar] [CrossRef]
- Tilley, J.; Terry, D.R. A two-stage technique for the in vitro digestion of forage crops. Grass and forage science 1963, 18, 104–111. [Google Scholar] [CrossRef]
- Rymer, C.; Huntington, J.; Williams, B.; Givens, D. In vitro cumulative gas production techniques: History, methodological considerations and challenges. Animal Feed Science and Technology 2005, 123, 9–30. [Google Scholar] [CrossRef]
- Navarro-Villa, A.; O’brien, M.; López, S.; Boland, T.; O’kiely, P. Modifications of a gas production technique for assessing in vitro rumen methane production from feedstuffs. Animal feed science and technology 2011, 166, 163–174. [Google Scholar] [CrossRef]
- Pellikaan, W.; Hendriks, W.; Uwimana, G.; Bongers, L.; Becker, P.; Cone, J. A novel method to determine simultaneously methane production during in vitro gas production using fully automated equipment. Animal Feed Science and Technology 2011, 168, 196–205. [Google Scholar] [CrossRef]
- Menke, K.; Raab, L.; Salewski, A.; Steingass, H.; Fritz, D.; Schneider, W. The estimation of the digestibility and metabolizable energy content of ruminant feedingstuffs from the gas production when they are incubated with rumen liquor in vitro. The Journal of Agricultural Science 1979, 93, 217–222. [Google Scholar]
- Tagliapietra, F.; Cattani, M.; Bailoni, L.; Schiavon, S. In vitro rumen fermentation: effect of headspace pressure on the gas production kinetics of corn meal and meadow hay. Animal Feed Science and Technology 2010, 158, 197–201. [Google Scholar] [CrossRef]
- Blümmel, M.; Orskov, E. Comparison of in vitro gas production and nylon bag degradability of roughages in predicting of food intake in cattle. Animal Feed Science and Technology 1993, 40, 109–119. [Google Scholar]
- Makkar, H.; Blümmel, M.; Becker, K. Formation of complexes between polyvinyl pyrrolidones or polyethylene glycols and tannins, and their implication in gas production and true digestibility in in vitro techniques. British Journal of Nutrition 1995, 73, 897–913. [Google Scholar] [CrossRef]
- Pell, A.; Schofield, P. Computerized monitoring of gas production to measure forage digestion in vitro. Journal of dairy science 1993, 76, 1063–1073. [Google Scholar] [CrossRef]
- Theodorou, M.K.; Williams, B.A.; Dhanoa, M.S.; McAllan, A.B.; France, J. A simple gas production method using a pressure transducer to determine the fermentation kinetics of ruminant feeds. Animal feed science and technology 1994, 48, 185–197. [Google Scholar] [CrossRef]
- Mauricio, R.M.; Mould, F.L.; Dhanoa, M.S.; Owen, E.; Channa, K.S.; Theodorou, M.K. A semi-automated in vitro gas production technique for ruminant feedstuff evaluation. Animal Feed Science and Technology 1999, 79, 321–330. [Google Scholar] [CrossRef]
- Massaro, S.; Jantzen, B.; Axel, A.M.D.; Tagliapietra, F.; Hansen, H.H. Effect of Iodoform in Maize and Clover Grass Silages: An In Vitro Study. Ruminants 2024, 4, 418–432. [Google Scholar] [CrossRef]
- Pellikaan, W.F.; Hendriks, W.H.; Uwimana, G.; Bongers, L.J.G.M.; Becker, P.M.; Cone, J.W. A novel method to determine simultaneously methane production during in vitro gas production using fully automated equipment. Animal Feed Science and Technology 2011, 168, 196–205. [Google Scholar] [CrossRef]
- Cone, J.W.; van Gelder, A.H.; Visscher, G.J.; Oudshoorn, L. Influence of rumen fluid and substrate concentration on fermentation kinetics measured with a fully automated time related gas production apparatus. Animal Feed Science and Technology 1996, 61, 113–128. [Google Scholar]
- Ramin, M.; Huhtanen, P. Development of an in vitro method for determination of methane production kinetics using a fully automated in vitro gas system—A modelling approach. Animal Feed Science and Technology 2012, 174, 190–200. [Google Scholar] [CrossRef]
- Ankom. ANKOM RF Gas Production System. Available online: https://www.ankom.com/product-catalog/ankom-rf-gas-production-system (accessed on March 18).
- Jouany, J.; Lassalas, B. Gas pressure inside a rumen in vitro system stimulates the use of hydrogen. 2002.
- Hess, P.A.; Giraldo, P.; Williams, R.; Moate, P.; Beauchemin, K.; Eckard, R. A novel method for collecting gas produced from the in vitro ankom gas production system. Journal of Animal Science 2016, 94, 570–570. [Google Scholar] [CrossRef]
- Tunkala, B.Z.; DiGiacomo, K.; Hess, P.S.A.; Dunshea, F.R.; Leury, B.J. Rumen fluid preservation for in vitro gas production systems. Animal Feed Science and Technology 2022, 292, 115405. [Google Scholar] [CrossRef]
- Regadas Filho, J.; Tedeschi, L.; Fonseca, M.; Cavalcanti, L. 1629 (M343) Comparison of fermentation kinetics of four feedstuffs using an in vitro gas production system and the ANKOM gas production system.
- Cornou, C.; Storm, I.M.D.; Hindrichsen, I.K.; Worgan, H.; Bakewell, E.; Ruiz, D.R.Y.; Abecia, L.; Tagliapietra, F.; Cattani, M.; Ritz, C. A ring test of a wireless in vitro gas production system. Animal Production Science 2013, 53, 585–592. [Google Scholar]
- Muetzel, S.; Hunt, C.; Tavendale, M.H. A fully automated incubation system for the measurement of gas production and gas composition. Animal Feed Science and Technology 2014, 196, 1–11. [Google Scholar] [CrossRef]
- Braidot, M.; Sarnataro, C.; Romanzin, A.; Spanghero, M. A new equipment for continuous measurement of methane production in a batch in vitro rumen system. Journal of Animal Physiology and Animal Nutrition 2023, 107, 747–753. [Google Scholar] [PubMed]
- Mauricio, R.M.; Owen, E.; Mould, F.L.; Givens, I.; Theodorou, M.K.; France, J.; Davies, D.R.; Dhanoa, M.S. Comparison of bovine rumen liquor and bovine faeces as inoculum for an in vitro gas production technique for evaluating forages. Animal Feed Science and Technology 2001, 89, 33–48. [Google Scholar] [CrossRef]
- Mutimura, M.; Myambi, C.; Gahunga, P.; Mgheni, D.; Laswai, G.; Mtenga, L.; Gahakwa, D.; Kimambo, A.; Ebong, C. Rumen liquor from slaughtered cattle as a source of inoculum for in vitro gas production technique in forage evaluation. Agricultural Journal 2013, 8, 173–180. [Google Scholar]
- Stocker, T. Climate change 2013: the physical science basis: Working Group I contribution to the Fifth assessment report of the Intergovernmental Panel on Climate Change; Cambridge university press: 2014.
- Spanghero, M.; Chiaravalli, M.; Colombini, S.; Fabro, C.; Froldi, F.; Mason, F.; Moschini, M.; Sarnataro, C.; Schiavon, S.; Tagliapietra, F. Rumen Inoculum Collected from Cows at Slaughter or from a Continuous Fermenter and Preserved in Warm, Refrigerated, Chilled or Freeze-Dried Environments for In Vitro Tests. Animals 2019, 9, 815. [Google Scholar] [CrossRef]
- Yáñez-Ruiz, D.R.; Bannink, A.; Dijkstra, J.; Kebreab, E.; Morgavi, D.P.; O’Kiely, P.; Reynolds, C.K.; Schwarm, A.; Shingfield, K.J.; Yu, Z.; et al. Design, implementation and interpretation of in vitro batch culture experiments to assess enteric methane mitigation in ruminants—a review. Animal Feed Science and Technology 2016, 216, 1–18. [Google Scholar] [CrossRef]
- Goering, H.K.; Van Soest, P.J. Forage fiber analyses (apparatus, reagents, procedures, and some applications); US Agricultural Research Service: 1970.
- Menke, K.H. Estimation of the energetic feed value obtained from chemical analysis and in vitro gas production using rumen fluid. Anim Res Dev 1988, 28, 7–55. [Google Scholar]
- Davies, Z.; Mason, D.; Brooks, A.; Griffith, G.; Merry, R.; Theodorou, M. An automated system for measuring gas production from forages inoculated with rumen fluid and its use in determining the effect of enzymes on grass silage. Animal Feed Science and Technology 2000, 83, 205–221. [Google Scholar]
- Mould, F.; Kliem, K.; Morgan, R.; Mauricio, R. In vitro microbial inoculum: A review of its function and properties. Animal Feed Science and Technology 2005, 123, 31–50. [Google Scholar]
- Mould, F.; Morgan, R.; Kliem, K.; Krystallidou, E. A review and simplification of the in vitro incubation medium. Animal Feed Science and Technology 2005, 123, 155–172. [Google Scholar]
- Instruments, B. Gas Endeavour III. Available online: https://bpcinstruments.com/bpc_products/gas- endeavour-iii/ (accessed on.
- Liu, J.; van Gorp, R.; Nistor, M. The new Gas Endeavour system from Bioprocess Control AB for in vitro assessment of animal feeds. In Proceedings of the Proceedings of the 9th Nordic Feed Science Conference, Uppsala, Sweden, 2018; pp. 12-13.
- Üveges, Z.; Damak, M.; Klátyik, S.; Ramay, M.W.; Fekete, G.; Varga, Z.; Gyuricza, C.; Székács, A.; Aleksza, L. Biomethane Potential in Anaerobic Biodegradation of Commercial Bioplastic Materials. Fermentation 2023, 9, 261. [Google Scholar] [CrossRef]
- Świechowski, K.; Rasaq, W.A.; Syguła, E. Anaerobic digestion of brewer’s spent grain with biochars—biomethane production and digestate quality effects. Frontiers in Energy Research 2023, 11. [Google Scholar] [CrossRef]
- Wu, H.; Li, J.; Liao, Q.; Fu, Q.; Liu, Z. Enhanced biohydrogen and biomethane production from Chlorella sp. with hydrothermal treatment. Energy Conversion and Management 2020, 205, 112373. [Google Scholar] [CrossRef]
- Carrillo-Reyes, J.; Buitrón, G.; Moreno-Andrade, I.; Tapia-Rodríguez, A.C.; Palomo-Briones, R.; Razo-Flores, E.; Aguilar-Juárez, O.; Arreola-Vargas, J.; Bernet, N.; Braga, A.F.M.; et al. Standardized protocol for determination of biohydrogen potential. MethodsX 2020, 7, 100754. [Google Scholar] [CrossRef]
- Dolci, G.; Venturelli, V.; Catenacci, A.; Ciapponi, R.; Malpei, F.; Romano Turri, S.E.; Grosso, M. Evaluation of the anaerobic degradation of food waste collection bags made of paper or bioplastic. Journal of Environmental Management 2022, 305, 114331. [Google Scholar] [CrossRef]
- Fernandes, T.M.D.; de Almeida, J.F.M.; Escócio, V.A.; da Silva, A.L.N.; de Sousa, A.M.F.; Visconte, L.L.Y.; Furtado, C.R.G.; Pacheco, E.B.A.V.; Leite, M.C.A.M. Evaluation of rheological behavior, anaerobic and thermal degradation, and lifetime prediction of polylactide/poly(butylene adipate-co-terephthalate)/powdered nitrile rubber blends. Polymer Bulletin 2019, 76, 2899–2913. [Google Scholar] [CrossRef]
- Liu, J.; Strömberg, S.; Gorp, J.v.; Nistor, M. Importance of accurate and correct quantitative measurements in a new volumetric gas measuring technique for in vitro assessment of ruminant feeds. 2016.
- Elgemark, E. Intensively processed silage using Bio-extruder. 2019.
- Psenovschi, G.; Vintila, A.C.N.; Neamtu, C.; Vlaicu, A.; Capra, L.; Dumitru, M.; Enascuta, C.-E. Biogas Production Using “Gas Endeavour” Automatic Gas Flow System. Chemistry Proceedings 2023, 13, 17. [Google Scholar]
- Dębowski, M.; Kisielewska, M.; Kazimierowicz, J.; Zieliński, M. Methane Production from Confectionery Wastewater Treated in the Anaerobic Labyrinth-Flow Bioreactor. Energies 2023, 16, 571. [Google Scholar] [CrossRef]
- Dębowski, M.; Zieliński, M.; Kazimierowicz, J.; Walery, M. Aquatic Macrophyte Biomass Periodically Harvested Form Shipping Routes and Drainage Systems in a Selected Region of Poland as a Substrate for Biogas Production. Applied Sciences 2023, 13, 4184. [Google Scholar]
- Chan, S.; Nishi, K.; Koyama, M.; Toda, T.; Matsuyama, T.; Ida, J. Combined effects of various conductive materials and substrates on enhancing methane production performance. Biomass and Bioenergy 2023, 178, 106977. [Google Scholar] [CrossRef]
- Syguła, E.; Rasaq, W.A.; Świechowski, K. Effects of Iron, Lime, and Porous Ceramic Powder Additives on Methane Production from Brewer’s Spent Grain in the Anaerobic Digestion Process. Materials 2023, 16, 5245. [Google Scholar] [CrossRef]
- Szwarc, D.; Nowicka, A.; Zieliński, M. Comparison of the Effects of Pulsed Electric Field Disintegration and Ultrasound Treatment on the Efficiency of Biogas Production from Chicken Manure. Applied Sciences 2023, 13, 8154. [Google Scholar]
- Kleinheinz, G.; Hernandez, J. Comparison of two laboratory methods for the determination of biomethane potential of organic feedstocks. Journal of microbiological methods 2016, 130, 54–60. [Google Scholar] [PubMed]
- Nolan, P.; Luostarinen, S.; Doyle, E.; O'kiely, P. Anaerobic digestion of perennial ryegrass prepared by cryogenic freezing versus thermal drying methods, using contrasting in vitro batch digestion systems. Renewable energy 2016, 87, 273–278. [Google Scholar]
- Yang, H.; Rustas, B.; Eriksson, T. Rumen in vitro total gas production of timothy, red clover and the mixed silage after extrusion. 2018.
- Quarantelli, A.; Renzi, M.; Simoni, M.; Martuzzi, F.; Rosato, M.; Zambini, E.M.; Righi, F. Evaluation of an additive capable to improve ruminal fermentations through the use of an automated gas production system. ITALIAN JOURNAL OF ANIMAL SCIENCE 2019, 18, 30–31. [Google Scholar]
- Bertignon, M. Analisi di additivi in grado di migliorare le fermentazioni ruminali attraverso l’uso di un sistema automatizzato di gas production. Università di Parma. Dipartimento di Scienze Medico-Veterinarie, 2020.
- Vigh, A.; Criste, A.; Gragnic, K.; Moquet, L.; Gerard, C. Ruminal solubility and bioavailability of inorganic trace mineral sources and effects on fermentation activity measured in vitro. Agriculture 2023, 13, 879. [Google Scholar] [CrossRef]
- Muragijeyezu, P.C. Investigating the potential of different dietary fibers to stimulate butyrate production in vitro. 2020.
- Diamond, L.W.; Akinfiev, N.N. Solubility of CO2 in water from− 1.5 to 100 C and from 0.1 to 100 MPa: evaluation of literature data and thermodynamic modelling. Fluid phase equilibria 2003, 208, 265–290. [Google Scholar]
- Sun, Q.; Tian, H.; Li, Z.; Guo, X.; Liu, A.; Yang, L. Solubility of CO2 in water and NaCl solution in equilibrium with hydrate. Part I: Experimental measurement. Fluid Phase Equilibria 2016, 409, 131–135. [Google Scholar]
- Jantzen, B.; Hansen, H.H. Differences in Donor Animal Production Stage Affect Repeatability of In Vitro Rumen Fermentation Kinetics. Animals 2023, 13, 2993. [Google Scholar] [CrossRef]
- Wang, M.; Wang, R.; Tang, S.; Tan, Z.; Zhou, C.; Han, X.; Kang, J. Comparisons of manual and automated incubation systems: effects of venting procedures on in vitro ruminal fermentation. Livestock Science 2016, 184, 41–45. [Google Scholar]
- Lucile, F.; Cézac, P.; Contamine, F.; Serin, J.-P.; Houssin, D.; Arpentinier, P. Solubility of Carbon Dioxide in Water and Aqueous Solution Containing Sodium Hydroxide at Temperatures from (293.15 to 393.15) K and Pressure up to 5 MPa: Experimental Measurements. Journal of Chemical & Engineering Data 2012, 57, 784–789. [Google Scholar] [CrossRef]
- Tagliapietra, F.; Cattani, M.; Bailoni, L.; Schiavon, S. In vitro rumen fermentation: Effect of headspace pressure on the gas production kinetics of corn meal and meadow hay. Animal Feed Science and Technology 2010, 158, 197–201. [Google Scholar] [CrossRef]
- Carroll, J.J.; Slupsky, J.D.; Mather, A.E. The solubility of carbon dioxide in water at low pressure. Journal of Physical and Chemical Reference Data 1991, 20, 1201–1209. [Google Scholar] [CrossRef]
- Longo, C.; Bueno, I.C.S.; Nozella, E.F.; Goddoy, P.B.; Cabral Filho, S.L.S.; Abdalla, A.L. The influence of head-space and inoculum dilution on in vitro ruminal methane measurements. International Congress Series 2006, 1293, 62–65. [Google Scholar] [CrossRef]
- Patra, A.K.; Yu, Z. Effects of gas composition in headspace and bicarbonate concentrations in media on gas and methane production, degradability, and rumen fermentation using in vitro gas production techniques. Journal of Dairy Science 2013, 96, 4592–4600. [Google Scholar] [CrossRef] [PubMed]
- Instruments, B. Gas Endeavour Anaerobic Testing Handbook. 2024, 1.0. 1.0.
- Rosato, M.; Quarantelli, A. Comparing ruminal activity measures performed with Gas Endeavour with literature data. Congress of Animal Nutrition ASPA 2016 - Perugia-Italy, 2017. [Google Scholar]
- Baffa, D.F.; Oliveira, T.S.; Fernandes, A.M.; Camilo, M.G.; Silva, I.N.; Meirelles Júnior, J.R.; Aniceto, E.S. Evaluation of Associative Effects of In Vitro Gas Production and Fermentation Profile Caused by Variation in Ruminant Diet Constituents. Methane 2023, 2, 344–360. [Google Scholar] [CrossRef]
- Boland, T.M.; Pierce, K.M.; Kelly, A.K.; Kenny, D.A.; Lynch, M.B.; Waters, S.M.; Whelan, S.J.; McKay, Z.C. Feed Intake, Methane Emissions, Milk Production and Rumen Methanogen Populations of Grazing Dairy Cows Supplemented with Various C 18 Fatty Acid Sources. Animals : an open access journal from MDPI 2020, 10. [Google Scholar] [CrossRef]
- Maccarana, L.; Cattani, M.; Tagliapietra, F.; Bailoni, L.; Schiavon, S. Influence of main dietary chemical constituents on the in vitro gas and methane production in diets for dairy cows. Journal of animal science and biotechnology 2016, 7, 1–8. [Google Scholar] [CrossRef]
- Rota Graziosi, A.; Colombini, S.; Crovetto, G.M.; Galassi, G.; Chiaravalli, M.; Battelli, M.; Reginelli, D.; Petrera, F.; Rapetti, L. Partial replacement of soybean meal with soybean silage in lactating dairy cows diet: part 1, milk production, digestibility, and N balance. Italian Journal of Animal Science 2022, 21, 634–644. [Google Scholar]
- Xu, H.; Li, Y.; Hua, D.; Mu, H.; Zhao, Y.; Chen, G. Methane production from the anaerobic digestion of substrates from corn stover: Differences between the stem bark, stem pith, and leaves. The Science of the total environment 2019, 694, 133641. [Google Scholar] [CrossRef]
- Blümmel, M.; Lebzien, P. Predicting ruminal microbial efficiencies of dairy rations by in vitro techniques. Livestock production science 2001, 68, 107–117. [Google Scholar]
- Council, N.R. Nutrient Requirements of Dairy Cattle: Seventh Revised Edition, 2001; The National Academies Press: Washington, DC, 2001; p. 405. [Google Scholar]
- Amodeo, C.; Hafner, S.D.; Teixeira Franco, R.; Benbelkacem, H.; Moretti, P.; Bayard, R.; Buffière, P. How different are manometric, gravimetric, and automated volumetric BMP results? Water 2020, 12, 1839. [Google Scholar] [CrossRef]
- Blümmel, M.; Steingass, H.; Becker, K. The relationship between in vitro gas production, in vitro microbial biomass yield and 15N incorporation and its implications for the prediction of voluntary feed intake of roughages. The British journal of nutrition 1997, 77, 911–921. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




