Submitted:
20 February 2025
Posted:
21 February 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Graphitized Soot
2.2. Knudsen Flow Reactor
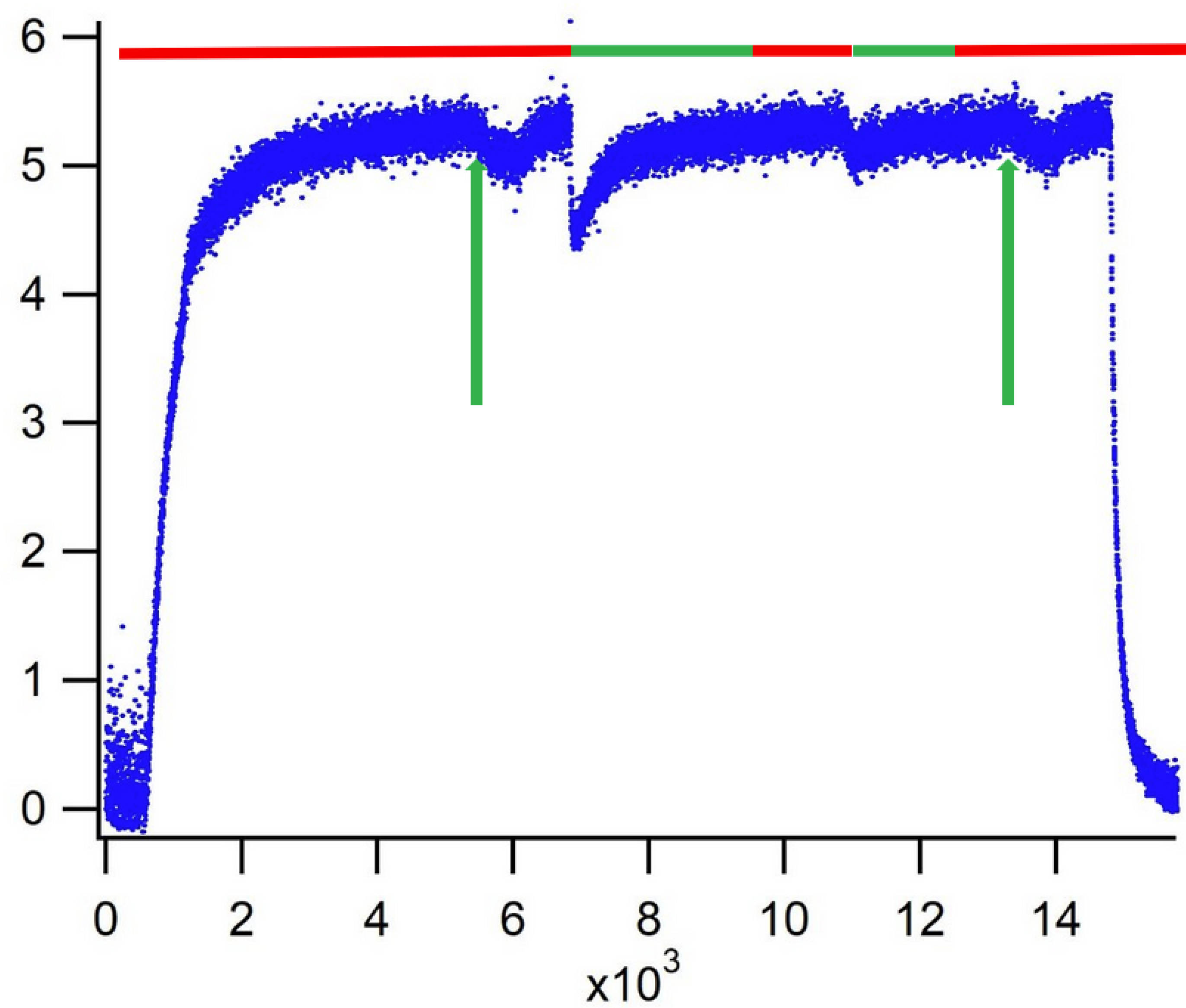
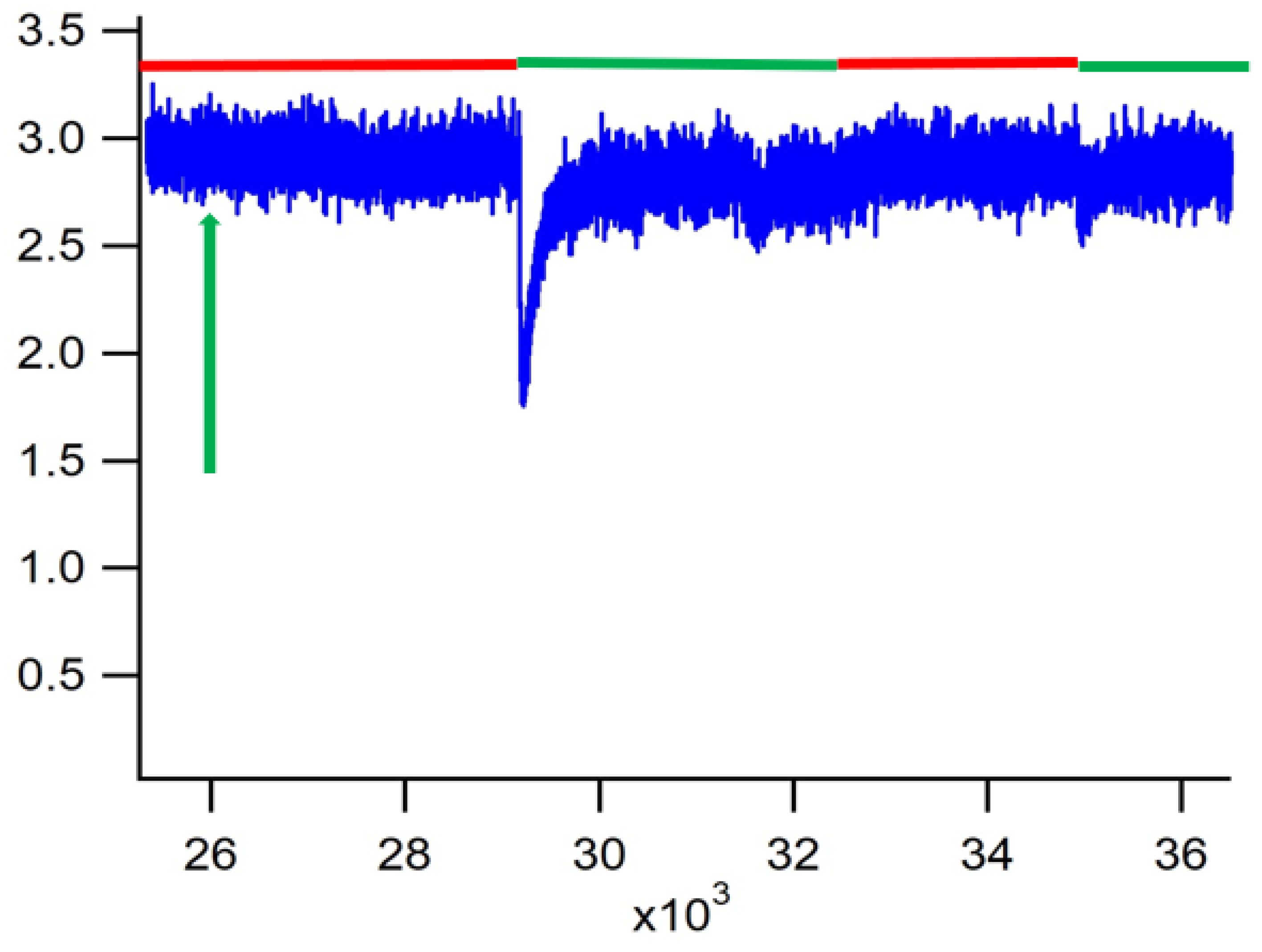
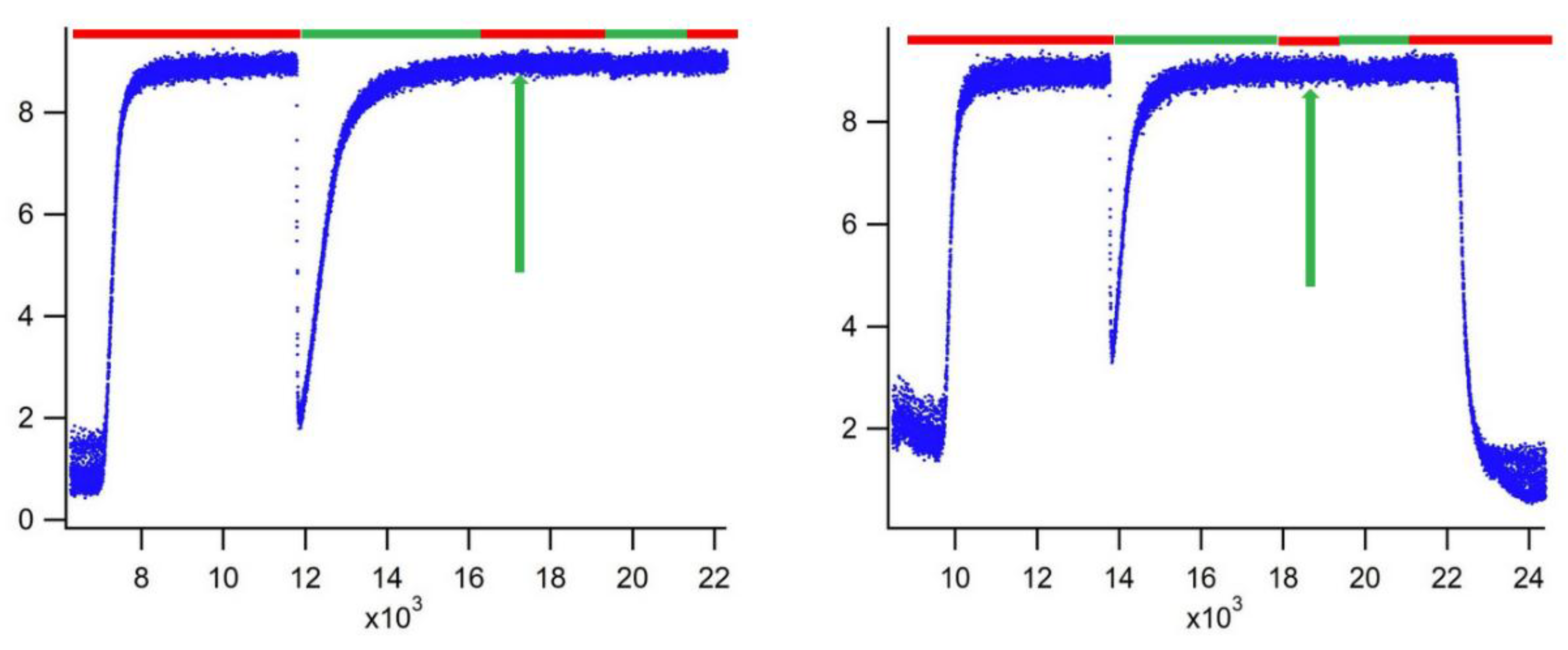
3. Results
4. Discussion
- -
- Considering the results of thermal amorphous carbon T900 and the HA probe we compare the entries for experiments s60#2 with s51#2 and come to the conclusion that previous ozone exposure increases the abundance of surface OH-groups at the interface by approximately 50%. We conclude that previous exposure to O3 irreversibly modifies the interface of T900.
- -
- For the case of BTCA-covered GTS80 probed by TMA we generally notice a “memory effect“ owing to exposure to NO2, TFA and HCl in that the uptake decreases upon exposure to these gases after probing with TMA. We take the average of runs s10#4 and s51#4 as the baseline for comparison, namely, an uptake of 9.2 1013 molecule.
- -
- Even for bare GTS80 we notice a “memory effect” when comparing run s20#3 with s21#3 owing to previous exposure to TFA.
- -
- Previous HA exposure of GTS6 and thermal carbon T900 samples enables marginal HCl uptake because adsorbed HA may act as a base as HA is a multifunctional molecule as seen in runs s71#1 and s61#2. The level of HCl uptake is larger than the “background level” of HCl uptake observed for bare GTS80, run s31#3 as seen in Table S2.
5. Conclusions
- -
- The sequence of abundance of the interfacial functional groups consisting of surface OH, reducing (oxidizable), basic and acidic surface sites is: GTS80 < GTS6 < Thermal Soot T900. High-temperature thermal processing (pyrolysis) under N2 significantly reduces surface functionality measured in sites cm-2 but does not eliminate altogether surface functionalities.
- -
- In going from T900 to bare GTS6 and GS80 through high-temperature processing (pyrolysis) one primarily loses the weakly reducing sites, the strongly reducing sites survive high-T pyrolysis much better than the weakly reducing sites.
- -
- Owing to the absence of HCl compared to TFA uptake we conclude that evidence points towards the exclusive presence of weak bases.
- -
- The comparison of O3/NO2 and HCl/TFA uptakes (the latter only for GTS80 + BTCA) leads to the conclusion that the interfacial reactivity is governed by strongly reducing groups. For the case of BTCA-covered GTS substrate we have in addition evidence for 86% weakly vs. 14% strongly basic surface functionalities.
- -
- The combination of strongly reducing power and weakly basic properties leads to the suggestion of the occurrence of pyrone bases coupled to hydroquinones, perhaps in a single highly conjugated aromatic hydrocarbon as presented in an earlier work making use of surface functionalities. See ESI section of reference [38] for structural formulas.
- -
- In many cases we may have to contend with irreversible memory effects upon exposure to probe gases, even when long-term rejuvenation through fast pumping is carried out. This effectively rules out multiple use of a single sample in the context of experimental investigations using several sequentially applied probe gases on the same sample.
Supplementary Materials
Funding
References
- IPCC, 2023: Summary for Policymakers. In: Climate Change 2023: Synthesis Report. Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team, H. Lee and J. Romero (eds.)]. IPCC, Geneva, Switzerland, pp. 1-34. [CrossRef]
- IPCC, 2023: Climate Change 2023: Synthesis Report. Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team, H. Lee and J. Romero (eds.)]. IPCC, Geneva, Switzerland, 184 pp.. [CrossRef]
- IPCC, 2001: Climate Change 2001: Synthesis Report. A Contribution of Working Groups I, II, and III to the Third Assessment Report of the Intergovernmental Panel on Climate Change [Watson, R.T. and the Core Writing Team (eds.)]. Cambridge University Press, Cambridge, United Kingdom, and New York, NY, USA, 398 pp. See pg. 48 and 156 for individual contributions of global average radiative forcing in W/m2.
- Petzold, A; Ogren, J.A.; Fiebig, M.; Laj, P.; Li, S.M.; Baltensperger, U.; Holzer-Popp, T.; Kinne,S.; Pappalardo, G.; Sugimoto N.; Wehrli, C.; Wiedensohler, A.; Zhang, X.Y; Recommendations for reporting“blackcarbon”measurements. Atmos. Chem. Phys. 2013, 13, 8365–8379.
- Bond, T.C. et al. Bounding the role of black carbon in the climate system: A scientific assessment. J. Geophys. Res. Atmos., 2013, 118(11), 5380-5552. [CrossRef]
- Jacobson, R.S.; Korte, A.R.; Vertes, A.; Miller, J.H. The molecular Composition of Soot. Angew. Chem. Int. Ed. 2020, 59, 4484-4490.
- Müller, J.-O.; Su, D.S.; Wild, U.; Schlögl, R. Bulk and surface structural investigations of Diesel engine soot and carbon black. Phys. Chem. Chem. Phys. 2007, 9, 4018-4025.
- Parent, P.; Laffon,C. ; Marhaba, I.; Ferry, D., Regier, T.Z.; Ortega, K.; Chazallon, B.; Carpentier, Y.; Focsa, C. Nanoscale characterization of aircraft soot: A high-resolution transmission electron microscopy, Raman spectroscopy, X-ray photoelectron and near-edge X-ray absorption spectroscopy study. Carbon, 2016, 101, 86-100.
- Kireeva, E.D. et al. Fractionation analysis of transport engine-generated soot particles with respect to hygroscopicity. J Atmos Chem 2009, 64, 129–147. [CrossRef]
- Popovicheva, O.B. et al. Small-Scale Study of Siberian Biomass Burning: I. Smoke Microstructure, Aerosol and Air Quality Research, 2015, 15, 117–128.
- Marhaba, I.; Ferry, D.; Laffon, C.; Regier, T.Z.; Ouf, F.-X ; Parent, P. Aircraft and MiniCAST soot at the nanoscale. Comb. and Flame, 2019, 204, 278-289.
- IPCC, 1999 – J.E. Penner, D.H. Lister, D.J. Griggs, D.J. Dokken, M. McFarland (Eds.) Prepared in collaboration with the Scientific Assessment Panel to the Montreal Protocol on Substances that Deplete the Ozone Layer Cambridge University Press, UK. pp 373 Available from Cambridge University Press, The Edinburgh Building Shaftesbury Road, Cambridge CB2 2RU ENGLAND.
- Lieske, L.-A.; Commodo, M.; Martin, J.W.; Kaiser, K.; Benekou, V.; Minutolo, P.; D’Anna, A.; Gross, L. Portraits of Soot Molecules Reveal Pathways to Large Aromatics, Five-/Seven-Membered Rings, and Inception through π-Radical Localization. ACS Nano 2023, 17, 13563−13574.
- Valavanidis, A.; Fiotakis, K.; Bakeas, E.; Vlahogianni, T. Electron paramagnetic resonance study of the generation of reactive oxygen species catalysed by transition metals and quinoid redox cycling by inhalable ambient particulate matter. Redox Reports, 2005, 10(1), 37-51.
- Li, M.; Bao, F.; Zhang, Y.; Song, W.; Chen, C.; Zhao, J. Role of elemental carbon in the photochemical aging of soot. PNAS (Proceedings of the National Academy of Sciences of the USA) 2018, 115, 7717-7722.
- Donnet, J.B. The Chemical Reactivity of Carbons. Carbon 1968, 6, 161-176.
- Papirer, E.; Li, S.; Donnet, J.-B. Contribution to the Study of Basic Surface Groups on Carbons. Carbon 1987, 25, 243-247.
- Boehm, H.P. Some Aspects of the Surface Chemistry of Carbon Blacks and other Carbons. Carbon, 1994, 32, 759-769.
- Boehm, H.P. Surface Oxides on Carbon and their Analysis: A critical Assessment. Carbon, 2002, 40, 145-149.
- Boehm, H.P. Acidic and Basic Properties of Hydroxylated Metal Oxide Surfaces. Discuss. Faraday Soc. 1971, 52, 264-275.
- Mirghaffary, N.; Iannarelli, R.; Ludwig, C., Rossi, M.J. Coexistence of reactive functional groups at the interface of a powdered activated amorphous carbon: A molecular view. Molecular Physics 2021, e1966110.
- Setyan, A.; Sauvain, J.-J.; Guillemin, M.; Riediker, M.; Demirdjian, B.; Rossi, M.J. Probing Functional Groups at the Gas-Aerosol Interface using Heterogeneous Titration Reactions: A Tool for Predicting Aerosol Health Effects? ChemPhysChem 2010, 11, 3823-3835.
- Setyan, A.; Sauvain, J.-J.; Rossi, M.J. The use of heterogeneous chemistry for the characterization of functional groups at the gas/particle interface of soot and TiO2 nanoparticles. Phys. Chem. Chem. Phys. 2009, 11, 6205-6217.
- Popovicheva, O.B.; Baumgardner, D.; Subramanian, R.; Kok, G.; Cary, R.; Vlasenko, E.; Khokhlova, T.; Shonija, N.; Kireeva, E. Tailored graphitized soot as reference material for EC/OC measurement validation. Atmos. Meas. Tech. 2011, 4, 923-932.
- Popovicheva, O.B.; Persiantseva, N.M.; Shonija, N.K.; DeMott, P.; Koehler, K.; Petters, M.; Kreidenweis, S.; Tishkova, V.; Demirdjian, B.; Suzanne, J. Water interaction with hydrophobic and hydrophilic soot particles. Phys. Chem. Chem. Phys. 2008, 10, 2332-2344.
- Popovicheva, O.B.; Kireeva, E.D.; Shonija, N.K.; Khokhlova, T.D. Water interaction with laboratory-simulated fossil fuel combustion particles. J. Phys. Chem. A 2009, 113, 10503-10511.
- Peri, J.B.; Hensley Jr., A.L. The Surface Structure of Silica Gel. J. Phys. Chem. 1966, 72, 2926-2933.
- Van Der Voort, P.; Gillis-D’Hamers, I.; Vrancken, K.C.; Vansant, E.F. Effect of Porosity on the Distribution and Reactivity of Hydroxy Groups on the Surface of Silica Gel. J. Chem. Soc. Faraday Trans. 1991, 87, 3899-3905.
- Griffith, P.R.; de Haseth, J.A. Fourier Transform Infrared Spectrometry, Vol. 83 in Chemical Analysis. A Series of Monography on Analytical Chemistry and its Applications, John Wiley & Sons, 1986, pg. 194.
- Tabor, K.; Gutzwiller, L.; Rossi, M.J. Heterogeneous Chemical Kinetics of NO2 on Amorphous Carbon at Ambient Temperature. J. Phys. Chem. 1994, 98, 6172-6186.
- Stadler, D.; Rossi, M.J. The reactivity of NO2 and HONO on Flame soot at ambient temperature: The influence of combustion conditions. Phys. Chem. Chem. Phys. 2000, 2, 5420-5429.
- Tapia, A.; Salgado, M.S.; Martin, M.P.; Lapuerta, M.; Rodriquez-Fernandez, J.; Rossi, M.J.; Cabanas, B. Molecular Characterization of the Gas-Particle Interface of Soot Sampled from a Diesel Engine Using a Titration Method. Environ. Sci. Technol. 2016, 50, 2946-2955.
- Maters, E.C.; Delmelle, P.; Rossi, M.J.; Ayris, P.M. Reactive Uptake of Sulfur Dioxide and Ozone on Volcanic Glass and Ash at Ambient Temperature. J. Geophys. Res.: Atmospheres, 2017, 122, 10077-10088.
- Borgmeyer, T.; Zhou, L.; Breider, F.; Rossi, M.J.; Ludwig, C. Natural and simulated weathering of polystyrene: A molecular view. Sci. Tot. Environ. 2024, 947, 174609.
- Borgmeyer, T.; Kupper, Y.; Rossi, M.J.; Luterbacher, J.; Ludwig, C. Characterization of Biobased Polymers at the Gas-Solid Interface – Analysis of Surface and Bulk Properties during Artificial Degradation. Environ. Sci. Technol. submitted (Nov. 2024).
- Rossi, M.J.; Gandolfo, A.; Lostier, A.; Roose, A.; George, C.; Salameh, T.; Thévenet, F.; Chen, H.; Romanias, M.N. Uptake of Atmospheric Pollutants on Road Asphalt Pavements: An Underestimated Sink in Urban Environments. Environ. Sci. Technol. 2024, submitted (Oct. 2024).
- Avgul, N. N; Kiselev, A. V. Physical Adsorption of Gases and Vapours on Graphitized Carbon Blacks, Chemistry and Physics of Carbons, edited by Walker, P. L., 6, Dekker, New York, 1–124, 1970.
- Iannarelli, R.; Ludwig, C.; Rossi, M.J. Flowing Gas Experiments Reveal Mechanistic Details of Interfacial Reactions on a Molecular Level at Knudsen Flow Conditions. Front. Astron. Space Sci. 2022, 9, 891177.
- Iannarelli, R.; Ludwig, C.; Rossi, M.J. The Kinetics of Adsorption and Desorption of Selected Semivolatile Hydrocarbons and H2O vapor on Two Mineral Dust Materials: A Molecular View. J. Phys. Chem. A 2022, 126, 8711-8726.
- Zelenay, V.; Monge, M.E.; D’Anna, B.; George, C.; Styler, S.A.; Huthwelker, T.; Ammann, M. Increased steady-state uptake of ozone on soot due to UV/Vis radiation. J. Geophys. Res. 2011, 116, D1130141.
- George, C.; Ammann, M.; D’Anna, B.; Donaldson. J.; Nizkorodov, S.A. Heterogeneous Photochemistry in the Atmosphere. Chem. Rev. 2015, 115, 4218-4258.
- Zogka, A.G.; Lostier, A.; Papadimitriou, V.C.; Thevenet, F.; Formenti, P.; Rossi, M.J.; Chen, H.; Romanias, M.N. Unraveling the Uptake of Glyoxal on a Diversity of Natural Dusts and Surrogates: Linking Dust Composition to Glyoxal Uptake and Estimation of Atmospheric Lifetimes. ACS Earth Space Chem. 2024, 8, 1165−1178.
- Diebold, U. Structure and properties of TiO2 surfaces: a brief review. Appl. Phys. A 2003, 76, 681–687.
| SAMPLES → ↓ Probe Gases |
(#1) GTS6 Graphitized Thermal Soot (5.6 ± 0.3 m2/g) |
(#2) T900 Thermal Soot-commercial (6.5 ± 0.3 m2/g) |
(#3) GTS80 Graphitized Thermal Soot (79 ± 2 m2/g) | (#4) GTS80 + 4.88% (wt) BTCAh (79 ± 2 m2/g) |
|---|---|---|---|---|
| N(CH3)3 (TMA) a | 5.5 1011 1.5 10-3 |
None - |
8.8 1011 2.4 10-3 |
9.2 1013 0.52 |
| NH2OH (HA) b | 1.4 1013 7.8 10-2 |
2.9 1013 3.7 10-2 |
4.0 1012 5.1 10-3 |
2.9 1013 16.4 10-2 |
| HCl c | None - |
None - |
None - |
7.7 1011 4.4 10-3 |
| CF3COOH (TFA) d | 3.5 1012 8.9 10-3 |
1.3 1013 3.3 10-2 |
6.5 1012 1.6 10-2 |
5.8 1012 3.3 10-2 |
| NO2 e | 1.4 1013 1.9 10-2 |
2.1 1013 3.0 10-2 |
2.4 1012 4.7 10-3 |
1.1 1013 6.3 10-2 |
| O3 f | 7.3 1013 9.9 10-2 |
3.4 1014 0.46 |
6.6 1012 8.9 10-3 |
1.5 1013 8.5 10-2 |
| SAMPLES → ↓ Probe Gases |
(#1) GTS6 Graphitized Thermal Soot (5.6 ± 0.3 m2/g) |
(#2) T900 Thermal Soot Commercial (6.5 ± 0.3 m2/g) |
(#3) GTS80 Graphitized Thermal Soot (79 ± 2 m2/g) | (#4) GTS80 + 4.88% wt BTCA (79 ± 2 m2/g) |
|---|---|---|---|---|
| NO2 | 3.4 10-5 (s50#1) |
2.5 10-4 (s10#2; s40#2) |
- | 1.7 10-4 (s50#4) |
| O3 | 8.7 10-5 (s60#1) |
1.2 10-3 (s50#2) |
6.8 10-4 (s70#3) |
7.4 10-4 (s60#4) |
| NH2OH (HA) | 1.3 10-4 (s70#1) |
1.6 10-4 (s60#2) |
2.1 10-5 b,d (s50#3) |
8.1 10-5 b (s40#4) |
| CF3COOH (TFA) | - | 3.5 10-4 b (s30#2) |
- | - |
| N(CH3)3 (TMA) |
- | - | - | 2.9 10-4 c (s10#4) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




