Submitted:
01 February 2025
Posted:
20 February 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Methodology
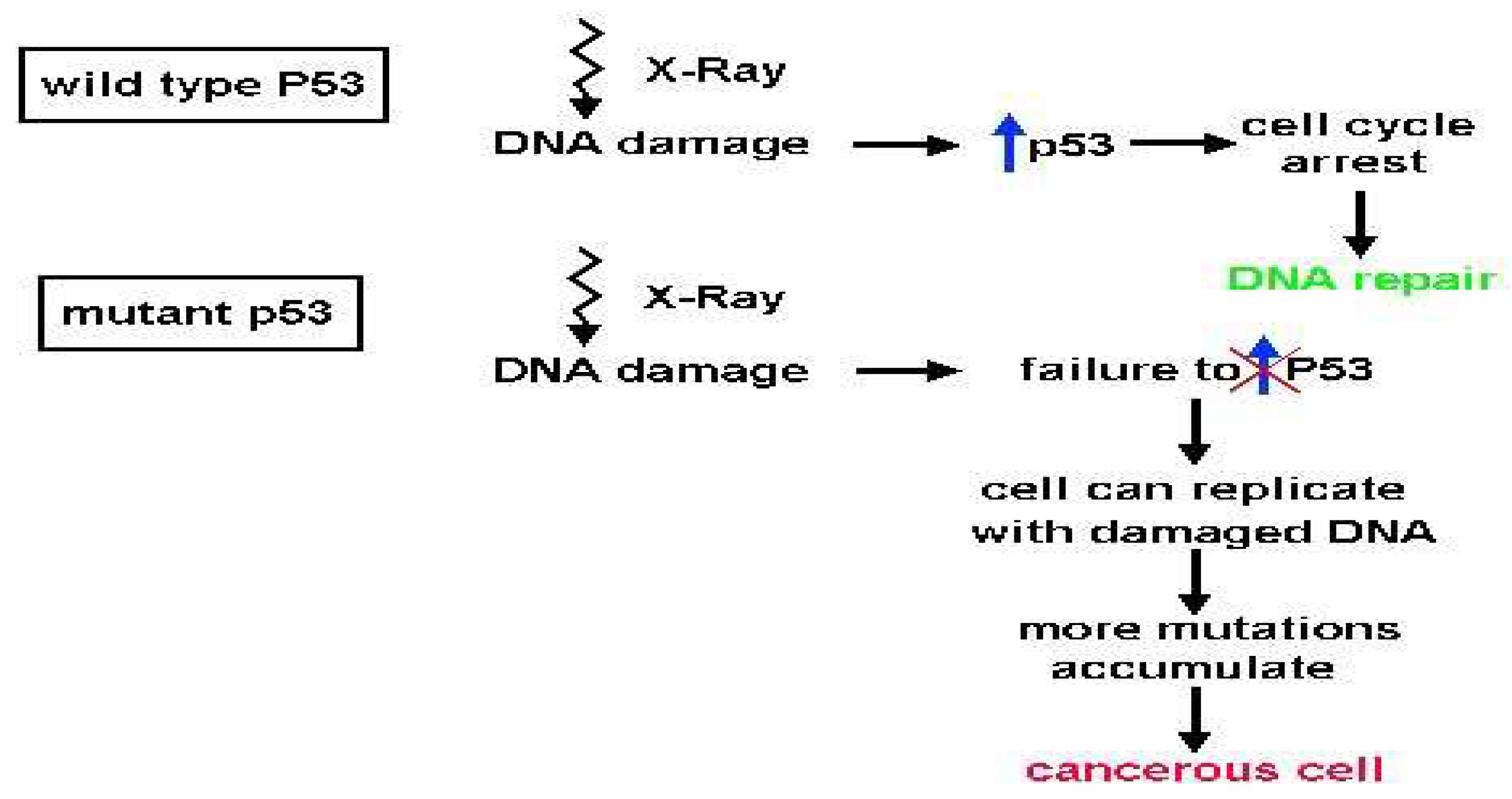
Results
Discussions
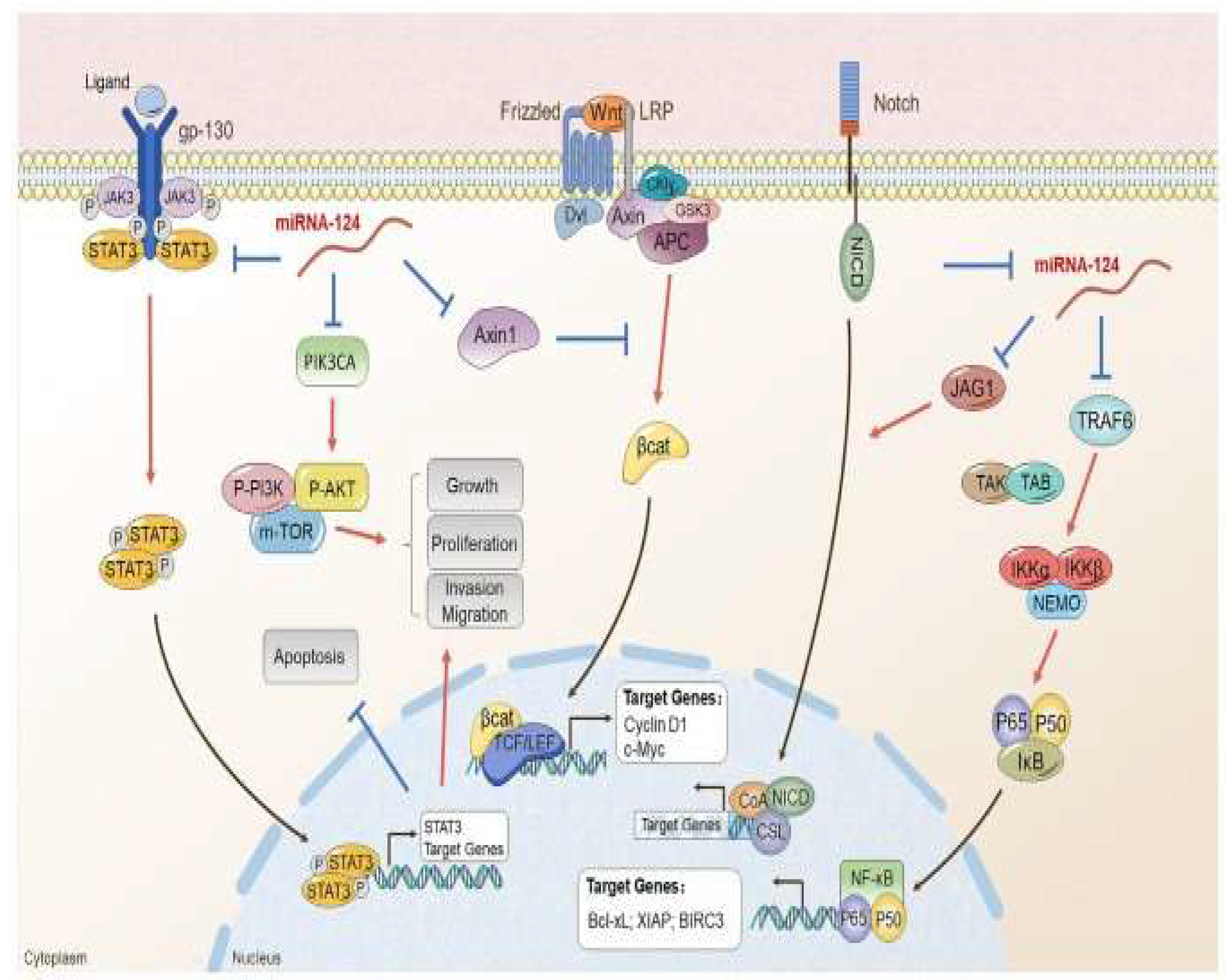
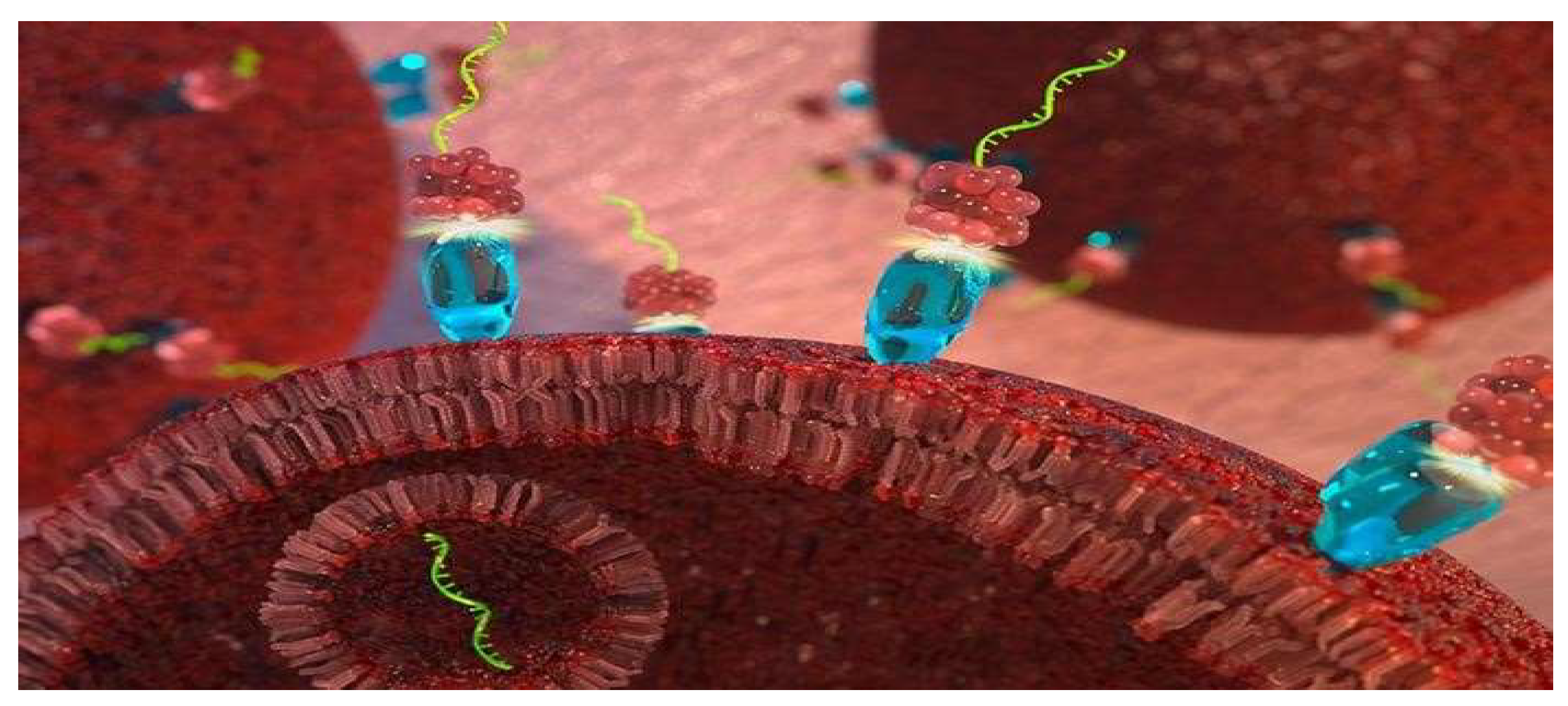
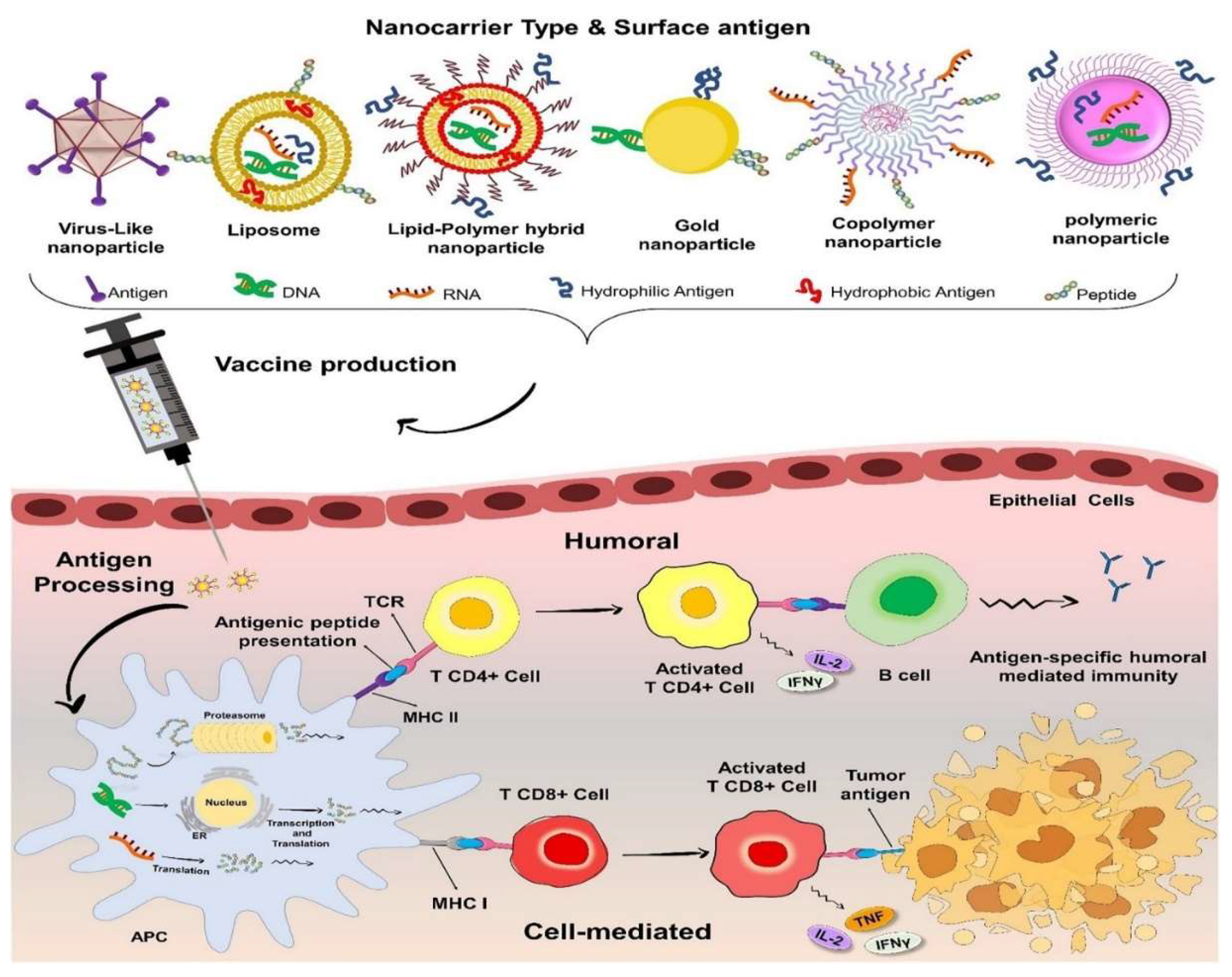
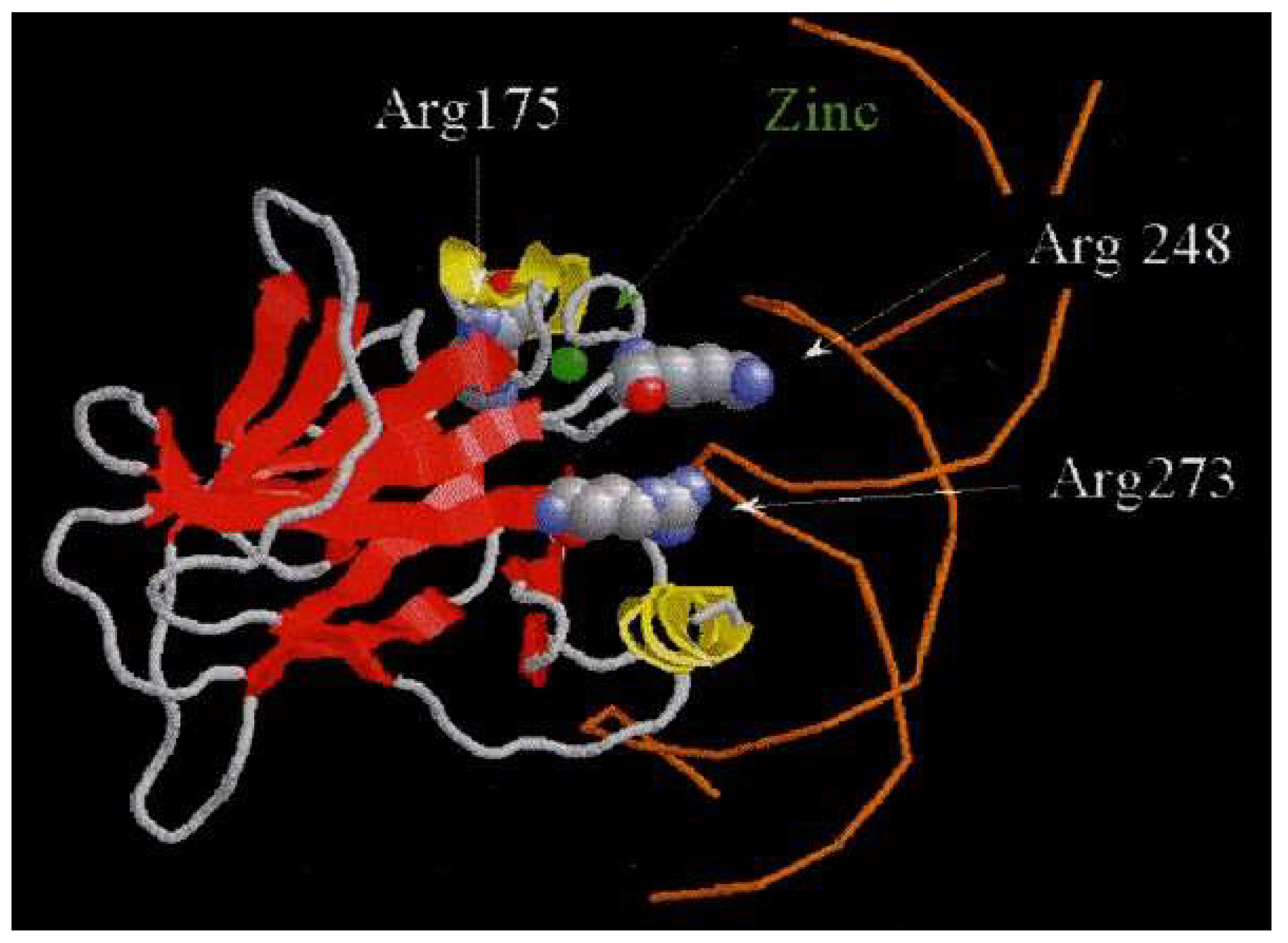
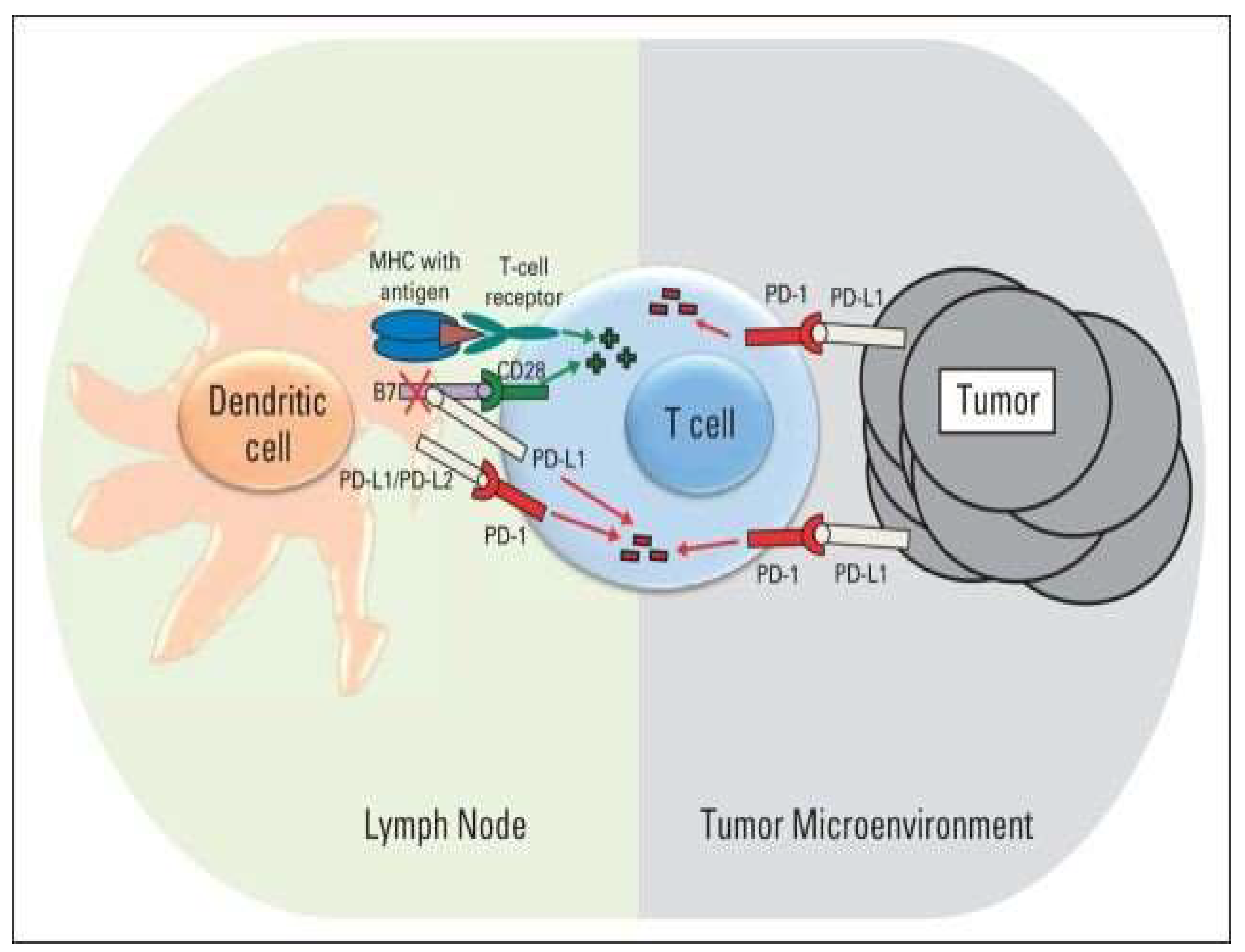
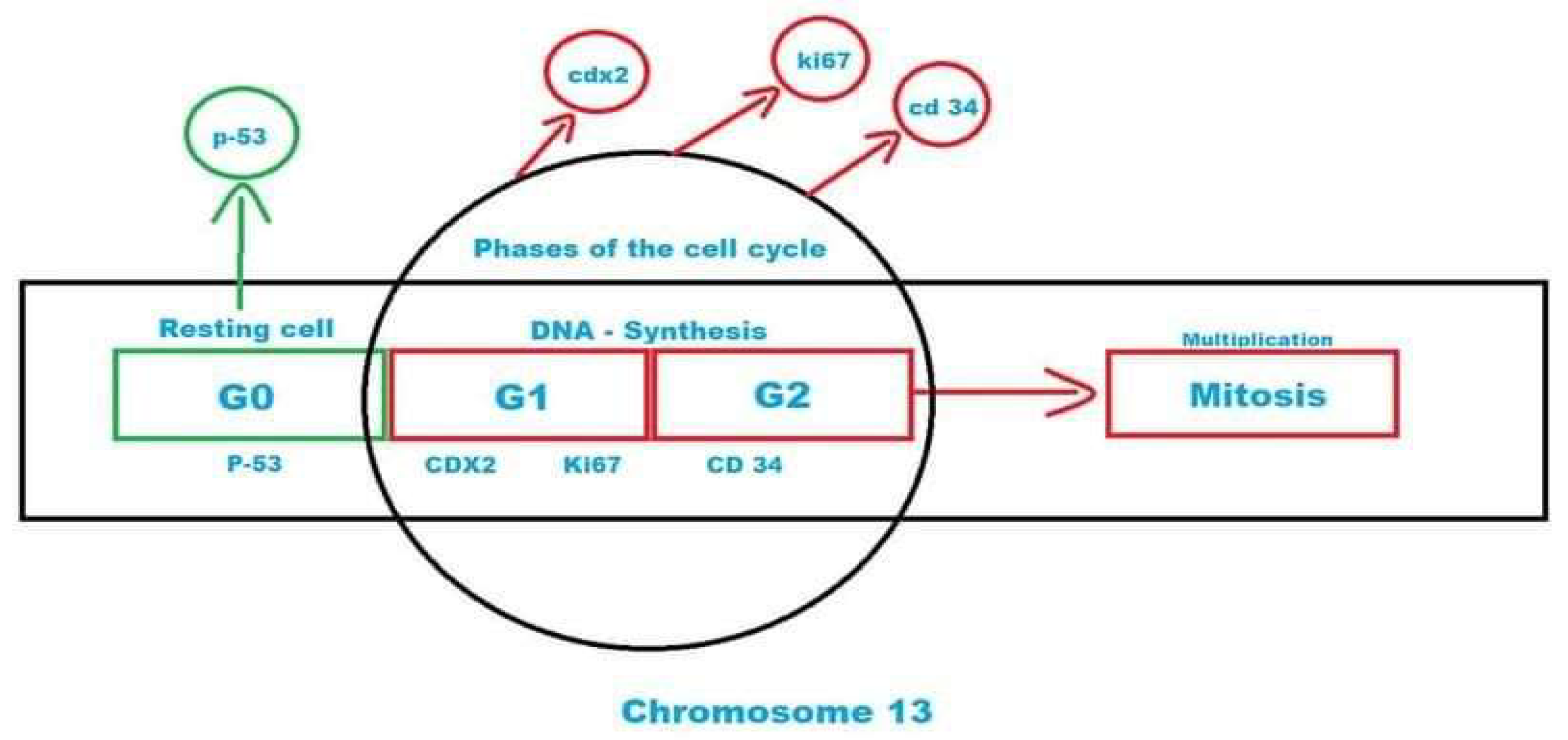
5. Conclusions
References
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424.
- Arnold, M. , Morgan, E., Rumgay, H., Mafra, A., Singh, D. et al. Current and future burden of breast cancer: Global statistics for 2020 and 2040. The Breast 2022; 66: 15–23.
- Dai X, Xiang L, Li T, Bai Z. Cancer Hallmarks, Biomarkers and Breast Cancer Molecular Subtypes. J Cancer. 2016;7(10):1281-1294. [CrossRef]
- Zaimy MA, Saffarzadeh N, Mohammadi A, Pourghadamyari H, Izadi P, Sarli A, Moghaddam LK, Paschepari SR, Azizi H, Torkamandi S, Tavakkoly-Bazzaz J. New methods in the diagnosis of cancer and gene therapy of cancer based on nanoparticles. Cancer Gene Ther. 2017; 24:233–243.
- Hung SK, Yang HJ, Lee MS. et al. Molecular subtypes of breast cancer predicting clinical benefits of radiotherapy after breast-conserving surgery: a propensity-score-matched cohort study. Breast Cancer Res 2023; (25); 149. [CrossRef]
- Dogra A, Mehta A, Doval DC. Are Basal-Like and Non-Basal-Like Triple-Negative Breast Cancers Really Different? J Oncol. 2020:4061063. [CrossRef]
- 7 Ismail A, El-Mahdy HA, Abuldsoud A et al. Beneficial and detrimental aspects of miRNAs as chief players in breast cancer: A comprehensive review. International Journal of Biological Macromolecules 2023; 224: 1541-1565.
- Si W, Shen J, Zheng H. et al. The role and mechanisms of action of microRNAs in cancer drug resistance. Clin Epigenetics. 2019; 11; 25. [CrossRef]
- Li JP, Zhang XM, Zhang Z, Zheng LH, Jindal S, Liu YJ. Association of p53 expression with poor prognosis in patients with triple-negative breast invasive ductal carcinoma. Medicine (Baltimore). 2019;98(18): e15449.
- Liu Y, Yang Y, Wang X, Yin S, Liang B, Zhang Y, Fan M, Fu Z, Shen C, Han Y, Chen B, Zhang Q. Function of microRNA-124 in the pathogenesis of cancer (Review). Int J Oncol. 2024 Jan;64(1):6. [CrossRef]
- Si, W. , Shen, J., Zheng, H. et al. The role and mechanisms of action of microRNAs in cancer drug resistance. Clin Epigenetics. 2019; 11; 25. [CrossRef]
- Amin MMJ, Trevelyan CJ, Turner NA. MicroRNA-214 in Health and Disease. Cells. 2021;10(12):3274. [CrossRef]
- Liu Y, Yang Y, Wang X, Yin S, Liang B, Zhang Y, Fan M, Fu Z, Shen C, Han Y, Chen B, Zhang Q. Function of microRNA-124 in the pathogenesis of cancer (Review). Int J Oncol. 2024;64(1):6. [CrossRef]
- Sarah, DG. Early Data for a Novel MiRNA Therapy Yield Promising Results. Oncology Times 2023; 45(23):p 17; http://doi/10.1097/01.COT.0000997480.29005.09.
- 15.. H, Torkamandi S, Tavakkoly-Bazzaz J. New methods in the diagnosis of cancer and gene therapy of cancer-based on nanoparticles. Cancer Gene Ther. 2017; 24:233–243.
- Vineis P, Wild CP. Global cancer patterns: Causes and prevention. Lancet. 2014; 383:549–557.
- Varna M, Bousquet G, Plassa LF, Bertheau P, Janin A. TP53 status and response to treatment in breast cancers. J Biomed Biotechnol.2011:284584. [CrossRef]
- Fatemeh-Davodabadi et al, Breast cancer vaccines: New insights into immunomodulatory and nano-therapeutic approach. Journal of Controlled Release. 2022; 349(4). https://www.researchgate.net/.
- Jia X, Wang X, Guo X, Ji J, Lou G, Zhao J, Zhou W, Guo M, Zhang M, Li C, Tai S, Yu S. MicroRNA-124: An emerging therapeutic target in cancer. Cancer Med. 2019 Sep;8(12):5638-5650. http://doi/10.1002/cam4.2489.
- Sarah, DG. Early Data for a Novel MiRNA Therapy Yield Promising Results. Oncology Times. 2023; 45(23): p 17; http://doi/10.1097/01.
- Sahar MA, Paish M, Powe DG et al. Tumor-Infiltrating CD8÷ Lymphocytes Predict Clinical Outcome in Breast Cancer Journal of Clinical Oncology 2011; (29)15. [CrossRef]
- Phillips KA, Nichol K, Ozcelik H, Knight J, Done SJ, Goodwin PJ et al. Frequency of p53 Mutations in Breast Carcinomas from Ashkenazi Jewish Carriers of BRCA1 Mutations, JNCI: Journal of the National Cancer Institute. 1999; (91)5;3: 69–473. [CrossRef]
- Jabir FA, Hoidy WH. No Evaluation of Serum P53 Levels in Iraqi Female Breast Cancer Patients. Asian Pac J Cancer Prev. 2017;18(9):2551-2553. doi. [CrossRef]
- Guptaa A, Shahb K, Ozac MJ, Behld T. Reactivation of the p53 gene by MDM2 inhibitors: A novel therapy for cancer treatment. Biomedicine & Pharmacotherapy. 2019; 109: 484–94.
- Pereira JA, Lanzar Z, Clark JT, et al. PD-1 and CTLA-4 exert additive control of effector regulatory T cells at homeostasis. Front. Immunol. 2023; 14: 20-23. [CrossRef]
- Paukena KE, Torchiac JA, Chaudhric A, Sharpea AH, Freeman GJ. Emerging concepts in PD-1 checkpoint biology. Seminars in Immunology. 2021: 101480. In Press,. [CrossRef]
- Ghosh C, Luong G, Sun Y. A snapshot of the PD-1/PD-L1 pathway. J Cancer. 2021;12(9):2735-46. [CrossRef]
- Torsin LI, Petrescu GED, Sabo AA, Chen B, Brehar FM, Dragomir MP et al. Editing and Chemical Modifications on Non-Coding RNAs in Cancer: A New Tale with Clinical Significance. Int. J. Mol. Sci. 2021; 22: 581. [CrossRef]
- Park M, Kim D, Ko S, Kim A, Mo K, Yoon H. Breast Cancer Metastasis: Mechanisms and Therapeutic Implications. Int J Mol Sci. 2022;23(12):6806. [CrossRef]
- Patanaphan V, Salazar OM, Risco R. Breast cancer: metastatic patterns and their prognosis. South Med J. 1988; 81(9):1109-1112.
- Romero Q, Bendahl PO, Fernö M, et al. A novel model for Ki67 assessment in breast cancer. Diagn Pathol. 2014; 9: 118. [CrossRef]
- Sobecki M, Mrouj K, Camasses A, et al. The cell proliferation antigen Ki-67 organises heterochromatin. Elife. 2016;5: e13722. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




