1. Introduction
Hydrogen plays a key role in the transition to a more sustainable energy model. Hydrogen is the fuel with the highest energy content per unit mass (120 MJ/kg), and its utilization releases water and oxygen into the atmosphere [
1]. However, due to its low volumetric density under ambient conditions (0.0824 kg/m
3), which is one-third that of the natural gas [
2], it is not competitive with fossil fuels without altering its state during the transport and storage phase.
High-pressure compression is currently the most developed and widely used method for storing and transporting hydrogen in metallic or composite cylindrical vessels at pressures up to 700 bar [
1]. However, it requires significant energy, consuming approximately 13–18% of hydrogen's lower heating value [
1]. Alternatively, liquefaction increases volumetric density to 71 kg/m³ at -253 °C and atmospheric pressure [
3], but it requires 30–40% of hydrogen’s lower heating value [
1]. These extremely low temperatures and high pressures represent significant challenges to large-scale commercialization. Therefore, developing an alternative that enables hydrogen transportation under milder and safer liquid-phase conditions, similar to traditional fuels, is crucial to gain public trust and attracting both private and public investment in the hydrogen supply chain [
4].
In recent times, liquid organic hydrogen carriers (LOHCs) have attracted considerable attention as a solution for transporting and storing hydrogen because of their favourable attributes:
a) LOHCs are oil-like organic system that can remain in liquid form under ambient conditions, so they are easy and safe to store and transport. This also facilitates the gradual integration of LOHC systems into existing petroleum-based industries, allowing the utilization of their existing infrastructure in a hydrogen-based economy [
5].
b) The concept of LOHC is based on the reversible hydrogenation and dehydrogenation of carbon double bonds through well-known catalytic reactions [
5].
c) LOHCs have been shown to have a high hydrogen storage capacity, ranging up to 7.3 wt
H2% [
6].
Hydrogen reversibly binds to LOHCs through a catalytic hydrogenation process, an exothermic reaction that occurs at moderate temperatures (100–300°C) and pressures (30–50 bar) [
5,
7,
8]. The hydrogenated LOHC exhibit a significant increase in volumetric energy density compared to molecular hydrogen and can be stored under ambient conditions for long periods without energy losses [
9]. In contrast, dehydrogenation is an endothermic process that typically occurs at atmospheric pressure and high temperatures (200-400°C) depending on the LOHC-catalyst combination [
8,
9]. The heat needed for dehydrogenation can be either supplied internally, through partial combustion of the released hydrogen, or externally from available waste heat sources or fuel combustion [
5]. In addition, if the two reactors are close together, it may be possible to use the heat produced by the exothermic reactor to power part of the endothermic one.
Over the years, various compounds have been studied, each with distinct chemical properties and significantly different transport and storage performances [
5]. Research has predominantly focused on compounds derived from hydrocarbons commonly used in petrochemicals [
5]. According to Niermann et al. [
9], an ideal LOHC should have the following characteristics: high boiling point, low melting point, high hydrogen storage capacity, wide availability, cost-effectiveness, low dehydrogenation temperature, environmental and human safety, stability, and high technology readiness level (TRL). Many aromatic hydrocarbons, such as benzene, toluene, naphthalene and dibenzyl-toluene can be used as LOHCs due to the interaction of aromatic rings, which makes them more prone to hydrogenation and dehydrogenation than other organic molecules [
7]. However, some of the aromatic compounds are carcinogenic or flammable while others are solid at room temperature [
10]. Generally, the hydrogen storage capacities of cyclic hydrocarbons are between 6-8 wt
H2 %, and the heat required by dehydrogenation is significant (62-71 kJ/mol) [
10]. In addition to cyclic aromatic hydrocarbons, N-heterocyclic base compounds [
11], such as N-ethyl carbazole, chinolina, indolo, phenazine, 1,2-Dihydro-1,2-Azaborine, have attracted much attention in LOHC research. The incorporation of heteroatoms such as N, P and O into aromatics significantly influence the thermodynamics of dehydrogenation [
7]. As shown by Clot et al. [
12], the substitution of heteroatoms at specific ring positions can effectively lower the dehydrogenation temperature of LOHCs and considerably reduce the enthalpy of reaction [
7,
12], resulting in thermodynamic conditions to fully release hydrogen at low temperatures [
13]. However, such compounds suffer from low TRL, high toxicity and complicated hydrogenation/dehydrogenation mechanics, which limits their scalability at large scale [
14].
Among all the possible aromatic candidates, dibenzyl-toluene (DBT) / perhydro-dibenzyl-toluene (PDBT) and toluene (TOL) / methylcyclohexane (MCH) systems are the most promising candidates for large-scale applications. TOL/MCH benefits from a low cost of 0.3 €/kg and a high mature TRL of 8, making it attractive for large scale applications, particularly in Asia, where pilot projects in Japan and Southeast Asia have demonstrated its viability [
15].
Conversely, DBT/PDBT has gained increasing attention in Europe, driven its non-flammability, low toxicity (13.8 TPI/mg), and low vapor pressure, which facilitate hydrogen separation. Its cost, currently ranging between 2-4 €/kg, suggests feasibility for large-scale deployment [
6,
8,
16]. The German company Hydrogenious GmbH has pioneered DBT-PDBT commercialization in Europe, reaching a TRL of 9 and engaging in multiple European hydrogen projects [
17]. Given the regional market distinction—TOL/MCH in Asia and DBT/PDBT in Europe—this study focuses on the techno-economic evaluation of the DBT/PDBT system for hydrogen transportation, aligning with the growing European interest in this LOHC technology.
1.1. Objective of the Work
In literature, numerous studies explore the use of LOHCs for transporting hydrogen over long distances by ship from countries rich in renewable energy to those with potentially high future hydrogen demand [
18,
19]. However, limited research has focused on the use of LOHCs for short-distance transport via road. Hurskainen et al. [
20] compared hydrogen road delivery costs via the DBT/PDBT concept and compressed gas. They found that the LOHC concept is more advantageous at higher hydrogen demands, particularly over longer transport distances. However, the analysis focuses on DBT/PDBT without modeling the hydrogenation and dehydrogenation reactors. Additionally, the liquid hydrogen option is not considered.
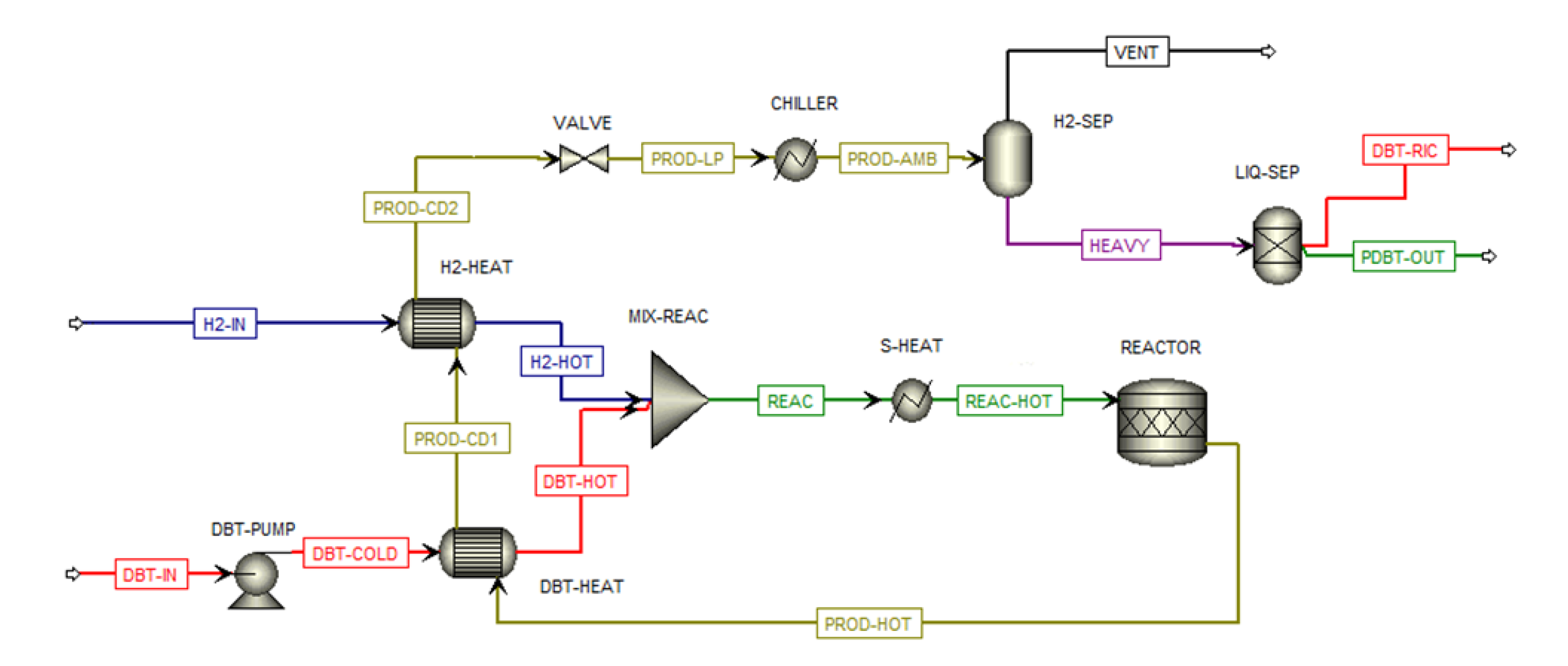
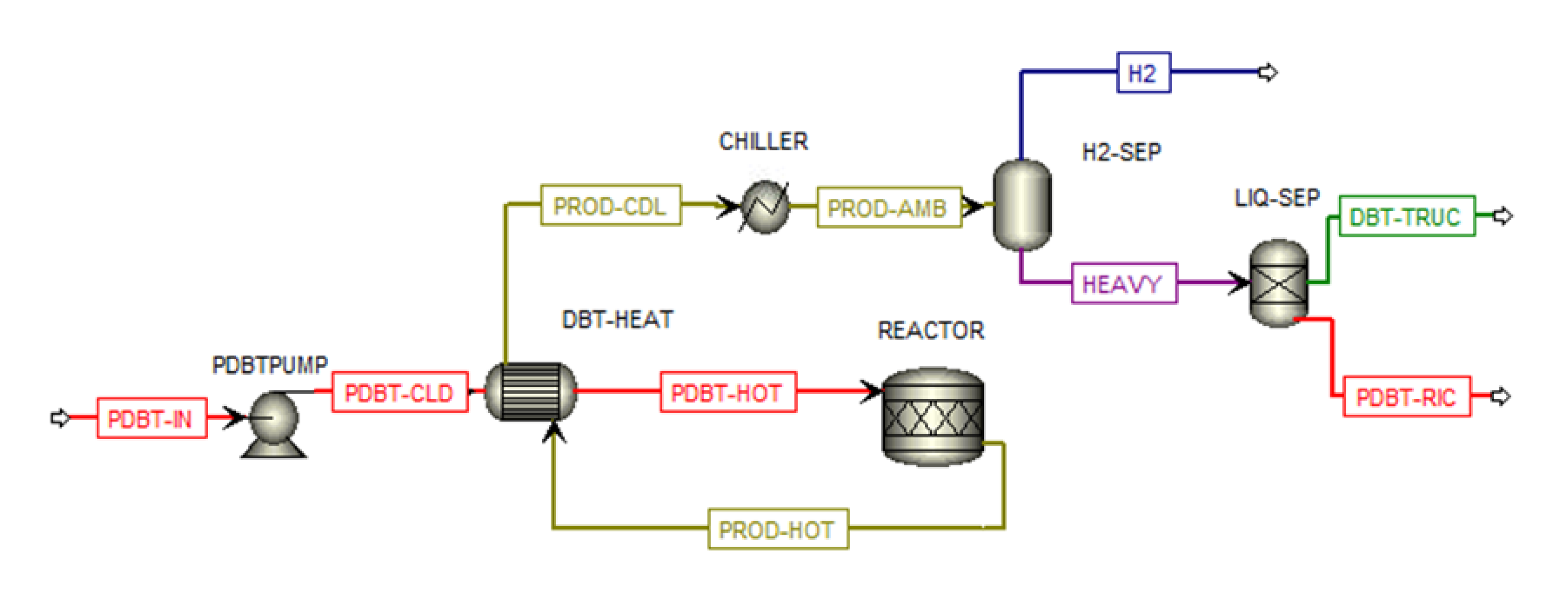
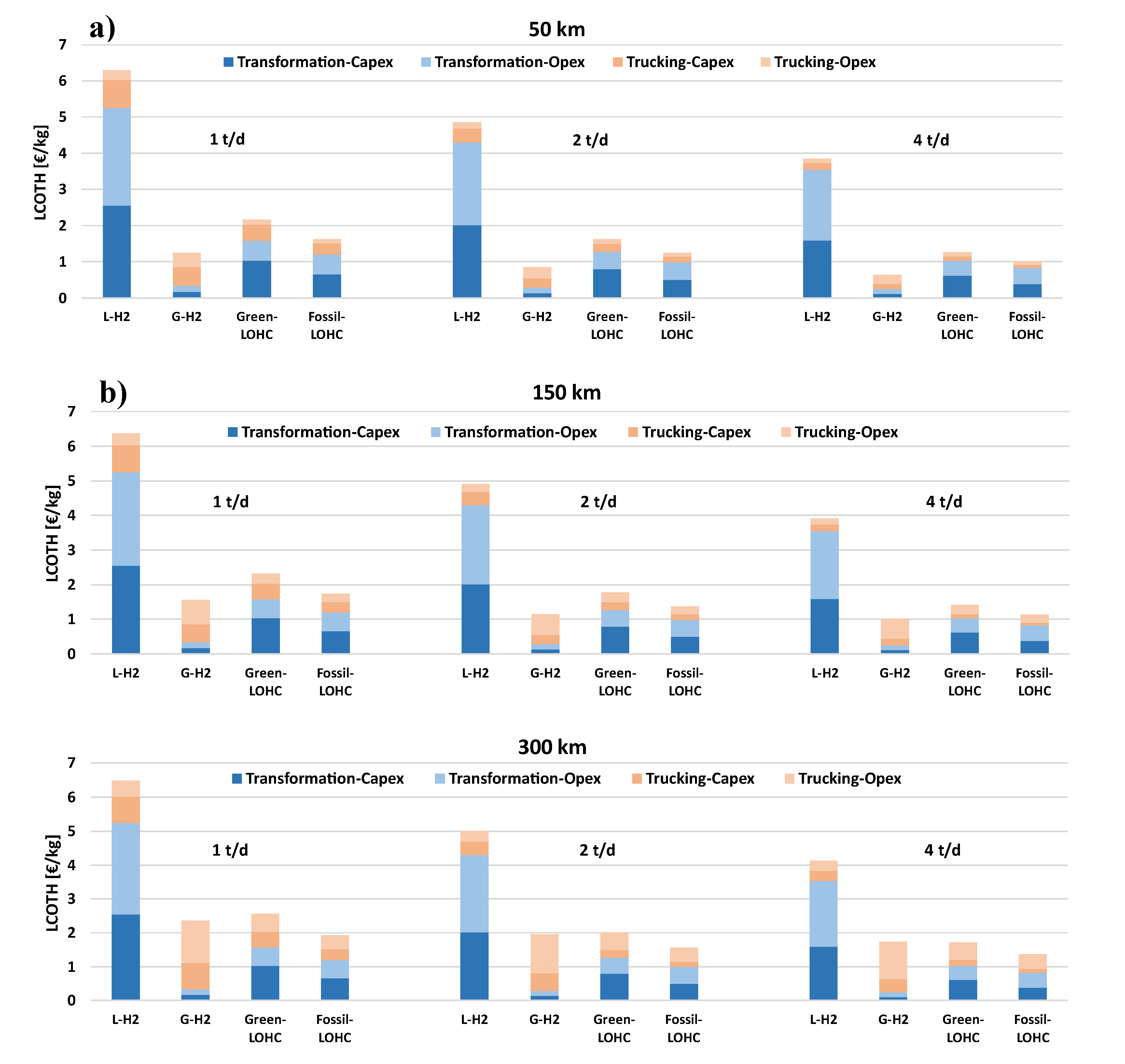
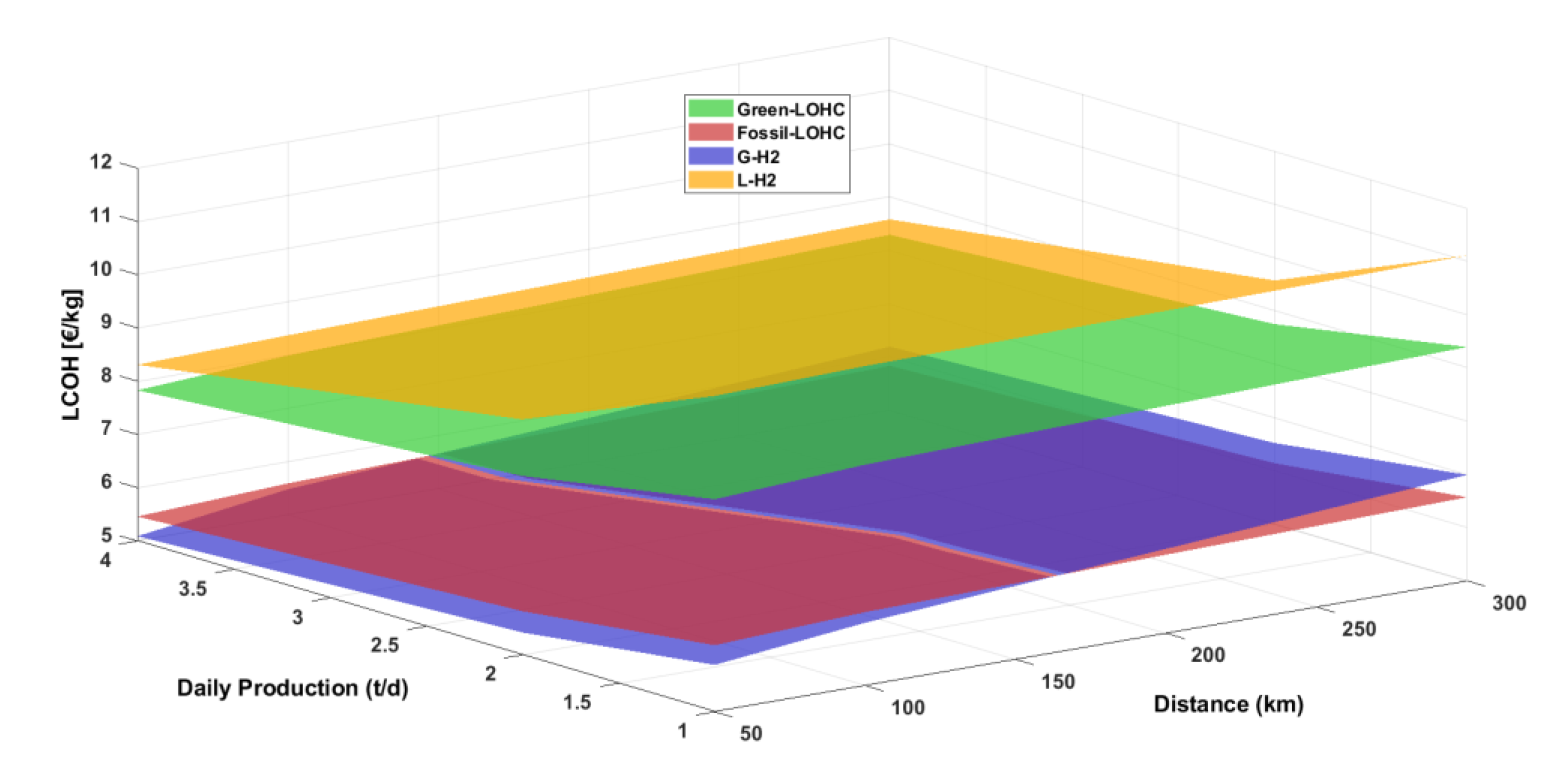
Therefore, this study presents a techno-economic analysis of hydrogen transportation via LOHCs by road, also simulating hydrogenation and dehydrogenation plants for DBT/PDBT systems in ASPEN Plus. The results are compared with hydrogen transported by using 350-bar compressed and cryogenic vessel tanks, so aiming at having a whole overview of all storage systems. Moreover, a sensitivity analysis evaluates the feasibility of transporting 1 t/d, 2 t/d, and 4 t/d of hydrogen over distances of 50, 150, and 300 km, from the production/hydrogenation site to the dehydrogenation/use point, focusing on the levelized cost of hydrogen (LCOH) and its cost components, particularly the levelized cost of transported hydrogen (LCOTH).
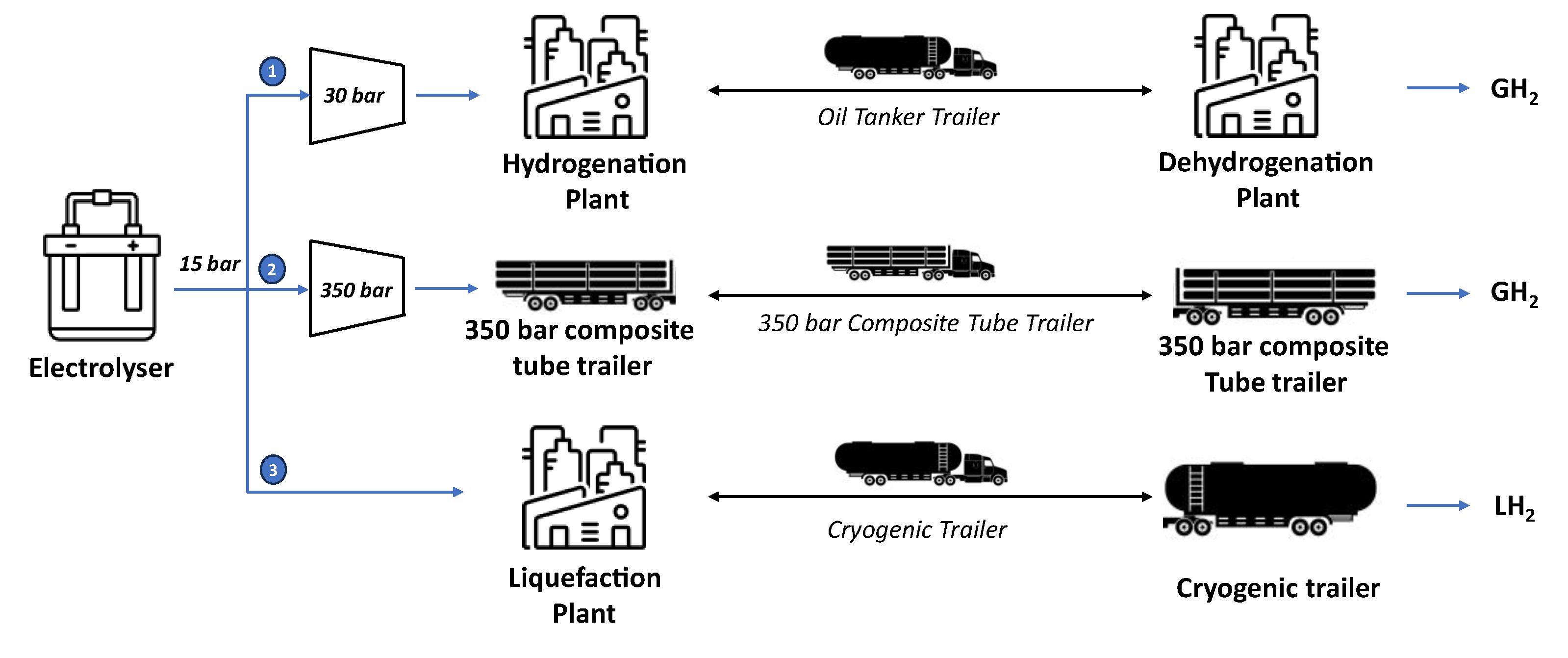
Figure 1 illustrates the three hydrogen transportation options considered in the analysis, in each case, hydrogen is produced by an alkaline electrolyser (AEL) at 15 bars. In the first pathway, hydrogen reacts with the LOHC in the hydrogenation unit after a compression step up to 30 bars. The hydrogenated product is then loaded into an oil tank trailer, transported by road, and unloaded at the dehydrogenation unit, where hydrogen is separated from the LOHC at 1.1 bar. In the second alternative pathway, hydrogen is compressed and loaded into a 350-bar composite tube trailer, transported by road, and unloaded at the destination. In the third pathway, hydrogen is liquefied and loaded into a cryogenic tube trailer. It is then transported by road and delivered to the destination point. As shown in
Figure 1, the two physical transportation ways (compressed and liquified) are shorter than the chemical one. Additionally, in physical-based transport, trucks return with an empty trailer, whereas in LOHC-based transport, they return with a full trailer containing the depleted LOHC. Since the analysis focuses on hydrogen transportation, the final user is not considered, and therefore, no further hydrogen transformations are considered once hydrogen reaches the delivery point.