Submitted:
12 February 2025
Posted:
13 February 2025
You are already at the latest version
Abstract
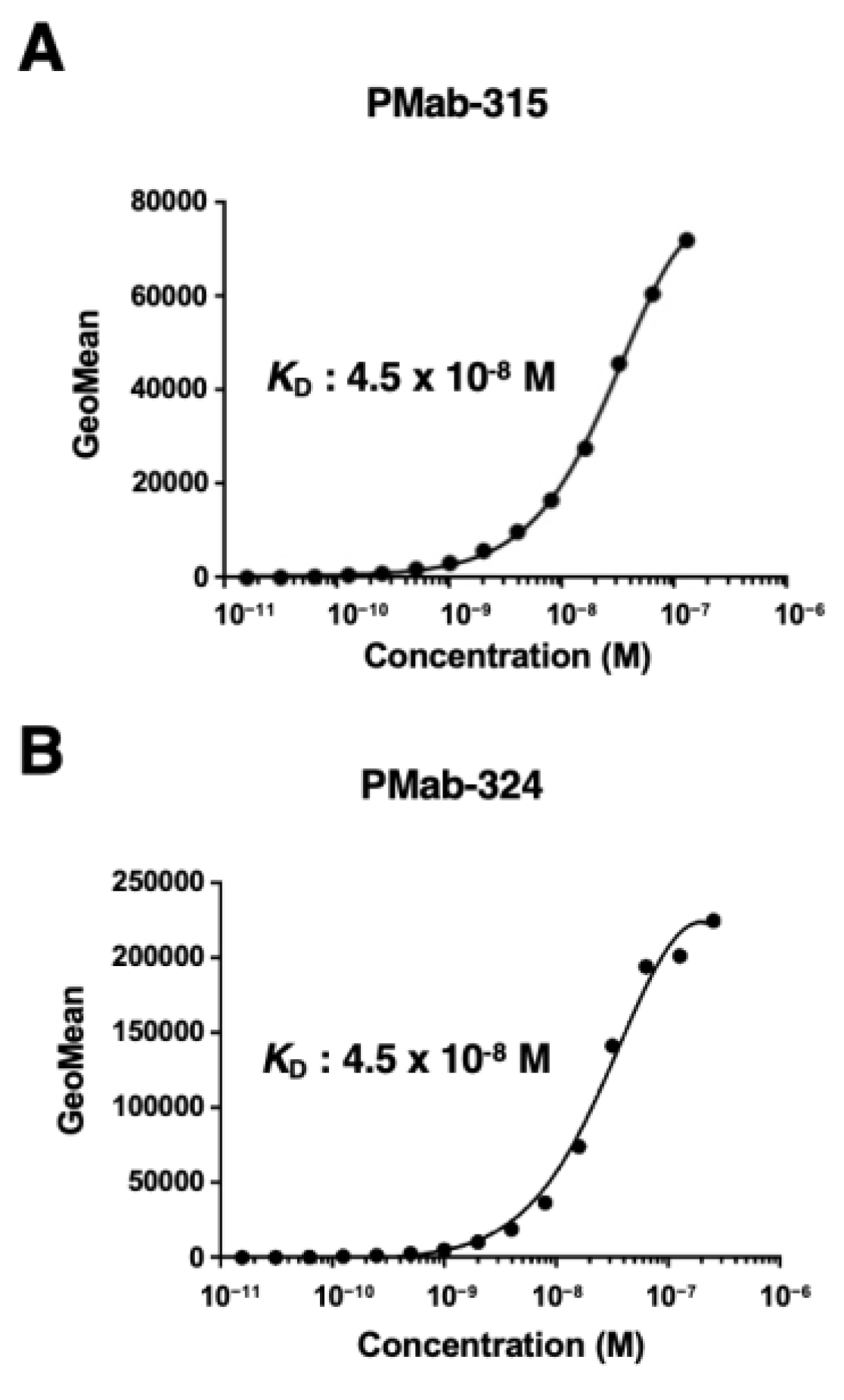
Podoplanin (PDPN) is a type I transmembrane protein with a highly glycosylated extracellular domain, a transmembrane domain, and a short intracellular domain. PDPN expression is observed in normal tissue and cells, including lung type I alveolar epithelial cells, kidney podocytes, and lymphatic endothelial cells. Therefore, anti-PDPN monoclonal antibodies (mAbs) in various animal species helped to identify PDPN-positive cells.We have developed anti-PDPN mAbs in more than 20 species. These anti-PDPN mAbs are applicable to flow cytometry, western blot, and immunohistochemistry. In this study, anti-rhinoceros PDPN (rhiPDPN) mAbs, PMab-315 (mouse IgG2a, kappa) and PMab-324 (mouse IgG2b, kappa) were established using the Cell-Based Immunization and Screening (CBIS) method. PMab-315 and PMab-324 strongly recognized the rhiPDPN-overexpressed Chinese hamster ovary K1 (CHO/rhiPDPN) and exhibited the same KD value (4.5 × 10−8 M) for CHO/rhiPDPN by a flow cytometry-based assay. Both mAbs can detect rhiPDPN in western blot and immunohistochemistry. These results suggest the usefulness of PMab-315 and PMab-324 for the pathological analysis of rhinoceros-derived tissues and cells.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Plasmids
2.2. Production of Hybridomas
2.3. Flow Cytometric Analysis
2.4. Determination of Dissociation Constant (KD) by Flow Cytometry
2.5. Western Blot Analysis
2.6. Immunohistochemical Analysis
3. Results
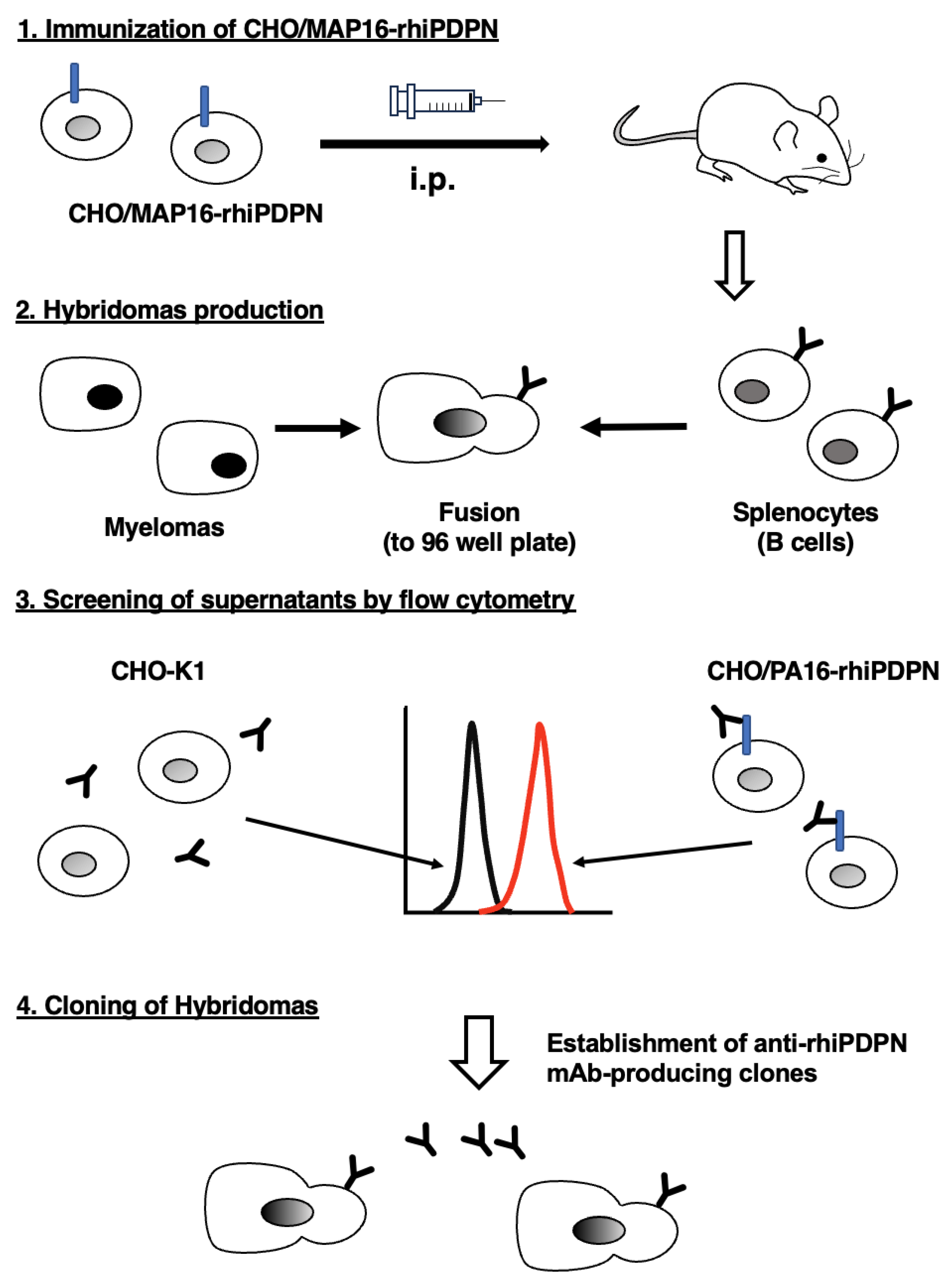
3.1. Development of Anti-rhiPDPN mAbs Using the CBIS Method
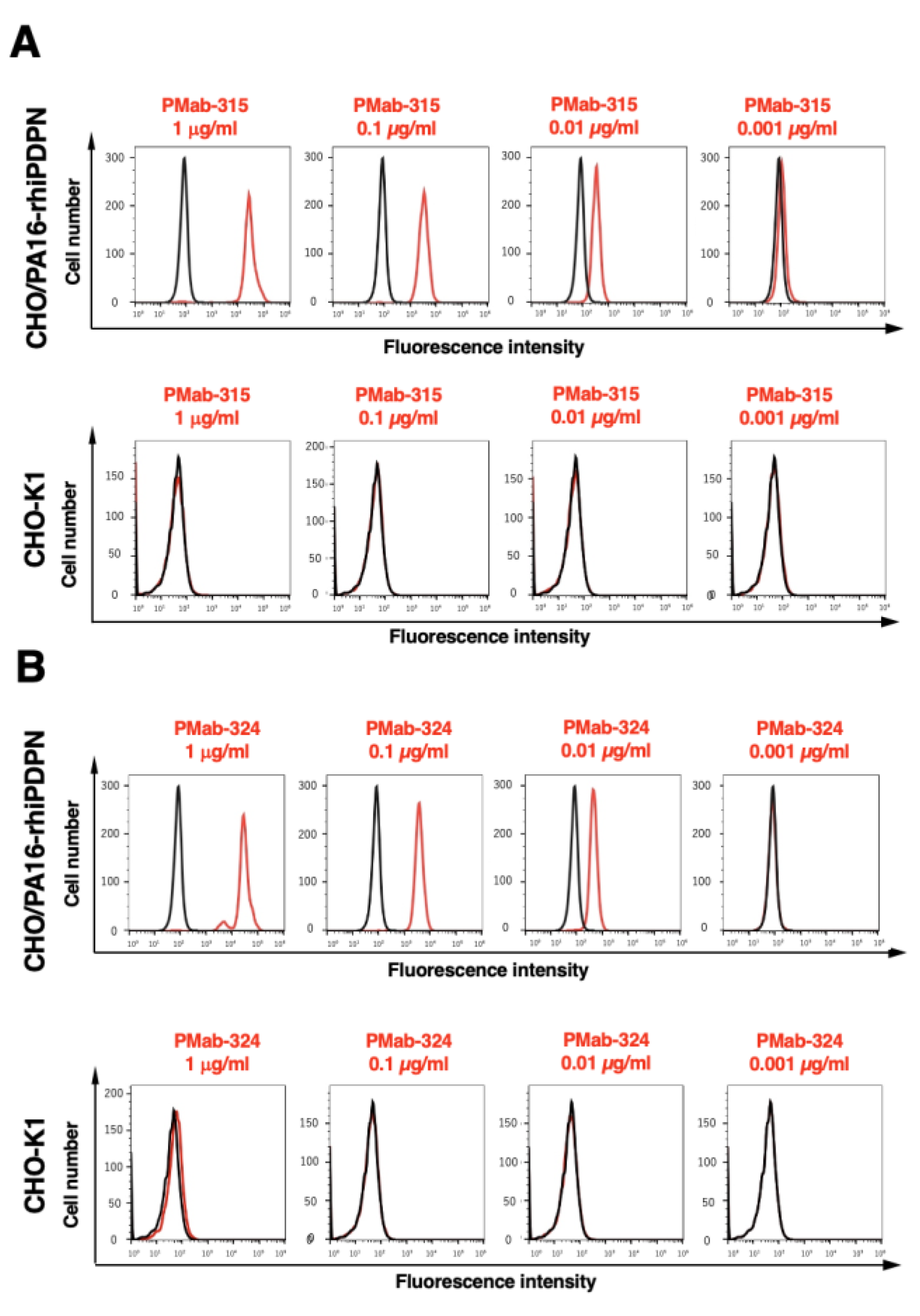
3.2. Flow Cytometry Using PMab-315 and PMab-324
3.3. The Binding Affinity of PMab-315 and PMab-324
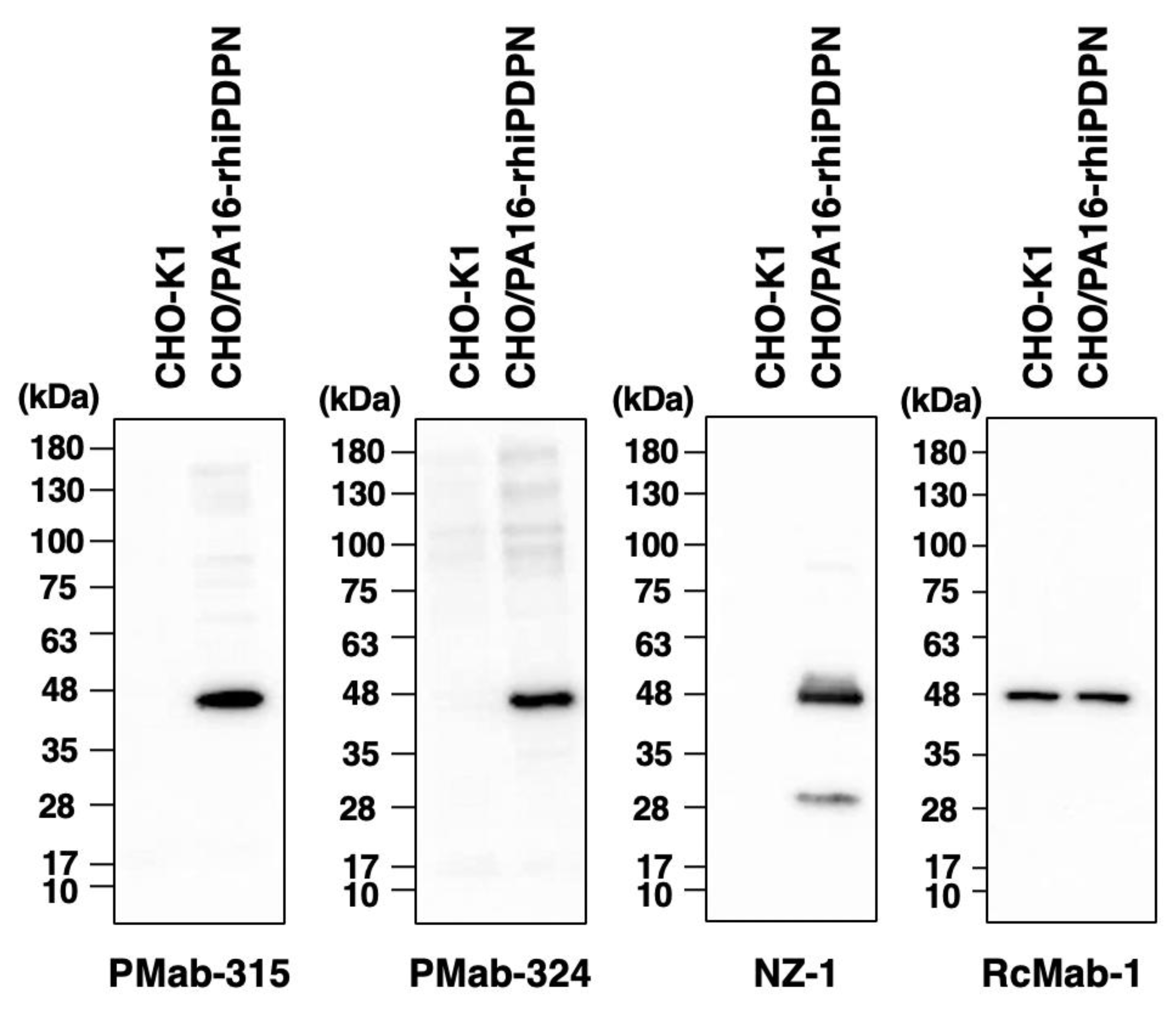
3.4. Western Blot Analysis Using PMab-315 and PMab-324
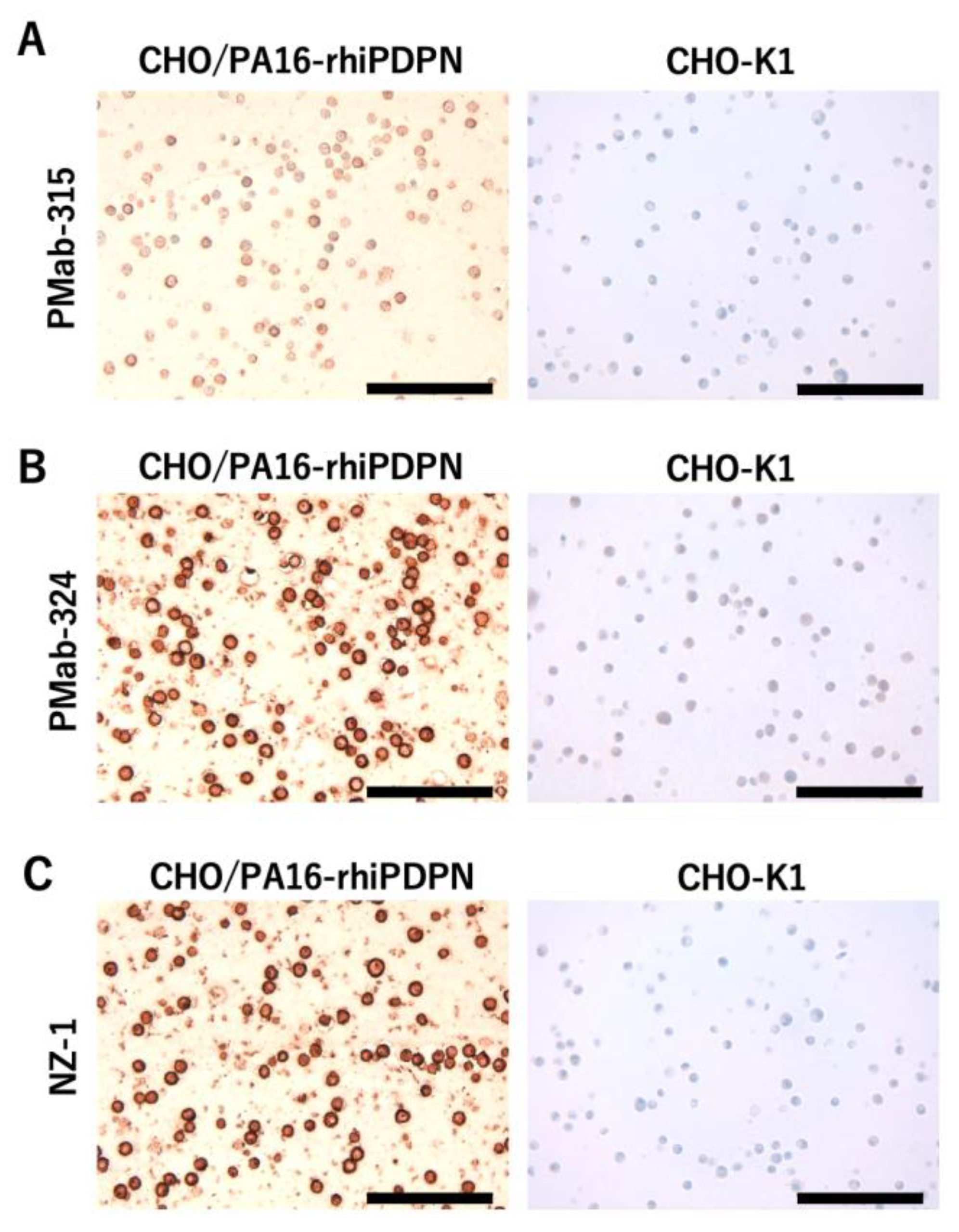
3.5. Immunohistochemistry Using PMab-315 and PMab-324
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kato, Y.; Fujita, N.; Kunita, A.; et al. Molecular identification of Aggrus/T1alpha as a platelet aggregation-inducing factor expressed in colorectal tumors. J Biol Chem 2003;278(51): 51599-51605. [CrossRef]
- Suzuki, H.; Kaneko, M.K.; Kato, Y. Roles of Podoplanin in Malignant Progression of Tumor. Cells 2022;11(3). [CrossRef]
- Dobbs, L.G.; Williams, M.C.; Gonzalez, R. Monoclonal antibodies specific to apical surfaces of rat alveolar type I cells bind to surfaces of cultured, but not freshly isolated, type II cells. Biochim Biophys Acta 1988;970(2): 146-156. [CrossRef]
- Rishi, A.K.; Joyce-Brady, M.; Fisher, J.; et al. Cloning, characterization, and development expression of a rat lung alveolar type I cell gene in embryonic endodermal and neural derivatives. Dev Biol 1995;167(1): 294-306.
- Breiteneder-Geleff, S.; Matsui, K.; Soleiman, A.; et al. Podoplanin, novel 43-kd membrane protein of glomerular epithelial cells, is down-regulated in puromycin nephrosis. Am J Pathol 1997;151(4): 1141-1152.
- Hirakawa, S.; Hong, Y.K.; Harvey, N.; et al. Identification of vascular lineage-specific genes by transcriptional profiling of isolated blood vascular and lymphatic endothelial cells. Am J Pathol 2003;162(2): 575-586. [CrossRef]
- Petrova, T.V.; Mäkinen, T.; Mäkelä, T.P.; et al. Lymphatic endothelial reprogramming of vascular endothelial cells by the Prox-1 homeobox transcription factor. Embo j 2002;21(17): 4593-4599. [CrossRef]
- Schacht, V.; Ramirez, M.I.; Hong, Y.K.; et al. T1alpha/podoplanin deficiency disrupts normal lymphatic vasculature formation and causes lymphedema. Embo j 2003;22(14): 3546-3556.
- Asif, P.J.; Longobardi, C.; Hahne, M.; Medema, J.P. The Role of Cancer-Associated Fibroblasts in Cancer Invasion and Metastasis. Cancers (Basel) 2021;13(18). [CrossRef]
- Sakai, T.; Aokage, K.; Neri, S.; et al. Link between tumor-promoting fibrous microenvironment and an immunosuppressive microenvironment in stage I lung adenocarcinoma. Lung Cancer 2018;126: 64-71. [CrossRef]
- Hoshino, A.; Ishii, G.; Ito, T.; et al. Podoplanin-positive fibroblasts enhance lung adenocarcinoma tumor formation: podoplanin in fibroblast functions for tumor progression. Cancer Res 2011;71(14): 4769-4779. [CrossRef]
- Sasaki, K.; Sugai, T.; Ishida, K.; et al. Analysis of cancer-associated fibroblasts and the epithelial-mesenchymal transition in cutaneous basal cell carcinoma, squamous cell carcinoma, and malignant melanoma. Hum Pathol 2018;79: 1-8. [CrossRef]
- Suzuki, J.; Aokage, K.; Neri, S.; et al. Relationship between podoplanin-expressing cancer-associated fibroblasts and the immune microenvironment of early lung squamous cell carcinoma. Lung Cancer 2021;153: 1-10. [CrossRef]
- Pula, B.; Jethon, A.; Piotrowska, A.; et al. Podoplanin expression by cancer-associated fibroblasts predicts poor outcome in invasive ductal breast carcinoma. Histopathology 2011;59(6): 1249-1260. [CrossRef]
- Shindo, K.; Aishima, S.; Ohuchida, K.; et al. Podoplanin expression in cancer-associated fibroblasts enhances tumor progression of invasive ductal carcinoma of the pancreas. Mol Cancer 2013;12(1): 168. [CrossRef]
- Kamath, P.L. Conserving rhinoceros in the face of disease. Proc Natl Acad Sci U S A 2022;119(25): e2206438119.
- Miller, M.A.; Buss, P.; Parsons, S.D.C.; et al. Conservation of White Rhinoceroses Threatened by Bovine Tuberculosis, South Africa, 2016-2017. Emerg Infect Dis 2018;24(12): 2373-2375.
- Miller, M.A.; Buss, P.E.; van Helden, P.D.; Parsons, S.D. Mycobacterium bovis in a Free-Ranging Black Rhinoceros, Kruger National Park, South Africa, 2016. Emerg Infect Dis 2017;23(3): 557-558.
- Miller, M.; Michel, A.; van Helden, P.; Buss, P. Tuberculosis in Rhinoceros: An Underrecognized Threat? Transbound Emerg Dis 2017;64(4): 1071-1078.
- Dwyer, R.; Goosen, W.; Buss, P.; et al. Epidemiology of Mycobacterium bovis infection in free-ranging rhinoceros in Kruger National Park, South Africa. Proc Natl Acad Sci U S A 2022;119(24): e2120656119.
- Kudo, Y.; Suzuki, H.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Development of a Novel Anti-CD44 Variant 5 Monoclonal Antibody C(44)Mab-3 for Multiple Applications against Pancreatic Carcinomas. Antibodies (Basel) 2023;12(2). [CrossRef]
- Okada, Y.; Suzuki, H.; Kaneko, M.K.; Kato, Y. Development of a Sensitive Anti-Mouse CD39 Monoclonal Antibody (C(39)Mab-1) for Flow Cytometry and Western Blot Analyses. Monoclon Antib Immunodiagn Immunother 2024;43(1): 24-31. [CrossRef]
- Kato, Y.; Kaneko, M.K. A cancer-specific monoclonal antibody recognizes the aberrantly glycosylated podoplanin. Sci Rep 2014;4: 5924. [CrossRef]
- Kaneko, M.K.; Honma, R.; Ogasawara, S.; et al. PMab-38 Recognizes Canine Podoplanin of Squamous Cell Carcinomas. Monoclon Antib Immunodiagn Immunother 2016;35(5): 263-266. [CrossRef]
- Kato, Y.; Ito, Y.; Ohishi, T.; et al. Antibody-Drug Conjugates Using Mouse-Canine Chimeric Anti-Dog Podoplanin Antibody Exerts Antitumor Activity in a Mouse Xenograft Model. Monoclon Antib Immunodiagn Immunother 2020;39(2): 37-44. [CrossRef]
- Fujii, Y.; Kaneko, M.K.; Kato, Y. MAP Tag: A Novel Tagging System for Protein Purification and Detection. Monoclon Antib Immunodiagn Immunother 2016;35(6): 293-299. [CrossRef]
- Fujii, Y.; Kaneko, M.; Neyazaki, M.; et al. PA tag: a versatile protein tagging system using a super high affinity antibody against a dodecapeptide derived from human podoplanin. Protein Expr Purif 2014;95: 240-247. [CrossRef]
- Kaji, C.; Tsujimoto, Y.; Kato Kaneko, M.; Kato, Y.; Sawa, Y. Immunohistochemical Examination of Novel Rat Monoclonal Antibodies against Mouse and Human Podoplanin. Acta. Histochem. Cytochem. 2012;45(4): 227-237. [CrossRef]
- Kato, Y.; Kaneko, M.K.; Kuno, A.; et al. Inhibition of tumor cell-induced platelet aggregation using a novel anti-podoplanin antibody reacting with its platelet-aggregation-stimulating domain. Biochem Biophys Res Commun 2006;349(4): 1301-1307. [CrossRef]
- Bernitz, N.; Kerr, T.J.; Goosen, W.J.; et al. Review of Diagnostic Tests for Detection of Mycobacterium bovis Infection in South African Wildlife. Front Vet Sci 2021;8: 588697. [CrossRef]
- Hlokwe, T.M.; van Helden, P.; Michel, A.L. Evidence of increasing intra and inter-species transmission of Mycobacterium bovis in South Africa: are we losing the battle? Prev Vet Med 2014;115(1-2): 10-17.
- Michel, A.L.; Bengis, R.G.; Keet, D.F.; et al. Wildlife tuberculosis in South African conservation areas: implications and challenges. Vet Microbiol 2006;112(2-4): 91-100. [CrossRef]
- Espie, I.W.; Hlokwe, T.M.; Gey van Pittius, N.C.; et al. Pulmonary infection due to Mycobacterium bovis in a black rhinoceros (Diceros bicornis minor) in South Africa. J Wildl Dis 2009;45(4): 1187-1193. [CrossRef]
- Shadwick, R.E.; Russell, A.P.; Lauff, R.F. The structure and mechanical design of rhinoceros dermal armour. Philos Trans R Soc Lond B Biol Sci 1992;337(1282): 419-428. [CrossRef]
- Plochocki, J.H.; Ruiz, S.; Rodriguez-Sosa, J.R.; Hall, M.I. Histological study of white rhinoceros integument. PLoS One 2017;12(4): e0176327. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



