Submitted:
10 February 2025
Posted:
11 February 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
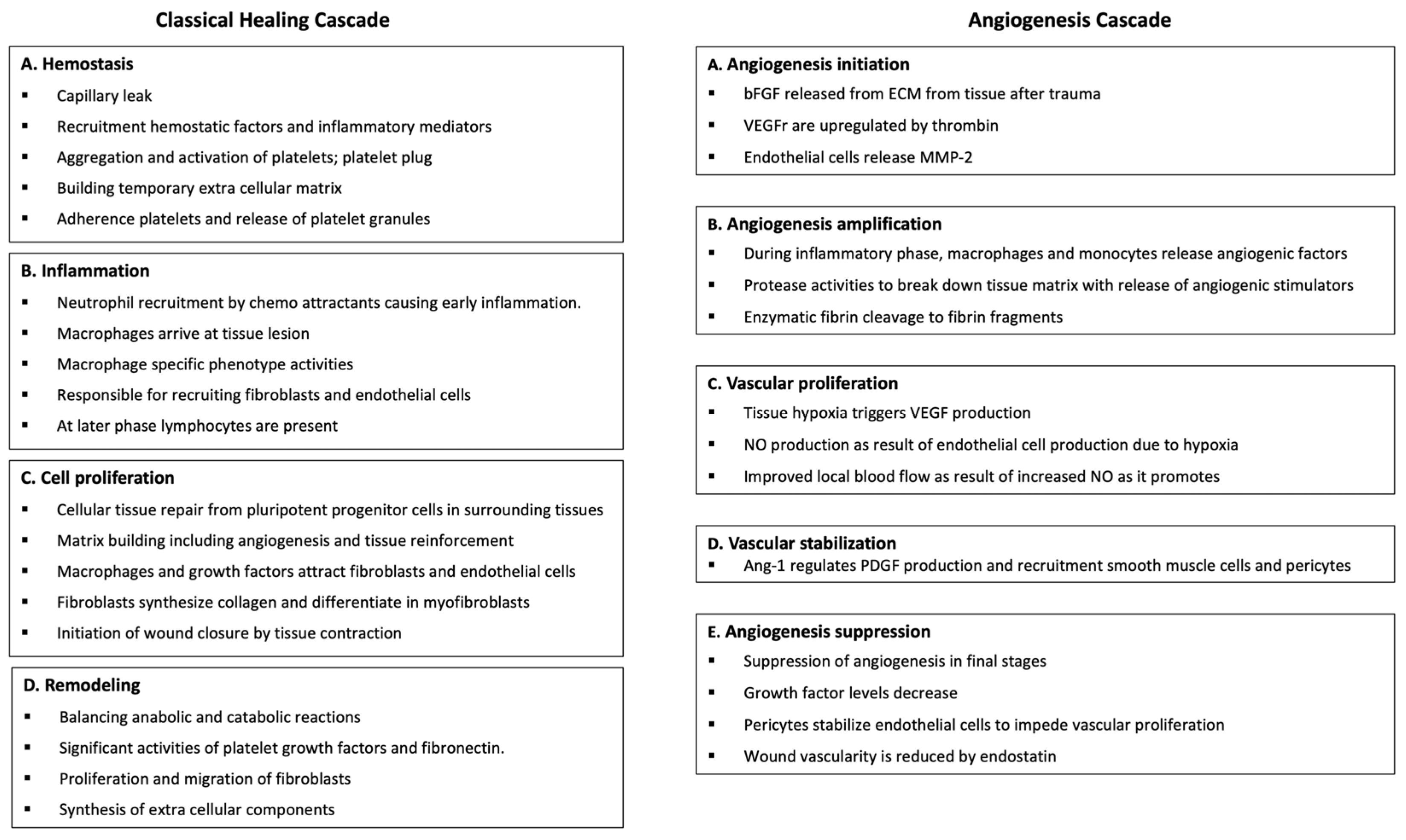
2. Healing Cascades
3. HD-PRP Definition
4. A Brief Overview of Platelets
5. PRP Variability and Its Consequences
5.1. PRP Classification
5.2. Variables in PRP Formulation
6. Defining the Biological Content of PRP and Platelets
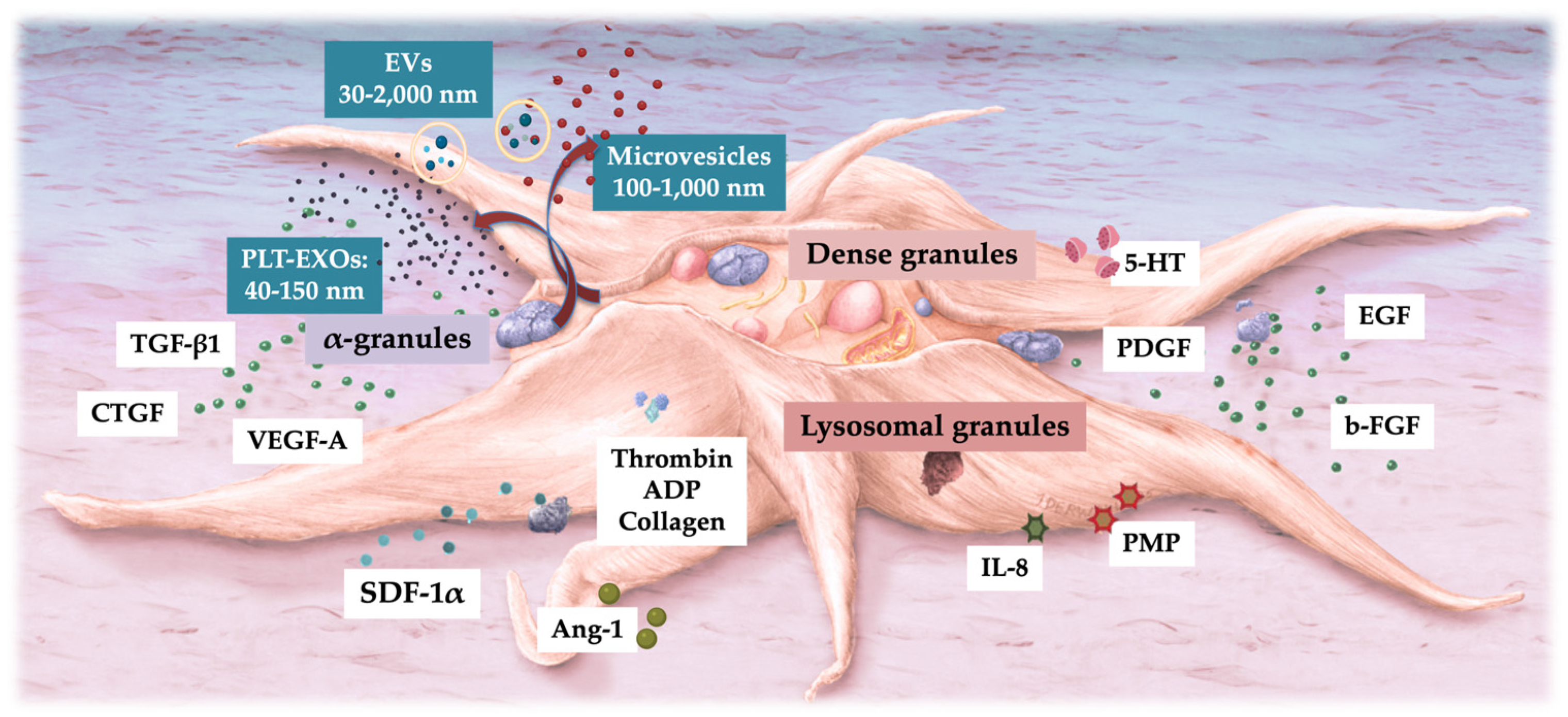
6.1. Platelet α-Granules

6.2. Platelet Dense Granules
6.3. Platelet Lysosomal Granules
6.4. Platelet Exosomes
7. Identifying and Comprehending HD-PRP Characteristics
7.1. Platelet Dose
7.2. Leukocytes in PRP
7.2.1. Platelet-Leukocyte Interactions in HD-PRP
7.3. Platelet-Derived EVs (PLT-EVs) and Exosomes (EXOs)
7.3.1. Autologous PLT-EXOs
7.4. Immunomodulation
7.5. Angiogenesis
7.6. Nociception
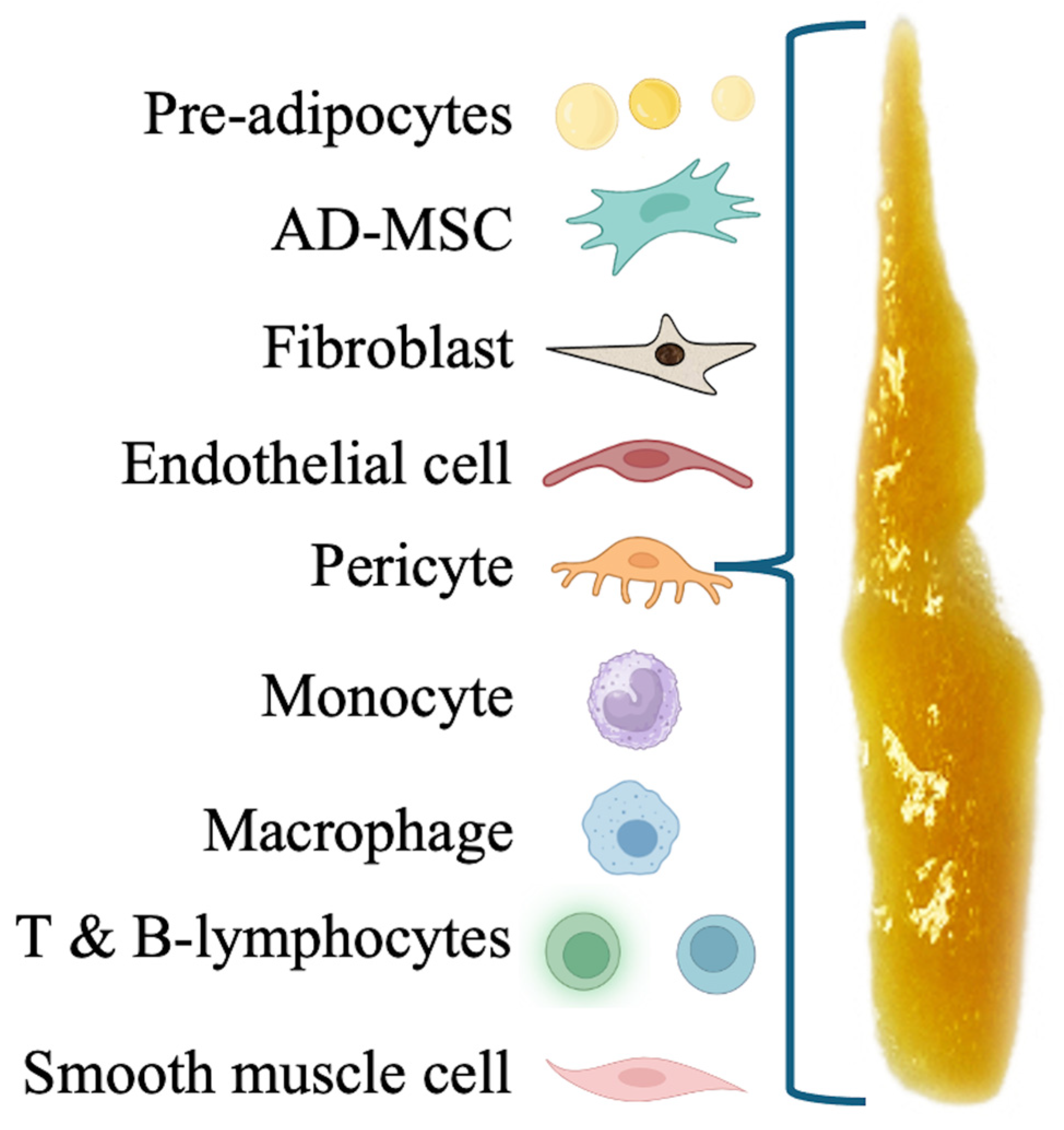
8. Aspects of Adipose Tissue Biology
8.1. Adipose t-SVF Hallmarks
8.2. t-SVF Components
8.2.1. AD-MSCs
8.2.2. Pericyte Stem Cells (PSCs)
8.2.3. Endothelial Cells
8.2.4. Fibroblasts
8.2.5. Immune Cells
9. The Regenerative Marriage Between HD-PRP and Adipose Tissue
9.1. Rationale for Compounding HD-PRP with t-SVF
9.2. Synergistic Effects
9.2.1. HD-PRP and t-SVF Matrix Formation
9.2.2. Optimizing Cell Migration
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ABP | autologous biological preparation |
| AD-MSCs | adipose-derived mesenchymal stem cells |
| ADP | adenosine diphosphate |
| b-FGF | basic fibroblast growth factor |
| BMAC | bone marrow aspirate concentrates |
| CTGF | connective tissue growth facto |
| DC | dendritic cells |
| ECM | extracellular matrix |
| EGF | epidermal growth factor |
| EV | extracellular vesicle |
| HD-PRP | high-density platelet-rich plasma |
| HGF | hepatocyte growth factor |
| IGF-1 | insulin-like growth factor-1 |
| IL | interleukin |
| ILV | intraluminal vesicle |
| LP-PRP | leukocyte-poor PRP |
| LR-PRP | leukocyte-rich PRP |
| MSC | mesenchymal stem cell |
| MSK | musculoskeletal |
| MVB | multivesicular bodies |
| PDGF | platelet-derived growth factor |
| PGF | platelet growth factor |
| PLT-EXO | platelet-derived exosomes |
| PPX | patient pure exosomes |
| PRF | platelet rich fibrin |
| PRP | platelet-rich plasma |
| PSC | pericyte stem cell |
| SVF | stromal vascular fractions |
| t-SVF | tissue stromal vascular fraction |
| TDP | deliverable platelets |
| TGF-b | transforming growth factor-b |
| Th-cell | T helper cell |
| Treg | regulatory T cell |
| VEGF | vascular endothelial growth factor |
References
- Everts P, Onishi K, Jayaram P, Lana JF, Mautner and K. Platelet-Rich Plasma: New Performance Understandings and Therapeutic Considerations in 2020 [Internet]. MEDICINE & PHARMACOLOGY; 2020 Oct [cited 2020 Oct 21]. Available online: https://www.preprints.org/manuscript/202010.0069/v1.
- A. Everts P. Autologous Platelet-Rich Plasma and Mesenchymal Stem Cells for the Treatment of Chronic Wounds. In: Hakan Dogan K, editor. Wound Healing - Current Perspectives [Internet]. IntechOpen; 2019 [cited 2020 Sep 10]. Available online: https://www.intechopen.com/books/wound-healing-current-perspectives/autologous-platelet-rich-plasma-and-mesenchymal-stem-cells-for-the-treatment-of-chronic-wounds.
- Calvi LM, Link DC. The hematopoietic stem cell niche in homeostasis and disease. Blood. 2015 Nov 26;126[22]:2443–51.
- Lana JF, Purita J, Everts PA, De Mendonça Neto PAT, De Moraes Ferreira Jorge D, Mosaner T, et al. Platelet-Rich Plasma Power-Mix Gel (ppm)—An Orthobiologic Optimization Protocol Rich in Growth Factors and Fibrin. Gels. 2023 Jul 7;9[7]:553.
- Alexander RW. Adipose Tissue Complex (ATC): Cellular and Biocellular Uses of Stem/Stromal Cells and Matrix in Cosmetic Plastic, Reconstructive Surgery and Regenerative Medicine. In: Duscher D, Shiffman MA, editors. Regenerative Medicine and Plastic Surgery [Internet]. Cham: Springer International Publishing; 2019 [cited 2024 Mar 24]. p. 45–69. Available online: https://link.springer.com/10.1007/978-3-030-19962-3_5.
- Everts P, M. Hoogbergen M, A.Weber T, J.J. Devilee R, van Monftort G, H.J.T. de Hingh I. Is the Use of Autologous Platelet-Rich Plasma Gels in Gynecologic, Cardiac, and General, Reconstructive Surgery Beneficial? Curr Pharm Biotechnol. 2012 May 1;13[7]:1163–72.
- Gentile P, Garcovich S. Systematic Review—The Potential Implications of Different Platelet-Rich Plasma (PRP) Concentrations in Regenerative Medicine for Tissue Repair. Int J Mol Sci. 2020 Aug 9;21[16]:5702.
- Everts PA, Sadeghi P, Smith DR. Basic Science of Autologous Orthobiologics. Phys Med Rehabil Clin N Am. 2023 Feb;34[1]:1–23.
- Honnegowda TM, Kumar P, Udupa EGP, Kumar S, Kumar U, Rao P. Role of angiogenesis and angiogenic factors in acute and chronic wound healing. 2015;2(5):7.
- Tonnesen MG, Feng X, Clark RAF. Angiogenesis in Wound Healing.
- Marx, RE. Platelet-Rich Plasma (PRP): What Is PRP and What Is Not PRP?: Implant Dent. 2001 Dec;10(4):225–8.
- Tate-Oliver K, Alexander RW. Combination of Autologous Adipose-Derived Tissue Stromal Vascular Fraction Plus High Density Platelet-Rich Plasma or Bone Marrow Concentrates in Achilles Tendon Tears.
- Pokrovskaya ID, Yadav S, Rao A, McBride E, Kamykowski JA, Zhang G, et al. 3D ultrastructural analysis of α-granule, dense granule, mitochondria, and canalicular system arrangement in resting human platelets. Res Pract Thromb Haemost. 2020 Jan;4(1):72–85.
- Day RB, Link DC. Megakaryocytes in the hematopoietic stem cell niche. Nat Med. 2014 Nov;20(11):1233–4.
- Roh YH, Kim W, Park KU, Oh JH. Cytokine-release kinetics of platelet-rich plasma according to various activation protocols. Bone Jt Res. 2016 Feb;5(2):37–45.
- Hurley ET, Fat DL, Moran CJ, Mullett H. The Efficacy of Platelet-Rich Plasma and Platelet-Rich Fibrin in Arthroscopic Rotator Cuff Repair: A Meta-analysis of Randomized Controlled Trials. Am J Sports Med. :8.
- Bennell KL, Paterson KL, Metcalf BR, Duong V, Eyles J, Kasza J, et al. Effect of Intra-articular Platelet-Rich Plasma vs Placebo Injection on Pain and Medial Tibial Cartilage Volume in Patients With Knee Osteoarthritis: The RESTORE Randomized Clinical Trial. JAMA. 2021 Nov 23;326(20):2021.
- Rodeo SA, Delos D, Williams RJ, Adler RS, Pearle A, Warren RF. The Effect of Platelet-Rich Fibrin Matrix on Rotator Cuff Tendon Healing: A Prospective, Randomized Clinical Study. Am J Sports Med. 2012 Jun;40(6):1234–41.
- Korpershoek JV, Vonk LA, De Windt TS, Admiraal J, Kester EC, Van Egmond N, et al. Intra-articular injection with Autologous Conditioned Plasma does not lead to a clinically relevant improvement of knee osteoarthritis: a prospective case series of 140 patients with 1-year follow-up. Acta Orthop. 2020 Nov 1;91(6):743–9.
- Everts PA, Mazzola T, Mautner K, Randelli PS, Podesta L. Modifying Orthobiological PRP Therapies Are Imperative for the Advancement of Treatment Outcomes in Musculoskeletal Pathologies. Biomedicines. 2022 Nov 15;10(11):2933.
- Ehrenfest DMD, Andia I, Zumstein MA, Zhang CQ, Pinto NR, Bielecki T. Classification of platelet concentrates (Platelet-Rich Plasma-PRP, Platelet-Rich Fibrin-PRF) for topical and infiltrative use in orthopedic and sports medicine: current consensus, clinical implications and perspectives. :7.
- DeLong JM, Russell RP, Mazzocca AD. Platelet-Rich Plasma: The PAW Classification System. Arthrosc J Arthrosc Relat Surg. 2012 Jul;28(7):998–1009.
- Mishra A, Harmon K, Woodall J, Vieira A. Sports Medicine Applications of Platelet Rich Plasma. Curr Pharm Biotechnol. 2012 May 1;13(7):1185–95.
- Mautner K, Malanga GA, Smith J, Shiple B, Ibrahim V, Sampson S, et al. A Call for a Standard Classification System for Future Biologic Research: The Rationale for New PRP Nomenclature. PM&R. 2015 Apr;7:S53–9.
- Magalon J, Chateau AL, Bertrand B, Louis ML, Silvestre A, Giraudo L, et al. DEPA classification: a proposal for standardising PRP use and a retrospective application of available devices. BMJ Open Sport Exerc Med. 2016 Feb;2(1):e000060.
- Lana JFSD, Purita J, Paulus C, Huber SC, Rodrigues BL, Rodrigues AA, et al. Contributions for classification of platelet rich plasma – proposal of a new classification: MARSPILL. Regen Med. 2017 Jul;12(5):565–74.
- Kon E, Di Matteo B, Delgado D, Cole BJ, Dorotei A, Dragoo JL, et al. Platelet-rich plasma for the treatment of knee osteoarthritis: an expert opinion and proposal for a novel classification and coding system. Expert Opin Biol Ther. 2020 Dec 1;20(12):1447–60.
- Everts PAM, van Zundert A, Schönberger JPAM, Devilee RJJ, Knape JTA. What do we use: Platelet-rich plasma or platelet-leukocyte gel? J Biomed Mater Res A. 2008 Jun 15;85A(4):1135–6.
- Chahla J, Cinque ME, Piuzzi NS, Mannava S, Geeslin AG, Murray IR, et al. A Call for Standardization in Platelet-Rich Plasma Preparation Protocols and Composition Reporting: A Systematic Review of the Clinical Orthopaedic Literature. J Bone Jt Surg. 2017 Oct 18;99(20):1769–79.
- Everts PA, Flanagan G, Podesta L. Autologous Orthobiologics. In: Mostoufi SA, George TK, Tria AJ, editors. Clinical Guide to Musculoskeletal Medicine [Internet]. Cham: Springer International Publishing; 2022 [cited 2022 Sep 22]. p. 651–79. Available online: https://link.springer.com/10.1007/978-3-030-92042-5_62.
- Miron RJ, Chai J, Fujioka-Kobayashi M, Sculean A, Zhang Y. Evaluation of 24 protocols for the production of platelet-rich fibrin. BMC Oral Health. 2020 Dec;20(1):310.
- Magalon J, Brandin T, Francois P, Degioanni C, De Maria L, Grimaud F, et al. Technical and biological review of authorized medical devices for platelets-rich plasma preparation in the field of regenerative medicine. Platelets. 2021 Feb 17;32(2):200–8.
- Marathe A, Patel SJ, Song B, Sliepka JM, Shybut TS, Lee BH, et al. Double-Spin Leukocyte-Rich Platelet-Rich Plasma Is Predominantly Lymphocyte Rich With Notable Concentrations of Other White Blood Cell Subtypes. Arthrosc Sports Med Rehabil. 2022 Apr;4(2):e335–41.
- Fitzpatrick J, Bulsara MK, McCrory PR, Richardson MD, Zheng MH. Analysis of Platelet-Rich Plasma Extraction: Variations in Platelet and Blood Components Between 4 Common Commercial Kits. Orthop J Sports Med. 2017 Jan;5(1):232596711667527.
- Piao L, Park H, Jo CH. Theoretical prediction and validation of cell recovery rates in preparing platelet-rich plasma through a centrifugation. Wang JHC, editor. PLOS ONE. 2017 Nov 2;12(11):e0187509.
- Cherian C, Malanga G, Mautner K. OPTIMIZING PLATELET-RICH PLASMA (PRP) INJECTIONS: A NARRATIVPEroceedings. 2:17.
- Guo SC, Tao SC, Yin WJ, Qi X, Yuan T, Zhang CQ. Exosomes derived from platelet-rich plasma promote the re-epithelization of chronic cutaneous wounds via activation of YAP in a diabetic rat model. Theranostics. 2017;7(1):81–96.
- Everts PA, Lana JF, Onishi K, Buford D, Peng J, Mahmood A, et al. Angiogenesis and Tissue Repair Depend on Platelet Dosing and Bioformulation Strategies Following Orthobiological Platelet-Rich Plasma Procedures: A Narrative Review. 2023.
- Hayashi S, Aso H, Watanabe K, Nara H, Rose MT, Ohwada S, et al. Sequence of IGF-I, IGF-II, and HGF expression in regenerating skeletal muscle. Histochem Cell Biol. 2000;122:427–34.
- Everts PA, Jakimowicz JJ, van Beek M, Schönberger JPAM, Devilee RJJ, Overdevest EP, et al. Reviewing the Structural Features of Autologous Platelet-Leukocyte Gel and Suggestions for Use in Surgery. Eur Surg Res. 2007;39(4):199–207.
- Kaiko GE, Horvat JC, Beagley KW, Hansbro PM. Immunological decision-making: how does the immune system decide to mount a helper T-cell response? Immunology. 2008 Mar;123(3):326–38.
- Heijnen H, van der Sluijs P. Platelet secretory behaviour: as diverse as the granules … or not? J Thromb Haemost. 2015 Dec;13(12):2141–51.
- Gray MA, Choy CH, Dayam RM, Ospina-Escobar E, Somerville A, Xiao X, et al. Phagocytosis Enhances Lysosomal and Bactericidal Properties by Activating the Transcription Factor TFEB. Curr Biol. 2016 Aug;26(15):1955–64.
- Kalra H, Drummen G, Mathivanan S. Focus on Extracellular Vesicles: Introducing the Next Small Big Thing. Int J Mol Sci. 2016 Feb 6;17(2):170.
- Rui S, Yuan Y, Du C, Song P, Chen Y, Wang H, et al. Comparison and Investigation of Exosomes Derived from Platelet-Rich Plasma Activated by Different Agonists. Cell Transplant. 2021 Jan 1;30:096368972110178.
- Spakova T, Janockova J, Rosocha J. Characterization and Therapeutic Use of Extracellular Vesicles Derived from Platelets. Int J Mol Sci. 2021 Sep 8;22(18):9701.
- Fadadu PP, Mazzola AJ, Hunter CW, Davis TT. Review of concentration yields in commercially available platelet-rich plasma (PRP) systems: a call for PRP standardization. Reg Anesth Pain Med. 2019 Jun;44(6):652–9.
- Giusti I, D’Ascenzo S, Mancò A, Di Stefano G, Di Francesco M, Rughetti A, et al. Platelet Concentration in Platelet-Rich Plasma Affects Tenocyte Behavior In Vitro. BioMed Res Int. 2014;2014:1–12.
- Berger DR, Centeno CJ, Steinmetz NJ. Platelet lysates from aged donors promote human tenocyte proliferation and migration in a concentration-dependent manner. Bone Jt Res. 2019 Jan;8(1):32–40.
- Bansal H, Leon J, Pont JL, Wilson DA, St SM, Bansal A, et al. Clinical Efficacy of a Single-dose of Platelet Rich Plasma in the Management of Early Knee Osteoarthritis: A Randomized Controlled Study With Mri Assessment and Evaluation of Optimal Dose [Internet]. In Review; 2020 Jun [cited 2020 Jul 7]. Available online: https://www.researchsquare.com/article/rs-33058/v1.
- Melo, Luzo, Lana, Santana. Centrifugation Conditions in the L-PRP Preparation Affect Soluble Factors Release and Mesenchymal Stem Cell Proliferation in Fibrin Nanofibers. Molecules. 2019 Jul 27;24(15):2729.
- Lana, JF. Leukocyte-rich PRP versus leukocyte-poor PRP - The role of monocyte/macrophage function in the healing cascade. J Clin Orthop Trauma. 2019;6.
- Zhou Y, Zhang J, Wu H, Hogan MV, Wang JHC. The differential effects of leukocyte-containing and pure platelet-rich plasma (PRP) on tendon stem/progenitor cells - implications of PRP application for the clinical treatment of tendon injuries. Stem Cell Res Ther. 2015 Dec;6(1):173.
- N. Serhan C, Krishnamoorthy S, Recchiuti A, Chiang N. Novel Anti-Inflammatory-Pro-Resolving Mediators and Their Receptors. Curr Top Med Chem. 2011 Mar 1;11(6):629–47.
- Kargarpour Z, Panahipour L, Mildner M, Miron RJ, Gruber R. Lipids of Platelet-Rich Fibrin Reduce the Inflammatory Response in Mesenchymal Cells and Macrophages. Cells. 2023 Feb 16;12(4):634.
- Serhan CN, Sheppard KA. Lipoxin formation during human neutrophil-platelet interactions. Evidence for the transformation of leukotriene A4 by platelet 12-lipoxygenase in vitro. J Clin Invest. 1990 Mar 1;85(3):772–80.
- Sadtler K, Estrellas K, Allen BW, Wolf MT, Fan H, Tam AJ, et al. Developing a pro-regenerative biomaterial scaffold microenvironment requires T helper 2 cells. Science. 2016 Apr 15;352(6283):366–70.
- Uchiyama R, Toyoda E, Maehara M, Wasai S, Omura H, Watanabe M, et al. Effect of Platelet-Rich Plasma on M1/M2 Macrophage Polarization. Int J Mol Sci. 2021 Feb 26;22(5):2336.
- Andia I, Martin JI, Maffulli N. Advances with platelet rich plasma therapies for tendon regeneration. Expert Opin Biol Ther. 2018 Apr 3;18(4):389–98.
- Lin KY, Chen P, Chen ACY, Chan YS, Lei KF, Chiu CH. Leukocyte-Rich Platelet-Rich Plasma Has Better Stimulating Effects on Tenocyte Proliferation Compared With Leukocyte-Poor Platelet-Rich Plasma. Orthop J Sports Med. 2022 Mar 1;10(3):232596712210847.
- Romandini I, Boffa A, Di Martino A, Andriolo L, Cenacchi A, Sangiorgi E, et al. Leukocytes Do Not Influence the Safety and Efficacy of Platelet-Rich Plasma Injections for the Treatment of Knee Osteoarthritis: A Double-Blind Randomized Controlled Trial. Am J Sports Med. 2024 Nov;52(13):3212–22.
- Liu X, Wang L, Ma C, Wang G, Zhang Y, Sun S. Exosomes derived from platelet-rich plasma present a novel potential in alleviating knee osteoarthritis by promoting proliferation and inhibiting apoptosis of chondrocyte via Wnt/β-catenin signaling pathway. J Orthop Surg. 2019 Dec;14(1):470.
- Colombo M, Raposo G, Théry C. Biogenesis, Secretion, and Intercellular Interactions of Exosomes and Other Extracellular Vesicles. Annu Rev Cell Dev Biol. 2014 Oct 11;30(1):255–89.
- Gupta AK, Wang T, Rapaport JA, Talukder M. Therapeutic Potential of Extracellular Vesicles (Exosomes) Derived From Platelet-Rich Plasma: A Literature Review. J Cosmet Dermatol. 2024 Dec;e16709.
- Murphy C, Withrow J, Hunter M, Liu Y, Tang YL, Fulzele S, et al. Emerging role of extracellular vesicles in musculoskeletal diseases. Mol Aspects Med. 2018 Apr;60:123–8.
- Cano A, Muñoz-Morales Á, Sánchez-López E, Ettcheto M, Souto EB, Camins A, et al. Exosomes-Based Nanomedicine for Neurodegenerative Diseases: Current Insights and Future Challenges. Pharmaceutics. 2023 Jan 16;15(1):298.
- Heidarzadeh M, Zarebkohan A, Rahbarghazi R, Sokullu E. Protein corona and exosomes: new challenges and prospects. Cell Commun Signal. 2023 Mar 27;21(1):64.
- Heidarzadeh M, Gürsoy-Özdemir Y, Kaya M, Eslami Abriz A, Zarebkohan A, Rahbarghazi R, et al. Exosomal delivery of therapeutic modulators through the blood–brain barrier; promise and pitfalls. Cell Biosci. 2021 Jul 22;11(1):142.
- Rajabi H, Konyalilar N, Erkan S, Mortazavi D, Korkunc SK, Kayalar O, et al. Emerging role of exosomes in the pathology of chronic obstructive pulmonary diseases; destructive and therapeutic properties. Stem Cell Res Ther. 2022 Apr 4;13(1):144.
- Penna F, Garcia-Castillo L, Costelli P. Extracellular Vesicles and Exosomes in the Control of the Musculoskeletal Health. Curr Osteoporos Rep. 2024 Apr;22(2):257–65.
- Wan R, Liu S, Feng X, Luo W, Zhang H, Wu Y, et al. The Revolution of exosomes: From biological functions to therapeutic applications in skeletal muscle diseases. J Orthop Transl. 2024 Mar;45:132–9.
- Chaudhary PK, Kim S, Kim S. An Insight into Recent Advances on Platelet Function in Health and Disease. Int J Mol Sci. 2022 May 27;23(11):6022.
- Torreggiani E, Perut F, Roncuzzi L, Zini N, Baglìo SR, Baldini N. Exosomes: novel effectors of human platelet lysate activity. Eur Cell Mater. 2014;28:137–51.
- Szunerits S, Chuang EY, Yang JC, Boukherroub R, Burnouf T. Platelet extracellular vesicles-loaded hydrogel bandages for personalized wound care. Trends Biotechnol. 2025 Jan;S0167779924003937.
- Institute of Orthopaedic Research and Biomechanics, University of Ulm, Helmholtzstr. 14, D-89081, Ulm, Germany, Kovtun A, Bergdolt S, Wiegner R, Radermacher P, Huber-Lang M, et al. The crucial role of neutrophil granulocytes in bone fracture healing. Eur Cell Mater. 2016 Jul 25;32:152–62.
- Iberg CA, Hawiger D. Natural and Induced Tolerogenic Dendritic Cells. J Immunol. 2020 Feb 15;204(4):733–44.
- Koupenova M, Clancy L, Corkrey HA, Freedman JE. Circulating Platelets as Mediators of Immunity, Inflammation, and Thrombosis. Circ Res. 2018 Jan 19;122(2):337–51.
- Shibuya, M. Vascular Endothelial Growth Factor (VEGF) and Its Receptor (VEGFR) Signaling in Angiogenesis: A Crucial Target for Anti- and Pro-Angiogenic Therapies. Genes Cancer. 2011 Dec 1;2(12):1097–105.
- Everts PA, Devilee RJJ, Brown Mahoney C, van Erp A, Oosterbos CJM, Stellenboom M, et al. Exogenous Application of Platelet-Leukocyte Gel during Open Subacromial Decompression Contributes to Improved Patient Outcome. Eur Surg Res. 2008;40(2):203–10.
- Johal H, Khan M, Yung S hang P, Dhillon MS, Fu FH, Bedi A, et al. Impact of Platelet-Rich Plasma Use on Pain in Orthopaedic Surgery: A Systematic Review and Meta-analysis. Sports Health Multidiscip Approach. 2019 Jul;11(4):355–66.
- Kuffler, DP. Platelet-Rich Plasma Promotes Axon Regeneration, Wound Healing, and Pain Reduction: Fact or Fiction. Mol Neurobiol. 2015;27.
- Kuffler, D. Variables affecting the potential efficacy of PRP in providing chronic pain relief. J Pain Res. 2018 Dec;Volume 12:109–16.
- Kuffler DP, Foy C. Restoration of Neurological Function Following Peripheral Nerve Trauma. Int J Mol Sci. 2020 Mar 6;21(5):1808.
- Mohammadi S, Nasiri S, Mohammadi MH, Malek Mohammadi A, Nikbakht M, Zahed Panah M, et al. Evaluation of platelet-rich plasma gel potential in acceleration of wound healing duration in patients underwent pilonidal sinus surgery: A randomized controlled parallel clinical trial. Transfus Apher Sci. 2017 Apr;56(2):226–32.
- Lutz C, Cheng J, Prysak M, Zukofsky T, Rothman R, Lutz G. Clinical outcomes following intradiscal injections of higher-concentration platelet-rich plasma in patients with chronic lumbar discogenic pain. Int Orthop [Internet]. 2022 Mar 28 [cited 2022 Apr 26]. Available online: https://link.springer.com/10.1007/s00264-022-05389-y.
- Migliorini F, Rath B, Tingart M, Baroncini A, Quack V, Eschweiler J. Autogenic mesenchymal stem cells for intervertebral disc regeneration. Int Orthop. 2019 Apr;43(4):1027–36.
- Gupta A, Jeyaraman M, Potty A. Leukocyte-Rich vs. Leukocyte-Poor Platelet-Rich Plasma for the Treatment of Knee Osteoarthritis. Biomedicines. 2023 Jan 6;11(1):141.
- Jeyaraman M, Maffulli N, Gupta A. Stromal Vascular Fraction in Osteoarthritis of the Knee. Biomedicines. 2023 May 16;11(5):1460.
- Van Boxtel J, Vonk LA, Stevens HP, Van Dongen JA. Mechanically Derived Tissue Stromal Vascular Fraction Acts Anti-inflammatory on TNF Alpha-Stimulated Chondrocytes In Vitro. Bioengineering. 2022 Jul 27;9(8):345.
- Alexander, R. Overview of cellular stromal vascular fraction (cSVF) & biocellular uses of stem/stromal cells & matrix (tSVF+ HD-PRP) in regenerative medicine, aesthetic medicine and plastic surgery. J Stem Cell Res Dev Ther. 2019;2:304–12.
- Klein, JA. The Tumescent Technique: Anesthesia and Modified Liposuction Technique. Dermatol Clin. 1990;8(3):425–37.
- Simonacci F, Bertozzi N, Grieco MP, Raposio E. From liposuction to adipose-derived stem cells: indications and technique. Acta Bio Medica Atenei Parm. 2019 May 23;90(2):197–208.
- Kurita M, Matsumoto D, Shigeura T, Sato K, Gonda K, Harii K, et al. Influences of Centrifugation on Cells and Tissues in Liposuction Aspirates: Optimized Centrifugation for Lipotransfer and Cell Isolation: Plast Reconstr Surg. 2008 Mar;121(3):1033–41.
- Williams SK, Wang TF, Castrillo R, Jarrell BE. Liposuction-derived human fat used for vascular graft sodding contains endothelial cells and not mesothelial cells as the major cell type. J Vasc Surg. 1994 May;19(5):916–23.
- Alexander, RW. Understanding Mechanical Emulsification (Nanofat) Versus Enzymatic Isolation of Tissue Stromal Vascular Fraction (tSVF) Cells from Adipose Tissue: Potential Uses in Biocellular Regenerative Medicine. :14.
- Vasilyev VS, Borovikova AA, Vasilyev SA, Khramtsova NI, Plaksin SA, Kamyshinsky RA, et al. Features and Biological Properties of Different Adipose Tissue Based Products. Milli-, Micro-, Emulsified (Nano-) Fat, SVF, and AD-Multipotent Mesenchymal Stem Cells. In: Plastic and Aesthetic Regenerative Surgery and Fat Grafting: Clinical Application and Operative Techniques. Springer; 2022. p. 91–107.
- Caplan AI, Correa D. The MSC: An Injury Drugstore. Cell Stem Cell. 2011 Jul;9(1):11–5.
- Li L, Xie T. STEM CELL NICHE: Structure and Function. 2005;30.
- Dallo I, Bernáldez P, Santos GS, Lana JF, Everts PA. Defining, optimizing, and measuring bone marrow aspirate and bone marrow concentrate. In: OrthoBiologics [Internet]. Elsevier; 2025 [cited 2025 Jan 28]. p. 47–59. Available online: https://linkinghub.elsevier.com/retrieve/pii/B9780128229026000210.
- Mohamed-Ahmed S, Fristad I, Lie SA, Suliman S, Mustafa K, Vindenes H, et al. Adipose-derived and bone marrow mesenchymal stem cells: a donor-matched comparison. Stem Cell Res Ther. 2018 Dec;9(1):168.
- Charbord, P. Bone Marrow Mesenchymal Stem Cells: Historical Overview and Concepts. Hum Gene Ther. 2010 Sep;21(9):1045–56.
- Izadpanah R, Trygg C, Patel B, Kriedt C, Dufour J, Gimble JM, et al. Biologic properties of mesenchymal stem cells derived from bone marrow and adipose tissue. J Cell Biochem. 2006 Dec 1;99(5):1285–97.
- Centeno C, Pitts J, Al-Sayegh H, Freeman M. Efficacy of Autologous Bone Marrow Concentrate for Knee Osteoarthritis with and without Adipose Graft. BioMed Res Int. 2014;2014:1–9.
- da Silva Meirelles L, Fontes AM, Covas DT, Caplan AI. Mechanisms involved in the therapeutic properties of mesenchymal stem cells. Cytokine Growth Factor Rev. 2009 Oct;20(5–6):419–27.
- Cucchiarini M, Venkatesan JK, Ekici M, Schmitt G, Madry H. Human mesenchymal stem cells overexpressing therapeutic genes: From basic science to clinical applications for articular cartilage repair. Biomed Mater Eng. 2012;22(4):197–208.
- Centeno CJ, Pastoriza SM. PAST, CURRENT AND FUTURE INTERVENTIONAL ORTHOBIOLOGICS TECHNIQUES AND HOW THEY RELATE TO REGENERATIVE REHABILITATION: A CLINICAL COMMENTARY. Int J Sports Phys Ther. 2020 Apr;15(2):301–25.
- Lana JF, da Fonseca LF, Azzini G, Santos G, Braga M, Cardoso Junior AM, et al. Bone Marrow Aspirate Matrix: A Convenient Ally in Regenerative Medicine. Int J Mol Sci. 2021 Mar 9;22(5):2762.
- Liu C, Xiao K, Xie L. Advances in the Regulation of Macrophage Polarization by Mesenchymal Stem Cells and Implications for ALI/ARDS Treatment. Front Immunol. 2022 Jul 8;13:928134.
- Lee S, Chae DS, Song BW, Lim S, Kim SW, Kim IK, et al. ADSC-Based Cell Therapies for Musculoskeletal Disorders: A Review of Recent Clinical Trials. Int J Mol Sci. 2021 Sep 30;22(19):10586.
- Caplan, AI. New MSC: MSCs as pericytes are Sentinels and gatekeepers: MSCs, PERICYTES, METASTASIS, REGENERATIVE MEDICINE. J Orthop Res. 2017 Jun;35(6):1151–9.
- Wu Y, Fu J, Huang Y, Duan R, Zhang W, Wang C, et al. Biology and function of pericytes in the vascular microcirculation. Anim Models Exp Med. 2023 Aug;6(4):337–45.
- Dias Moura Prazeres PH, Sena IFG, Borges IDT, De Azevedo PO, Andreotti JP, De Paiva AE, et al. Pericytes are heterogeneous in their origin within the same tissue. Dev Biol. 2017 Jul;427(1):6–11.
- Dimmeler S, Zeiher AM. Endothelial Cell Apoptosis in Angiogenesis and Vessel Regression.
- Festa J, AlZaim I, Kalucka J. Adipose tissue endothelial cells: insights into their heterogeneity and functional diversity. Curr Opin Genet Dev. 2023 Aug;81:102055.
- Kakudo N, Minakata T, Mitsui T, Kushida S, Notodihardjo FZ, Kusumoto K. Proliferation-Promoting Effect of Platelet-Rich Plasma on Human Adipose–Derived Stem Cells and Human Dermal Fibroblasts: Plast Reconstr Surg. 2008 Nov;122(5):1352–60.
- Guo J, Nguyen A, Banyard DA, Fadavi D, Toranto JD, Wirth GA, et al. Stromal vascular fraction: A regenerative reality? Part 2: Mechanisms of regenerative action. J Plast Reconstr Aesthet Surg. 2016 Feb;69(2):180–8.
- Man K, Kallies A, Vasanthakumar A. Resident and migratory adipose immune cells control systemic metabolism and thermogenesis. Cell Mol Immunol. 2022 Mar;19(3):421–31.
- Cipolletta D, Feuerer M, Li A, Kamei N, Lee J, Shoelson SE, et al. PPAR-γ is a major driver of the accumulation and phenotype of adipose tissue Treg cells. Nature. 2012 Jun;486(7404):549–53.
- Lynch L, Michelet X, Zhang S, Brennan PJ, Moseman A, Lester C, et al. Regulatory iNKT cells lack expression of the transcription factor PLZF and control the homeostasis of Treg cells and macrophages in adipose tissue. Nat Immunol. 2015 Jan;16(1):85–95.
- Davies LC, Jenkins SJ, Allen JE, Taylor PR. Tissue-resident macrophages. Nat Immunol. 2013 Oct;14(10):986–95.
- Lacy P, Stow JL. Cytokine release from innate immune cells: association with diverse membrane trafficking pathways. Blood. 2011 Jul 7;118(1):9–18.
- Smith OJ, Jell G, Mosahebi A. The use of fat grafting and platelet-rich plasma for wound healing: A review of the current evidence. Int Wound J. 2019 Feb;16(1):275–85.
- Tatsis D, Vasalou V, Kotidis E, Anestiadou E, Grivas I, Cheva A, et al. The Combined Use of Platelet-Rich Plasma and Adipose-Derived Mesenchymal Stem Cells Promotes Healing. A Review of Experimental Models and Future Perspectives. Biomolecules. 2021 Sep 24;11(10):1403.
- Dallo I, Morales M, Gobbi A. Platelets and Adipose Stroma Combined for the Treatment of the Arthritic Knee. Arthrosc Tech. 2021 Nov;10(11):e2407–14.
- Eto H, Suga H, Inoue K, Aoi N, Kato H, Araki J, et al. Adipose Injury–Associated Factors Mitigate Hypoxia in Ischemic Tissues through Activation of Adipose-Derived Stem/Progenitor/Stromal Cells and Induction of Angiogenesis. Am J Pathol. 2011 May;178(5):2322–32.
- Mashiko T, Yoshimura K. How Does Fat Survive and Remodel After Grafting? Clin Plast Surg. 2015 Apr;42(2):181–90.
- Yu W, Wang Z, Dai Y, Zhao S, Chen H, Wang S, et al. Autologous fat grafting for postoperative breast reconstruction: A systemic review. Regen Ther. 2024 Jun;26:1010–7.
- Ding P, Lu E, Li G, Sun Y, Yang W, Zhao Z. Research Progress on Preparation, Mechanism, and Clinical Application of Nanofat. J Burn Care Res. 2022 Sep 1;43(5):1140–4.
- Hersant B, Sid-Ahmed M, Braud L, Jourdan M, Baba-Amer Y, Meningaud JP, et al. Platelet-Rich Plasma Improves the Wound Healing Potential of Mesenchymal Stem Cells through Paracrine and Metabolism Alterations. Stem Cells Int. 2019 Oct 31;2019:1–14.
- Chen W, He Z, Li S, Wu Z, Tan J, Yang W, et al. The Effect of Tissue Stromal Vascular Fraction as Compared to Cellular Stromal Vascular Fraction to Treat Anal Sphincter Incontinence. Bioengineering. 2022 Dec 26;10(1):32.
- Blanton MW, Hadad I, Johnstone BH, Mund JA, Rogers PI, Eppley BL, et al. Adipose Stromal Cells and Platelet-Rich Plasma Therapies Synergistically Increase Revascularization during Wound Healing: Plast Reconstr Surg. 2009 Feb;123(Supplement):56S-64S.
- Siegel KR, Clevenger TN, Clegg DO, Proctor DA, Proctor CS. Adipose Stem Cells Incorporated in Fibrin Clot Modulate Expression of Growth Factors. Arthrosc J Arthrosc Relat Surg. 2018 Feb;34(2):581–91.
- Everts PA, Lana JF, Alexander RW, Dallo I, Kon E, Ambach MA, et al. Profound Properties of Protein-Rich, Platelet-Rich Plasma Matrices as Novel, Multi-Purpose Biological Platforms in Tissue Repair, Regeneration, and Wound Healing. Int J Mol Sci. 2024 Jul 19;25(14):7914.
- Trepat X, Chen Z, Jacobson K. Cell Migration. In: Prakash YS, editor. Comprehensive Physiology [Internet]. 1st ed. Wiley; 2012 [cited 2025 Jan 29]. p. 2369–92. Available online: https://onlinelibrary.wiley.com/doi/10.1002/cphy.c110012.
- Merino-Casallo F, Gomez-Benito MJ, Hervas-Raluy S, Garcia-Aznar JM. Unravelling cell migration: defining movement from the cell surface. Cell Adhes Migr. 2022 Dec 31;16(1):25–64.
- Berzat A, Hall A. Cellular responses to extracellular guidance cues. EMBO J. 2010 Aug 18;29(16):2734–45.
- Wagh K, Ishikawa M, Garcia DA, Stavreva DA, Upadhyaya A, Hager GL. Mechanical Regulation of Transcription: Recent Advances. Trends Cell Biol. 2021 Jun;31(6):457–72.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



