Introduction
Triple-negative breast cancer (TNBC) constitutes around 10-15% of total breast cancer cases[
1]. It is named for its histological presentation by the lack of favorable staining for estrogen and progesterone receptors, along with the absence of HER2 amplification[
2]. These tumors are highly aggressive, displaying substantial genomic instability, and are associated with an unfavorable prognosis and early visceral metastasis[
3]. The survival rates for women experiencing relapse within five years of treatment are notably lower compared to those with breast cancer positive for one or two hormone receptors[
2,
3].
Currently, there are no targeted therapies for triple-negative breast cancer (TNBC). Radiation therapy and chemotherapy have been the main treatment options for locally advanced and metastatic TNBC. However, the effectiveness of radiotherapy or chemotherapeutic strategies has been hindered by inherent or acquired treatment resistance in TNBC[
4]. This emphasizes the need for more sensitive and specific therapeutic approaches for this challenging breast cancer subtype[
5].
Aldehyde dehydrogenases (ALDH) are a family of enzymes that catalyze the oxidation of aldehydes to carboxylic acids, playing a crucial role in detoxification, biosynthesis, and various other biological processes[
6].. Many ALDH isoforms have been identified in solid tumors underscoring their relevance in tumor progression and therapy resistance[
7]. Among the ALDH isoforms present in breast cancer, members of the ALDH1 family, including ALDH1A1 and ALDH1A3, contribute to cancer stemness, progression and resistance[
7,
8,
9].
ALDH1A1 is particularly significant for its association with treatment resistance and poor outcomes in TNBC[
3,
10,
11,
12,
13,
14]. ALDH1A1 expression in breast cancer is correlated estrogen receptor negativity, high grade, tumor size and stage, lymph node metastasis, and drug resistance [
3,
12]. ALDH1A1 is best known as a marker of breast cancer stem cells[
10], primarily through catalyzing the oxidation of retinaldehyde to retinoic acid—a key regulator of cellular differentiation[
8]. ALDH1A1 and ALDH1A3 both drive retinoic acid production in breast cancer, and have been linked to angiogenesis, tumor invasion and metastasis [
8,
15]. Additionally, ALDH1A1 plays a direct role in treatment resistance through metabolism of reactive aldehydes[
8,
9].
ALDH1A1 contributes to chemoresistance by modulating cellular metabolism and regulating reactive oxygen species (ROS)[
9,
16,
17,
18,
19,
20]. Its expression is upregulated in response to oxidative stress, serving as a protective mechanism to enhance ROS detoxification during aldehyde metabolism and maintain redox balance in both normal and malignant tissues[
20] [
21,
22,
23]. By detoxifying aldehyde byproducts of lipid peroxidation, such as 4-hydroxynonenal, ALDH1A1 shields cells from oxidative damage induced by chemotherapy. [
24]. However, ALDH1A1's role in mitigating these effects enhances chemoresistance, protecting cells under therapeutic stress[
8,
19,
25] .
Although ALDH1A1 has been reported to induce radiation resistance in solid tumors including breast cancer[
8,
26], the exact mechanism is not clear. Our group has demonstrated that ALDH1A1 activity and expression increase in a dose-dependent manner following ionizing radiation, suggesting a role in managing radiation-induced redox stress[
27] . Based on these findings, we hypothesize that targeting ALDH1A1 could disrupt cellular redox balance and sensitize TNBC cells to radiation therapy. To investigate this, we generated genetic knockouts of ALDH1A1 to further explore its role in redox stress and radiation resistance.
Materials and Methods
CRISPR/Cas9 Targeting
To generate ALDH1A1 knockout cell lines, we chose three single guide RNAs (sgRNAs 2, 3, and 4) from the Synthego website targeting exon 2 of the ALDH1A1 gene, each containing the requisite NGG PAM sequence. The gRNAs with the lowest off target effect according to a prescreen done by Synthego was chosen (Table 1 and 2). These guide RNAs were then complexed with Cas9 endonuclease to form ribonucleoprotein (RNP) complexes. SUM159 cells were cultured under standard conditions until 70-80% confluence, after which they were subjected to nucleofection with the RNP complexes to facilitate nuclear entry using the Lonza 4D nucleofector. Following a 72-hour recovery period, initial validation of ALDH1A1 knockout efficiency was performed via Western blot analysis. The most effective guide, as determined by protein knockdown levels, was selected for subsequent experiments. The bulk population of targeted cells was then sorted into 96-well plates using fluorescence-activated cell sorting (FACS). Individual clones were screened and expanded under standard culture conditions, followed by comprehensive validation of ALDH1A1 knockout at the DNA and protein levels. We selected 2-3 clones exhibiting complete ALDH1A1 knockout for further experimentation. These validated knockout clones were then preserved for future use. Throughout the process, appropriate controls were maintained, and all experimental parameters were thoroughly documented to ensure reproducibility.
Western Blot Analysis
SUM159PT cells and the clonally derived cell lines were treated and incubated as indicated. When cells reached 70-80% confluency they were harvested in RIPA buffer (Sigma E-Life sciences SLBG8489) with phosphatase and protease inhibitors EDTA-free (ThermoFisher Scientific, USA). BCA assays were performed to determine protein concentration using Pierce BCA protein assay kit (ThermoFisher). Western blot was performed using 10% ten-well comb precast gel from Biorad (Bio-Rad Mini protean TGX gel 4561033). Gels were transferred to membranes (LI-COR biosciences) The membranes were blocked in 3% BSA in TBS-T and blotted for ALDH1A1 primary antibody at room temperature (RT). The following antibodies were used. ALDH1A1 Rabbit monoclonal antibody (Cell signaling technology. ECL western blotting detection reagent (protein biology, thermoscientific.com/protein) was used to detect signals. Images were acquired and densitometry was performed using a LI-COR imager.
Sanger Sequencing
DNA was extracted from SUM159 parent cells and the clonally derived cell lines using DNeasy blood and tissue kit (Qiagen, Germany) DNA concentrations were checked using mass spectrometry (Thermo Scientific, Nanodrop one) and DNA was diluted to 25ng/uL or less. Primers were obtained from Integrated DNA technologies (Primer sequence in Table) and then a gradient PCR was run to obtain the optimum annealing temperature of the primers. After this amplicon PCR was done and the PCR clean up with the QIAquick PCR purification kit (250) (Qiagen, Germany) according to the recommended protocol by the company. Agarose gel electrophoresis. Samples were then sent to Azenta GeneWiz for Sanger sequencing. Sanger sequencing data was analyzed using ICE software, DECODR, and benchling.
Aldefluor Assay.
Aldefluor assay was done according to the manufacturers protocol as described in [
27]. DEAB gates were set to 0.2%. Assay was done in three biological replicates
Apoptosis Assay (Annexin v Pi)
Cells were plated at a low density of 150,000 cells per well in a 6-well plate. Cells were left for 72 hours and then trypsinized, washed in 1X PBS, and incubated with 1X annexin-binding buffer, Alexa fluor 488 Annexin V component, and propidium iodide solution according to the manufacturer's protocol. (Invitrogen by ThermoScientific) results were collected by flow cytometry. For radiation response one million cells were plated in a 100mm dish and allowed to attach and treated with 8Gy radiation. The cells were then replated at 150,00 cells per well and the assay was performed as described above.
Live/Dead Assay (Viability/Cytotoxicity Kit)
The Invitrogen Live/Dead viability/cytotoxicity kit was used according to the manufacturer’s instructions, including Calcein AM visibility dye cat no :65-0853-39 and EthD-1 dye cat no: E1169. Cells were plated at a density of 500 cells per well in a 96-well black clear bottom plate and allowed to grow for 5 days, it was stained with Calcien AM and Ethidium homodimer-1 and read on a plate reader (Tecan) at their respective fluorescence wavelengths. Alternatively, 1000 cells were used on cover slips and stained. Images were captured using a Zeiss Apotome 2 microscope using Zen Blue software, (Zeiss)
Reactive Oxygen Species (DCFDA/H2DCFDA)
2',7'-dichlorofluorescein diacetate (DCFDA) Abcam kit (ab113851) was used to detect reactive oxygen species (ROS). 250,000 cells were plated and allowed to attach. The next day we incubated 10um of DCFDA for 30 minutes and 100um of TBHP as positive control. The ROS levels are then measured by flow cytometry.
Alamar Blue Viability Assay
1000 cells were plated in 96 well plates and incubated with alamar blue (Invitrogen Catalog number: DAL1025). This was read at the Nanoquant Infinite Pro plate reader on days one and five. At an absorbance or 570nm
Lentiviral Transduction
Lentiviral overexpression of ALDH1A1 was used to rescue ALDH1A1 in the knockout cell lines. The lentiviral construct was made by the ALDH1A1 gene under the control of a CMV promoter. This construct also includes Avi and flag tags for protein detection, an SV40 element, and a puromycin resistance gene for selection. The cells of each cell line were seeded and incubated with the lentivirus for 72 hours to allow for viral entry and integration into the host genome. Successfully transduced cells were selected using puromycin for 1 week at 10ug/ul. The overexpression of ALDH1A1 was verified by western blotting.
Statistical Analysis
Statistical comparisons between two groups (e.g., parent and bulk) were analyzed using Welch's t-test. For all other analyses involving multiple groups, one-way ANOVA was performed with subsequent Dunnett's post-hoc test. All statistical analyses were conducted using GraphPad Prism. version 10.3.0 (507)
Results:
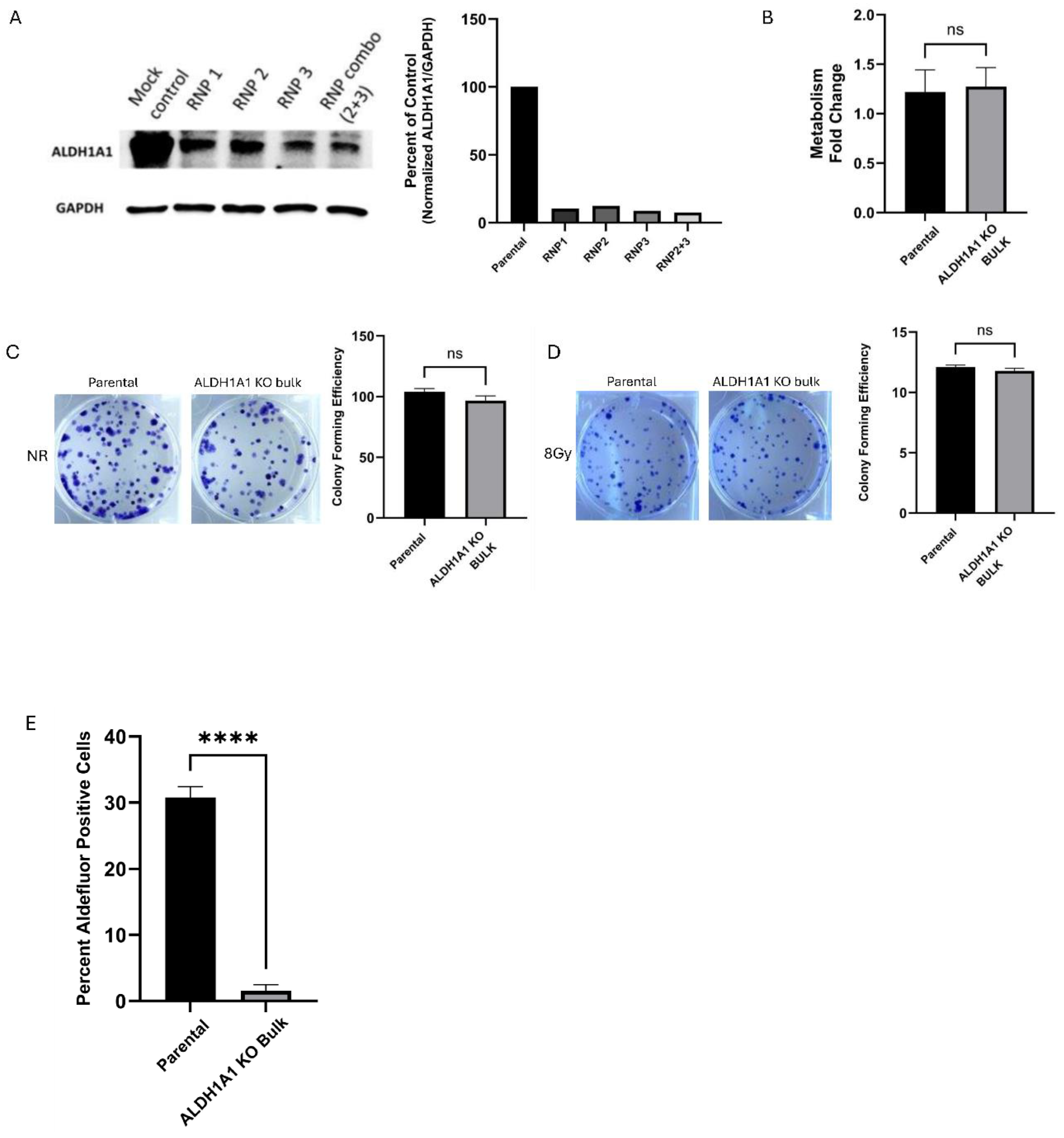
Effect of CRISPR/Cas9 Knock Out (KO) of ALDH1A1 SUM159 Bulk Cells
To investigate the role of ALDH1A1 in radiation resistance, we used CRISPR/Cas9 to generate genetic knock out (KO) of ALDH1A1 in the triple-negative breast cancer (TNBC) cell line SUM159. This cell line was chosen due to its constitutive ALDH1A1 expression and documented ALDH1A1 upregulation following radiation exposure [
27]. Three guide RNAs (gRNAs) were designed to target exon 2 of ALDH1A1, which encodes the active domain critical for the ALDH1A1 enzyme's catalytic function (
Figure S1A). Targeting at exon 2 occurs prior to the substrate binding domain [
28]. These mutations result in a frameshift, disrupting the reading frame and impairing the production of a functional protein. These guides were complexed to Cas 9 as single guides or in pairs and the ribonuclear complexes (RNP1, RNP2 and RNP3 for single guides or RNP 2+3 for combination guides) were delivered to SUM159 cells via nucleofection.
We confirmed the knockdown of ALDH1A1 at the protein level (
Figure 1A) and at the genomic level in the bulk population of transfected cells. We confirmed that using a combination of two different gRNAs to form our ribonucleoprotein complex (RNP2+3) was more efficient than the targeting done with the single gRNAs and reduced ALDH1A1 expression by 92%. These cells were selected for further study (
Figure 1A).
To assess the functional impact of ALDH1A1 KO in the bulk SUM159 cells, we performed a metabolic assay to determine cell growth and viability. Cells were plated and cellular metabolism was measured on day 1 and day 5 using an Alamar Blue assay. No significant difference was observed in bulk ALDH1A1 knock out (ALDH1A1KO) cells compared to control cells (
Figure 1B). Additionally, colony formation assays were performed to evaluate reproductive integrity, revealing no significant difference in colony-forming ability between SUM159 parental and ALDH1A1KO bulk cells (
Figure 1C). We also tested colony-forming efficiency in response to 8Gy radiation. This dose was chosen as it was found to have the highest increase in ALDEFLUOR activity and ALDH1A1 expression after radiation in our previous study[
29]. As shown in
Figure 1D, no significant differences in radiation response were observed between SUM159 control and ALDH1A1KO bulk cells. We tested for Aldefluor activity in the ALDH1A1 ko bulk cell lines and we found that the loss of ALDH1A1 in the bulk cell lines reduced aldefluor activity significantly compared to the parent cell line (
Figure 1E, S1C)
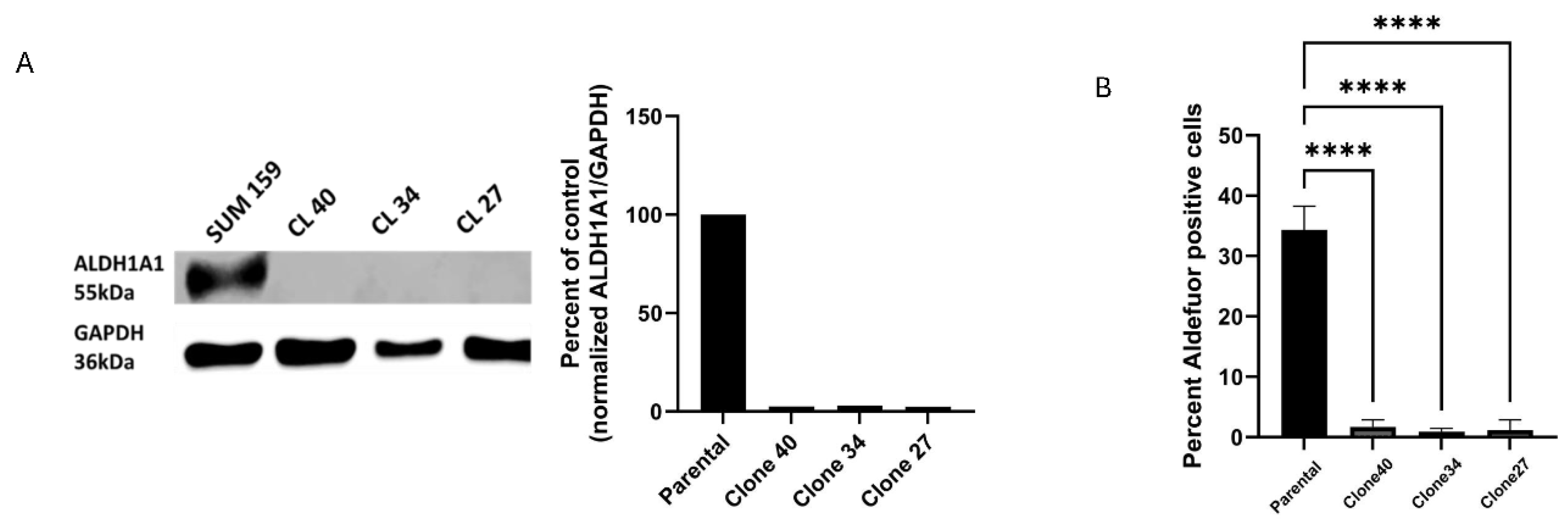
Clonal Cell Lines Derived from Bulk Knock-Out Cells Have Decreased ALDEFLUOR Activity.
The bulk population of targeted cells contains a mixture of cells with varying levels of ALDH1A1 knockout. To ensure a complete knock-down of ALDH1A1, we isolated single-cell clones from the ALDH1A1KO bulk population. Three clonal lines (clone 40, clone 34, and clone 27) were confirmed to be complete knockouts via western blotting (
Figure 2A) and Sanger sequencing (
Figure S1B). Analysis of ALDEFLUOR activity in all three clonal lines revealed significant decreases in ALDEFLUOR positivity compared to the parental line (
Figure 2B and
Figure S2). These results indicate a critical role for ALDH1A1 in ALDEFLUOR activity in the three knock-out lines.
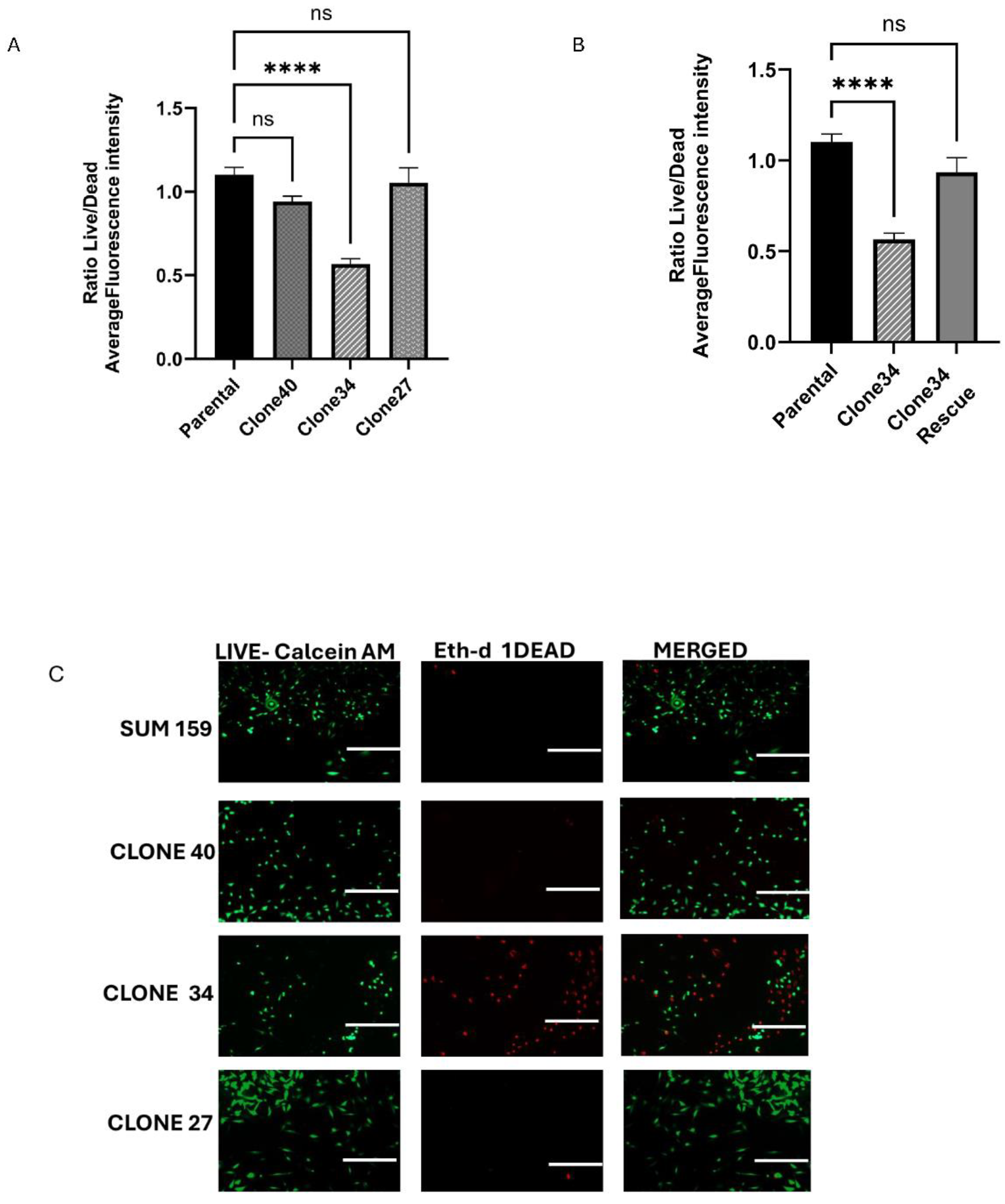
Clonal Differences in the Effect of ALDH1A1 Knockout on Viability, Apoptosis, and Reactive Oxygen Species
To investigate the effect of ALDH1A1 knockout on cell viability in clonal lines, we performed Calcein AM/ETHD-1 staining to detect live and dead cells. While no significant changes in viability were observed in Clones 27 and 40, Clone 34 exhibited a significantly lower number of live cells, and a higher number of dead cells compared to the parental SUM159 line (
Figure 4A,
Figure 4C). This reduction in viability in Clone 34 was rescued by overexpression of ALDH1A1 (
Figure 4B and
Figure S4A).
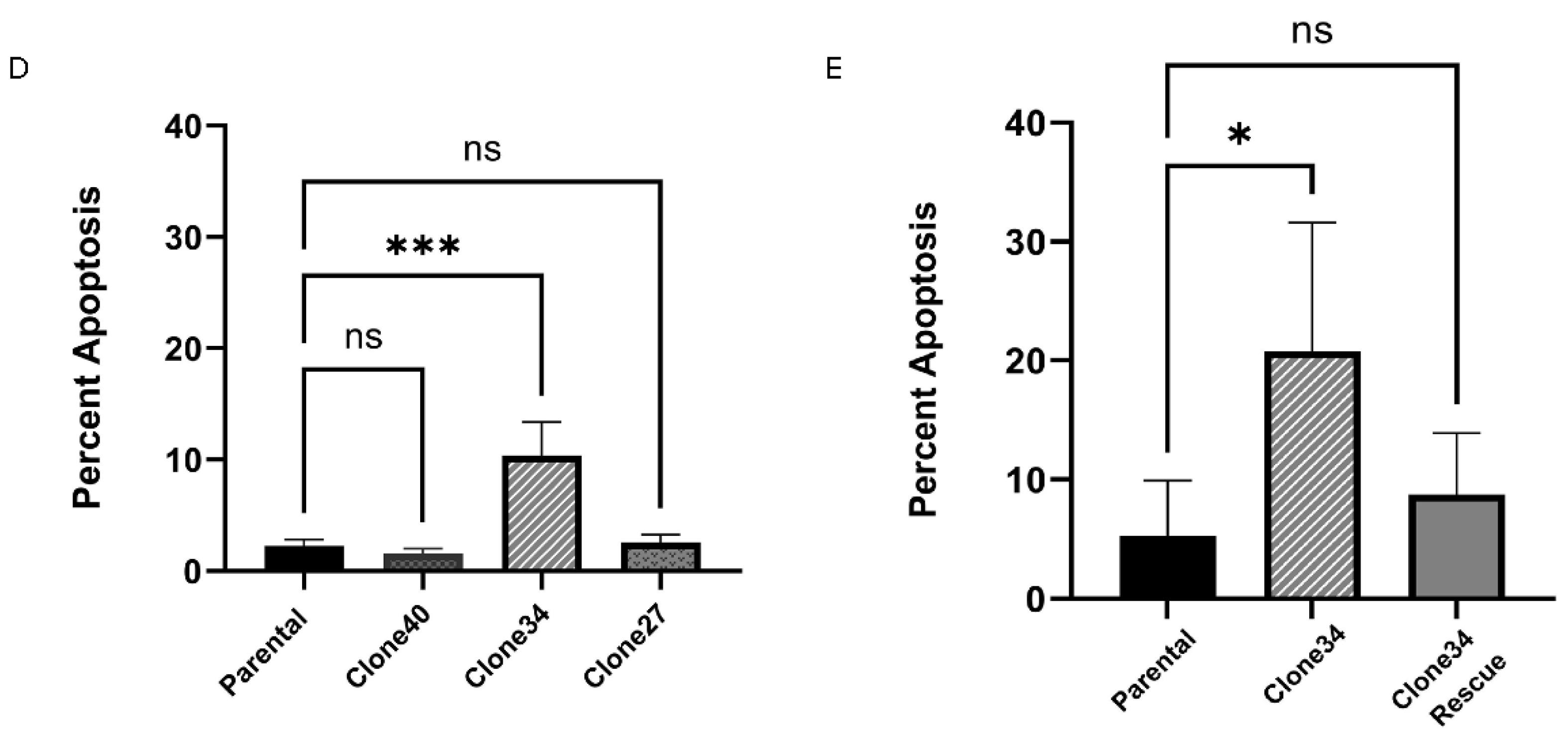
Furthermore, Annexin-V/PI staining revealed an increased percentage of apoptotic cells in Clone 34 compared to the parental line, which was similarly rescued by ALDH1A1 overexpression (
Figure 4D-E and
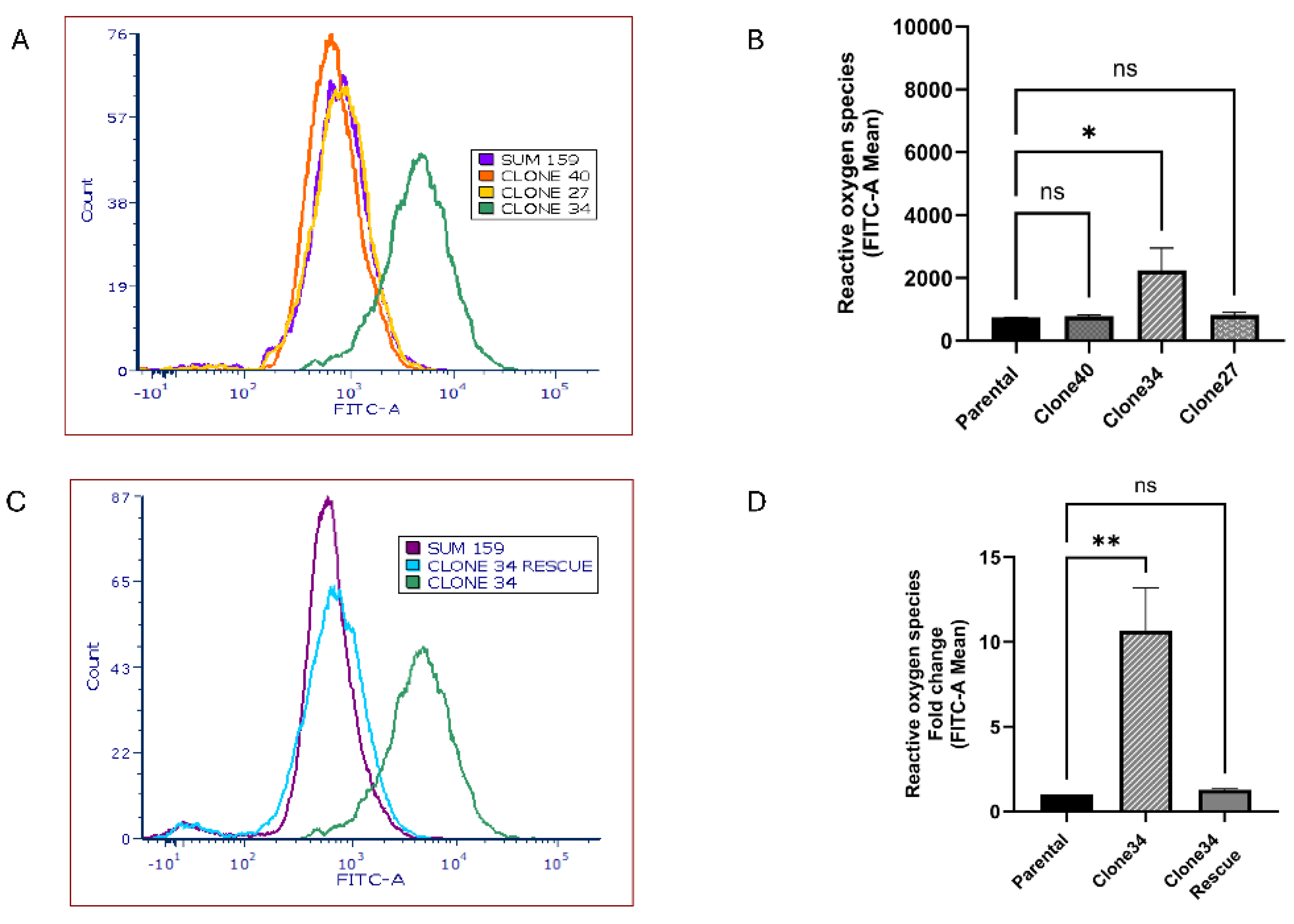
Figure S5). Given ALDH1A1’s role in ROS metabolism, we also measured ROS levels in the clonal lines. As shown in
Figure 5A-B, ROS levels were significantly elevated in Clone 34 compared to the parental line, and this effect was reversed by rescuing ALDH1A1 expression (
Figure 5C-D). These findings suggest that Clone 34 depends on ALDH1A1 for maintaining redox balance and cell viability, unlike other clonal lines.
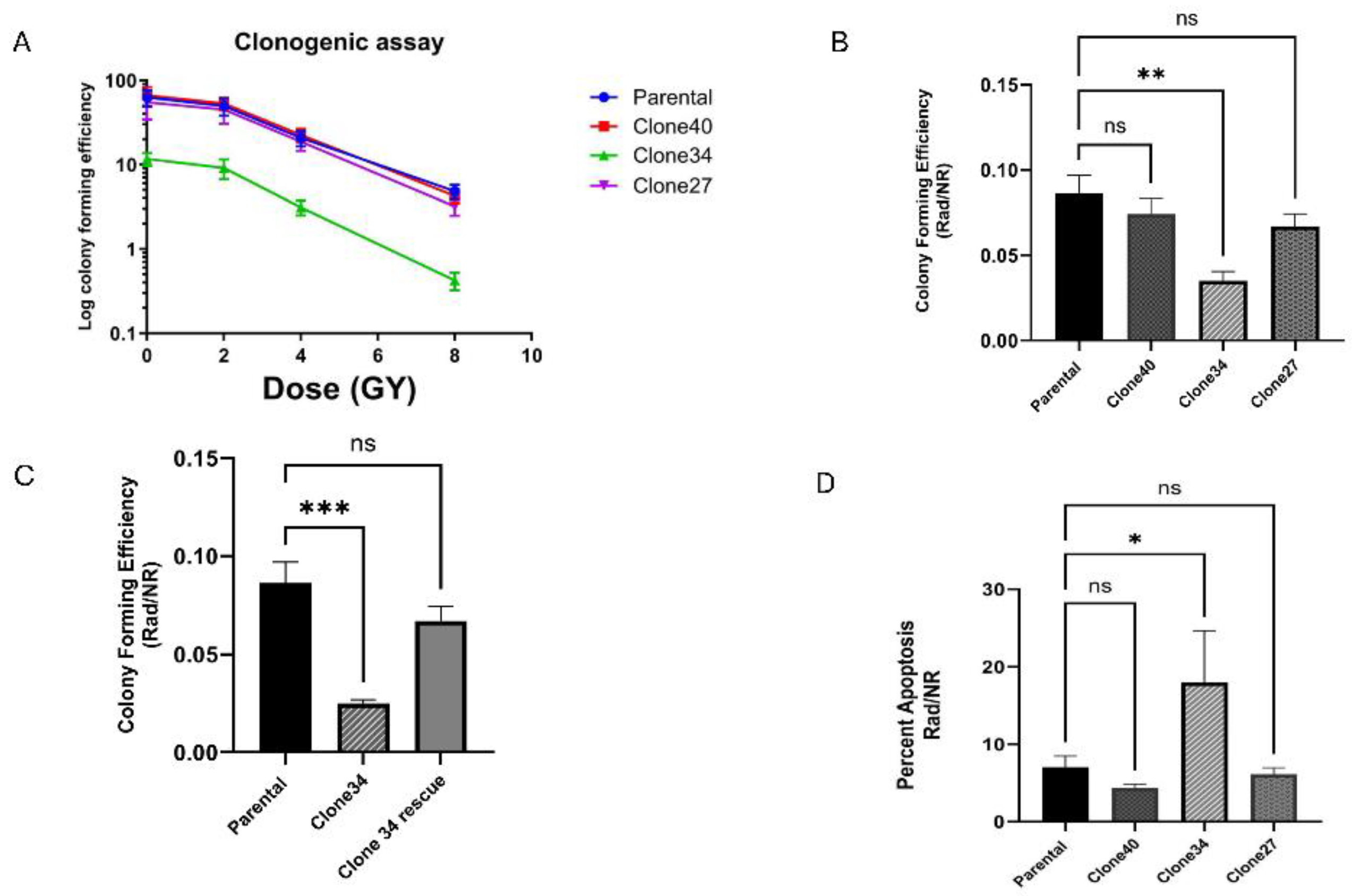
Radiation Sensitivity in ALDH1A1 Knockout Clones
To evaluate the effects of radiation on ALDH1A1 knockout clones, we performed clonogenic assays on the parental SUM159 line and knockout clones at radiation doses of 2, 4, and 8 Gy. Only Clone 34 showed a significant reduction in colony-forming ability upon radiation exposure (
Figure 6A and
Figure S6A-B), with the greatest reduction observed at 8 Gy. This effect persisted even after normalization to non-irradiated colony formation (
Figure 6B) and was attenuated upon rescue of ALDH1A1 expression in Clone 34 (
Figure 6C and S C). Clone 27, which exhibited reduced colony formation under non-irradiated conditions, showed no additional sensitization to radiation (
Figure 6B).
To further investigate the role of ALDH1A1 in radiation resistance, we assessed radiation-induced apoptosis. Following 8 Gy radiation, apoptosis levels were measured via flow cytometry 3 days post-treatment. Clone 34 exhibited a marked increase in apoptosis compared to other clones and the parental line, an effect that was rescued by ALDH1A1 expression (
Figure 6D and
Figure S6D). These findings suggest that ALDH1A1 is particularly critical for radiation resistance in Clone 34.
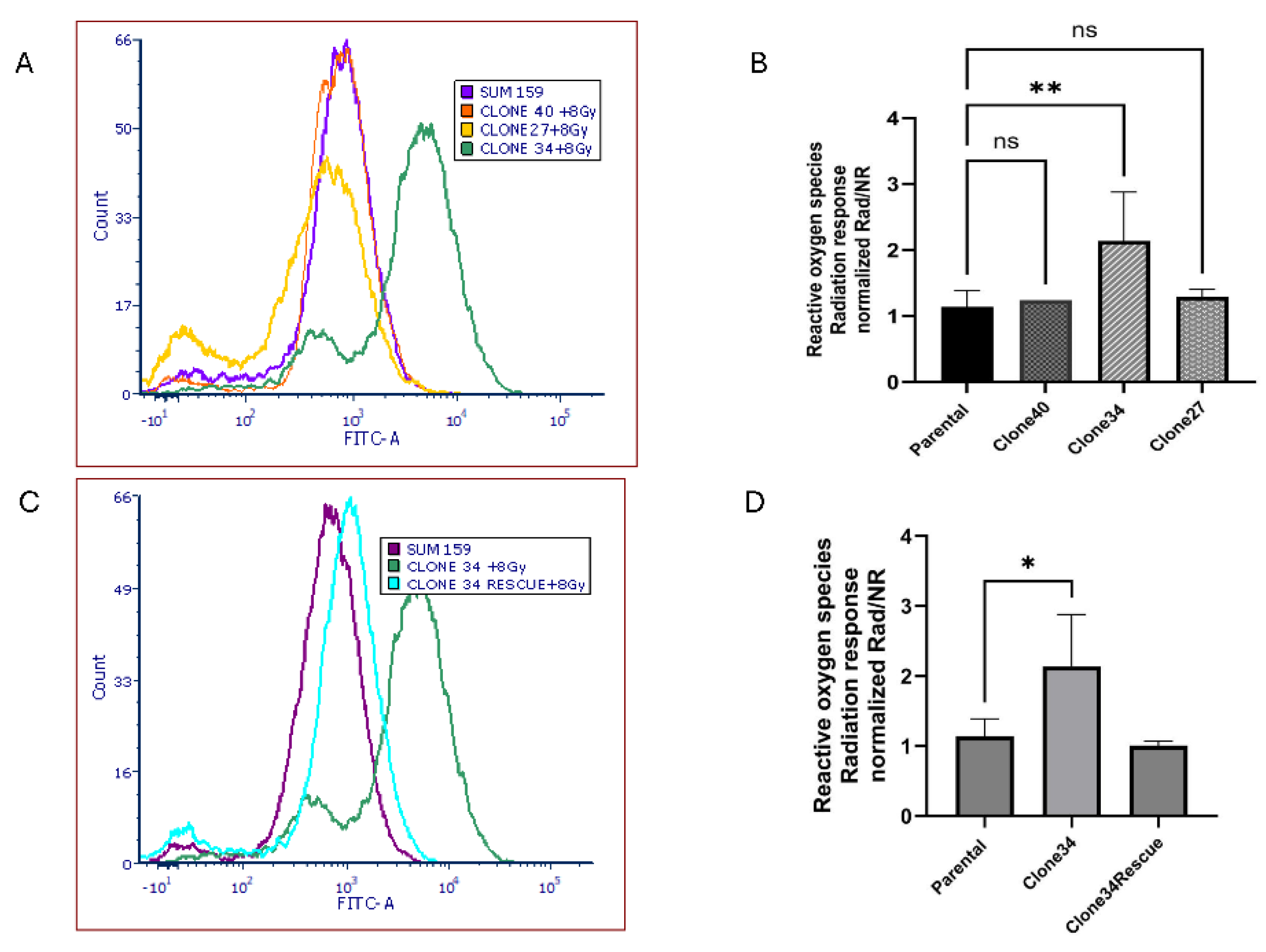
Radiation is known to increase ROS levels[
30], so we examined ROS accumulation in ALDH1A1 knockout clones after 8 Gy radiation. As shown in
Figure 7A-B, ROS levels significantly increased in Clone 34 upon radiation, even when normalized to pre-treatment levels. This effect was reversed by restoring ALDH1A1 expression (
Figure 7C-D). No significant changes in radiation-induced ROS levels or sensitivity were observed in other clonal lines (
Figure S7A-B).
Discussion
This study used CRISPR/Cas9-generated functional knockouts of ALDH1A1 in TNBC cells to investigate the role of this ALDH isoform in radiation resistance. We observed that the bulk population of ALDH1A1 KO cells did not exhibit significant changes in radiation response upon ALDH1A1 KO. However, analysis of clonal lines obtained from the bulk ALDH1A1 KO population revealed distinct variations upon loss of ALDH1A1. Notably, Clone 34 demonstrated a significant reduction in metabolic activity, colony-forming ability, and viability driven by increased reactive oxygen species (ROS) accumulation and apoptosis under normal culture conditions. These effects were amplified by radiation with significant increases in ROS and cell death after 8 Gy radiation. Reintroduction of ALDH1A1 expression by viral transfection rescued these effects, confirming that they were specifically related to the loss of ALDH1A1.In contrast, other clones, Clones 27 and 40, did not exhibit significant changes upon loss of ALDH1A1, even after radiation, suggesting a heterogeneous dependency on ALDH1A1 among different clonal populations. These findings suggest that ALDH1A1 plays distinct roles in different subpopulations, particularly in relation to cellular metabolism and redox stress.
Interestingly, ALDEFLUOR activity, a correlate of retinal metabolism [
31,
32], was significantly reduced in the bulk knockout population as well as all three clonal populations upon ALDH1A1 knockdown. This finding indicates that while ALDH1A1 may play a universal role in retinal metabolism across tumor cells, its function in managing redox stress may be context-dependent, varying based on the cellular state or genetic background.
Our finding that loss of ALDH1A1 significantly reduces ALDEFLUOR activity contrasts with a prior study that attributed ALDEFLUOR activity primarily to the ALDH1A3 isoform [
8]. Notably, this study employed knockdown approaches that left residual ALDH1A1 expression. However, while ALDEFLUOR activity is correlated with retinoic acid production, further research is needed to elucidate the roles and potential interactions of ALDH isoforms in retinal metabolism.
Radiation is known to induce cell death by increasing oxidative stress [
33]. Our study is the first to demonstrate that the loss of ALDH1A1 leads to enhanced radiation-induced cell death through increased ROS accumulation in a TNBC clonal line. This finding is consistent with studies in prostate cancer which have reported increased ROS in response to radiation upon inhibition of ALDH family activity with the competitive inhibitor DEAB [
34]. Likewise, a previous study reported increased radiation-induced death in bulk SUM159 cells upon partial loss of ALDH1A1 siRNA knockdown. This study utilized fractionated radiation, resulting in significantly higher cumulative doses compared to the single-dose approach in our study [
8]. While we did not see a radiation effect in all clonal lines, it is possible that compensatory mechanisms for ALDH1A1 loss exist in some populations at lower radiation doses but may be lost at higher cumulative doses observed with fractionated radiation.
Our group and others have shown that ALDH1-positive cells are resistant to radiation in TNBC and that ALDEFLUOR activity is upregulated in both ALDH-positive and negative cells post-irradiation
3,26. We previously observed a dose-dependent upregulation of ALDEFLUOR activity correlating with increased expression of both ALDH1A1 and ALDH1A3 [
27]. In prostate cancer, knockdown of both ALDH1A1 and ALDH1A3 enhanced radio sensitization, but only ALDH1A1 knockdown impaired DNA repair, suggesting a unique role for ALDH1A1in in maintaining cellular homeostasis upon radiation [
26]. Although our findings are consistent with this study, we did not investigate the role of ALDH1A3, and it remains unclear if ALDH1A3 can compensate for ALDH1A1 loss in TNBC.
Our study indicates that ALDH1A1 primarily promotes radiation resistance in a distinct subpopulation of cells. We observed that one clonal line relies on ALDH1A1 activity, both under normal conditions and following radiation exposure, as evidenced by reduced colony formation, increased ROS accumulation, and heightened apoptosis in the absence of ALDH1A1. Cancer stem-like cells have been reported to have lower ROS [
35], and previous research has established that ALDH1A1 is highly expressed in cancer stem-like cells and plays a critical role in mitigation of redox stress in these cells [
19,
36,
37,
38,
39]. We have previously shown that cancer stem cells are more sensitive to inhibition of STAT3-driven ALDH activity upon radiation than their non-stem cell components [
27]. However, the phenotypes of our clonal populations remain uncharacterized. Studies are underway to profile these clones and to expand functional knockout studies to include other TNBC cell lines and patient-derived samples to understand ALDH1A1 function across different tumor microenvironments and genetic backgrounds.
In conclusion, our study shows the diversity of ALDH1A1’s role in redox balance and resistance to radiation-induced stress among TNBC clonal populations. The variable responses among TNBC subpopulations to loss of ALDH1A1 highlight the complexity of its role in TNBC and have implications for the development of ALDH1A1 drug-targeting strategies.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on
Preprints.org.
Financial support
This work was supported by a grant from the Lisa Dean Moseley Foundation, and the Delaware INBRE (IDeA Networks of Biomedical Research Excellence) program, with a grant from the National Institute of General Medical Sciences – NIGMS (P20 GM103446) from the National Institutes of Health and the State of Delaware.
Conflicts of interest
The authors declare no conflicts of interest.
References
- Du, X.L.; Li, Z. Incidence trends in triple-negative breast cancer among women in the United States from 2010 to 2019 by race/ethnicity, age and tumor stage. Am J Cancer Res 2023, 13, 678–691. [Google Scholar]
- Foulkes, W.D.; Smith, I.E.; Reis-Filho, J.S. Triple-negative breast cancer. N Engl J Med 2010, 363, 1938–1948. [Google Scholar] [CrossRef]
- Althobiti, M.; El Ansari, R.; Aleskandarany, M.; Joseph, C.; Toss, M.S.; Green, A.R.; Rakha, E.A. The prognostic significance of ALDH1A1 expression in early invasive breast cancer. Histopathology 2020, 77, 437–448. [Google Scholar] [CrossRef] [PubMed]
- Echeverria, G.V.; Ge, Z.; Seth, S.; Zhang, X.; Jeter-Jones, S.; Zhou, X.; Cai, S.; Tu, Y.; McCoy, A.; Peoples, M.; et al. Resistance to neoadjuvant chemotherapy in triple-negative breast cancer mediated by a reversible drug-tolerant state. Sci Transl Med 2019, 11. [Google Scholar] [CrossRef]
- Chaudhuri, A.; Kumar, D.N.; Dehari, D.; Patil, R.; Singh, S.; Kumar, D.; Agrawal, A.K. Endorsement of TNBC Biomarkers in Precision Therapy by Nanotechnology. Cancers (Basel) 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Tomita, H.; Tanaka, K.; Tanaka, T.; Hara, A. Aldehyde dehydrogenase 1A1 in stem cells and cancer. Oncotarget 2016, 7, 11018–11032. [Google Scholar] [CrossRef] [PubMed]
- Marcato, P.; Dean, C.A.; Giacomantonio, C.A.; Lee, P.W. Aldehyde dehydrogenase: its role as a cancer stem cell marker comes down to the specific isoform. Cell Cycle 2011, 10, 1378–1384. [Google Scholar] [CrossRef]
- Croker, A.K.; Rodriguez-Torres, M.; Xia, Y.; Pardhan, S.; Leong, H.S.; Lewis, J.D.; Allan, A.L. Differential Functional Roles of ALDH1A1 and ALDH1A3 in Mediating Metastatic Behavior and Therapy Resistance of Human Breast Cancer Cells. Int J Mol Sci 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Olsson, M.; Larsson, P.; Johansson, J.; Sah, V.R.; Parris, T.Z. Cancer stem cells are prevalent in the basal-like 2 and mesenchymal triple-negative breast cancer subtypes in vitro. Front Cell Dev Biol 2023, 11, 1237673. [Google Scholar] [CrossRef]
- Ginestier, C.; Hur, M.H.; Charafe-Jauffret, E.; Monville, F.; Dutcher, J.; Brown, M.; Jacquemier, J.; Viens, P.; Kleer, C.G.; Liu, S.; et al. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell 2007, 1, 555–567. [Google Scholar] [CrossRef]
- Liu, Y.; Baglia, M.; Zheng, Y.; Blot, W.; Bao, P.P.; Cai, H.; Nechuta, S.; Zheng, W.; Cai, Q.; Shu, X.O. ALDH1A1 mRNA expression in association with prognosis of triple-negative breast cancer. Oncotarget 2015, 6, 41360–41369. [Google Scholar] [CrossRef] [PubMed]
- Ma, F.; Li, H.; Li, Y.; Ding, X.; Wang, H.; Fan, Y.; Lin, C.; Qian, H.; Xu, B. Aldehyde dehydrogenase 1 (ALDH1) expression is an independent prognostic factor in triple negative breast cancer (TNBC). Medicine (Baltimore) 2017, 96, e6561. [Google Scholar] [CrossRef] [PubMed]
- Maosa, S.F., L, Schug, Z., Siegel, S. Sims-Mourtada, J.. Strong Tumor Expression of ALDH1A1 is Associated with Black Race, Metabolic Disorders, and Poor Breast Cancer Outcomes. Cancer Health Disparities 2024, 7, 1-14.
- Panigoro, S.S.; Kurnia, D.; Kurnia, A.; Haryono, S.J.; Albar, Z.A. ALDH1 Cancer Stem Cell Marker as a Prognostic Factor in Triple-Negative Breast Cancer. Int J Surg Oncol 2020, 2020, 7863243. [Google Scholar] [CrossRef] [PubMed]
- Ciccone, V.; Terzuoli, E.; Donnini, S.; Giachetti, A.; Morbidelli, L.; Ziche, M. Stemness marker ALDH1A1 promotes tumor angiogenesis via retinoic acid/HIF-1alpha/VEGF signalling in MCF-7 breast cancer cells. J Exp Clin Cancer Res 2018, 37, 311. [Google Scholar] [CrossRef] [PubMed]
- Allison, S.E.; Chen, Y.; Petrovic, N.; Zhang, J.; Bourget, K.; Mackenzie, P.I.; Murray, M. Activation of ALDH1A1 in MDA-MB-468 breast cancer cells that over-express CYP2J2 protects against paclitaxel-dependent cell death mediated by reactive oxygen species. Biochem Pharmacol 2017, 143, 79–89. [Google Scholar] [CrossRef]
- Calleja, L.F.; Yoval-Sanchez, B.; Hernandez-Esquivel, L.; Gallardo-Perez, J.C.; Sosa-Garrocho, M.; Marin-Hernandez, A.; Jasso-Chavez, R.; Macias-Silva, M.; Salud Rodriguez-Zavala, J. Activation of ALDH1A1 by omeprazole reduces cell oxidative stress damage. FEBS J 2021, 288, 4064–4080. [Google Scholar] [CrossRef]
- Xiao, Z.; Ding, L.; Yu, Y.; Ma, C.; Lei, C.; Liu, Y.; Chang, X.; Chen, Y.; He, Y.; Zhu, Y.; et al. Tanreqing injection inhibits stemness and enhances sensitivity of non-small cell lung cancer models to gefitinib through ROS/STAT3 signaling pathway. J Cancer 2024, 15, 4259–4274. [Google Scholar] [CrossRef]
- Yue, H.; Hu, Z.; Hu, R.; Guo, Z.; Zheng, Y.; Wang, Y.; Zhou, Y. ALDH1A1 in Cancers: Bidirectional Function, Drug Resistance, and Regulatory Mechanism. Front Oncol 2022, 12, 918778. [Google Scholar] [CrossRef]
- Granit Mizrahi, A.; Gugenheim, A.; Hamad, H.; Hamed, R.; Tetro, N.; Maimon, O.; Khutsurauli, S.; Nechushtan, H.; Nisman, B.; Duran, D.; et al. Valproic acid reprograms the metabolic aberration of cisplatin treatment via ALDH modulation in triple-negative breast cancer cells. Front Cell Dev Biol 2023, 11, 1217149. [Google Scholar] [CrossRef] [PubMed]
- Marchitti, S.A.; Chen, Y.; Thompson, D.C.; Vasiliou, V. Ultraviolet radiation: cellular antioxidant response and the role of ocular aldehyde dehydrogenase enzymes. Eye Contact Lens 2011, 37, 206–213. [Google Scholar] [CrossRef]
- Zhou, J.; Sun, C.; Yang, L.; Wang, J.; Jn-Simon, N.; Zhou, C.; Bryant, A.; Cao, Q.; Li, C.; Petersen, B.; et al. Liver regeneration and ethanol detoxification: A new link in YAP regulation of ALDH1A1 during alcohol-related hepatocyte damage. FASEB J 2022, 36, e22224. [Google Scholar] [CrossRef] [PubMed]
- Ahmed Laskar, A.; Younus, H. Aldehyde toxicity and metabolism: the role of aldehyde dehydrogenases in detoxification, drug resistance and carcinogenesis. Drug Metab Rev 2019, 51, 42–64. [Google Scholar] [CrossRef]
- Jiang, H.; Zuo, J.; Li, B.; Chen, R.; Luo, K.; Xiang, X.; Lu, S.; Huang, C.; Liu, L.; Tang, J.; et al. Drug-induced oxidative stress in cancer treatments: Angel or devil? Redox Biol 2023, 63, 102754. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Shoeb, M.; Goswamy, J.; Liu, P.; Xiao, T.L.; Hogan, D.; Campbell, G.A.; Ansari, N.H. Overexpression of aldehyde dehydrogenase 1A1 reduces oxidation-induced toxicity in SH-SY5Y neuroblastoma cells. J Neurosci Res 2010, 88, 686–694. [Google Scholar] [CrossRef]
- Gorodetska, I.; Offermann, A.; Puschel, J.; Lukiyanchuk, V.; Gaete, D.; Kurzyukova, A.; Freytag, V.; Haider, M.T.; Fjeldbo, C.S.; Di Gaetano, S.; et al. ALDH1A1 drives prostate cancer metastases and radioresistance by interplay with AR- and RAR-dependent transcription. Theranostics 2024, 14, 714–737. [Google Scholar] [CrossRef] [PubMed]
- Arnold, K.M.; Opdenaker, L.M.; Flynn, N.J.; Appeah, D.K.; Sims-Mourtada, J. Radiation induces an inflammatory response that results in STAT3-dependent changes in cellular plasticity and radioresistance of breast cancer stem-like cells. Int J Radiat Biol 2020, 96, 434–447. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.J.; Wang, M.F.; Han, C.L.; Wang, S.L. Substrate binding pocket structure of human aldehyde dehydrogenases. A substrate specificity approach. Adv Exp Med Biol 1995, 372, 9–16. [Google Scholar] [CrossRef]
- Arnold, K.M.; Flynn, N.J.; Raben, A.; Romak, L.; Yu, Y.; Dicker, A.P.; Mourtada, F.; Sims-Mourtada, J. The Impact of Radiation on the Tumor Microenvironment: Effect of Dose and Fractionation Schedules. Cancer Growth Metastasis 2018, 11, 1179064418761639. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Luo, X.; Li, J.; Zhang, T.; Gao, X.; Hua, J.; Li, Y.; Ding, N.; He, J.; Zhang, Y.; et al. Ionizing Radiation Upregulates Glutamine Metabolism and Induces Cell Death via Accumulation of Reactive Oxygen Species. Oxid Med Cell Longev 2021, 2021, 5826932. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.J.; Cai, J.; Gao, L.; Yu, S.C. ALDEFLUOR activity, ALDH isoforms, and their clinical significance in cancers. J Enzyme Inhib Med Chem 2023, 38, 2166035. [Google Scholar] [CrossRef] [PubMed]
- Moreb, J.S.; Ucar-Bilyeu, D.A.; Khan, A. Use of retinoic acid/aldehyde dehydrogenase pathway as potential targeted therapy against cancer stem cells. Cancer Chemother Pharmacol 2017, 79, 295–301. [Google Scholar] [CrossRef]
- Azzam, E.I.; Jay-Gerin, J.P.; Pain, D. Ionizing radiation-induced metabolic oxidative stress and prolonged cell injury. Cancer Lett 2012, 327, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Cojoc, M.; Peitzsch, C.; Kurth, I.; Trautmann, F.; Kunz-Schughart, L.A.; Telegeev, G.D.; Stakhovsky, E.A.; Walker, J.R.; Simin, K.; Lyle, S.; et al. Aldehyde Dehydrogenase Is Regulated by beta-Catenin/TCF and Promotes Radioresistance in Prostate Cancer Progenitor Cells. Cancer Res 2015, 75, 1482–1494. [Google Scholar] [CrossRef]
- Shi, X.; Zhang, Y.; Zheng, J.; Pan, J. Reactive oxygen species in cancer stem cells. Antioxid Redox Signal 2012, 16, 1215–1228. [Google Scholar] [CrossRef] [PubMed]
- Bo, Y.; Zhou, J.; Cai, K.; Wang, Y.; Feng, Y.; Li, W.; Jiang, Y.; Kuo, S.H.; Roy, J.; Anorma, C.; et al. Leveraging intracellular ALDH1A1 activity for selective cancer stem-like cell labeling and targeted treatment via in vivo click reaction. Proc Natl Acad Sci U S A 2023, 120, e2302342120. [Google Scholar] [CrossRef]
- Cui, B.; Luo, Y.; Tian, P.; Peng, F.; Lu, J.; Yang, Y.; Su, Q.; Liu, B.; Yu, J.; Luo, X.; et al. Stress-induced epinephrine enhances lactate dehydrogenase A and promotes breast cancer stem-like cells. J Clin Invest 2019, 129, 1030–1046. [Google Scholar] [CrossRef] [PubMed]
- Anorma, C.; Hedhli, J.; Bearrood, T.E.; Pino, N.W.; Gardner, S.H.; Inaba, H.; Zhang, P.; Li, Y.; Feng, D.; Dibrell, S.E.; et al. Surveillance of Cancer Stem Cell Plasticity Using an Isoform-Selective Fluorescent Probe for Aldehyde Dehydrogenase 1A1. ACS Cent Sci 2018, 4, 1045–1055. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, C.H.; Zhang, Y.F.; Zhu, L.; Lei, H.M.; Tang, Y.B. UPLC-MS-based metabolomics reveals metabolic dysregulation in ALDH1A1-overexpressed lung adenocarcinoma cells. Metabolomics 2019, 15, 52. [Google Scholar] [CrossRef]
Figure 1.
ALDH1A1 Knockout in TNBC Bulk Cells Does Not Impact Metabolism, Colony Formation, or Radiation Response. (A) Western blot validation and quantification of ALDH1A1 KO in targeted CRISPR/Cas9 RNP targeted bulk cell populations. (B) Alamar blue assay measured cell metabolism in bulk ALDH1A1 knockout cell line. Fold change was expressed as a ratio of day 5/day 1. Knockout of ALDH1A1 showed no change in colony formation without (C) or with 8Gy radiation (D), as compared to the parental cell line. (E) ALDEFLUOR assay in ALDH1A1 KO bulk cells compared to the control cell line.Data represents the mean ± SEM normalized to respective control cells. ****p<0.0001, ns = not significant.
Figure 1.
ALDH1A1 Knockout in TNBC Bulk Cells Does Not Impact Metabolism, Colony Formation, or Radiation Response. (A) Western blot validation and quantification of ALDH1A1 KO in targeted CRISPR/Cas9 RNP targeted bulk cell populations. (B) Alamar blue assay measured cell metabolism in bulk ALDH1A1 knockout cell line. Fold change was expressed as a ratio of day 5/day 1. Knockout of ALDH1A1 showed no change in colony formation without (C) or with 8Gy radiation (D), as compared to the parental cell line. (E) ALDEFLUOR assay in ALDH1A1 KO bulk cells compared to the control cell line.Data represents the mean ± SEM normalized to respective control cells. ****p<0.0001, ns = not significant.
Figure 2.
Clonally derived ALDH1A1 knock cell lines have decreased ALDEFLUOR activity. (A) Western blotshows loss of ALDH1A1 protein expression in the clonally derived KO cell lines compared to the SUM159 parental cell line. (B) ALDEFLUOR assay shows a significant decrease in ALDEFLUOR activity in all three clonal lines compared to the parental cell line. Data represents the mean ± SEM normalized to respective control cells. ****p<0.0001, ***p< 0.005, **<0.005.
Figure 2.
Clonally derived ALDH1A1 knock cell lines have decreased ALDEFLUOR activity. (A) Western blotshows loss of ALDH1A1 protein expression in the clonally derived KO cell lines compared to the SUM159 parental cell line. (B) ALDEFLUOR assay shows a significant decrease in ALDEFLUOR activity in all three clonal lines compared to the parental cell line. Data represents the mean ± SEM normalized to respective control cells. ****p<0.0001, ***p< 0.005, **<0.005.
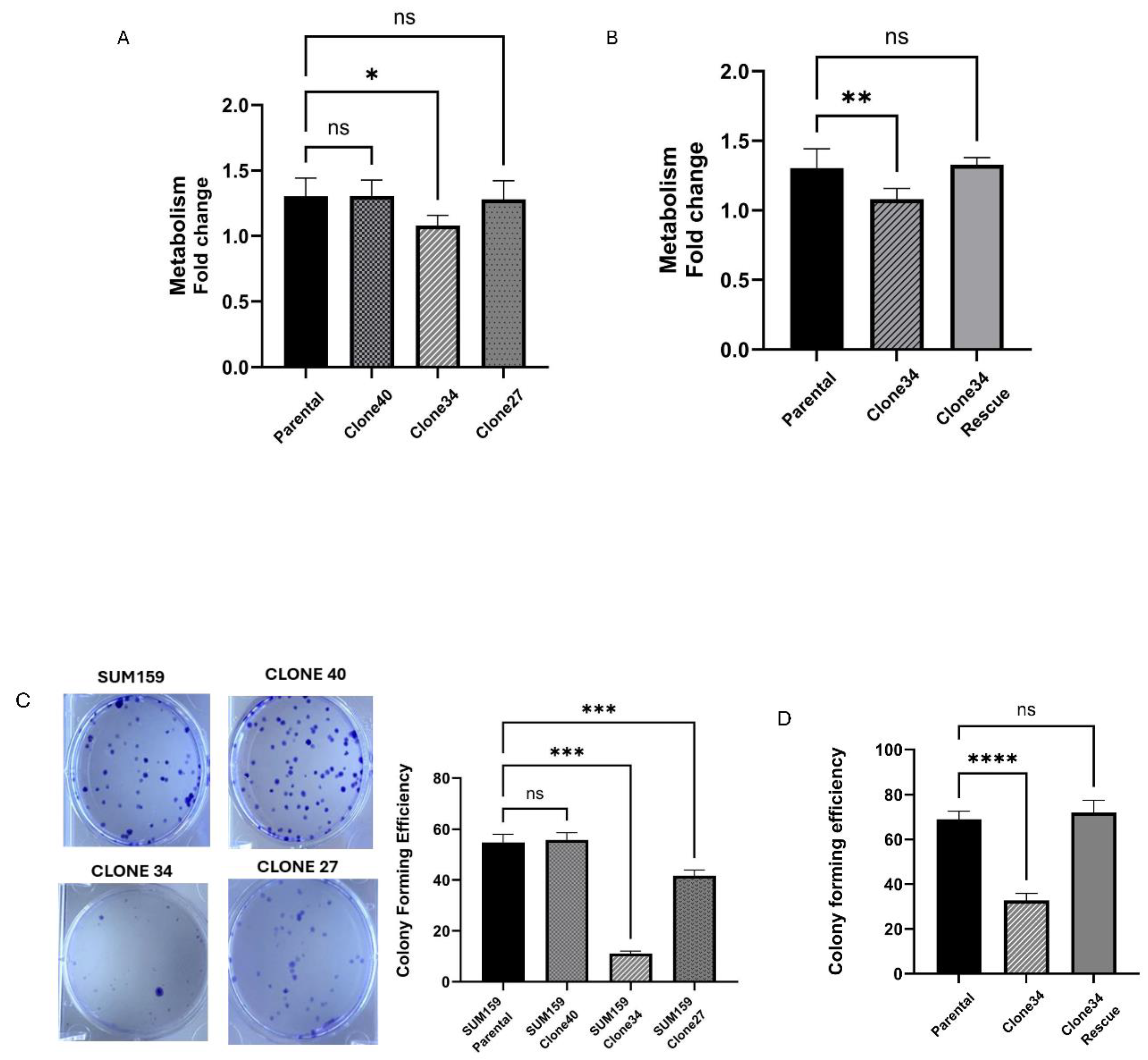
Figure 3.
Effect of ALDH1A1 loss on metabolism and survival. (A)Alamar blue assay measured cell metabolism in ALDH1A1 knockout clonal cell lines. Fold change was expressed as a ratio of day 5/day 1. (B) Rescue of ALDH1A1 expression restored cellular metabolism levels in clone 34. (C) Colony forming efficiency of SUM159 and ALDH1A1 knock out cell lines. The pictures are representative images of each cell line. (D) Rescue of ALDH1A1 expression in clone 34 restored colony-forming efficiency. Data represents the mean ± SEM normalized to respective parental cells. *p<0.05, **p<0.01 ***p< 0.001 ****p< 0.0001, ns = not significant.
Figure 3.
Effect of ALDH1A1 loss on metabolism and survival. (A)Alamar blue assay measured cell metabolism in ALDH1A1 knockout clonal cell lines. Fold change was expressed as a ratio of day 5/day 1. (B) Rescue of ALDH1A1 expression restored cellular metabolism levels in clone 34. (C) Colony forming efficiency of SUM159 and ALDH1A1 knock out cell lines. The pictures are representative images of each cell line. (D) Rescue of ALDH1A1 expression in clone 34 restored colony-forming efficiency. Data represents the mean ± SEM normalized to respective parental cells. *p<0.05, **p<0.01 ***p< 0.001 ****p< 0.0001, ns = not significant.
Figure 4.
Effect of ALDH1A1 loss on cell viability and survival. (A) Cell viability determined using Calcein-AM (green) and Ethd-1 (red) and expressed as a ratio of live/dead. (B) Rescuing ALDH1A1 expression in clone 34 overcomes loss of viability as detected by Calcein-AM and Ethd-1 fluorescence reading. (C) Representative images of live/dead staining in SUM159 cell, ALDH1A1 knockout clones, and upon rescue of ALDH1A1 expression in clone 34. Magnification 100X (D) Annexin V positive cell analysis in ALDH1A1 KO clones (percent apoptosis). (E) Rescue of ALDH1A1 reduces apoptosis measured by Annexin V positive cells in Clone 34. Data represents the mean ± SEM normalized to respective parental cells. *p<0.05, ***p< 0.001 ****p< 0.0001, ns = not significant.
Figure 4.
Effect of ALDH1A1 loss on cell viability and survival. (A) Cell viability determined using Calcein-AM (green) and Ethd-1 (red) and expressed as a ratio of live/dead. (B) Rescuing ALDH1A1 expression in clone 34 overcomes loss of viability as detected by Calcein-AM and Ethd-1 fluorescence reading. (C) Representative images of live/dead staining in SUM159 cell, ALDH1A1 knockout clones, and upon rescue of ALDH1A1 expression in clone 34. Magnification 100X (D) Annexin V positive cell analysis in ALDH1A1 KO clones (percent apoptosis). (E) Rescue of ALDH1A1 reduces apoptosis measured by Annexin V positive cells in Clone 34. Data represents the mean ± SEM normalized to respective parental cells. *p<0.05, ***p< 0.001 ****p< 0.0001, ns = not significant.
Figure 5.
Knockout of ALDH1A1 increases reactive oxygen species level radiation response. ROS generation in the different clones as measured by their mean fluorescence intensity shown as a representative histogram (A) and graphical representation of replicates (B). ROS generation in ALDH1A1 KO clone 34 before and after rescue as measured by their mean fluorescence intensity as shown as a representative histogram (C) and graphical representation of replicates (D). rescue of ALDH1A1 reduced reactive oxygen species accumulation in Clone 34. Data represents the mean ± SEM normalized to respective parental cells. *p<0.05, **p<0.01, ns = not significant.
Figure 5.
Knockout of ALDH1A1 increases reactive oxygen species level radiation response. ROS generation in the different clones as measured by their mean fluorescence intensity shown as a representative histogram (A) and graphical representation of replicates (B). ROS generation in ALDH1A1 KO clone 34 before and after rescue as measured by their mean fluorescence intensity as shown as a representative histogram (C) and graphical representation of replicates (D). rescue of ALDH1A1 reduced reactive oxygen species accumulation in Clone 34. Data represents the mean ± SEM normalized to respective parental cells. *p<0.05, **p<0.01, ns = not significant.
Figure 6.
Loss of ALDH1A1 significantly affects colony-forming efficiency and increases radiation sensitivity in ALDH1A1 knock out clonally derived cell lines. (A) Clonogenic curve shows a radiation response to the loss of ALDH1A1. (B) Colony formation at 8 Gy radiation (Rad) normalized to no radiation (NR) in ALDH1A1 KO clones compared to SUM159 parental cells. (C) Colony forming efficiency in response to radiation inALDH1A1 clone 34 rescue. (D) Significant increase in apoptosis seen in clone 34 after radiation exposure. Data represents the mean ± SEM normalized to respective parental cells. *p<0.05, **p<0.01 ***p< 0.001, ns = not significant.
Figure 6.
Loss of ALDH1A1 significantly affects colony-forming efficiency and increases radiation sensitivity in ALDH1A1 knock out clonally derived cell lines. (A) Clonogenic curve shows a radiation response to the loss of ALDH1A1. (B) Colony formation at 8 Gy radiation (Rad) normalized to no radiation (NR) in ALDH1A1 KO clones compared to SUM159 parental cells. (C) Colony forming efficiency in response to radiation inALDH1A1 clone 34 rescue. (D) Significant increase in apoptosis seen in clone 34 after radiation exposure. Data represents the mean ± SEM normalized to respective parental cells. *p<0.05, **p<0.01 ***p< 0.001, ns = not significant.
Figure 7.
Knockout of ALDH1A1 increases reactive oxygen species levels in radiation response. (A) ROS generation in the different clones after 8Gy radiation as measured by their mean fluorescence intensity shown as a representative histogram (A) and graphical representation of replicates (B). ROS generation in ALDH1A1 KO clone 34 before and after rescue with 8Gy radiation as measured by their mean fluorescence intensity shown as a representative histogram (C) and graphical representation of replicates (D) Rescue of ALDH1A1 reduced ROS accumulation in Clone 34 irradiated cells. Data represents the mean ± SEM normalized to respective parental cells. *p<0.05, **p<0.01 ***p< 0.001 ****p< 0.0001, ns = not significant.
Figure 7.
Knockout of ALDH1A1 increases reactive oxygen species levels in radiation response. (A) ROS generation in the different clones after 8Gy radiation as measured by their mean fluorescence intensity shown as a representative histogram (A) and graphical representation of replicates (B). ROS generation in ALDH1A1 KO clone 34 before and after rescue with 8Gy radiation as measured by their mean fluorescence intensity shown as a representative histogram (C) and graphical representation of replicates (D) Rescue of ALDH1A1 reduced ROS accumulation in Clone 34 irradiated cells. Data represents the mean ± SEM normalized to respective parental cells. *p<0.05, **p<0.01 ***p< 0.001 ****p< 0.0001, ns = not significant.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).