Submitted:
20 January 2025
Posted:
21 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
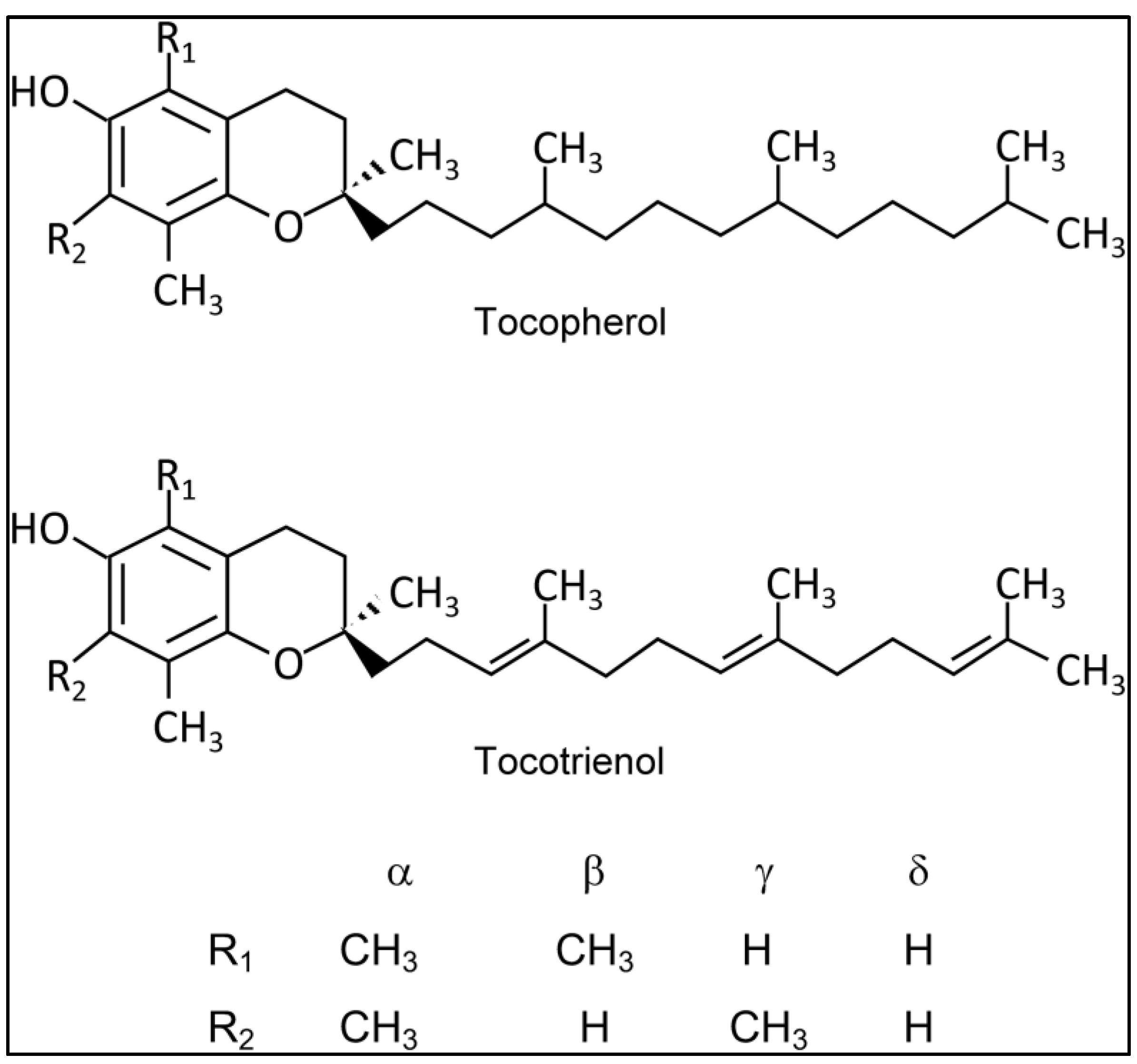
2. Materials and Methods
Materials
Instrumentation
Sample Preparation
Assay Validation
Stability
3. Results & Discussion
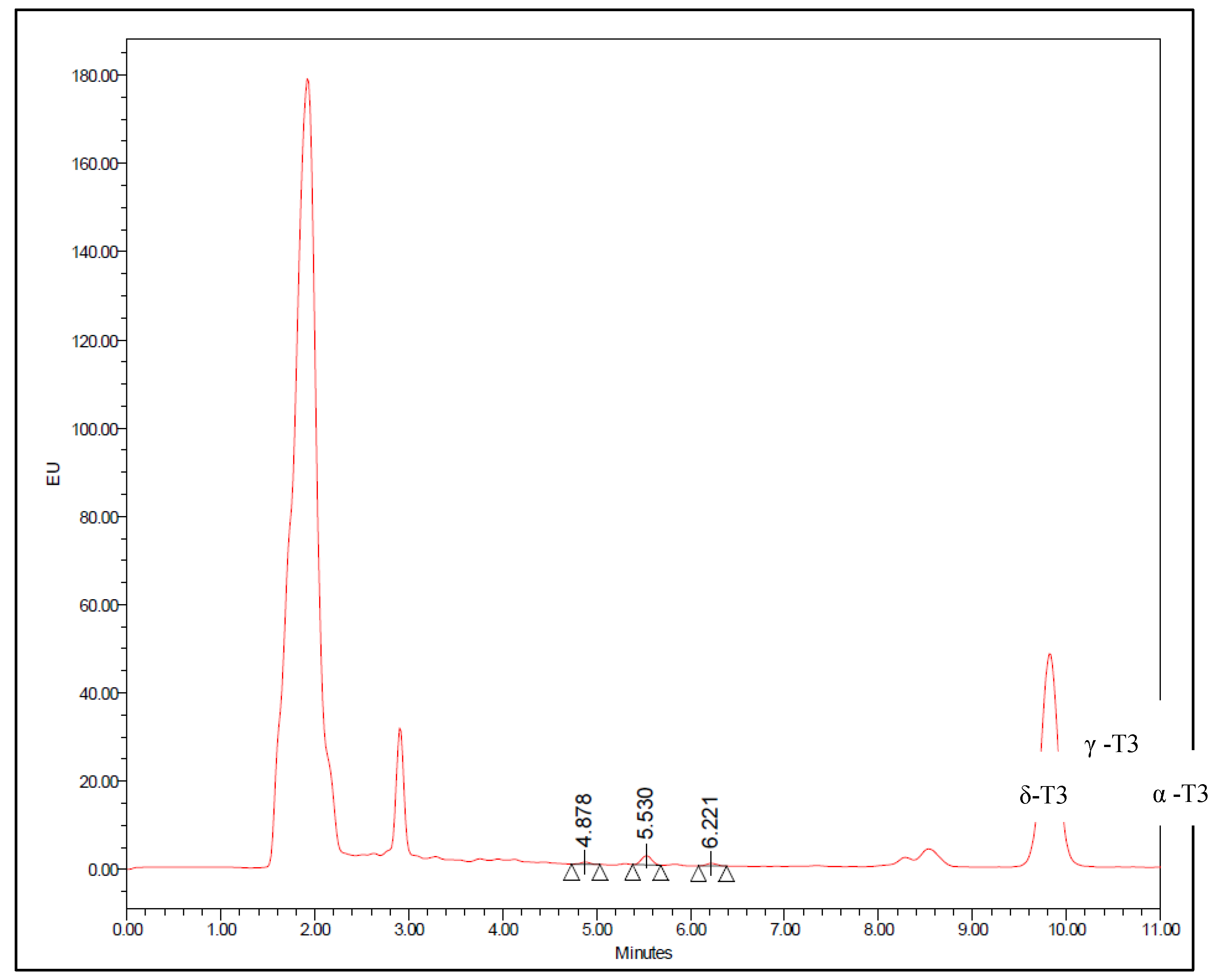
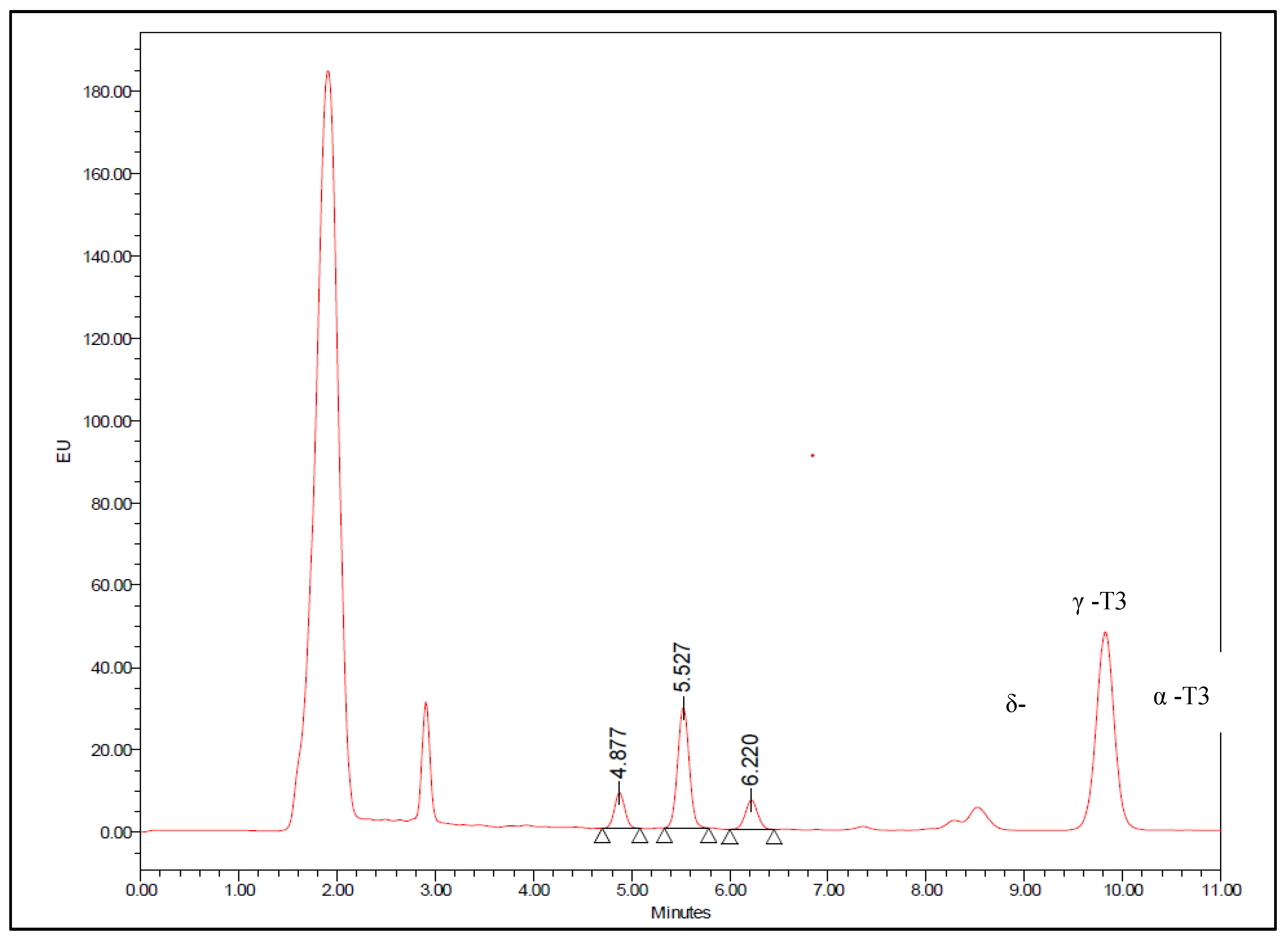
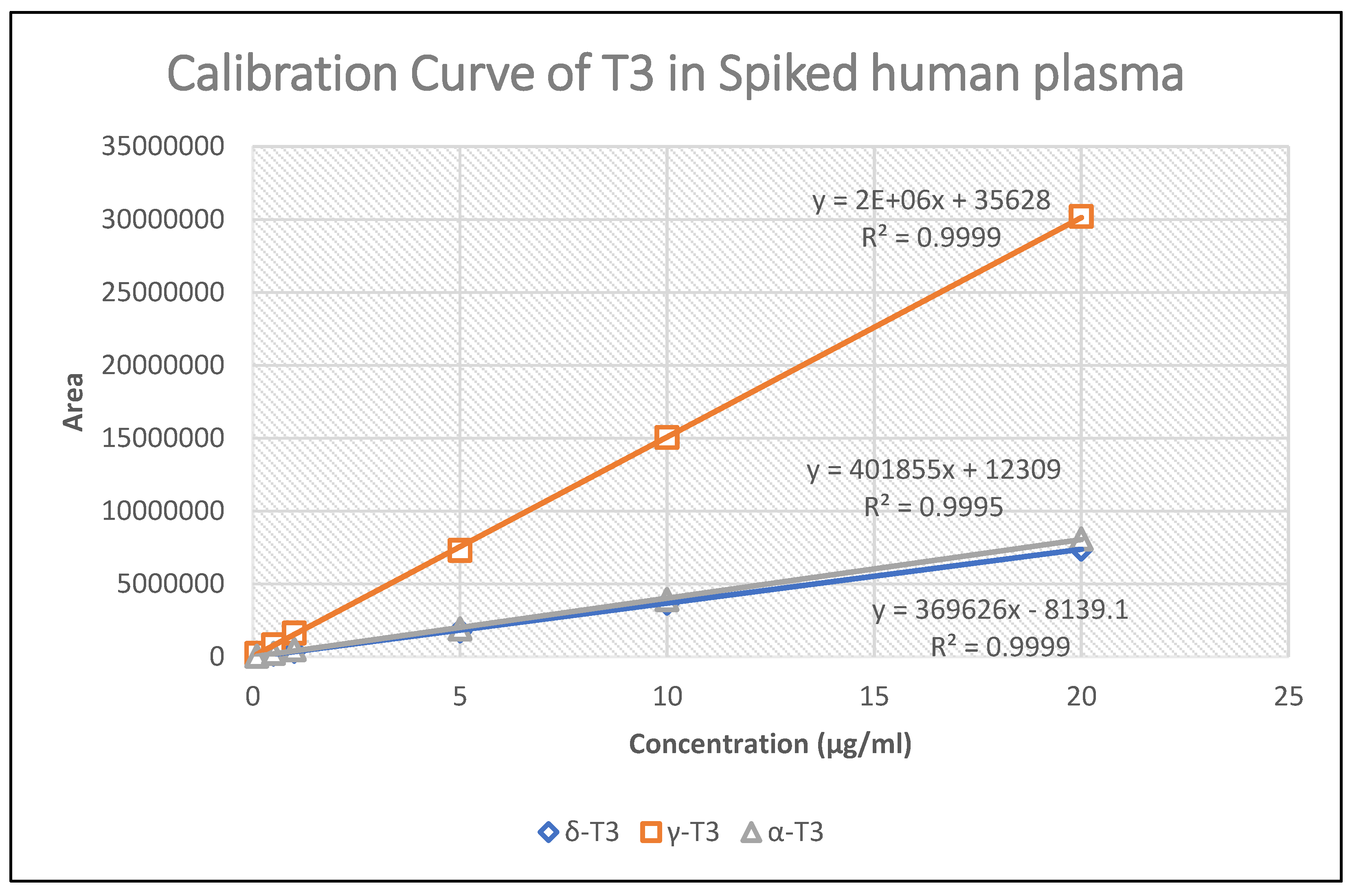
| Concentration (ng/ml) | Recovery | Intra-day (Within) | Inter-day (Between) | |
|---|---|---|---|---|
| C.V. | Accuracy (%) | Precision (C.V.) | Precision (C.V.) | |
| 4.6 | 4.3 | 102.8 | 4.7 | 12.5 |
| 23.1 | 1.7 | 108.8 | 1.6 | 12.8 |
| 46.2 | 2.7 | 97.7 | 2.9 | 5.9 |
| 230.8 | 4.3 | 99.6 | 5.4 | 5.8 |
| 461.6 | 2.9 | 109.1 | 6.1 | 8.3 |
| 923.1 | 0.8 | 108.2 | 3.0 | 6.4 |
| Concentration (ng/ml) | Recovery | Intra-day (Within) | Inter-day (Between) | |
|---|---|---|---|---|
| C.V. | Accuracy (%) | Precision (C.V.) | Precision (C.V.) | |
| 19.2 | 1.3 | 94.7 | 7.7 | 12.7 |
| 96.0 | 2.0 | 104.4 | 1.6 | 6.3 |
| 192.0 | 3.5 | 90.9 | 2.7 | 6.2 |
| 959.8 | 4.3 | 97.6 | 6.3 | 7.1 |
| 1919.7 | 3.0 | 106.3 | 7.4 | 8.6 |
| 3839.3 | 0.8 | 106.2 | 1.7 | 7.0 |
| Concentration (ng/ml) | Recovery | Intra-day (Within) | Inter-day (Between) | |
|---|---|---|---|---|
| C.V. | Accuracy (%) | Precision (C.V.) | Precision (C.V.) | |
| 12.7 | 1.3 | 102.1 | 7.0 | 10.2 |
| 63.5 | 0.2 | 111.2 | 1.8 | 10.2 |
| 126.9 | 0.8 | 91.1 | 1.9 | 6.4 |
| 634.6 | 1.6 | 85.0 | 4.9 | 8.5 |
| 1269.3 | 1.2 | 97.1 | 4.9 | 10.9 |
| 2538.6 | 0.1 | 93.5 | 2.0 | 8.3 |
| Concentration (µg/ml) | Stability mean percentage (%) | |||
|---|---|---|---|---|
| Freeze and Thaw | delta | gamma | alpha | |
| 0.1 | 111.3 | 109.5 | 111.6 | |
| 20 | 104.6 | 100.7 | 101.9 | |
| Short-term | delta | gamma | alpha | |
| 0.1 | 109.8 | 112.3 | 103.0 | |
| 20 | 104.1 | 104.2 | 100.1 | |
| Long-term | delta | gamma | alpha | |
| 0.1 | 107.2 | 113.5 | 111.0 | |
| 20 | 100.9 | 103.7 | 102.7 | |
| Post-operative | delta | gamma | alpha | |
| 0.1 | 107.1 | 114.6 | 114.4 | |
| 20 | 100.6 | 100.2 | 101.1 | |
| Concentration (µg/ml) | Stability Mean Percentage (%) | |||
|---|---|---|---|---|
| Short-term | delta | gamma | alpha | |
| 0.1 | 98.8 | 100.9 | 98.9 | |
| 20 | 101.8 | 101.9 | 101.8 | |
| Long-term | delta | gamma | alpha | |
| 0.1 | 98.3 | 102.0 | 101.2 | |
| 20 | 98.9 | 102.0 | 99.3 | |
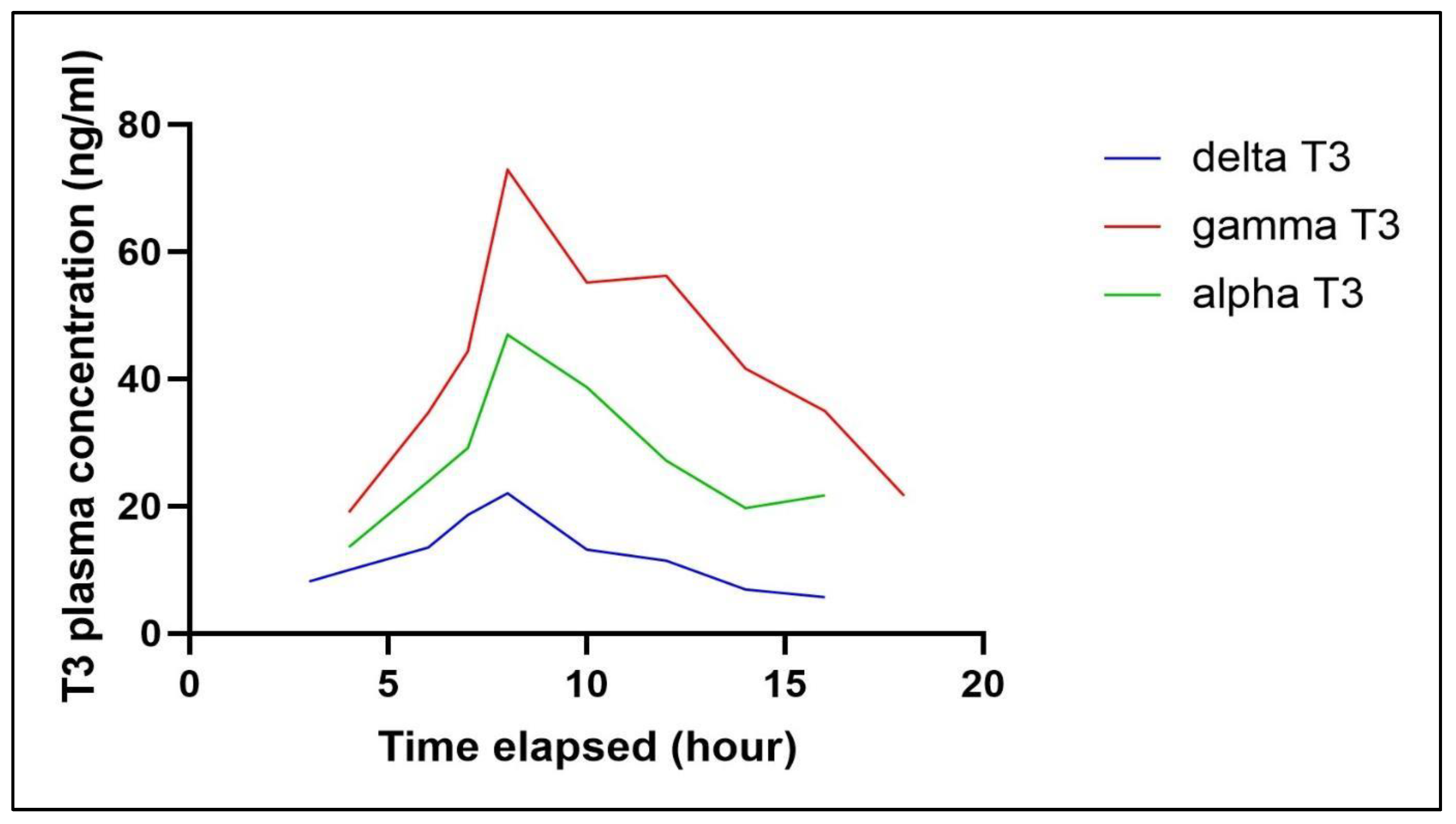
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Garg, A.; Lee, J.C.Y. Vitamin E: Where Are We Now in Vascular Diseases? Life 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Zainal, Z.; Khaza’ai, H.; Kutty Radhakrishnan, A.; Chang, S.K. Therapeutic potential of palm oil vitamin E-derived tocotrienols in inflammation and chronic diseases: Evidence from preclinical and clinical studies. Food Research International 2022, 156, 111175. [Google Scholar] [CrossRef] [PubMed]
- Ismail, M.; Alsalahi, A.; Imam, M.U.; Ooi, D.J.; Khaza’ai, H.; Aljaberi, M.A.; Shamsudin, M.N.; Idrus, Z. Safety and Neuroprotective Efficacy of Palm Oil and Tocotrienol-Rich Fraction from Palm Oil: A Systematic Review. Nutrients 2020, 12, 521. [Google Scholar] [CrossRef] [PubMed]
- Ranasinghe, R.; Mathai, M.; Zulli, A. Revisiting the therapeutic potential of tocotrienol. BioFactors 2022, 48, 813–856. [Google Scholar] [CrossRef] [PubMed]
- Abuasal, B.; Thomas, S.; Sylvester, P.W.; Kaddoumi, A. Development and validation of a reversed-phase HPLC method for the determination of γ-tocotrienol in rat and human plasma. Biomedical Chromatography 2011, 25, 621–627. [Google Scholar] [CrossRef]
- Beretta, G.; Gelmini, F.; Fontana, F.; Moretti, R.M.; Montagnani Marelli, M.; Limonta, P. Semi-preparative HPLC purification of δ-tocotrienol (δ-T3) from Elaeis guineensis Jacq. and Bixa orellana L. and evaluation of its in vitro anticancer activity in human A375 melanoma cells. Nat Prod Res 2018, 32, 1130–1135. [Google Scholar] [CrossRef]
- Bartosińska, E.; Jacyna, J.; Borsuk-De Moor, A.; Kaliszan, M.; Kruszewski, W.J.; Jankowski, Z.; Siluk, D. HPLC-APCI-MS/MS method development and validation for determination of tocotrienols in human breast adipose tissue. Talanta 2018, 176, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Nagy, K.; Courtet-Compondu, M.C.; Holst, B.; Kussmann, M. Comprehensive analysis of vitamin E constituents in human plasma by liquid chromatography-mass spectrometry. Anal Chem 2007, 79, 7087–7096. [Google Scholar] [CrossRef] [PubMed]
- Strohschein, S.; Rentel, C.; Lacker, T.; Bayer, E.; Albert, K. Separation and identification of tocotrienol isomers by HPLC-MS and HPLC-NMR coupling. Anal Chem 1999, 71, 1780–1785. [Google Scholar] [CrossRef]
- Lee, M.J.; Feng, W.; Yang, L.; Chen, Y.K.; Chi, E.; Liu, A.; Yang, C.S. Methods for efficient analysis of tocopherols, tocotrienols and their metabolites in animal samples with HPLC-EC. J Food Drug Anal 2018, 26, 318–329. [Google Scholar] [CrossRef]
- Shelly, D.C.; Warner, I.M. Fluorescence Detectors in High-Performance Liquid Chromatography. In Liquid Chromatography Detectors; CRC Press: Boca Raton, FL, USA, 2020; pp. 87–123. [Google Scholar]
- Che, H.L.; Tan, D.M.Y.; Meganathan, P.; Gan, Y.L.; Abdul Razak, G.; Fu, J.Y. Validation of a HPLC/FLD Method for Quantification of Tocotrienols in Human Plasma. Int J Anal Chem 2015, 2015, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Katsanidis, E.; Addis, P.B. Novel HPLC analysis of tocopherols, tocotrienols, and cholesterol in tissue. Free Radic Biol Med 1999, 27, 1137–1140. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.; Nazzal, S. Development and validation of a reversed-phase HPLC method for the simultaneous analysis of simvastatin and tocotrienols in combined dosage forms. J Pharm Biomed Anal 2009, 49, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Yap, S.P.; Julianto, T.; Wong, J.W.; Yuen, K.H. Simple high-performance liquid chromatographic method for the determination of tocotrienols in human plasma. J Chromatogr B Biomed Sci Appl 1999, 735, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Skoog, D.A.; West, D.M.F.; Holler, J.; Crouch, S.R. Fundamentals of Analytical Chemistry, 10th ed.; Cengage Learning: Boston, MA, USA, 2021. [Google Scholar]
- Górnaś, P.; Siger, A.; Czubinski, J.; Dwiecki, K.; Segliņa, D.; Nogala-Kalucka, M. An alternative RP-HPLC method for the separation and determination of tocopherol and tocotrienol homologues as butter authenticity markers: A comparative study between two European countries. European Journal of Lipid Science and Technology 2014, 116, 895–903. [Google Scholar] [CrossRef]
- Grebenstein, N.; Frank, J. Rapid baseline-separation of all eight tocopherols and tocotrienols by reversed-phase liquid-chromatography with a solid-core pentafluorophenyl column and their sensitive quantification in plasma and liver. J Chromatogr A 2012, 1243, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Verma, P.; Mishra, A.P.; Omar, N.; Mathur, R. A Review on Novel Analytical Method Development and Validation by RP-HPLC Method. Indian Journal of Forensic Medicine & Toxicology 2021, 15, 3476–3486. [Google Scholar]
- Fairus, S.; Nor, R.M.; Cheng, H.M.; Sundram, K. Postprandial metabolic fate of tocotrienol-rich vitamin E differs significantly from that of α-tocopherol. American Journal of Clinical Nutrition 2006, 84, 835–842. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




