Figure 1.
Multiple peripheral-systemic injurious species affect the systemic endothelial cells (SECs) and brain endothelial cells (BECs). Notably, these injurious species contribute to SECs and vascular remodeling to result in vascular arterial stiffening (VAS). These injurious species activate not only SECs but also the BECs of the neurovascular unit (NVU), resulting in BEC activation and dysfunction (BEC
act/dys) and blood–brain barrier dysfunction/disruption (BBB
dd). BEC
act/dys and BBB
dd are biomarkers for the development of cerebral small vessel disease (SVD). Importantly, SVDs may serve as biomarkers of BEC
act/dys and BBB
dd). Note the red-dashed line at the top of this image, which designates the EC plasmalemma location of the multiple injurious species that are responsible for initial SEC and BEC injury in multiple clinical diseases and structural remodeling abnormalities including SVD. SEC injurious species result in a SEC fragmentation and attenuation and/or loss of elastin that is replaced with stiffened collagen and accumulation of proteoglycans along with vascular smooth muscle cell hypertrophy and migratory phenotypes in addition to SEC stiffening contributing to VAS. BEC insulin resistance associated with an increase in glucose and the AGE receptor (RAGE) interaction is also important in the development of BEC
act/dys, BBB
dd, and neurodegeneration (not shown). Also, note that angiotensin II (AngII) denotes the activation of the renin-angiotensin-aldosterone system (RAAS) including the endothelial cell mineralocorticoid receptor (ECMR) for aldosterone and that both play a role in the development of arterial stiffening. This modified image is provided with permission by CC 4.0 [
3,
4,
5,
6]. AGE, advanced glycation end products; Ang II, angiotensin two; BBB, blood–brain barrier; BEC, brain endothelial cell; BBB
dd, blood–brain barrier dysfunction and disruption; BEC
act/dys, brain endothelial cell activation/dysfunction; BH4, tetrahydrobiopterin; CCL2, chemokine (C-C motif) ligand 2; Cox-2, cyclo-oxygenase-2; Cox-2/PGE2 axis, cyclo-oxygenase-2/prostaglandin E2; downward arrows, arrowheads, denote call outs; ecGCx, endothelial glycocalyx; ICAM-1, intercellular adhesion molecule-1; IL-1β, interleukin-1β; IL-6, interleukin-6; JAMs, junctional adhesion molecules; LDL, low-density lipoprotein cholesterol; LPa, lipoprotein little a; MCP-1, monocyte chemotactic protein-1; NO, nitric oxide; Nox2, nicotinamide adenine dinucleotide phosphate oxidase (NADPH Ox); peroxinitrite (ONOO-); pnsCC, peripheral nervous system cytokines/chemokines; pp, pulsatile-pulse pressure;, NVU, neurovascular unit; RBC, red blood cell; Red arrows, denote increase; RONSS, reactive oxygen, nitrogen, sulfur species; ROS, reactive oxygen species; RSI, reactive species interactome; T, transcytosis; TNFα, tumor necrosis factor alpha; underlining, denotes emphasis; upward arrows, denote increase; VCAM-1, vascular cellular adhesion molecule-1; WBC, white blood cell.
Figure 1.
Multiple peripheral-systemic injurious species affect the systemic endothelial cells (SECs) and brain endothelial cells (BECs). Notably, these injurious species contribute to SECs and vascular remodeling to result in vascular arterial stiffening (VAS). These injurious species activate not only SECs but also the BECs of the neurovascular unit (NVU), resulting in BEC activation and dysfunction (BEC
act/dys) and blood–brain barrier dysfunction/disruption (BBB
dd). BEC
act/dys and BBB
dd are biomarkers for the development of cerebral small vessel disease (SVD). Importantly, SVDs may serve as biomarkers of BEC
act/dys and BBB
dd). Note the red-dashed line at the top of this image, which designates the EC plasmalemma location of the multiple injurious species that are responsible for initial SEC and BEC injury in multiple clinical diseases and structural remodeling abnormalities including SVD. SEC injurious species result in a SEC fragmentation and attenuation and/or loss of elastin that is replaced with stiffened collagen and accumulation of proteoglycans along with vascular smooth muscle cell hypertrophy and migratory phenotypes in addition to SEC stiffening contributing to VAS. BEC insulin resistance associated with an increase in glucose and the AGE receptor (RAGE) interaction is also important in the development of BEC
act/dys, BBB
dd, and neurodegeneration (not shown). Also, note that angiotensin II (AngII) denotes the activation of the renin-angiotensin-aldosterone system (RAAS) including the endothelial cell mineralocorticoid receptor (ECMR) for aldosterone and that both play a role in the development of arterial stiffening. This modified image is provided with permission by CC 4.0 [
3,
4,
5,
6]. AGE, advanced glycation end products; Ang II, angiotensin two; BBB, blood–brain barrier; BEC, brain endothelial cell; BBB
dd, blood–brain barrier dysfunction and disruption; BEC
act/dys, brain endothelial cell activation/dysfunction; BH4, tetrahydrobiopterin; CCL2, chemokine (C-C motif) ligand 2; Cox-2, cyclo-oxygenase-2; Cox-2/PGE2 axis, cyclo-oxygenase-2/prostaglandin E2; downward arrows, arrowheads, denote call outs; ecGCx, endothelial glycocalyx; ICAM-1, intercellular adhesion molecule-1; IL-1β, interleukin-1β; IL-6, interleukin-6; JAMs, junctional adhesion molecules; LDL, low-density lipoprotein cholesterol; LPa, lipoprotein little a; MCP-1, monocyte chemotactic protein-1; NO, nitric oxide; Nox2, nicotinamide adenine dinucleotide phosphate oxidase (NADPH Ox); peroxinitrite (ONOO-); pnsCC, peripheral nervous system cytokines/chemokines; pp, pulsatile-pulse pressure;, NVU, neurovascular unit; RBC, red blood cell; Red arrows, denote increase; RONSS, reactive oxygen, nitrogen, sulfur species; ROS, reactive oxygen species; RSI, reactive species interactome; T, transcytosis; TNFα, tumor necrosis factor alpha; underlining, denotes emphasis; upward arrows, denote increase; VCAM-1, vascular cellular adhesion molecule-1; WBC, white blood cell.
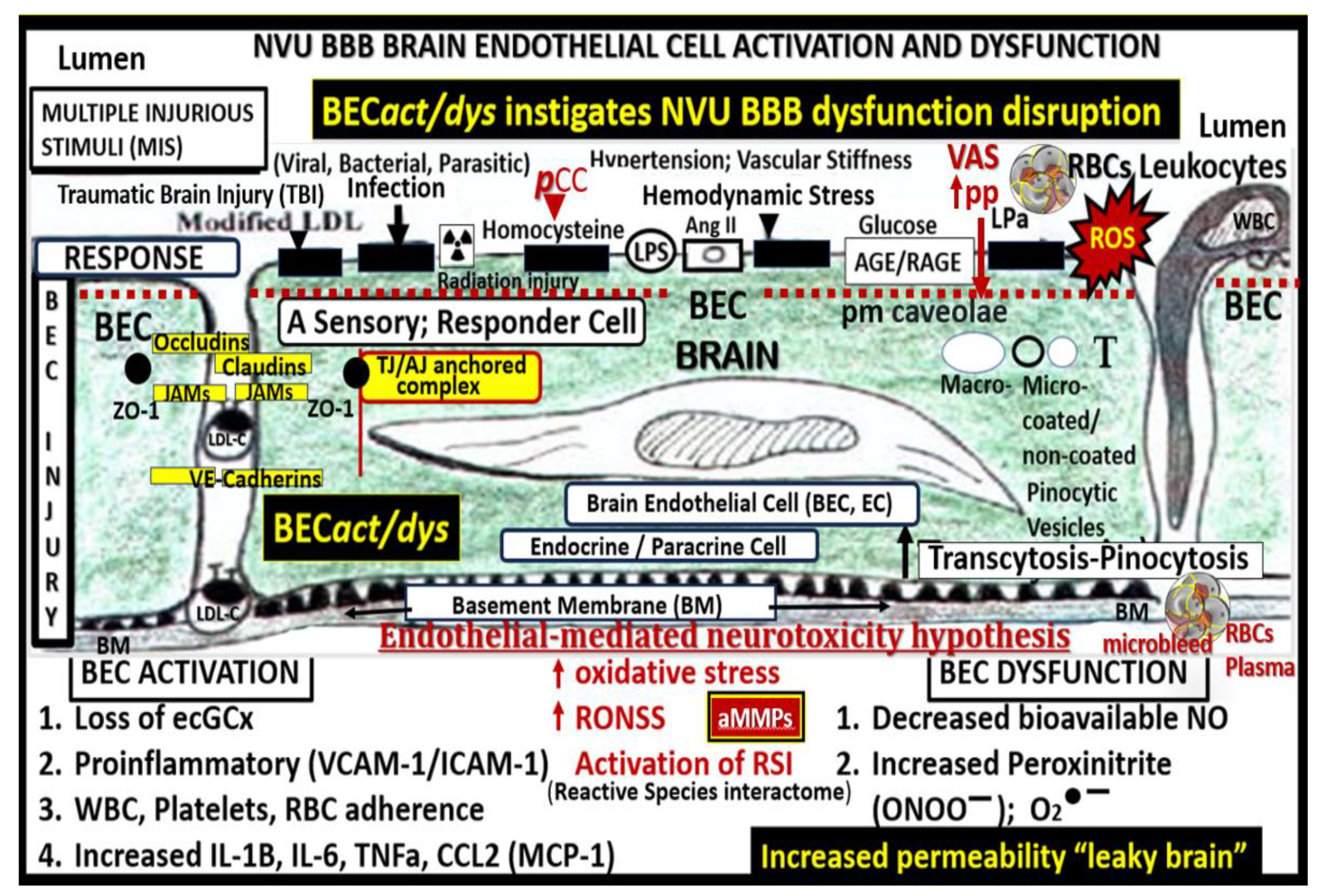
Figure 2.
Possible four-step sequence of events (S1–S4) implicating extracranial vascular arterial stiffening (VAS) in the development and progression of neurodegeneration and late-onset Alzheimer’s disease (LOAD). Step 1. Includes aging, genetics, hypertension inclusive of essential hypertension (eHTN) and the more predominant isolated systolic hypertension (ISH) in the elderly, and atherosclerosis that is inclusive of vascular ossification calcification (VOC), metabolic syndrome (MetS), insulin resistance (IR), and type 2 diabetes mellitus (T2DM). Step 2. Includes extracranial vascular arterial stiffening/stiffness (VAS), oxidative redox stress and vascular inflammation, stroke (primarily ischemic and secondarily hemorrhagic), and vascular contributions to impaired cognition and dementia (VCID). Step 3. Includes small vessel disease (SVD), which includes lacunes, white matter hyperintensities, enlarged perivascular spaces, and cerebral microbleeds that contribute to chronic cerebral hypoperfusion (CCH). Step 4. Includes neurodegeneration, atrophy, and LOAD. ECM, extracellular matrix including both collagen and proteoglycans, CCH, chronic cerebral hypoperfusion and hypometabolism; MCI, mild cognitive impairment; open arrows, represent icons meaning leading to; SECact/dys, systemic endothelial cell activation and dysfunction; VSMC, vascular smooth muscle cell(s).
Figure 2.
Possible four-step sequence of events (S1–S4) implicating extracranial vascular arterial stiffening (VAS) in the development and progression of neurodegeneration and late-onset Alzheimer’s disease (LOAD). Step 1. Includes aging, genetics, hypertension inclusive of essential hypertension (eHTN) and the more predominant isolated systolic hypertension (ISH) in the elderly, and atherosclerosis that is inclusive of vascular ossification calcification (VOC), metabolic syndrome (MetS), insulin resistance (IR), and type 2 diabetes mellitus (T2DM). Step 2. Includes extracranial vascular arterial stiffening/stiffness (VAS), oxidative redox stress and vascular inflammation, stroke (primarily ischemic and secondarily hemorrhagic), and vascular contributions to impaired cognition and dementia (VCID). Step 3. Includes small vessel disease (SVD), which includes lacunes, white matter hyperintensities, enlarged perivascular spaces, and cerebral microbleeds that contribute to chronic cerebral hypoperfusion (CCH). Step 4. Includes neurodegeneration, atrophy, and LOAD. ECM, extracellular matrix including both collagen and proteoglycans, CCH, chronic cerebral hypoperfusion and hypometabolism; MCI, mild cognitive impairment; open arrows, represent icons meaning leading to; SECact/dys, systemic endothelial cell activation and dysfunction; VSMC, vascular smooth muscle cell(s).
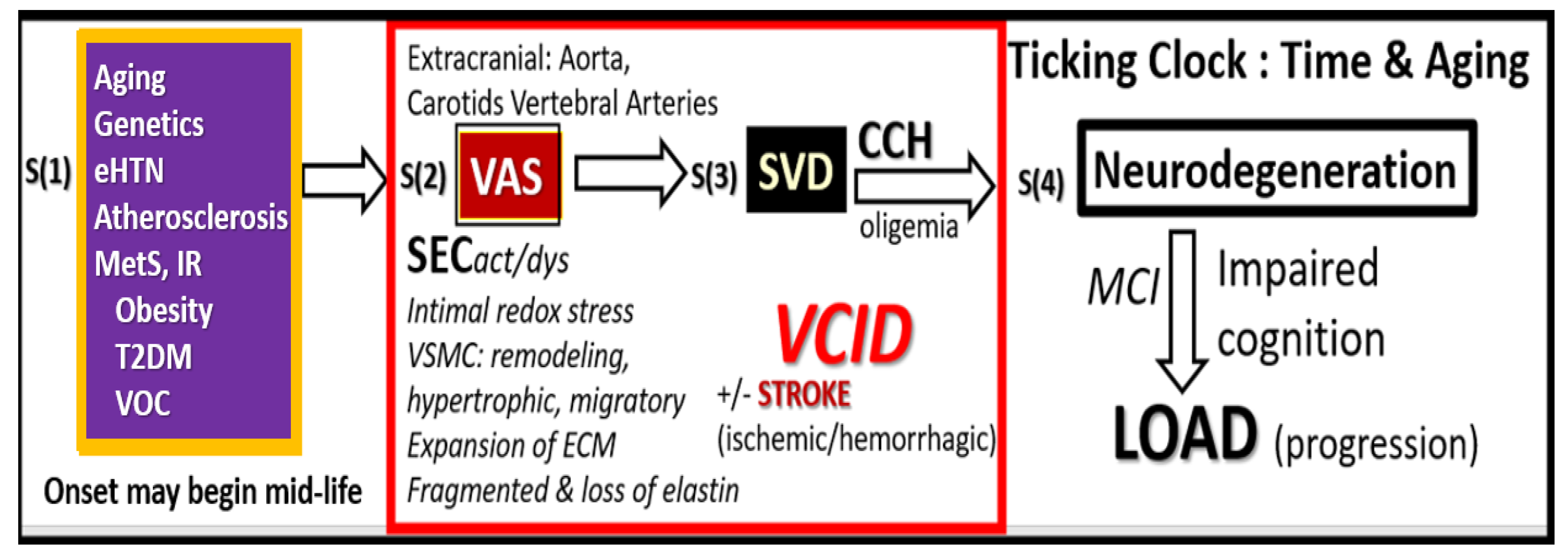
Figure 3.
The essential hypertension (HTN) wheel. This HTN wheel depicts insulin resistance (IR) as a central core feature of the wheel as it is also the central core feature in the metabolic syndrome (MetS). IR is a central mediator for the development of HTN, impaired fasting glucose (IFG), impaired glucose tolerance (IGT), and type two diabetes mellitus (T2DM) along with multiple metabolic and clinical boxed-in conditions that surround the outer positions of this hypertension wheel. Extracranial vascular arterial stiffening (VAS) is placed at the top of the wheel because it is believed to be a driving force behind the subsequent clinical end-organ cerebral small vessel disease (SVD) remodeling in addition to the development of HTN. Systemic and brain endothelial cell activation and dysfunction (SEC and BEC respectively) are represented by EC
act/dys at the bottom of the wheel result in increased peroxynitrite (ONOO
−) and decreased nitric oxide (NO) bioavailability (asterisk). Also, peroxynitrite is generated by the reaction between superoxide and NO. Note the multiple diseases that are associated with HTN (black boxes surrounding the HTN wheel), and the wheel also depicts the interconnectedness between HTN and the multiple disease states, including vascular stiffening. Thus, obesity, IR, HTN, and T2DM are not to be underestimated. Atheroscleropathy is a term that may be used when discussing accelerated atherosclerosis and macrovascular disease in those individuals with T2DM and the MetS. The wheel was chosen as a background icon because it goes round and round, and over time it just keeps on turning and results in vascular arterial stiffening (VAS) and end-organ damage in the heart-vessel-brain axis that has high capillary flow with low resistance and increased vulnerability to the increased pulsatile pulse pressure and pulse wave velocity associated with vascular stiffening, HTN, and microvascular disease. Modified image provided with permission by CC 4.0 [
34]. Asterisk, equals emphasis; CAD, coronary artery disease; CCVD, cerebrocardiovascular disease; CHF, congestive heart failure; CKD, chronic kidney disease; Dd, diastolic dysfunction
; eNOS, endothelial nitric oxide synthase; ESRD, end-stage renal disease; LOAD, late-onset Alzheimer’s disease; MASLD, metabolic dysfunction-associated liver disease; MI, myocardial infarction; mtROS, mitochondrial reactive oxygen species; O
2•–, superoxide; PAD, peripheral artery disease; TIA, transient ischemic attack.
Figure 3.
The essential hypertension (HTN) wheel. This HTN wheel depicts insulin resistance (IR) as a central core feature of the wheel as it is also the central core feature in the metabolic syndrome (MetS). IR is a central mediator for the development of HTN, impaired fasting glucose (IFG), impaired glucose tolerance (IGT), and type two diabetes mellitus (T2DM) along with multiple metabolic and clinical boxed-in conditions that surround the outer positions of this hypertension wheel. Extracranial vascular arterial stiffening (VAS) is placed at the top of the wheel because it is believed to be a driving force behind the subsequent clinical end-organ cerebral small vessel disease (SVD) remodeling in addition to the development of HTN. Systemic and brain endothelial cell activation and dysfunction (SEC and BEC respectively) are represented by EC
act/dys at the bottom of the wheel result in increased peroxynitrite (ONOO
−) and decreased nitric oxide (NO) bioavailability (asterisk). Also, peroxynitrite is generated by the reaction between superoxide and NO. Note the multiple diseases that are associated with HTN (black boxes surrounding the HTN wheel), and the wheel also depicts the interconnectedness between HTN and the multiple disease states, including vascular stiffening. Thus, obesity, IR, HTN, and T2DM are not to be underestimated. Atheroscleropathy is a term that may be used when discussing accelerated atherosclerosis and macrovascular disease in those individuals with T2DM and the MetS. The wheel was chosen as a background icon because it goes round and round, and over time it just keeps on turning and results in vascular arterial stiffening (VAS) and end-organ damage in the heart-vessel-brain axis that has high capillary flow with low resistance and increased vulnerability to the increased pulsatile pulse pressure and pulse wave velocity associated with vascular stiffening, HTN, and microvascular disease. Modified image provided with permission by CC 4.0 [
34]. Asterisk, equals emphasis; CAD, coronary artery disease; CCVD, cerebrocardiovascular disease; CHF, congestive heart failure; CKD, chronic kidney disease; Dd, diastolic dysfunction
; eNOS, endothelial nitric oxide synthase; ESRD, end-stage renal disease; LOAD, late-onset Alzheimer’s disease; MASLD, metabolic dysfunction-associated liver disease; MI, myocardial infarction; mtROS, mitochondrial reactive oxygen species; O
2•–, superoxide; PAD, peripheral artery disease; TIA, transient ischemic attack.
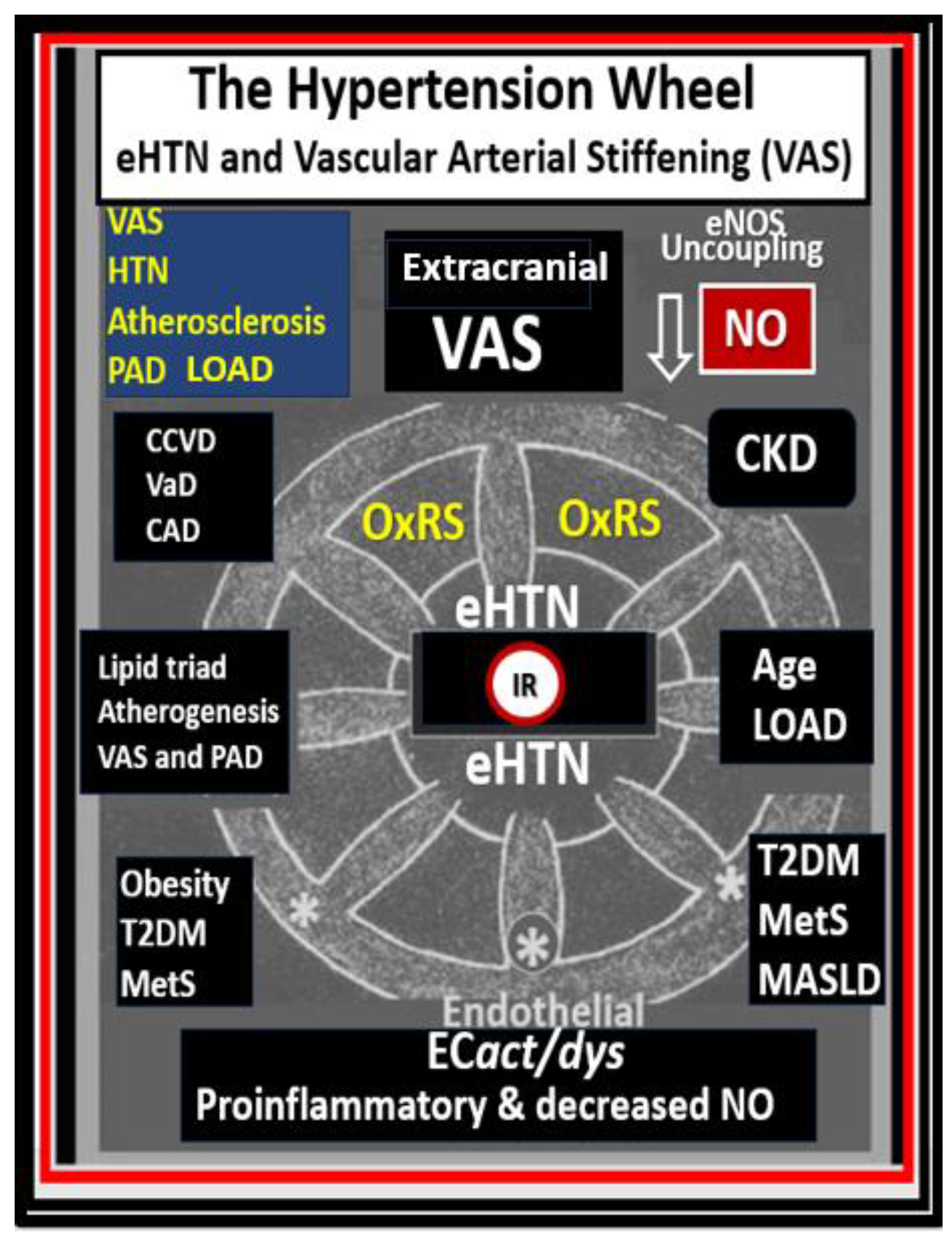
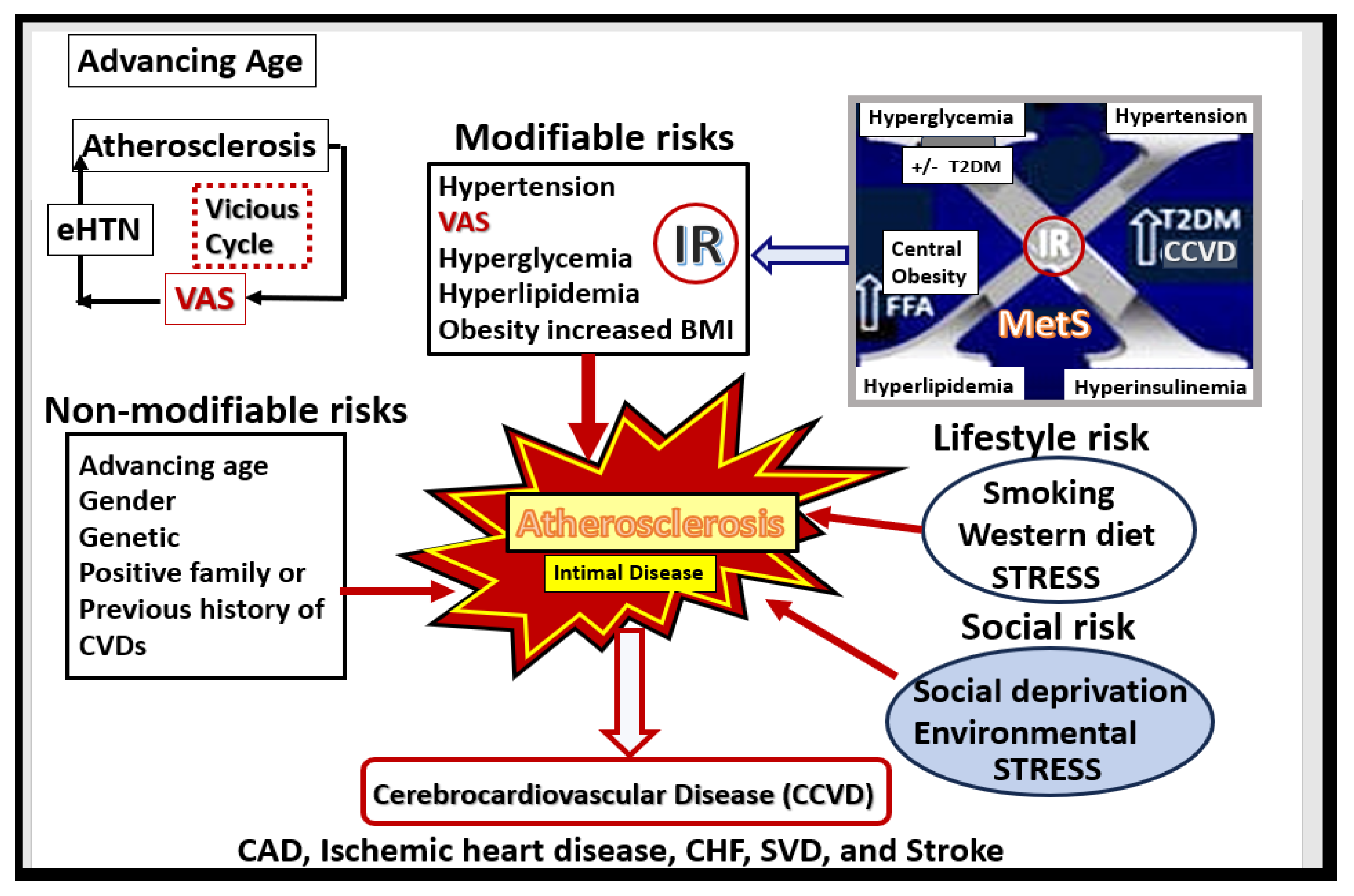
Figure 4.
Multiple risk factors for the development and progression of atherosclerosis. Note the important role of the MetS upper right and also the vicious cycle between atherosclerosis, eHTN and VAS that associates with advancing age. BMI, body mass index; CAD, coronary artery disease; CCVD, cerebrocardiovascular disease; CHF, congestive heart failure; CVDs, cardiovascular diseases; eHTN, essential hypertension; FFA, free fatty acids; IR, insulin resistance; MetS, metabolic syndrome; SVD, small vessel disease. T2DM, type 2 diabetes mellitus; VAS, extracranial vascular arterial stiffness/stiffening. .
Figure 4.
Multiple risk factors for the development and progression of atherosclerosis. Note the important role of the MetS upper right and also the vicious cycle between atherosclerosis, eHTN and VAS that associates with advancing age. BMI, body mass index; CAD, coronary artery disease; CCVD, cerebrocardiovascular disease; CHF, congestive heart failure; CVDs, cardiovascular diseases; eHTN, essential hypertension; FFA, free fatty acids; IR, insulin resistance; MetS, metabolic syndrome; SVD, small vessel disease. T2DM, type 2 diabetes mellitus; VAS, extracranial vascular arterial stiffness/stiffening. .
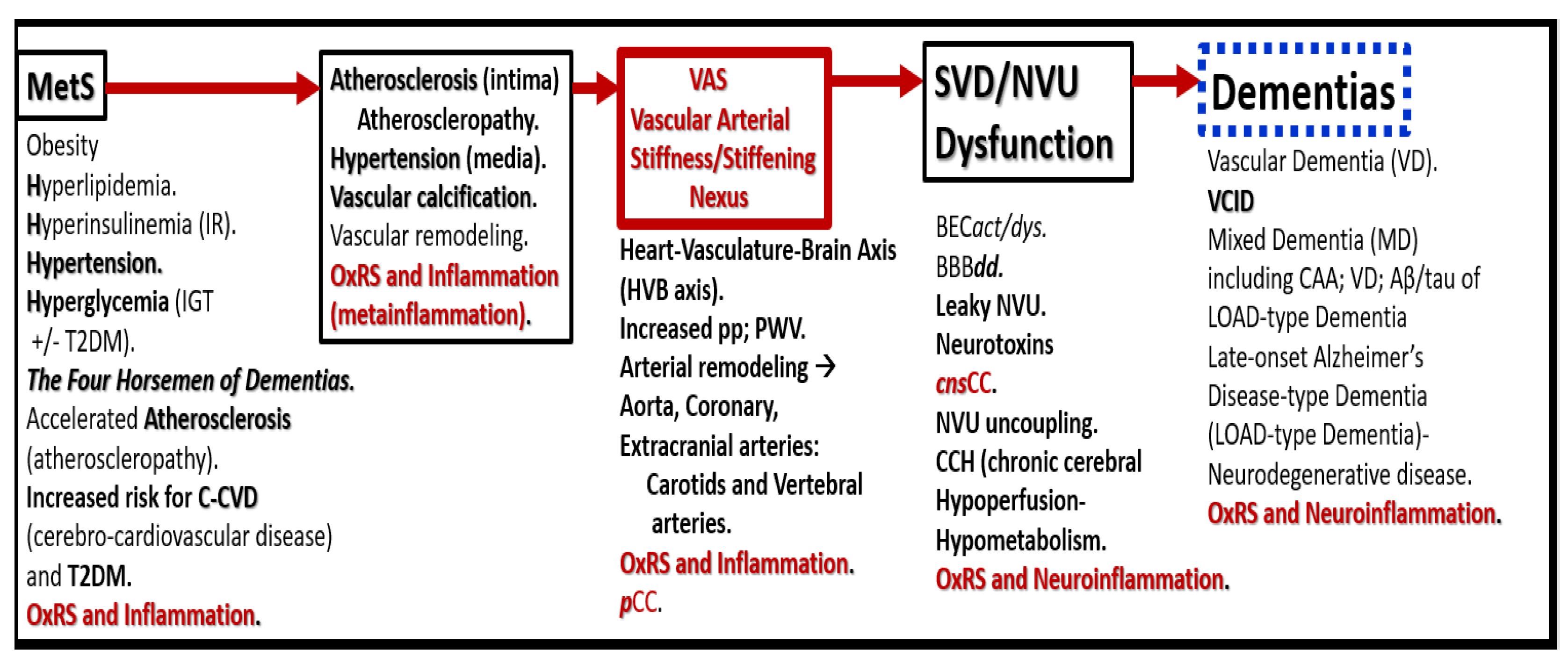
Figure 5.
Possible sequence of events in the development and progression of dementias. Vascular arterial stiffening/stiffness (VAS) may be the Nexus. This figure serves to further expand the information presented in
Figure 2. Aβ, amyloid beta, BEC
act/dys, brain endothelial cell activation/dysfunction; BBB
dd, blood-brain barrier dysfunction and disruption; CAA, cerebral amyloid angiopathy;
cnsCC, central nervous system cytokines/chemokines; IGT, impaired glucose tolerance; MetS, metabolic syndrome; OxRS, oxidative redox stress; NVU, neurovascular unit;
pCC, peripheral cytokines/chemokines; pp, pulse pressure; PWV, pulse wave velocity; SVD, small vessel disease; T2DM, type 2 diabetes mellitus; VCID, vascular contributions to impaired cognition and dementia.
Figure 5.
Possible sequence of events in the development and progression of dementias. Vascular arterial stiffening/stiffness (VAS) may be the Nexus. This figure serves to further expand the information presented in
Figure 2. Aβ, amyloid beta, BEC
act/dys, brain endothelial cell activation/dysfunction; BBB
dd, blood-brain barrier dysfunction and disruption; CAA, cerebral amyloid angiopathy;
cnsCC, central nervous system cytokines/chemokines; IGT, impaired glucose tolerance; MetS, metabolic syndrome; OxRS, oxidative redox stress; NVU, neurovascular unit;
pCC, peripheral cytokines/chemokines; pp, pulse pressure; PWV, pulse wave velocity; SVD, small vessel disease; T2DM, type 2 diabetes mellitus; VCID, vascular contributions to impaired cognition and dementia.
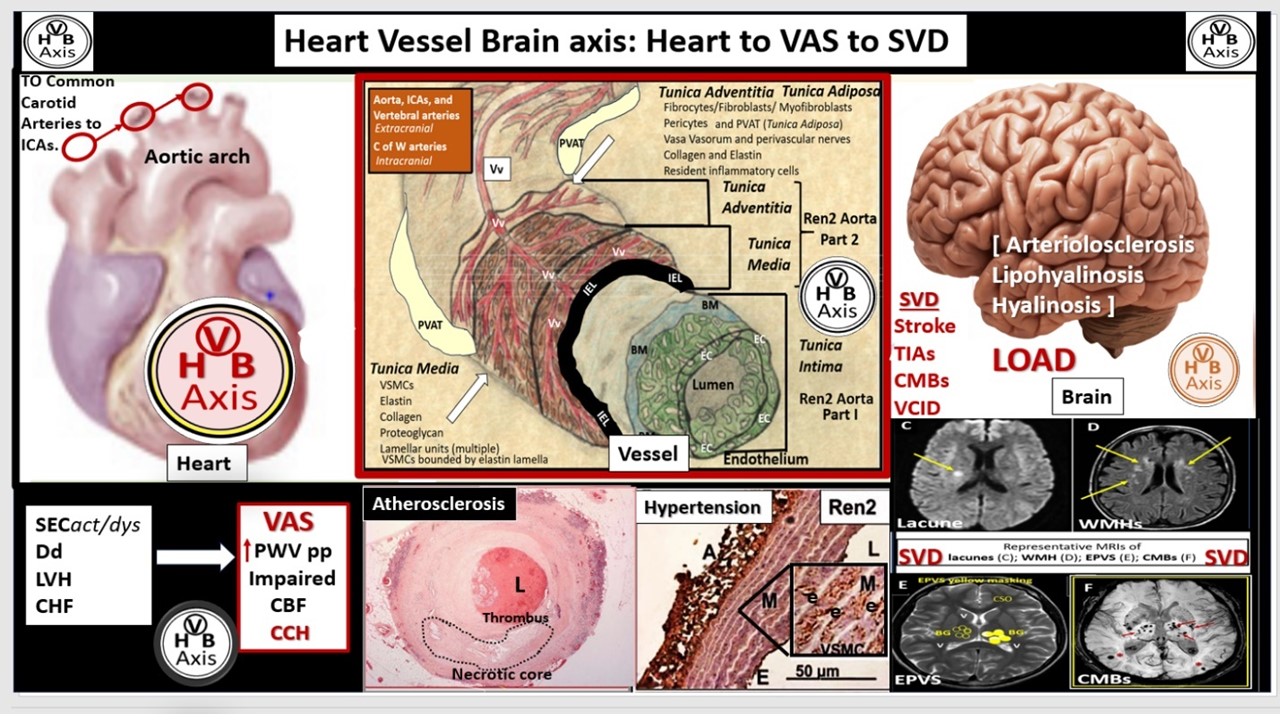
Figure 6.
Arterial pull-out model illustrating the three major tunica layers of arteries and how they are structurally interrelated. This illustration demonstrates the lumen that is surrounded by the monolayer of systemic and brain endothelial cells (SECs and BECs) (pseudo-colored green) that have a basement membrane (BM) pseudo-colored blue, and the proteoglycan (PGN)-rich and hyaluronan subintimal space (SiS noted by double arrows). These three tunica layers begin most luminally with the tunica intima where atherosclerosis is known to begin. Next the thickened black line forms internal elastic lamina (IEL), which is demarcated by the bold white line, which separates the intima from the tunica media that encircles the artery and serves to allow elastic recoil of the artery once it is stretched along with multiple layers of the elastin lamella that bound the lamellar units containing the vascular smooth muscle cell(s) (VSMCs) pseudo-colored red and denoted by open white arrows that comprise the second tunica that is bound abluminally by the external elastic lamina (yellow dashed line). The most abluminal and third tunica is the tunica adventitia, which is demarcated by the yellow dashed line luminally and the perivascular adipose tissue (PVAT) of the tunica adventitia that contains a loose connective tissue with multiple cells including pericytes, mast cells and occasional leukocytes through which, the vasa vasorum (Vv) flows as it penetrates the media to be referred to as the vessel within the vessel. The perivascular adipose tissue (PVAT underlined and pseudo-colored yellow) contains visceral adipocytes and extracellular matrix that is an extension of the tunica adventitia; however, some feel that it merits the distinction to be called the tunica adiposa due to its proinflammatory (metainflammation) potential to result in aberrant arterial remodeling from the outside in. Modified image provided with permission by CC 4.0 [
14,
34]. C of W, circle of Willis; EL, elastin lamina; HVB, heart-vessel-brain axis; ICAs, internal carotid arteries; PGN, proteoglycan; Ren2 rat part I and II, ultrastructure study of the young male transgenic heterozygous (mRen2)27 (Ren2) rat hypertensive model: Aorta intima Part I and media-adventitia Part II with elevated renin, angiotensin II, angiotensin type-1 (AT-1) receptors, and aldosterone; Vv, vasa vasorum.
Figure 6.
Arterial pull-out model illustrating the three major tunica layers of arteries and how they are structurally interrelated. This illustration demonstrates the lumen that is surrounded by the monolayer of systemic and brain endothelial cells (SECs and BECs) (pseudo-colored green) that have a basement membrane (BM) pseudo-colored blue, and the proteoglycan (PGN)-rich and hyaluronan subintimal space (SiS noted by double arrows). These three tunica layers begin most luminally with the tunica intima where atherosclerosis is known to begin. Next the thickened black line forms internal elastic lamina (IEL), which is demarcated by the bold white line, which separates the intima from the tunica media that encircles the artery and serves to allow elastic recoil of the artery once it is stretched along with multiple layers of the elastin lamella that bound the lamellar units containing the vascular smooth muscle cell(s) (VSMCs) pseudo-colored red and denoted by open white arrows that comprise the second tunica that is bound abluminally by the external elastic lamina (yellow dashed line). The most abluminal and third tunica is the tunica adventitia, which is demarcated by the yellow dashed line luminally and the perivascular adipose tissue (PVAT) of the tunica adventitia that contains a loose connective tissue with multiple cells including pericytes, mast cells and occasional leukocytes through which, the vasa vasorum (Vv) flows as it penetrates the media to be referred to as the vessel within the vessel. The perivascular adipose tissue (PVAT underlined and pseudo-colored yellow) contains visceral adipocytes and extracellular matrix that is an extension of the tunica adventitia; however, some feel that it merits the distinction to be called the tunica adiposa due to its proinflammatory (metainflammation) potential to result in aberrant arterial remodeling from the outside in. Modified image provided with permission by CC 4.0 [
14,
34]. C of W, circle of Willis; EL, elastin lamina; HVB, heart-vessel-brain axis; ICAs, internal carotid arteries; PGN, proteoglycan; Ren2 rat part I and II, ultrastructure study of the young male transgenic heterozygous (mRen2)27 (Ren2) rat hypertensive model: Aorta intima Part I and media-adventitia Part II with elevated renin, angiotensin II, angiotensin type-1 (AT-1) receptors, and aldosterone; Vv, vasa vasorum.
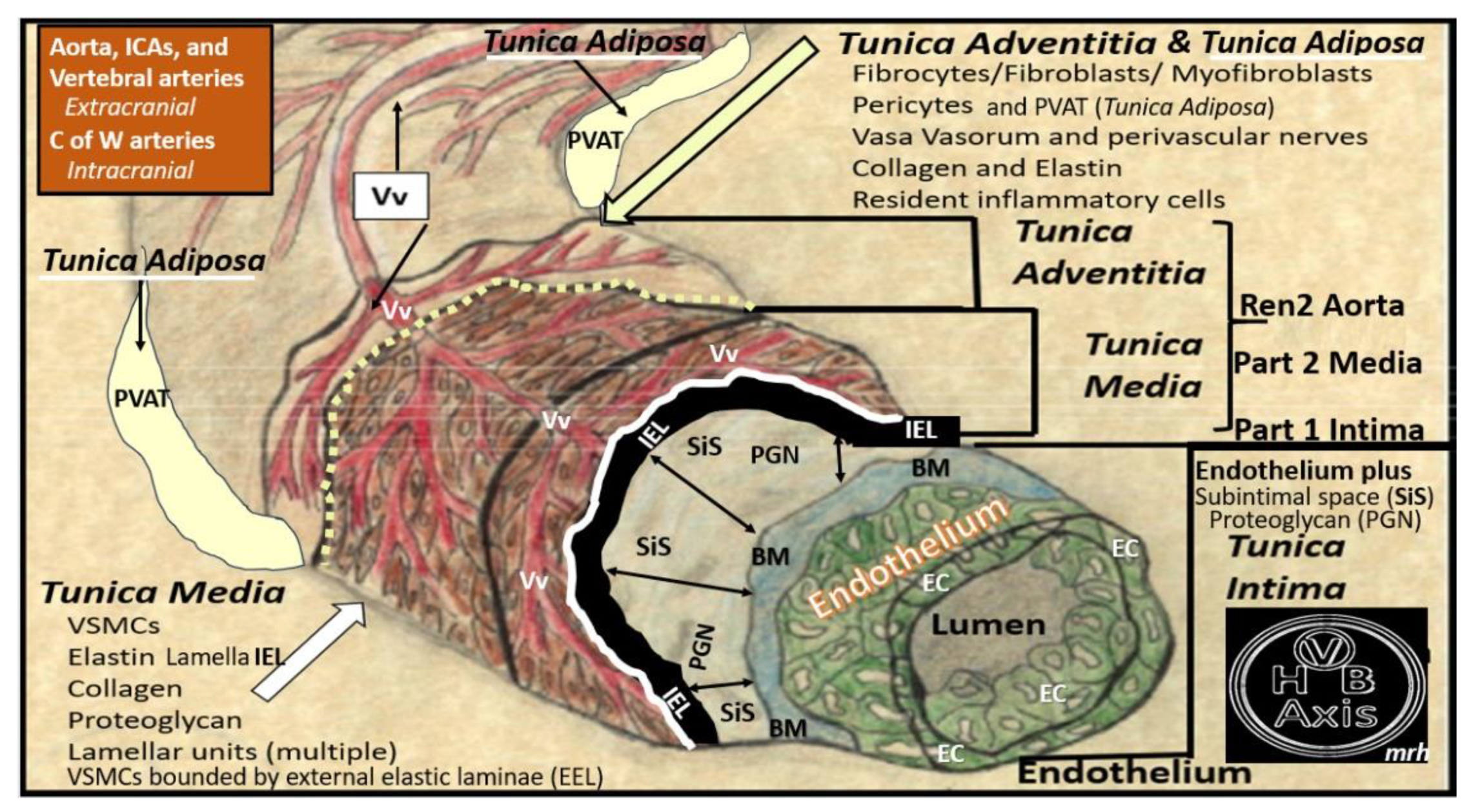
Figure 7.
Comparison of non-diabetic plaque rupture to diabetic (T2DM) plaque erosion to result in atherothrombosis and sudden death. Panel A depicts an erosive plaque in a 58-year-old female individual with history of T2DM, obesity, hypertension, and previous stroke who presented with acute ventricular tachycardia and sudden death due to a left mid-anterior descending coronary occlusion with an erosive concentric plaque [
56]. Note the extensive mononuclear vasculitic type adventitial infiltrate associated with this concentric erosive thrombotic plaque. While each of these vulnerable plaques in panels B and A’ associate with increased extracranial vascular arterial stiffness they may result in similar clinical outcomes, i.e. sudden death due to atherothrombosis. However, non-diabetic plaques are more prone to rupture while T2DM individuals are more prone to develop thromboerosion and result in sudden death. Additionally, T2DM individuals are known to have increased vascular stiffness as compared to non-diabetic individuals. Panels A and A’ are hematoxylin and eosin (H&E) and Panel B is H&E stained (pseudo-colored blue in order to contrast colors with A and A’). Modified images provided with permission by CC 4.0 [
56]. Asterisks, vasculitic lymphocytic infiltrate within the adventitia; CAD, coronary artery disease; P22 Phox (underlined in red), is the light chain of the NADPHOx enzyme and its positive staining strongly suggests increased oxidative stress within the plaque and the adventitia that may contribute to plaque instability and erosion in T2DM; Vv, vasa vasorum.
Figure 7.
Comparison of non-diabetic plaque rupture to diabetic (T2DM) plaque erosion to result in atherothrombosis and sudden death. Panel A depicts an erosive plaque in a 58-year-old female individual with history of T2DM, obesity, hypertension, and previous stroke who presented with acute ventricular tachycardia and sudden death due to a left mid-anterior descending coronary occlusion with an erosive concentric plaque [
56]. Note the extensive mononuclear vasculitic type adventitial infiltrate associated with this concentric erosive thrombotic plaque. While each of these vulnerable plaques in panels B and A’ associate with increased extracranial vascular arterial stiffness they may result in similar clinical outcomes, i.e. sudden death due to atherothrombosis. However, non-diabetic plaques are more prone to rupture while T2DM individuals are more prone to develop thromboerosion and result in sudden death. Additionally, T2DM individuals are known to have increased vascular stiffness as compared to non-diabetic individuals. Panels A and A’ are hematoxylin and eosin (H&E) and Panel B is H&E stained (pseudo-colored blue in order to contrast colors with A and A’). Modified images provided with permission by CC 4.0 [
56]. Asterisks, vasculitic lymphocytic infiltrate within the adventitia; CAD, coronary artery disease; P22 Phox (underlined in red), is the light chain of the NADPHOx enzyme and its positive staining strongly suggests increased oxidative stress within the plaque and the adventitia that may contribute to plaque instability and erosion in T2DM; Vv, vasa vasorum.
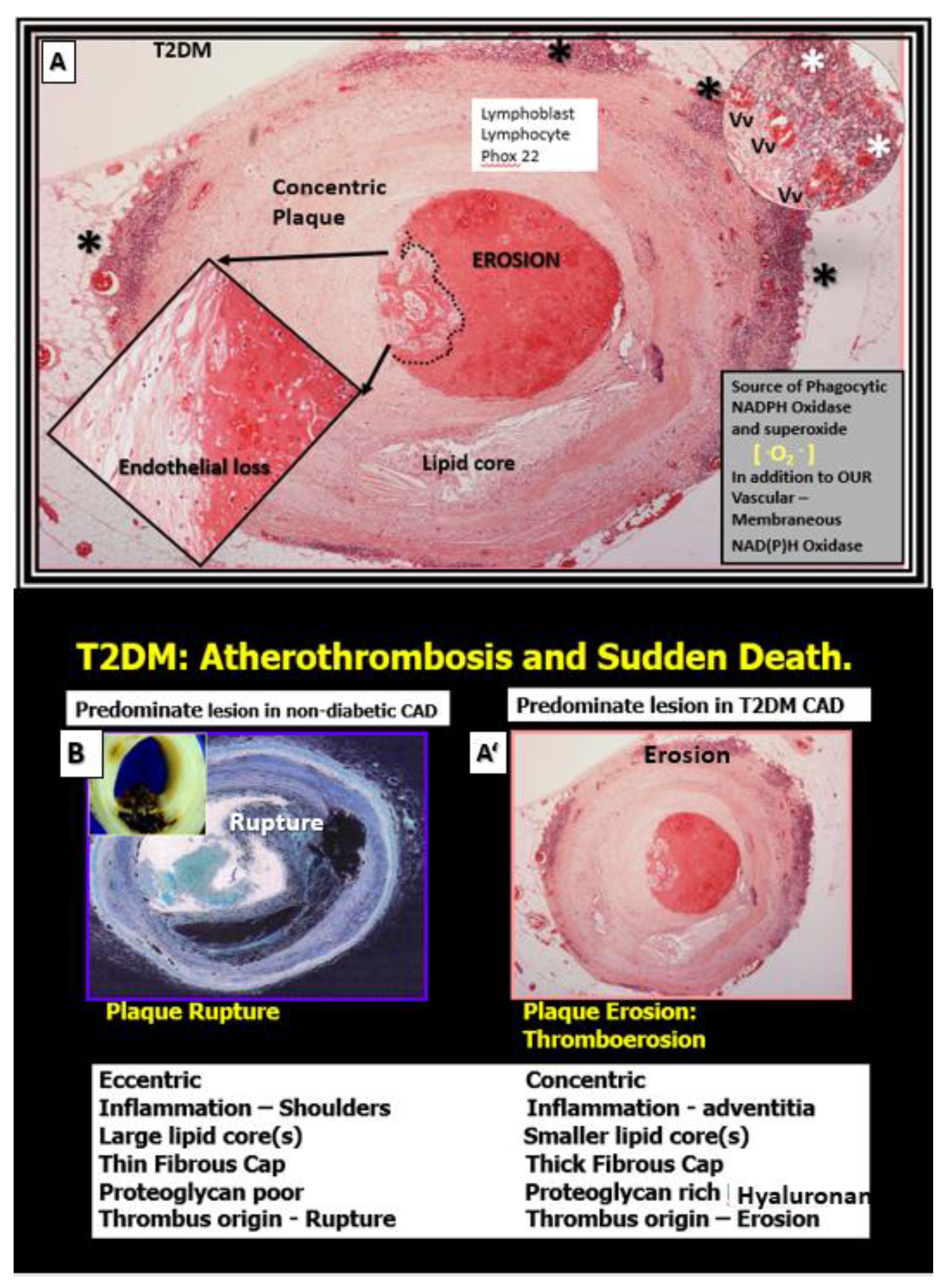
Figure 8.
Potential mechanisms involving uremic toxins and reactive oxygen species (ROS) in vascular calcification. Uremic toxins such as increased parathyroid hormone (PTH), phosphorus (Pi) and phosphate (PO4
3−), calcium, calcium x phosphorus product, vitamin D3, and oxidative redox stress (OxRS) via reactive oxygen species (ROS) significantly contribute to vascular smooth muscle cell (VSMC) differentiation into osteoblast-like phenotypes. Phosphate absorption into these cells is facilitated by the sodium phosphate cotransporter (Pit-1) resulting in an osteogenic switch due to activation of transcription factors: osteoblast-specific cis-acting element (Osf2)—core binding factor alpha1 (Cbfa-1/Runx2). Osteocalcin, osteonectin, bone morphogenic protein-2alpha and alkaline phosphatase (ALP) are also known inducers of calcification. In contrast, the systemic and local inhibitors of calcification fetuin-A (alpha2-Heremans-Schmid glycoprotein (AHSG) and matrix Gla protein (MGP) are decreased in uremia and calciphylaxis. Further, ROS and inflammatory cytokine surges may contribute to decreased hepatic synthesis of fetuin-A (insert a). Uremic toxins—ROS promote uncoupling of endothelial nitric oxide synthase (eNOS) enzyme via the oxidation of the requisite tetrahydrobiopterin (BH4) cofactor and results in the endothelium becoming a net producer of superoxide—ROS (insert b). Additionally, decreased bioavailable NO due to eNOS enzyme uncoupling promotes a proinflammatory, proconstrictive, prothrombotic vascular endothelium and note the constricted collapse arteriole (insert b). OxRS including ROS are also capable of promoting VSMC apoptosis in the arterial vascular wall (AVW) and when this occurs the matrix vesicles and apoptotic bodies serve as nucleating sites for further calcium deposition in the extracellular matrix of the arteriole media (inserts c–e). Image reproduced with permission by CC 4.0 [
68].
Figure 8.
Potential mechanisms involving uremic toxins and reactive oxygen species (ROS) in vascular calcification. Uremic toxins such as increased parathyroid hormone (PTH), phosphorus (Pi) and phosphate (PO4
3−), calcium, calcium x phosphorus product, vitamin D3, and oxidative redox stress (OxRS) via reactive oxygen species (ROS) significantly contribute to vascular smooth muscle cell (VSMC) differentiation into osteoblast-like phenotypes. Phosphate absorption into these cells is facilitated by the sodium phosphate cotransporter (Pit-1) resulting in an osteogenic switch due to activation of transcription factors: osteoblast-specific cis-acting element (Osf2)—core binding factor alpha1 (Cbfa-1/Runx2). Osteocalcin, osteonectin, bone morphogenic protein-2alpha and alkaline phosphatase (ALP) are also known inducers of calcification. In contrast, the systemic and local inhibitors of calcification fetuin-A (alpha2-Heremans-Schmid glycoprotein (AHSG) and matrix Gla protein (MGP) are decreased in uremia and calciphylaxis. Further, ROS and inflammatory cytokine surges may contribute to decreased hepatic synthesis of fetuin-A (insert a). Uremic toxins—ROS promote uncoupling of endothelial nitric oxide synthase (eNOS) enzyme via the oxidation of the requisite tetrahydrobiopterin (BH4) cofactor and results in the endothelium becoming a net producer of superoxide—ROS (insert b). Additionally, decreased bioavailable NO due to eNOS enzyme uncoupling promotes a proinflammatory, proconstrictive, prothrombotic vascular endothelium and note the constricted collapse arteriole (insert b). OxRS including ROS are also capable of promoting VSMC apoptosis in the arterial vascular wall (AVW) and when this occurs the matrix vesicles and apoptotic bodies serve as nucleating sites for further calcium deposition in the extracellular matrix of the arteriole media (inserts c–e). Image reproduced with permission by CC 4.0 [
68].
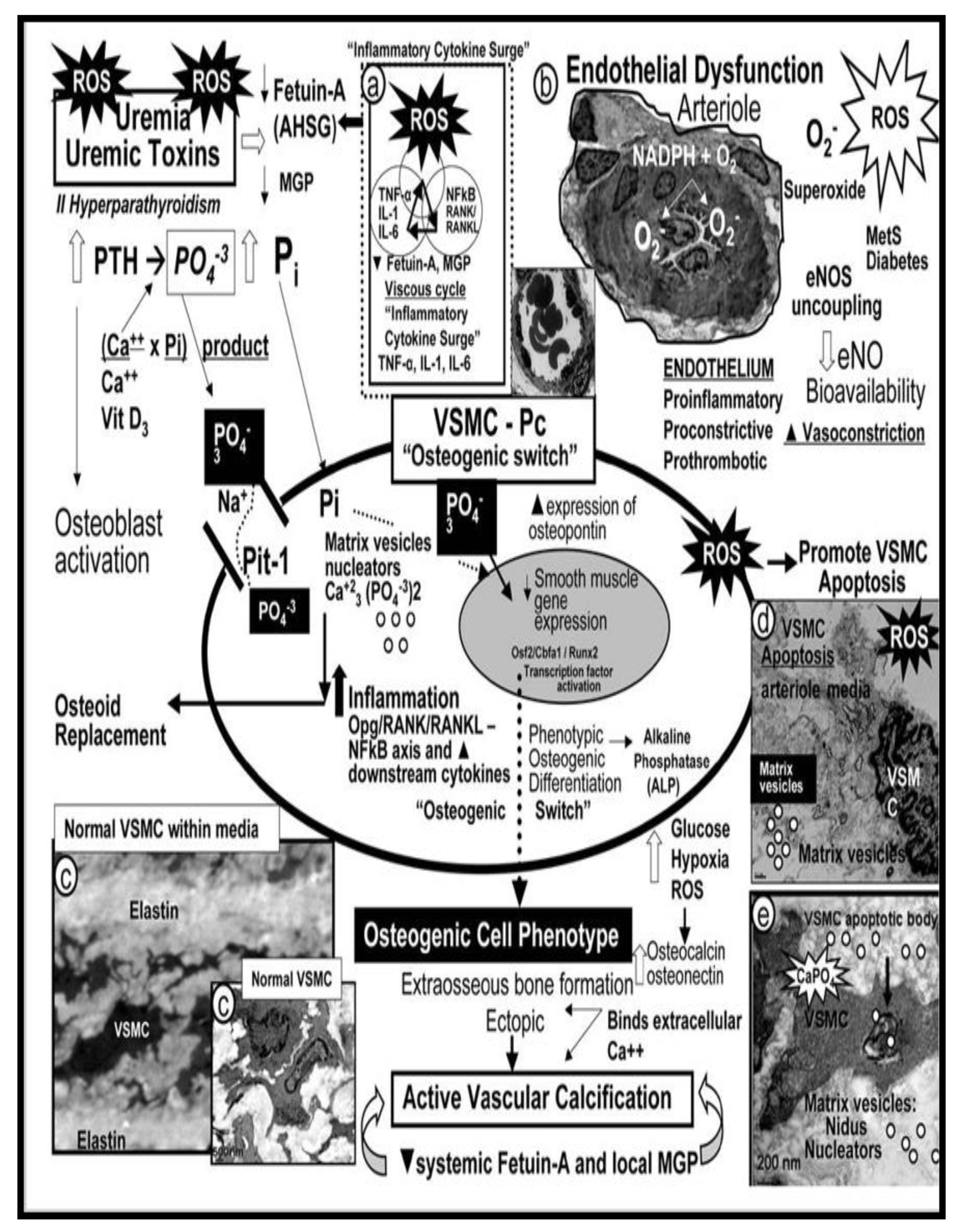
Figure 9.
Double vicious endothelial activation and dysfunction cycles are in play during the development of vascular stiffening and small vessel disease. Panel A depicts the vicious cycle between oxidative redox stress (OxRS), which includes (reactive oxygen (ROS), nitrogen (RONS), sulfur RONSS species of the reactive species interactome (RSI). The systemic endothelial cells (SECs) can be activated via either OxRS or metainflammation induced by peripheral excessive inflammation/metainflammation from the visceral adipose tissue or the visceral perivascular adipose tissue (PVAT) of the vessels as in figures 1 and 4 and gut dysbiosis (pCC, LPS, oxLDL-C plus others). Likewise, this vicious cycle may also be instigated by OxRS. This peripheral inflammation is additionally capable of instigating the vicious cycle in panel B (downward red arrowhead). Panel B depicts the vicious cycle that exists in the development of SECs and/or brain endothelial cells (BECs) activation (proinflammatory endothelium) and in turn cycles with EC dysfunction that is associated with decreased levels of nitric oxide (NO) as a result of endothelial nitric oxide synthase (eNOS) enzyme uncoupling due to a decrease in the essential cofactor of totally reduced BH4, which results from excessive OxRS to oxidize BH4 to BH2 that will not run the eNOS reaction and hence results in eNOS uncoupling making the ECs a net producer of ROS instead of the antioxidant, anti-inflammatory, antithrombotic, antiatherosclerotic, and vasodilation effects of NO in the vasculature. BH2, dihydrobiopterin; BH4, tetrahydrobiopterin; EC, endothelial cell; LPS, lipopolysaccharide; NVU, neurovascular unit; oxLDL-C, oxidized low-density lipoprotein-cholesterol: pCC, peripheral cytokines/chemokines.
Figure 9.
Double vicious endothelial activation and dysfunction cycles are in play during the development of vascular stiffening and small vessel disease. Panel A depicts the vicious cycle between oxidative redox stress (OxRS), which includes (reactive oxygen (ROS), nitrogen (RONS), sulfur RONSS species of the reactive species interactome (RSI). The systemic endothelial cells (SECs) can be activated via either OxRS or metainflammation induced by peripheral excessive inflammation/metainflammation from the visceral adipose tissue or the visceral perivascular adipose tissue (PVAT) of the vessels as in figures 1 and 4 and gut dysbiosis (pCC, LPS, oxLDL-C plus others). Likewise, this vicious cycle may also be instigated by OxRS. This peripheral inflammation is additionally capable of instigating the vicious cycle in panel B (downward red arrowhead). Panel B depicts the vicious cycle that exists in the development of SECs and/or brain endothelial cells (BECs) activation (proinflammatory endothelium) and in turn cycles with EC dysfunction that is associated with decreased levels of nitric oxide (NO) as a result of endothelial nitric oxide synthase (eNOS) enzyme uncoupling due to a decrease in the essential cofactor of totally reduced BH4, which results from excessive OxRS to oxidize BH4 to BH2 that will not run the eNOS reaction and hence results in eNOS uncoupling making the ECs a net producer of ROS instead of the antioxidant, anti-inflammatory, antithrombotic, antiatherosclerotic, and vasodilation effects of NO in the vasculature. BH2, dihydrobiopterin; BH4, tetrahydrobiopterin; EC, endothelial cell; LPS, lipopolysaccharide; NVU, neurovascular unit; oxLDL-C, oxidized low-density lipoprotein-cholesterol: pCC, peripheral cytokines/chemokines.
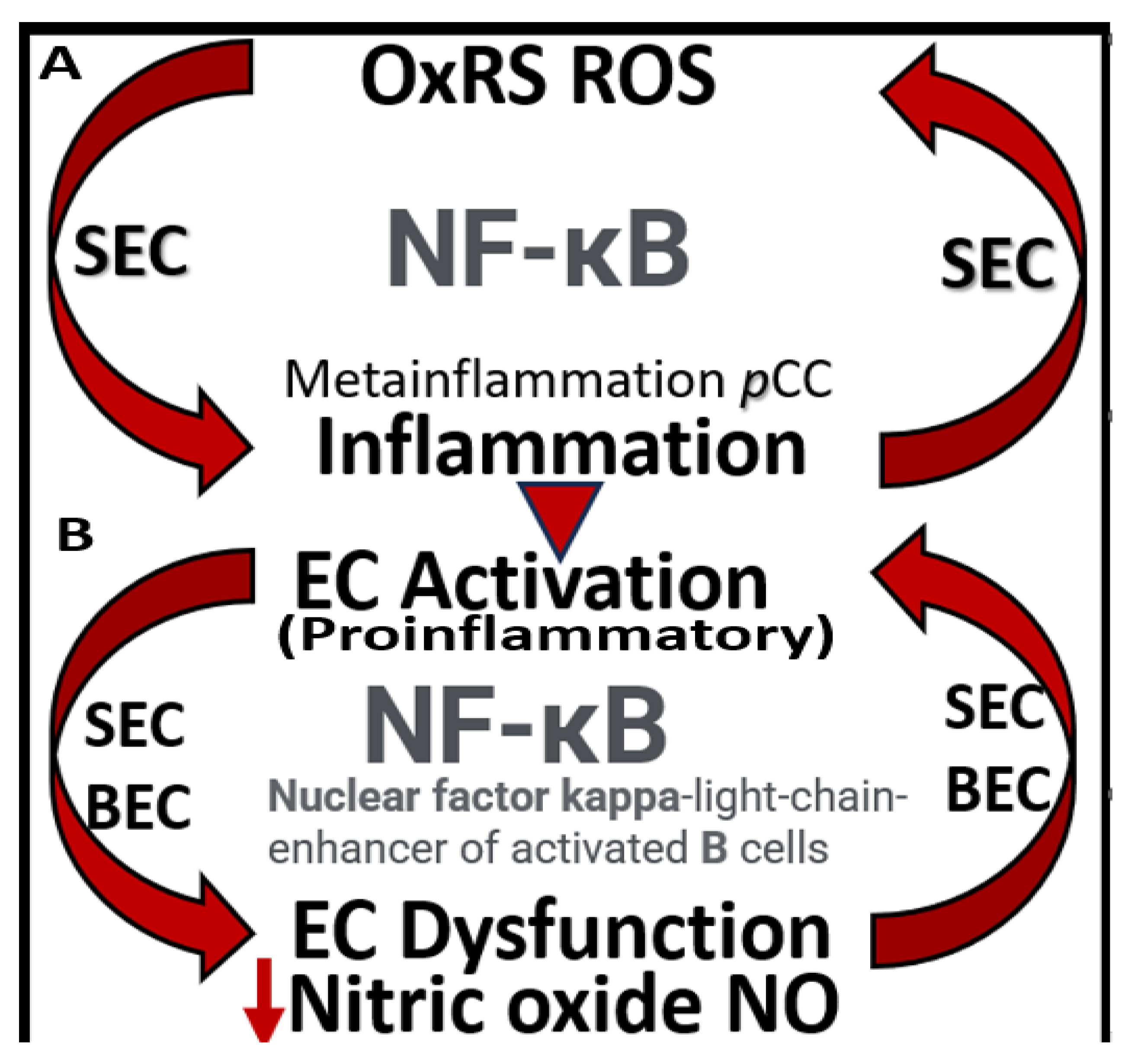
Figure 10.
Systemic endothelial cell and brain endothelial cell activation and dysfunction (BEC
act/dys) associate with and contribute to the evolution of enlarged perivascular spaces (EPVS)-cerebral small vessel disease (SVD), and hypoperfusion/hypometabolism with ischemia and nutrient deficiency to result in neurodegeneration. Mechanisms 1-4 detail and outline the process of developing the formation via BECact/dys through the development of EPVS-SVD and eventual neurodegeneration that include the increase in pulse wave velocity (PWV) and increased pulsatile pulse pressure (pp) that act as an injurious species to result in neurodegeneration via OxRS vicious cycles to result in neuroinflammation and SVD. Note that the injurious stimuli image of the endothelial cell now contains sodium-salt (red box) that results in EC act/dys resulting in a proinflammatory endothelium and a decrease in nitric oxide (NO) due to eNOS uncoupling as a result of the essential cofactor tetrahydrobiopterin (BH4) oxidation. Modified image provided by CC 4.0 [
25]. AGE, advanced glycation end-products; Ang II, angiotensin II; asterisks, denotes and emphasizes that reactive oxygen species are responsible for the activation matrix metalloproteinases (MMPs); BECact/dys, brain endothelial cell activation and dysfunction; BEC, brain endothelial cell; downward arrow, decreased; eNOS, endothelial nitric oxide synthase; ICAM-1, intercellular adhesion molecule; LDL, low density lipoprotein-cholesterol; Lpa, lipoprotein little a; LPS, lipopolysaccharide; MMPs, matrix metalloproteinases; MtROS, mitochondrial reactive oxygen species; NADPH Ox, nicotinamide adenine dinucleotide phosphate reduced oxidase; pnsCC, peripheral nervous system cytokines chemokines RAGE, receptor for AGE: T, transcytosis; upward arrow, increased;VCAM-1. vascular cellular adhesion molecule.
Figure 10.
Systemic endothelial cell and brain endothelial cell activation and dysfunction (BEC
act/dys) associate with and contribute to the evolution of enlarged perivascular spaces (EPVS)-cerebral small vessel disease (SVD), and hypoperfusion/hypometabolism with ischemia and nutrient deficiency to result in neurodegeneration. Mechanisms 1-4 detail and outline the process of developing the formation via BECact/dys through the development of EPVS-SVD and eventual neurodegeneration that include the increase in pulse wave velocity (PWV) and increased pulsatile pulse pressure (pp) that act as an injurious species to result in neurodegeneration via OxRS vicious cycles to result in neuroinflammation and SVD. Note that the injurious stimuli image of the endothelial cell now contains sodium-salt (red box) that results in EC act/dys resulting in a proinflammatory endothelium and a decrease in nitric oxide (NO) due to eNOS uncoupling as a result of the essential cofactor tetrahydrobiopterin (BH4) oxidation. Modified image provided by CC 4.0 [
25]. AGE, advanced glycation end-products; Ang II, angiotensin II; asterisks, denotes and emphasizes that reactive oxygen species are responsible for the activation matrix metalloproteinases (MMPs); BECact/dys, brain endothelial cell activation and dysfunction; BEC, brain endothelial cell; downward arrow, decreased; eNOS, endothelial nitric oxide synthase; ICAM-1, intercellular adhesion molecule; LDL, low density lipoprotein-cholesterol; Lpa, lipoprotein little a; LPS, lipopolysaccharide; MMPs, matrix metalloproteinases; MtROS, mitochondrial reactive oxygen species; NADPH Ox, nicotinamide adenine dinucleotide phosphate reduced oxidase; pnsCC, peripheral nervous system cytokines chemokines RAGE, receptor for AGE: T, transcytosis; upward arrow, increased;VCAM-1. vascular cellular adhesion molecule.
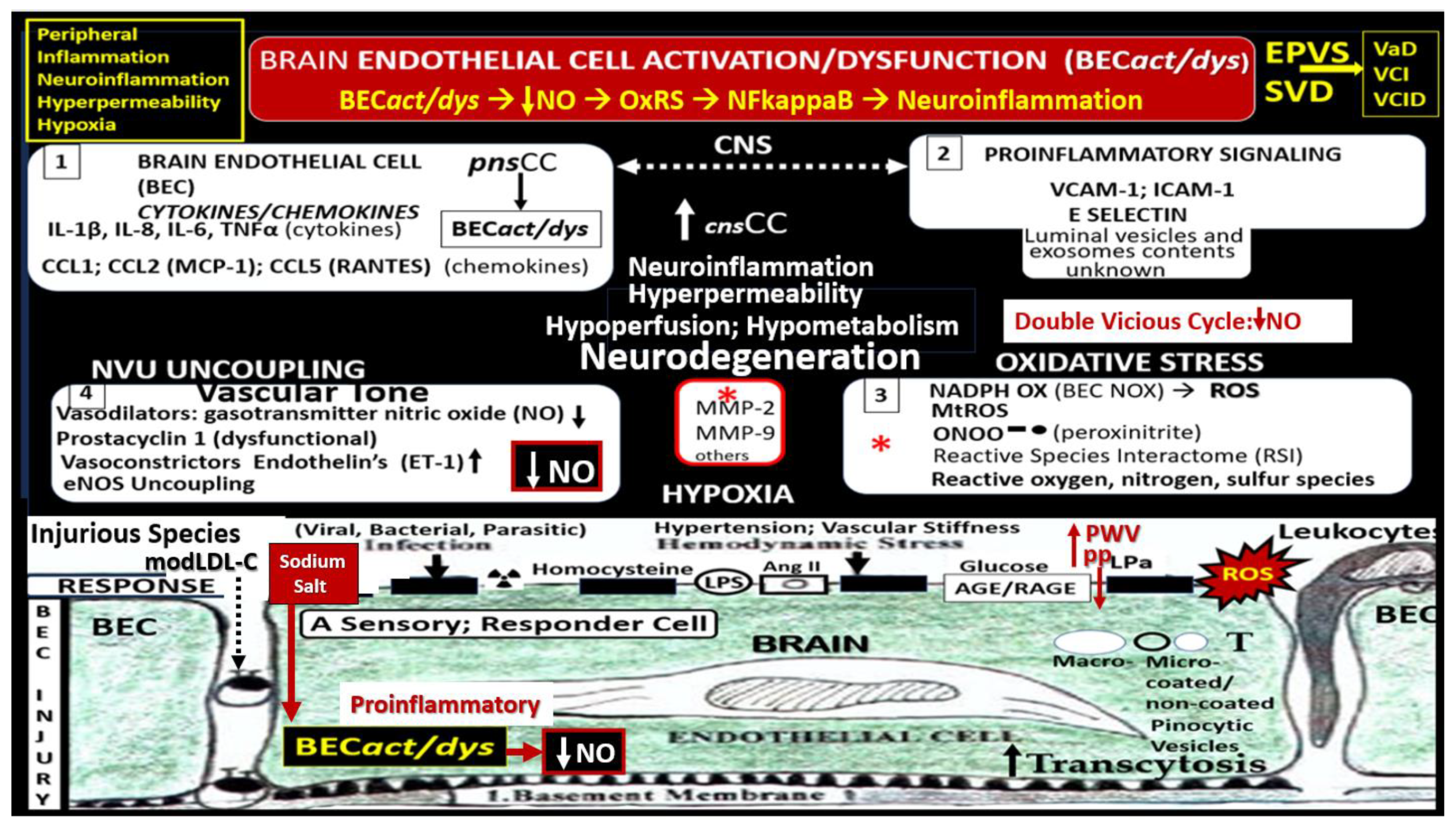
Figure 11.
10 major transmission electron microscopic ultrastructural changes to systemic and brain endothelial cells (SECs and BECs) in various preclinical female rodent models including diet-induced obesity (Western models), obese, insulin resistant, diabetic (
db/db mice), lipopolysaccharide neuroinflammatory male mice, and genetic male hypertensive (transgenic heterozygous (mRen2)27 (Ren2) rats.). Notably, BEC stiffening was determined by atomic force microscopy in number 9. Image provided by CC 4.0 [
25,
26,
27]. ecGCx, endothelial cell glycocalyx; LPS, lipopolysaccharide; mtROS, mitochondrial-derived reactive oxygen species. .
Figure 11.
10 major transmission electron microscopic ultrastructural changes to systemic and brain endothelial cells (SECs and BECs) in various preclinical female rodent models including diet-induced obesity (Western models), obese, insulin resistant, diabetic (
db/db mice), lipopolysaccharide neuroinflammatory male mice, and genetic male hypertensive (transgenic heterozygous (mRen2)27 (Ren2) rats.). Notably, BEC stiffening was determined by atomic force microscopy in number 9. Image provided by CC 4.0 [
25,
26,
27]. ecGCx, endothelial cell glycocalyx; LPS, lipopolysaccharide; mtROS, mitochondrial-derived reactive oxygen species. .
Figure 12.
The “H” phenomenon of the MetS (hyperlipidemia (obesity lipid triad), hyperinsulinemia (IR), hypertension (HTN), and hyperglycemia (impaired glucose tolerance (IGT) +/- type 2 diabetes mellitus (T2DM), as well as, the clustering effect of each of these variables of the MetS itself contribute to extracranial vascular arterial stiffening (VAS). Additionally, IR and LR contribute to vascular stiffening primarily through the actions of obesity on the development of VAS. Modified graphic abstract image provided with permission by CC 4.0 [38Hay]. AAA, abdominal aortic aneurysm; aMt, aberrant mitochondria; CCVD, cerebrocardiovascular disease; CAD, coronary artery disease; CKD, chronic kidney disease; CL, capillary lumen; CVD, cardiovascular disease; BHK, brain-heart-kidney axis; CHF, congestive heart failure; Dd, diastolic dysfunction; EC, endothelial cell; FFA, free fatty acids; H, hyperlipidemia, hyperinsulinemia, hypertension, and hyperglycemia; HCY, homocysteine; HHCY, hyperhomocyteinemia; HVB, heart-vessel-brain axis; IR, insulin resistance; LR, leptin resistance; lncRNAs, long non-coding ribonucleic acids MI, myocardial infarction; miRNAs, micro ribonucleic acids; MΦ, macrophage; NO, nitric oxide; ROS, reactive oxygen species; RONS, reactive oxygen, nitrogen species; RONSS, reactive oxygen, nitrogen, sulfur species; RSI, reactive species interactome; VAS, extracranial vascular arterial stiffness. .
Figure 12.
The “H” phenomenon of the MetS (hyperlipidemia (obesity lipid triad), hyperinsulinemia (IR), hypertension (HTN), and hyperglycemia (impaired glucose tolerance (IGT) +/- type 2 diabetes mellitus (T2DM), as well as, the clustering effect of each of these variables of the MetS itself contribute to extracranial vascular arterial stiffening (VAS). Additionally, IR and LR contribute to vascular stiffening primarily through the actions of obesity on the development of VAS. Modified graphic abstract image provided with permission by CC 4.0 [38Hay]. AAA, abdominal aortic aneurysm; aMt, aberrant mitochondria; CCVD, cerebrocardiovascular disease; CAD, coronary artery disease; CKD, chronic kidney disease; CL, capillary lumen; CVD, cardiovascular disease; BHK, brain-heart-kidney axis; CHF, congestive heart failure; Dd, diastolic dysfunction; EC, endothelial cell; FFA, free fatty acids; H, hyperlipidemia, hyperinsulinemia, hypertension, and hyperglycemia; HCY, homocysteine; HHCY, hyperhomocyteinemia; HVB, heart-vessel-brain axis; IR, insulin resistance; LR, leptin resistance; lncRNAs, long non-coding ribonucleic acids MI, myocardial infarction; miRNAs, micro ribonucleic acids; MΦ, macrophage; NO, nitric oxide; ROS, reactive oxygen species; RONS, reactive oxygen, nitrogen species; RONSS, reactive oxygen, nitrogen, sulfur species; RSI, reactive species interactome; VAS, extracranial vascular arterial stiffness. .

Figure 13.
Media vascular smooth muscle cells (VSMCs), collagen, proteoglycan (PG) and the phenotypic switch from contractile in control models to synthetic phenotypes in the lean hypertensive male transgenic heterozygous (mRen2)27 (Ren2) rats in the upper 1/3 of the descending thoracic aorta at 10-weeks of age. Panel A demonstrates the normal ultrastructure of the endothelium, intima, and media. Note the regular arrangement of internal elastic lamina (IEL) alternating with VSMCs and the elastic lamina (EL) with arrows pointing to aorta endothelial cells. The elastin lamellar unit (dashed yellow oval) is considered to be the basic contractile structural unit of the media. Note that the collagen in control groups is of the loose areolar collagen type. Panel B illustrates the VSMC layer interspersed between the layers of the EL. Panel C depicts the increased organized stiffer collagen 1 in the Ren2 hypertensive models. Panel D also depicts an increase in the matrix proteoglycans (asterisks). Panel E depicts the ‘phenotypic switch’ associated with VAS. The contractile normal phenotype (C) has an abundance of focal adhesion plaques (arrows) that firmly attach to the extracellular matrix (ECM) proteins; however, the phenotypic switch to the synthetic VSMCs (S) have no adhesions and are known to be quite mobile within the media and subintimal spaces of the vascular wall and are also capable of increased synthesis of stiffer collagen and PGN. These synthetic VSMC have a more electron dense cytoplasm that is supportive of increased endoplasmic reticulum (see comparative insets). Also, note the nuclear separation in the synthetic phenotype. Modified images provided with permission by 4.0 [
50]. .
Figure 13.
Media vascular smooth muscle cells (VSMCs), collagen, proteoglycan (PG) and the phenotypic switch from contractile in control models to synthetic phenotypes in the lean hypertensive male transgenic heterozygous (mRen2)27 (Ren2) rats in the upper 1/3 of the descending thoracic aorta at 10-weeks of age. Panel A demonstrates the normal ultrastructure of the endothelium, intima, and media. Note the regular arrangement of internal elastic lamina (IEL) alternating with VSMCs and the elastic lamina (EL) with arrows pointing to aorta endothelial cells. The elastin lamellar unit (dashed yellow oval) is considered to be the basic contractile structural unit of the media. Note that the collagen in control groups is of the loose areolar collagen type. Panel B illustrates the VSMC layer interspersed between the layers of the EL. Panel C depicts the increased organized stiffer collagen 1 in the Ren2 hypertensive models. Panel D also depicts an increase in the matrix proteoglycans (asterisks). Panel E depicts the ‘phenotypic switch’ associated with VAS. The contractile normal phenotype (C) has an abundance of focal adhesion plaques (arrows) that firmly attach to the extracellular matrix (ECM) proteins; however, the phenotypic switch to the synthetic VSMCs (S) have no adhesions and are known to be quite mobile within the media and subintimal spaces of the vascular wall and are also capable of increased synthesis of stiffer collagen and PGN. These synthetic VSMC have a more electron dense cytoplasm that is supportive of increased endoplasmic reticulum (see comparative insets). Also, note the nuclear separation in the synthetic phenotype. Modified images provided with permission by 4.0 [
50]. .

Figure 14.
Descending thoracic aorta endothelial cell lifting and separation from the internal elastic lamina (IEL) and IEL and elastin lamella fragmentation in the female obese insulin resistant diabetic
db/db mouse models at 20-weeks of age. Panel A demonstrates the normal tight adherence of the aortic endothelial cells to the underlying IEL in control models. Panel B depicts the abnormal structural remodeling of EC lifting and separation from the IEL in the obese, diabetic
db/db models at 10-weeks of age. Panel C depicts the elastolysis of the IEL (outlined by white dashed line) that provides a space in which there resides elastin-derived fragments (a rich source of elastin-derived peptides (EDPs)) in the subendothelial space in the db/db models. Panel D also depicts the EC separation and lifting from the IEL with evidence of elastolysis and elastin fragments. Panel E demonstrates the intact elastin lamina (EL) adjacent to a VSMC with its homogeneous electron dense construct. Panel F depicts elastin lamella that has undergone elastin degradation with separation of elastin filaments with degradation. Importantly once elastin is fragmented or undergoes splitting-separation it loses its ability to lengthen and its important ability to undergo its recoil functions to resume its original length with increased stiffness and less compliance. Revised images provided with permission by 4.0 [
94]. .
Figure 14.
Descending thoracic aorta endothelial cell lifting and separation from the internal elastic lamina (IEL) and IEL and elastin lamella fragmentation in the female obese insulin resistant diabetic
db/db mouse models at 20-weeks of age. Panel A demonstrates the normal tight adherence of the aortic endothelial cells to the underlying IEL in control models. Panel B depicts the abnormal structural remodeling of EC lifting and separation from the IEL in the obese, diabetic
db/db models at 10-weeks of age. Panel C depicts the elastolysis of the IEL (outlined by white dashed line) that provides a space in which there resides elastin-derived fragments (a rich source of elastin-derived peptides (EDPs)) in the subendothelial space in the db/db models. Panel D also depicts the EC separation and lifting from the IEL with evidence of elastolysis and elastin fragments. Panel E demonstrates the intact elastin lamina (EL) adjacent to a VSMC with its homogeneous electron dense construct. Panel F depicts elastin lamella that has undergone elastin degradation with separation of elastin filaments with degradation. Importantly once elastin is fragmented or undergoes splitting-separation it loses its ability to lengthen and its important ability to undergo its recoil functions to resume its original length with increased stiffness and less compliance. Revised images provided with permission by 4.0 [
94]. .

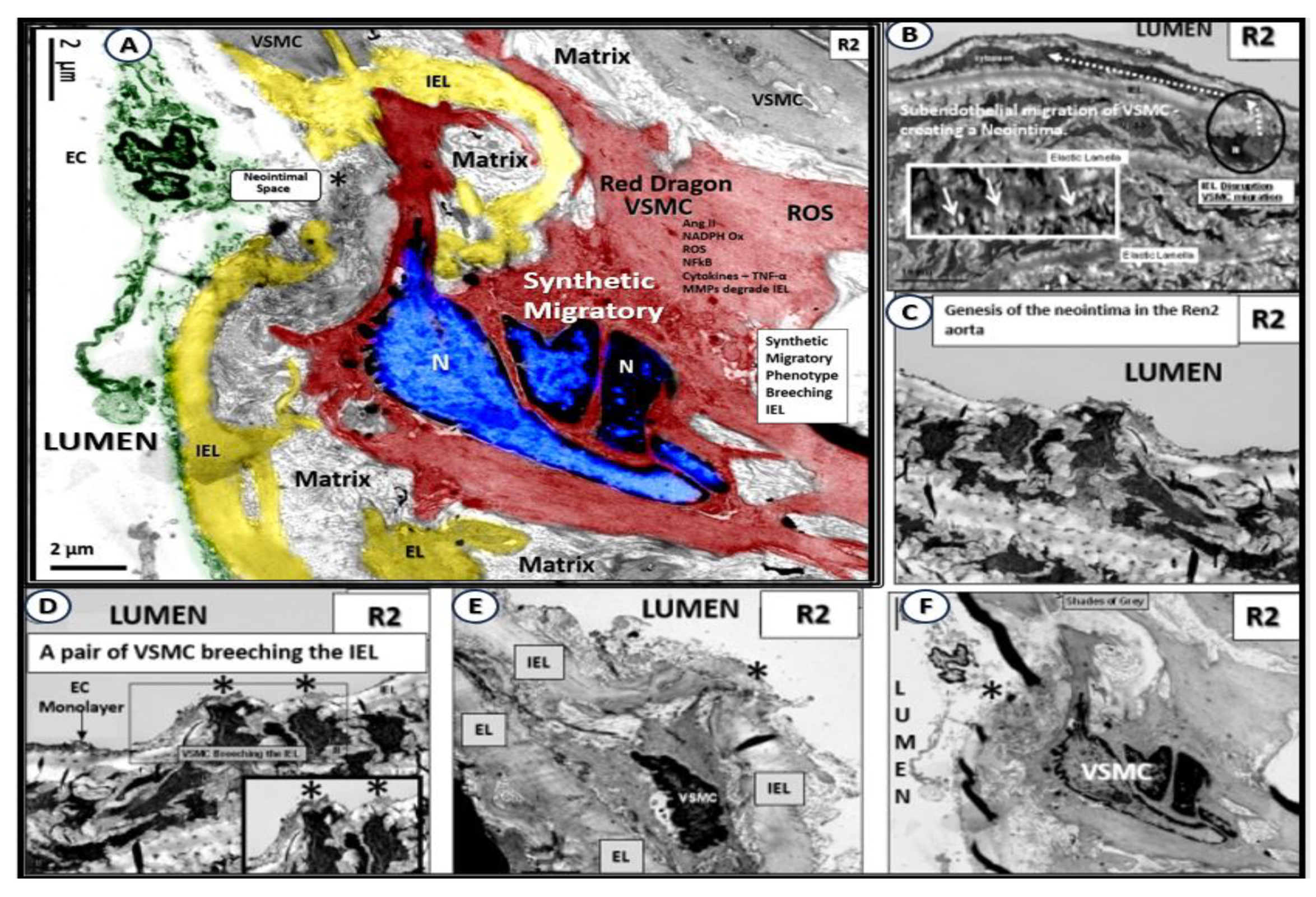
Figure 15.
Genesis of the neointima via the synthetic, migratory vascular smooth muscle cell (VSMC) in the lean hypertensive male transgenic heterozygous (mRen2)27 (Ren2) rat at 10-weeks of age. Panel A depicts the migratory synthetic vascular smooth muscle cell (VSMC) (pseudo-colored red) migrating to the internal elastic lamina (IEL) with elastolysis and breeching of the IEL (pseudo-colored yellow) to form the neointima. Also, note the lifting and separation of the aorta endothelial cell (pseudo-colored green). Panels B through F also depict the migration of VSMCs to create and reside within the neointimal space. Note that panel F is the original TEM of panel A that is pseudo-colored red and nicknamed the ‘red dragon’. Asterisks, denote the VSMCs creating and entering the neointimal-subintimal space. Modified image provided with permission by CC 4.0 [
50].
Figure 15.
Genesis of the neointima via the synthetic, migratory vascular smooth muscle cell (VSMC) in the lean hypertensive male transgenic heterozygous (mRen2)27 (Ren2) rat at 10-weeks of age. Panel A depicts the migratory synthetic vascular smooth muscle cell (VSMC) (pseudo-colored red) migrating to the internal elastic lamina (IEL) with elastolysis and breeching of the IEL (pseudo-colored yellow) to form the neointima. Also, note the lifting and separation of the aorta endothelial cell (pseudo-colored green). Panels B through F also depict the migration of VSMCs to create and reside within the neointimal space. Note that panel F is the original TEM of panel A that is pseudo-colored red and nicknamed the ‘red dragon’. Asterisks, denote the VSMCs creating and entering the neointimal-subintimal space. Modified image provided with permission by CC 4.0 [
50].
Figure 16.
Panel A illustrates the heart-vessel-brain (HVB) axis and vascular arterial stiffness (VAS) contributions to small vessel disease (SVD) via the damaging pulsatile pulse pressure (pp) to the carotids, vertebral (V) and basilar arteries, and the circle of Willis (C of W) to the central nervous system (CNS) microvessels. Panel B depicts cardio-cerebrovascular diseases including stroke (ischemic or hemorrhagic) that associate with and contribute to brain endothelial cell activation and dysfunction (BEC
act/dys) with concurrent blood–brain barrier dysfunction and disruption (BBB
dd) induced ischemia, hypoxia, and chronic cerebral hypoperfusion (CCH) and hypometabolism via oxidative redox stress (OxRS) and neuroinflammation of the microvessels to result in neurodegeneration, impaired cognition, and dementia in LOAD and MD. Importantly, note how VCID and the brain are centrally placed within the overlapping Venn diagrams representing cardio-cerebrovascular disease (in red circle) and progressive neurodegeneration, cognitive impairment, LOAD (Alzheimer’s dementia), and mixed dementias (LOAD plus VAD) in aging individuals (in blue circle). Modified image in panel B provided with permission by CC 4.0 [
27]. CCA, common carotid arteries; ICA, internal carotid; T, thoracic; VAD, vascular dementia.
Figure 16.
Panel A illustrates the heart-vessel-brain (HVB) axis and vascular arterial stiffness (VAS) contributions to small vessel disease (SVD) via the damaging pulsatile pulse pressure (pp) to the carotids, vertebral (V) and basilar arteries, and the circle of Willis (C of W) to the central nervous system (CNS) microvessels. Panel B depicts cardio-cerebrovascular diseases including stroke (ischemic or hemorrhagic) that associate with and contribute to brain endothelial cell activation and dysfunction (BEC
act/dys) with concurrent blood–brain barrier dysfunction and disruption (BBB
dd) induced ischemia, hypoxia, and chronic cerebral hypoperfusion (CCH) and hypometabolism via oxidative redox stress (OxRS) and neuroinflammation of the microvessels to result in neurodegeneration, impaired cognition, and dementia in LOAD and MD. Importantly, note how VCID and the brain are centrally placed within the overlapping Venn diagrams representing cardio-cerebrovascular disease (in red circle) and progressive neurodegeneration, cognitive impairment, LOAD (Alzheimer’s dementia), and mixed dementias (LOAD plus VAD) in aging individuals (in blue circle). Modified image in panel B provided with permission by CC 4.0 [
27]. CCA, common carotid arteries; ICA, internal carotid; T, thoracic; VAD, vascular dementia.
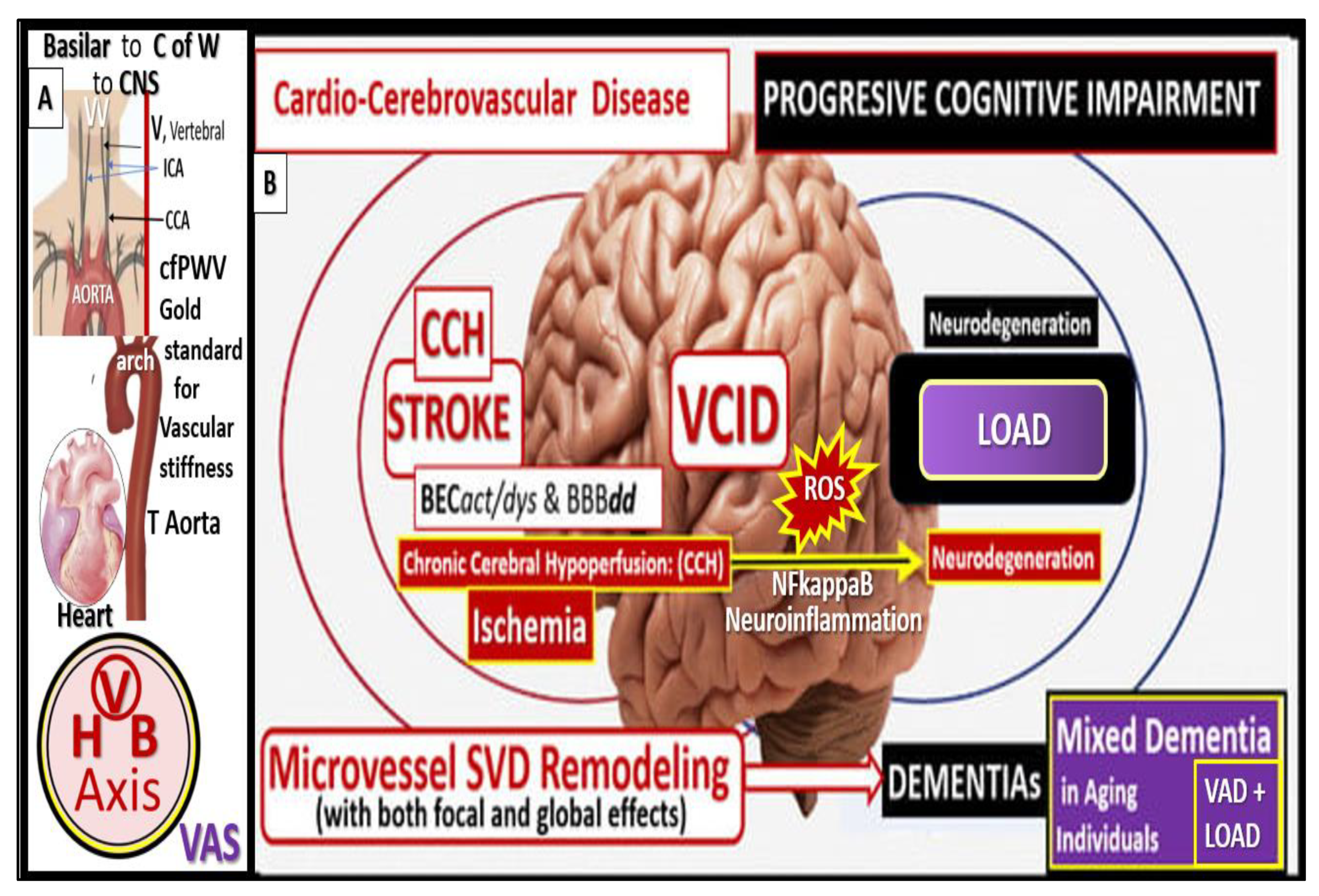
Figure 17.
Possible sequence of events involving increased pulse wave velocity (PWV) and increased pulsatile pulse pressure (pp) of extracranial vascular stiffening (VAS) to brain microvessels and subsequent neurodegeneration. The injurious stimuli of increased pp (upper left-hand insert of a cleaned capillary from the
db/db female diabetic model; red arrows) are due to the direct effect of extracranial vascular arterial stiffening (VAS) and this injury to the brain endothelial cell(s) (BECs) results in a response to injury wound healing mechanism of BEC, hyperlucency, and thickening of the basement membrane (upper left). This injury also associates with increased oxidative redox stress (OxRS) via reactive oxygen species (ROS) and the reactive species interactome (RSI) and neuroinflammation that gives rise to BEC activation and dysfunction (BEC
act/dys) and blood-brain barrier dysfunction and disruption (BBB
dd). Importantly BECact/dys results in neuroinflammation and decreased nitric oxide (NO) bioavailability in addition to increased neurovascular unit (NVU) permeability and NVU uncoupling. The NVU uncoupling gives rise to chronic cerebral hypoperfusion (CCH) and hypometabolism to regional neurons with resulting neurodegeneration, impaired cognition with mild cognitive impairment (MCI), dementia, and late-onset Alzheimer’s disease (LOAD). Concurrently, the cerebral small vessel disease (SVD) that associates with BEC
act/dys and BBB
dd, eNOS uncoupling, decreased NO and endothelial nitric oxide synthase (eNOS) uncoupling can also give rise to neurodegeneration. Additionally, VAS may potentiate amyloid and tau accumulation and deposition in the brain through pulsatile damage to not only the NVU capillaries but also to the perivascular spaces and contribute to enlarged perivascular spaces that associate with impaired glymphatic waste removal of toxic oligomers of Aβ and tau [
29]. Aβ, amyloid beta; BH2, dihydrobiopterin; BH4, tetrahydrobiopterin; CL, capillary lumen; CMBs, cerebral microbleeds; EPVS, enlarged perivascular spaces; HVB, heart-vessel-vascular-brain axis (upper right-hand logo); MD, mixed dementia; MRI, magnetic resonance imaging; NADPH Ox, nicotinamide adenine dinucleotide phosphate oxidase reduced; RSI, reactive species interactome; VAD, vascular dementia; WMH, white mater hyperintensities. .
Figure 17.
Possible sequence of events involving increased pulse wave velocity (PWV) and increased pulsatile pulse pressure (pp) of extracranial vascular stiffening (VAS) to brain microvessels and subsequent neurodegeneration. The injurious stimuli of increased pp (upper left-hand insert of a cleaned capillary from the
db/db female diabetic model; red arrows) are due to the direct effect of extracranial vascular arterial stiffening (VAS) and this injury to the brain endothelial cell(s) (BECs) results in a response to injury wound healing mechanism of BEC, hyperlucency, and thickening of the basement membrane (upper left). This injury also associates with increased oxidative redox stress (OxRS) via reactive oxygen species (ROS) and the reactive species interactome (RSI) and neuroinflammation that gives rise to BEC activation and dysfunction (BEC
act/dys) and blood-brain barrier dysfunction and disruption (BBB
dd). Importantly BECact/dys results in neuroinflammation and decreased nitric oxide (NO) bioavailability in addition to increased neurovascular unit (NVU) permeability and NVU uncoupling. The NVU uncoupling gives rise to chronic cerebral hypoperfusion (CCH) and hypometabolism to regional neurons with resulting neurodegeneration, impaired cognition with mild cognitive impairment (MCI), dementia, and late-onset Alzheimer’s disease (LOAD). Concurrently, the cerebral small vessel disease (SVD) that associates with BEC
act/dys and BBB
dd, eNOS uncoupling, decreased NO and endothelial nitric oxide synthase (eNOS) uncoupling can also give rise to neurodegeneration. Additionally, VAS may potentiate amyloid and tau accumulation and deposition in the brain through pulsatile damage to not only the NVU capillaries but also to the perivascular spaces and contribute to enlarged perivascular spaces that associate with impaired glymphatic waste removal of toxic oligomers of Aβ and tau [
29]. Aβ, amyloid beta; BH2, dihydrobiopterin; BH4, tetrahydrobiopterin; CL, capillary lumen; CMBs, cerebral microbleeds; EPVS, enlarged perivascular spaces; HVB, heart-vessel-vascular-brain axis (upper right-hand logo); MD, mixed dementia; MRI, magnetic resonance imaging; NADPH Ox, nicotinamide adenine dinucleotide phosphate oxidase reduced; RSI, reactive species interactome; VAD, vascular dementia; WMH, white mater hyperintensities. .
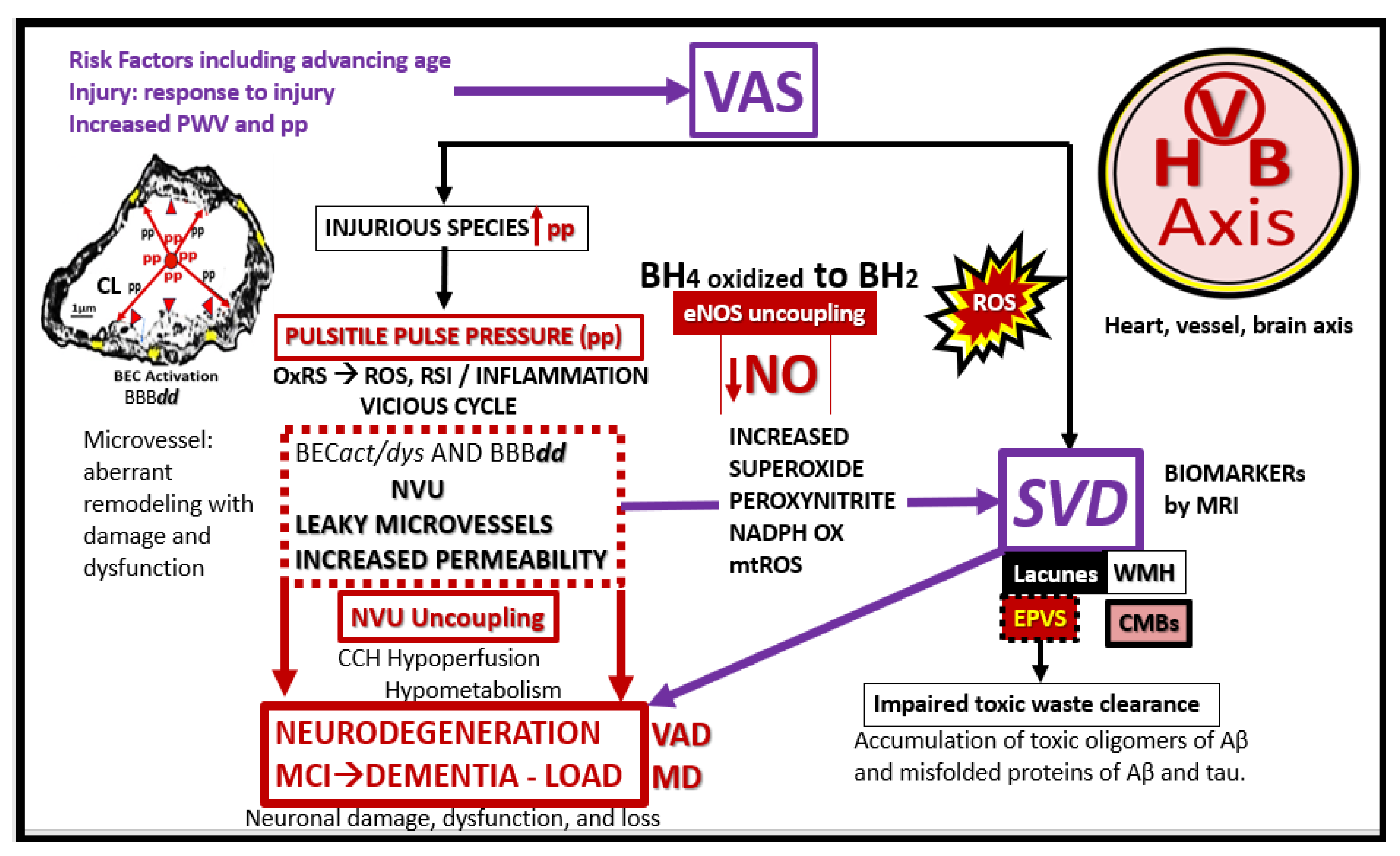
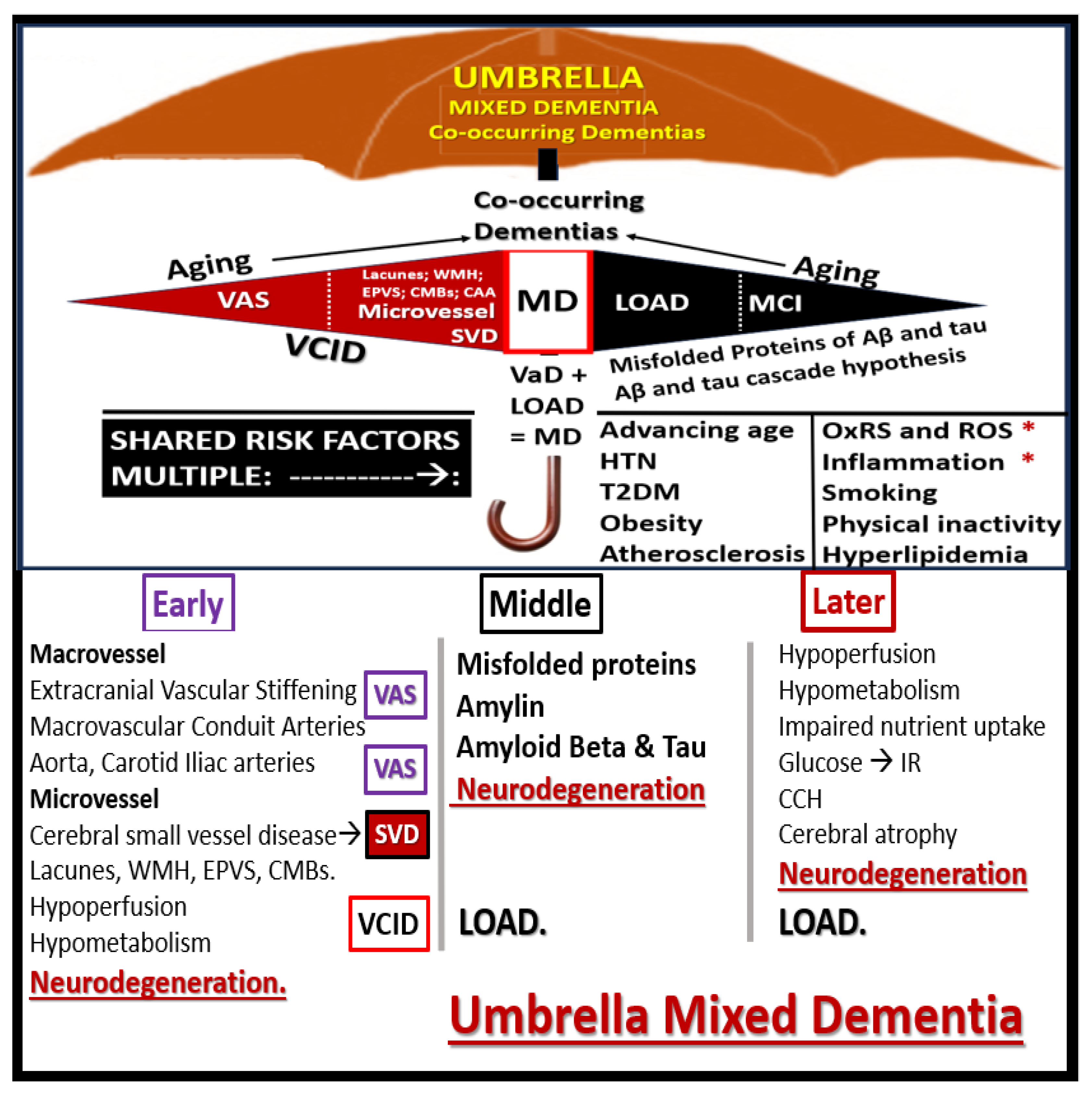
Figure 18.
An umbrella overarching approach of mixed dementias including the vascular contributions to impaired cognition and dementia (VCID). Importantly, the shared risk factors for each of these spectrum diseases (VCID, VaD and LOAD) may help to explain their shared coexistence in mixed co-occurring dementias under the umbrella. Below the umbrella spectrum diseases of mixed dementias illustration these processes are divided into early, middle, and later manifestations in the development of dementias as follows: Early remodeling neurovascular remodeling hypothesis (vascular remodeling changes occur prior to the deposition of amyloid beta); middle amyloid beta cascade and tau hypothesis; later hypoperfusion-hypometabolism with impaired glucose/nutrient uptake relationship to chronic cerebral hypoperfusion and hypometabolism to result in neuronal dysfunction, impaired cognition, neurodegeneration, and LOAD. Even though MCI is placed on the neurodegenerative blackened side of the spectrum, it may also occur on the left-hand side of the red spectrum. Concepts utilized in this modified illustration were provided with permission by CC 4.0 [
104]. Aβ, amyloid beta; asterisks, indicate emphasis; CAA, cerebral amyloid angiopathy; CCH, chronic cerebral hypoperfusion; CMBs, cerebral microbleeds; EPVS, enlarged perivascular spaces; HTN, hypertension; IR, insulin resistance; OxRS, oxidative redox stress; ROS, reactive oxygen species; T2DM, type 2 diabetes mellitus; WMH, white matter hyperintensities.
Figure 18.
An umbrella overarching approach of mixed dementias including the vascular contributions to impaired cognition and dementia (VCID). Importantly, the shared risk factors for each of these spectrum diseases (VCID, VaD and LOAD) may help to explain their shared coexistence in mixed co-occurring dementias under the umbrella. Below the umbrella spectrum diseases of mixed dementias illustration these processes are divided into early, middle, and later manifestations in the development of dementias as follows: Early remodeling neurovascular remodeling hypothesis (vascular remodeling changes occur prior to the deposition of amyloid beta); middle amyloid beta cascade and tau hypothesis; later hypoperfusion-hypometabolism with impaired glucose/nutrient uptake relationship to chronic cerebral hypoperfusion and hypometabolism to result in neuronal dysfunction, impaired cognition, neurodegeneration, and LOAD. Even though MCI is placed on the neurodegenerative blackened side of the spectrum, it may also occur on the left-hand side of the red spectrum. Concepts utilized in this modified illustration were provided with permission by CC 4.0 [
104]. Aβ, amyloid beta; asterisks, indicate emphasis; CAA, cerebral amyloid angiopathy; CCH, chronic cerebral hypoperfusion; CMBs, cerebral microbleeds; EPVS, enlarged perivascular spaces; HTN, hypertension; IR, insulin resistance; OxRS, oxidative redox stress; ROS, reactive oxygen species; T2DM, type 2 diabetes mellitus; WMH, white matter hyperintensities.