Submitted:
05 January 2025
Posted:
06 January 2025
You are already at the latest version
Abstract
Immunotherapy using dendritic cell-derived exosomes (DEXs) represents an innovative strategy for cancer treatment. While cur-rent clinical monitoring methods, such as RECIST and iRECIST, provide tools for evaluating outcomes through imaging and clin-ical parameters, there remains a gap in comprehensive molecular evaluation of immunotherapy quality. This work reviews exist-ing tools and develops an integrated design that structures these tools into a systematic monitoring protocol to optimize the de-velopment of DEX-based therapies. Methods: The protocol includes flow cytometry for immune characterization, ELISA for cy-tokine profiling (e.g., IFN-γ and IL-12), and Western blotting for exosomal markers (CD63, CD9, and CD81). These molecular tools complement current clinical evaluations, providing comprehensive insights into the quality of DEXs and patient immune responses. Results: Molecular analyses validated the consistency and reproducibility of DEXs through stable expression of exo-somal markers. Furthermore, cytokine profiling revealed significant increases in Th1 responses, including elevated levels of IFN-γ and IL-12, correlated with enhanced T cell activation and tumor cell apoptosis. Conclusions: This work not only reviews existing tools and methodologies but also proposes an original design that organizes and structures these tools into an integral monitor-ing protocol. This system connects clinical and molecular data, optimizing precision, reproducibility, and personalization in DEX-based treatments, establishing a robust foundation to enhance the impact of immunotherapy in oncopathology.
Keywords:
1. Introduction
2. Bases of Monitoring in DC Immunotherapy
3. Optimization and Characterization in the Molecular Laboratory
3.1. Optimizing Progenitor Cell Isolation and DC Differentiation
3.2. Structural and Functional Characterization of Exosomes
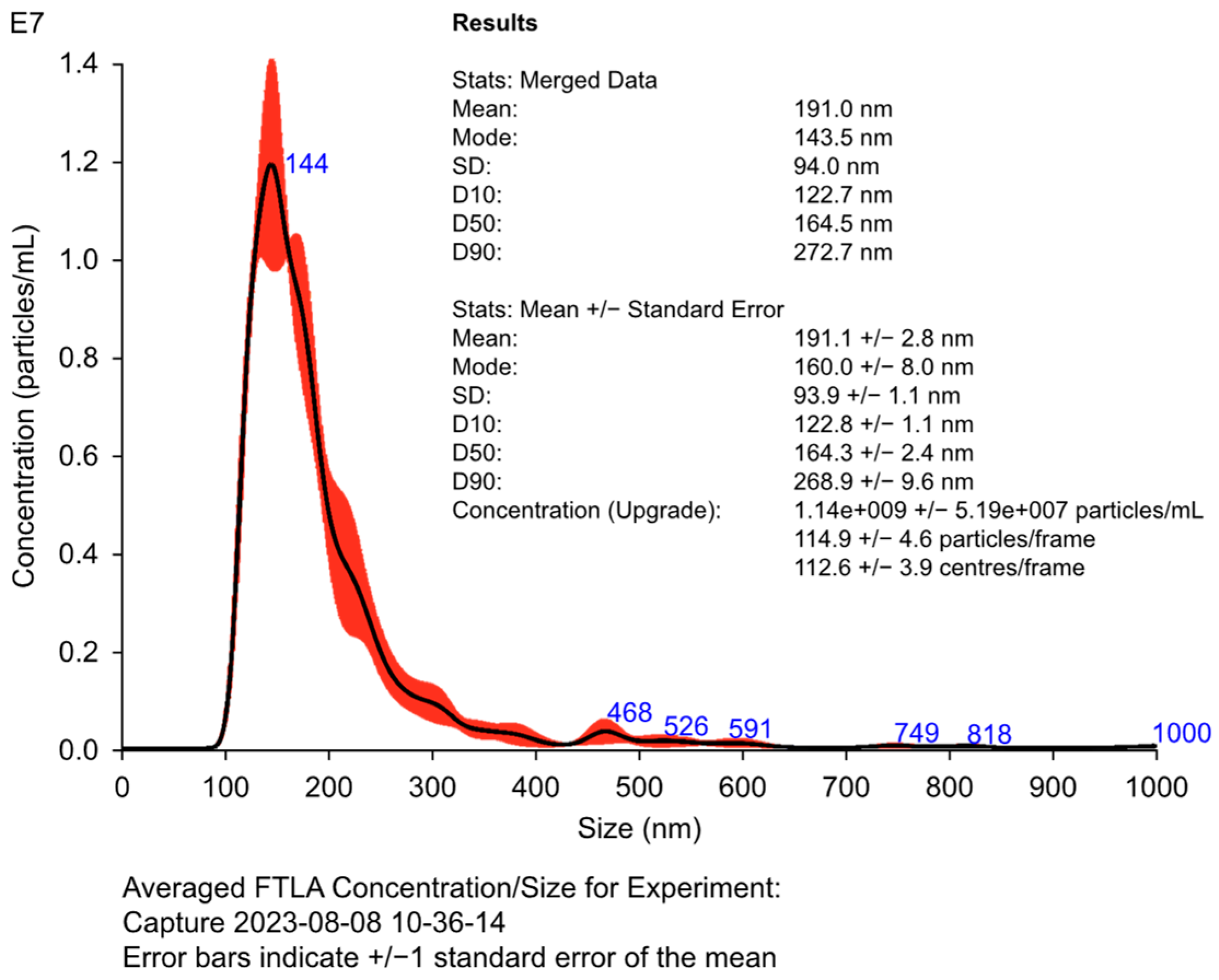
3.3. Advanced Quality and Functionality Assessment
4. Immune Monitoring Protocol
5. Complementary Clinical Follow-Up
6. Impact of Laboratory Results on Treatment Personalization
6.1. T-Cell Activation and Treatment Adjustments
6.2. Th1, Th2, and Th17 Immune Profiles: Influence on Response
6.3. Evaluation of the Quality and Functionality of Exosomes
6.4. Adjustments in Immunotherapy Administration
7. Cost Analysis and Protocol Scalability
7.1. Cost Analysis
7.2. Scalability
8. Conclusions
9. Limitations and Future Directions
9.1. Limitations in the Immune Response
9.2. Future Directions in Research
9.3. Interdisciplinary Collaborations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DC | Dendritic Cell |
| DEX | Dendritic Cell-Derived Exosome |
| PBMC | Peripheral Blood Mononuclear Cell |
| GM-CSF | Granulocyte–Macrophage Colony-Stimulating Factor |
| TNF-α | Tumor Necrosis Factor Alpha |
| IL-4 | Interleukin 4 |
| IL-1β | Interleukin 1 Beta |
| RECIST | Response Evaluation Criteria in Solid Tumors |
| iRECIST | Immune Response Evaluation Criteria in Solid Tumors |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| PET-CT | Positron Emission Tomography–Computed Tomography |
| CEA | Carcinoembryonic Antigen |
| CA-125 | Cancer Antigen 125 |
| PSA | Prostate-Specific Antigen |
| CD | Cluster of Differentiation |
| Th1, Th2, Th17 | T Helper Cell Subtypes |
| IFN-γ | Interferon Gamma |
| LDH | Lactate Dehydrogenase |
| NTA | Nanoparticle Tracking Analysis |
| HLA-DR | Human Leukocyte Antigen–DR Isotype |
| CD80, CD83, CD63, CD81 | Cell Differentiation Markers |
| TLR | Toll-Like Receptor |
| FSC-A | Forward Scatter Area |
| SSC-A | Side Scatter Area |
| APC-A | Allophycocyanin Area |
| PE | Phycoerythrin |
| CBA | Cytometric Bead Array |
| pg/mL | Picograms per milliliter |
References
- Zhang, H.; Wang, S.; Sun, M.; Cui, Y.; Xing, J.; Teng, L.; Xi, Z.; Yang, Z. Exosomes as smart drug delivery vehicles for cancer immunotherapy. Front. Immunol. 2023, 13, 1093607. [CrossRef]
- Lee KW; Yam JWP; Mao X. Dendritic cell vaccines: A shift from conventional approach to new generations. Cells 2023, 12, 2147. [CrossRef]
- Tojjari A; Saeed A; Singh M; Cavalcante L; Sahin IH; Saeed A. A comprehensive review on cancer vaccines and vaccine strategies in hepatocellular carcinoma. Vaccines 2023, 11, 1357. [CrossRef]
- Gardner A; de Mingo Pulido Á; Ruffell B. Dendritic cells and their role in immunotherapy. Front. Immunol. 2020, 11, 924. [CrossRef]
- Cai Y; Prochazkova M; Kim YS; Jiang C; Ma J; Moses L; et al. Assessment and comparison of viability assays for cellular products. Cytotherapy 2024, 26, 201–209. [CrossRef]
- Seymour L; Bogaerts J; Perrone A; Ford R; Schwartz LH; Mandrekar S; Lin NU; Litière S; Dancey J; Chen A; Hodi FS; Therasse P; Hoekstra OS; Shankar LK; Wolchok JD; Ballinger M; Caramella C; de Vries EGE; RECIST working group. iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol. 2017, 18, e143-e152.
- Vladimirov N; Perlman O. Molecular MRI-based monitoring of cancer immunotherapy treatment response. Int. J. Mol. Sci. 2023, 24, 3151. [CrossRef]
- Gu YZ; Zhao X; Song XR. Ex vivo pulsed dendritic cell vaccination against cancer. Acta Pharmacol. Sin. 2020, 41, 959-969. [CrossRef]
- Liu C; Yang M; Zhang D; Chen M; Zhu D. Clinical cancer immunotherapy: Current progress and prospects. Front. Immunol. 2022, 13, 961805. [CrossRef]
- Connor L; Dean J; McNett M; Tydings DM; Shrout A; Gorsuch PF; Hole A; Moore L; Brown R; Melnyk BM; Gallagher-Ford L. Evidence-based practice improves patient outcomes and healthcare system return on investment: Findings from a scoping review. Worldviews Evid. Based Nurs. 2023, 20, 6-15. [CrossRef]
- Couchoud C; Fagnoni P; Aubin F; Westeel V; Maurina T; Thiery-Vuillemin A; Gerard C; Kroemer M; Borg C; Limat S; Nerich V. Economic evaluations of cancer immunotherapy: a systematic review and quality evaluation. Cancer Immunol. Immunother. 2020, 69, 1947-1958.
- Singh S; Paul D; Nath V. Exosomes: Current knowledge and future perspectives. Tissue Barriers 2024, 12, 2232248. [CrossRef]
- Marques HS; de Brito BB; da Silva FAF; Santos MLC; de Souza JCB; Correia TML; Lopes LW; Neres NSM; Dórea RSDM; Dantas ACS; Morbeck LLB; Lima IS; de Almeida AA; Dias MRJ; de Melo FF. Relationship between Th17 immune response and cancer. World J. Clin. Oncol. 2021, 12, 845-867.
- Masucci M; Karlsson C; Blomqvist L; Ernberg I. Bridging the divide: A review on the implementation of personalized cancer medicine. J. Pers. Med. 2024, 14, 561. [CrossRef]
- Jin P; Han TH; Ren J; et al. Molecular signatures of maturing dendritic cells: implications for testing the quality of dendritic cell therapies. J. Transl. Med. 2010, 8, 4. [CrossRef]
- Leng SX; McElhaney JE; Walston JD; Xie D; Fedarko NS; Kuchel GA. ELISA and multiplex technologies for cytokine measurement in inflammation and aging research. J. Gerontol. A Biol. Sci. Med. Sci. 2008, 63, 879-884. [CrossRef]
- Mucherino S; Lorenzoni V; Orlando V; Triulzi I; Del Re M; Capuano A; Danesi R; Turchetti G; Menditto E. Cost-effectiveness of treatment optimisation with biomarkers for immunotherapy in solid tumours: a systematic review protocol. BMJ Open 2021, 11, e048141. [CrossRef]
- Liu R; Zhao Y; Shi F; Zhu J; Wu J; Huang M; Qiu K. Cost-effectiveness analysis of immune checkpoint inhibitors as first-line therapy in advanced biliary tract cancer. Immunotherapy 2024, 16, 669-678. [CrossRef]
- Gutierrez-Sandoval R; Gutierrez-Castro F; Rivadeneira I; Krakowiak F; Iturra J. Advances in the translational application of immunotherapy with pulsed dendritic cell-derived exosomes. J. Clin. Biomed. Res. 2024, 6, 1-8.
- Araujo-Abad S; Berna JM; Lloret-Lopez E; López-Cortés A; Saceda M; de Juan Romero C. Exosomes: from basic research to clinical diagnostic and therapeutic applications in cancer. Cell Oncol. 2024, 47, -. [CrossRef]
- Tian H; Li W. Dendritic cell-derived exosomes for cancer immunotherapy: hope and challenges. Ann. Transl. Med. 2017, 5, 221. [CrossRef]
- Fu P; Yin S; Cheng H; Xu W; Jiang J. Engineered exosomes for drug delivery in cancer therapy: A promising approach and application. Curr. Drug Deliv. 2024, 21, 817-827. [CrossRef]
- Marciscano AE; Anandasabapathy N. The role of dendritic cells in cancer and anti-tumor immunity. Semin. Immunol. 2021, 52, 101481. [CrossRef]
- Ganjalikhani Hakemi M; Yanikkaya Demirel G; Li Y; Jayakumar N. The immunosuppressive tumor microenvironment and strategies to revert its immune regulatory milieu for cancer immunotherapy. Front. Immunol. 2023, 14, 1238698.
- Huangfu S; Pan J. Novel biomarkers for predicting response to cancer immunotherapy. Front. Immunol. 2023, 14, 1179913. [CrossRef]
- Karlsen W; Akily L; Mierzejewska M; Teodorczyk J; Bandura A; Zaucha R; Cytawa W. Is 18F-FDG-PET/CT an optimal imaging modality for detecting immune-related adverse events after immune-checkpoint inhibitor therapy? Pros and cons. Cancers (Basel) 2024, 16, 1990. [CrossRef]
- Bi WL; Hosny A; Schabath MB; Giger ML; Birkbak NJ; Mehrtash A; Allison T; Arnaout O; Abbosh C; Dunn IF; Mak RH; Tamimi RM; Tempany CM; Swanton C; Hoffmann U; Schwartz LH; Gillies RJ; Huang RY; Aerts HJWL. Artificial intelligence in cancer imaging: Clinical challenges and applications. CA Cancer J. Clin. 2019, 69, 127-157.
- Sidiropoulos DN; Stein-O'Brien GL; Danilova L; Gross NE; Charmsaz S; Xavier S; Leatherman J; Wang H; Yarchoan M; Jaffee EM; Fertig EJ; Ho WJ. Integrated T cell cytometry metrics for immune-monitoring applications in immunotherapy clinical trials. JCI Insight 2022, 7, e160398.
- Schmitt A; Hus I; Schmitt M. Dendritic cell vaccines for leukemia patients. Expert Rev. Anticancer Ther. 2007, 7, 275-283. [CrossRef]
- Safaei S; Fadaee M; Farzam OR; Yari A; Poursaei E; Aslan C; Samemaleki S; Shanehbandi D; Baradaran B; Kazemi T. Exploring the dynamic interplay between exosomes and the immune tumor microenvironment: implications for breast cancer progression and therapeutic strategies. Breast Cancer Res. 2024, 26, 57. [CrossRef]
- Louie AD; Huntington K; Carlsen L; Zhou L; El-Deiry WS. Integrating molecular biomarker inputs into development and use of clinical cancer therapeutics. Front. Pharmacol. 2021, 12, 747194. [CrossRef]
- Zou W; Restifo NP. T(H)17 cells in tumour immunity and immunotherapy. Nat. Rev. Immunol. 2010, 10, 248-256.
- Bergholz JS; Wang Q; Kabraji S; Zhao JJ. Integrating immunotherapy and targeted therapy in cancer treatment: Mechanistic insights and clinical implications. Clin. Cancer Res. 2020, 26, 5557-5566. [CrossRef]
- Abou-El-Enein M; Elsallab M; Feldman SA; Fesnak AD; Heslop HE; Marks P; Till BG; Bauer G; Savoldo B. Scalable manufacturing of CAR T cells for cancer immunotherapy. Blood Cancer Discov. 2021, 2, 408-422.
- Bol KF; Mensink HW; Aarntzen EH; Schreibelt G; Keunen JE; Coulie PG; de Klein A; Punt CJ; Paridaens D; Figdor CG; de Vries IJ. Long overall survival after dendritic cell vaccination in metastatic uveal melanoma patients. Am. J. Ophthalmol. 2014, 158, 939-947.
- Palucka K; Banchereau J. Dendritic-cell-based therapeutic cancer vaccines. Immunity 2013, 39, 38-48. [CrossRef]
- Pitt JM; Charrier M; Viaud S; André F; Besse B; Chaput N; Zitvogel L. Dendritic cell-derived exosomes as immunotherapies in the fight against cancer. J. Immunol. 2014, 193, 1006-1011. [CrossRef]
- Waldmann TA. Cytokines in cancer immunotherapy. Cold Spring Harb. Perspect. Biol. 2018, 10, a028472. [CrossRef]
- Xu Z; Zeng S; Gong Z; et al. Exosome-based immunotherapy: a promising approach for cancer treatment. Mol. Cancer 2020, 19, 160. [CrossRef]
- Escudier B; Dorval T; Chaput N; André F; Caby MP; Novault S; Flament C; Leboulaire C; Borg C; Amigorena S; Boccaccio C; Bonnerot C; Dhellin O; Movassagh M; Piperno S; Robert C; Serra V; Valente N; Le Pecq JB; Spatz A; Lantz O; Tursz T; Angevin E; Zitvogel L. Vaccination of metastatic melanoma patients with autologous dendritic cell (DC) derived-exosomes: Results of the first phase I clinical trial. J. Transl. Med. 2005, 3, 10.
- Wu J; Shen Z. Exosomal miRNAs as biomarkers for diagnostic and prognostic in lung cancer. Cancer Med. 2020, 9, 6909-6922. [CrossRef]
- Saida Y; Watanabe S; Koyama S; Togashi Y; Kikuchi T. Strategies to overcome tumor evasion and resistance to immunotherapies by targeting immune suppressor cells. Front. Oncol. 2023, 13, 1240926. [CrossRef]
- Sheikhlary S; Lopez DH; Moghimi S; Sun B. Recent findings on therapeutic cancer vaccines: An updated review. Biomolecules 2024, 14, 503. [CrossRef]
- Lim HX; Hong HJ; Jung MY; Cho D; Kim TS. Principal role of IL-12p40 in the decreased Th1 and Th17 responses driven by dendritic cells of mice lacking IL-12 and IL-18. Cytokine 2013, 63, 179-186. [CrossRef]
- Kim CR; Kim B; Ning MS; Reddy JP; Liao Z; Tang C; Welsh JW; Mott FE; Shih YT; Gomez DR. Cost analysis of PET/CT versus CT as surveillance for stage III non-small-cell lung cancer after definitive radiation therapy. Clin. Lung Cancer 2018, 19, e517-e528. [CrossRef]
- Olson BM; McNeel DG. Monitoring regulatory immune responses in tumor immunotherapy clinical trials. Front. Oncol. 2013, 3, 109. [CrossRef]
- Deng M; Wu S; Huang P; Liu Y; Li C; Zheng J. Engineered exosomes-based theranostic strategy for tumor metastasis and recurrence. Asian J. Pharm. Sci. 2023, 18, 100870. [CrossRef]
- Lee KW; Yam JWP; Mao X. Dendritic cell vaccines: A shift from conventional approach to new generations. Cells 2023, 12, 2147. [CrossRef]
- Shalaby N; Dubois VP; Ronald J. Molecular imaging of cellular immunotherapies in experimental and therapeutic settings. Cancer Immunol. Immunother. 2022, 71, 1281-1294. [CrossRef]
- Sathyanarayanan V; Neelapu SS. Cancer immunotherapy: Strategies for personalization and combinatorial approaches. Mol. Oncol. 2015, 9, 2043-2053. [CrossRef]
- Pitt JM; André F; Amigorena S; Soria JC; Eggermont A; Kroemer G; Zitvogel L. Dendritic cell-derived exosomes for cancer therapy. J. Clin. Invest. 2016, 126, 1224-1232.
- Das S; Dey MK; Devireddy R; Gartia MR. Biomarkers in cancer detection, diagnosis, and prognosis. Sensors (Basel) 2023, 24, 37.
- Bhavsar D; Raguraman R; Kim D; Ren X; Munshi A; Moore K; Sikavitsas V; Ramesh R. Exosomes in diagnostic and therapeutic applications of ovarian cancer. J. Ovarian Res. 2024, 17, 113.
- Pham TD; Teh MT; Chatzopoulou D; Holmes S; Coulthard P. Artificial intelligence in head and neck cancer: Innovations, applications, and future directions. Curr. Oncol. 2024, 31, 5255-5290. [CrossRef]
- Wandrey M; Jablonska J; Stauber RH; Gül D. Exosomes in cancer progression and therapy resistance: Molecular insights and therapeutic opportunities. Life (Basel) 2023, 13, 2033. [CrossRef]
- Yao Y; Fu C; Zhou L; Mi QS; Jiang A. DC-derived exosomes for cancer immunotherapy. Cancers (Basel) 2021, 13, 3667. [CrossRef]
- Lee S; Margolin K. Cytokines in cancer immunotherapy. Cancers (Basel) 2011, 3, 3856-3893. [CrossRef]
- Subbiah V; Murthy R; Hong DS; Prins RM; Hosing C; Hendricks K; Kolli D; Noffsinger L; Brown R; McGuire M; Fu S; Piha-Paul S; Naing A; Conley AP; Benjamin RS; Kaur I; Bosch ML. Cytokines produced by dendritic cells administered intratumorally correlate with clinical outcome in patients with diverse cancers. Clin. Cancer Res. 2018, 24, 3845-3856.
- Gonzalez-Angulo AM; Hennessy BT; Mills GB. Future of personalized medicine in oncology: a systems biology approach. J. Clin. Oncol. 2010, 28, 2777-2783. [CrossRef]
- Yu X; Ibrahim SM. Evidence of a role for Th17 cells in the breach of immune tolerance in arthritis. Arthritis Res. Ther. 2011, 13, 132. [CrossRef]
- Shang N; Figini M; Shangguan J; Wang B; Sun C; Pan L; Ma Q; Zhang Z. Dendritic cells based immunotherapy. Am. J. Cancer Res. 2017, 7, 2091-2102.
- Pathania AS; Prathipati P; Challagundla KB. New insights into exosome mediated tumor-immune escape: Clinical perspectives and therapeutic strategies. Biochim. Biophys. Acta Rev. Cancer 2021, 1876, 188624. [CrossRef]
- Vu SH; Vetrivel P; Kim J; Lee MS. Cancer resistance to immunotherapy: Molecular mechanisms and tackling strategies. Int. J. Mol. Sci. 2022, 23, 10906. [CrossRef]
- Zanotta S; Galati D; De Filippi R; Pinto A. Enhancing dendritic cell cancer vaccination: The synergy of immune checkpoint inhibitors in combined therapies. Int. J. Mol. Sci. 2024, 25, 7509. [CrossRef]
- Ghaffari Laleh N; Ligero M; Perez-Lopez R; Kather JN. Facts and hopes on the use of artificial intelligence for predictive immunotherapy biomarkers in cancer. Clin. Cancer Res. 2023, 29, 316-323. [CrossRef]
- Lai JJ; Chau ZL; Chen SY; Hill JJ; Korpany KV; Liang NW; Lin LH; Liu JK. Exosome processing and characterization approaches for research and technology development. Adv. Sci. (Weinh.) 2022, 9, e2103222. [CrossRef]
- Bhinder B; Gilvary C; Madhukar NS; Elemento O. Artificial intelligence in cancer research and precision medicine. Cancer Discov. 2021, 11, 900-915. [CrossRef]
- van Gulijk M; Dammeijer F; Aerts JGJV; Vroman H. Combination strategies to optimize efficacy of dendritic cell-based immunotherapy. Front. Immunol. 2018, 9, 2759. [CrossRef]
- Zhou Z; Wang J; Wang J; Yang S; Wang R; Zhang G; Li Z; Shi R. Deciphering the tumor immune microenvironment from a multidimensional omics perspective: Insight into next-generation CAR-T cell immunotherapy and beyond. Mol. Cancer 2024, 23, 131. [CrossRef]
- Song MS; Nam JH; Noh KE; Lim DS. Dendritic cell-based immunotherapy: The importance of dendritic cell migration. J. Immunol. Res. 2024, 2024, 7827246. [CrossRef]
- Lyakh L; Trinchieri G; Provezza L; Carra G; Gerosa F. Regulation of interleukin-12/interleukin-23 production and the T-helper 17 response in humans. Immunol. Rev. 2008, 226, 112-131.
- Makler A; Asghar W. Exosomal biomarkers for cancer diagnosis and patient monitoring. Expert Rev. Mol. Diagn. 2020, 20, 387-400. [CrossRef]
- Dumbrava EI; Meric-Bernstam F. Personalized cancer therapy-leveraging a knowledge base for clinical decision-making. Cold Spring Harb. Mol. Case Stud. 2018, 4, a001578. [CrossRef]
- Liu Q; Li S; Dupuy A; Mai HL; Sailliet N; Logé C; Robert JH; Brouard S. Exosomes as new biomarkers and drug delivery tools for the prevention and treatment of various diseases: Current perspectives. Int. J. Mol. Sci. 2021, 22, 7763. [CrossRef]
- Hato L; Vizcay A; Eguren I; Pérez-Gracia JL; Rodríguez J; Gállego Pérez-Larraya J; Sarobe P; Inogés S; Díaz de Cerio AL; Santisteban M. Dendritic cells in cancer immunology and immunotherapy. Cancers (Basel) 2024, 16, 981.
- Sajan A; Lamane A; Baig A; Floch KL; Dercle L. The emerging role of AI in enhancing intratumoral immunotherapy care. Oncotarget 2024, 15, 635-637. [CrossRef]
- Wang Z; Wang Q; Qin F; Chen J. Exosomes: a promising avenue for cancer diagnosis beyond treatment. Front. Cell Dev. Biol. 2024, 12, 1344705. [CrossRef]
- Zhao N; Yi Y; Cao W; Fu X; Mei N; Li C. Serum cytokine levels for predicting immune-related adverse events and the clinical response in lung cancer treated with immunotherapy. Front. Oncol. 2022, 12, 923531. [CrossRef]
- Kamigaki T; Takimoto R; Okada S; Ibe H; Oguma E; Goto S. Personalized dendritic-cell-based vaccines targeting cancer neoantigens. Anticancer Res. 2024, 44, 3713-3724. [CrossRef]
- Sabado RL; Balan S; Bhardwaj N. Dendritic cell-based immunotherapy. Cell Res. 2017, 27, 74-95.
- Luo S; Chen J; Xu F; Chen H; Li Y; Li W. Dendritic cell-derived exosomes in cancer immunotherapy. Pharmaceutics 2023, 15, 2070. [CrossRef]
| Marker | Description | Evaluation Method | Expected Value |
|---|---|---|---|
| CD69 | Early marker of T-cell activation | Flow cytometry | >70% activated lymphocytes |
| CD25 | Late activation marker and regulatory function | Flow cytometry | >60% activated lymphocytes |
| HLA-DR | DC maturation marker | Flow cytometry | High expression (>80%) |
| IFN-γ | Key cytokine for Th1 polarization | ELISA | >100 pg/mL |
| IL-12 | Cytokine for the induction of Th1 response | ELISA | >80 pg/mL |
| Parameter | Description | Evaluation Method | Optimal Range |
|---|---|---|---|
| Size | Average diameter of exosomes | Nanoparticle tracking analysis (NTA) | 100–150 nm |
| Concentration | Number of exosomes per mL of sample | NTA | >109 particles/mL |
| CD63 | Exosome-specific surface marker | Western blotting | Positive expression |
| CD81 | Extracellular vesicle marker | Western blotting | Positive expression |
| Alix | Marker of exosome integrity and biogenesis | Western blotting | Positive expression |
| Monitoring Technique | Approximate Cost | Benefits |
|---|---|---|
| Flow cytometry | High | High precision in the quantification of cellular markers |
| PET-CT with 18F-FDG | Very high | Early detection of metabolic changes in the tumor |
| NTA | Moderate | Accurate assessment of exosome size and concentration |
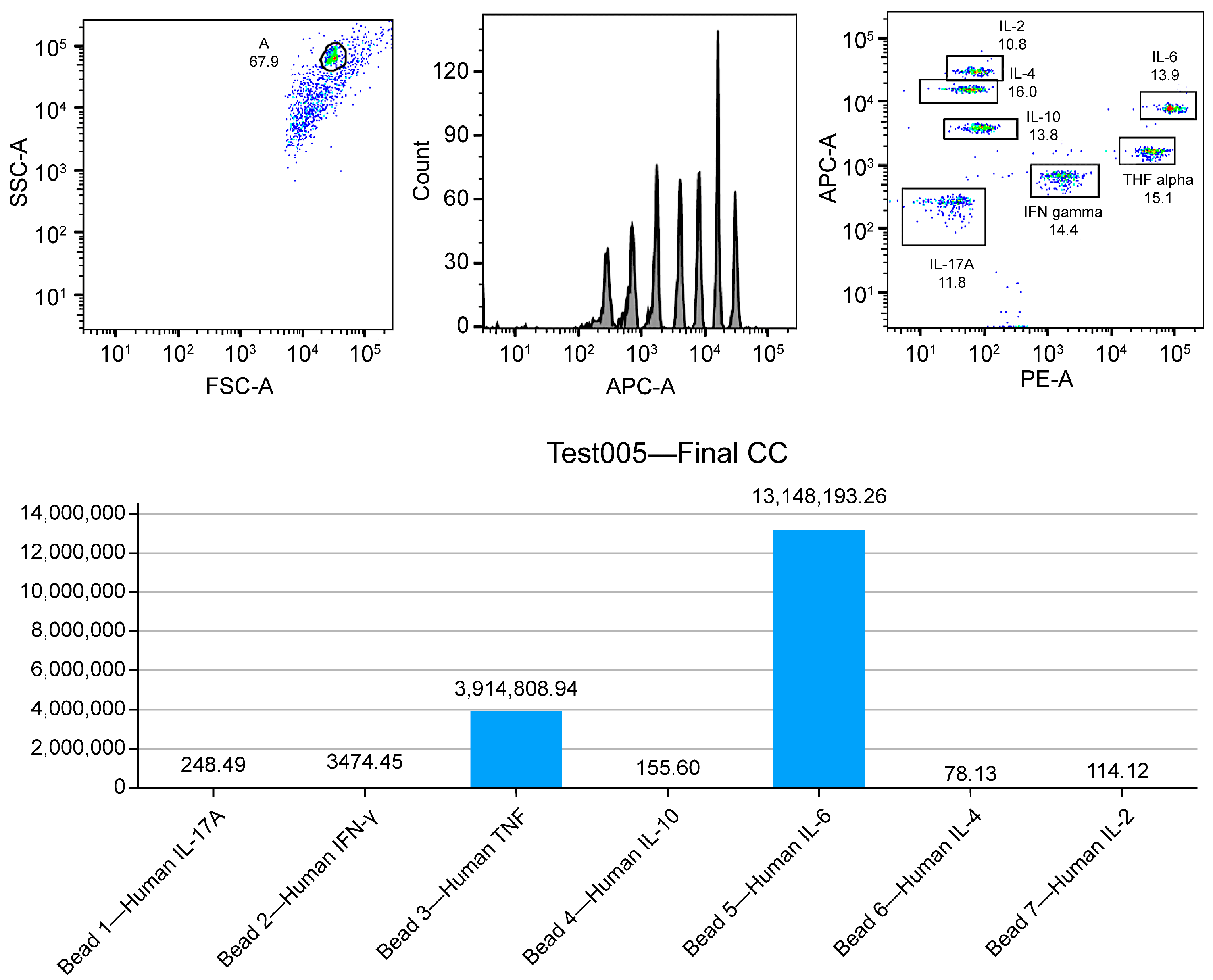
| Immune Profile | Key Cytokines | Effect on Therapy |
|---|---|---|
| Th1 | IFN-γ, IL-12 | Drive a robust cytotoxic response, enhancing T-cell activation and tumor clearance. |
| Th2 | IL-4, IL-10 | May suppress cytotoxic responses, potentially compromising therapy efficacy. |
| Th17 | IL-6, IL-17A | Linked to pro-inflammatory responses, necessitating careful modulation to prevent excessive inflammation and associated side effects. |
| No. | Stage | Day | Sample | Exam | Purpose |
|---|---|---|---|---|---|
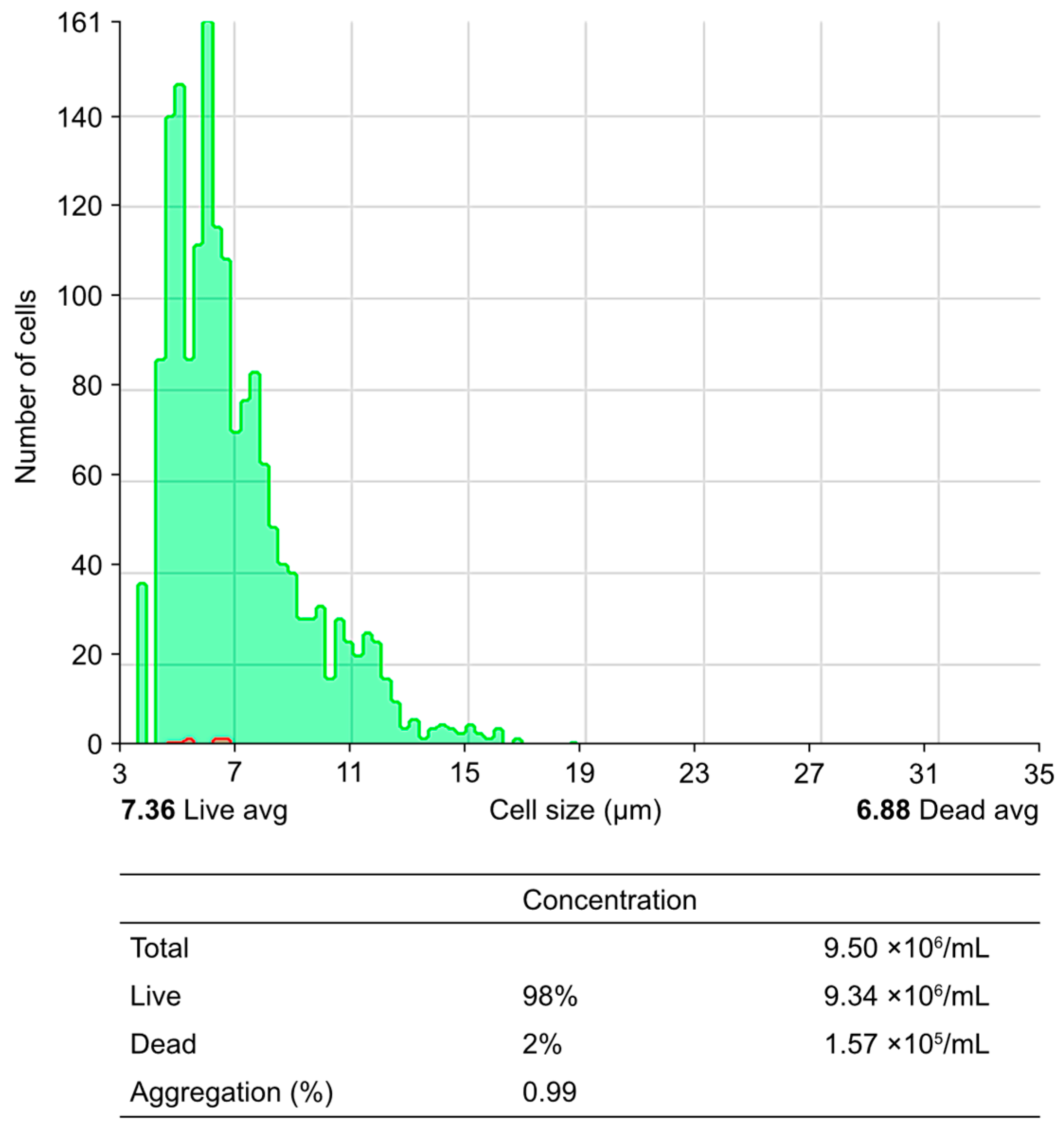
| 1 | Isolation of PBMCs | Day 1 | Peripheral blood | Separation of PBMCs using the Ficoll density gradient technique. | Isolate PBMCs with high viability and functionality to ensure suitability for subsequent differentiation and activation protocols. |
| 2 | Cell viability and integrity test | Day 1 | Isolated PBMNCs | Cell viability (Trypan blue or Annexin 5 assay) | Assess viability (>95%) and integrity of PBMCs, ensuring optimal vitality and functional competence |
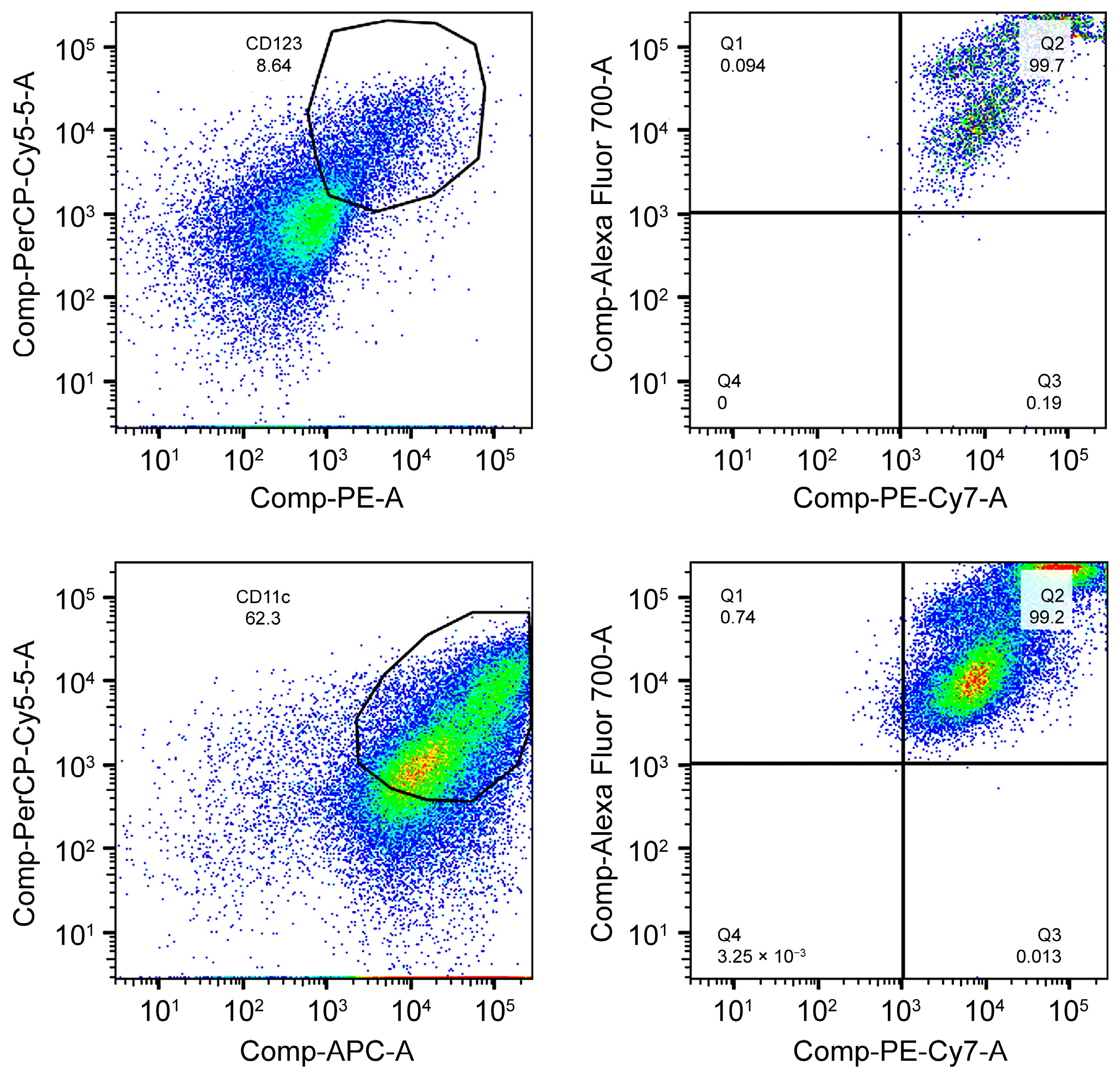
| 3 | DC differentiation | Day 7 | PBMC culture | Expression of HLA-DR, CD123, and CD11c via flow cytometry | Confirm efficient differentiation into DCs through high expression of phenotypic markers |
| 4 | DC maturation | Day 10 | Immature DCs | Evaluation of maturation markers CD80, CD83, and CD86 via flow cytometry | Verify the complete maturation of DCs, demonstrating the capacity for lymphocyte activation |
| 5 | Obtaining and characterizing exosomes | Day 12 | DC secretome | Concentration and size via NanoSight | Quantify the concentration and characterize the size of exosomes (90–120 nm), ensuring uniformity and stability |
| 6 | Immunopotency assessment | Day 12 | DC secretome | Evaluation of the Th1, Th2, and Th17 cytokine profiles using ELISA and CBA | Confirm the ability to produce proinflammatory cytokines capable of generating an effective immune response and respective lymphocyte activation (TNF-Q, IFN-y, IL-6, IL-17a, and IL-1b) |
| 7 | Lymphocyte activation | Day 14 | Co-culture of exosomes and/or DCs with T lymphocytes | Determination of lymphocyte activation (CD69 and CD25, via flow cytometry) | Confirm the functional activation of T lymphocytes, evaluating their proliferative capacity, activation, and production of cytotoxic cytokines |
| 8 | Induction of tumor apoptosis | Day 14 | Co-culture of activated T cells and tumor cells | Apoptosis assay (LDH, caspase activation) | Quantify the cytotoxic capacity of activated T lymphocytes to induce apoptosis in tumor cells, demonstrating the efficacy of the immunotherapeutic protocol |
| 9 | Final product characterization | Day 14 | Enriched exosome concentrate | Safety, viability, and membrane integrity tests, and immunological markers | Confirm the quality and safety of the product before its clinical administration, complying with regulatory standards |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




