Submitted:
02 January 2025
Posted:
03 January 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board and Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Seale, A.C.; Bianchi-Jassir, F.; Russell, N.J.; Kohli-Lynch, M.; Tann, C.J.; Hall, J.; Madrid, L.; Blencowe, H.; Cousens, S.; Baker, C.J.; et al. Estimates of the Burden of Group B Streptococcal Disease Worldwide for Pregnant Women, Stillbirths, and Children. Clinical Infectious Diseases 2017, 65, S200–S219. [Google Scholar] [CrossRef] [PubMed]
- Decoster, L.; Frans, J.; Blanckaert, H.; Lagrou, K.; Verhaegen, J. Antimicrobial Susceptibility of Group B Streptococci Collected in Two Belgian Hospitals. Acta Clin Belg 2005, 60. [Google Scholar] [CrossRef] [PubMed]
- Lamagni, T.; Wloch, C.; Broughton, K.; Collin, S.M.; Chalker, V.; Coelho, J.; Ladhani, S.N.; Brown, C.S.; Shetty, N.; Johnson, A.P. Assessing the Added Value of Group B Streptococcus Maternal Immunisation in Preventing Maternal Infection and Fetal Harm: Population Surveillance Study. BJOG 2022, 129, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Raabe, V.N.; Shane, A.L. Streptococcus Agalactiae (Group B Streptococcus). Microbiology S 2018, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Busch, L.M.; Kadri, S.S. Antimicrobial Treatment Duration in Sepsis and Serious Infections. Journal of Infectious Diseases 2020, 222, S142–S155. [Google Scholar] [CrossRef] [PubMed]
- Paul, M.; Shani, V.; Muchtar, E.; Kariv, G.; Robenshtok, E.; Leibovici, L. Systematic Review and Meta-Analysis of the Efficacy of Appropriate Empiric Antibiotic Therapy for Sepsis. Antimicrob Agents Chemother 2010, 54, 4851–4863. [Google Scholar] [CrossRef] [PubMed]
- Hughes, R.; Brocklehurst, P.; Steer, P.; Heath, P.T.; Stenson, B. Prevention of Early-Onset Neonatal Group B Streptococcal Disease: Green-Top Guideline No. 36. BJOG 2017, 124, e280–e305. [Google Scholar] [CrossRef]
- Fuchs, A.; Bielicki, J.; Mathur, S.; Sharland, M.; Van Den Anker, J.N. Reviewing the WHO Guidelines for Antibiotic Use for Sepsis in Neonates and Children. Paediatr Int Child Health 2018, 38, S3–S15. [Google Scholar] [CrossRef] [PubMed]
- CLSI M100 Performance Standards for Antimicrobial Susceptibility Testing; 30th ed.; Clinical and Laboratory Standards Institute: Pennsylvania, 2020; Vol. 8; ISBN 978-1-68440-067-6.
- EUCAST EUCAST V11: The European Committee on Antimicrobial Susceptibility Testing. Breakpoint Tables for Interpretation of MICs and Zone Diameters. Version 11.0, 2021; 2021.
- ISO 20776-1: 2019. Susceptibility Testing of Infectious Agents and Evaluation of Performance of Antimicrobial Susceptibility Test Devices—Part 1: Broth Micro-Dilution Reference Method for Testing the In Vitro Activity of Antimicrobial Agents Against Rapidly G; Geneva, Switzerland, 2019.
- Decoster, L.; Frans, J.; Blanckaert, H.; Lagrou, K.; Verhaegen, J. Antimicrobial Susceptibility of Group B Streptococci Collected in Two Belgian Hospitals. Acta Clin Belg 2005, 60, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Seki, T.; Kimura, K.; Reid, M.E.; Miyazaki, A.; Banno, H.; Jin, W.; Wachino, J.I.; Yamada, K.; Arakawa, Y. High Isolation Rate of MDR Group B Streptococci with Reduced Penicillin Susceptibility in Japan. Journal of Antimicrobial Chemotherapy 2015, 70, 2725–2728. [Google Scholar] [CrossRef] [PubMed]
- Khan, U.B.; Portal, E.A.R.; Sands, K.; Lo, S.; Chalker, V.J.; Jauneikaite, E.; Spiller, O.B. Genomic Analysis Reveals New Integrative Conjugal Elements and Transposons in GBS Conferring Antimicrobial Resistance. Antibiotics 2023, 12, 544. [Google Scholar] [CrossRef] [PubMed]
- EUCAST "The European Committee on Antimicrobial Susceptibility Testing. Breakpoint Tables for Interpretation of MICs and Zone Diameters. Version 14.0, 2024. Http://Www.Eucast.Org 2024, 0–77.
- Kardos, S.; Tóthpál, A.; Laub, K.; Kristóf, K.; Ostorházi, E.; Rozgonyi, F.; Dobay, O. High Prevalence of Group B Streptococcus ST17 Hypervirulent Clone among Non-Pregnant Patients from a Hungarian Venereology Clinic. BMC Infect Dis 2019, 19, 1–10. [Google Scholar] [CrossRef]
- Li, P.; Wei, Y.; Li, G.; Cheng, H.; Xu, Z.; Yu, Z.; Deng, Q.; Shi, Y. Comparison of Antimicrobial Efficacy of Eravacycline and Tigecycline against Clinical Isolates of Streptococcus Agalactiae in China: In Vitro Activity, Heteroresistance, and Cross-Resistance. Microb Pathog 2020, 149, 104502. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, R.; Shackcloth, J.; Felmingham, D.; MacGowan, A.P.; Booth, J.; Brown, D.F.J.; Coles, S.; Harding, I.; Livermore, D.M.; Reed, V.; et al. Comparison of BSAC Agar Dilution and NCCLS Broth Microdilution MIC Methods for in Vitro Susceptibility Testing of Streptococcus Pneumoniae, Haemophilus Influenzae and Moraxella Catarrhalis: The BSAC Respiratory Resistance Surveillance Programme. Journal of Antimicrobial Chemotherapy 2003, 52, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Amsler, K.; Santoro, C.; Foleno, B.; Bush, K.; Flamm, R. Comparison of Broth Microdilution, Agar Dilution, and Etest for Susceptibility Testing of Doripenem against Gram-Negative and Gram-Positive Pathogens. J Clin Microbiol 2010, 48, 3353–3357. [Google Scholar] [CrossRef] [PubMed]
- Lallemand, E.A.; Lacroix, M.Z.; Toutain, P.L.; Boullier, S.; Ferran, A.A.; Bousquet-Melou, A. In Vitro Degradation of Antimicrobials during Use of Broth Microdilution Method Can Increase the Measured Minimal Inhibitory and Minimal Bactericidal Concentrations. Front Microbiol 2016, 7, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Cunha, V. Da; Davies, M.R.; Douarre, P.; Rosinski-, I. Streptococcus Agalactiae Clones Infecting Humans Were Selected and Fixed through the Extensive Use of Tetracycline. 2015, 337549, 1–23. [CrossRef]
| NCTC number | Resistance genes | Original reference | Serotype | Sequence type |
|---|---|---|---|---|
| 14894 | erm(A) tet(M) | PHEGBS0044 | Ia | ST23 |
| 14895 | erm(A) tet(M) | PHEGBS0066 | III | ST17 |
| 14896 | mef(A) msr(D) tet(M) | PHEGBS0067 | Ia | ST23 |
| 14897 | mef(A) msr(D) tet(M) | PHEGBS0070 | Ia | ST23 |
| 14898 | lsa(C) erm(B) tet(M) tet(O) | PHEGBS0071 | III | ST19 |
| 14899 | erm(A) tet(M) | PHEGBS0082 | V | ST1 |
| 14900 | erm(A) tet(O) | PHEGBS0091 | II | ST12 |
| 14901 | erm(A) tet(M) | PHEGBS0098 | V | ST1 |
| 14902 | erm(B) tet(M) | PHEGBS0128 | V | ST1 |
| 14903 | aac(6') aph(2"), erm(A) lnu(C) tet(M) | PHEGBS0139 | V | ST19 |
| 14904 | None | PHEGBS0408 | II | ST28 |
| 14905 | None | PHEGBS0446 | VI | ST1 |
| 14906 | None | PHEGBS0491 | II | ST12 |
| 14907 | lsa(C) tet(M) | PHEGBS0511 | IV | ST297 |
| 14908 | ant(6-Ia) aph(3'-III), aadE , erm(B) msr(D) mef(A) tet(O) | PHEGBS0577 | III | ST17 |
| 14909 | tet(L) tet(M) | PHEGBS0586 | II | ST652 |
| 14910 | erm(B) tet(M) | PHEGBS0592 | V | ST1 |
| 14911 | erm(A) msr(D) mef(A) tet(M) catQ | PHEGBS0599 | V | ST19 |
| 14912 | erm(B) tet(O) tet(M) | PHEGBS0608 | II | ST28 |
| 14913 | erm(B) ant(6-Ia) aph(3'-III) tet(S) | PHEGBS0624 | VI | ST1 |
| 14914 | erm(B) tet(M) ant(6-Ia) aph(3'-III) cat(C194) | PHEGBS0662 | V | ST19 |
| 14915 | erm(B) msr(D) mef(A) tet(O) ant(6-Ia) aph(3'-III)aadE | PHEGBS0738 | III | ST130 |
| n/a | None | PHEGBS0359 | II | ST19 |
| n/a | erm(B) tet(M) | PHEGBS0595 | V | ST19 |
| ATCC 49619 1 | S. pneumoniae susceptible control strain | |||
| ATCC 700677 | S. pneumoniae resistant control for macrolides, penicillin, tetracycline |
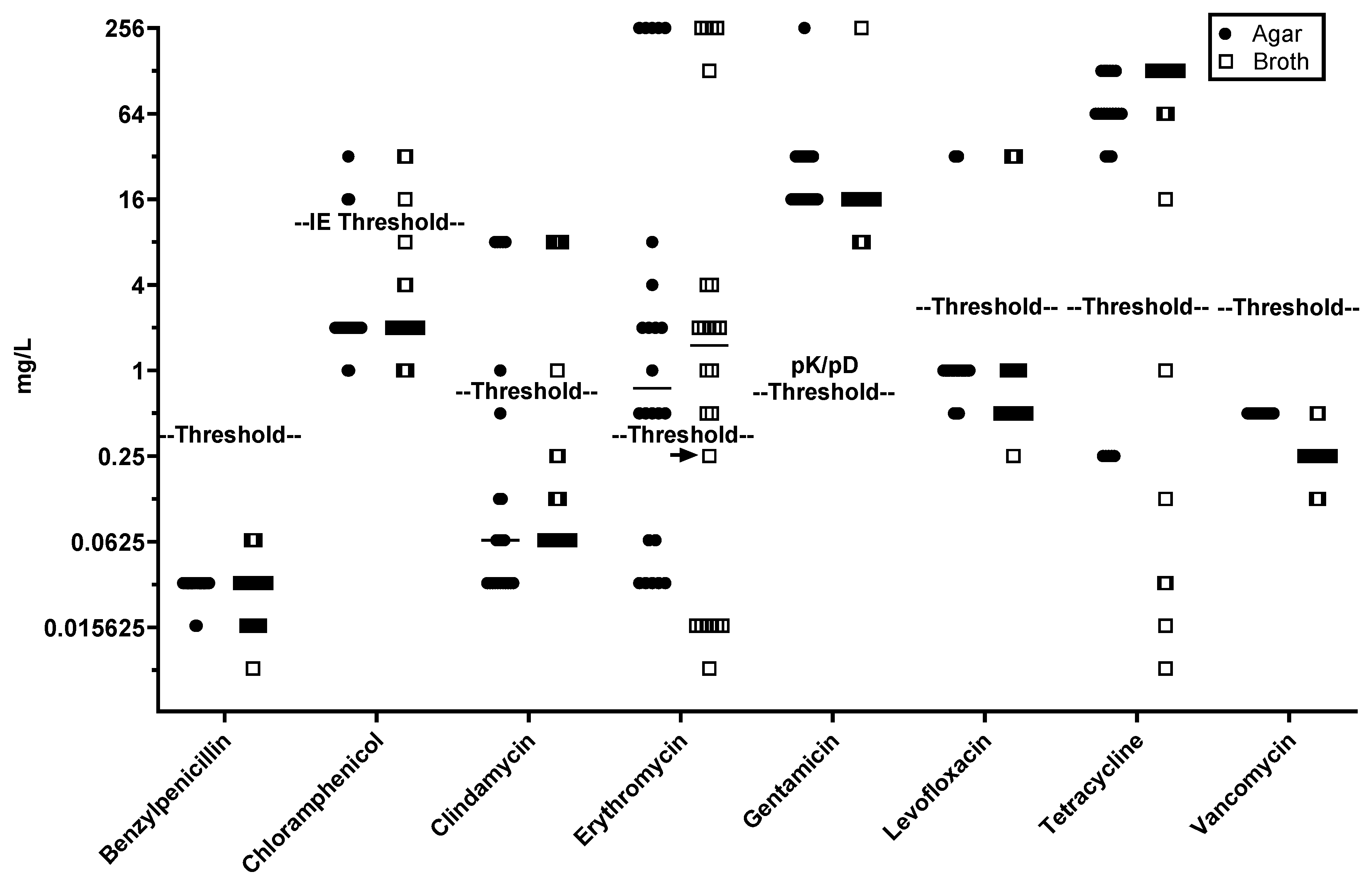
| MIC fold dilution for agar versus BMD | |||||||||
| Antimicrobial | -3 | -2 | -1 | 0 | 1 | 2 | 3 | >+3 | % within ± log2 dilution |
| Benzylpenicillin | 10 | 36 | 19 | 7 | 90.28 | ||||
| Chloramphenicol | 3 | 8 | 56 | 5 | 95.83 | ||||
| Clindamycin | 2 | 30 | 34 | 3 | 3 | 93.06 | |||
| Erythromycin | 2 | 8 | 31 | 21 | 10 | 83.33 | |||
| Gentamicin | 4 | 30 | 38 | 100.0 | |||||
| Levofloxacin | 3 | 39 | 29 | 1 | 98.61 | ||||
| Tetracycline | 3 | 19 | 24 | 9 | 5 | 1 | 5 | 6 | 52.78 |
| Vancomycin | 15 | 48 | 9 | 87.50 | |||||
| Antibiotic | Susceptibility profiles (Agar-Broth) |
% Concordance (SS, II, RR) | Kappa | Agreement Strength | ||||
| SS | RS | SR | RR | II | ||||
| Chloramphenicol | 62 | 1 | 9 | 98.61 | 0.94 | High | ||
| Clindamycin | 54 | 1 | 17 | 98.61 | 0.96 | High | ||
| Erythromycin | 22 | 4 | 46 | 94.44 | 0.88 | Moderate | ||
| Levofloxacin | 9 | 63 | 100.00 | 1 | High | |||
| Tetracycline | 15 | 57 | 100.00 | 1 | High | |||
| Primary stock mg/L | μL of stock added to 20 ml | End concentration mg/L |
|---|---|---|
| 2560 | 2000 | 256 |
| 2560 | 1000 | 128 |
| 2560 | 500 | 64 |
| 2560 | 250 | 32 |
| 2560 | 125 | 16 |
| 2560 | 62.5 | 8 |
| 80 | 1000 | 4 |
| 80 | 500 | 2 |
| 80 | 250 | 1 |
| 80 | 125 | 0.5 |
| 80 | 62.5 | 0.25 |
| 2.5 | 1000 | 0.125 |
| 2.5 | 512 | 0.064 |
| 2.5 | 256 | 0.032 |
| 2.5 | 128 | 0.016 |
| 2.5 | 64 | 0.008 |
| Antimicrobial | Sensitive | Increased | Resistant |
|---|---|---|---|
| Benzylpenicillin | ≤0.25 | - | >0.25 |
| Chloramphenicol* | ≤8 | - | >8 |
| Clindamycin | ≤0.5 | - | >0.5 |
| Erythromycin | ≤0.25 | - | >0.25 |
| Levofloxacin | ≤0.001 | 0.002-2 | >2 |
| Gentamicin* | >0.5 | ||
| Tetracycline | ≤1 | - | >1 |
| Vancomycin | ≤2 | - | >2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




