Submitted:
27 December 2024
Posted:
30 December 2024
You are already at the latest version
Abstract
Autoimmune diseases, characterized by chronic immune-mediated tissue damage, pose significant therapeutic challenges due to their complexity and limited long-term solutions. Recent advancements in cell immunotherapy, particularly CAR-T cells and CAR-Treg therapies, present groundbreaking avenues to directly modulate immune dysfunction. This review examines the mechanisms, therapeutic applications, and ongoing developments in these innovative strategies, detailing how CAR-T cells target autoreactive immune components and CAR-Tregs reestablish immune homeostasis. By addressing critical challenges such as safety, scalability, and clinical integration, this work highlights the potential of these therapies to redefine the landscape of autoimmune disease management.
Keywords:
Introduction
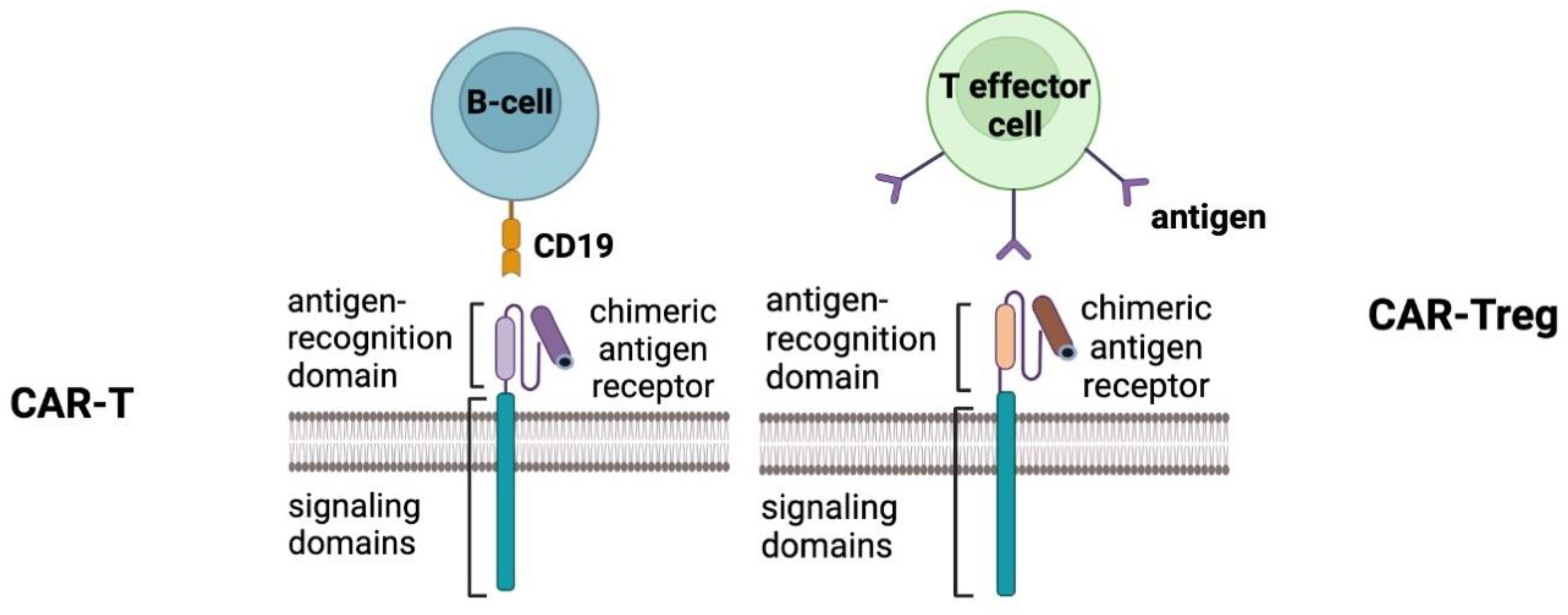
CAR-T Cell and Regulatory T-Cell (Treg) Therapy for Autoimmune Diseases
Mechanism of Action
Applications
Challenges
| Therapy | Mechanism of Action | Targeted Immune Components | Applications in Autoimmune Diseases | Challenges | Clinical Outcomes/Progress |
|---|---|---|---|---|---|
| CAR-T Cells | Genetically engineered T cells that express a chimeric receptor to recognize and target specific antigens on autoreactive cells. | Autoreactive B cells, plasma cells, autoantibodies. | Systemic lupus erythematosus (SLE), systemic sclerosis (SSc), rheumatoid arthritis (RA) | Antigen heterogeneity, cytokine release syndrome (CRS), off-target effects | Documented clinical remission in SLE and SSc. Ongoing trials. |
| Regulatory T Cells (Tregs) | Isolated Tregs are expanded and infused to restore immune tolerance by suppressing autoreactive immune responses. | Treg cells themselves target various immune cells and pathways. | Rheumatoid arthritis (RA), systemic lupus erythematosus (SLE) | Ensuring long-term stability of infused Tregs, potential conversion into pathogenic Th17 cells | Reduced disease activity and biomarkers of immune tolerance. |
Conclusion
Funding
Acknowledgments
Conflicts of Interest
References
- Vukovic J, Abazovic D, Vucetic D, Medenica S. CAR-engineered T cell therapy as an emerging strategy for treating autoimmune diseases. Front. Med. 2024, 11, 1447147. [Google Scholar]
- Chasov, V.; Ganeeva, I.; Zmievskaya, E.; Davletshin, D.; Gilyazova, E.; Valiullina, A.; Bulatov, E. Cell-Based Therapy and Genome Editing as Emerging Therapeutic Approaches to Treat Rheumatoid Arthritis. Cells 2024, 13, 1282. [Google Scholar] [CrossRef]
- Liu, J.; Zhao, Y.; Zhao, H. Chimeric antigen receptor T-cell therapy in autoimmune diseases. Front. Immunol. 2024, 15, 1492552. [Google Scholar] [CrossRef] [PubMed]
- Arjomandnejad M, Kopec AL, Keeler AM. CAR-T regulatory (CAR-Treg) cells: engineering and applications. Biomedicines 2022, 10, 287. [Google Scholar] [CrossRef]
- Yu J, Yang Y, Gu Z, Shi M, La Cava A, Liu A. CAR immunotherapy in autoimmune diseases: promises and challenges. Front. Immunol. 2024, 15, 1461102. [Google Scholar]
- Requejo Cier, C.J.; Valentini, N.; Lamarche, C. Unlocking the potential of Tregs: innovations in CAR technology. Front. Mol. Biosci. 2023, 10, 1267762. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




