Submitted:
26 December 2024
Posted:
27 December 2024
You are already at the latest version
Abstract
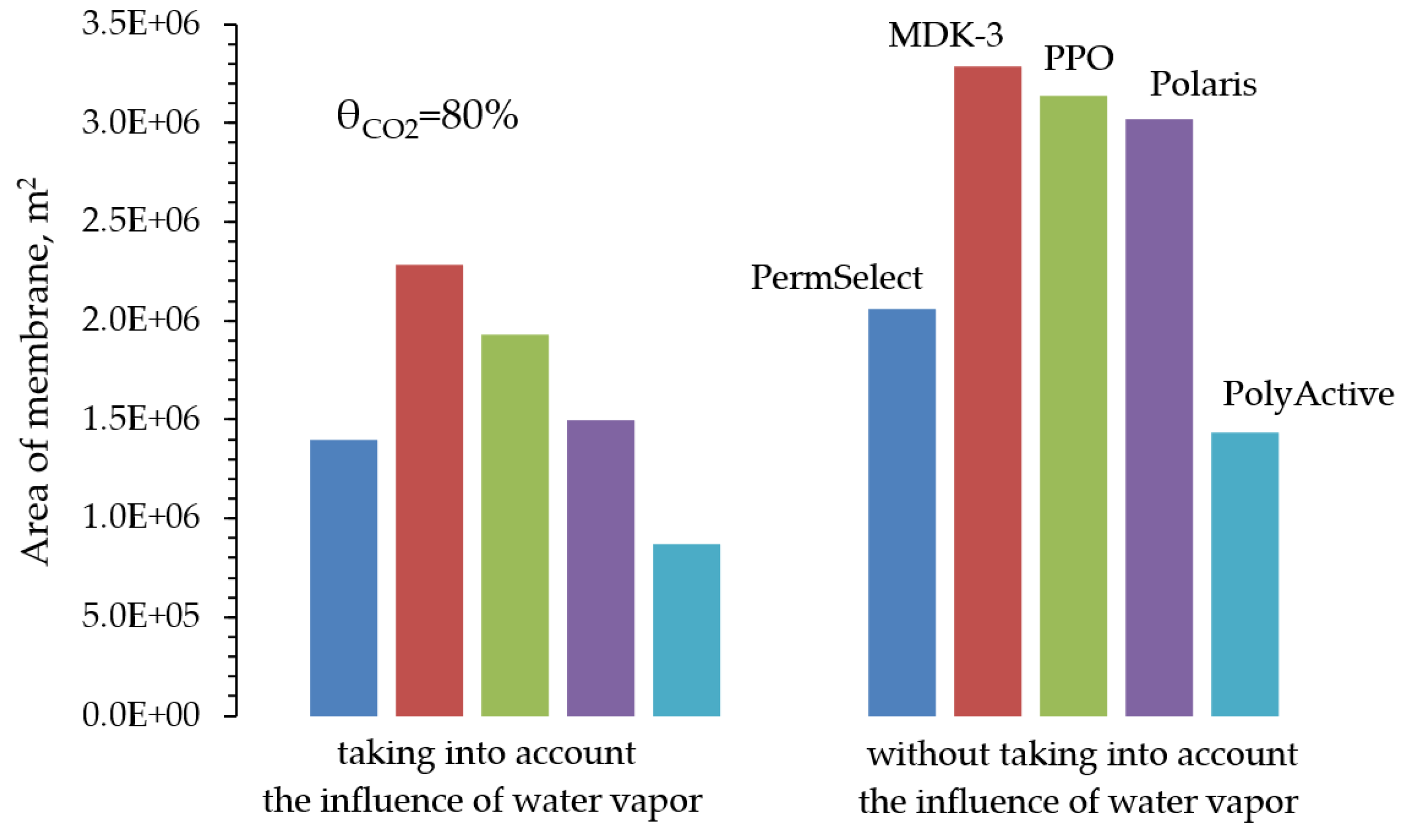
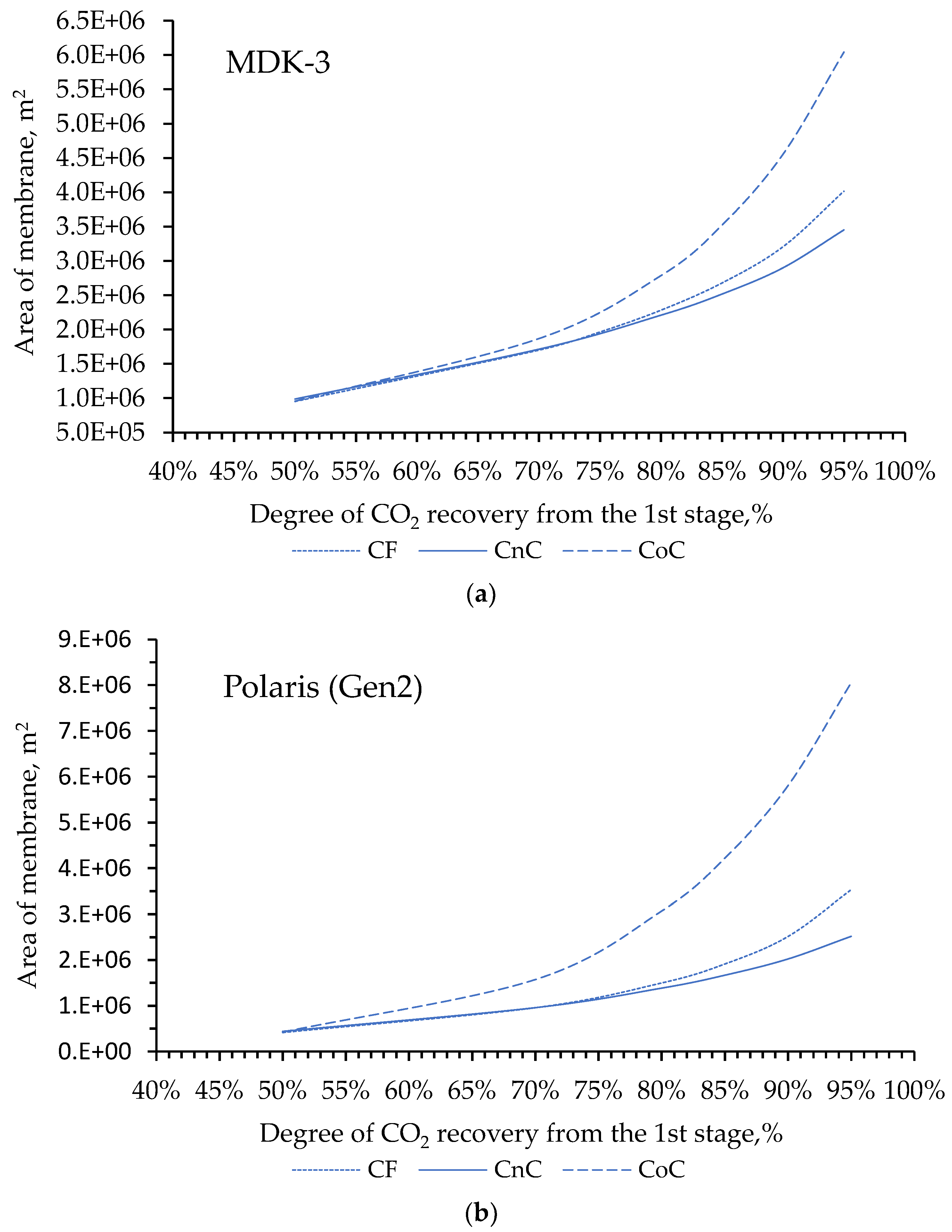
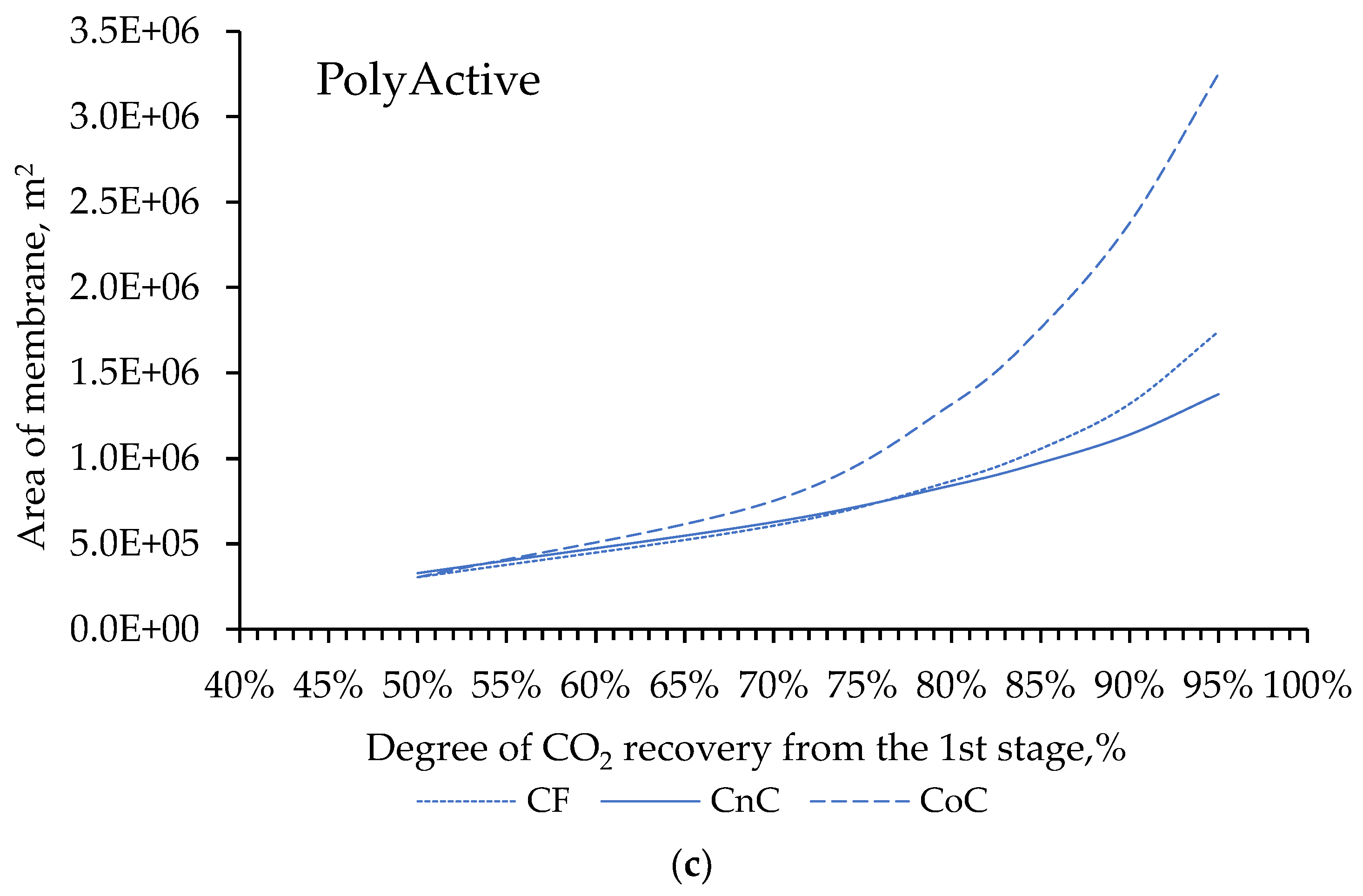
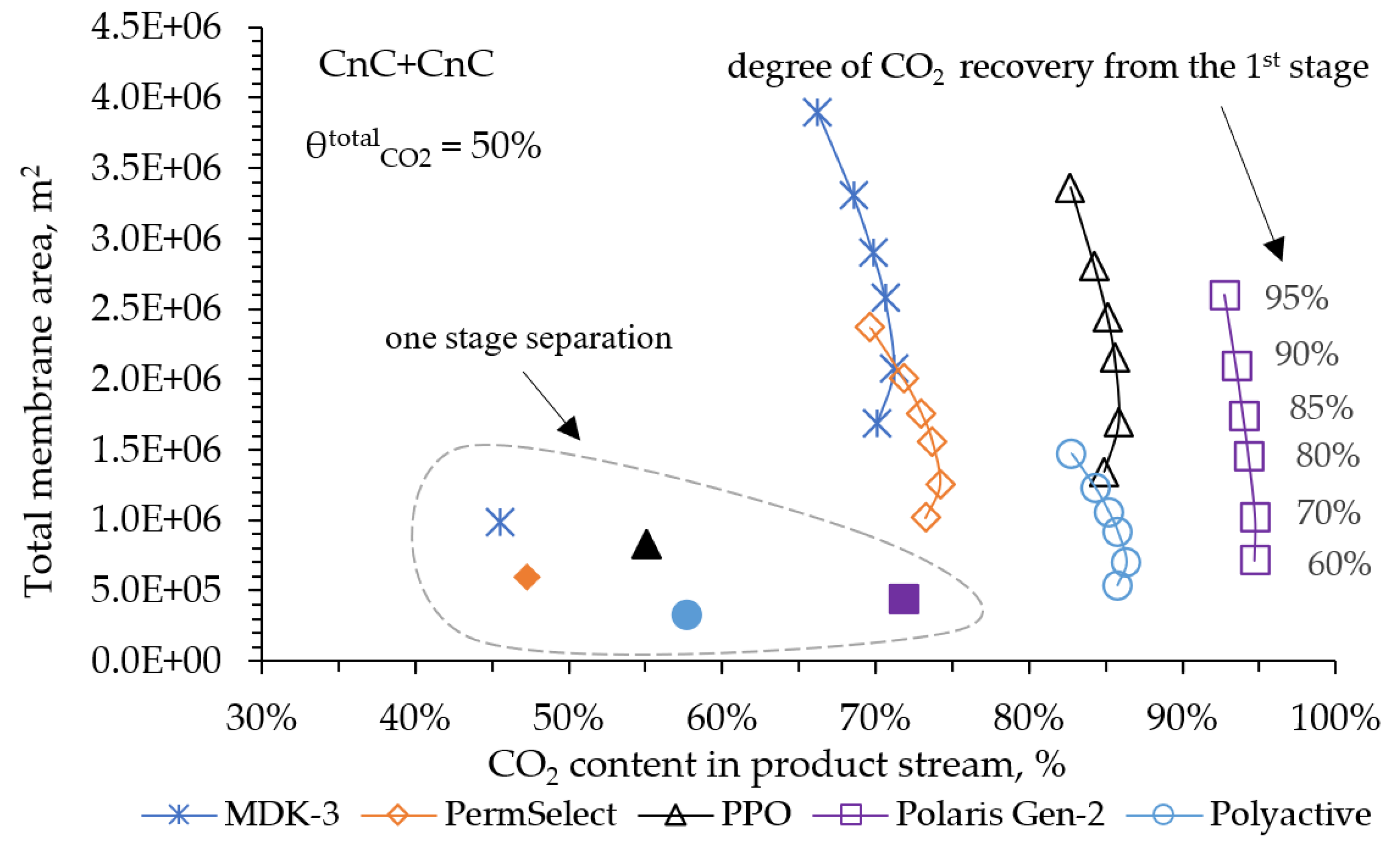
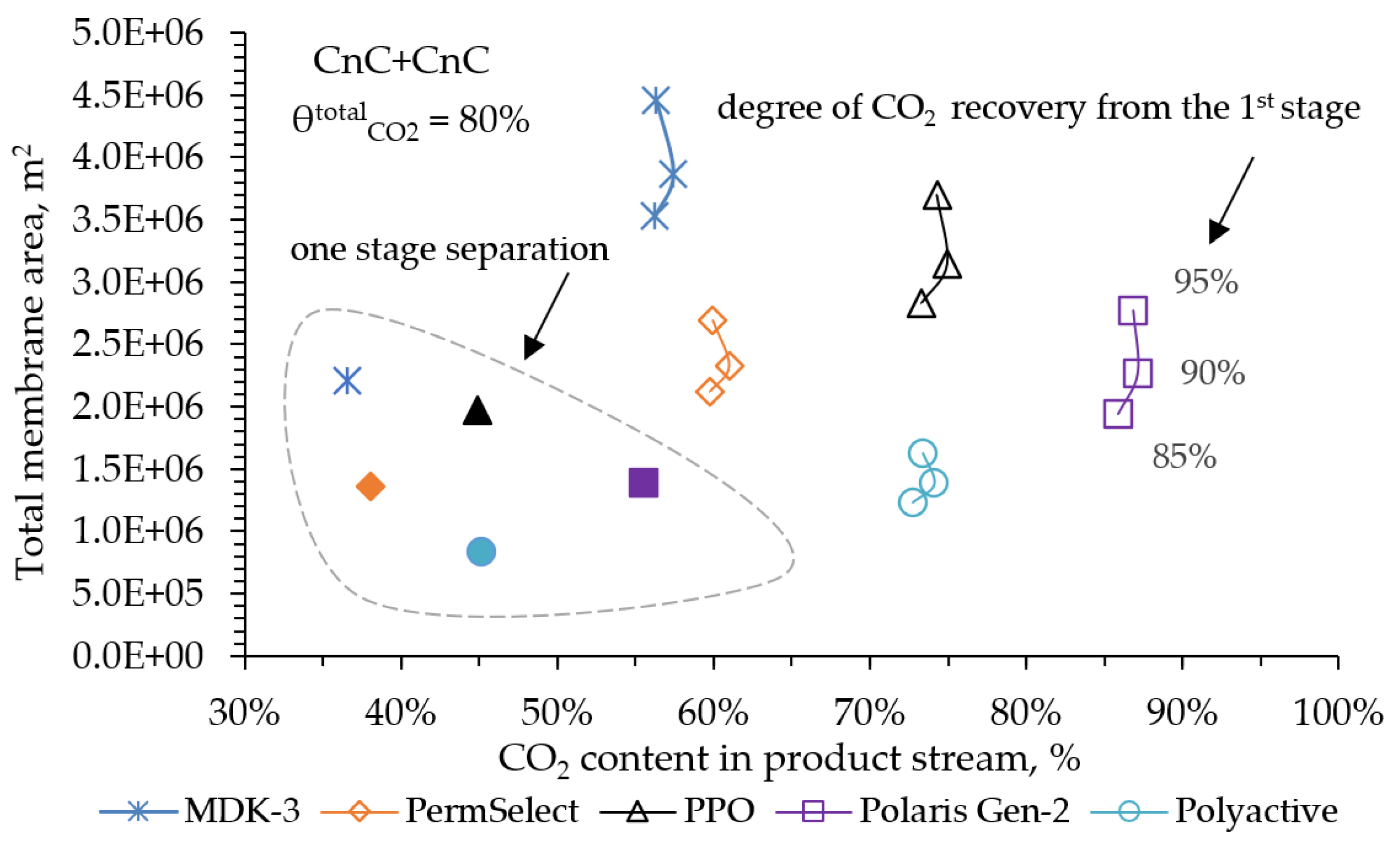
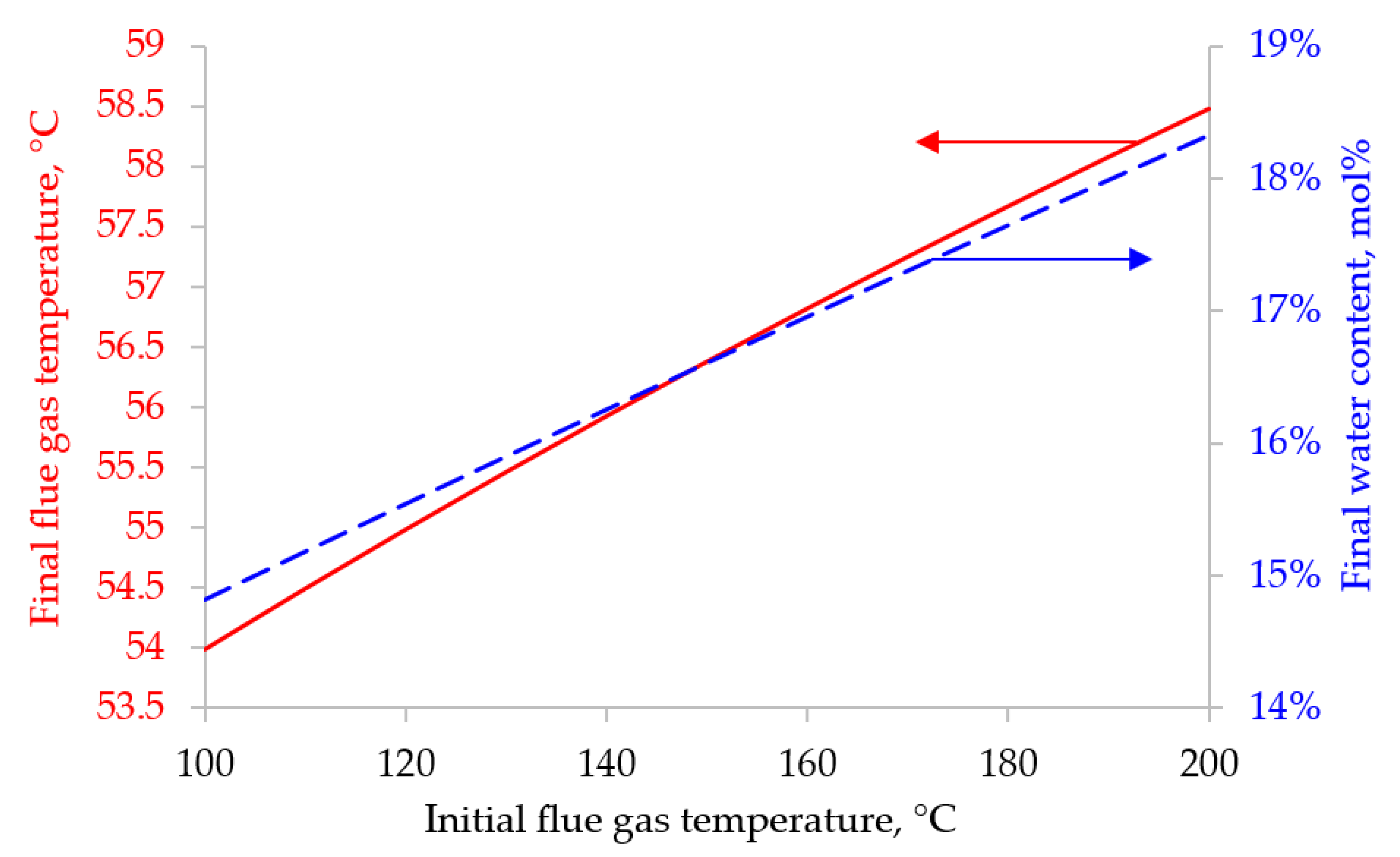
Despite the development of nuclear and alternative energy, thermal power plants operating by burning fossil fuels (coal, petroleum products or natural gas) will retain a significant share in the energy balance for a long time. In this regard, it is of particular interest to reduce CO2 emissions from the combustion of organic fuels through its capture and subsequent use or burial. In our work, mathematical modeling of the two-stage process of membrane extraction of CO2 from the flue gases of a thermal power plant was carried out, taking into account the presence of water vapor and various operating modes of the membrane module. We used commercially available polymer membranes for gas separation in our simulations. The calculations showed: Taking into account the presence of water vapor makes it possible to reduce the required membrane area by 1.6 times; For the degree of CO2 extraction < 80% in one stage, cross-flow and counter-current modes provide equal indicators for the required membrane area, and the co-current mode turns out to be less advantageous already with a degree of CO2 extraction > 60%. In this regard, in the area of low CO2 extraction values at the first stage, any flow organization mode in the membrane module can be selected, and in the high area, a counter-current has a slight advantage over the cross-flow mode; An optimal combination of membrane areas in the first and second stages is shown to achieve the maximum CO2 concentration in the product stream; Polaris Gen-2 membranes provide the best performance after two-stage separation: the CO2 content in the product stream was > 85 mol% and > 90 mol% with a total recovery rate of 80 and 50%, respectively; PolyActive and PPO membranes provide equal indicators for the CO2 content in the product stream, but in the use of PolyActive, the required membrane area is 2.3 times less.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials and Manufacturers of Commercial Gas Separation Membranes
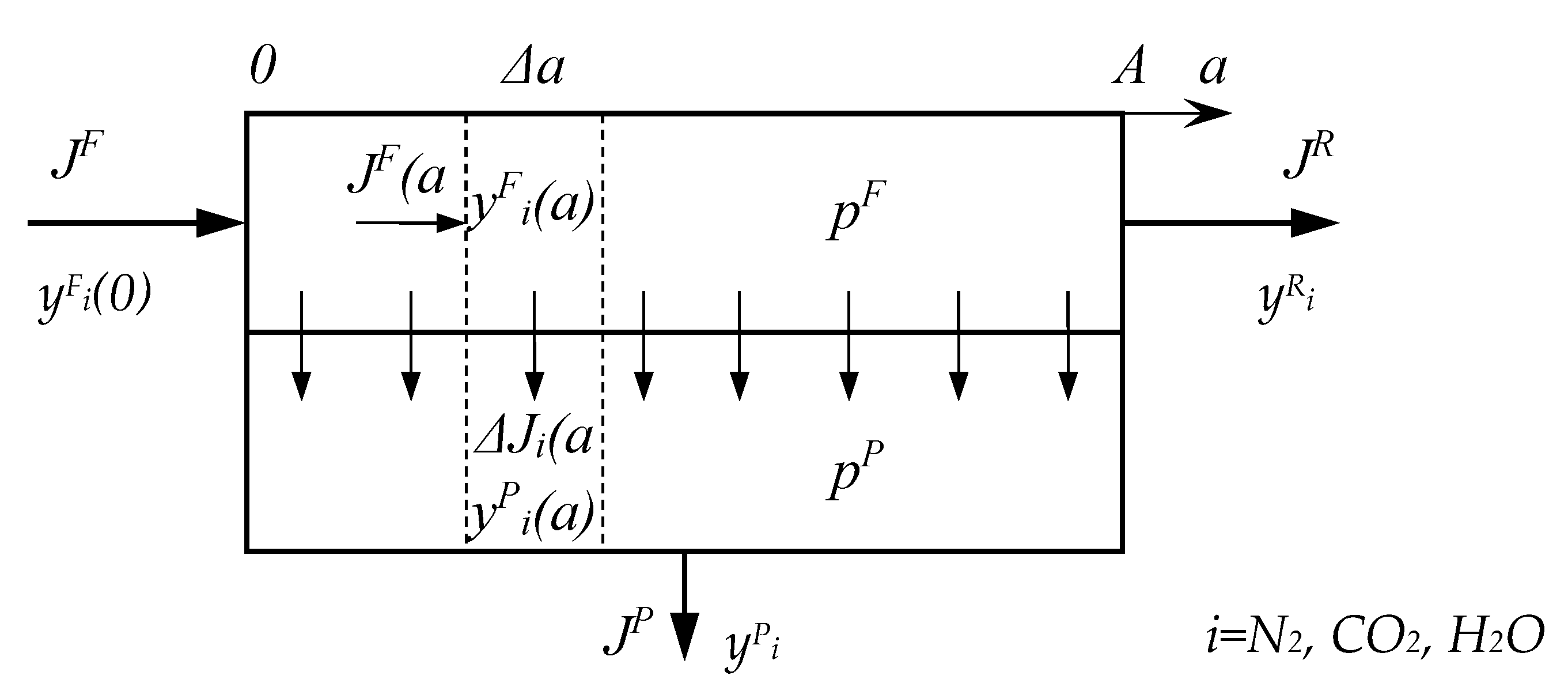
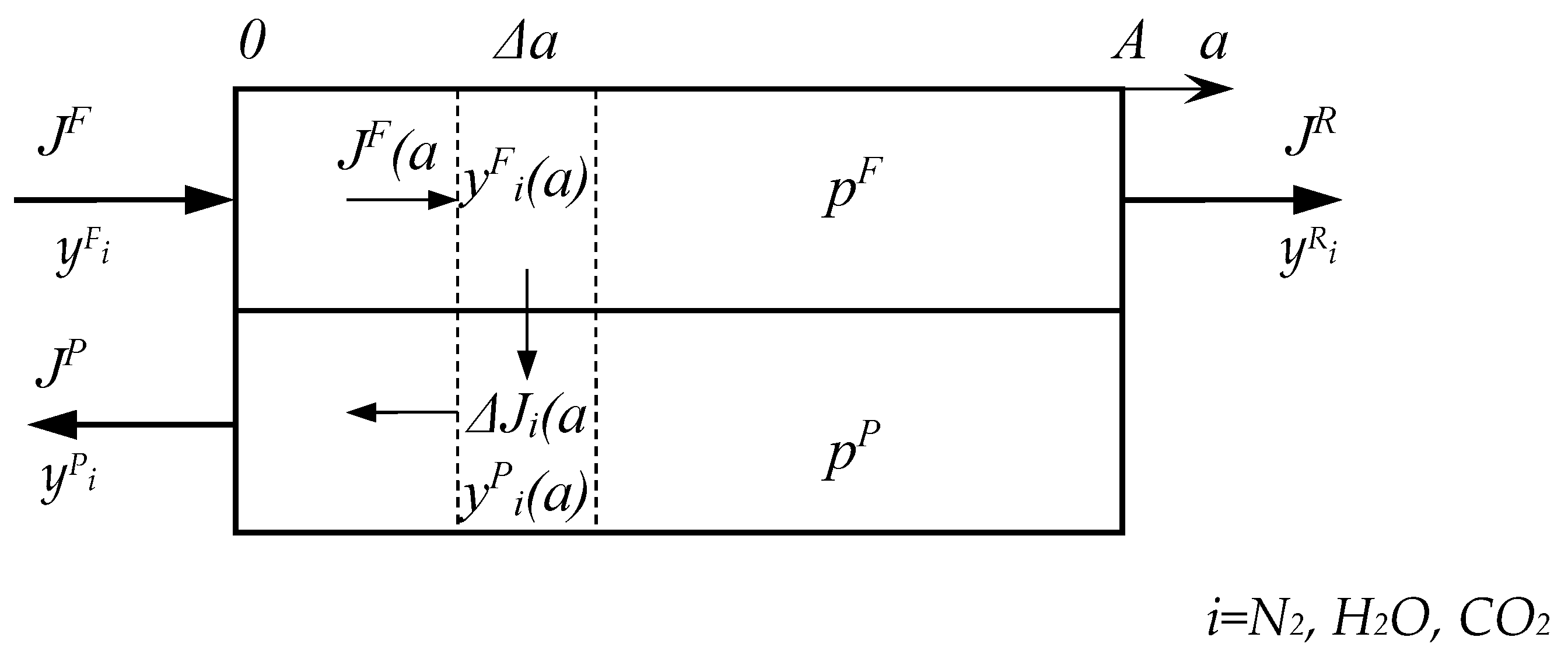
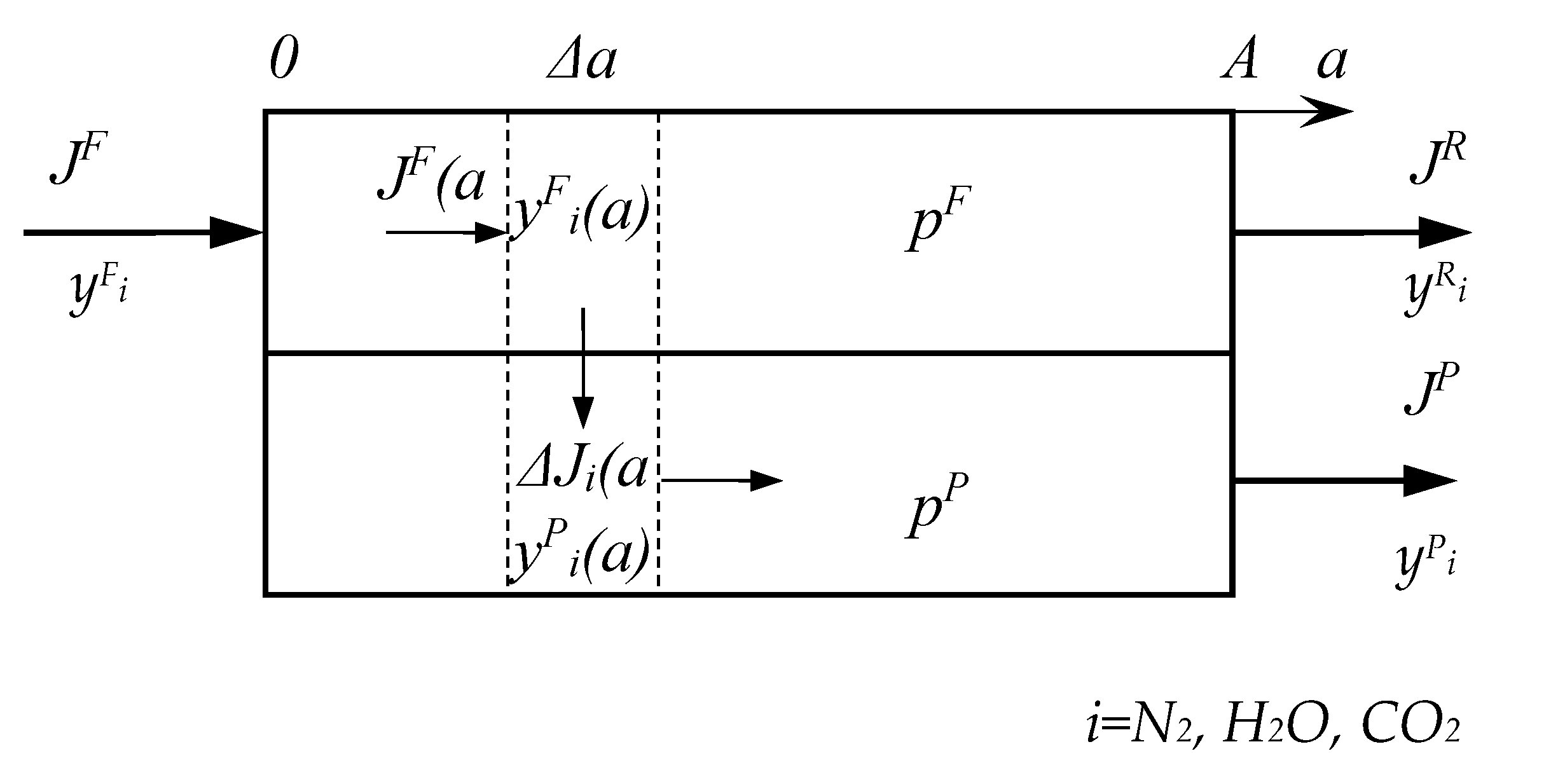
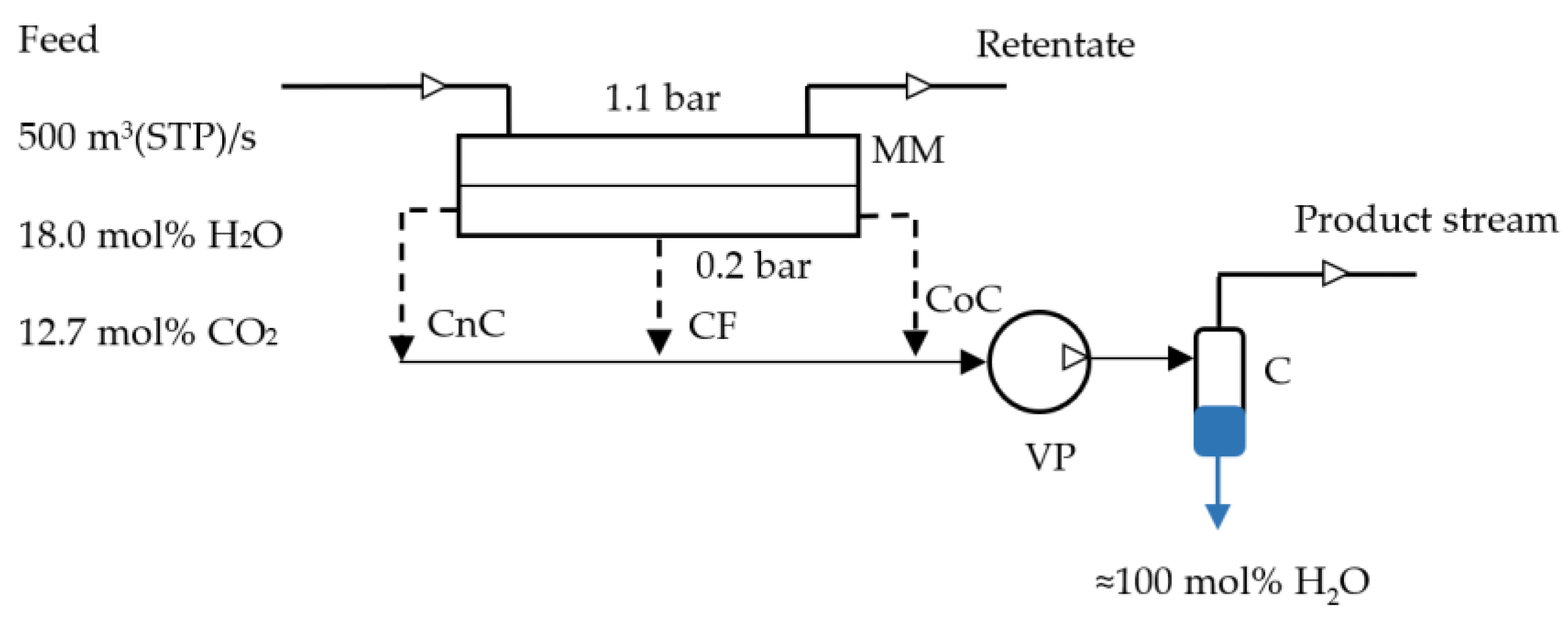
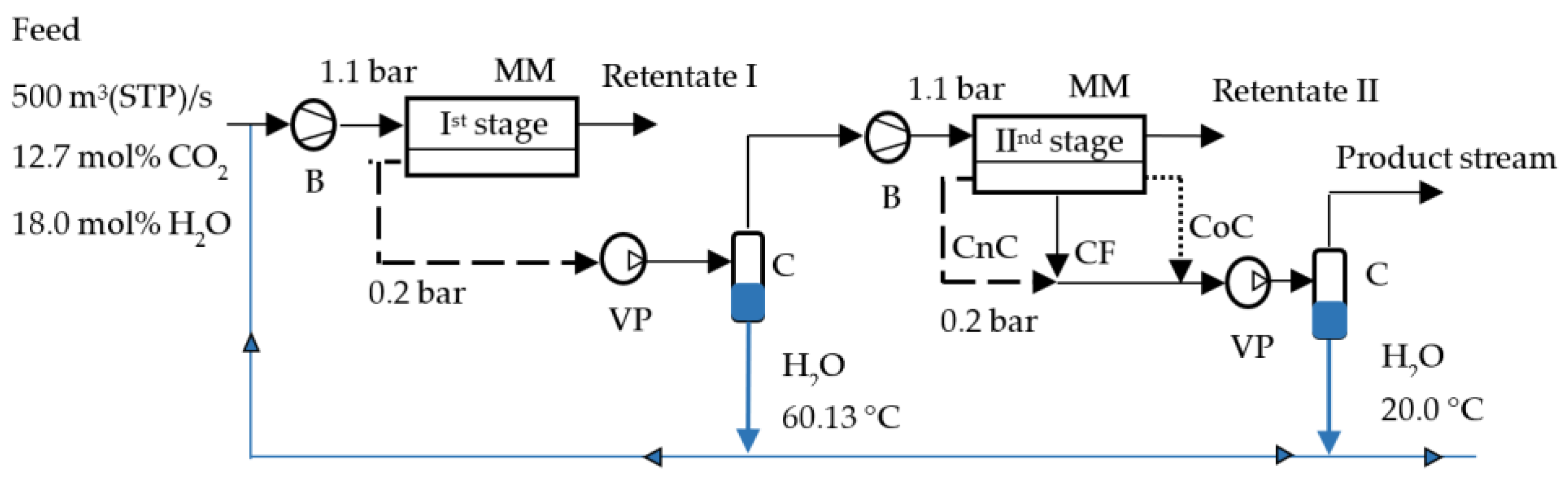
2.2. Mathematical Modeling of the Process of CO2 Capture from Flue Gas Using Membranes
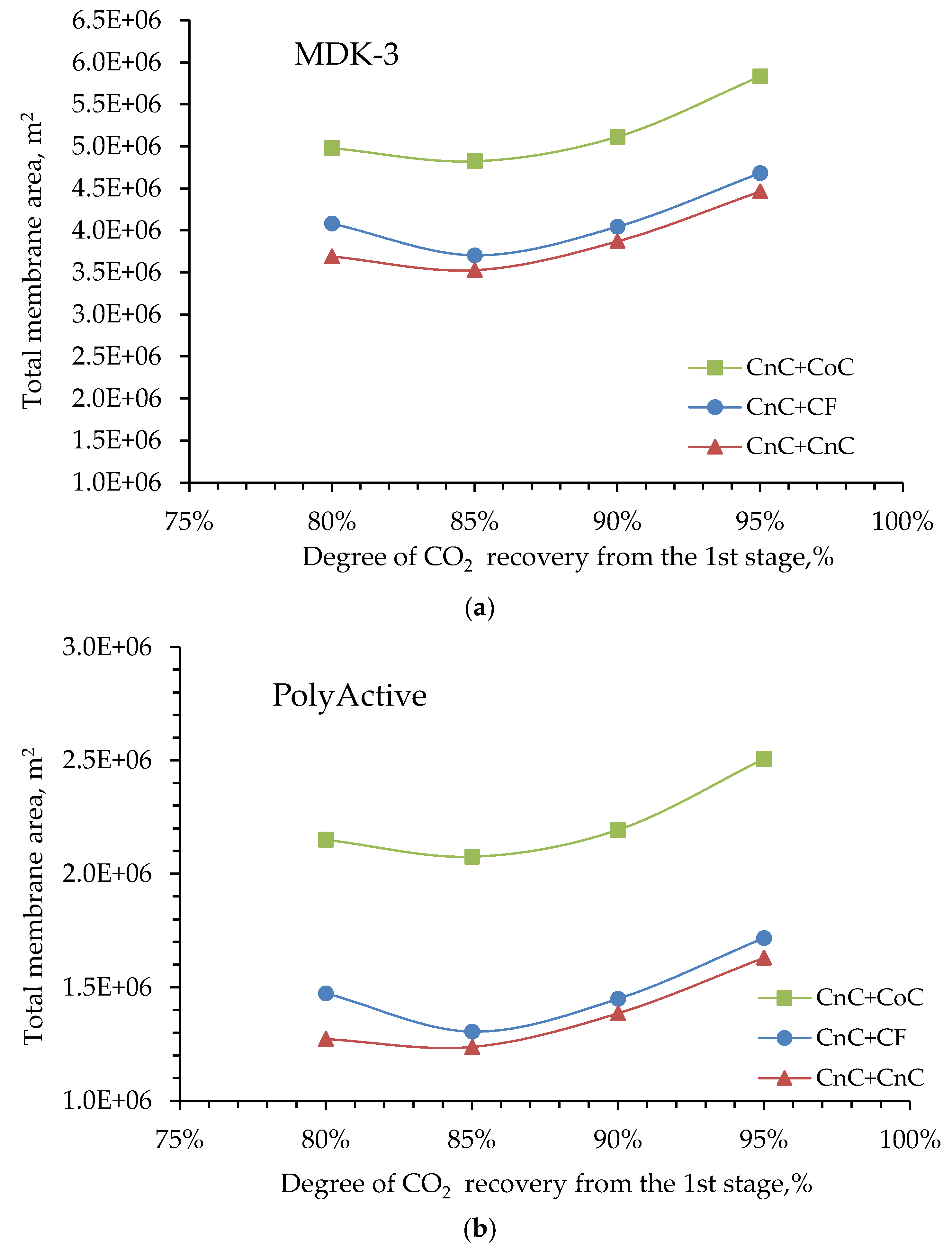
3. Results and Discussion
4. Conclusions
- (1)
- when a higher CO2 content in the stream is required, and
- (2)
- when the amount of CO2 capture is more important.
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Nomenclature
| A | area of membrane, m2 |
| a | coordinate of membrane area, m2 |
| J | flow rate, mol/s |
| P | permeability coefficient, barrer |
| p | pressure, bar |
| Q | permeance, mol/(m2∙s∙kPa) |
| y | molar fraction, mol% |
| α | selectivity of polymer/membrane |
| θ | stage cut, % |
| θi | recovery of component i, % |
| 0 | initial value |
| F | feed |
| P | permeate |
| R | retentate |
| i | component |
Appendix A
References
- Olivier, J.; Janssens-Maenhout, G.; Peters, J. Trends in global CO2 emissions: 2012 Report; PBL Netherlands Environmental Assessment Agency: The Hague, The Netherlands, 2012.
- Emissions trading system (ETS), 2020 climate & energy package; Available online: https://ec.europa.eu/clima/policies/strategies/2020_en (accessed 15.03.2024).
- E. Commission, EU Emissions TradingSystem (EU ETS); Available online: https://ec.europa.eu/clima/sites/clima/files/factsheet_ets_en (accessed 15.03.2024).
- Kárászováa, M.; Zacha, B.; Petrusováa, Z.; Červenkaa, V.; Bobákb, M.; Šyca, M.; Izák, P. Post-combustion carbon capture by membrane separation. Separation and Purification Technology 2020, 238, 116448. [CrossRef]
- Bui, M.; Adjiman, C.S.; Bardow, A.; Anthony, E.J.; Boston, A.; Brown, S.; Fennell, P.S.; Fuss, S.; Galindo, A.; Hackett, L.A.; Hallett, J.P.; Herzog, H.J.; Jackson, G.; Kemper, J.; Krevor, S.; Maitland, G.C.;, Matuszewski, M.; Metcalfe, I.S.; Petit, C.; Puxty, G.; Reimer, J.; Reiner, D.M.; Rubin, E.S.; Scott, S.A.; Shah, N.; Smit, B.; Trusler, J.P.M.; Webley, P.; Wilcox, J.; Mac Dowell, N. Carbon capture and storage (CCS): the way forward. Energy & Environmental Science 2018, 11(5), 1062-1176.
- Karnwiboon, K.; Krajangpit, W.; Supap, T.; Muchan, P.; Saiwan, C.; Idem, R.; Koiwanit, J. Solvent extraction based reclaiming technique for the removal of heat stable salts (HSS) and neutral degradation products from amines used during the capture of carbon dioxide (CO2) from industrial flue gases. Separation and Purification Technology 2019, 228, 115744. [CrossRef]
- Coleman. A policy analysis of the driving factors behind carbon capture and storage facilities. LSU Journal of Energy Law and Resources 2018, 6.
- Elmabrouk, H.E.B.S.K.; Mahmud, W.M. An overview of power plant CCS and CO2 EOR projects. IEOM, 2017, Rabat Morroco.
- Schlissel, D.W.D. Holy Grail of Carbon Capture Continues to Elude Coal Industry. IEEFA, 2018, Cleveland.
- Merkel, Tim C.; Lin, Haiqing; Wei, Xiaotong; Baker, Richard. Power plant post-combustion carbon dioxide capture: An opportunity for membranes. Journal of Membrane Science 2010, 359, 126-139.
- Li, Qinghua; Wu, Hongyu; Wang, Zhi; Wang, Jixiao. Analysis and optimal design of membrane processes for flue gas CO2 capture. Separation and Purification Technology 2022, 298, 121584. [CrossRef]
- Han, Yang; Yang, Yutong; Winston Ho, W. S. Recent Progress in the Engineering of Polymeric Membranes for CO2 Capture from Flue Gas. Membranes 2020, 10(11), 365. [CrossRef]
- Hussain, Arshad; Hägg, May-Britt. Afeasibility study of CO2 capture from flue gas by a facilitated transport membrane. Journal of Membrane Science 2010, 359, 140–148. [CrossRef]
- He, X.; Chen, D.; Liang, Zh.; Yang F. Insight and Comparison of Energy-efficient Membrane Processes for CO2 Capture from Flue Gases in Power Plant and Energy-intensive Industry. Carbon Capture Science & Technology 2022, 2, 100020.
- Miroshnichenko, D.; Shalygin M.; Bazhenov S. Simulation of the Membrane Process of CO2 Capture from Flue Gas via Commercial Membranes While Accounting for the Presence of Water Vapor. Membranes 2023, 13, 692. [CrossRef]
- PermSelect. Available online: https://www.permselect.com/membranes (accessed on 15.04.2024).
- STC “Vladipor”. Available online: http://www.vladipor.ru (accessed on 15.04.2024).
- Puri, P.S. Commercial Applications of Membranes in Gas Separations. Membrane Engineering for the Treatment of Gases 2011, Volume 1: Gas-separation Problems with Membranes, 215-244.
- Brinkmann, T.; Lillepärg, J.; Notzke, H.; Pohlmann, J.; Shishatskiy, S.; Wind, J.; Wolff, T. Development of CO2 Selective Poly(Ethylene Oxide)-Based Membranes: From Laboratory to Pilot Plant Scale. Engineering 2017, 3 (4), 485-493.
- Baker R.; Freeman, B. Large Pilot Testing of the MTR Membrane Post-Combustion CO2 Capture Process. U.S. DEPARTMENT OF ENERGY NATIONAL ENERGY TECHNOLOGY LABORATORY 2018.
- Atlaskin, A.A.; Trubyanov, M.M.; Yanbikov, N.R.; Kryuchkov, S.S.; Chadov, A.A.; Smorodin, K.A.; Drozdov, P.N.; Vorotyntsev, V.M.; Vorotyntsev, I.V. Experimental Evaluation of the Efficiency of Membrane Cascades Type of “Continuous Membrane Column” in the Carbon Dioxide Capture Applications. Membranes and Membrane Technologies 2020, 2 (1), 35–44. [CrossRef]
- Metz, S.J.; W.J.C. van de Ven; Potreck, J.; Mulder, M.H.V.; Wessling, M. Transport of water vapor and inert gas mixtures through highly selective and highly permeable polymer membranes. Journal of Membrane Science 2005, 251, 29–41.
- Alentiev, A.Y.; Levin, I.S.; Belov, N.A.; Nikiforov, R.Y.; Chirkov, S.V.; Bezgin, D.A.; Ryzhikh, V.E.; Kostina, J.V.; Shantarovich, V.P.; Grunin, L.Y. Features of the Gas-Permeable Crystalline Phase of Poly-2,6-dimethylphenylene Oxide. Polymers 2022, 14, 120. [CrossRef]
- Merkel, Tim C.; Wei, Xiaotong; He, Zhenjie; White, Lloyd S.; Wijmans, J.G.; Baker, Richard W. Selective Exhaust Gas Recycle with Membranes for CO2 Capture from Natural Gas Combined Cycle Power Plants. Industrial and Engineering Chemistry Research 2013, 52, 1150-1159.
- Brinkmann, T. Pilot scale investigations of the removal of carbon dioxide from hydrocarbon gas streams using poly(ethylene oxide)-poly(butylene terephthalate) PolyActive™ thin film composite membranes. Journal of membrane science 2015. [CrossRef]
- Bounaceur, R.; Lape, N.; Roizard, D.; Vallieres, C.; Favre, E. Membrane processes for post-combustion carbon dioxide capture: a parametric study. Energy 2006, 31, 2556. [CrossRef]


| Manufacturer | Сommercial name | Polymer | Ref. |
|---|---|---|---|
| PermSelect, Ann Arbor, MI, USA | PermSelect | PDMS* | [16] |
| STC “Vladipor“, Vladimir, Russia | MDK-3 | PDMS-based copolymer | [17] |
| MTR, Newark, CA, USA | Polaris Gen-2 | n/a | [18] |
| Parker Hannifin, Cleveland, OH, USA | Parker | PPO | [18] |
| Helmholtz-Zentrum Geesthacht, Geesthacht, Germany | PolyActive | PEO** | [4,19] |
| Membrane | Q(N2), GPU | Q(CO2), GPU | Q(H2O), GPU | α (CO2/N2) | Ref. |
|---|---|---|---|---|---|
| PermSelect | 93.3 | 1083 | 12,000 | 11.6 | [16] |
| MDK-3 | 61.8 | 616.6 | 4203 | 10.0 | [21] |
| Parker | 50 | 1120 | 81,200 | 22.4 | [22,23] |
| Polaris Gen-2 | 44 | 2200 | 8800 | 50.0 | [10,24] |
| PolyActive | 111 | 2350 | 17,390 | 21.2 | [25] |
| Parameters | Values |
|---|---|
| Flue gas feed flow rate, m3(STP)/s | 500 |
| Initial flue gas composition, mol%: | |
| N2 | 69.3 |
| CO2 | 12.7 |
| H2O | 18.0 |
| Feed pressure (absolute), bar | 1.1 |
| Permeate pressure (absolute), bar | 0.2 |
| Temperature, °C | 60.13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




