Submitted:
27 December 2024
Posted:
27 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Influence of Dietary Mechanisms on Imids
1.1.1. Diet Composition, Intestinal Microbiota, and Dysbiosis
1.1.2. Diet Composition and Intestinal Permeability
1.1.3. Diet Composition, Inflammation, and Immune System
Dietary Inflammatory Index (DII)
1.2. Obesity
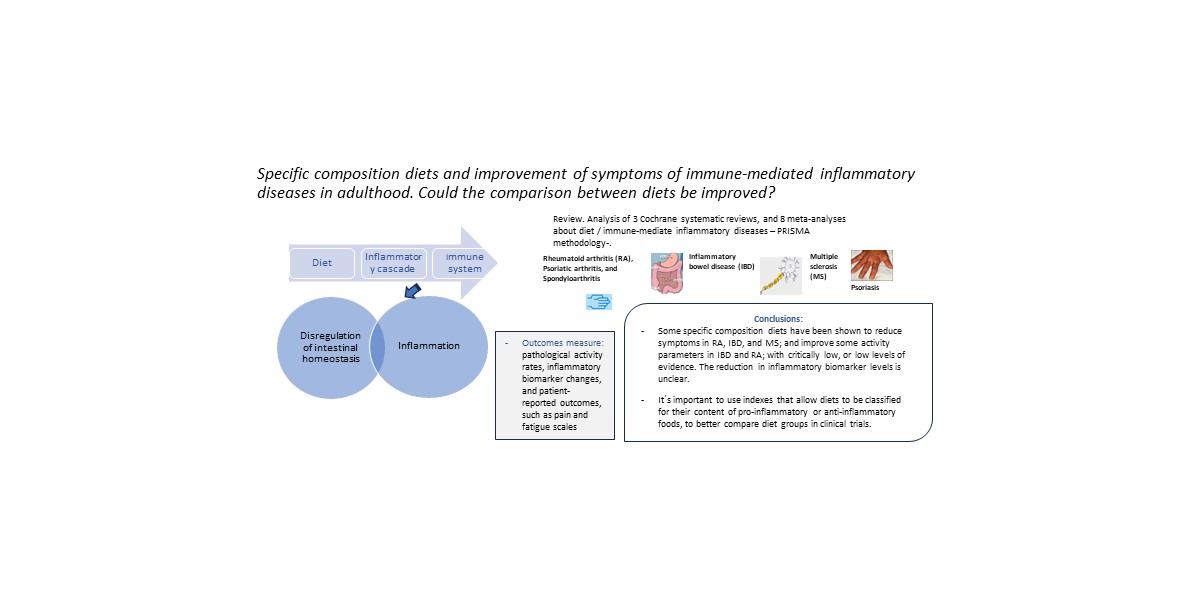
2. Methods
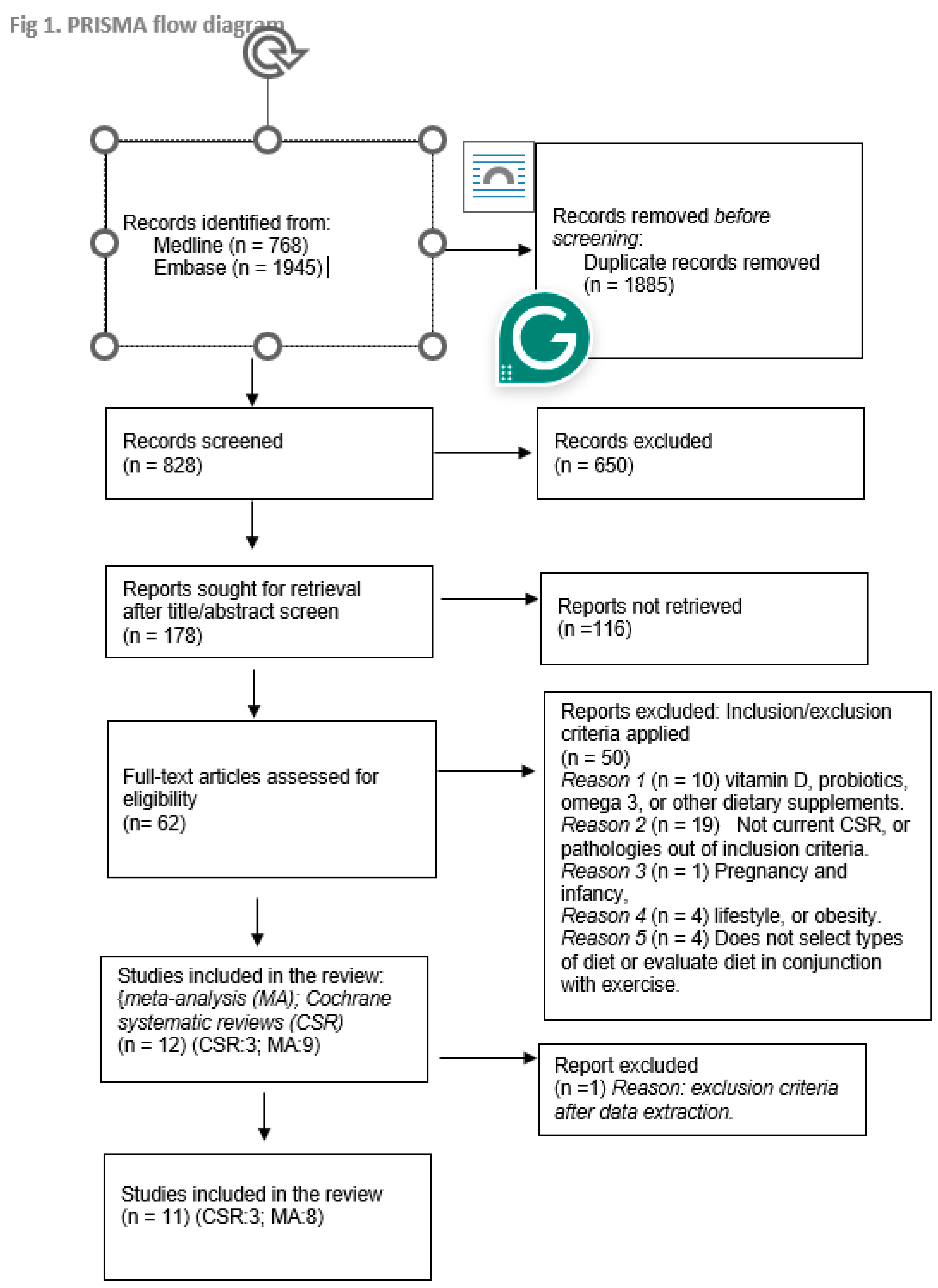
2.1. Search Methods for Identifying Studies
2.2. Eligibility Criteria Used for Literature Search and Screening
2.3. Data Extraction
2.4. Outcomes Measures
2.5. Quality of Meta-Analyses
3. Results
3.1. Characteristics of Eligible Studies
3.2. Cochrane SRs
3.2.1. Cochrane SR and Meta-Analyses on the Outcomes of Diet on Rheumatic and Musculoskeletal Diseases
3.2.2. Cochrane SR on the Outcomes of Diets in IBD
3.2.3. Cochrane SR on the Outcomes of Diets and MS
3.3. Meta-Analyses
3.3.1. RA, PsA, and SpA
3.3.2. IBD
3.3.3. MS
3.3.4. Psoriasis
4. Discussion
4.1. Efficacy of Diets with Specific Compositions to Reduce IMID Symptoms. Quality of Evidence
4.2. Measurement of the Inflammatory Effect of Different Components of the Diet
4.3. DII May Be Correlated with Circulating Inflammatory Marker Levels
4.4. Restriction of Common Components in Many Diets
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Schett G: McInnes IB, Neurath MF. Reframing immune-mediated inflammatory diseases through signature cytokine hubs. N Engl J Med 2021 Aug 12;385(7): 628–39. [CrossRef]
- El-Gabalawy, Hani, et al. “Epidemiology of immune-mediated inflammatory diseases: incidence, prevalence, natural history, and comorbidities.” The Journal of rheumatology. Supplement vol. 85 (2010): 2-10. [CrossRef]
- Monteleone, G., Moscardelli, A., Colella, A., Marafini, I., & Salvatori, S. (2023). Immune-mediated inflammatory diseases: Common and different pathogenic and clinical features. Autoimmunity reviews, 22(10), 103410. [CrossRef]
- Burisch, Johan et al. “Incidence of Immune-Mediated Inflammatory Diseases Among Patients With Inflammatory Bowel Diseases in Denmark.” Clinical gastroenterology and hepatology : the official clinical practice journal of the American Gastroenterological Association vol. 17,13 (2019): 2704-2712.e3. [CrossRef]
- Mazzucca, Camilla Barbero et al. “How to Tackle the Relationship between Autoimmune Diseases and Diet: Well Begun Is Half-Done.” Nutrients vol. 13,11 3956. 5 Nov. 2021. [CrossRef]
- Nezamoleslami, Shokufeh, et al. “The relationship between dietary patterns and rheumatoid arthritis: a case-control study.” Nutrition & metabolism vol. 17 75. 17 Sep. 2020. [CrossRef]
- Li, Tong, et al. “Systematic review and meta-analysis: Association of a pre-illness Western dietary pattern with the risk of developing inflammatory bowel disease.” Journal of digestive diseases vol. 21,7 (2020): 362-371. [CrossRef]
- Zitvogel, Laurence, and Guido Kroemer. “Immunostimulatory gut bacteria.” Science (New York, N.Y.) vol. 366,6469 (2019): 1077-1078. [CrossRef]
- Meade, Susanna, et al. “Gut Microbiome-Associated Predictors as Biomarkers of Response to Advanced Therapies in Inflammatory Bowel Disease: A Systematic Review.” Gut Microbes, vol. 15, no. 2, Dec. 2023. [CrossRef]
- Zhao, Ting et al. “Gut microbiota and rheumatoid arthritis: From pathogenesis to novel therapeutic opportunities.” Frontiers in immunology vol. 13 1007165. 8 Sep. 2022. [CrossRef]
- Mousa, Walaa K et al. “Microbial dysbiosis in the gut drives systemic autoimmune diseases.” Frontiers in immunology vol. 13 906258. 20 Oct. 2022. [CrossRef]
- Sorboni, Shokufeh Ghasemian et al. “A Comprehensive Review on the Role of the Gut Microbiome in Human Neurological Disorders.” Clinical microbiology reviews vol. 35,1 (2022): e0033820. [CrossRef]
- Zhang, Ping. “Influence of Foods and Nutrition on the Gut Microbiome and Implications for Intestinal Health.” International Journal of Molecular Sciences, vol. 23, no. 17, Aug. 2022, p. 9588. [CrossRef]
- Arroyo Hornero, R., Hamad, I., Côrte-Real, B., Kleinewietfeld, M., 2020. The Impact of Dietary Components on Regulatory T-cells and Disease. Front. Immunol. 11, 253. [CrossRef]
- Bäcklund, Reecka, et al. “Diet and the risk of rheumatoid arthritis - A systematic literature review.” Seminars in arthritis and rheumatism vol. 58 (2023): 152118. [CrossRef]
- Khademi, Zeinab, et al. “Dietary Intake of Total Carbohydrates, Sugar and Sugar-Sweetened Beverages, and Risk of Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis of Prospective Cohort Studies.” Frontiers in Nutrition, vol. 8, Oct. 2021. [CrossRef]
- Bolte, Laura A., et al. “Long-Term Dietary Patterns Are Associated with pro-Inflammatory and Anti-Inflammatory Features of the Gut Microbiome.” Gut, vol. 70, no. 7, Apr. 2021, pp. 1287–98. [CrossRef]
- Rastall, R. A., et al. “Structure and Function of Non-Digestible Carbohydrates in the Gut Microbiome.” Beneficial Microbes, vol. 13, no. 2, June 2022, pp. 95–168. [CrossRef]
- Serrano Fernandez, Victor, et al. “High-Fiber Diet and Crohn’s Disease: Systematic Review and Meta-Analysis.” Nutrients, vol. 15, no. 14, July 2023, p. 3114. [CrossRef]
- Mallon, K., et al. “P862 Dietary Risk Factors Associated with Onset of IBD: A Systematic Literature Review and Meta-Analysis.” Journal of Crohn’s and Colitis, vol. 17, no. Supplement_1, Jan. 2023, pp. i985–86. [CrossRef]
- Wagenaar, Carlijn A et al. “The Effect of Dietary Interventions on Chronic Inflammatory Diseases in Relation to the Microbiome: A Systematic Review.” Nutrients vol. 13,9 3208. 15 Sep. 2021. [CrossRef]
- Oliver, J. E., and A. J. Silman. “Risk Factors for the Development of Rheumatoid Arthritis.” Scandinavian Journal of Rheumatology, vol. 35, no. 3, Jan. 2006, pp. 169–74. [CrossRef]
- Graff, Erica, et al. “Dietary Intake and Systemic Inflammation: Can We Use Food as Medicine?”. Current nutrition reports, 10.1007/s13668-023-00458-z. 20 Jan. 2023.
- García-Montero, Cielo et al. “Nutritional Components in Western Diet Versus Mediterranean Diet at the Gut Microbiota-Immune System Interplay. Implications for Health and Disease.” Nutrients vol. 13,2 699. 22 Feb. 2021. [CrossRef]
- Fu, Yawei et al. “Associations among Dietary Omega-3 Polyunsaturated Fatty Acids, the Gut Microbiota, and Intestinal Immunity.” Mediators of inflammation vol. 2021 8879227. 2 Jan. 2021. [CrossRef]
- Gowen, Rachael et al. “Modulating the Microbiome for Crohn's Disease Treatment.” Gastroenterology vol. 164,5 (2023): 828-840. [CrossRef]
- Camara-Lemarroy, C.R., Metz, L., Meddings, J.B., Sharkey, K.A., Wee Yong, V., 2018. The intestinal barrier in multiple sclerosis: Implications for pathophysiology and therapeutics. Brain 141, 1900–1916. [CrossRef]
- Mu, Qinghui, et al. “Leaky Gut As a Danger Signal for Autoimmune Diseases.” Frontiers in Immunology, vol. 8, May 2017. [CrossRef]
- Jiang S, Miao Z. High-fat diet induces intestinal mucosal barrier dysfunction in ulcerative colitis: emerging mechanisms and dietary intervention perspective. Am J Transl Res. 2023 Feb 15;15(2):653-677. PMCID, 1000. [PubMed] [PubMed Central]
- de Punder K, Pruimboom L. The dietary intake of wheat and other cereal grains and their role in inflammation. Nutrients. 2013 Mar 12;5(3):771-87. [CrossRef] [PubMed] [PubMed Central]
- Huang X, Schuppan D, Rojas Tovar LE, Zevallos VF, Loponen J, Gänzle M. Sourdough Fermentation Degrades Wheat Alpha-Amylase/Trypsin Inhibitor (ATI) and Reduces Pro-Inflammatory Activity. Foods. 2020 Jul 16;9(7):943. [CrossRef] [PubMed] [PubMed Central]
- Esposito, S., Bonavita, S., Sparaco, M., Gallo, A., & Tedeschi, G. (2018). The role of diet in multiple sclerosis: A review. Nutritional neuroscience, 21(6), 377–390. [CrossRef]
- Bilal, Muhammad et al. “Dietary Component-Induced Inflammation and Its Amelioration by Prebiotics, Probiotics, and Synbiotics.” Frontiers in nutrition vol. 9 931458. 22 Jul. 2022. [CrossRef]
- Gershteyn, Iosif M., et al. “Immunodietica: Interrogating the Role of Diet in Autoimmune Disease.” International Immunology, vol. 32, no. 12, Aug. 2020, pp. 771–83. [CrossRef]
- Yang, Wenjing, and Yingzi Cong. “Gut Microbiota-Derived Metabolites in the Regulation of Host Immune Responses and Immune-Related Inflammatory Diseases.” Cellular & Molecular Immunology, vol. 18, no. 4, Mar. 2021, pp. 866–77. [CrossRef]
- Shivappa, Nitin, et al. “Designing and Developing a Literature-Derived, Population-Based Dietary Inflammatory Index.” Public Health Nutrition, vol. 17, no. 8, Aug. 2013, pp. 1689–96. [CrossRef]
- Hébert JR, Shivappa N, Wirth MD, Hussey JR, Hurley TG. Perspective: The Dietary Inflammatory Index (DII)-Lessons Learned, Improvements Made, and Future Directions. Adv Nutr. 2019 Mar 1;10(2):185-195. [CrossRef] [PubMed] [PubMed Central]
- Kwiat VR, Reis G, Valera IC, Parvatiyar K, Parvatiyar MS. Autoimmunity as a sequela to obesity and systemic inflammation. Front Physiol. 2022 Nov 21;13:887702. [CrossRef] [PubMed] [PubMed Central]
- Roman, S.N., Fitzgerald, K.C., Beier, M., Mowry, E.M., 2020. Safety and feasibility of various fasting-mimicking diets among people with multiple sclerosis. Mult. Scler. Relat. Disord. 42, 102149. [CrossRef]
- Ye, Ding, et al. “Lifestyle Factors Associated with Incidence of Rheumatoid Arthritis in US Adults: Analysis of National Health and Nutrition Examination Survey Database and Meta-Analysis.” BMJ Open, vol. 11, no. 1, Jan. 2021, p. e038137. [CrossRef]
- Ortolan, Augusta, et al. “Do Obesity and Overweight Influence Disease Activity Measures in Axial Spondyloarthritis? A Systematic Review and Meta-Analysis.” Arthritis care & research vol. 73,12 (2021): 1815-1825. [CrossRef]
- Liu, Yang et al. “Impact of Obesity on Remission and Disease Activity in Rheumatoid Arthritis: A Systematic Review and Meta-Analysis.” Arthritis care & research vol. 69,2 (2017): 157-165. [CrossRef]
- Moroni L, Farina N, Dagna L. Obesity and its role in the management of rheumatoid and psoriatic arthritis. Clin Rheumatol. 2020 Apr;39(4):1039-1047. [CrossRef] [PubMed]
- Ortolan, Augusta, et al. “The Impact of Diet on Disease Activity in Spondyloarthritis: A Systematic Literature Review.” Joint Bone Spine, vol. 90, no. 2, Mar. 2023, p. 105476. [CrossRef]
- Upala, S., and A. Sanguankeo. “Effect of Lifestyle Weight Loss Intervention on Disease Severity in Patients with Psoriasis: A Systematic Review and Meta-Analysis.” International Journal of Obesity, vol. 39, no. 8, Apr. 2015, pp. 1197–202. [CrossRef]
- Caputo, Marina, et al. “Targeting Microbiota in Dietary Obesity Management: A Systematic Review on Randomized Control Trials in Adults.” Critical Reviews in Food Science and Nutrition, June 2022, pp. 1–33. [CrossRef]
- “Index”. Cochrane Handbook for Systematic Reviews of Interventions, pp. 633–49. [CrossRef]
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. [CrossRef]
- Kurtzke JF. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology. 1983 Nov;33(11):1444-52. [CrossRef] [PubMed]
- Cohen, Jeffrey A., et al. “Disability Outcome Measures in Multiple Sclerosis Clinical Trials: Current Status and Future Prospects.” The Lancet Neurology, vol. 11, no. 5, May 2012, pp. 467–76. [CrossRef]
- Vickrey, B. G., et al. “A Health-Related Quality of Life Measure for Multiple Sclerosis.” Quality of Life Research, vol. 4, no. 3, June 1995, pp. 187–206. [CrossRef]
- Fisk, John D., et al. “Measuring the Functional Impact of Fatigue: Initial Validation of the Fatigue Impact Scale.” Clinical Infectious Diseases, vol. 18, no. Supplement_1, Jan. 1994, pp. S79–83. [CrossRef]
- Harvey, R F, and J M Bradshaw. “A simple index of Crohn's-disease activity.” Lancet (London, England) vol. 1,8167 (1980): 514. [CrossRef]
- Prevoo ML, van 't Hof MA, Kuper HH, van Leeuwen MA, van de Putte LB, van Riel PL. Modified disease activity scores that include twenty-eight joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum. 1995;38(1):44–8. [CrossRef]
- Statistical Power Analysis for the Behavioral Sciences. 1977. [CrossRef]
- Lu, Cuncun, et al. “Use of AMSTAR-2 in the methodological assessment of systematic reviews: protocol for a methodological study.” Annals of Translational Medicine vol. 8,10 (2020): 652. [CrossRef]
- Gwinnutt, James M., et al. “Effects of Diet on the Outcomes of Rheumatic and Musculoskeletal Diseases (RMDs): Systematic Review and Meta-Analyses Informing the 2021 EULAR Recommendations for Lifestyle Improvements in People with RMDs.” RMD Open, vol. 8, no. 2, June 2022, p. e002167. [CrossRef]
- Cramp, Fiona, et al. “Non-Pharmacological Interventions for Fatigue in Rheumatoid Arthritis.” Cochrane Database of Systematic Reviews, Aug. 2013. [CrossRef]
- Limketkai, Berkeley N., et al. “Dietary Interventions for Induction and Maintenance of Remission in Inflammatory Bowel Disease.” Cochrane Database of Systematic Reviews, Feb. 2019. [CrossRef]
- Limketkai, Berkeley N., et al. “Dietary Interventions for the Treatment of Inflammatory Bowel Diseases: An Updated Systematic Review and Meta-analysis.” Clinical Gastroenterology and Hepatology, Dec. 2022. [CrossRef]
- Parks, Natalie E., et al. “Dietary Interventions for Multiple Sclerosis-Related Outcomes.” Cochrane Database of Systematic Reviews, vol. 2020, no. 5, May 2020. [CrossRef]
- Schönenberger, Katja A., et al. “Effect of Anti-Inflammatory Diets on Pain in Rheumatoid Arthritis: A Systematic Review and Meta-Analysis.” Nutrients, vol. 13, no. 12, Nov. 2021, p. 4221. [CrossRef]
- Genel, Furkan, et al. “Health Effects of a Low-Inflammatory Diet in Adults with Arthritis: A Systematic Review and Meta-Analysis.” Journal of Nutritional Science, vol. 9, 2020. [CrossRef]
- Turk, Matthew A et al. “Non-pharmacological interventions in the treatment of rheumatoid arthritis: A systematic review and meta-analysis.” Autoimmunity reviews vol. 22,6 (2023): 103323. [CrossRef]
- Ortolan, Augusta, et al. “Efficacy and Safety of Non-Pharmacological and Non-Biological Interventions: A Systematic Literature Review Informing the 2022 Update of the ASAS/EULAR Recommendations for the Management of Axial Spondyloarthritis.” Annals of the Rheumatic Diseases, vol. 82, no. 1, Oct. 2022, pp. 142–52. [CrossRef]
- Comeche, José M., et al. “Predefined Diets in Patients with Inflammatory Bowel Disease: Systematic Review and Meta-Analysis.” Nutrients, vol. 13, no. 1, Dec. 2020, p. 52. [CrossRef]
- Zhan, Yong-le, et al. “Is a Low FODMAP Diet Beneficial for Patients with Inflammatory Bowel Disease? A Meta-Analysis and Systematic Review.” Clinical Nutrition, vol. 37, no. 1, Feb. 2018, pp. 123–29. [CrossRef]
- Peng, Ziheng, et al. “A Low-FODMAP Diet Provides Benefits for Functional Gastrointestinal Symptoms but Not for Improving Stool Consistency and Mucosal Inflammation in IBD: A Systematic Review and Meta-Analysis.” Nutrients, vol. 14, no. 10, May 2022, p. 2072. [CrossRef]
- Guerrero Aznar, María Dolores et al. “Efficacy of diet on fatigue, quality of life and disability status in multiple sclerosis patients: rapid review and meta-analysis of randomized controlled trials.” BMC neurology vol. 22,1 388. 20 Oct. 2022. [CrossRef]
- Bohlouli, Jalal, et al. “Modified Mediterranean Diet v. Traditional Iranian Diet: Efficacy of Dietary Interventions on Dietary Inflammatory Index Score, Fatigue Severity and Disability in Multiple Sclerosis Patients.” British Journal of Nutrition, Aug. 2021, pp. 1–11. [CrossRef]
- Snetselaar, Linda, et al. “Efficacy of Diet on Fatigue and Quality of Life in Multiple Sclerosis: A Systematic Review and Network Meta-Analysis of Randomized Trials.” Neurology, Oct. 2022, p. 10.1212/WNL.0000000000201371. [CrossRef]
- Ford, Adam R., et al. “Dietary Recommendations for Adults With Psoriasis or Psoriatic Arthritis From the Medical Board of the National Psoriasis Foundation.” JAMA Dermatology, vol. 154, no. 8, Aug. 2018, p. 934. [CrossRef]
- Gill, Paul A., et al. “The Role of Diet and Gut Microbiota in Regulating Gastrointestinal and Inflammatory Disease.” Frontiers in Immunology, vol. 13, Apr. 2022. [CrossRef]
- Johnston, Bradley C., et al. “The Philosophy of Evidence-Based Principles and Practice in Nutrition.” Mayo Clinic Proceedings: Innovations, Quality & Outcomes, vol. 3, no. 2, June 2019, pp. 189–99. [CrossRef]
- Nelson, Josefine et al. “Do Interventions with Diet or Dietary Supplements Reduce the Disease Activity Score in Rheumatoid Arthritis? A Systematic Review of Randomized Controlled Trials.” Nutrients vol. 12,10 2991. 29 Sep. 2020. [CrossRef]
- Lomer MCE, Wilson B, Wall CL. British Dietetic Association consensus guidelines on the nutritional assessment and dietary management of patients with inflammatory bowel disease. J Hum Nutr Diet. 2023 Feb;36(1):336-377. [CrossRef] [PubMed] [PubMed Central]
- Zhu, Zhanhui, et al. “Effects of Crohn’s Disease Exclusion Diet on Remission: A Systematic Review.” Therapeutic Advances in Gastroenterology, vol. 16, Jan. 2023. [CrossRef]
- Bischoff, S.C.; Bager, P.; Escher, J.; Forbes, A.; Hébuterne, X.; Hvas, C.L.; Joly, F.; Klek, S.; Krznaric, Z.; Ockenga, J.; et al. ESPEN guideline on Clinical Nutrition in inflammatory bowel disease. Clin. Nutr. 2023, 42, 352–379 [CrossRef] [PubMed]. [Google Scholar] [CrossRef] [PubMed]
- Correia, Inês et al. “Is There Evidence of Crohn's Disease Exclusion Diet (CDED) in Remission of Active Disease in Children and Adults? A Systematic Review.” Nutrients vol. 16,7 987. 28 Mar. 2024. [CrossRef]
- Herrador-López, Marta, et al. “Dietary Interventions in Ulcerative Colitis: A Systematic Review of the Evidence with Meta-Analysis.” Nutrients, vol. 15, no. 19, Sept. 2023, p. 4194. [CrossRef]
- Weissman, Simcha, et al. “The Overall Quality of Evidence of Recommendations Surrounding Nutrition and Diet in Inflammatory Bowel Disease.” International Journal of Colorectal Disease, vol. 38, no. 1, Apr. 2023. [CrossRef]
- Chiba M, Hosoba M, Yamada K. Plant-Based Diet Recommended for Inflammatory Bowel Disease. Inflamm Bowel Dis. 2023 May 2;29(5):e17-e18. [CrossRef] [PubMed] [PubMed Central]
- Kheirouri, Sorayya, and Mohammad Alizadeh. “Dietary Inflammatory Potential and the Risk of Neurodegenerative Diseases in Adults.” Epidemiologic Reviews, vol. 41, no. 1, 2019, pp. 109–20. [CrossRef]
- Hajianfar, Hossein, et al. “Association between Dietary Inflammatory Index and Risk of Demyelinating Autoimmune Diseases.” International Journal for Vitamin and Nutrition Research, Mar. 2022. [CrossRef]
- Rad, Esmaeil Yousefi et al. “A systematic review and meta-analysis of Dietary Inflammatory Index and the likelihood of multiple sclerosis/ demyelinating autoimmune disease.” Clinical nutrition ESPEN vol. 62 (2024): 108-114. [CrossRef]
- Mirhosseini, Seyed Mohsen et al. “What is the link between the dietary inflammatory index and the gut microbiome? A systematic review.” European journal of nutrition, 10.1007/s00394-024-03470-3. 28 Jul. 2024. [CrossRef]
- Lerner, Aaron, et al. “Gluten-Free Diet Can Ameliorate the Symptoms of Non-Celiac Autoimmune Diseases.” Nutrition Reviews, vol. 80, no. 3, Aug. 2021, pp. 525–43. [CrossRef]
- Bhatia, Bhavnit K., et al. “Diet and Psoriasis, Part II: Celiac Disease and Role of a Gluten-Free Diet.” Journal of the American Academy of Dermatology, vol. 71, no. 2, Aug. 2014, pp. 350–58. [CrossRef]
- Bell, Katheryn A., et al. “The Effect of Gluten on Skin and Hair: A Systematic Review.” Dermatology Online Journal, vol. 27, no. 4, 2021. [CrossRef]
- Dieterich, Walburga, and Yurdagül Zopf. “Gluten and FODMAPS—Sense of a Restriction/When Is Restriction Necessary?” Nutrients, vol. 11, no. 8, Aug. 2019, p. 1957. [CrossRef]
- Cenni S, Sesenna V, Boiardi G, Casertano M, Russo G, Reginelli A, Esposito S, Strisciuglio C. The Role of Gluten in Gastrointestinal Disorders: A Review. Nutrients. 2023 Mar 27;15(7):1615. [CrossRef] [PubMed] [PubMed Central]
- Zhu, Zhanhui et al. “Effects of Crohn's disease exclusion diet on remission: a systematic review.” Therapeutic advances in gastroenterology vol. 16 17562848231184056. 30 Aug. 2023. [CrossRef]
| Reference | Study design | Studies | Pathology | Intervention/exposure/control | Outcomes | Main Results |
|---|---|---|---|---|---|---|
| (Genel et al., 2020) [63] | SR and MA | 5 RCT (2 RA, 3 OA), 2 PPT (RA). 468 P. I: 259; C:226. |
RA, OA, Seronegative arthropathies (psoriatic, reactive, ankylosing spondylitis or IBD-related) | Low-inflammatory diet, Anti-inflammatory diet, Mediterranean diet -caloric or non-caloric restricted-, to the usual diet. |
Meta-analysis (not including ketogenic diet) of 1) weight change 2) CRP, Interleukin 6. 3)Pain score |
1)Weight SMD −0⋅45 (CI −0⋅71, −0⋅18). 2) CRP: - 50,65 mg/l (-83,4, -18,26), in RA patients. Inflammatory biomarkers (SMD -2,33. (CI −3⋅82, −0⋅84), P=0,002, OA+RA patients. RA group (SMD −1⋅10 [95 % CI −1⋅71, −0⋅49], P = 0⋅0004 3)Joint pain (SMD −0⋅98; CI −2⋅90, 0⋅93), P=0,31. In a subgroup analysis pooled pain scores in a RA group were (SMD -2,81 CI -3,6, -2,02), P<0,00001. |
| (Schönenbergeret al., 2021) [62] | SR and MA | 7 RCT. 326 P. (92% female P.). | RA | Mediterranean, vegetarian, vegan, and ketogenic diets, to usual diet. | Meta-analysis of RA pain, and CRP. | Anti-inflammatory diets resulted in significantly lower pain than ordinary diets (−9.22 mm; 95% CI −14.15 to −4.29; p = 0.0002. There were no significant differences in CRP levels (SMD -2,51 CI -6,10, -1,08). |
| (Turk et al., 2023) [64] | SR and MA | 11 RCT. | RA | Dietary changes. | Meta-analyses of RA DAS28, and pain. | DAS28 was significantly improved in the group treated with diet (SMD − 0.46 CI− 0.91, − 0.02), p = 0.04, with a very low level of evidence. Pain (SMD -1,23 CI -1,96, -0,5), P=0,001. |
| (Comeche et al., 2020) [66] | SR and MA | 31 research studies. MA: 10 trials: RCT, UNRCT, NRCT. 279 P. |
IBD (CD, UC) | Low microparticle diet, semi-vegetarian diet (lacto-ovo-vegetarian diet- fish once a week and meat once every 2 weeks, both of them at half the average amount, bread rarely), IgG4-guided exclusion diet (CD), and others. to the usual diet. | Meta-analyses of CDAI for CD patients. FC, CRP, and ALB, for IBD patients. | A tendency to CDAI reduction (SMD -107,62 CI -182,12; -33,12), p<0,01. No differences were observed for CRP (SMD -0,45 CI-1,06, 0,16), ALB (SMD 0,16 CI 0,09, 0,41), and FC (SMD -7,40 CI -54,12, 39,32). |
| (Zhan et al., 2018) [67] | SR and MA | 2 RCTs and 4 before-after studies. 319 P. | IBD | LFD to the usual diet. | The effect of diets on diarrhea, abdominal bloating, abdominal pain, fatigue, and nausea. | Improvement in other symptoms: diarrhea response (OR: 0.24, 95% CI: 0.11–0.52, p = 0.0003), abdominal bloating (OR: 0.10, 95% CI: 0.06–0.16, p < 0.00001), abdominal pain (OR: 0.24, 95% CI: 0.16–0.35, p < 0.00001), fatigue (OR: 0.40, 95% CI: 0.24–0.66, p = 0.0003) and nausea (OR: 0.51, 95% CI: 0.31–0.85, p = 0.009). Very low level of evidence. |
| (Peng et al, 2022) [68] | SR and MA | 4 RCTs and 5 before– after studies. 446 P. (96% in remission). | IBD | LFD to the usual diet. | FGSs. QoL -SIBDQ-(only 2 studies). | Overall FGSs (RR: 0.47, 95% CI: 0.33–0.66, p = 0.0000). QoL -SIBDQ (MD = 11.24, 95% CI 6.61 to 15.87, p = 0.0000). CD patients (RR: 0,44 CI 0,34-0,55) UC (RR: 0,43 CI 0,33-0,56). No statistically significant differences in normal stool consistency and mucosal inflammation. |
| (Guerrero Aznar et al., 2022) [69] | RR and MA | 8 RCT. 515 P. | MS | Modified anti-inflammatory diet, Modified Mediterranean diet, Fasting-Mimicking-diet + Mediterranean diet, Modified Paleolithic diet -gluten-free-, Co- hot nature diet, very low-fat plant-based Diet, Supplemented Mediterranean-type diet, to usual diet. | The effect of some anti-inflammatory diets on EDSS, MFIS, and QOL. | MA: A trend of reduction in fatigue (MFIS) (308 pat.) SMD -2,033, 95%-IC (-3,195, -0,152), p-value: 0.0341)-, increase in QOL Physical (77 pat.), SMD 1.297, (0.2454,2.3485). p-value of 0,01; and in QOL mental (44 pat.), SMD 1.1086, 95%-CI (0.6143,1.6029). p < 0,0001. No significant effect on EDSS (337 pat.), and no severe adverse events. |
| (Snetselaar et al., 2023) [71] | SR and NWMA | 12 RT. 608 P. | MS | Low-fat, Mediterranean, ketogenic, anti-inflammatory, Paleolithic, fasting, calorie restriction, to the usual diet. | The effect of diets on MFIS, and QOL. | NWMA: The Paleolithic (SMD -1.27; 95% CI -1.81 to -0.74), low-fat (SMD -0.90; 95% CI -1.39 to -0.42), and Mediterranean (SMD -0.89; 95% CI -1.15 to -0.64) diets showed greater reductions in fatigue compared with control. The Paleolithic and Mediterranean diets showed greater improvements in physical and mental QoL compared with the control. Very low level of evidence. |
| References | ITEMS | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | Final rating | |
| (Genel et al., 2020) [63] | Y | Y | Y | pY | Y | Y | Y | Y | Y | N | N | Y | N | N | NA | Y | Y:10, pY:1, N:4, NA:1 |
| (Schönenberger et al., 2021) [62] | Y | Y | Y | pY | Y | Y | Y | Y | Y | N | Y | Y | Y | N | NA | Y | Y:12, pY:1, N:2, NA:1 |
| (Turk et al., 2023) [64] | Y | pY | Y | pY | Y | N | pY | y | Y | N | Y | Y | Y | N | Y | Y | Y:9, pY:4, N:3 |
| (Comeche et al., 2020) [66] | Y | pY | Y | pY | Y | Y | Y | Y | Y | Y | Y | Y | Y | N | NA | Y | Y:11, pY:3, N:1, NA:1 |
| (Zhan et al., 2018) [67] | Y | pY | Y | pY | N | Y | pY | Y | pY | N | Y | Y | N | N | NA | Y | Y:8, pY:4, N:3, NA:1 |
| (Peng et al, 2022) [68] | Y | Y | Y | pY | Y | Y | pY | Y | Y | N | Y | Y | Y | Y | y | Y | Y:13, pY:2, N:1, |
| (Guerrero Aznar et al., 2022) [69] | Y | pY | Y | pY | Y | Y | Y | Y | pY | N | Y | Y | Y | Y | NA | Y | Y:10, pY:3, N:2, NA:1 |
| (Snetselaar et al., 2023) [71] | Y | Y | Y | Y | Y | Y | Y | Y | Y | N | N | Y | N | N | Y | Y | Y:12, N:4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





