Submitted:
15 December 2024
Posted:
16 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
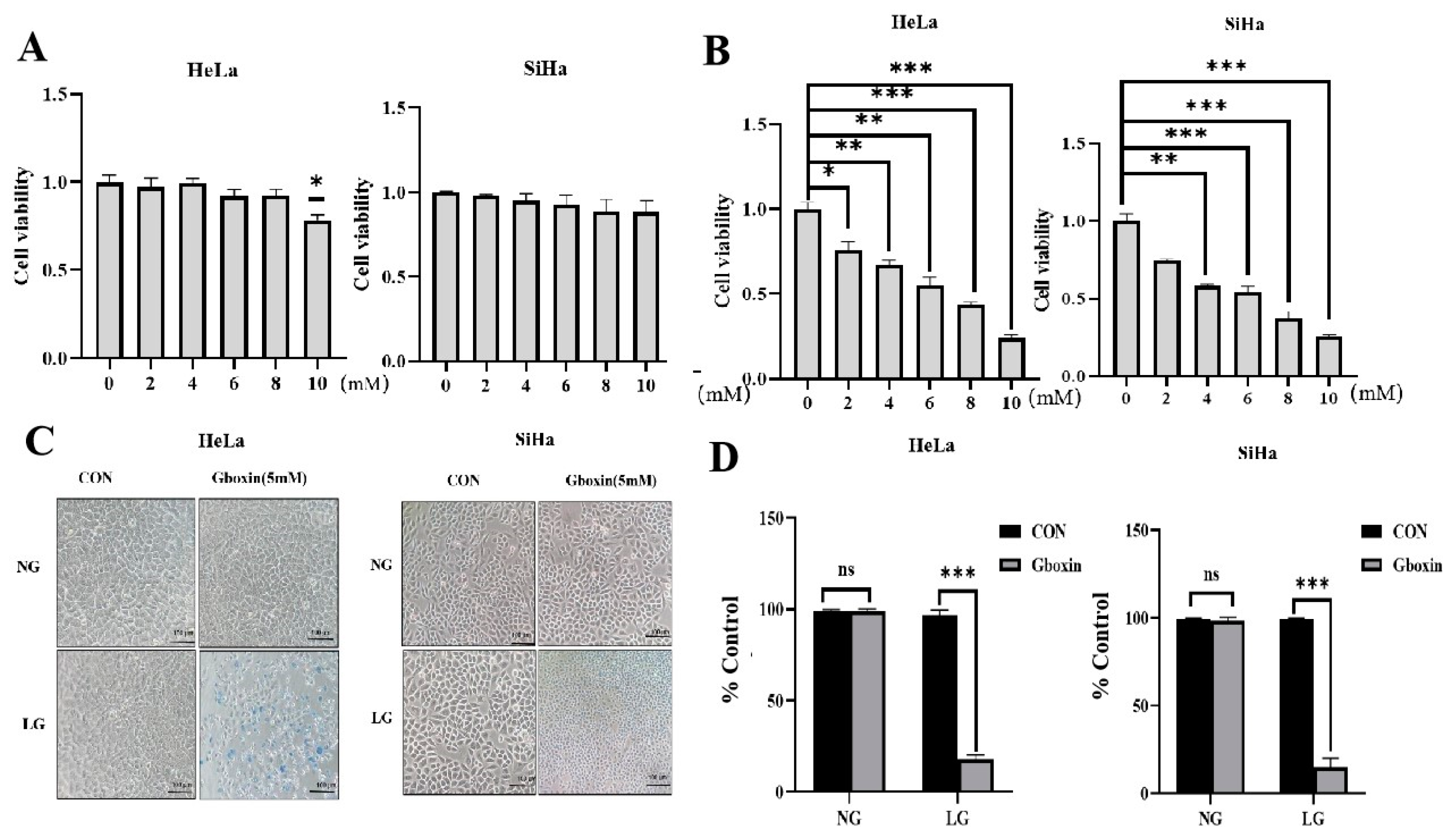
2.1. Gboxin Inhibited the Viability of Cervical Cancer Cells Under Low-Glucose Conditions
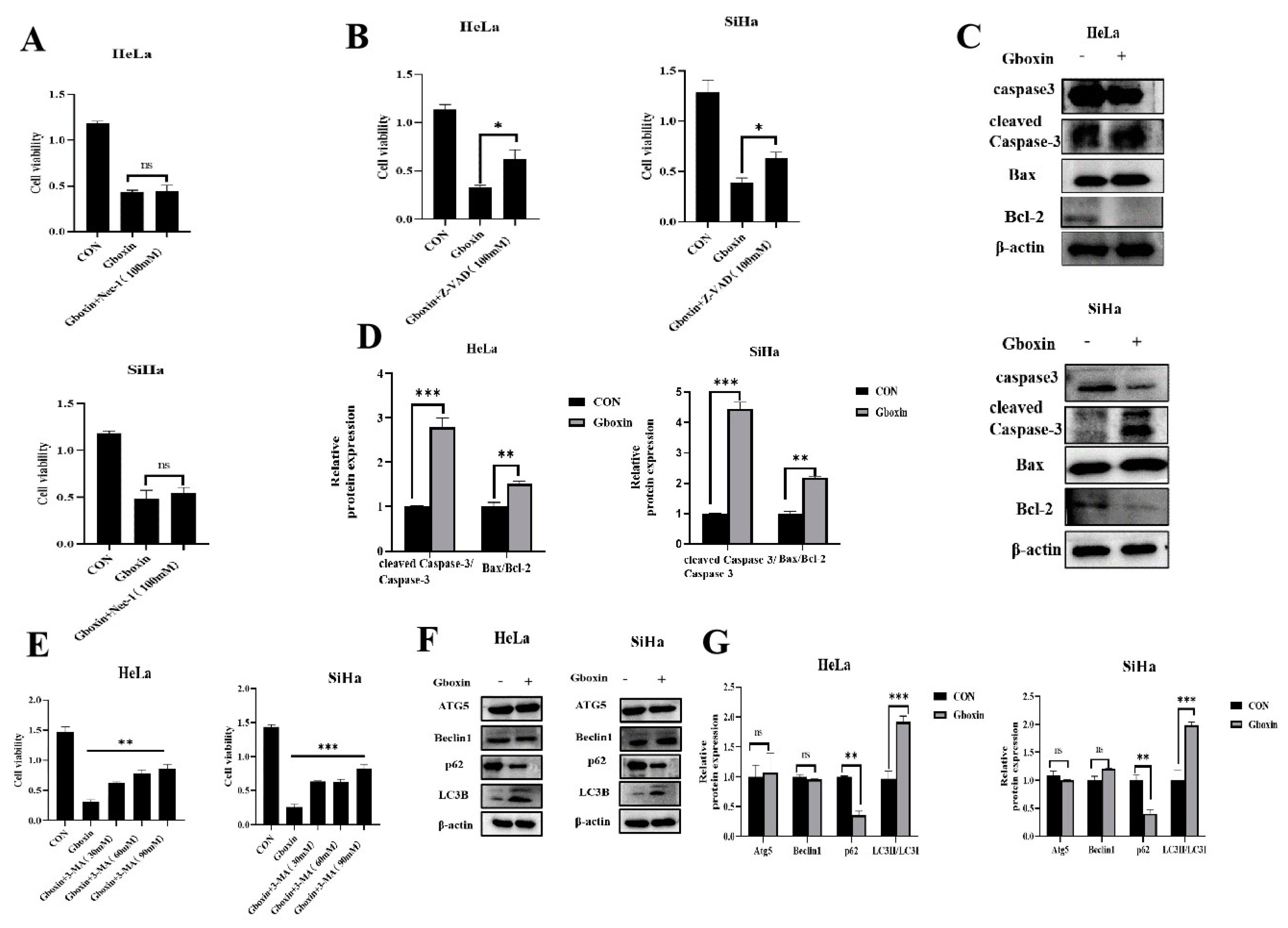
2.2. Gboxin Induced Autophagy and Apoptosis of Cervical Cancer Cells
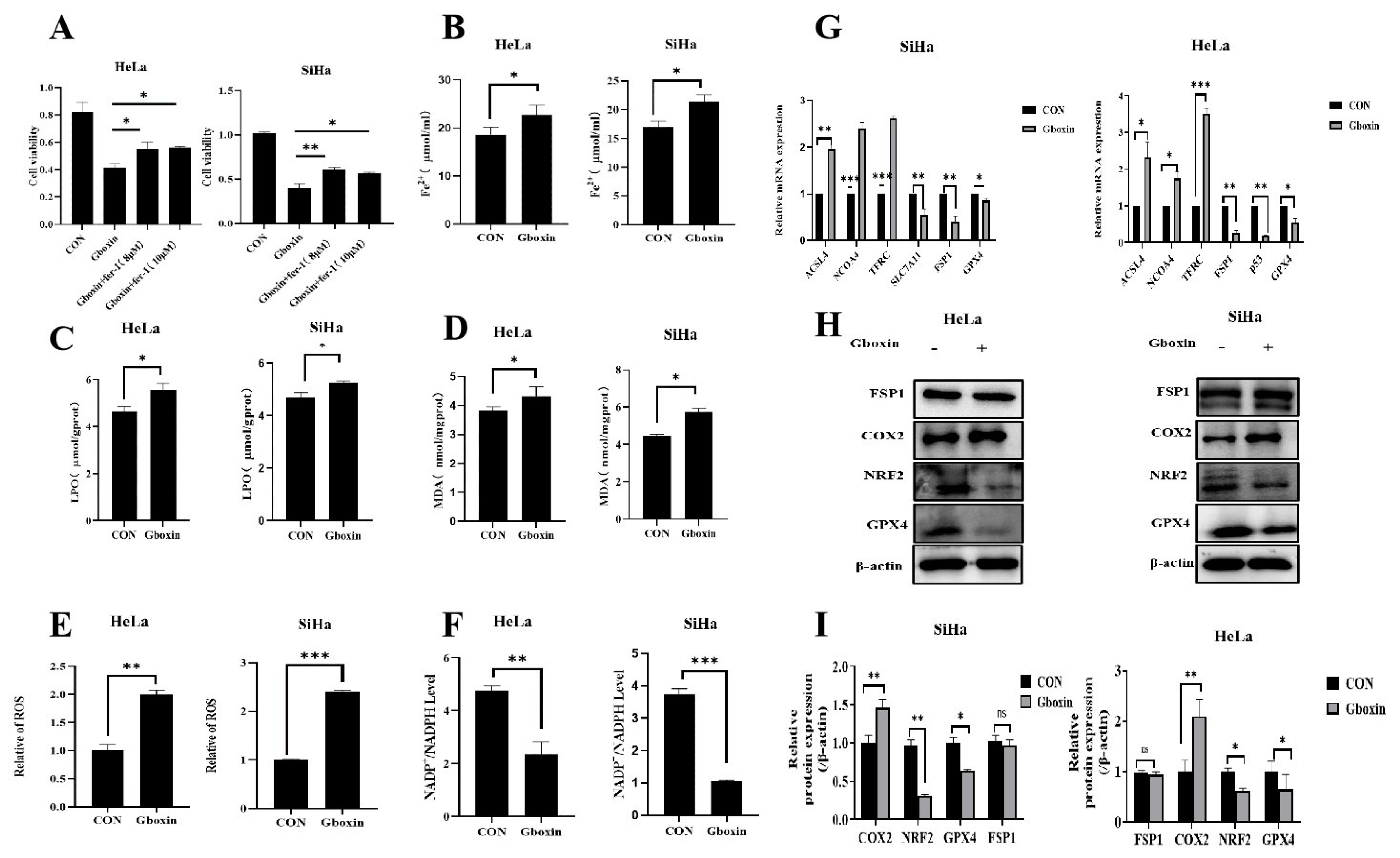
2.3. Gboxin Induced Ferroptosis of Cervical Cancer Cells
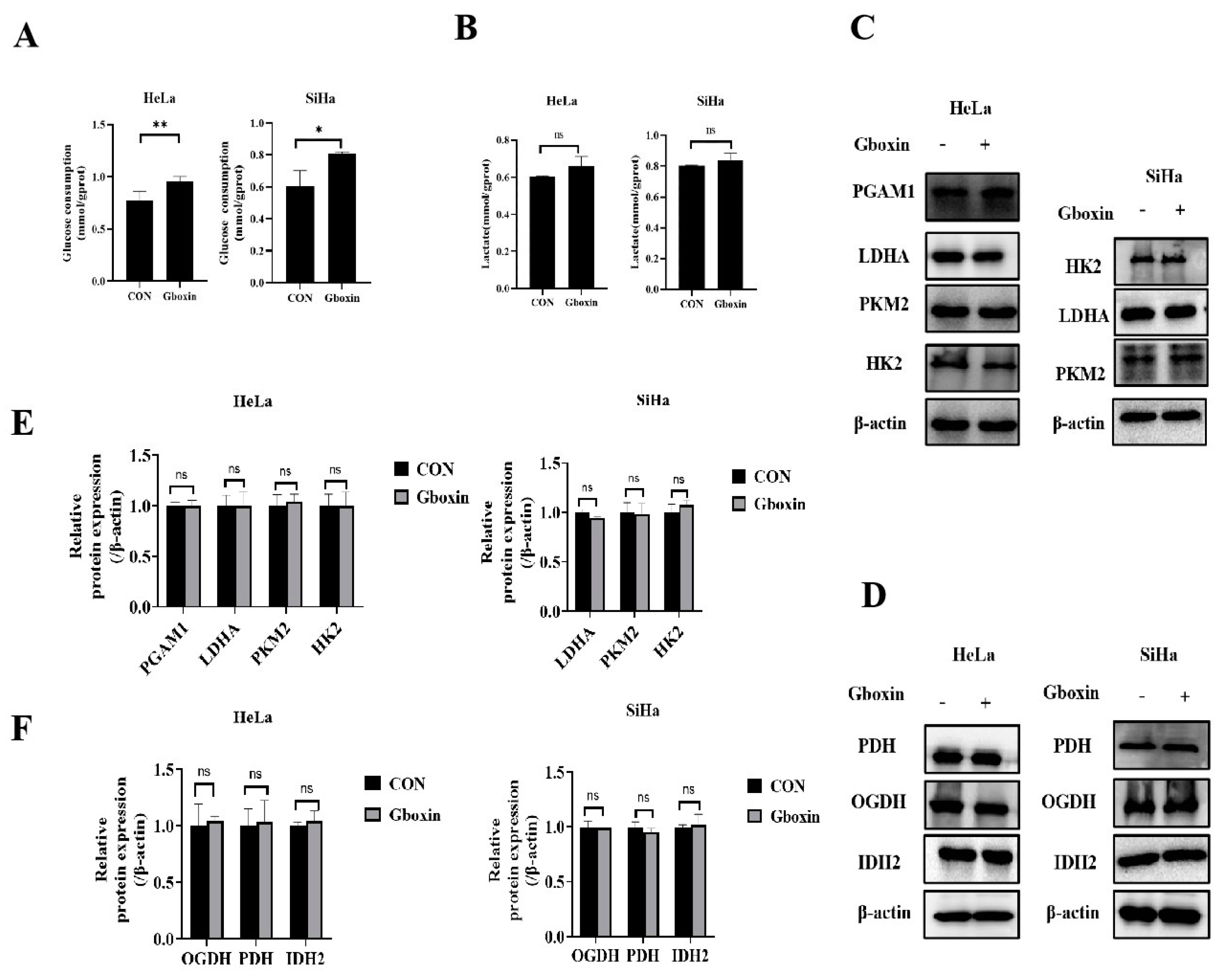
2.4. Gboxin Had no Effect on Glycolysis and Expression of Rate-Limiting Enzymes in TCA Cycle
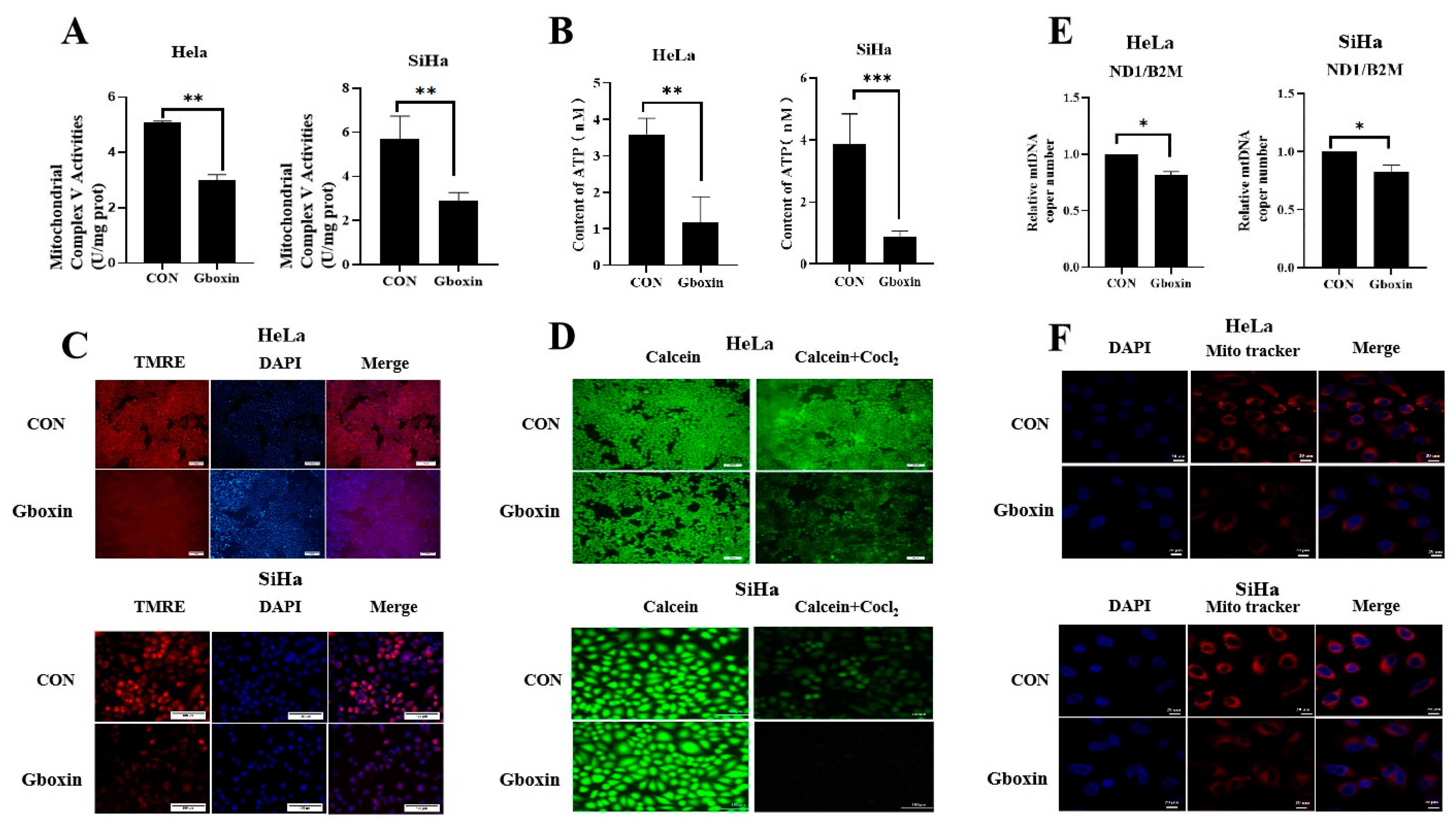
2.5. Gboxin Treatment Resulted in Mitochondrial Dysfunction Under Low-Glucose Conditions
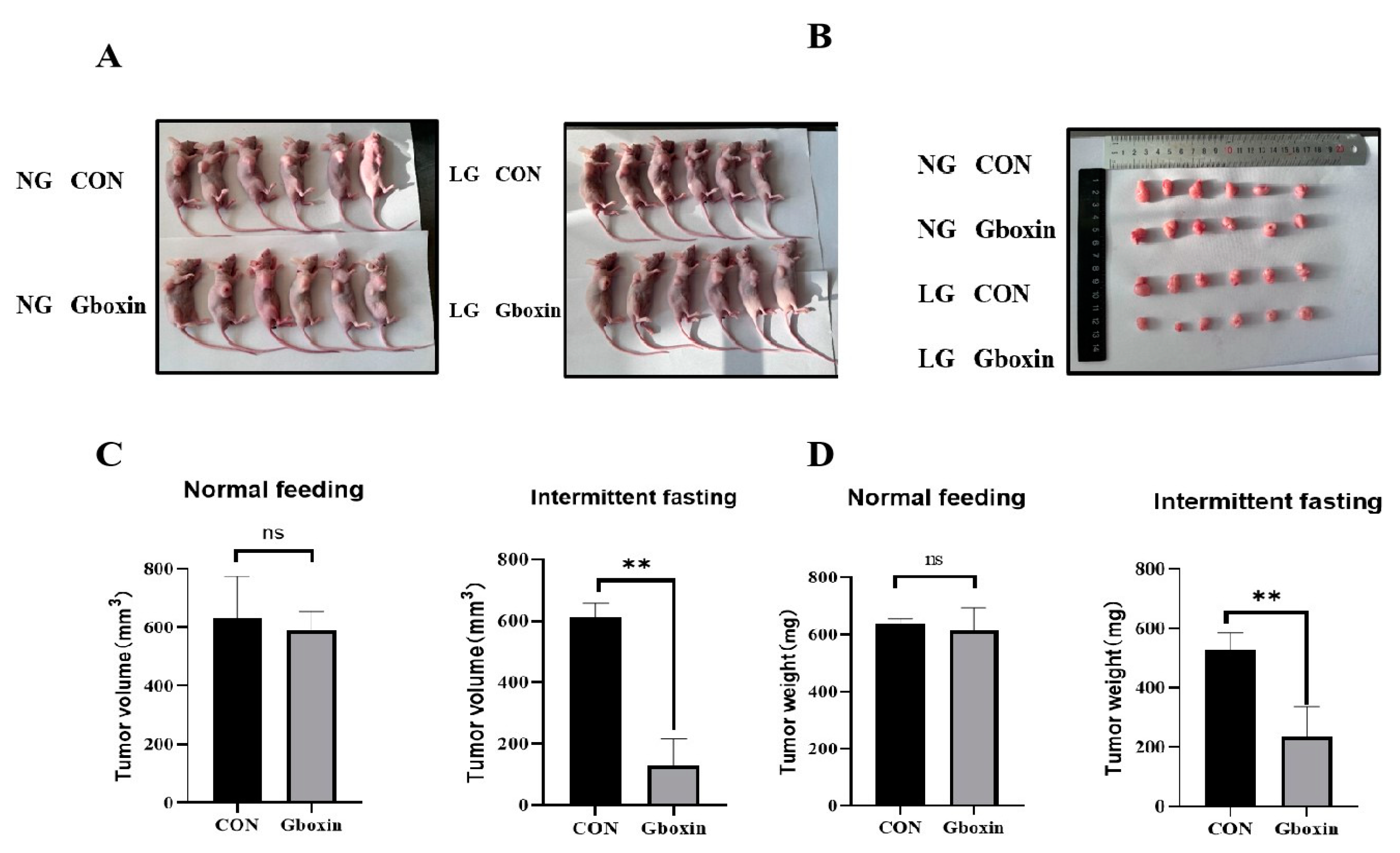
2.6. Gboxin Inhibited Tumor Growth Under Nutrient-Deprivation Conditions
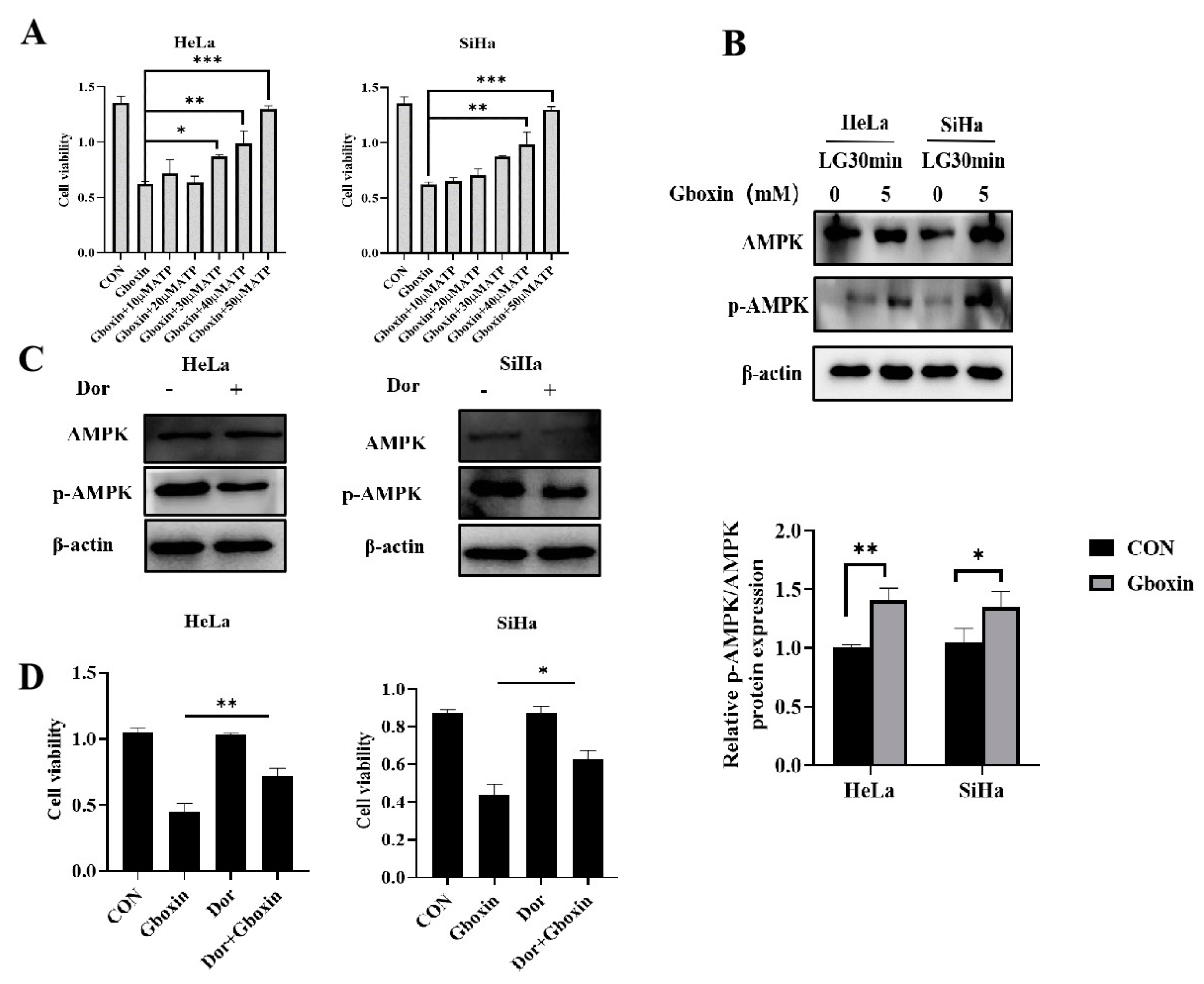
2.7. AMPK Signaling Pathway Was Involved in Gboxin-Induced Cell Death
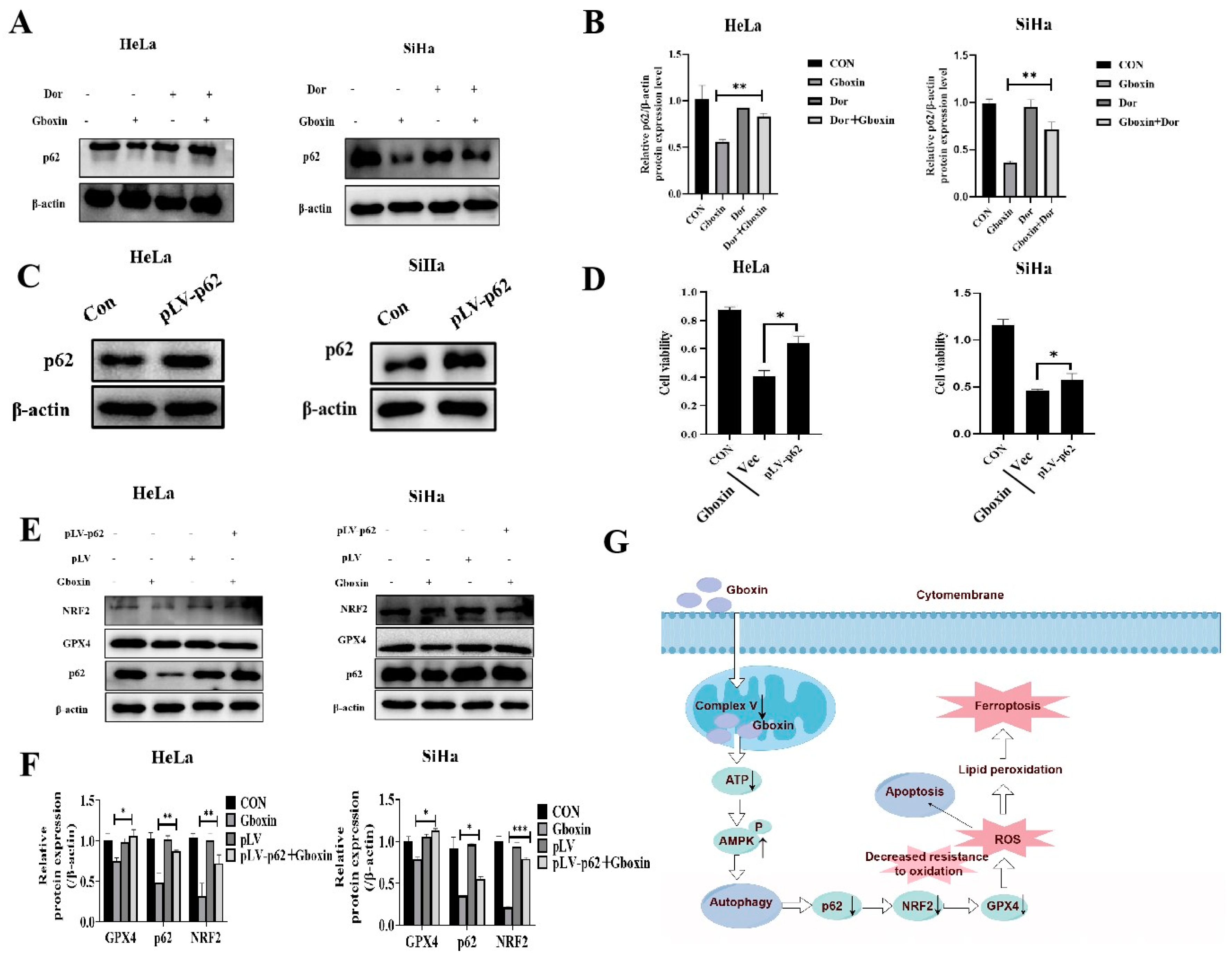
2.8. Overexpression of p62 Protein Reversed the Inhibitory Effect of Gboxin
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Cell Transfection
4.3. Cell Protein Extraction and Western Blotting Analysis
4.4. Trypan Blue Staining
4.5. MTT Assay
4.6. Antibodies
4.7. Measurement of NADP+/NADPH Ratio
4.8. Measurement of Lipid Peroxidation(LPO) and Malondialdehyde(MDA)
4.9. Intracellular Fe2+ Assay
4.10. Intracellular ROS Measurement
4.11. RNA Extraction and qRT-PCR Detection
| T | Title 2 | Title 3 |
| β-actin | F-Primer | CGTGCGTGACATTAAGGAGAAG |
| R-Primer | GGAAGGAAGGCTGGAAGAGTG | |
| p53 | F-Primer | CAGCACATGACGGAGGTTGT |
| R-Primer | TCATCCAAATACTCCACACGC | |
| ACSL4 | F-Primer | CATCCCTGGAGCAGATACTCT |
| R-Primer | TCACTTAGGATTTCCCTGGTCC | |
| NCOA4 | F-Primer | GAGGTGTAGTGATGCACGGAG |
| R-Primer | GACGGCTTATGCAACTGTGAA | |
| GPX4 | F-Primer | GAGGCAAGACCGAAGTAAACTAC |
| R-Primer | CCGAACTGGTTACACGGGAA | |
| FSP1 | F-Primer | AGACAGGGTTCGCCAAAAAGA |
| R-Primer | CAGGTCTATCCCCACT ACTAGC | |
| TFRC | F-Primer | ACCATTGTCATATACCCGGTTCA |
| R-Primer | CAATAGCCCAAGTAGCCAATCAT | |
| SLC7A11 | F-Primer | TCTCCAAAGGAGGTTACCTGC |
| R-Primer | AGACTCCCCTCAGTAAAGTGAC | |
| ND1 | F-Primer | CCCTAAAACCCGCCACATCT |
| R-Primer | GAGCGATGGTGAGAGCTAAGGT | |
| B2M | F-Primer | TGCTGTCTCCATGTTTGATGTATCT |
| R-Primer | TCTCTGCTCCCCACCTCTAAGT |
4.12. Lactate Production Assay
4.13. ATP Production Assay
4.14. Glucose Consumption Assay
4.15. Measurement of Mitochondrial Respiratory Chain Complex V Activity
4.16. Membrane Potential Measurement
4.17. MPTP Assay
4.18. Mitochondrial DNA (mtDNA) Copy Number Detection
4.19. Tumor Xenograft Studies
4.20. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hanahan, D., Hallmarks of Cancer: New Dimensions. Cancer Discovery, 2022. 12(1): p. 31-46. [CrossRef]
- Christine L. Chaffer, Robert A. Weinberg ,A Perspective on Cancer Cell Metastasis. Science331, 1559-1564(2011). [CrossRef]
- Pajak, B., et al., 2-Deoxy-d-Glucose and Its Analogs: From Diagnostic to Therapeutic Agents. International Journal of Molecular Sciences, 2019. 21(1). [CrossRef]
- McGuirk, S., Y. Audet-Delage, and J. St-Pierre, Metabolic Fitness and Plasticity in Cancer Progression. Trends in Cancer, 2020. 6(1): p. 49-61. [CrossRef]
- Chaube B, Malvi P, Singh SV, Mohammad N, Meena AS, Bhat MK. Targeting metabolic flexibility by simultaneously inhibiting respiratory complex I and lactate generation retards melanoma progression. Oncotarget. 2015 Nov 10;6(35):37281-99. [CrossRef]
- Sun, L., H. Zhang, and P. Gao, Metabolic reprogramming and epigenetic modifications on the path to cancer. Protein & Cell, 2021. 13(12): p. 877-919. [CrossRef]
- Bonnay, F., et al., Oxidative Metabolism Drives Immortalization of Neural Stem Cells during Tumorigenesis. Cell, 2020. 182(6): p. 1490-1507.e19. [CrossRef]
- Zhao Z, Fu A. [Mitochondrial therapy: a new strategy for treating mitochondrion-associated diseases]. Sheng Wu Gong Cheng Xue Bao. 2021 Apr 25;37(4):1168-1177. [CrossRef]
- Whitehall, J.C. and L.C. Greaves, Aberrant mitochondrial function in ageing and cancer. Biogerontology, 2019. 21(4): p. 445-459. [CrossRef]
- Tsuji, A., et al., IACS-010759, a potent inhibitor of glycolysis-deficient hypoxic tumor cells, inhibits mitochondrial respiratory complex I through a unique mechanism. Journal of Biological Chemistry, 2020. 295(21): p. 7481-7491. [CrossRef]
- Janku, F., et al., First-in-human evaluation of the novel mitochondrial complex I inhibitor ASP4132 for treatment of cancer. Investigational New Drugs, 2021. 39(5): p. 1348-1356. [CrossRef]
- Baccelli, I., et al., Mubritinib Targets the Electron Transport Chain Complex I and Reveals the Landscape of OXPHOS Dependency in Acute Myeloid Leukemia. Cancer Cell, 2019. 36(1): p. 84-99.e8. [CrossRef]
- Nuevo-Tapioles, C., et al., Coordinate β-adrenergic inhibition of mitochondrial activity and angiogenesis arrest tumor growth. Nature Communications, 2020. 11(1). [CrossRef]
- Shi, Y., et al., Gboxin is an oxidative phosphorylation inhibitor that targets glioblastoma. Nature, 2019. 567(7748): p. 341-346. [CrossRef]
- Lv, X., et al., The crosstalk between ferroptosis and autophagy in cancer. Autoimmunity, 2023. 56(1). [CrossRef]
- Pekson, R., et al., The mitochondrial ATP synthase is a negative regulator of the mitochondrial permeability transition pore. Proceedings of the National Academy of Sciences, 2023. 120(51). [CrossRef]
- Luo, Y., et al., Characterization of Shy1, the Schizosaccharomyces pombe homolog of human SURF1. Scientific Reports, 2024. 14(1). [CrossRef]
- Jia, C., et al., The opposite role of lactate dehydrogenase a (LDHA) in cervical cancer under energy stress conditions. Free Radical Biology and Medicine, 2024. 214: p. 2-18. [CrossRef]
- Yang, Y., et al., Arenobufagin regulates the p62-Keap1-Nrf2 pathway to induce autophagy-dependent ferroptosis in HepG2 cells. Naunyn-Schmiedeberg's Archives of Pharmacology, 2024. 397(7): p. 4895-4909. [CrossRef]
- Sung, H., et al., Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: A Cancer Journal for Clinicians, 2021. 71(3): p. 209-249. [CrossRef]
- Arneth, B., Tumor Microenvironment. Medicina, 2019. 56(1). [CrossRef]
- Wu, H., et al., The Multifaceted Regulation of Mitochondria in Ferroptosis. Life, 2021. 11(3). [CrossRef]
- Zhang, B., et al., Naringenin Alleviates Renal Ischemia Reperfusion Injury by Suppressing ER Stress-Induced Pyroptosis and Apoptosis through Activating Nrf2/HO-1 Signaling Pathway. Oxidative Medicine and Cellular Longevity, 2022. 2022: p. 1-24. [CrossRef]
- Ding, Y., et al., Identification of a small molecule as inducer of ferroptosis and apoptosis through ubiquitination of GPX4 in triple negative breast cancer cells. Journal of Hematology & Oncology, 2021. 14(1). [CrossRef]
- Lee, Y.-S., et al., BAX-dependent mitochondrial pathway mediates the crosstalk between ferroptosis and apoptosis. Apoptosis, 2020. 25(9-10): p. 625-631. [CrossRef]
- Adjemian, S., et al., Ionizing radiation results in a mixture of cellular outcomes including mitotic catastrophe, senescence, methuosis, and iron-dependent cell death. Cell Death & Disease, 2020. 11(11). [CrossRef]
- Noguchi, M., et al., Autophagy as a modulator of cell death machinery. Cell Death & Disease, 2020. 11(7). [CrossRef]
- Yang M, Chen P, Liu J, Zhu S, Kroemer G, Klionsky DJ, Lotze MT, Zeh HJ, Kang R, Tang D. Clockophagy is a novel selective autophagy process favoring ferroptosis. Sci Adv. 2019 Jul 24;5(7):eaaw2238. [CrossRef]
- Watanabe, R., et al., Autophagy plays a protective role as an anti-oxidant system in human T cells and represents a novel strategy for induction of T-cell apoptosis. European Journal of Immunology, 2014. 44(8): p. 2508-2520. [CrossRef]
- Shibui, Y., et al., MPTP-induced neuroblast apoptosis in the subventricular zone is not regulated by dopamine or other monoamine transporters. NeuroToxicology, 2009. 30(6): p. 1036-1044. [CrossRef]
- Shackelford, D.B. and R.J. Shaw, The LKB1-AMPK pathway: metabolism and growth control in tumour suppression. Nature Reviews Cancer, 2009. 9(8): p. 563-575. [CrossRef]
- Yang, L., et al., FFAR4 improves the senescence of tubular epithelial cells by AMPK/SirT3 signaling in acute kidney injury. Signal Transduction and Targeted Therapy, 2022. 7(1). [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




