Submitted:
13 December 2024
Posted:
14 December 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Minimum Entropy Production Principle (MinEPP)
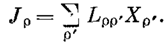
3. MinEPP in an Exactly Solvable Rayleigh Gas Model
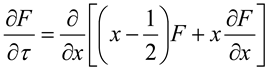
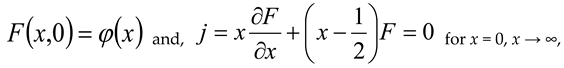
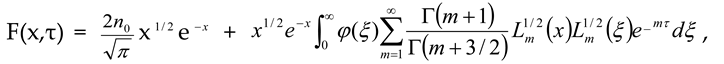
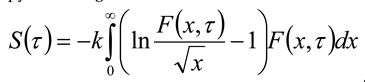
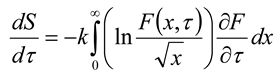
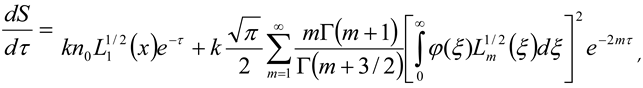
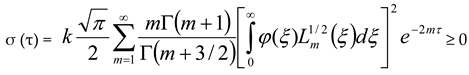
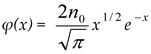 , then σ(τ) = 0.
, then σ(τ) = 0.4. The Principle of Maximum Entropy Production (MEPP)
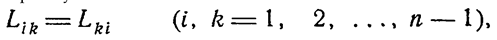
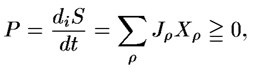
5. MEPP in an Exactly Solvable Model of a Rayleigh Gas with Sources
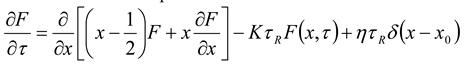
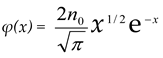 – initial equilibrium distribution function, ε0 – energies of particles δ – source, k – Boltzmann constant, n0 - the initial number of particles, K is the chemical reaction constant and η - is the power of the δ source. For this model, we obtained an analytical solution in the form of an expansion in Laguerre polynomials and showed the entropy balance in [9]. Provided that the number of particles is balanced n = η/K, with the initial equilibrium DF, this solution has the form
– initial equilibrium distribution function, ε0 – energies of particles δ – source, k – Boltzmann constant, n0 - the initial number of particles, K is the chemical reaction constant and η - is the power of the δ source. For this model, we obtained an analytical solution in the form of an expansion in Laguerre polynomials and showed the entropy balance in [9]. Provided that the number of particles is balanced n = η/K, with the initial equilibrium DF, this solution has the form
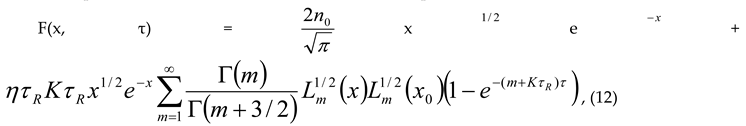
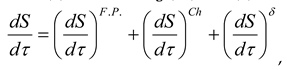
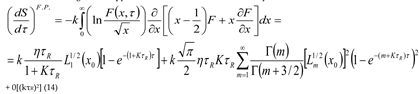
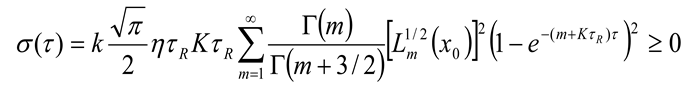
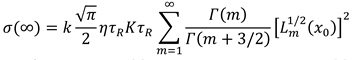
6. Conclusion
- We confirm that neither the manuscript nor any part of its contents are currently under consideration for publication in another journal or are being published in one.
- All authors have approved the manuscript and agree to its submission to Preprits.org.
References
- Prigogine, I. Etude thermodynamique des phenoměněs irreversible: Thesis. ̶ University of Brussels, 1945.
- Prigogine, I. Introduction to thermodynamics of irreversible processes. Springfild, Illinois, U.S.A., 1955.
- Ziegler, H. Some Extremum Principles in Irreversible Thermodynamics, with Application to Continuum Mechanics. Zurich: Swiss Federal Institute of Technology, 1962.
- Ziegler von, H. Zwei Extremalprinzipien der irreversiblen Thermodynamik Ing. – Arch., 30 (1961), 410. [CrossRef]
- Tadjibaev, P. , Tojiboev O. The second law of thermodynamics for open systems. Journal of Engineering Mathematics (2023) 142:9. [CrossRef]
- Osipov, A.I. , Tadzhibaev P.A. Chemical physics of combustion and explosion processes. Kinetics of chemical reactions. // Materials of the All- Union. Symp. On Fire and Explosion. Chernogolovka, 1989, pp. 60-63.
- Delas, N. The principle of maximum entropy production in the evolution of macrosystems: New results. East European Journal of Advanced Technologies. 6/4(72)2014. Kyiv, Ukraine, 03680 (in russian). [CrossRef]
- Martyushev L., M. The principle of maximum entropy production: history of origin and current state, Physics-Uspekhi (Advances in Physical Sciences), 2021, volume 191, number 6, p. 586–613.
- Prigogine, I. Time, Structure, and Fluctuations. Science, 1978, v. 201, No. 4358, pp. 777—785.
- Anderson, K. , Shuler K. E. On the Relaxation of the Hard – Sphere Rayleigh and Lorentz Gas. J. Chem. Phys., 1964, 40, p. 633–650. [Google Scholar]
- Bensah Y.D., Sekhar J.A. Interfacial instability of a planar Interface and diffuseness at the solid-liquid interface for pure and binary materials. arXiv.org; 1605.05005.
- Sánchez-Cañizares, J. Is the maximum entropy production just a heuristic principle. Metaphysics on natural determination. Synthese. (2023) 201:121. [CrossRef]
- Сasimiг H. В., O. On Onsager’s Principle of Microscopic Reversibility. Rev. Mod. Phys., 17, 343 (1945).
- de Groot, S.R. .Thermodynamics of Irreversible Processes, Amsterdam, 1952.
- Onsager, L. Repciprocal Relations in Irreversible Processes. I. Phys. Rev. Febrary 15, 1931,vol. 37, 405 – 426.
- Biot M.A. Variational Principles in Irreversible Thermodynamics with Application to Viscoelasticity. Phys. Rev. March 15, 1955,vol. 97, No. 6, 1463-1469.
- Martyushev, L.M. , Seleznev V.D. Maximum entropy production principle in physics, chemistry and biology. Phys. Rep, v. 426(1), pp. 1-45, 2006.
- Jaynes, E.T. The minimum entropy production principle. Ann. Rev. Phys. Chem. 1980. v. 31. p. 579.
- Endres R.G. Entropy production selects nonequilibrium states in multistable systems. Scientific Reports. 7: 14437 . [CrossRef]
- Evans, D.J. , Williams S.R., Searles D.J., Rondoni L. On the Relaxation to Nonequilibrium Steady Steates. arXiv.org;1602.05808.
- Cocconi L, Garcia-Millan R., Zhen Z., Buturca B., Pruessner G. Entropy Production in Exactly Solvable Systems. Entropy 2020, 22, 1252. [CrossRef]
- Plank, M. Vorlesungen ȕber Thermodynamik. ̶ Lpz.: Teubner, 1930.
- https://www.goodreads.com/author/show/821936.Niels_Bohr.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



