1. Introduction
Microbial exopolysaccharides (EPS), so called for the first time by Sutherland, are high molecular weight polymers secreted by different types of microorganisms, mainly halophiles. Some EPS are associated to the cell membrane (capsular EPS), while other types are secreted into the extracellular environment [
1]. These biopolymers are responsible for the aggregation between cells thanks to the formation of a biofilm that keeps them attached; furthermore, biofilm plays also a role in the interaction between microorganisms by promoting the horizontal transfer of metabolites and genes and assuring survival in conditions with low nutrient availability [
2]. EPS also possess additional biological roles. Lack of water is a stress factor for cells with consequent decrease of membrane fluidity and loss of functions for several macromolecules as proteins. EPS protect from the harmful effects of desiccation most likely through the ability of the hydroxyl groups to retain water [
3]. They also defend cells against the stress caused by high salt concentrations and extreme temperatures [
4].
The chemical composition and structures are very varied. EPS can be homo- or hetero-polysaccharides, branched or unbranched and, in addition, they can have organic or inorganic substituents on the sugar moieties, such as pyruvate, acetate, sulphate or phosphate. Example of microbial homo-polysaccarides are levan, composed of D-fructose, and alternan, consisting in D-glucose units [
5]. Natural hetero-polysaccharides are many, due to the high possibilities of combining the different sugars with each other. Here, two of the best-known microbial hetero-polysaccharides are cited as an example. Hyaluronic acid, mainly used in the cosmetic industry, is made up of units of D-N-acetyl-glucosamine and D-glucuronic acid, and alginate, used in the food industry as gelling ingredient, consists of D-mannuronate/L-guluronate units [
6,
7].
EPS-producing halophilic microorganisms belong to both Bacteria and Archaea domains. Among Bacteria, the most common microorganisms able to synthesize EPS are those belonging to the genus
Halomonas.
H. stenophila produces a sulphated EPS with potential biological properties as it is proved that this type of polysaccharides are inhibitors of several viruses [
8,
9]. Recently, a novel EPS was characterized from
H. smyrnensis K2. It is a hetero-polysaccharide provided with anti-biofilm activity against
Escherichia coli and
Enterococcus faecalis [
10].
In the Archaea domain, extreme halophiles (haloarchaea), so called because they grow optimally in environment characterized by high salt concentrations (2.0-5.2 M NaCl), are able to produce EPS [
11]. The first description regarding the synthesis of EPS by a haloarchaeon dates back to 1988, and the
Haloferax genus is undoubtedly the most studied. Antón et al. reported on the production of a polymer secreted by
Hfx. mediterranei who contributed to a mucous appearance to the colonies [
12]. Other EPS-producing haloarchaea belong to the genera
Halococcus,
Halobacterium,
Natronococcus,
Haloarcula, and
Haloterrigena [
13,
14].
Plants are also a source of polysaccharides, but production by microorganisms can offer several advantages. Extraction of plant polysaccharides is complex and time-consuming, while recovery from a microbial culture medium consists of a rapid precipitation step with alcohol [
15,
16]. In addition, the yields of plant products are often affected by the variability of the growing conditions, whereas once established the growing parameters for microbial EPS production, the yield should not vary. Finally, the high salt amount required for the growth makes extreme halophiles more advantageous to cultivate because non-sterile conditions are allowed.
EPS can have a wide range of industrial applications thanks to properties that make them suitable for uses in cosmetic, pharmaceutical and food sectors. Dextran, xanthan, and gellan are EPS that find application in food and beverages industries for their gelling, thickening and emulsifying capacities [
17]. Tarannum and co-workers recently described the anti-inflammatory, and antimicrobial properties of EPS isolated from some lactic acid bacteria present in bovine milk, thus showing that these natural polymers are endowed with characteristics of considerable interest for applications in medicine, such as wound healing [
18]. EPS isolated from halophiles also exhibit such valuable properties, as that produced by
Bacillus axarquiensison, isolated from sea sediment samples in Porto-Elsokhna beach (Egypt). EPSBa3 showed notable antioxidant power when assessed as DPPH radical scavenger, and was able to reduce the carrageen-induced paw edema in rats [
19].
A further characteristic possessed by several EPS is the ability to bind heavy metals and toxic dyes. The hetero-polysaccharide from the marine bacterium
Alteromonas sp. JL2810 showed high adsorption capacity towards Cu
++ and Cr
6+ at low pH, and the anionic EPS R040 from
Lactobacillus plantarum was highly effective in removing methylene blue (MB) used to dye fabrics [
20,
21]. As the presence of heavy metals and dyes in wastewaters coming from anthropic activities is responsible of ecological and health damages to the environment and humans, the use of microbial EPS for an effective detoxification of polluted waters is gaining increasing consideration.
Haloterrigena turkmenica is an extreme halophilic archaeon assigned to the family of
Natrialbaceae [
22]. It is Gram-negative with a variable shape (from coccoid to rod), and requires a minimum of 2.0 M NaCl for the growth. In a previous paper, it was ascertained the ability of
Htg. turkmenica to produce an anionic hetero-polysaccharide when grown in medium supplemented with glucose. The antioxidant, moisture-absorption/retention, and emulsifying properties of the isolated and characterized EPS were investigated [
23]. In this paper, a further characterization of the EPS is presented. We report on properties of relevant importance in determining possible future applications of the biopolymer. In detail, the heavy metals and textile dyes uptake capacity of the EPS from
Htg. turkmenica, its capability to scavenge the radical cation and the superoxide radical anion generated by ABTS and pyrogallol, respectively, and the anti-inflammatory activity were investigated.
2. Materials and Methods
2.1. Chemicals
Chemicals for preparation of the growth medium, 2,2’-azino-bis(3-ethylbenzthiazoline-6-sulfonic) acid (ABTS), potassium persulphate, pyrogallol, EDTA-Na2, citric acid, bovine serum albumin (BSA), cadmium nitrate tetrahydrate, copper (II) sulfate pentahydrate, lead(II) chloride, methyl orange (MO), methylene blue (MB) monohydrate, and nitric acid (65% v/v) for metal ions analysis were purchased from Merck (Milano, Italy). Ethanol was from Carlo Erba Reagents (Arese, Milano, Italy).
2.2. EPS Production
For EPS production,
Htg. turkmenica (DSM-5511), obtained from Deutsche Sammlung von Mikroorganismen und Zellkulturen (DSMZ) (Braunschweig, Germany), was grown in halobacteria medium M372 (DSMZ catalogue) added with 1 % (w/v) glucose up to 4 liters (batch cultures of 1 liter each). The EPS was recovered from the culture medium, freeze-dried and stored at RT until analyses as previously described [
23].
2.3. Antioxidant Assays
2.3.1. Radical Scavenging Activity (RSA)
RSA was estimated by measuring the discoloration of the ABTS radical in accordance with the method described by Cheng et al. with some modifications [
24]. To generate ABTS radical, ABTS aqueous solution (7mM final concentration) was mixed with potassium persulphate (2.45 mM final concentration). This solution was incubated in the dark for 16 h at RT, and then diluted with 50 mM Tris-HCl pH 7.0 buffer until reaching an absorbance of 1.0 at 734 nm. A volume of EPS aqueous solution (20 mg/mL), opportunely diluted to 0.1 mL and containing 0.2, 0.5, 1.0, 1.5, and 2.0 mg was added to 0.9 mL ABTS radical solution. The absorbance was measured at 734 nm after 10 min against a blank containing 0.1 mL water and 0.9 mL Tris-HCl buffer. A control sample made of 0.1 mL water and 0.9 mL ABTS radical solution was prepared and the absorbance was recorded as reported above. RSA% was calculated according to the following formula:
The results were compared with RSA% obtained with 2 mg of citric acid used as positive control.
2.3.2. Superoxide Scavenging Activity (SSA)
The SSA was evaluated by the pyrogallol autoxidation method as described in Serino et al. with modifications [
25]. A volume of EPS aqueous solution (20 mg/mL) containing 0.2, 0.5, 1.0, 1.5, and 2.0 mg was added to 50 mM Tris-HCl (pH 7.4)/1 mM EDTA-Na
2 buffer solution (final volume 983 μL) in a quartz cuvette. Then, 60 mM pyrogallol in 1 mM HCl (17 μL) was added, and the absorbance was measured at 325 nm after 10 min against a blank containing the buffer solution (1 mL). A control sample made of buffer solution (983 μL) and 60 mM pyrogallol in 1 mM HCl (17 μL) was prepared, and the absorbance was recorded as reported above. SSA% was calculated according to the following formula:
The results were compared with SSA% obtained with 0.2 mg of citric acid used as positive control.
2.4. Anti-Inflammatory Activity
The anti-inflammatory activity was estimated by the BSA denaturation assay [
26]. A volume of EPS aqueous solution (20 mg/mL), opportunely diluted to 0.1 mL and containing 0.02, 0.05, 0.1, 0.2, 0.5, 1.0, and 2.0 mg was added to 1.0 mL of 0.2% BSA in 50 mM Tris-HCl pH 6.5 buffer. Blank containing 1.1 mL Tris-HCl buffer and control containing 1.0 mL of 0.2% BSA in 50 mM Tris-HCl pH 6.5 buffer plus 0.1 mL Tris-HCl buffer were also prepared. All samples were incubated for 15 min at 37 °C (first step), and successively at 72 °C for 5 min (second step). After cooling, the turbidity due to BSA denaturation was recorded at 660 nm. The inhibition of BSA denaturation (ID
BSA%) was calculated according to the following formula:
The results were compared with IDBSA% obtained with 0.2 mg of ibuprofen used as positive control.
2.5. Heavy Metals Uptake
To evaluate the heavy metal uptake ability, dialysis tubes (12.0 kDa MWCO) containing 2.5 mL of EPS aqueous solution (5 g/L) were placed in 100 mL of the following metal solutions: Pb++, Cd++, and Cu++. Each experiment was performed with 10 and 50 mg/L of each metal and pH were adjusted to 5.32, 5.51, and 4.28, respectively to assure the complete dissolution of the salts. Control experiments with distilled water instead of EPS in the dialysis tubes were also run. All preparations were maintained at RT under stirring, and samples for metal analysis were withdrawn at 0 and 24 h. The quantitative determination of Pb++, Cd++, and Cu++ in the aqueous samples was carried out using an Agilent ICP-MS 7500ce mass spectrometer. Before ICP-MS analysis, the samples were diluted, and then acidified until 2% (w/w) nitric acid solution was obtained. The amount of the investigated metals was obtained by an external calibration (standard Periodic table mix 1 for ICP, Merck; product code: 92091).
The adsorption capacity (
q, mg/g EPS) and the metal removal efficiency (MRE%) were calculated as follows:
CF CONTROL
Where V (L) is the volume of the metal solution, CI and CF (mg/L) are the metal concentrations at the beginning of the experiment and at equilibrium, and mEPS (g) is the amount of EPS.
2.6. Dyes Uptake
The dyes uptake ability was assessed using MB and MO. Dialysis tubes (12.0 kDa MWCO) containing 2.5 mL of EPS aqueous solution (5 g/L) were placed in 100 mL of MB or MO solutions (0.10, 0.25 and 0.50 g/L). The pH of the dye solutions was adjusted to 4.78 and 4.45, respectively. Control experiments were performed by using distilled water instead of EPS in the dialysis tubes. All preparations were maintained at RT under stirring. Samples for the estimation of the residual dyes in solution were withdrawn at 0 and 24 h, and the absorbance was measured at 663 and 464 nm, for MB and MO, respectively. The dyes were quantified by calibration curves built with increasing quantities of MB (range 2-6 μg) and MO (range 2-12 μg).
The adsorption capacity and the dye removal efficiency (DRE%) were calculated according to equations 4 and 5.
2.7. Statistical Analysis
All analyses were performed in triplicate and expressed as mean ± standard deviation (SD). Significant statistical differences were determined using analysis of variance (ANOVA) completed by Bonferroni post-test. A p value of < 0.05 was considered to indicate a significant result.
3. Results and Discussion
3.1. EPS Production
For the EPS production,
Htg. turkmenica was grown in M372 medium supplemented with 1% glucose for 96 h. This sugar was selected because provided the highest EPS yield among several sugars tested as carbon source in previous experiments [
23]. The growth was stopped at the stationary phase, and under the selected conditions
Htg. turkmenica produced 217 mg/L EPS.
3.2. Antioxidant Activity
The capability of theEPS to act as radical scavenger was investigated by means of two assays based on the use of radical species: the ABTS radical cation ((ABTS•+) and the superoxide radical anion ((O2•−).
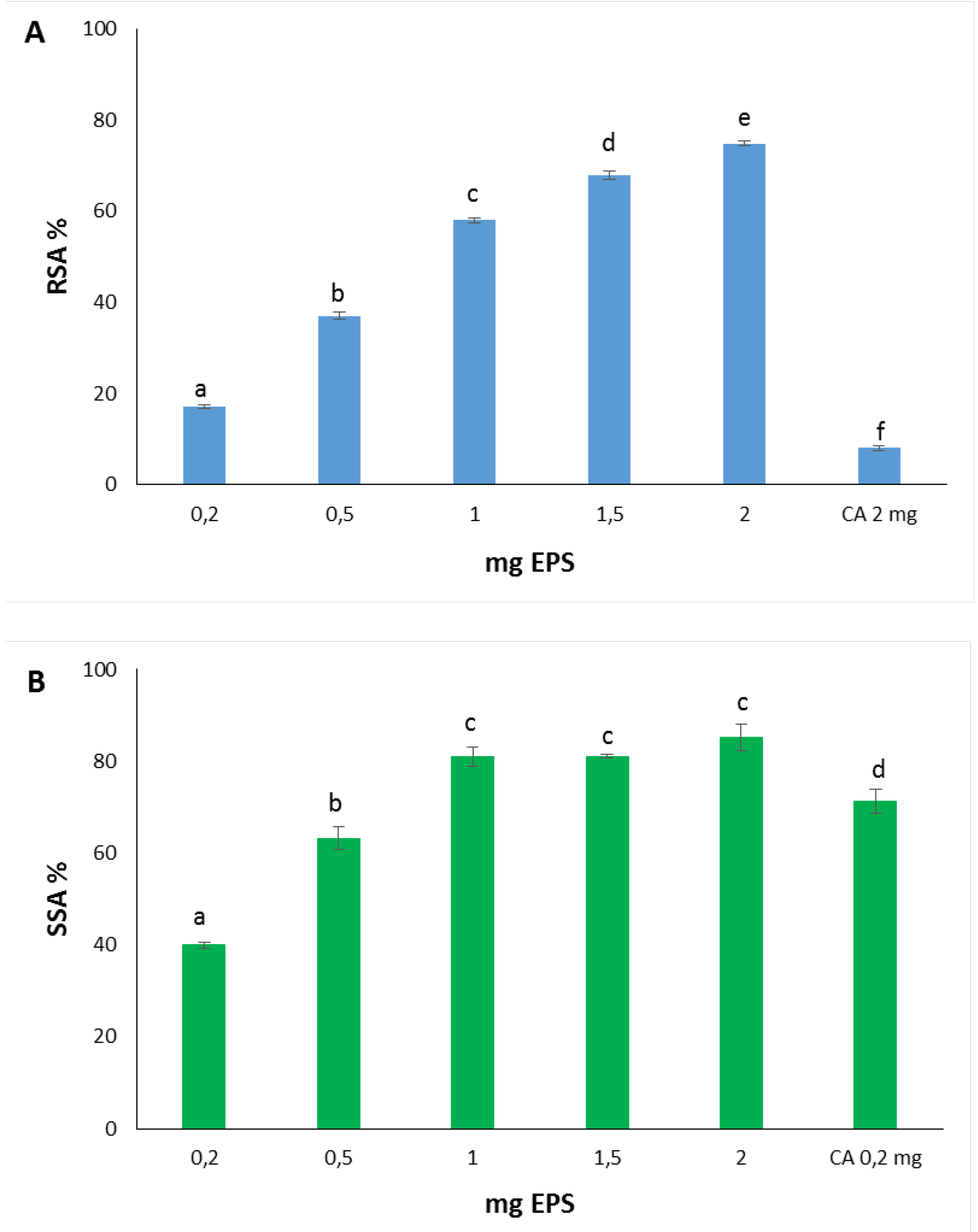
The EPS from
Htg. turkmenica was able to decolorize the ABTS radical cation showing a dose-dependent increase of the antioxidant power (
Figure 1A). The RSA%, measured after 10 min of assay, ranged from 17.16% ± 0.39 to 74.81% ± 0.56 with 0.2 and 2.0 mg EPS, respectively. The IC
50 was 0.82 mg/mL. RSA% differences were highly significant with
p value < 0.0001.
The scavenging capacity was also estimated through an assay based on the inhibition of the pyrogallol autoxidation, which produces the superoxide radical anion in alkaline solution [
27]. EPS was endowed with high scavenging activity showing 40% ± 0.65 SSA at the lowest dose tested (0.2 mg) (
Figure 1B). SSA% increased as concentration increased, reaching 85.28% ± 2.89 with 2.0 mg EPS. The IC
50 was 0.34 mg/mL. It should be noted that the plateau was reached at 1.0 mg EPS (SSA% equal to 81.02% ± 2.15), and this value did not increase considerably by analyzing higher quantities of the polysaccharide (
p > 0.05). Statistically significant differences were measured among SSA% of all EPS doses tested and SSA% of citric acid selected as antioxidant reference.
The antioxidant power of EPS from
Htg. turkmenica was previously assessed by assays based on diverse mechanisms. It showed radical scavenging capacity against the free stable radical 2,2′-diphenyl-1-picrylhydrazyl (DPPH), and also ability to react with Fe
3+ and Mo
6+ ions in a dose-dependent manner. Furthermore, it was able to stabilize emulsions prepared with vegetable oils such as olive oil and sunflower oil [
23]. This remarkable feature, along with the antioxidant power, makes the EPS a potential active ingredient to use as emulsifier and preservative at the same time in food products.
To compare the radical scavenging capacity of the EPS with an antioxidant positive control, citric acid was selected because widely used in food products. This additive, known as E330, acts both as acidity regulator and as antioxidant by enhancing the antioxidant effect of other commonly used antioxidants of synthetic origin whose safety is questioned, such as BHA and BHT [
28]. Citric acid was endowed with SSA% higher than EPS, reaching 71.23% ± 2.56 with 0.2 mg tested, but was much less antioxidant toward ABTS, exhibiting RSA% value of 8.05% ± 0.52 at 2.0 mg dose.
Free radicals are very reactive species produced in the presence of oxidative stress. These molecules are capable of causing damage to cells and are responsible for the onset of various diseases such as diabetes, cardiovascular diseases and aging [
29]. Blocking the propagation chain of free radicals is therefore an important mechanism for stopping or reducing the spread of harmful events and many antioxidant molecules possess this ability. For the above-mentioned reason, in the present work it was decided to further investigate the EPS capability to block the action of radical species, despite many types of antioxidant assays based on other types of action mechanisms exist.
The findings were in agreement with data reported, since several microbial EPS exhibits radical scavenging ability. The EPS from the bacterium
Levilactobacillus brevis NCCP 963, isolated from a traditional Indian drink prepared with fermented purple carrots (Kanji), showed strong ABTS scavenging activity at 0.8 mg/mL (65% RSA with IC
50=0.31 mg/mL), but lesser SSA% than
Htg. turkmenica, with 82.45% at 10 mg/mL and IC
50=6.7 mg/mL [
30]. The EPS produced by the Arctic bacterium
Polaribacter sp. SM1127 exhibited values of 55.40% and 28.2% at 10 mg/mL against DPPH and superoxide radical anion, respectively [
31].
To the best of our knowledge, the EPS produced by Htg. turkmenica is the first exopolysaccharide from extreme halophilic Archaea whose antioxidant power was studied in such detail.
3.3. Anti-Inflammatory Activity
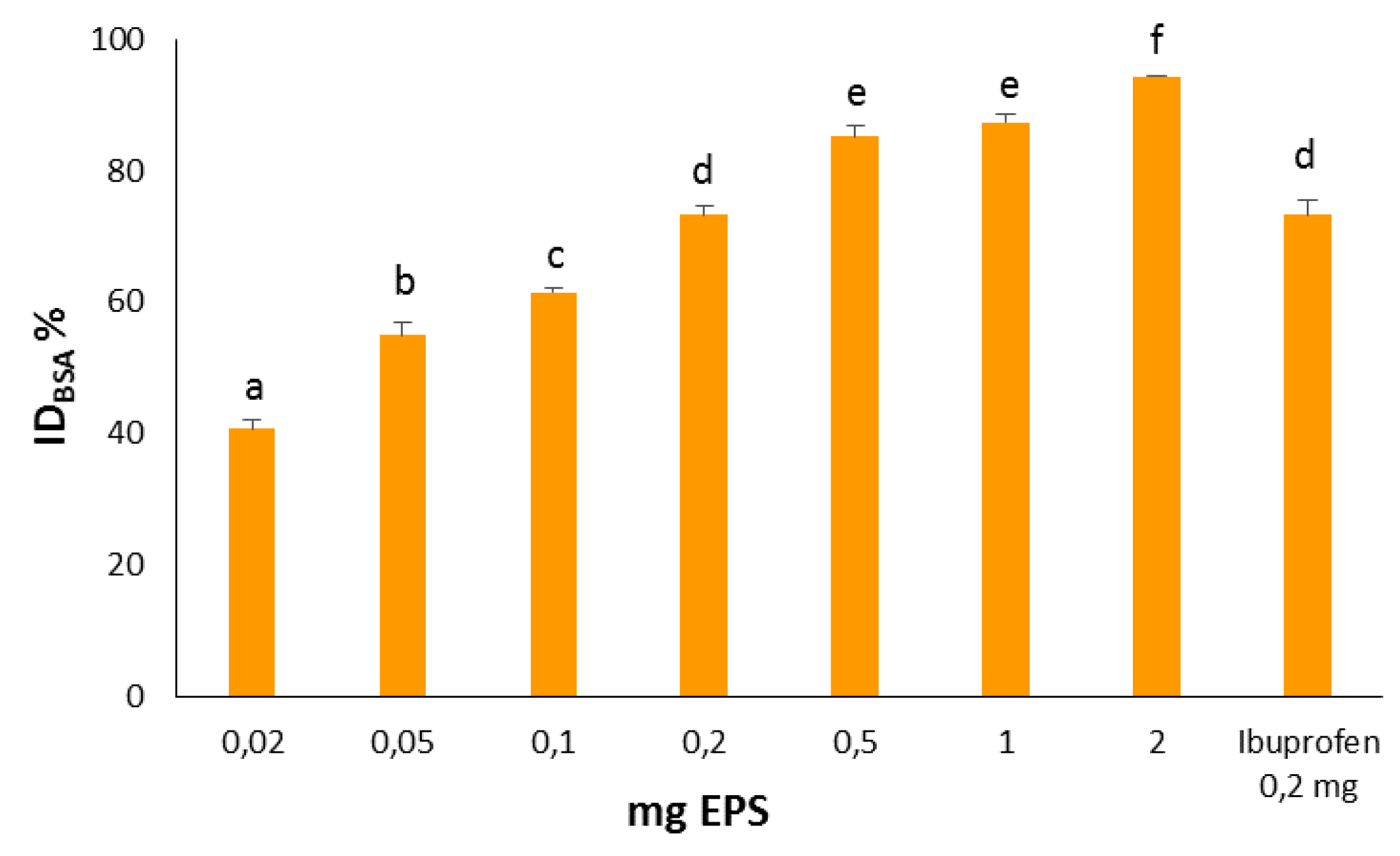
The anti
-inflammatory activity of the EPS was assessed by measuring the rate of inhibition of BSA denaturation. The polysaccharide showed remarkable anti-inflammatory activity with a directly proportional dose-effect ratio. At the lowest dose tested (0.02 mg), the ID
BSA% was 40.71 ± 1.40 and reached 94.26 ± 0.22 with 2.0 mg (
Figure 2). The differences of anti-inflammatory effect measured with the diverse doses of EPS were statistically significant (
p < 0.001) with the exception of 0.5 and 1.0 mg (
p > 0.05). The IC
50 was 0.046 mg/mL. Ibuprofen, a well-known and widely used anti-inflammatory drug, was used as positive control. It showed ID
BSA% of 73.19 ± 2.30 when tested at 0.2 mg. The same effect (ID
BSA% = 73.29% ± 1.30) was obtained with the same dose of EPS, thus confirming its strong anti-inflammatory activity.
The inhibition of BSA denaturation assay was chosen to test the anti-inflammatory effect of the halophilic EPS for its quickness. Furthermore, it is much simpler to set up than assays using cell line models. The assay is based on the observation that well-known non-steroidal drugs with anti-inflammatory action, such as ibuprofen and indomethacin, have the ability to prevent the denaturation of BSA at pathological pH [
32]. When a protein denatures, it loses its three-dimensional structure, generating self-antigens that cause autoimmune reactions such as inflammation. Therefore, molecules capable of inhibiting protein denaturation are potential anti-inflammatory substances [
33].
Anti-inflammatory activity is a characteristic attributed to many EPS. The one synthesized by
Bacillus circulans, a microorganism isolated from slimy layer of coconut, showed 93% ID
BSA at a concentration of 1 mg/mL [
34]. EPS produced by
Cyanobacteria also exhibited notable anti-inflammatory power, and the sulphated EPS isolated from the red seaweed
Spyrida showed 51% ID
BSA at 0.1 mg/mL [
35,
36]. However, the most described EPS in literature derive from lactic acid bacteria (LAB). In a study by Tarannum et al., four strains of LAB, isolated from bovine milk produced EPS with high anti-inflammatory power whose ID
BSA ranged from 80.67% (strain ME2b) to 84% (strain ME7) [
37].
Although it is very likely that EPS from halophiles (including haloarchaea) are also endowed with anti-inflammatory activity, no EPS from such microorganisms have been studied to verify this property to date. Therefore, EPS from Htg. turkmenica is the first EPS derived from an extreme halophile whose anti-inflammatory power has been proven.
3.4. Heavy Metals Uptake
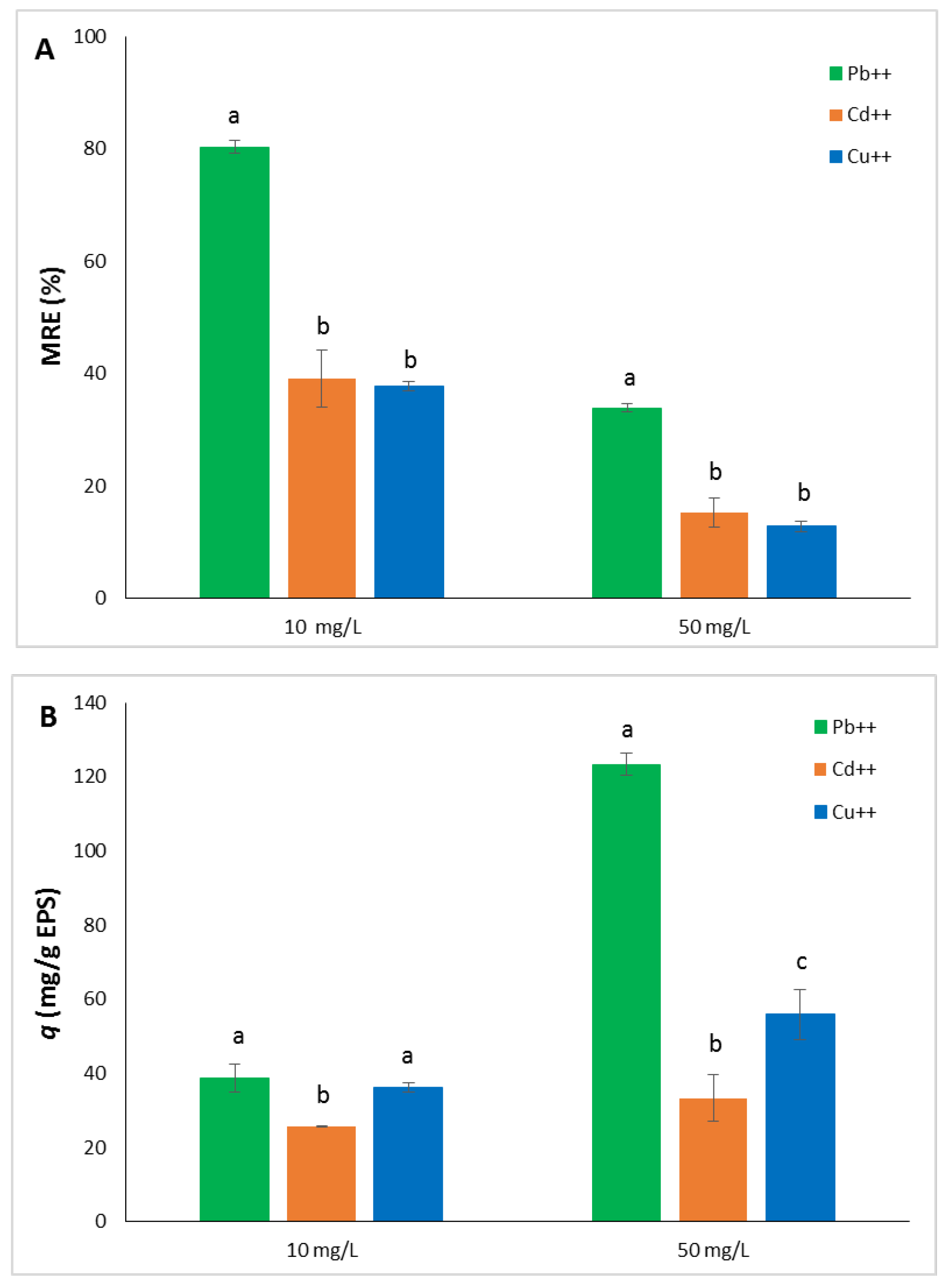
The heavy metals uptake capacity of the EPS from
Htg. turkmenica was evaluated by incubating the exopolysaccharide with Pb
++, Cd
++, and Cu
++ at 10 and 50 mg/L for 24h. These concentrations were selected because it was reported that some of the conventional methods used for heavy metals removal, such as electrochemical method and precipitation, are not effective at concentrations lower than 100 mg/L [
38]. The EPS was able to bind the three heavy metal ions although in different quantities. The MRE% followed the order Pb
++>>Cd
++>Cu
++ for both the concentrations of metals tested (
Figure 3A). MRE% ranged from 37.76 ± 0.75 (Cu
++) to 80.36 ± 1.16 (Pb
++) at 10 mg/L, and from 12.83 ± 0.97 (Cu
++) to 33.9 ± 0.67 (Pb
++) at 50 mg/L. The differences of the metal removal values between Pb
++ and the other two ions were statistically significant at both 10 and 50 mg/L (
p < 0.0001), whereas no significant differences were measured between Cd
++ and Cu
++ (
p > 0.05).
Pb
++ also showed the highest value of adsorption capacity (
q), but the order was reversed between Cd
++ and Cu
++ with respect to the MRE% results. In particular, Pb
++ exhibited
q value much higher than Cd
++ and Cu
++ at 50 mg/L with 123.36 ± 2.91, 33.28 ± 6.34, and 55.89 ± 6.7 mg/g EPS respectively (
Figure 3B). At 10 mg/L ion concentration,
q was quite similar for Pb
++ and Cu
++ with 38.63 ± 3.69 and 36.27 ± 1.2 mg/g EPS, respectively (
p > 0.05), whereas lower adsorption capacity was exhibited by the EPS towards Cd
++ (25.72 ± 0.06).
The adsorption capacity of the metal ions here considered increased as their concentration increased. In particular, q was more than 3 times higher for Pb++ at 50 mg/L in comparison to 10 mg/L. The increase of metal ions amount per g of EPS could determine the saturation of the polysaccharide sites, and this could be the reason for the decrease of MRE% from 10 to 50 mg/L.
The EPS of
Htg. turkmenica has an anionic nature, but the bio-sorption mechanism depends, in addition to the characteristics of the EPS, also on the type of ion. The charge of the metals examined is the same while the ionic radii are Pb
++ 1.12 Å, Cd
++ 0.97 Å, and Cu
++ 0.74 Å. The ionic radius of Pb
++ is the largest and, generally, anionic EPS tend to bind preferably ions with large radii [
39]. Furthermore, the bio-sorption process is usually mediated by different types of interactions between EPS and metal, since there are various ligand species on EPS that can contribute to the binding in different ways. Excluding flocculation, since no precipitates were observed in our experiments, metal sorption could occur thanks to electrostatic interactions, chelation, and complexation, due to the presence on the EPS of carboxylic, hydroxyl, amino (amino sugars) and anionic (sulfate) groups [
40].
Our findings were in accordance with some data published, where the best metal absorption was reached with Pb
++. FucoPol, an exopolysaccharide secreted by the bacterium
Enterobacter A47, removed about 94% Pb
++ from a 10 mg/L solution and showed adsorption capacity of 41.1 mg/g EPS [
41]. Similarly, the EPS from
Bacillus cereus KMS3-1 exhibited the same trend of increasing the absorption capacity of the EPS from
Htg. turkmenica (Cd
++ < Cu
++ < Pb
++ with 54.05, 71.42, and 78.74 mg/g EPS, respectively in 50 mg/L metal solutions) [
42]. Even EPS produced by some halophiles showed the same behavior. The moderately halophilic bacteria
Halomonas nitroreducens WB1 and the strain D, belonging to genus
Halomonas, isolated at a salt site in Iran, secreted EPS that preferentially bound lead over other metals tested. The absorption capacity of the EPS secreted by
H. nitroreducens followed the order Cu
++ < Cd
++ < Pb
++ with
q value for Pb
++ 2.6 times higher than Cd
++. Furthermore, the EPS from strain D removed 39% Pb
++ and 25% Cd
++ from 100 mg/L metal solutions [
43,
44].
Based on the results obtained, it can be stated that the EPS from
Htg. turkmenica has a good capacity to bind Pb
++. This metal is highly toxic and its widespread use has caused massive environmental contamination with consequent damage to health. For this reason, Pb
++ was chosen among the metals to be tested. For the same reason, Cd
++ and Cu
++ were selected. The first is a by-product of the zinc production process, it is contained in rechargeable batteries, and it is the seventh most toxic heavy metal [
45]. The second is a cofactor of many enzymes and an important nutrient for plants. However, its accumulation in the environment and soil, caused by a large use of copper-containing pesticides, determines toxicity for plants and humans [
46,
47].
3.5. Dyes Uptake
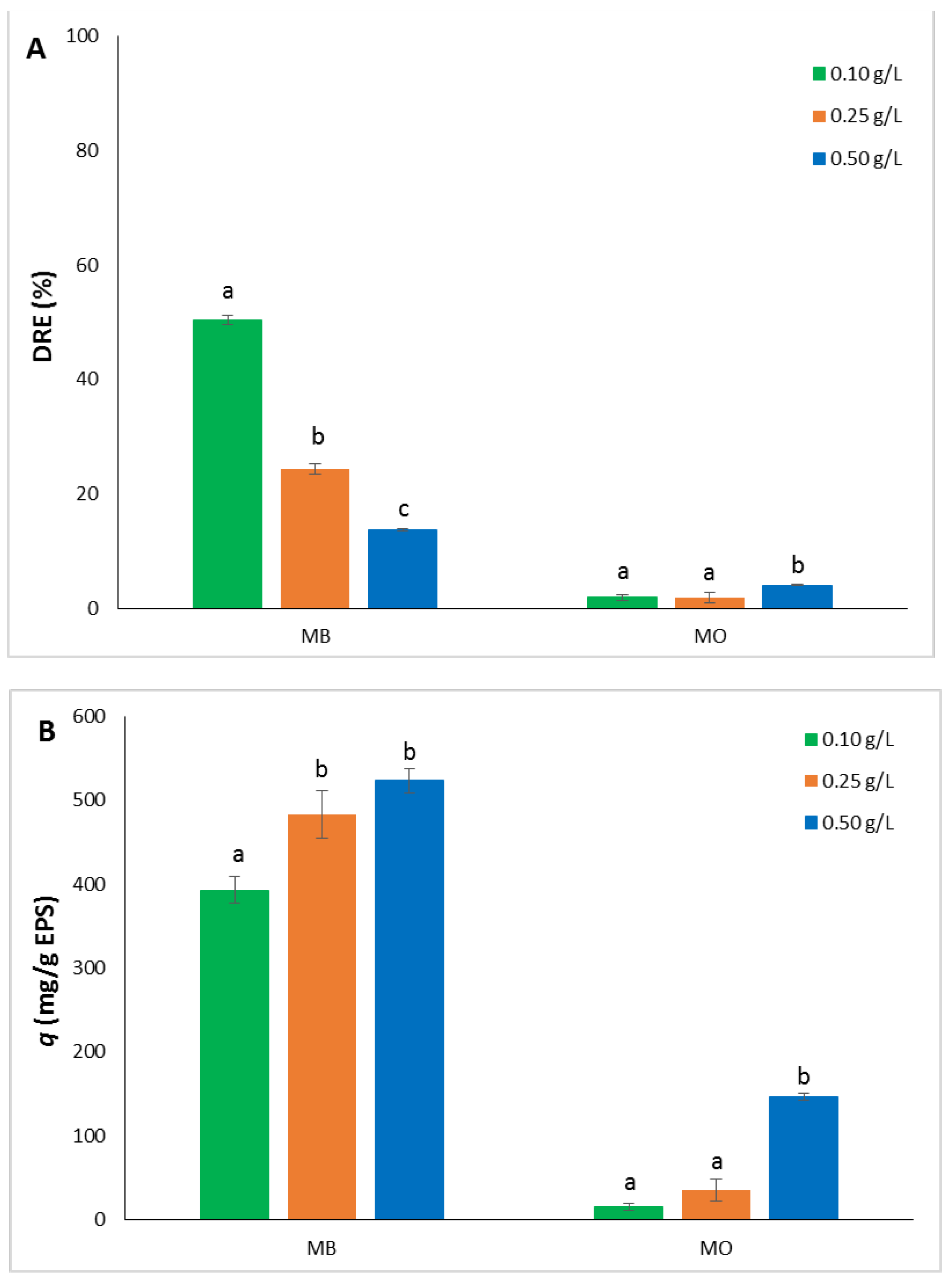
MB and MO at different concentrations were used as model dyes to assess the uptake capacity of the EPS. It showed binding capacity towards MB, although to a different extent in relation to the diverse concentrations analyzed, while the binding efficiency toward MO was very poor. In fact, the highest DRE% after 24h were 50.43 ± 0.77 for MB at 0.10 g/L and 4.08 ± 0.11 for MO at 0.50 g/L (
Figure 4A). The uptake differences between the values measured for MB were highly significant with
p value < 0.0001.
The trend of the adsorption capacity (
q) was opposite, increasing from the lowest to the highest concentration for both the dyes used.
Q values for MB were much higher than those recorded with MO. They ranged from 393.17 ± 15.41 to 523.33 ± 14.14 mg/g EPS for MB, and from 15.57 ± 4.46 to 146.84 ± 3.79 for MO (
Figure 4B). Differences measured between 0.25 and 0.50 g/L for MB and between 0.10 and 0.25 g/L for MO were not statistically significant (
p > 0.05), whereas significant differences were observed in the other cases (
p < 0.006).
The findings demonstrated that the EPS was able to remove MB from the solution, while it was much less effective against MO. The reason could be the different chemical structure of the two dyes used in the tests: MB is a cationic dye while MO has an anionic nature [
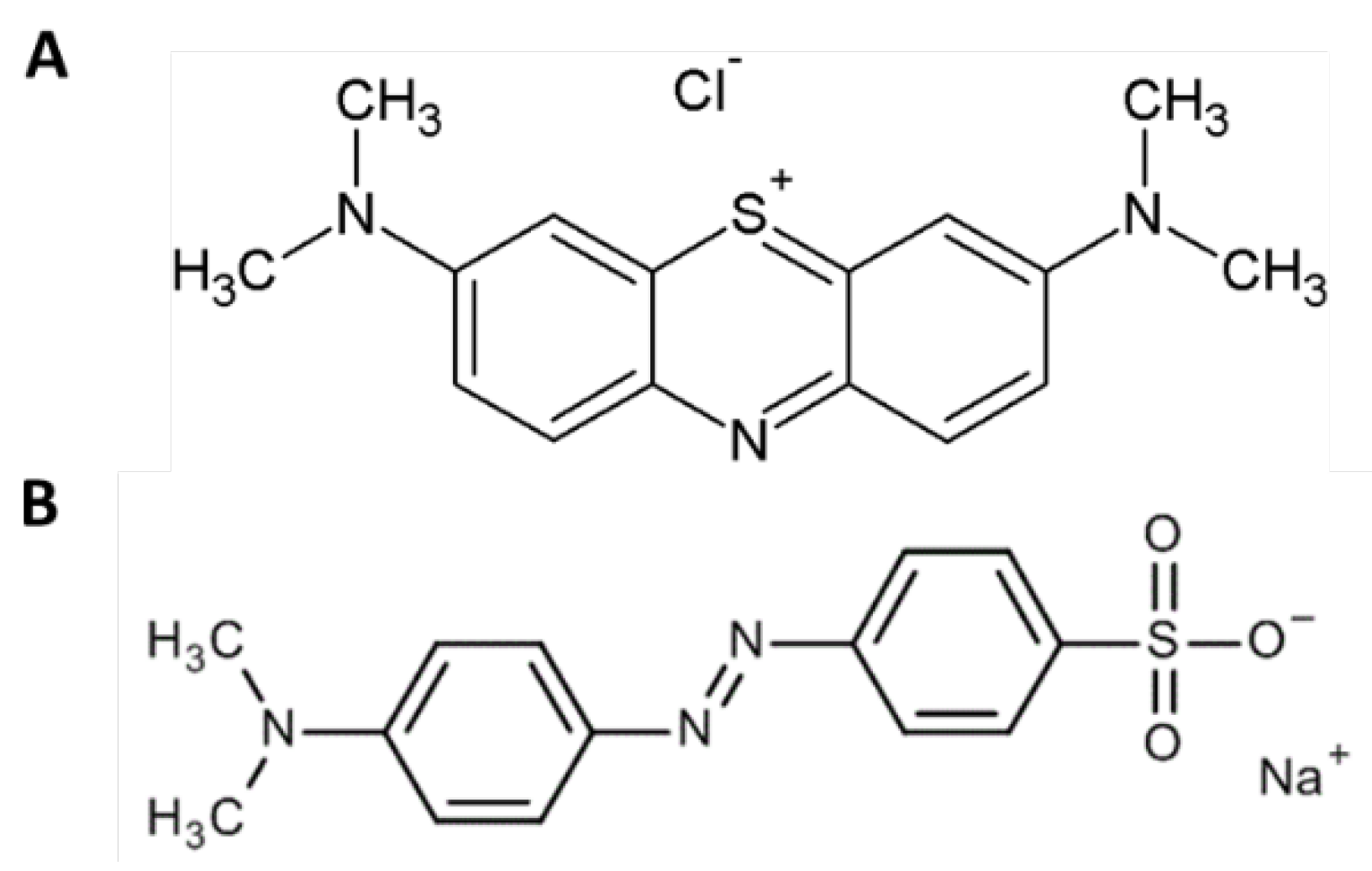
48]. The removal tests were performed at acidic pH, in order to ensure the presence of positive charges on the two structures to favor the interaction with the sulphate groups of the EPS. As it can be observed in
Figure 5, MO has a sulphate group that possesses characteristics of a strong acid. This means that even at low pH values, it is dissociated and therefore, the negative charge of the sulphate group most likely interferes with the EPS, which is also negatively charged, preventing the binding. From the results obtained and the characteristics of the two dyes, it can be assumed that the interaction between the EPS and the dyes was preferably of the electrostatic type.
MB and MO were chosen as model dyes because they are very often found in industrial wastewater generating environmental issues. MB is widely used as a dye for fabrics, it is difficult to degrade in the environment and its accumulation in water is harmful to humans and aquatic fauna. This dye also has a use in medicine as a diagnostic reagent and therapeutic agent; however, it is dangerous in high doses and its toxicity derives from the large quantities found in textile processing effluents. MB can cause respiratory and digestive problems, mental disorders and irritation of eyes and skin [
49]. MO is usually used as coloring agent for fabrics and leather items. This dye is also cause of environmental pollution and harmful effects on exposed organisms because of losses that occur during the dyeing processes and spillage into water. It may cause vomiting and diarrhea and has carcinogenic effects [
50].
Conventional technologies used to remove dyes from wastewater include electrocoagulation, oxidation processes, flocculation and photocatalytic reactions. Unfortunately, these methods are not free from negative effects, such as the production of unwanted products and, in some cases, more than one technique must be used to achieve the desired target [
51]. Alternative methods are represented by the use of absorption materials. Among them, the most used is the activated carbon (AC), whose preparation is expensive. Watermelon shells were subjected to pyrolysis to produce AC for MB removal. The adsorbent showed
q of 303.30 mg/g in a 0.1 g/L MB solution, a value lower than that achieved by the EPS of
Htg. turkmenica at the same dye concentration [
52]. Many other types of adsorbents, derived from agricultural by-products, were tested to remove dyes with the purpose of using cheaper adsorbents. However, the adsorption capacity towards MB was usually lower than the EPS of the present paper [
53].
Microbial EPS are also considered as valuable agents to detoxify waters from harmful dyes, since they are naturally produced. They are considered safe and environmentally friendly detoxifying agents. The EPS from the halophilic bacterium
Exiguobacterium sp.VK1, isolated from a salt pan soil, was able to remove the toxic dye Malachite Green, showing an adsorption capacity of 684.38 mg/g [
54]. MB was efficiently removed by EPS-R040 from
Lactobacillus plantarum.
Q value was comparable to that of the EPS from
Htg. turkmenica at 0.10 g/L MB concentration, but increased to 1200 mg/g at 0.50 g/L [
21]. However, no studies are known on the removal of dyes by EPS from halophilic archaeobacteria up to now. To the best of our knowledge, the EPS from
Htg. turkmenica is the first exopolysaccharide produced by an extreme halophile to be studied for this purpose.
5. Conclusions
The EPS from Htg. turkmenica showed interesting properties for applications in sectors such as cosmetics, food and environment. The remarkable anti-inflammatory power, here attributed for the first time to an EPS produced by a halophile belonging to Archaea domain, together with the emulsifying capacity, antioxidant power, and moisture-absorption/retention ability, make it a valuable active ingredient to use in cosmetic formulations aimed at skin care.
The biopolymer also exhibited good ability to bind Pb++ ions in solution and fair binding capacity towards the MB dye. Therefore, the use of EPS in bio-sorption processes to remove contaminants from water, especially those that are difficult to biodegrade, such as metals and dyes, could be taken into consideration. Furthermore, being an anionic polymer containing sulphate groups, dissociated even at low pH values, the EPS could find use in the decontamination of acid wastewaters containing metals, such as those from batteries manufacturing.
Although EPS with the same properties, produced by other types of microorganisms, were described, the advantage in using EPS from extreme halophiles is due to the ease of cultivation; in fact, their extreme growth conditions, represented by the high salt concentrations in the culture medium, make contamination phenomena impossible. However, in order to be competitive with EPS from mesophilic or moderate halophilic microorganisms, there is a need to increase the production yield and decrease the costs of preparation of synthetic media. These goals can be achieved by using cheap substrates, such as wastewaters rich in sugars and salt, as those from some agro-food industrial processes, and by optimizing the growth parameters. This type of study is currently under investigation together with immobilization tests aimed at enabling the reuse of the EPS during toxic product removal processes.
Author Contributions
Conceptualization, A.M.; methodology, G.S.; validation, A.M.; formal analysis, G.S. and A.M.; investigation, L.B., L.S. and A.C.; data curation, G.S. and L.S.; writing—original draft preparation, G.S. and L.S.; writing—review and editing, A.M.; supervision, A.M.; project administration, G.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
Data are contained within the article.
Acknowledgments
The authors gratefully acknowledge Dr. Eng. Paola Giudicianni, from the Institute of Sciences and Technologies for Sustainable Energy and Mobility of the National Research Council of Italy (STEMS-CNR), Naples, for her contribution to heavy metals analyses.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Sutherland, I.W. Bacterial exopolysaccharides. Adv. Microb. Physiol. 1972, 8, 143–213. [CrossRef]
- Sutherland, I.W. Biofilm exopolysaccharides: A strong and sticky framework. Microbiology 2001, 147(1), 3–9. [CrossRef]
- Knowles, E.J.; Castenholz, R.W. Effect of exogenous extracellular polysaccharides on the desiccation and freezing tolerance of rock-inhabiting phototrophic microorganisms. FEMS Microbiol. Ecol. 2008, 66, 261–270. [CrossRef]
- Netrusov, A.I.; Liyaskina, E.V.; Kurgaeva, I.V.; Liyaskina, A.U.; Yang, G.; Revin, V.V. Exopolysaccharides Producing Bacteria: A Review. Microorganisms 2023, 11(6), 1541. [CrossRef]
- Monsan, P.; Bozonnet, S.; Albenne, C.; Joucla, G.; Willemot,R.M.; Remaud-Siméon, M. Homopolysaccharides from lactic acid bacteria. Int. Dairy J. 2001, 11, 675–685. [CrossRef]
- Juncan, A.M.; Moisă, D.G.; Santini, A.; Morgovan, C.; Rus, L.L.; Vonica-Țincu, A.V.; Loghin, F. Advantages of Hyaluronic Acid and Its Combination with Other Bioactive Ingredients in Cosmeceuticals. Molecules 2021, 26(15), 4429. [CrossRef]
- Sanderson, G.R.; Ortega, D. Alginates and Gellan Gum: Complementary Gelling Agents. In Food Hydrocolloids; Nishinari, K., Doi, E. Eds.; Springer: Boston, MA, 1994; pp. 83-89.
- Llamas, I.; Béjar, V.; Martínez-Checa, F.; Martínez-Cánovas, M.J.; Molina, I.; Quesada, E. Halomonas stenophila sp. nov., a halophilic bacterium that produces sulphate exopolysaccharides with biological activity. Int. J. Syst. Evol. Micr. 2011, 61, 2508–2514. [CrossRef]
- Chaisuwan, W.; Phimolsiripol, Y.; Chaiyaso, T.; Techapun, C.; Leksawasdi, N.; Jantanasakulwong, K.; Rachtanapun, P.; Wangtueai, S.; Sommano, S.R.; You, S.G.; Regenstein, J.M.; Barba, F.J.; Seesuriyachan, P. The Antiviral Activity of Bacterial, Fungal, and Algal Polysaccharides as Bioactive Ingredients: Potential Uses for Enhancing Immune Systems and Preventing Viruses. Front. Nutr. 2021, 8, 2021. [CrossRef]
- Joulak, I.; Finore, I.; Poli, A.; Abid, Y.; Bkhairia, I.; Nicolaus, B.; Di Donato, P.; Dal Poggetto, G; Gharsallaoui, A.; Attia, H.; Azabou, S. Hetero-exopolysaccharide from the extremely halophilic Halomonas smyrnensis K2: Production, characterization and functional properties in vitro. 3 Biotech 2020, 10, 395. [CrossRef]
- Kushner, D.J. Life in high salt and solute concentrations. In Microbial Life in Extreme Environments; Kushner, D.J., Ed.; Academic Press: London, UK, 1978; pp. 317-368.
- Antón, J.; Meseguer, I.; Rodríguez-Valera, F. Production of an Extracellular Polysaccharide by Haloferax mediterranei. Appl. Environ. Microbiol. 1988, 54, 2381–2386. [CrossRef]
- Biswas, J.; Paul, AK. Diversity and production of extracellular polysaccharide by halophilic microorganisms. Biodiversity Int. J. 2017, 1(2), 32‒39. [CrossRef]
- Squillaci, G.; Serino, I.; La Cara, F.; Morana, A. The wonderful halophiles: Microorganisms producing useful compounds. In Extreme Environments: Unique Ecosystems-Amazing Microbes; Pandey, A., Sharma, A., Eds.; CRC Press: Boca Raton, FL, USA, 2021; pp. 1-20.
- Fu, Y.; Li, F.; Ding, Y.; Li, H.Y.; Xiang, X.R.; Ye, Q.; Zhang, J.; Zhao, L.; Qin, W.; Gan, R.Y.; Wu, D.T. Polysaccharides from loquat (Eriobotrya japonica) leaves: Impacts of extraction methods on their physicochemical characteristics and biological activities. Int. J. Biol. Macromol. 2020, 146, 508-517. [CrossRef]
- Sharma, K.; Sharma, N.; Handa, S.; Pathania, S. Purification and characterization of novel exopolysaccharides produced from Lactobacillus paraplantarum KM1 isolated from human milk and its cytotoxicity. J. Genet. Eng. Biotechnol. 2020, 18, 56. [CrossRef]
- Suryawanshi, N.; Naik, S.; Eswari, J.S. Exopolysaccharides and their applications in food processing industries. Food Sci. Appl. Biotechnol. 2022, 5(1), 22-44. [CrossRef]
- Tarannum, N.; Hossain, T.J.; Ali, F.; Das, T.; Dhar, K.; Nafiz, I. H. Antioxidant, antimicrobial and emulsification properties of exopolysaccharides from lactic acid bacteria of bovine milk: Insights from biochemical and genomic analysis. LWT - Food Sci. Technol. 2023, 186, 115263. [CrossRef]
- Ibrahim, G.S.; Mahmoud, M.G.: EL Awady, M.E.; Ahmed, M.M.; Fathy, S.A. Characterization and anti-inflammatory activity effect of exopolysaccharide from Bacillus axarquiensison paw rats carrageenan model. J. Biosci. Appl. Res. 2019, 5(4), 501 -518. [CrossRef]
- Zhang, Z.; Cai, R.; Zhang, W.; Fu, Y.; Jiao, N. A Novel Exopolysaccharide with Metal Adsorption Capacity Produced by a Marine Bacterium Alteromonas sp. JL2810. Mar. Drugs, 2017, 15, 175. [CrossRef]
- Li, C.; Chen, D.; Ding, J.; Shi, Z. A novel hetero-exopolysaccharide for the adsorption of methylene blue from aqueous solutions: Isotherm, kinetic, and mechanism studies. J. Clean. Prod. 2020, 265, 121800. [CrossRef]
- Gupta, R.S.; Naushad, S.; Fabros, R.; Adeolu, M. A phylogenomic reappraisal of family-level divisions within the class Halobacteria: Proposal to divide the order Halobacteriales into the families Halobacteriaceae, Haloarculaceae fam. nov., and Halococcaceae fam. nov., and the order Haloferacales into the families, Haloferacaceae and Halorubraceae fam nov. Antonie Van Leeuwenhoek, 2016, 109, 565–587. [CrossRef]
- Squillaci, G.; Finamore, R.; Diana, P.; Restaino, O.F.; Schiraldi, C.; Arbucci, S.; Ionata, E.; La Cara, F.; Morana, A. Production and properties of an exopolysaccharide synthesized by the extreme halophilic archaeon Haloterrigena turkmenica. Appl. Microbiol. Biotechnol. 2016, 100, 613–623. [CrossRef]
- Cheng, H.; Feng, S.; Jia, X.; Li, Q.; Zhou, Y.; Ding, C. Structural characterization and antioxidant activities of polysaccharides extracted from Epimedium acuminatum. Carbohydr. Polym. 2013, 92, 63-68. [CrossRef]
- Serino, I.; Squillaci, G.; Errichiello, S.; Carbone, V.; Baraldi, L.; La Cara, F.; Morana, A. Antioxidant Capacity of Carotenoid Extracts from the Haloarchaeon Halorhabdus utahensis. Antioxidants 2023, 12, 1840. [CrossRef]
- Hamoudi, M.; Amrouni, D.; Baghiani, A.; Khennouf, S.; Dahamna, S. Antioxidant, Anti-inflammatory, and Analgesic Activities of Alcoholic Extracts of Ephedra nebrodensis From Eastern Algeria. Turk. J. Pharm. Sci. 2021, 18(5), 574-580. [CrossRef]
- Li, X.A. Improved Pyrogallol Autoxidation Method: A Reliable and Cheap Superoxide-Scavenging Assay Suitable for All Antioxidants. J. Agric. Food Chem. 2012, 60, 6418–6424. [CrossRef]
- Silva, M.M.; Cebola Lidon, F. An overview on applications and side effects of antioxidant food additives. Emir. J. Food Agric. 2016, 28(12), 823-832-. [CrossRef]
- Jomova, K.; Raptova, R.; Alomar, S.Y.; Alwasel, S.H.; Nepovimova, E.; Kuca, K.; Valko, M. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: Chronic diseases and aging. Arch. Toxicol. 2023, 97, 2499–2574. [CrossRef]
- Afreen, A.; Ahmed, Z.; Khalid, N. Optimization, fractional characterization, and antioxidant potential of exopolysaccharides from Levilactobacillus brevis NCCP 963 isolated from“kanji”. RSC Adv. 2023, 13, 19725. [CrossRef]
- Sun, M.; Zhao, F.; Shi, M.; Zhang, X.; Zhou, B.; Zhang, Y.; Chen, X. Characterization and Biotechnological Potential Analysis of a New Exopolysaccharide from the Arctic Marine Bacterium Polaribacter sp. SM1127. Sci. Rep. 2015, 5(1). [CrossRef]
- Grant, NH.; Alburn, HE.; Kryzanauska, C. Stabilization of serum albumin by anti-inflammatory drugs. Biochem. Pharmacol. 1970, 19, 715-722. [CrossRef]
- Mouffouk, C.; Hambaba, L.; Haba, H.; Mouffouk, S.; Bensouici, C.; Hachemi, M.; Khadraoui, H. Acute toxicity and in vivo anti-inflammatory effects and in vitro antioxidant and anti-arthritic potential of Scabiosa Stellata. Orient. Pharm. Exp. Med. 2018, 18, 335-348. [CrossRef]
- Vidhyalakshmi, R.; Valli Nachiyar, C.; Narendra Kumar, G., Sunkar, S. Bacillus circulans exopolysaccharide: Production, characterization and bioactivities. Int. J. Biol. Macromol. 2016, 87, 405-414. [CrossRef]
- Manoharan, D.; Natesan, S.; Billamboz, M.; Jawhara, S. Role of Bacteria-Derived Exopolysaccharides in Inflammatory Bowel Disease with a Special Focus on Cyanobacterial Exopolysaccharides. Appl. Microbiol. 2024, 4, 250–274. [CrossRef]
- Reddy, S.; Suresh, V.; Pitchiah, S.; Subramanian, B. Anti-inflammatory Activities of Sulfated Polysaccharides From Ethanol Crude Extract of Spyrida Species Red Seaweed. Cureus 2023, 15(12), e50284. [CrossRef]
- Tarannum, N.; Hossain, T.J.; Ali, F.; Das, T.; Dhar, K.; Nafiz, I.H. Antioxidant, antimicrobial and emulsification properties of exopolysaccharides from lactic acid bacteria of bovine milk: Insights from biochemical and genomic analysis. LWT - Food Sci. Technol. 2023, 186, 115263. [CrossRef]
- Wang, J.; Chen, C. Biosorbents for heavy metals removal and their future. Biotechnol. Adv. 2009, 27(2), 195-226. [CrossRef]
- Loaëc, M.; Olier, R.; Guezennec, J. Chelating properties of bacterial exopolysaccharides from deep-sea hydrothermal vents. Carbohydr. Polym. 1998, 35(1-2), 65-70. [CrossRef]
- Ghosh, A.; Sah, D.; Chakraborty, M.; Rai, J.P.N. Mechanism and application of bacterial exopolysaccharides: An advanced approach for sustainable heavy metal abolition from soil. Carbohydr. Res. 2024, 544, 109247. [CrossRef]
- Concórdio-Reis, P.; Reis, M.A.M.; Freitas, F. Biosorption of Heavy Metals by the Bacterial Exopolysaccharide FucoPol. Appl. Sci. 2020, 10(19), 6708. [CrossRef]
- Mathivanan, K.; Chandirika, J.U.; Mathimani, T.; Rajaram, R.; Annadurai, G.; Yin, H. Production and functionality of exopolysaccharides in bacteria exposed to a toxic metal environment. Ecotoxicol. Environ. Saf. 2021, 208, 111567. [CrossRef]
- Chikkanna, A.; Ghosh , D.; Kishore, A. Expression and characterization of a potential exopolysaccharide from a newly isolated halophilic thermotolerant bacteria Halomonas nitroreducens strain WB1. Peer J. 2018, 6, e4684. [CrossRef]
- Amoozegar, M. A.; Ghazanfari, N.; Didari, M. Lead and Cadmium Bioremoval by Halomonas sp., an Exopolysaccharide-Producing Halophilic Bacterium. Prog. Biol. Sci. 2012, 2, 1-11. [CrossRef]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, mechanism and health effects of some heavy metals. Interdiscip. Toxicol. 2014, 7(2), 60–72. [CrossRef]
- Kuehne, S.; Rossberg, D.; Röhrig, P.; Mehring, F.V.; Weihrauch, F.; Kanthak, S.; Kienzle, J.; Patzwahl, W.; Reiners, E.; Gitzel, J. The Use of Copper Pesticides in Germany and the Search for Minimization and Replacement Strategies. Org. Farm. 2017, 3, 66-75. [CrossRef]
- Royer, A.; Sharman, T. Copper Toxicity. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2024.
- Show, S.; Akhter, R.; Paul, I.; Das, P.; Bal, M.; Bhattacharya, R.; Bose, D.; Mondal, A.; Saha, S.; Halder, G. Efficacy of exopolysaccharide in dye-laden wastewater treatment: A comprehensive review. Chemosphere 2024, 355, 141753. [CrossRef]
- Khan, I.; Saeed, K.; Zekker, I.; Zhang, B.; Hendi, A.H.; Ahmad, A.; Ahmad, S.; Zada, N.; Ahmad, H.; Shah, L.A.; Shah, T.; Khan. I. Review on Methylene Blue: Its Properties, Uses, Toxicity and Photodegradation. Water 2022, 14, 242. [CrossRef]
- Hanafi, M.F.; Sapawe, N. A review on the water problem associate with organic pollutants derived from phenol, methyl orange, and remazol brilliant blue dyes. Materials Today: Proceedings 2020, 31, A141-A150. [CrossRef]
- El-gaayda, J.; Ezzahra, F.; Oukhrib, R.; Yap, P.; Liu, T. Natural flocculants for the treatment of wastewaters containing dyes or heavy metals : A state-of-the-art review. J. Environ. Chem. Eng. 2021, 9, 106060. [CrossRef]
- Rajendran, J.; Panneerselvam, A.; Ramasamy, S.; Palanisamy, P. Methylene blue and methyl orange removal from wastewater by magnetic adsorbent based on activated carbon synthesised from watermelon shell. Desalin. Water Treat. 2024, 317, 100040. [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid Interface Sci. 2014, 209, 172-184. [CrossRef]
- Kalpana, R.; Maheshwaran, M.; Vimali, E.; Soosai, M.R.; Shivamathi, C.S.; Moorthy, I.G.; Ashokkumar, B.; Varalakshmi, P. Decolorization of Textile Dye by Halophilic Exiguobacterium sp.VK1: Biomass and Exopolysaccharide (EPS) Enhancement for Bioremediation of Malachite Green. ChemistrySelect 2020, 5(28), 8787-8797. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).