Submitted:
10 December 2024
Posted:
12 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Material and Methods
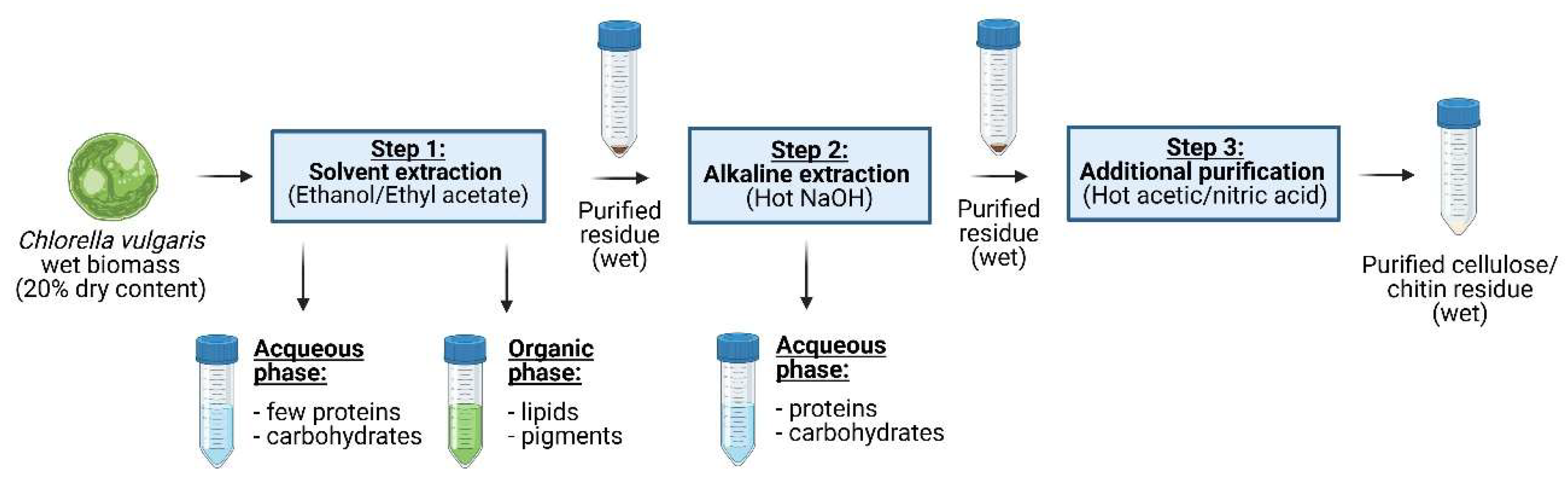
2.1. Microalgal Biomass
2.4. Biopolymer Purification
2.5. Yield of Co-Products
2.5.1. Pigments
2.5.2. Lipids
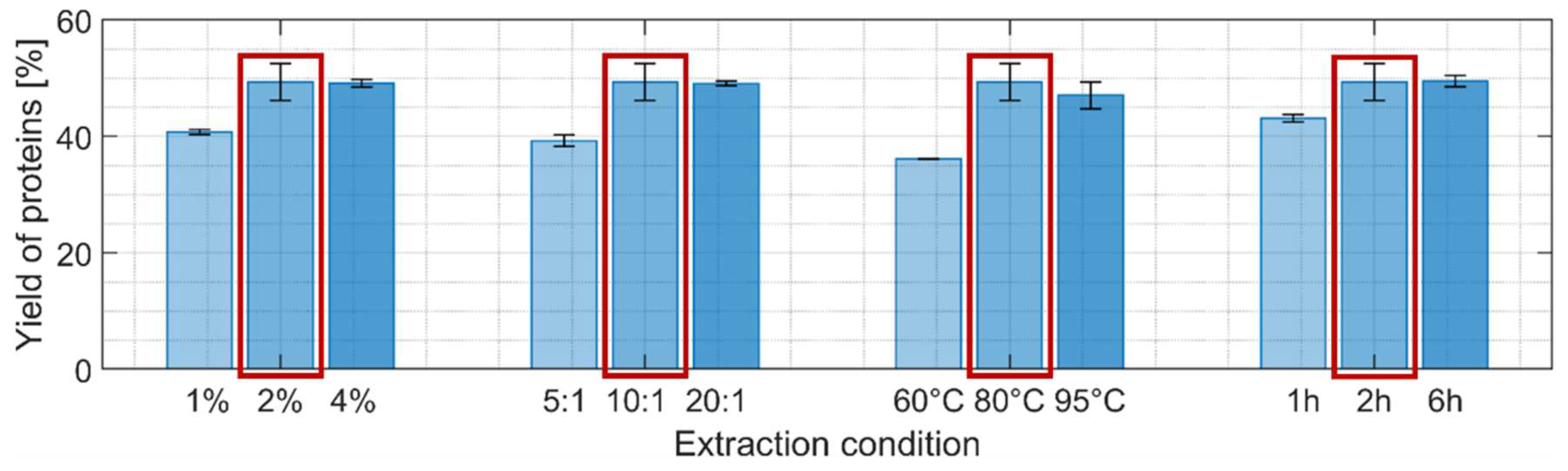
2.5.3. Proteins
2.6. Yield of the Residues
2.7. Scanning Electron Microscopy
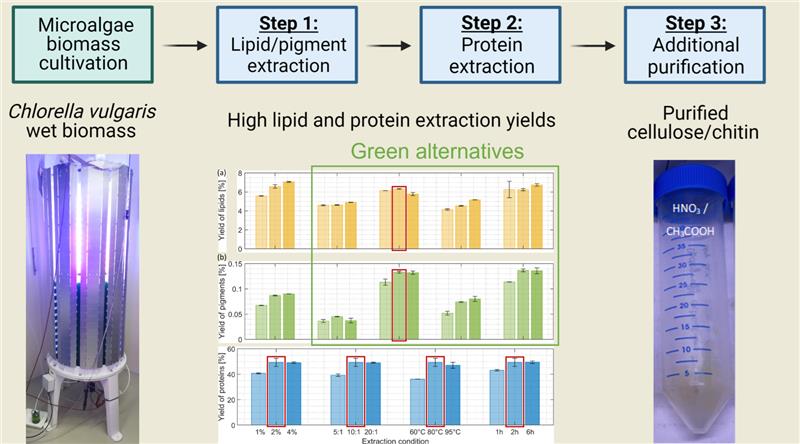
3. Results and Discussion
3.1. Experimental Design of the Biorefinery Process
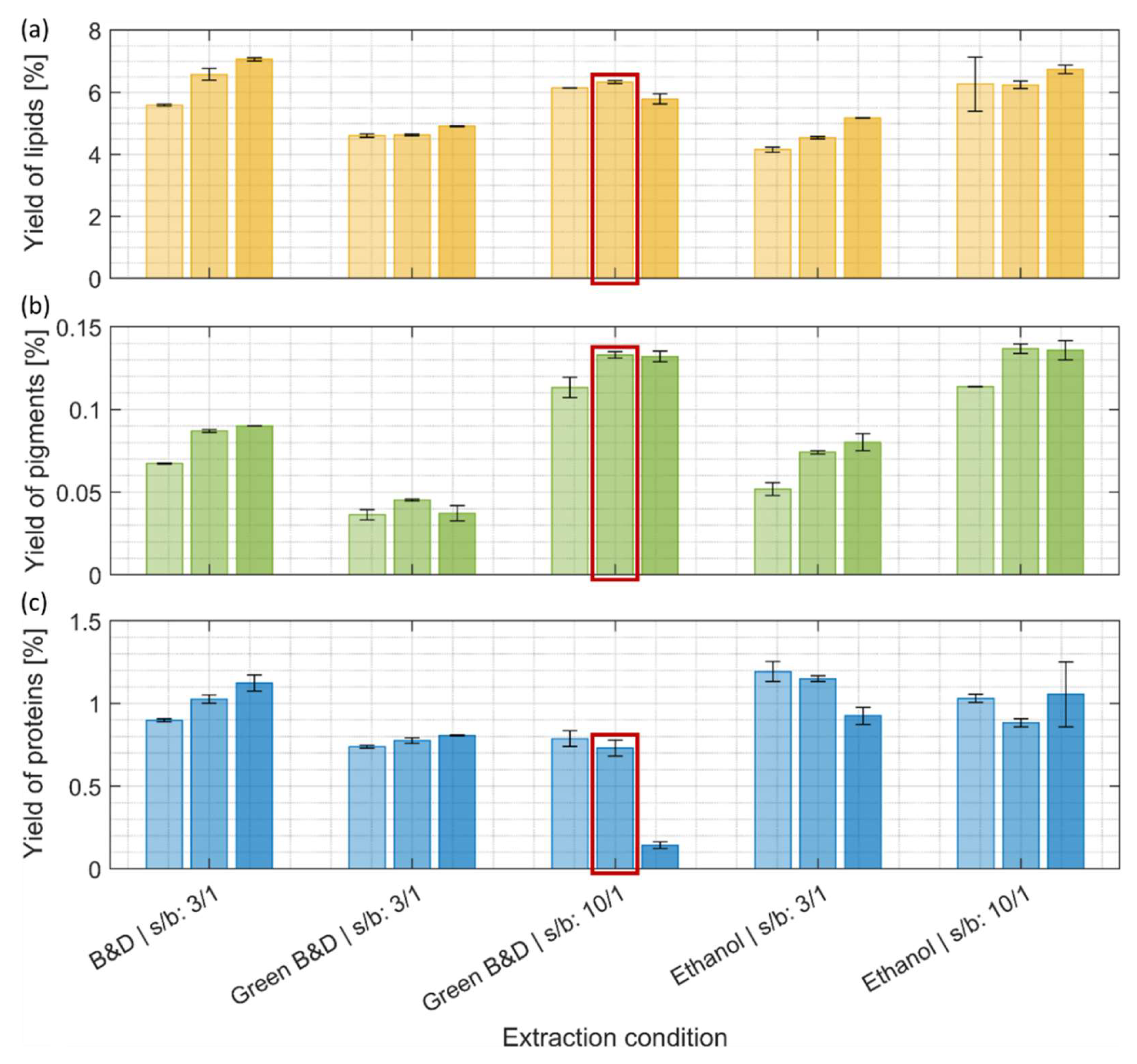
3.2. Yield of Co-Products
3.2.1. Lipids
3.2.2. Pigments
3.2.3. Proteins
3.3. Overall Yields and Purity of the Residue
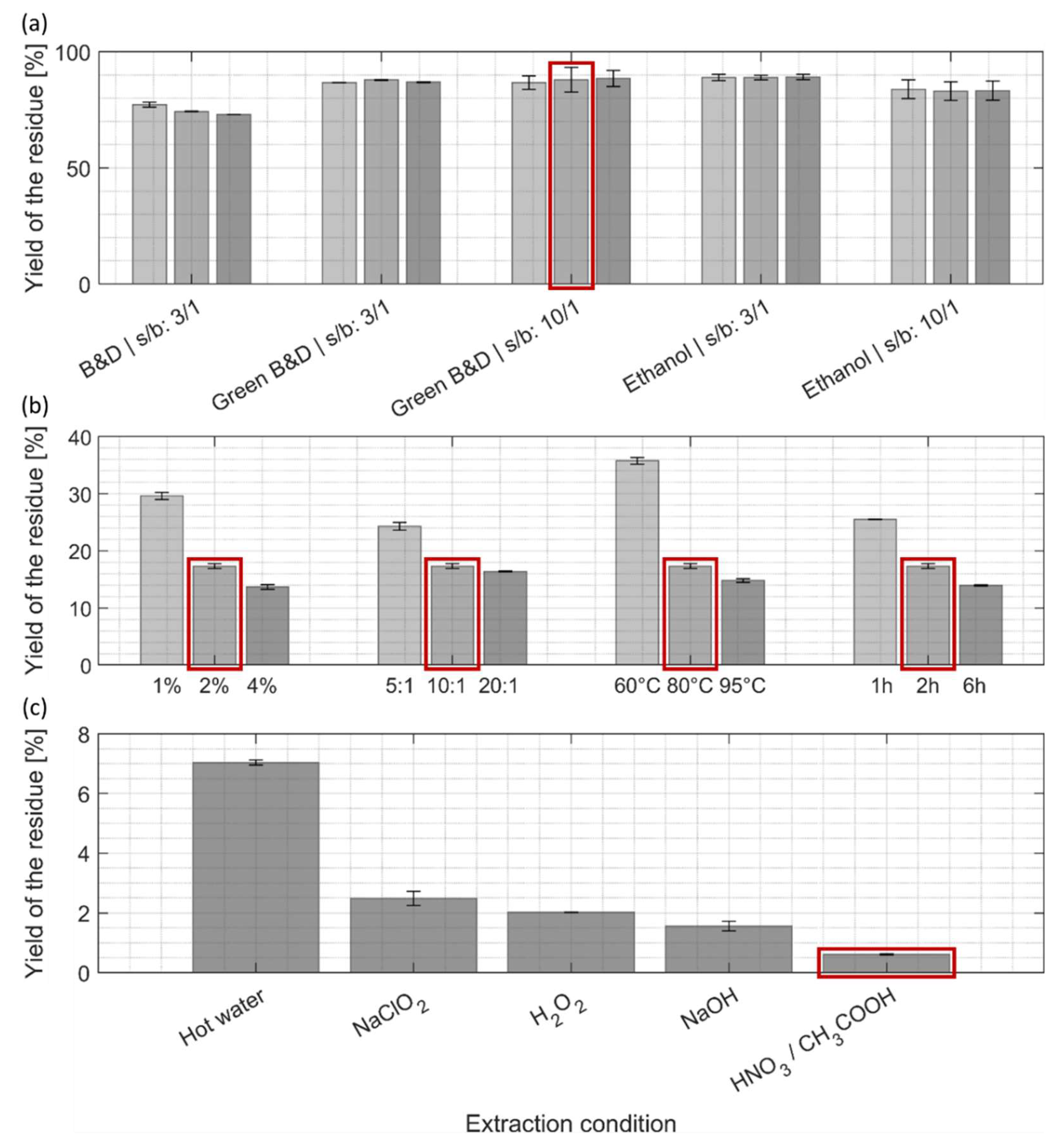
3.3.1. Yield of the Residue After Each Extraction Step
3.4.2. Purity of the Residue Containing Cellulose and Chitin Biopolymers
4. Conclusion
Acknowledgments
Appendix A
References
- Zanchetta, E.; Damergi, E.; Patel, B.; Borgmeyer, T.; Pick, H.; Pulgarin, A.; Ludwig, C. Algal Cellulose, Production and Potential Use in Plastics: Challenges and Opportunities. Algal Res 2021, 56, 102288. [Google Scholar] [CrossRef]
- Serventi, L.; He, Q.; Huang, J.; Mani, A.; Subhash, A.J. Advances in the Preparations and Applications of Nanochitins. Food Hydrocolloids for Health 2021, 1, 100036. [Google Scholar] [CrossRef]
- Trache, D.; Tarchoun, A.F.; Derradji, M.; Hamidon, T.S.; Masruchin, N.; Brosse, N.; Hussin, M.H. Nanocellulose: From Fundamentals to Advanced Applications. Front Chem 2020, 8, 392. [Google Scholar] [CrossRef]
- Isogai, A. Wood Nanocelluloses: Fundamentals and Applications as New Bio-Based Nanomaterials. Journal of Wood Science 2013, 59, 449–459. [Google Scholar] [CrossRef]
- Lawrence, D.; Coe, M.; Walker, W.; Verchot, L.; Vandecar, K. The Unseen Effects of Deforestation: Biophysical Effects on Climate. Frontiers in Forests and Global Change 2022, 5, 49. [Google Scholar] [CrossRef]
- Hamed, I.; Özogul, F.; Regenstein, J.M. Industrial Applications of Crustacean By-Products (Chitin, Chitosan, and Chitooligosaccharides): A Review. Trends Food Sci Technol 2016, 48, 40–50. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications. Mar Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef] [PubMed]
- Kapaun, E.; Reisser, W. A Chitin-like Glycan in the Cell Wall of a Chlorella Sp. (Chlorococcales, Chlorophyceae). Planta 1995, 197, 577–582. [Google Scholar] [CrossRef]
- Burczyk, J.; Śmietana, B.; Termińska-Pabis, K.; Zych, M.; Kowalowski, P. Comparison of Nitrogen Content Amino Acid Composition and Glucosamine Content of Cell Walls of Various Chlorococcalean Algae. Phytochemistry 1999, 51, 491–497. [Google Scholar] [CrossRef]
- Durkin, C.A.; Mock, T.; Armbrust, E.V. Chitin in Diatoms and Its Association with the Cell Wall. Eukaryot Cell 2009, 8, 1038–1050. [Google Scholar] [CrossRef] [PubMed]
- Latil de Ros, D.G.; Lòpez Cerro, M.T.; Ruiz Canovas, E.; Durany Turk, O.; Segura de Yebra, J.; Mercadé Roca, J. Chitin and Chitosan Producing Methods 2016.
- Adamczyk, M.; Lasek, J.; Skawińska, A. CO2 Biofixation and Growth Kinetics of Chlorella Vulgaris and Nannochloropsis Gaditana. Appl Biochem Biotechnol 2016, 179, 1248–1261. [Google Scholar] [CrossRef]
- Koyande, A.K.; Chew, K.W.; Rambabu, K.; Tao, Y.; Chu, D.T.; Show, P.L. Microalgae: A Potential Alternative to Health Supplementation for Humans. Food Science and Human Wellness 2019, 8, 16–24. [Google Scholar] [CrossRef]
- Duval, J.F.L.; Razafitianamaharavo, A.; Bihannic, I.; Offroy, M.; Lesniewska, N.; Sohm, B.; Le Cordier, H.; Mustin, C.; Pagnout, C.; Beaussart, A. New Insights into the Effects of Growth Phase and Enzymatic Treatment on the Cell-Wall Properties of Chlorella Vulgaris Microalgae. Algal Res 2023, 69. [Google Scholar] [CrossRef]
- Zanchetta, E.; Ollivier, M.; Taing, N.; Damergi, E.; Agarwal, A.; Ludwig, C.; Pick, H. Abiotic Stress Approaches for Enhancing Cellulose and Chitin Production in Chlorella Vulgaris. Bioresour Technol In press. 2024. [Google Scholar]
- Bligh, E.G.; Dyer, W.J. A Rapid Method of Total Lipid Extraction and Purification. Can J Biochem Physiol 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Breil, C.; Abert Vian, M.; Zemb, T.; Kunz, W.; Chemat, F. “Bligh and Dyer” and Folch Methods for Solid–Liquid–Liquid Extraction of Lipids from Microorganisms. Comprehension of Solvatation Mechanisms and towards Substitution with Alternative Solvents. Int J Mol Sci 2017, 18, 708. [Google Scholar] [CrossRef]
- Lee, H.-R.; Kim, K.; Mun, S.C.; Chang, Y.K.; Choi, S.Q. A New Method to Produce Cellulose Nanofibrils from Microalgae and the Measurement of Their Mechanical Strength. Carbohydr Polym 2018, 180, 276–285. [Google Scholar] [CrossRef]
- Updegraff, D.M. Semimicro Determination of Cellulose in Biological Materials. Anal Biochem 1969, 32, 420–424. [Google Scholar] [CrossRef]
- Wellburn, A.R. The Spectral Determination of Chlorophylls a and b, as Well as Total Carotenoids, Using Various Solvents with Spectrophotometers of Different Resolution. J Plant Physiol 1994, 144, 307–313. [Google Scholar] [CrossRef]
- Kumar, P.; Ramakritinan, C.M.; Kumaraguru, A.K. Solvent Extraction and Spectrophotometric Determination of Pigments of Some Algal Species from the Shore of Puthumadam, Southeast Coast of India. International Journal of Oceans and Oceanography 2010, 4, 29–34. [Google Scholar]
- de Jesus, S.S.; Ferreira, G.F.; Moreira, L.S.; Maciel, M.R.W.; Maciel Filho, R. Comparison of Several Methods for Effective Lipid Extraction from Wet Microalgae Using Green Solvents. Renew Energy 2019, 143, 130–141. [Google Scholar] [CrossRef]
- Saini, R.K.; Prasad, P.; Shang, X.; Keum, Y.S. Advances in Lipid Extraction Methods—a Review. Int J Mol Sci 2021, 22, 13643. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wang, M.; Saraiva, J.A.; Martins, A.P.; Pinto, C.A.; Prieto, M.A.; Simal-Gandara, J.; Cao, H.; Xiao, J.; Barba, F.J. Extraction of Lipids from Microalgae Using Classical and Innovative Approaches. Food Chem 2022, 384, 132236. [Google Scholar] [CrossRef] [PubMed]
- Loyao, A.S.; Villasica, S.L.G.; Dela Peña, P.L.L.; Go, A.W. Extraction of Lipids from Spent Coffee Grounds with Non-Polar Renewable Solvents as Alternative. Ind Crops Prod 2018, 119, 152–161. [Google Scholar] [CrossRef]
- Yang, F.; Xiang, W.; Sun, X.; Wu, H.; Li, T.; Long, L. A Novel Lipid Extraction Method from Wet Microalga Picochlorum Sp. at Room Temperature. Mar Drugs 2014, 12, 1258–1270. [Google Scholar] [CrossRef]
- Ramalakshmi, K.; Raghavan, B. Caffeine in Coffee: Its Removal. Why and How? Crit Rev Food Sci Nutr 1999, 39, 441–456. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Wang, Z.; Yuan, Z. Characteristics of Lipid Extraction from Chlorella Sp. Cultivated in Outdoor Raceway Ponds with Mixture of Ethyl Acetate and Ethanol for Biodiesel Production. Bioresour Technol 2015, 191, 433–437. [Google Scholar] [CrossRef]
- Sarkar, S.; Manna, M.S.; Bhowmick, T.K.; Gayen, K. Extraction of Chlorophylls and Carotenoids from Dry and Wet Biomass of Isolated Chlorella Thermophila: Optimization of Process Parameters and Modelling by Artificial Neural Network. Process Biochemistry 2020, 96, 58–72. [Google Scholar] [CrossRef]
- Wood, L.W. Chloroform–Methanol Extraction of Chlorophyll a. Canadian journal of fisheries and aquatic sciences 1985, 42, 38–43. [Google Scholar] [CrossRef]
- Sun, H.; Wang, Y.; He, Y.; Liu, B.; Mou, H.; Chen, F.; Yang, S. Microalgae-Derived Pigments for the Food Industry. Mar Drugs 2023, 21, 82. [Google Scholar] [CrossRef]
- Becker, E.W. Micro-Algae as a Source of Protein. Biotechnol Adv 2007, 25, 207–210. [Google Scholar] [CrossRef]
- Wang, Y.; Tibbetts, S.M.; McGinn, P.J. Microalgae as Sources of High-Quality Protein for Human Food and Protein Supplements. Foods 2021, 10, 3002. [Google Scholar] [CrossRef]
- Bleakley, S.; Hayes, M. Algal Proteins: Extraction, Application, and Challenges Concerning Production. Foods 2017, 6, 33. [Google Scholar] [CrossRef]
- Phongthai, S.; Lim, S.T.; Rawdkuen, S. Ultrasonic-Assisted Extraction of Rice Bran Protein Using Response Surface Methodology. J Food Biochem 2017, 41, e12314. [Google Scholar] [CrossRef]
- Xie, T.; Xia, Y.; Zeng, Y.; Li, X.; Zhang, Y. Nitrate Concentration-Shift Cultivation to Enhance Protein Content of Heterotrophic Microalga Chlorella Vulgaris: Over-Compensation Strategy. Bioresour Technol 2017, 233, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Sun, J.; Fa, Y.; Liu, X.; Lindblad, P. Enhancing Microalgal Lipid Accumulation for Biofuel Production. Front Microbiol 2022, 13, 1024441. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Sun, B.; She, X.; Zhao, F.; Cao, Y.; Ren, D.; Lu, J. Lipid Production and Composition of Fatty Acids in Chlorella Vulgaris Cultured Using Different Methods: Photoautotrophic, Heterotrophic, and Pure and Mixed Conditions. Ann Microbiol 2014, 64, 1239–1246. [Google Scholar] [CrossRef]
- Markou, G.; Angelidaki, I.; Georgakakis, D. Microalgal Carbohydrates: An Overview of the Factors Influencing Carbohydrates Production, and of Main Bioconversion Technologies for Production of Biofuels. Appl Microbiol Biotechnol 2012, 96, 631–645. [Google Scholar] [CrossRef] [PubMed]
- Weber, S.; Grande, P.M.; Blank, L.M.; Klose, H. Insights into Cell Wall Disintegration of Chlorella Vulgaris. PLoS One 2022, 17, e0262500. [Google Scholar] [CrossRef]
- Hamed, S.B.; Hamed, M.B.B.; Kassouar, S.; Abi Ayad, S.E.-A. Physicochemical Analysis of Cellulose from Microalgae Nannochloropsis Gaditana. Afr J Biotechnol 2016, 15, 1201–1206. [Google Scholar]
- Baudelet, P.H.; Ricochon, G.; Linder, M.; Muniglia, L. A New Insight into Cell Walls of Chlorophyta. Algal Res 2017, 25, 333–371. [Google Scholar] [CrossRef]

| Extraction severity | Solvent-to-biomass ratio | Temperature [°C] | Time [h] | Concentration [%] |
| Mild | 5:1 | 60 | 1 | 1 |
| Intermediate | 10:1 | 80 | 2 | 2 |
| Harsh | 20:1 | 95 | 6 | 4 |
| Reagent | Solvent-to-biomass ratio | Temperature [°C] | Time [h] | Concentration [%] |
| NaClO2 | 20:1 | 80 | 6 | 0.85 |
| H2O2 | 20:1 | 80 | 1.5 | 30 |
| Hot water | 20:1 | 95 | 6 | - |
| NaOH | 20:1 | 95 | 6 | 4 |
| HNO3/CH3COOH | 20:1 | 95 | 1 | 6.36/72.73 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




