Submitted:
06 December 2024
Posted:
09 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
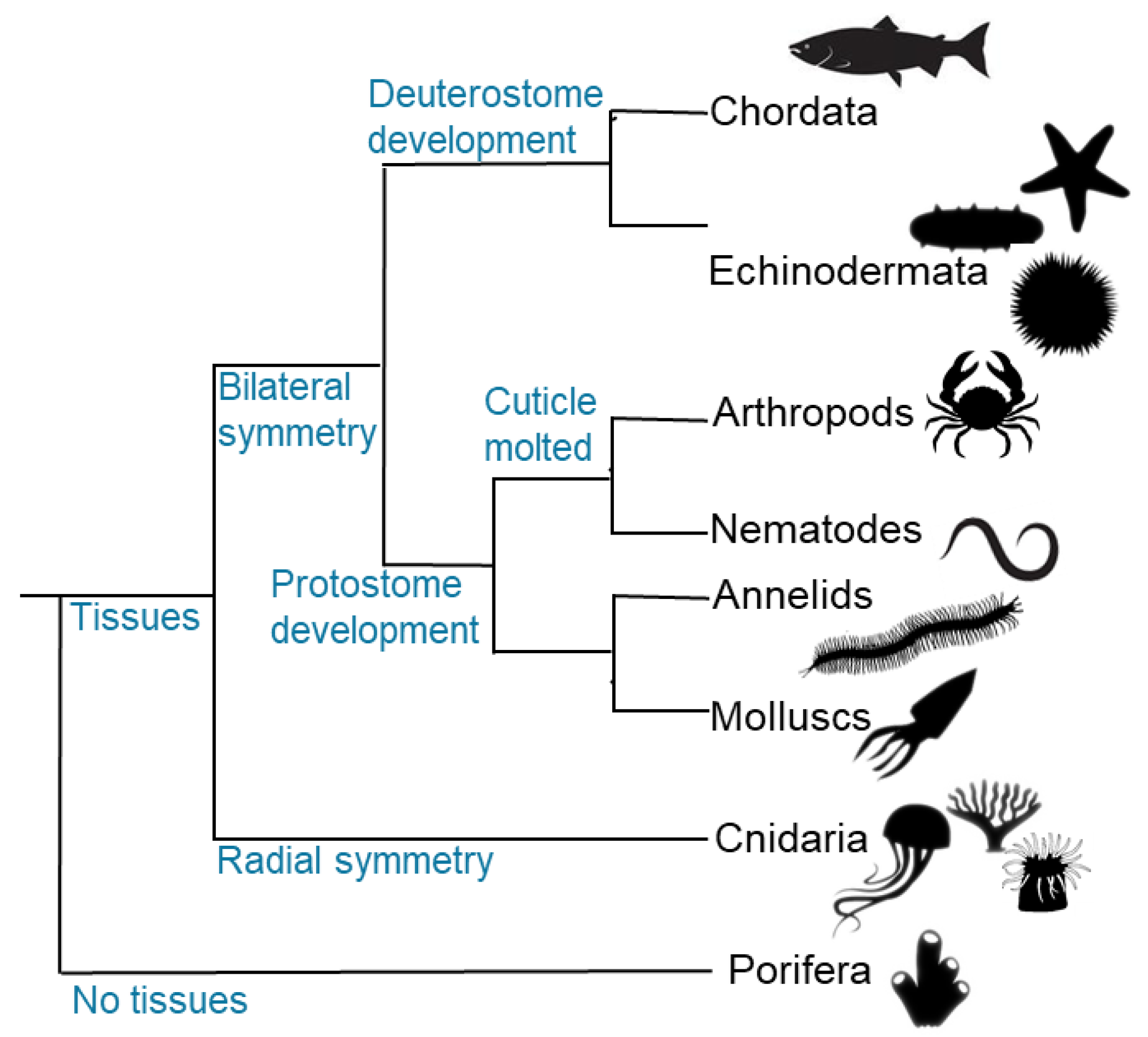
1.1. Background on Collagen Fundamentals of Porifera – Spongin and Collagen Diversity
1.2. Background on Collagen Fundamentals of Cnidaria – Mesoglea Collagens
1.3. Background on Collagen Fundamentals of Nematodes and Annelids - Cuticular Collagens
1.4. Background on Collagen Fundamentals of Molluscs and Crustaceans - Collagens from Muscle Tissues
1.5. Background on Collagen Fundamentals of Echinoderms – Mutable Collagen Tissues
2. Results
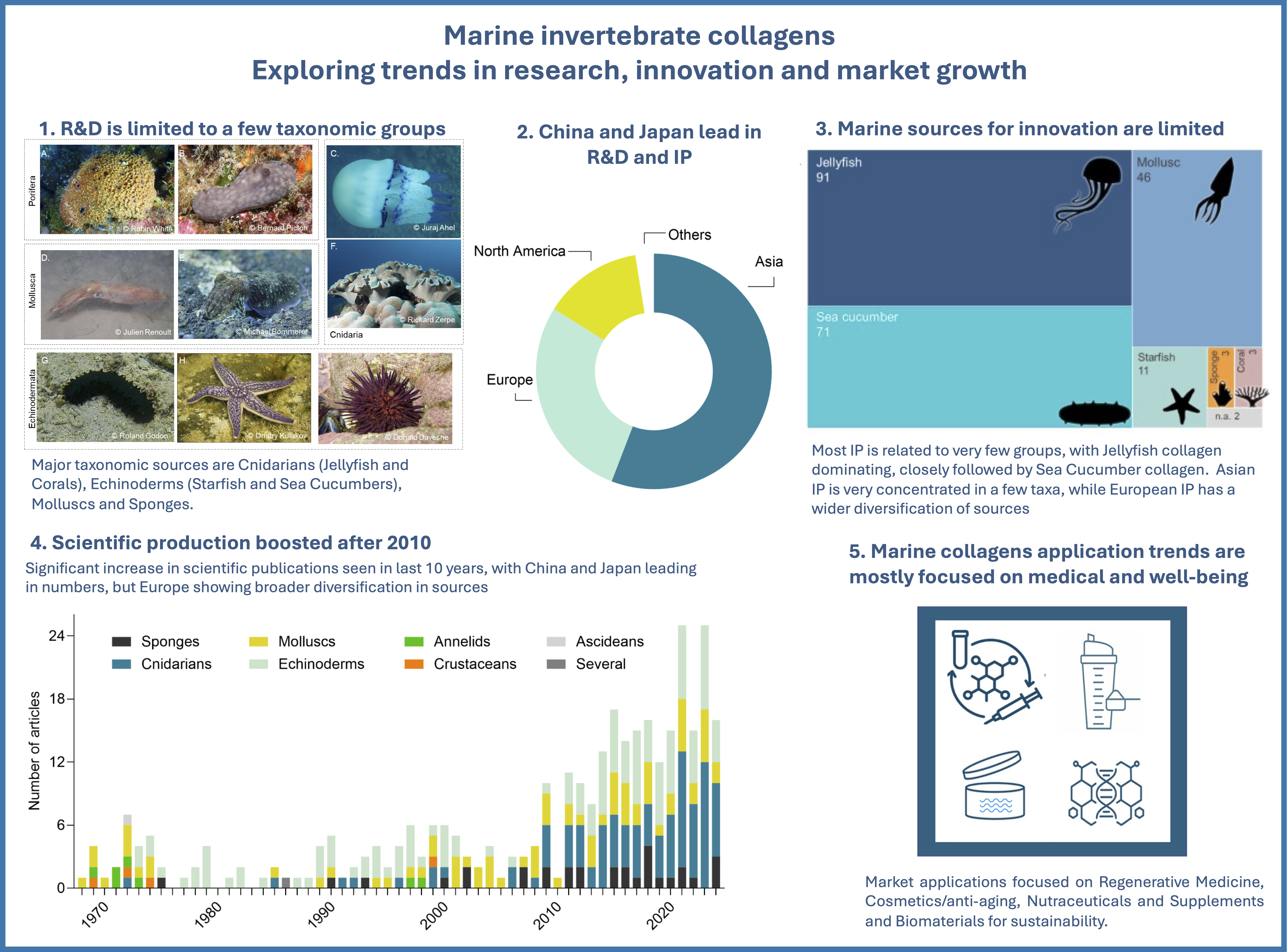
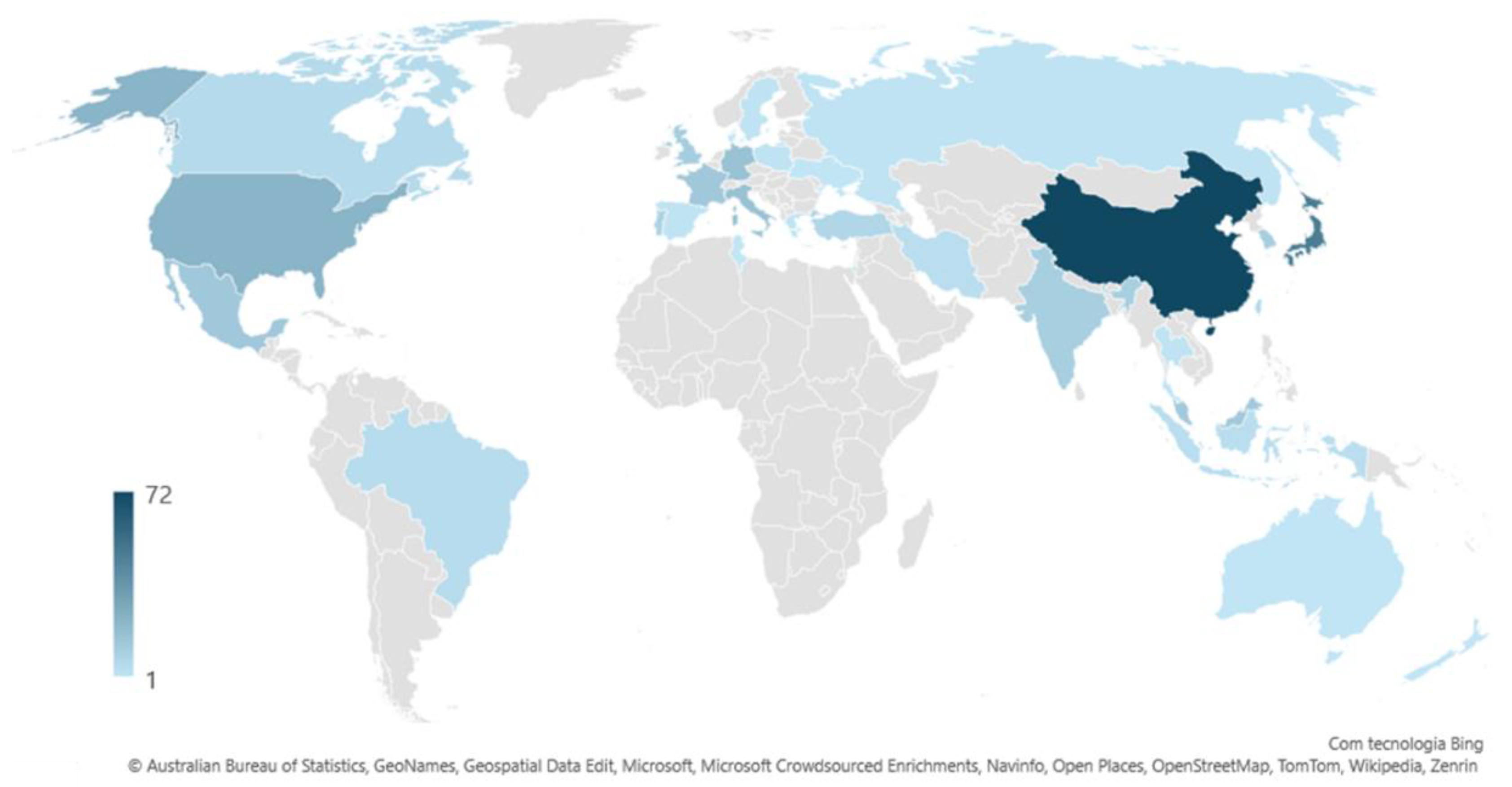
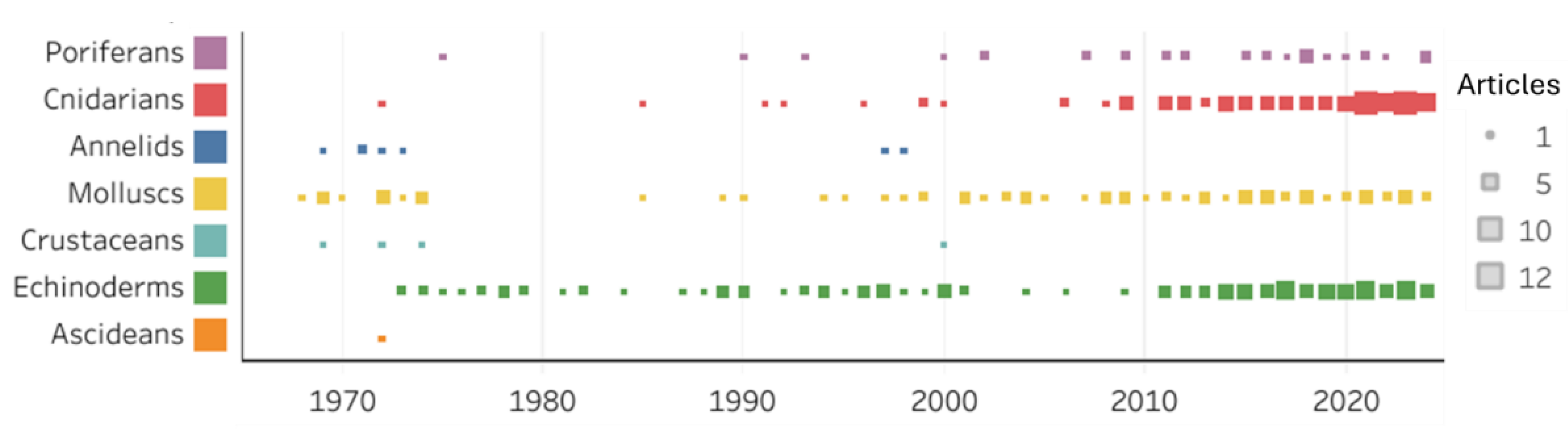
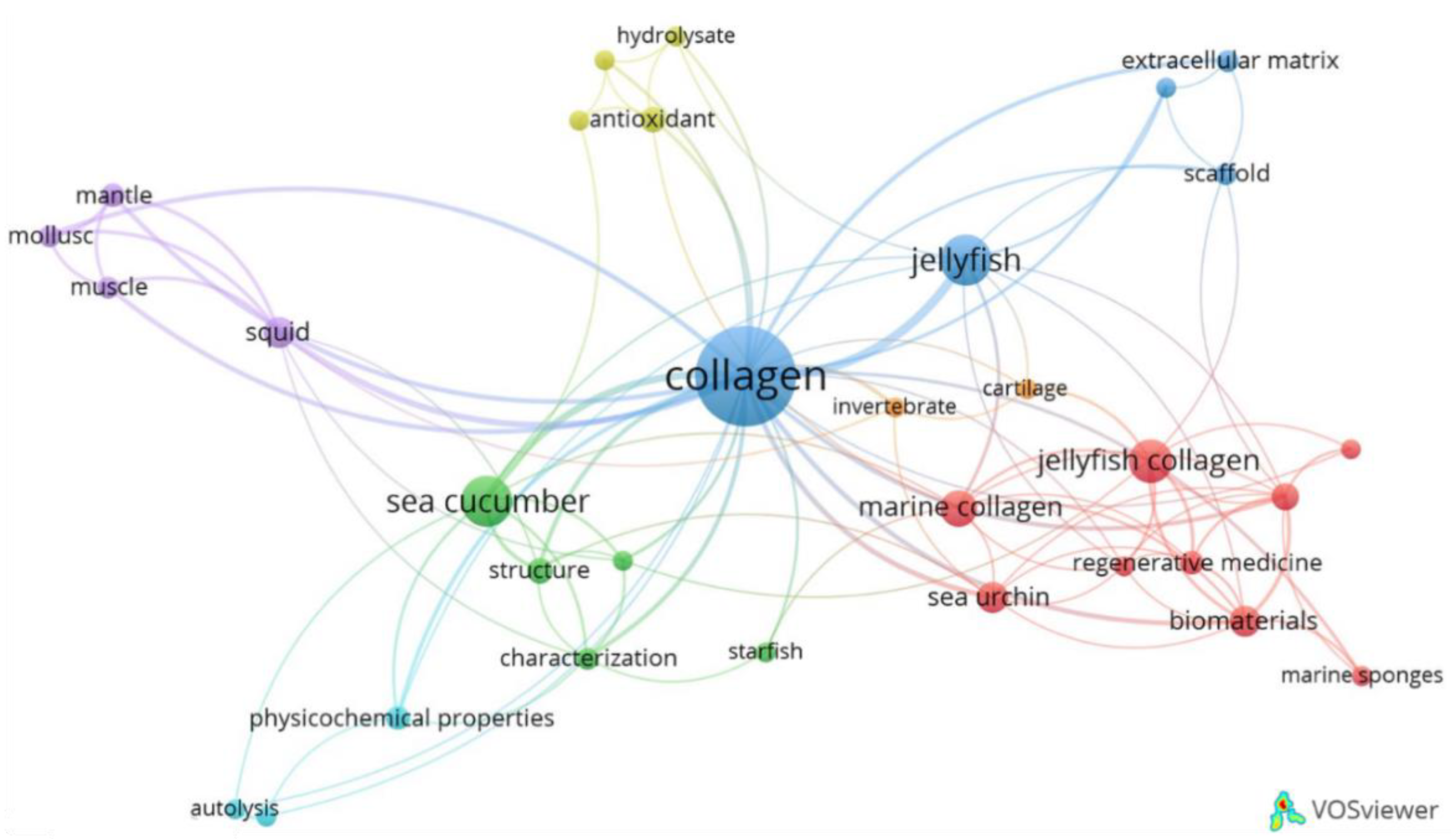
2.1. Temporal Growth, Geographic Distribution and Trends in Research Topics
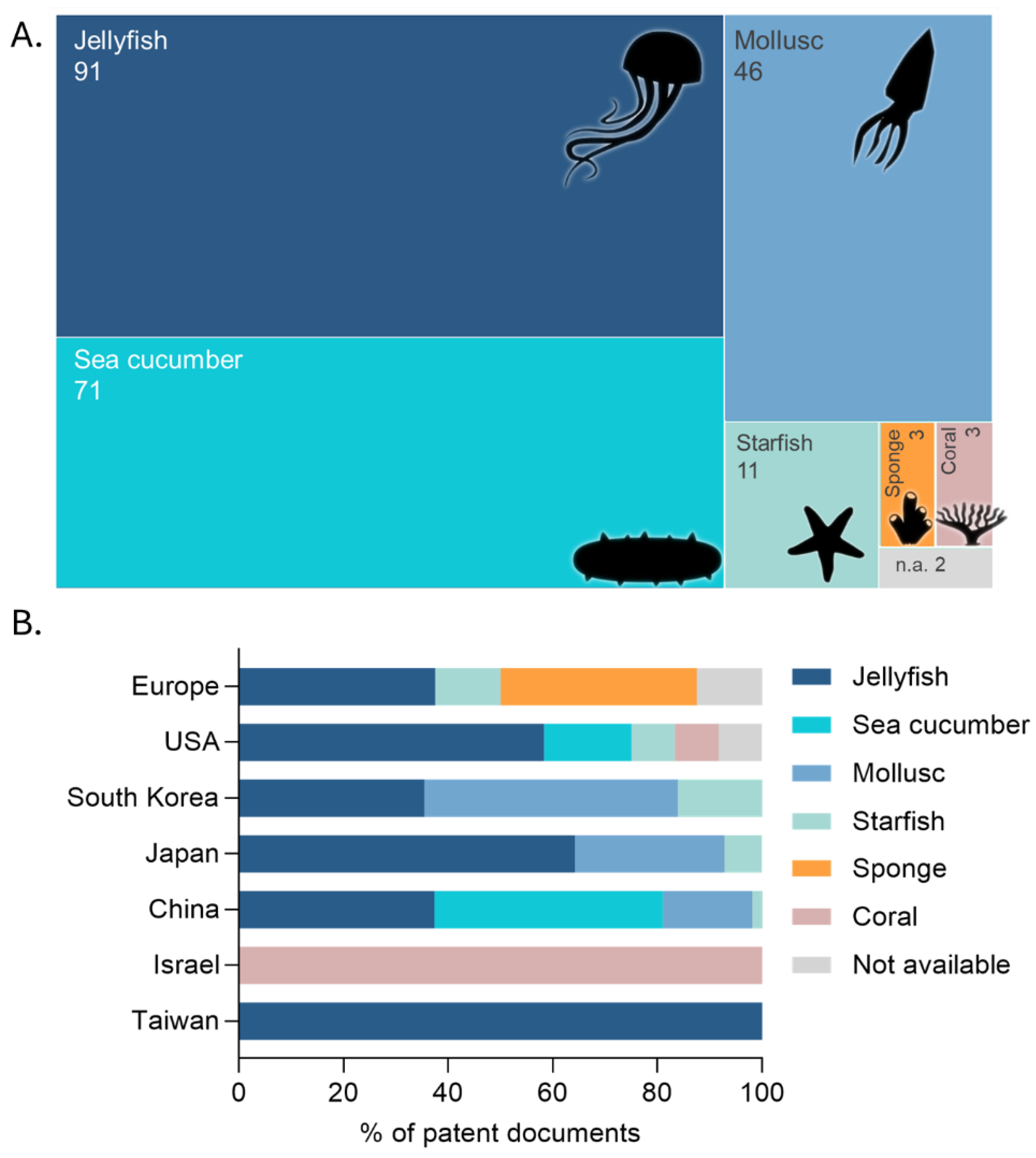
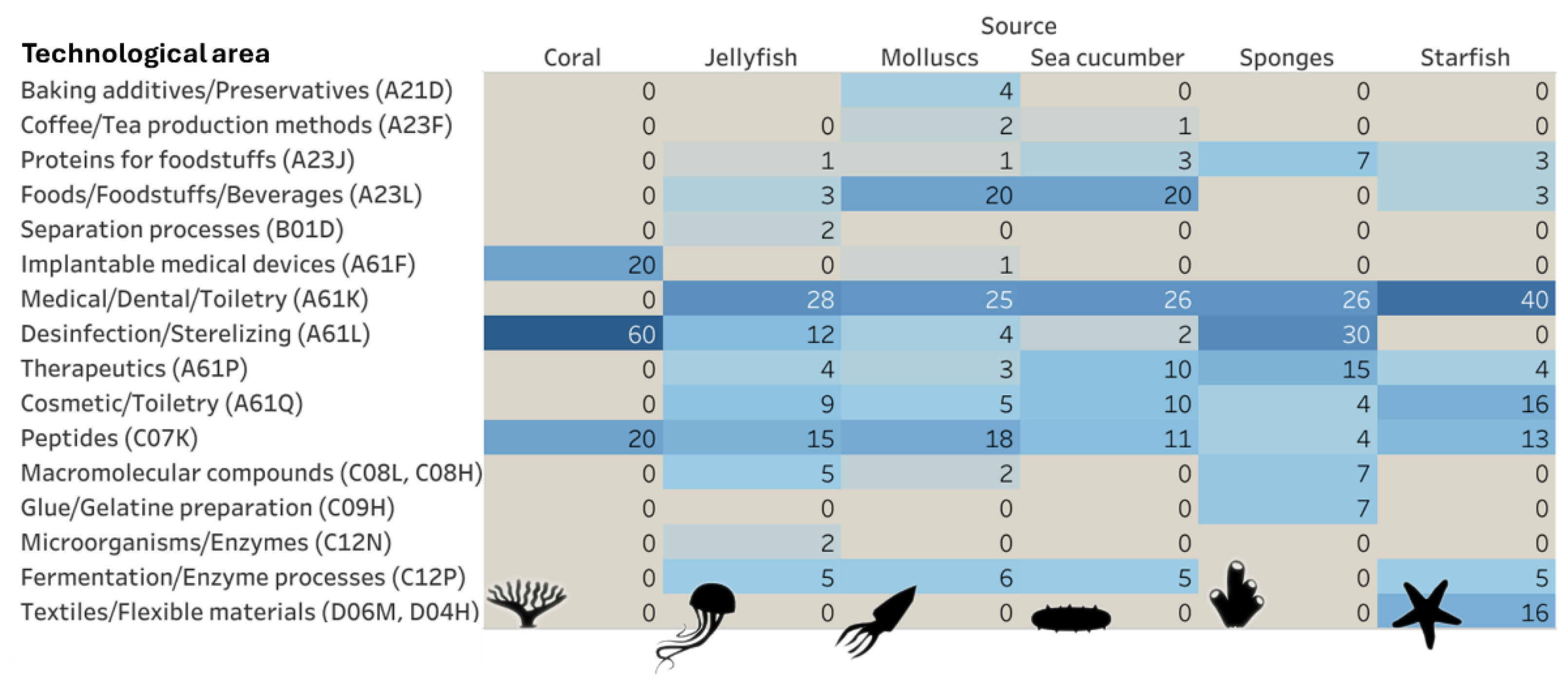
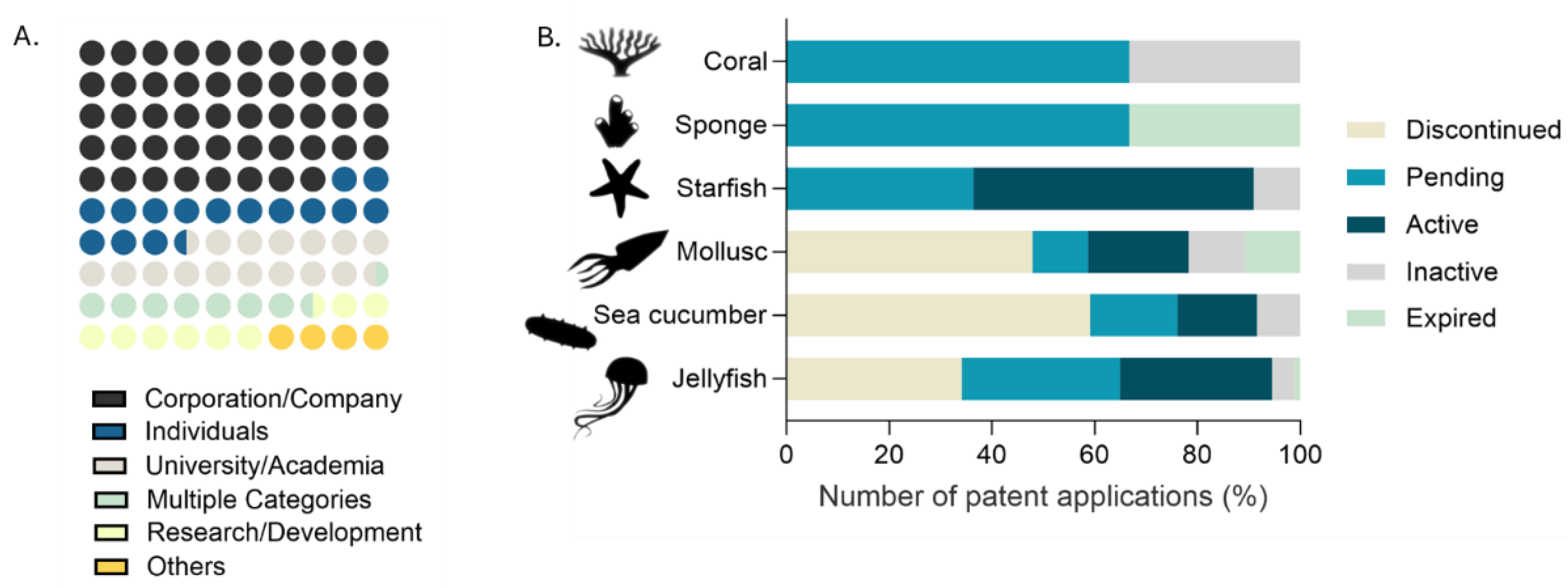
2.2. Technological Development Trends, Sources, Application Areas and Demography
2.3. Market Overview
3. Discussion
4. Materials and Methods
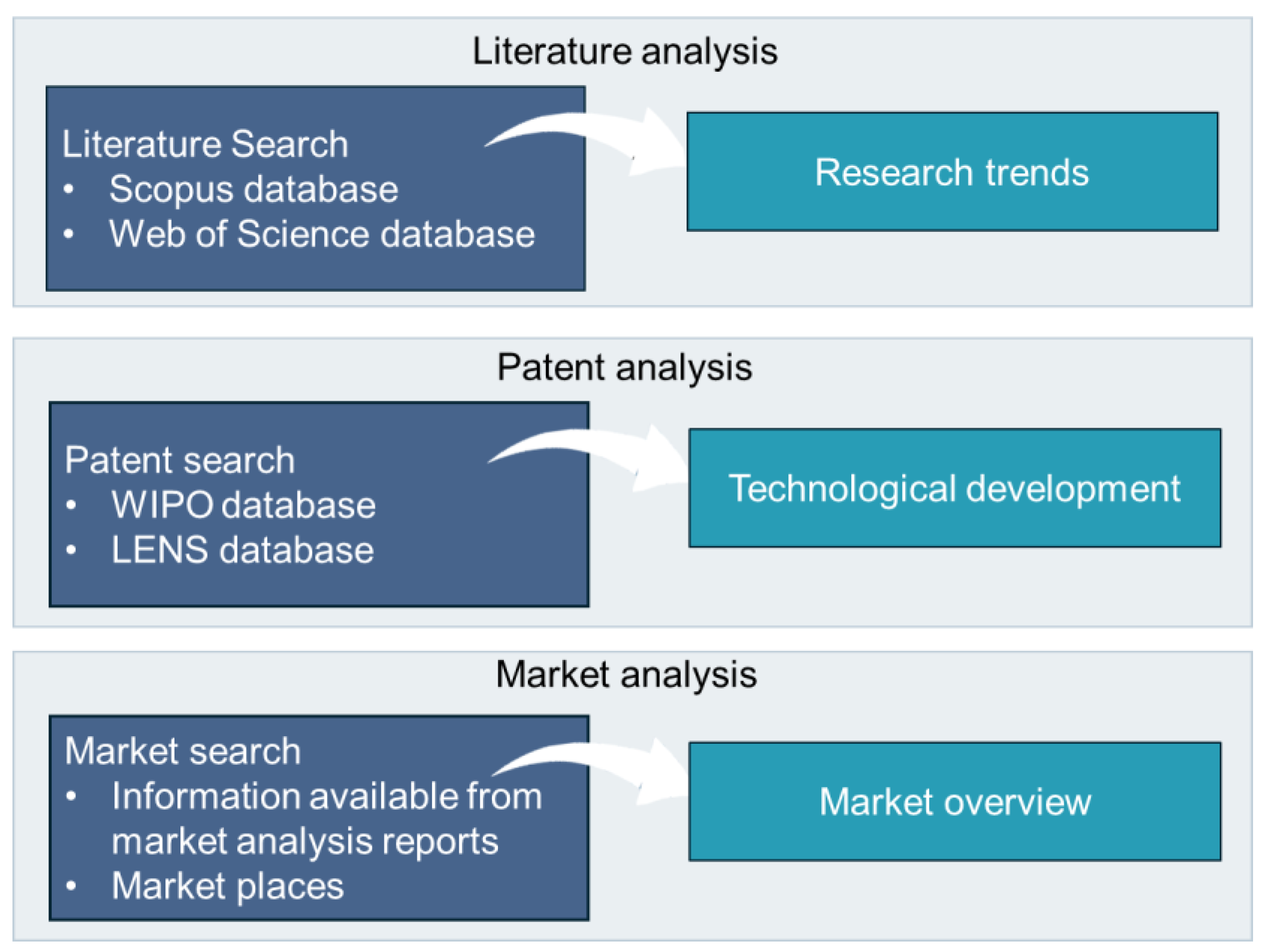
4.1. Literature Review and Patent Data Extraction and Analysis
4.2. Market Data Extraction and Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Exposito, J.-Y.; Cluzel, C.; Garrone, R.; Lethias, C. Evolution of collagens. Anat Rec 2002, 268, 302–316. [Google Scholar] [CrossRef] [PubMed]
- Shoulders, M.D.; Raines, R.T. Collagen Structure and Stability. Annu Rev Biochem 2009, 78, 929–958. [Google Scholar] [CrossRef]
- Rappu, P.; Salo, A.M.; Myllyharju, J.; Heino, J. Role of prolyl hydroxylation in the molecular interactions of collagens. Essays Biochem 2019, 63, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, M.; Sricholpech, M. Lysine post-translational modifications of collagen. Essays Biochem 2012, 52, 113–133. [Google Scholar] [CrossRef]
- Hennet, T. Collagen glycosylation. Curr Opin Struct Biol 2019, 56, 131–138. [Google Scholar] [CrossRef]
- Pokidysheva, E.; Zientek, K.D.; Ishikawa, Y.; Mizuno, K.; Vranka, J.A.; Montgomery, N.T.; Keene, D.R.; Kawaguchi, T.; Okuyama, K.; Bächinger, H.P. Posttranslational Modifications in Type I Collagen from Different Tissues Extracted from Wild Type and Prolyl 3-Hydroxylase 1 Null Mice*. Journal of Biological Chemistry 2013, 288, 24742–24752. [Google Scholar] [CrossRef]
- Sionkowska, A.; Skrzyński, S.; Śmiechowski, K.; Kołodziejczak, A. The review of versatile application of collagen. Polym Adv Technol 2017, 28, 4–9. [Google Scholar] [CrossRef]
- Fidler, A.L.; Darris, C.E.; Chetyrkin, S. V.; Pedchenko, V.K.; Boudko, S.P.; Brown, K.L.; Gray Jerome, W.; Hudson, J.K.; Rokas, A.; Hudson, B.G. Collagen iv and basement membrane at the evolutionary dawn of metazoan tissues. Elife 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Rudovica, V.; Rotter, A.; Gaudêncio, S.P.; Novoveská, L.; Akgül, F.; Akslen-Hoel, L.K.; Alexandrino, D.A.M.; Anne, O.; Arbidans, L.; Atanassova, M.; et al. Valorization of Marine Waste: Use of Industrial By-Products and Beach Wrack Towards the Production of High Added-Value Products. Front Mar Sci 2021, 8. [Google Scholar] [CrossRef]
- de Melo Oliveira, V.; Assis, C.R.D.; de Aquino Marques Costa, B.; de Araújo Neri, R.C.; Monte, F.T.D.; da Costa Vasconcelos Freitas, H.M.S.; França, R.C.P.; Santos, J.F.; de Souza Bezerra, R.; Porto, A.L.F. Physical, biochemical, densitometric and spectroscopic techniques for characterization collagen from alternative sources: A review based on the sustainable valorization of aquatic by-products. J Mol Struct 2021, 1224, 129023. [Google Scholar] [CrossRef]
- Sousa, R.O.; Alves, A.L.; Carvalho, D.N.; Martins, E.; Oliveira, C.; Silva, T.H.; Reis, R.L. Acid and enzymatic extraction of collagen from Atlantic cod (Gadus Morhua) swim bladders envisaging health-related applications. J Biomater Sci Polym Ed 2020, 31, 20–37. [Google Scholar] [CrossRef]
- Pozzolini, M.; Scarfì, S.; Gallus, L.; Castellano, M.; Vicini, S.; Cortese, K.; Gagliani, M.C.; Bertolino, M.; Costa, G.; Giovine, M. Production, Characterization and Biocompatibility Evaluation of Collagen Membranes Derived from Marine Sponge Chondrosia reniformis Nardo, 1847. Mar Drugs 2018, 16, 111–130. [Google Scholar] [CrossRef]
- Sousa, R.O.; Martins, E.; Carvalho, D.N.; Alves, A.L.; Oliveira, C.; Duarte, A.R.C.; Silva, T.H.; Reis, R.L. Collagen from Atlantic cod (Gadus morhua) skins extracted using CO2 acidified water with potential application in healthcare. Journal of Polymer Research 2020, 27. [Google Scholar] [CrossRef]
- Kendler, S.; Kobbenes, S.M.M.; Jakobsen, A.N.; Mukhatov, K.; Lerfall, J. The application of microwave and ultrasound technologies for extracting collagen from European plaice by-products. Front Sustain Food Syst 2023, 7. [Google Scholar] [CrossRef]
- Zilia, F.; Orsi, L.; Costantini, M.; Tedesco, D.E.A.; Sugni, M. Case study of Life Cycle Assessment and sustainable business model for sea urchin waste. Cleaner Environmental Systems 2023, 8, 100108. [Google Scholar] [CrossRef]
- Leonard, A.R.; Cumming, M.H.; Ali, M.A.; Cabral, J.D. Fish Collagen Cross-Linking Strategies to Improve Mechanical and Bioactive Capabilities for Tissue Engineering and Regenerative Medicine. Adv Funct Mater 2024, 34. [Google Scholar] [CrossRef]
- Carolo, A.; Melotti, L.; Zivelonghi, G.; Sacchetto, R.; Akyürek, E.E.; Martinello, T.; Venerando, A.; Iacopetti, I.; Sugni, M.; Martinelli, G.; et al. Mutable Collagenous Tissue Isolated from Echinoderms Leads to the Production of a Dermal Template That Is Biocompatible and Effective for Wound Healing in Rats. Mar Drugs 2023, 21, 506. [Google Scholar] [CrossRef] [PubMed]
- Langasco, R.; Cadeddu, B.; Formato, M.; Lepedda, A.J.; Cossu, M.; Giunchedi, P.; Pronzato, R.; Rassu, G.; Manconi, R.; Gavini, E. Natural collagenic skeleton of marine sponges in pharmaceutics: Innovative biomaterial for topical drug delivery. Materials Science and Engineering C 2017. [Google Scholar] [CrossRef]
- Kumar, A.; Elavarasan, K.; Hanjabam, M.D.; Binsi, P.K.; Mohan, C.O.; Zynudheen, A.A.; Kumar K, A. Marine collagen peptide as a fortificant for biscuit: Effects on biscuit attributes. LWT 2019, 109, 450–456. [Google Scholar] [CrossRef]
- Garrone, R. Collagen, a common thread in extracellular matrix evolution. Proceedings of the Indian Academy of Sciences - Chemical Sciences 1999, 111, 51–56. [Google Scholar] [CrossRef]
- Ehrlich, H.; Wysokowski, M.; Zółtowska-Aksamitowska, S.; Petrenko, I.; Jesionowski, T. Collagens of poriferan origin. Mar Drugs 2018, 16, 1–21. [Google Scholar] [CrossRef]
- Tziveleka, L.-A.; Ioannou, E.; Tsiourvas, D.; Berillis, P.; Foufa, E.; Roussis, V. Collagen from the Marine Sponges Axinella cannabina and Suberites carnosus: Isolation and Morphological, Biochemical, and Biophysical Characterization. Mar Drugs 2017, 15, 152. [Google Scholar] [CrossRef]
- Tassara, E.; Orel, B.; Ilan, M.; Cavallo, D.; Dodero, A.; Castellano, M.; Vicini, S.; Giovine, M.; Pozzolini, M. Seasonal Molecular Difference in Fibrillar Collagen Extracts Derived from the Marine Sponge Chondrosia reniformis (Nardo, 1847) and Their Impact on Its Derived Biomaterials. Mar Drugs 2023, 21. [Google Scholar] [CrossRef] [PubMed]
- Araújo, T.A.T.; de Souza, A.; Santana, A.F.; Braga, A.R.C.; Custódio, M.R.; Simões, F.R.; Araújo, G.M.; Miranda, A.; Alves, F.; Granito, R.N.; et al. Comparison of Different Methods for Spongin-like Collagen Extraction from Marine Sponges (Chondrilla caribensis and Aplysina fulva): Physicochemical Properties and In Vitro Biological Analysis. Membranes (Basel) 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Diehl-Seifert, B.; Kurelec, B.; Zahn, R.K.; Dorn, A.; JeriČevic, B.; Uhlenbruck, G.; Müller, W.E.G. Attachment of sponge cells to collagen substrata: Effect of a collagen assembly factor. J Cell Sci 1985, 79, 271–285. [Google Scholar] [CrossRef]
- Heinemann, S.; Ehrlich, H.; Douglas, T.; Heinemann, C.; Worch, H.; Schatton, W.; Hanke, T. Ultrastructural studies on the collagen of the marine sponge Chondrosia reniformis Nardo. Biomacromolecules 2007, 8, 3452–3457. [Google Scholar] [CrossRef] [PubMed]
- Gross, J.; SOKAL, Z.; ROUGVIE, M. STRUCTURAL AND CHEMICAL STUDIES ON THE CONNECTIVE TISSUE OF MARINE SPONGES. Journal of Histochemistry & Cytochemistry 1956, 4, 227–246. [Google Scholar] [CrossRef]
- Pallela, R.; Bojja, S.; Janapala, V.R. Biochemical and biophysical characterization of collagens of marine sponge, Ircinia fusca (Porifera: Demospongiae: Irciniidae). Int J Biol Macromol 2011, 49, 85–92. [Google Scholar] [CrossRef]
- Pozzolini, M.; Tassara, E.; Dodero, A.; Castellano, M.; Vicini, S.; Ferrando, S.; Aicardi, S.; Cavallo, D.; Bertolino, M.; Petrenko, I.; et al. Potential Biomedical Applications of Collagen Filaments derived from the Marine Demosponges Ircinia oros (Schmidt, 1864) and Sarcotragus foetidus (Schmidt, 1862). Mar Drugs 2021, 19. [Google Scholar] [CrossRef] [PubMed]
- Fassini, D.; Duarte, A.R.C.; Reis, R.L.; Silva, T.H. Bioinspiring chondrosia reniformis (Nardo, 1847) collagen-based hydrogel: A new extraction method to obtain a sticky and self-healing collagenous material. Mar Drugs 2017, 15. [Google Scholar] [CrossRef] [PubMed]
- Pozzolini, M.; Millo, E.; Oliveri, C.; Mirata, S.; Salis, A.; Damonte, G.; Arkel, M.; Scarfì, S. Elicited ROS Scavenging Activity, Photoprotective, and Wound-Healing Properties of Collagen-Derived Peptides from the Marine Sponge Chondrosia reniformis. Mar Drugs 2018, 16, 465. [Google Scholar] [CrossRef] [PubMed]
- Orel, B.; Giovine, M.; Ilan, M. On the Path to Thermo-Stable Collagen: Culturing the Versatile Sponge Chondrosia reniformis. Mar Drugs 2021, 19. [Google Scholar] [CrossRef]
- Rocha, M.S.; Marques, C.F.; Carvalho, A.C.; Martins, E.; Ereskovsky, A.; Reis, R.L.; Silva, T.H. The Characterization and Cytotoxic Evaluation of Chondrosia reniformis Collagen Isolated from Different Body Parts (Ectosome and Choanosome) Envisaging the Development of Biomaterials. Mar Drugs 2024, 22, 55. [Google Scholar] [CrossRef] [PubMed]
- Gökalp, M.; Chondrosia, S.; Kooistra, T.; Rocha, M.S.; Silva, T.H.; Osinga, R.; Murk, A.J.; Wijgerde, T. The Effect of Depth on the Morphology, Bacterial Clearance and Respiration of the Mediterranean Sponge Chondrosia reniformis (Nardo, 1847) Mert. Mar Drugs 2020, 18, 358. [Google Scholar] [CrossRef]
- Brusca, R.C.; Brusca, G.J. Invertebrates; Associates, S., Ed.; 2nd ed.; Sunderland, 2003.
- Deutzmann, R.; Fowler, S.; Zhang, X.; Boone, K.; Dexter, S.; Boot-Handford, R.P.; Rachel, R.; Sarras, M.P. Molecular, biochemical and functional analysis of a novel and developmentally important fibrillar collagen (Hcol-I) in hydra. Development 2000, 127, 4669–4680. [Google Scholar] [CrossRef] [PubMed]
- Nordwig, A.; Hayduk, U. Invertebrate collagens: Isolation, characterization and phylogenetic aspects. J Mol Biol 1969, 44, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Nordwig, A.; Nowack, H.; Hieber-Rogall, E. Sea anemone collagen: Further evidence for the existence of only oneα-chain type. J Mol Evol 1973, 2, 175–180. [Google Scholar] [CrossRef]
- Katzman, R.L. “Structural glycoprotein” from Metridium dianthus connective tissue. Life Sci 1972, 11, 131–136. [Google Scholar] [CrossRef]
- Tillet-Barret, E.; Franc, J.M.; Franc, S.; Garrone, R. Characterization of heterotrimeric collagen molecules in a sea-pen (Cnidaria, Octocorallia). Eur J Biochem 1992, 203, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Orgel, J.P.R.O.; Sella, I.; Madhurapantula, R.S.; Antipova, O.; Mandelberg, Y.; Kashman, Y.; Benayahu, D.; Benayahu, Y. Molecular and ultrastructural studies of a fibrillar collagen from octocoral (Cnidaria). Journal of Experimental Biology 2017, 220, 3327–3335. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A. Collagen of Extracellular Matrix from Marine Invertebrates and Its Medical Applications. Mar Drugs 2019, 17. [Google Scholar] [CrossRef] [PubMed]
- Miura, S.; Kimura, S. Jellyfish mesogloea collagen. Characterization of molecules as alpha 1 alpha 2 alpha 3 heterotrimers. Journal of Biological Chemistry 1985, 260, 15352–15356. [Google Scholar] [CrossRef] [PubMed]
- Addad, S.; Exposito, J.-Y.; Faye, C.; Ricard-Blum, S.; Lethias, C. Isolation, characterization and biological evaluation of jellyfish collagen for use in biomedical applications. Mar Drugs 2011, 9, 967–983. [Google Scholar] [CrossRef] [PubMed]
- Barzideh, Z.; Latiff, A.A.; Gan, C.-Y.; Benjakul, S.; Karim, A.A. Isolation and characterisation of collagen from the ribbon jellyfish (Chrysaora sp.). Int J Food Sci Technol 2014, 49, 1490–1499. [Google Scholar] [CrossRef]
- Nagai, T.; Worawattanamateekul, W.; Suzuki, N.; Nakamura, T.; Ito, T.; Fujiki, K.; Nakao, M.; Yano, T. Isolation and characterization of collagen from rhizostomous jellyfish (Rhopilema asamushi). Food Chem 2000, 70, 205–208. [Google Scholar] [CrossRef]
- Zhang, J.; Duan, R.; Huang, L.; Song, Y.; Regenstein, J.M. Characterisation of acid-soluble and pepsin-solubilised collagen from jellyfish (Cyanea nozakii Kishinouye). Food Chem 2014, 150, 22–26. [Google Scholar] [CrossRef]
- Nagai, T.; Ogawa, T.; Nakamura, T.; Ito, T.; Nakagawa, H.; Fujiki, K.; Nakao, M.; Yano, T. Collagen of edible jellyfish exumbrella. J. Sci. Food Agric. 1999, 79. [Google Scholar] [CrossRef]
- Khong, N.M.H.; Yusoff, F.Md.; Jamilah, B.; Basri, M.; Maznah, I.; Chan, K.W.; Armania, N.; Nishikawa, J. Improved collagen extraction from jellyfish (Acromitus hardenbergi) with increased physical-induced solubilization processes. Food Chem 2018, 251, 41–50. [Google Scholar] [CrossRef]
- Smith, I.P.; Domingos, M.; Richardson, S.M.; Bella, J. Characterization of the Biophysical Properties and Cell Adhesion Interactions of Marine Invertebrate Collagen from Rhizostoma pulmo. Mar Drugs 2023, 21. [Google Scholar] [CrossRef]
- Faruqui, N.; Williams, D.S.; Briones, A.; Kepiro, I.E.; Ravi, J.; Kwan, T.O.C.; Mearns-Spragg, A.; Ryadnov, M.G. Extracellular matrix type 0: From ancient collagen lineage to a versatile product pipeline – JellaGelTM. Mater Today Bio 2023, 22, 100786. [Google Scholar] [CrossRef]
- Palmieri, M.G.; Barausse, A.; Luisetti, T.; Turner, K. Jellyfish blooms in the Northern Adriatic Sea: Fishermen’s perceptions and economic impacts on fisheries. Fish Res 2014, 155, 51–58. [Google Scholar] [CrossRef]
- Özbek, S.; Pertz, O.; Schwager, M.; Lustig, A.; Holstein, T.; Engel, J. Structure/Function Relationships in the Minicollagen ofHydra Nematocysts. Journal of Biological Chemistry 2002, 277, 49200–49204. [Google Scholar] [CrossRef] [PubMed]
- Farajollahi, S.; Dennis, P.B.; Crosby, M.G.; Slocik, J.M.; Pelton, A.T.; Hampton, C.M.; Drummy, L.F.; Yang, S.J.; Silberstein, M.N.; Gupta, M.K.; et al. Disulfide Crosslinked Hydrogels Made From the Hydra Stinging Cell Protein, Minicollagen-1. Front Chem 2020, 7. [Google Scholar] [CrossRef]
- Cox, G.N. Molecular and Biochemical Aspects of Nematode Collagens. J Parasitol 1992, 78, 1. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, I.L. Cuticle collagen genes. Trends in Genetics 2000, 16, 21–27. [Google Scholar] [CrossRef]
- Gaill, F.; Mann, K.; Wiedemann, H.; Engel, J.; Timpl, R. Structural Comparison of Cuticle and Interstitial Collagens from Annelids Living in Shallow Sea-water and at Deep-sea Hydrothermal Vents. J Mol Biol 1995, 246, 284–294. [Google Scholar] [CrossRef]
- Mann, K.; Mechling, D.E.; Bächinger, H.P.; Eckerskorn, C.; Gaill, F.; Timpl, R. Glycosylated Threonine but not 4-Hydroxyproline Dominates the Triple Helix Stabilizing Positions in the Sequence of a Hydrothermal Vent Worm Cuticle Collagen. J Mol Biol 1996, 261, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Yoshinaka, R.; Mizuta, S.; Itoh, Y.; Sato, M. Two genetically distinct types of collagen in kuruma prawn Penaeus japonicus. Comparative Biochemistry and Physiology Part B: Comparative Biochemistry 1990, 96, 451–456. [Google Scholar] [CrossRef]
- Sivakumar, P.; Suguna, L.; Chandrakasan, G. Molecular species of collagen in the intramuscular connective tissues of the marine crab, Scylla serrata. Comp Biochem Physiol B Biochem Mol Biol 2000, 125, 555–562. [Google Scholar] [CrossRef]
- Sivakumar, P.; Suguna, L.; Chandrakasan, G. Similarity between the major collagens of cuttlefish cranial cartilage and cornea. Comp Biochem Physiol B Biochem Mol Biol 2003, 134, 171–180. [Google Scholar] [CrossRef]
- Veeruraj, A.; Arumugam, M.; Ajithkumar, T.; Balasubramanian, T. Isolation and characterization of collagen from the outer skin of squid (Doryteuthis singhalensis). Food Hydrocoll 2015, 43, 708–716. [Google Scholar] [CrossRef]
- Fu, W.; Wang, Y.; Zheng, B.; Liao, M.; Zhang, W. Isolation and Characterization of Pepsin-Soluble Collagen from the Skin of Peru Squid ( Dosidicus gigas ). Journal of Aquatic Food Product Technology 2013, 22, 270–280. [Google Scholar] [CrossRef]
- Hazeena, S.H.; Shih, M.-K.; Hsieh, S.-L.; Hsieh, C.-W.; Liu, T.T.; Chen, M.-H.; Huang, Y.-W.; Hou, C.-Y. Structural characteristics of collagen from cuttlefish skin waste extracted at optimized conditions. Int J Food Prop 2022, 25, 2211–2222. [Google Scholar] [CrossRef]
- Mizuta, S.; Yoshinaka, R.; Sato, M.; Sakaguchi, M. Isolation and Partial Characterization of Two Distinct Types of Collagen in the Squid Todarodes pacificus Fisheries science 1994, 60, 467–471. [CrossRef]
- Kittiphattanabawon, P.; Nalinanon, S.; Benjakul, S.; Kishimura, H. Characteristics of Pepsin-Solubilised Collagen from the Skin of Splendid Squid ( Loligo formosana ). J Chem 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- Cozza, N.; Bonani, W.; Motta, A.; Migliaresi, C. Evaluation of alternative sources of collagen fractions from Loligo vulgaris squid mantle. Int J Biol Macromol 2016, 87, 504–513. [Google Scholar] [CrossRef] [PubMed]
- Mizuta, S.; Miyagi, T.; Nishimiya, T.; Yoshinaka, R. Partial characterization of collagen in several bivalve molluscs. Food Chem 2004, 87, 83–88. [Google Scholar] [CrossRef]
- Mizuta, S.; MIYAGI, T.; YOSHINAKA, R. Characterization of the quantitatively major collagen in the mantle of oyster Crassostrea gigas. Fisheries Science 2005, 71, 229–235. [Google Scholar] [CrossRef]
- Mizuta, S.; Miyagi, T.; Nishimiya, T.; Yoshinaka, R. Partial characterization of collagen in mantle and adductor of pearl oyster (Pinctada fucata). Food Chem 2002, 79, 319–325. [Google Scholar] [CrossRef]
- Waite, J.H.; Qin, X.-X.; Coyne, K.J. The peculiar collagens of mussel byssus. Matrix Biology 1998, 17, 93–106. [Google Scholar] [CrossRef]
- Hagenau, A.; Scheidt, H.A.; Serpell, L.; Huster, D.; Scheibel, T. Structural Analysis of Proteinaceous Components in Byssal Threads of the Mussel Mytilus galloprovincialis. Macromol Biosci 2009, 9, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Wilkie, I.C. Mutable Collagenous Tissue: Overview and Biotechnological Perspective. In Echinodermata; Springer-Verlag: Berlin/Heidelberg, 2005; pp. 221–250. [Google Scholar]
- Senadheera, T.R.L.; Dave, D.; Shahidi, F. Sea Cucumber Derived Type I Collagen: A Comprehensive Review. Mar Drugs 2020, 18, 471. [Google Scholar] [CrossRef] [PubMed]
- Candia Carnevali, M.D.; Sugni, M.; Bonasoro, F.; Wilkie, I.C. Mutable Collagenous Tissue: A Concept Generator for Biomimetic Materials and Devices. Mar Drugs 2024, 22, 37. [Google Scholar] [CrossRef] [PubMed]
- Fassini, D.; Wilkie, I.C.; Pozzolini, M.; Ferrario, C.; Sugni, M.; Rocha, M.S.; Giovine, M.; Bonasoro, F.; Silva, T.H.; Reis, R.L. Diverse and Productive Source of Biopolymer Inspiration: Marine Collagens. Biomacromolecules 2021, 22, 1815–1834. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, T. Collagen Fibrils of the Sea Cucumber, Sticliopus Japonicus: Purification and Morphological Study. Connect Tissue Res 1974, 2, 117–125. [Google Scholar] [CrossRef]
- Tian, M.; Xue, C.; Chang, Y.; Shen, J.; Zhang, Y.; Li, Z.; Wang, Y. Collagen fibrils of sea cucumber (Apostichopus japonicus) are heterotypic. Food Chem 2020, 316, 126272. [Google Scholar] [CrossRef]
- Dong, X.; Zhu, B.; Sun, L.; Zheng, J.; Jiang, D.; Zhou, D.; Wu, H.; Murata, Y. Changes of collagen in sea cucumber (Stichopus japonicas) during cooking. Food Sci Biotechnol 2011, 20, 1137–1141. [Google Scholar] [CrossRef]
- Zhong, M.; Chen, T.; Hu, C.; Ren, C. Isolation and Characterization of Collagen from the Body Wall of Sea Cucumber Stichopus monotuberculatus. J Food Sci 2015, 80, C671–C679. [Google Scholar] [CrossRef] [PubMed]
- Vate, N.K.; Strachowski, P.; Undeland, I.; Abdollahi, M. Structural and functional properties of collagen isolated from lumpfish and starfish using isoelectric precipitation vs salting out. Food Chem X 2023, 18, 100646. [Google Scholar] [CrossRef]
- Li, L.; Yu, Y.; Wu, W.; Wang, P. Extraction, Characterization and Osteogenic Activity of a Type I Collagen from Starfish (Asterias amurensis). Mar Drugs 2023, 21, 274. [Google Scholar] [CrossRef]
- Burke, R.D.; Bouland, C.; Sanderson, A.I. Collagen diversity in the sea urchin, strongylocentrotus purpuratus. Comparative Biochemistry and Physiology Part B: Comparative Biochemistry 1989, 94, 41–44. [Google Scholar] [CrossRef]
- Tomita, M.; Kinoshita, T.; Izumi, S.; Tomino, S.; Yoshizato, K. Characterizations of sea urchin fibrillar collagen and its cDNA clone. Biochimica et Biophysica Acta (BBA) - Gene Structure and Expression 1994, 1217, 131–140. [Google Scholar] [CrossRef]
- Roncoroni, M.; Martinelli, G.; Farris, S.; Marzorati, S.; Sugni, M. Sea Urchin Food Waste into Bioactives: Collagen and Polyhydroxynaphtoquinones from P. lividus and S. granularis. Mar Drugs 2024, 22, 163. [Google Scholar] [CrossRef] [PubMed]
- Nhu Hieu, V.M.; Thanh Van, T.T.; Hang, C.T.T.; Mischenko, N.P.; Sergey A., F.; Truong, H.B. Polyhydroxynaphthoquinone Pigment From Vietnam Sea Urchins as a Potential Bioactive Ingredient in Cosmeceuticals. Nat Prod Commun 2020, 15. [Google Scholar] [CrossRef]
- Benedetto, C.; Barbaglio, A.; Martinello, T.; Alongi, V.; Fassini, D.; Cullorà, E.; Patruno, M.; Bonasoro, F.; Barbosa, M.; Carnevali, M.; et al. Production, Characterization and Biocompatibility of Marine Collagen Matrices from an Alternative and Sustainable Source: The Sea Urchin Paracentrotus lividus. Mar Drugs 2014, 12, 4912–4933. [Google Scholar] [CrossRef]
- Ferrario, C.; Rusconi, F.; Pulaj, A.; Macchi, R.; Landini, P.; Paroni, M.; Colombo, G.; Martinello, T.; Melotti, L.; Gomiero, C.; et al. From Food Waste to Innovative Biomaterial: Sea Urchin-Derived Collagen for Applications in Skin Regenerative Medicine. Mar Drugs 2020, 18, 414. [Google Scholar] [CrossRef]
- León-López, A.; Morales-Peñaloza, A.; Martínez-Juárez, V.M.; Vargas-Torres, A.; Zeugolis, D.I.; Aguirre-Álvarez, G. Hydrolyzed Collagen—Sources and Applications. Molecules 2019, 24, 4031. [Google Scholar] [CrossRef] [PubMed]
- FAO The State of World Fisheries and Aquaculture 2022; FAO, 2022; ISBN 978-92-5-136364-5.
- Transparency Market research Collagen Market (Source: Bovine, Porcine, Marine, and Poultry; Application: Pharmaceutical & Healthcare, Food & Beverage, Cosmetics, Nutraceuticals, and Others; and Product: Gelatin, Hydrolyzed Gelatin, and Native Collagen) - Global Industry Analysis, Size, Share, Growth, Trends, and Forecast, 2021-2028; Delaware, USA, 2022.
- Precedence Research Collagen Market Size, Share, and Trends 2024 to 2034; Canada/India, 2024.
- Markets and Markets Collagen Market by Product Type (Gelatin, Hydrolyzed Collagen, Native Collagen), Application, Source (Bovine, Porcine, Poultry, Marine, and Plant Sources), Form, Type, Extraction Process and Region—Global Forecast to 2030; Dublin, Ireland, 2023.
- Market Research Future Marine Collagen Market Research Report By Source, By Form, By Application, By Target Consumers, By Distribution Channel, By Regional Source; New York, USA, 2024.
- Plez, K.A.; Gross, J. The amino acid composition and morphology of some invertebrate and vertebrate collagens. Biochim Biophys Acta 1959, 34, 24–39. [Google Scholar] [CrossRef]
- Science and Engineering Indicators Publications Output: U.S. Trends and International Comparisons; 2023.
- Herbert-Read, J.E.; Thornton, A.; Amon, D.J.; Birchenough, S.N.R.; Côté, I.M.; Dias, M.P.; Godley, B.J.; Keith, S.A.; McKinley, E.; Peck, L.S.; et al. A global horizon scan of issues impacting marine and coastal biodiversity conservation. Nat Ecol Evol 2022, 6, 1262–1270. [Google Scholar] [CrossRef]
- Cheng, X.; Shao, Z.; Li, C.; Yu, L.; Raja, M.A.; Liu, C. Isolation, Characterization and Evaluation of Collagen from Jellyfish Rhopilema esculentum Kishinouye for Use in Hemostatic Applications. PLoS One 2017, 12(1), e0169731. [Google Scholar] [CrossRef]
- Zhao, J.; Yu, W.; Zhang, Q.; Li, X.; Huang, Y.; Zhao, S.; Li, T.; Liu, S.; Li, Y.; Shan, H. Structural and biofunctional evaluation of decellularized jellyfish matrices. J Mater Chem B 2023, 11, 3740–3751. [Google Scholar] [CrossRef] [PubMed]
- Cisneros-Mata, M.A.; Mangin, T.; Bone, J.; Rodriguez, L.; Smith, S.L.; Gaines, S.D. Fisheries governance in the face of climate change: Assessment of policy reform implications for Mexican fisheries. PLoS One 2019, 14, e0222317. [Google Scholar] [CrossRef]
- Chiarelli, P.G.; Suh, J.H.; Pegg, R.B.; Chen, J.; Mis Solval, K. The emergence of jellyfish collagen: A comprehensive review on research progress, industrial applications, and future opportunities. Trends Food Sci Technol 2023, 141, 104206. [Google Scholar] [CrossRef]
- Cadar, E.; Pesterau, A.-M.; Sirbu, R.; Negreanu-Pirjol, B.S.; Tomescu, C.L. Jellyfishes—Significant Marine Resources with Potential in the Wound-Healing Process: A Review. Mar Drugs 2023, 21, 201. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Lloréns, J.L.; Mouritsen, O.G. Sea cucumber: A scavenger overexploited, traded and turned into food (even a gastronomic delicacy). Int J Gastron Food Sci 2024, 37, 100996. [Google Scholar] [CrossRef]
- Mordor Intelligence China Collagen Market Size & Share Analysis - Growth Trends & Forecasts (2024 - 2029). Mordor Intelligence 2024.
- Vate, N.K.; Undeland, I.; Abdollahi, M. Resource efficient collagen extraction from common starfish with the aid of high shear mechanical homogenization and ultrasound. Food Chem 2022, 393, 133426. [Google Scholar] [CrossRef] [PubMed]
- Duarte A. R., C.; Moreira-Silva, J.; Silva T., H.; Osinga, R.; Ilan, M.; Marques A., P.; Mano J., F.; Reis, R. L Marine sponges as natural scaffolds: decellularization by supercritical fluid technology and cellularization with osteoblasts for tissue engineering applications. J Tissue Eng Regen Med 2012, 6, 171–172. [Google Scholar]
- Green, D.; Howard, D.; Yang, X.; Kelly, M.; Oreffo, R.O.C. Natural Marine Sponge Fiber Skeleton: A Biomimetic Scaffold for Human Osteoprogenitor Cell Attachment, Growth, and Differentiation. Tissue Eng 2003, 9, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Bierwirth, J.; Mantas, T.P.; Villechanoux, J.; Cerrano, C. Restoration of Marine Sponges—What Can We Learn from over a Century of Experimental Cultivation? Water (Basel) 2022, 14, 1055. [Google Scholar] [CrossRef]
- Sarabia-Sainz, H.M.; Ezquerra-Brauer, J.M.; Santacruz-Ortega, H.C.; Rouzaud-Sández, O.; Valenzuela-Soto, E.M.; Acosta-Elias, M.; Torres-Arreola, W. Muscle lysyl oxidase activity and structural/thermal properties of highly cross-linked collagen in jumbo squid (Dosidicus gigas) mantle, fins and arms. Food Sci Biotechnol 2018, 27, 57–64. [Google Scholar] [CrossRef]
- Ezquerra-Brauer, J.M.; Márquez-Ríos, E.; López-Corona, B.E.; Ocaño-Higuera, V.M.; Ramírez-Guerra, H.E.; Cota-Arriola, O.; Torres-Arreola, W. Physicochemical changes of pepsin-solubilized and insoluble collagen in jumbo squid ( Dosidicus gigas ) muscle after cooking process. Int J Food Prop 2018, 21, 821–834. [Google Scholar] [CrossRef]
- Zhao, Q.; Li, Z.; Liu, Z.; Zhao, X.; Fan, Y.; Dong, P.; Hou, H. Preparation, typical structural characteristics and relieving effects on osteoarthritis of squid cartilage type II collagen peptides. Food Research International 2024, 191, 114697. [Google Scholar] [CrossRef]
- Nakchum, L.; Kim, S.M. Preparation of squid skin collagen hydrolysate as an antihyaluronidase, antityrosinase, and antioxidant agent. Prep Biochem Biotechnol 2016, 46, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Mittal, A.; Benjakul, S. Full Utilization of Squid Meat and Its Processing By-products: Revisit. Food Reviews International 2022, 38, 455–479. [Google Scholar] [CrossRef]
- LUYUN, C.; MINJIE, C.; JIANRONG, L.; YANBO, W.; TAO, M. Production method of squid skin collagen peptide instant noodle sandwich biscuits capable of nourishing skin 2018.
- Quero, M.J.; Díaz-Méndez, M.; Ventura, R.; Gummesson, E. Co-patenting, co-ownership, and co-ideation as drivers for university business innovation: the case of public universities in Spain. TQM Journal 2022, 328–346. [Google Scholar] [CrossRef]
- Tan, C.C.; Karim, A.A.; Latiff, A.A.; Gan, C.Y.; Ghazali, F.C. Extraction and characterization of pepsin-solubilized collagen from the body wall of crown-of-thorns Starfish (Acanthaster planci). Int Food Res J 2013, 20. [Google Scholar]
- Cui, F.; Xue, C.; Li, Z.; Zhang, Y.; Dong, P.; Fu, X.; Gao, X. Characterization and subunit composition of collagen from the body wall of sea cucumber Stichopus japonicus. Food Chem 2007, 100, 1120–1125. [Google Scholar] [CrossRef]
- Saito, M.; Kunisaki, N.; Urano, N.; Kimura, S. Collagen as the Major Edible Component of Sea Cucumber (Stichopus japonicus). J Food Sci 2002, 67, 1319–1322. [Google Scholar] [CrossRef]
- Drake, J.L.; Mass, T.; Haramaty, L.; Zelzion, E.; Bhattacharya, D.; Falkowski, P.G. Proteomic analysis of skeletal organic matrix from the stony coral Stylophora pistillata. Proceedings of the National Academy of Sciences 2013, 110, 3788–3793. [Google Scholar] [CrossRef]
- Bielajew, B.J.; Hu, J.C.; Athanasiou, K.A. Collagen: quantification, biomechanics and role of minor subtypes in cartilage. Nat Rev Mater 2020, 5, 730–747. [Google Scholar] [CrossRef]
- Alves, AL.; Marques, C.F.; Almeida, M.; Pérez-Martín, R.I.; Reis, R.L.; Silva, T.H. Isolation and Characterization of Marine-Derived Collagens and Gelatins in the Perspective of Biomedical Application. In Handbook of the Extracellular Matrix; Springer International Publishing: Cham, 2023; pp. 1–28. [Google Scholar]
- Edelist, D.; Angel, D.L.; Canning-Clode, J.; Gueroun, S.K.M.; Aberle, N.; Javidpour, J.; Andrade, C. Jellyfishing in Europe: Current Status, Knowledge Gaps, and Future Directions towards a Sustainable Practice. Sustainability 2021, 13, 12445. [Google Scholar] [CrossRef]
- Lucas, C.H.; Pitt, K.A.; Purcell, J.E.; Lebrato, M.; Condon, R.H. What’s in a jellyfish? Proximate and elemental composition and biometric relationships for use in biogeochemical studies. Ecology 2011, 92, 1704–1704. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021, n71. [Google Scholar] [CrossRef] [PubMed]

| Brand/Company and Source | Country | Ingredient/Product | Application | |
|---|---|---|---|---|
 |
Jellagen1: Jellyfish | United Kingdom | Power (in pre- clinical stage) | Wound care |
 |
KliniPharm2: sponge | Germany | Boswellia Spongi®-Kollagen-Lecithin Capsules | Nutraceutical |
 |
KliniPharm: sponge | Germany | Spongi Sunflower Collagen-Lecithin Capsules | Nutraceutical |
 |
KliniPharm: sponge | Germany | SpongiCol® Collagen-Lecithin Capsules | Nutraceutical |
 |
KliniPharm: sponge | Germany | SpongiCol Collagen-Lecithin Granulate | Nutraceutical |
 |
KliniPharm: sponge | Germany | SpongiCol ECO Collagen-Lecithin Capsules | Nutraceutical |
 |
Certified Nutraceuticals3: Jellyfish | USA | kollaJell Brain Health & Cognitive function | Nutraceutical |
 |
MitoLife4: Jellyfish | USA | Jellyfish Jolt (Wild-caught collagen) - capsules | Nutraceutical |
 |
Neogenesis Health5: Jellyfish | South Africa | INFINITY JellyFish Collagencapsules | Nutraceutical |
 |
Nova Sea Atlantic6: Sea cucumber | Canada | Nova Sea Atlantic® Sea Cucumber Capsules | Nutraceutical |
 |
KliniPharm: sponge | Germany | ELEANA MARINE collagen creme nourishing sponge extract | Cosmetic |
 |
KliniPharm: sponge | Germany | ELEANA MARINE collagen serum$reconstructing sponge extract | Cosmetic |
 |
KliniPharm: sponge | Germany | ELEANA MARINE collagen gel nourishing sponge extract | Cosmetic |
 |
Nano Recipe7: sponge | South Korea | Korean Marin Collagen Concentrate Serum | Cosmetic |
 |
Joli Visage8 | USA | Joli Collagen Spongilla Cream | Cosmetic |
 |
Coseed BioPharm9Jellyfish and starfish | South Korea | Jellyfish extract$Starfish collagen | Cosmetic |
 |
Bescher10: Sea cucumber | Australia | Sea cucumber collagen Anti-Ageing Cream | Cosmetic |
 |
Bescher: Sea cucumber | Australia | Sea Cucumber Collagen Blemish Defence Serum | Cosmetic |
 |
Bescher: Sea cucumber | Australia | Sea Cucumber Collagen Regeneration Serum | Cosmetic |
 |
Bescher: Sea cucumber | Australia | Sea Cucumber Collagen Glow Toner | Cosmetic |
 |
Bescher: Sea cucumber | Australia | Sea Cucumber Collagen Gel Cleanser | Cosmetic |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





