Submitted:
04 December 2024
Posted:
05 December 2024
You are already at the latest version
Abstract
Keywords:
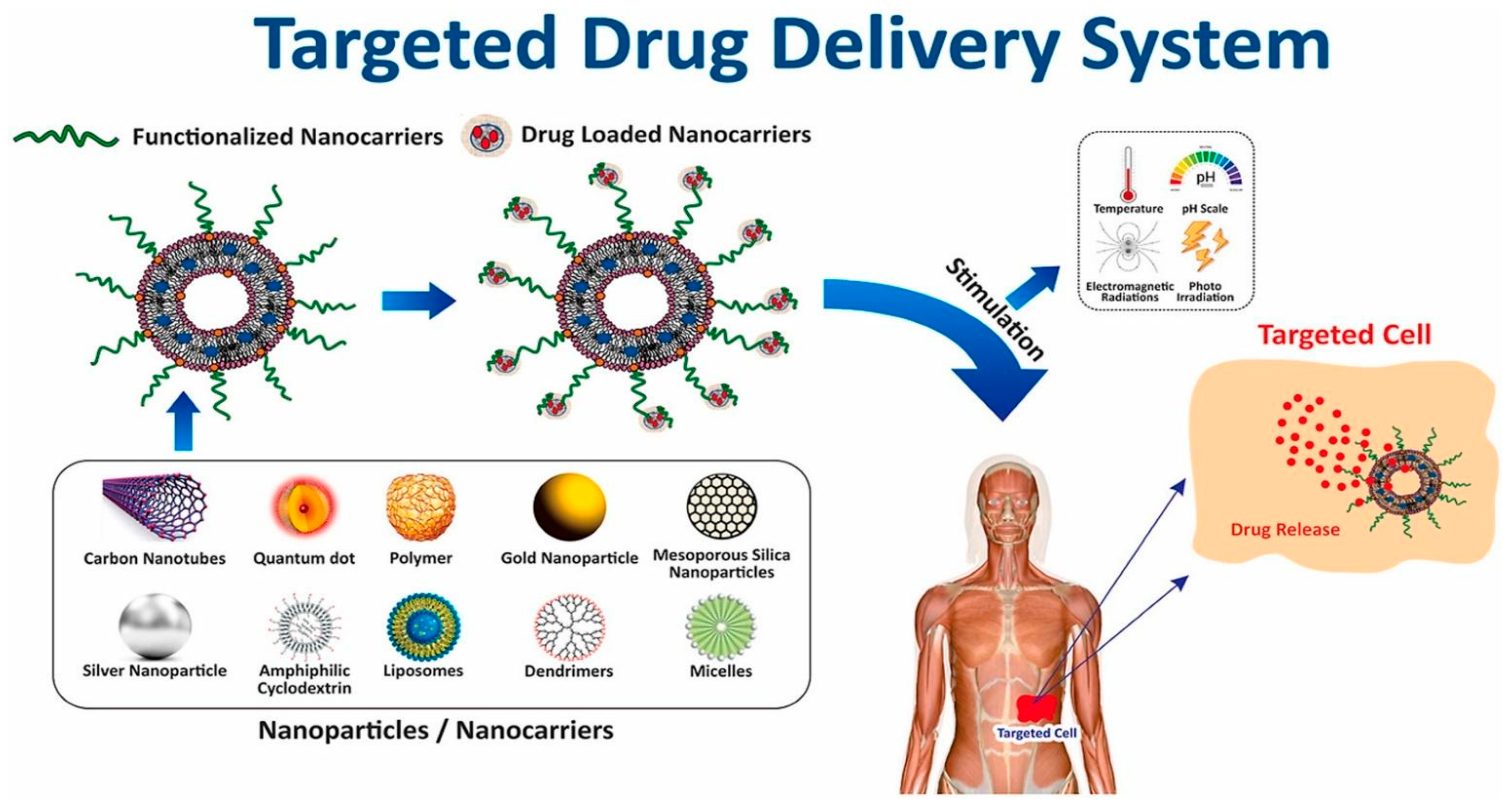
1. Introduction
2. Metallic Nanoparticles
3. Silica/Organosilica Nanoparticles
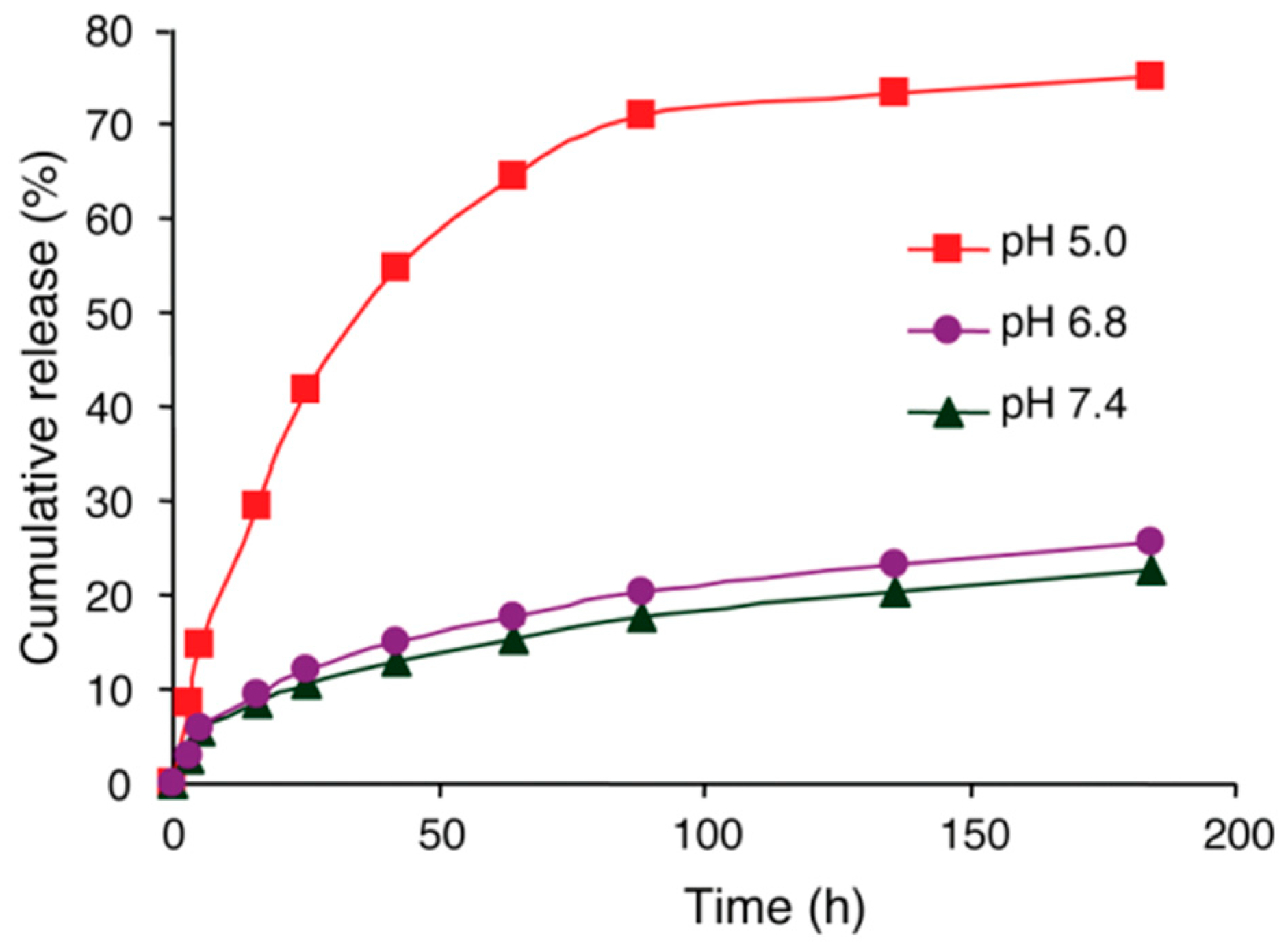
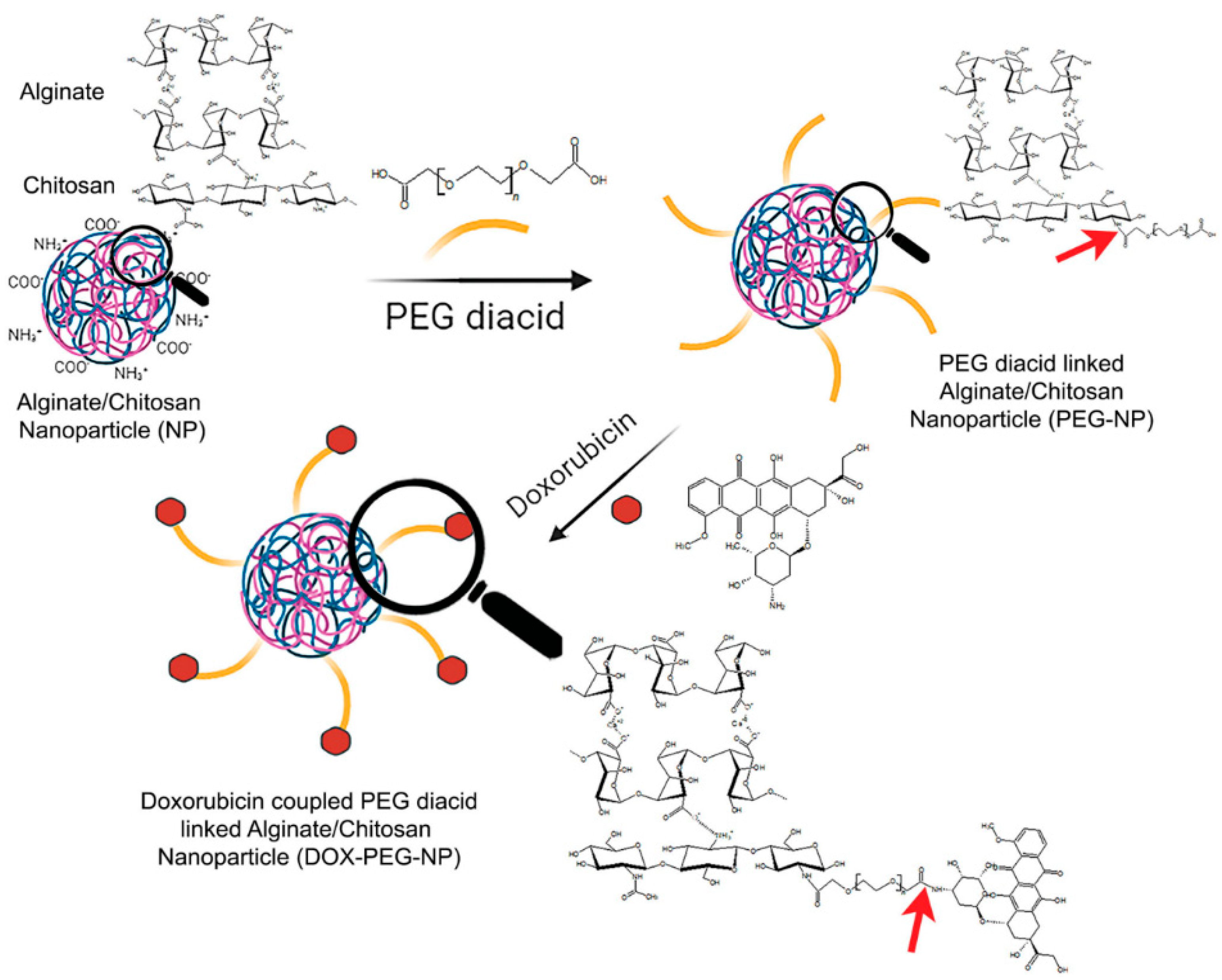
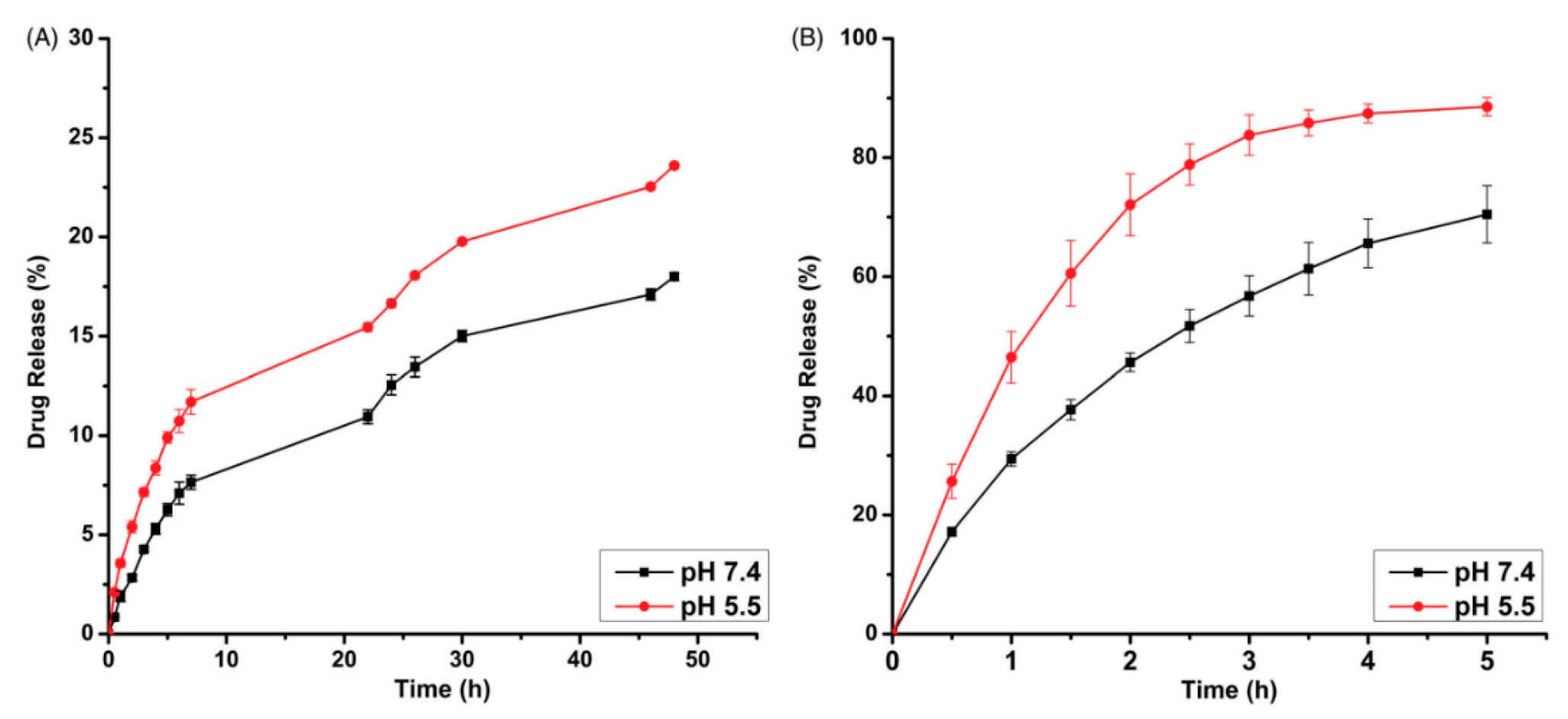
4. Polymeric Nanoparticles
5. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zaid, A. S; Aleissawy, A. E.; Yahia, I. S.; Yassien, M. A.; Hassouna, N. A.; Aboshanab, K. M. Streptomyces griseus KJ623766: A Natural Producer of Two Anthracycline Cytotoxic Metabolites β-and γ-Rhodomycinone. Molecules 2021, 26, 4009. [Google Scholar] [CrossRef]
- Rivankar, S. An overview of doxorubicin formulations in cancer therapy. J. Cancer Res. Ther. 2014, 10, 853–858. [Google Scholar] [CrossRef]
- Mohammadi, M.; Arabi, L.; Alibolandi, M. Doxorubicin-loaded composite nanogels for cancer treatment. J. Control. Release 2020, 328, 171–191. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, K.; Zhang, J.; Honbo, N.; Karliner J., S. Doxorubicin Cardiomyopathy. Cardiology 2010, 115, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Shim, M. K.; Kim, W. J.; Choi, J.; Nam, G. H.; Kim, J.; Kim, K. Cancer-activated doxorubicin prodrug nanoparticles induce preferential immune response with minimal doxorubicin-related toxicity. Biomater. 2021, 272, 120791. [Google Scholar] [CrossRef]
- Sultana, A.; Zare, M.; Thomas, V.; Kumar, T. S.; Ramakrishna, S. Nano-based drug delivery systems: Conventional drug delivery routes, recent developments and future prospects. Medicine Drug Discov. 2022, 100134. [Google Scholar] [CrossRef]
- Mun, E. A.; Zhaisanbayeva, B. A. Silica Nanoparticles for Biomedical Application: Challenges and Opportunities. EurAsian J. Appl. Biotechnol. 2020. [Google Scholar] [CrossRef]
- Shah, A.; Aftab, S.; Nisar, J.; Ashiq, M. N.; Iftikhar, F. J. Nanocarriers for targeted drug delivery. J. Drug Deliv. Sci. Technol. 2021, 62, 102426. [Google Scholar] [CrossRef]
- Chhikara, B. S.; Rathi, B.; Parang, K. Critical evaluation of pharmaceutical rational design of Nano-Delivery systems for Doxorubicin in Cancer therapy. J. Mater. NanoSci. 2019, 6, 47–66. [Google Scholar]
- Sahu, S.; Sinha, N.; Bhutia, S. K.; Majhi, M.; Mohapatra, S. Luminescent magnetic hollow mesoporous silica nanotheranostics for camptothecin delivery and multimodal imaging. J. Mater. Chem. B 2014, 2, 3799–3808. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, K. K.; Bhatt, A. N.; Mishra, A. K.; Dwarakanath, B. S.; Jain, S.; Schatz, C.; Lecommandoux, S. The intracellular drug delivery and anti tumor activity of doxorubicin loaded poly (γ-benzyl l-glutamate)-b-hyaluronan polymersomes. Biomater. 2010, 31, 2882–2892. [Google Scholar] [CrossRef] [PubMed]
- Meng, F.; Engbers, G. H.; Feijen, J. Biodegradable polymersomes as a basis for artificial cells: encapsulation, release and targeting. J. Control. Release 2005, 101, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Chandrakala, V.; Aruna, V.; Angajala, G. Review on metal nanoparticles as nanocarriers: Current challenges and perspectives in drug delivery systems. Emerg. Mater. 2022, 1–23. [Google Scholar] [CrossRef]
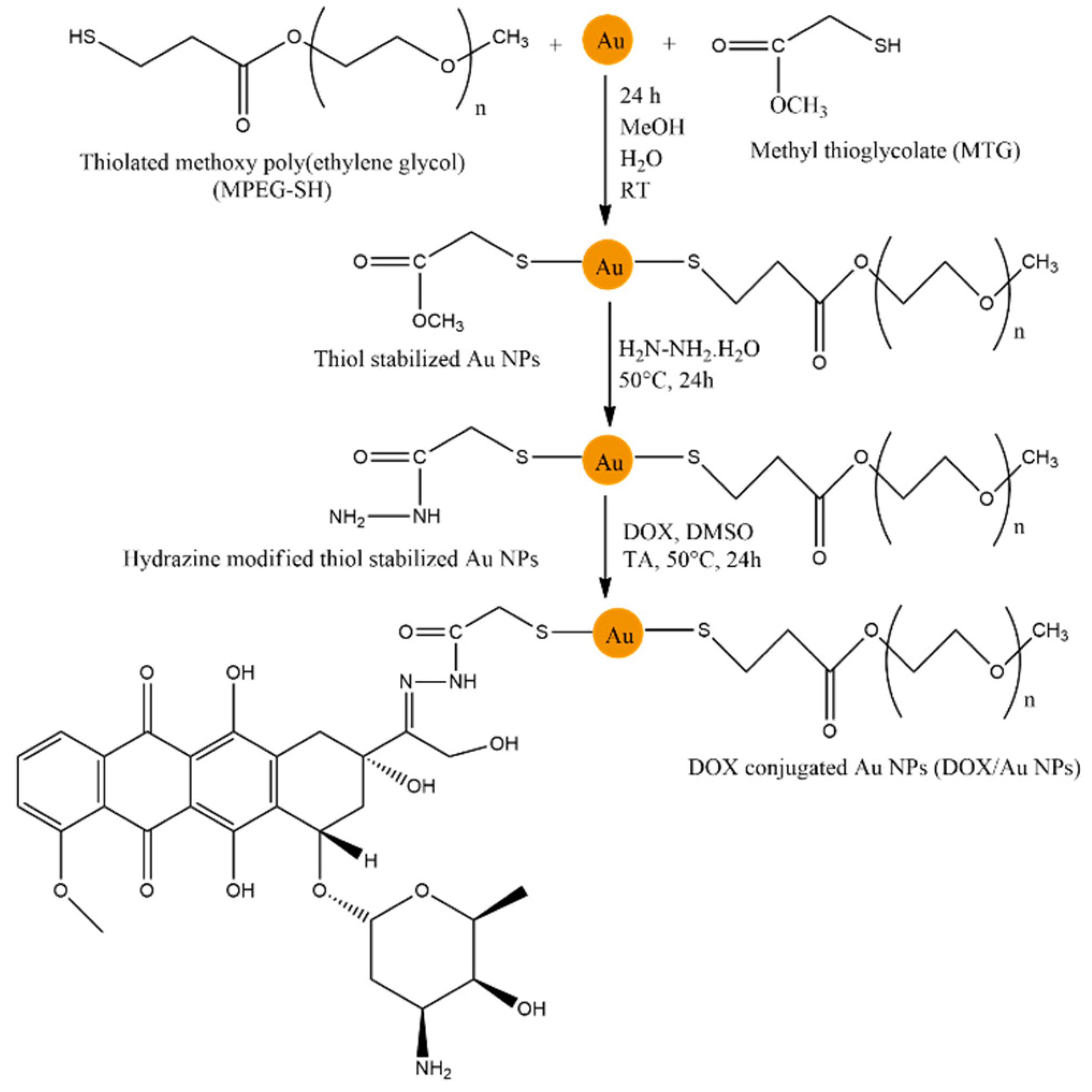
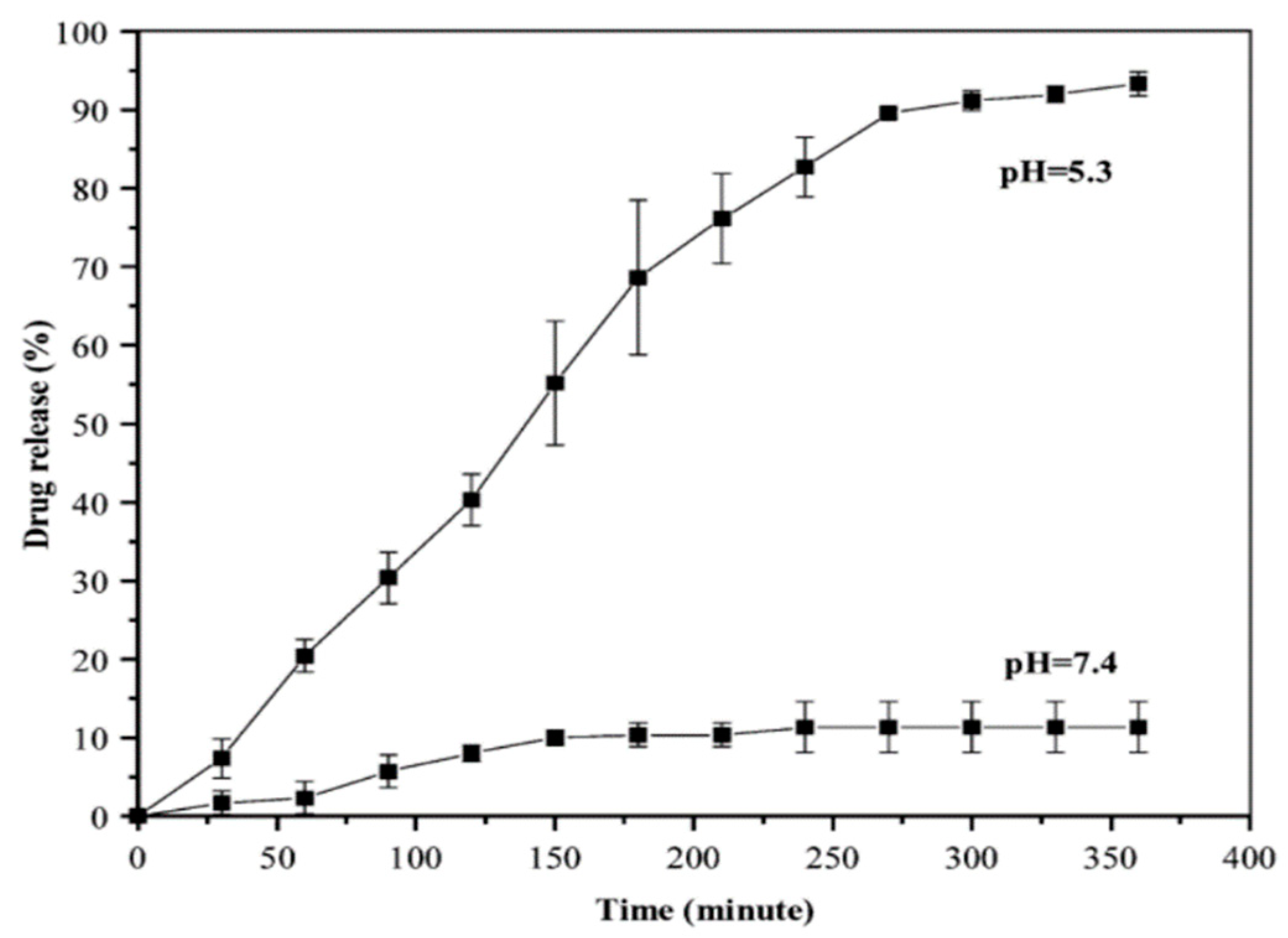
- Aryal, S.; Grailer, J. J.; Pilla, S.; Steeber, D. A.; Gong, S. Doxorubicin conjugated gold nanoparticles as water-soluble and pH-responsive anticancer drug nanocarriers. J. Mater. Chem. 2009, 19, 7879–7884. [Google Scholar] [CrossRef]
- Wang, J. X.; Choi, S. Y.; Niu, X.; Kang, N.; Xue, H.; Killam, J.; Wang, Y. Lactic acid and an acidic tumor microenvironment suppress anticancer immunity. Int. J. Mol. Sci. 2020, 21, 8363. [Google Scholar] [CrossRef] [PubMed]
- Cui, T.; Liang, J.-J.; Chen, H.; Geng, D.-D.; Jiao, L.; Yang, J.-Y.; Ding, Y. Performance of Doxorubicin-Conjugated Gold Nanoparticles: Regulation of Drug Location. ACS Appl. Mater. Interfaces 2017, 9, 8569–8580. [Google Scholar] [CrossRef]
- Wu, W.; He, Q.; Jiang, C. Magnetic Iron Oxide Nanoparticles: Synthesis and Surface Functionalization Strategies. Nanoscale Res. Lett. 2008, 3, 397–415. [Google Scholar] [CrossRef]
- Zhu, N.; Ji, H.; Yu, P.; Niu, J.; Farooq, M.; Akram, M.; Niu, X. Surface Modification of Magnetic Iron Oxide Nanoparticles. Nanomater. 2018, 8, 810. [Google Scholar] [CrossRef]
- Vangijzegem, T.; Stanicki, D.; Laurent, S. Magnetic iron oxide nanoparticles for drug delivery: applications and characteristics. Expert Opin. Drug Deliv. 2019, 16, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Majeed, M. I.; Lu, Q.; Yan, W.; Li, Z.; Hussain, I.; Tahir, M. N.; Tan, B. Highly water-soluble magnetic iron oxide (Fe3O4) nanoparticles for drug delivery: enhanced in vitro therapeutic efficacy of doxorubicin and MION conjugates. J. Mater. Chem. B 2013, 1, 2874–2884. [Google Scholar] [CrossRef]
- Guimarães, R. S.; Rodrigues, C. F.; Moreira, A. F.; Correia, I. J. Overview of stimuli-responsive mesoporous organosilica nanocarriers for drug delivery. Pharmacol. Res. 2020, 155, 104742. [Google Scholar] [CrossRef] [PubMed]
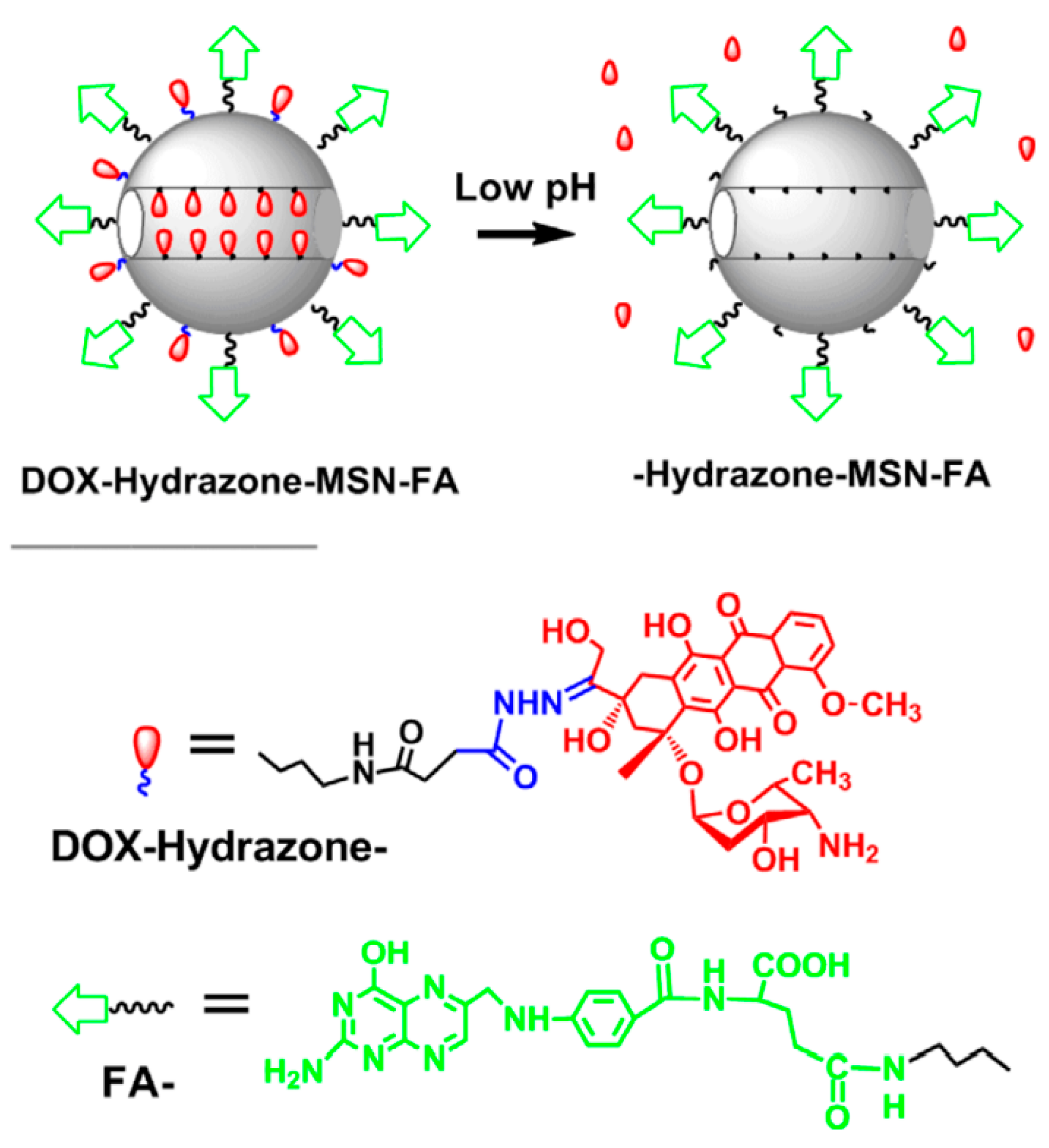
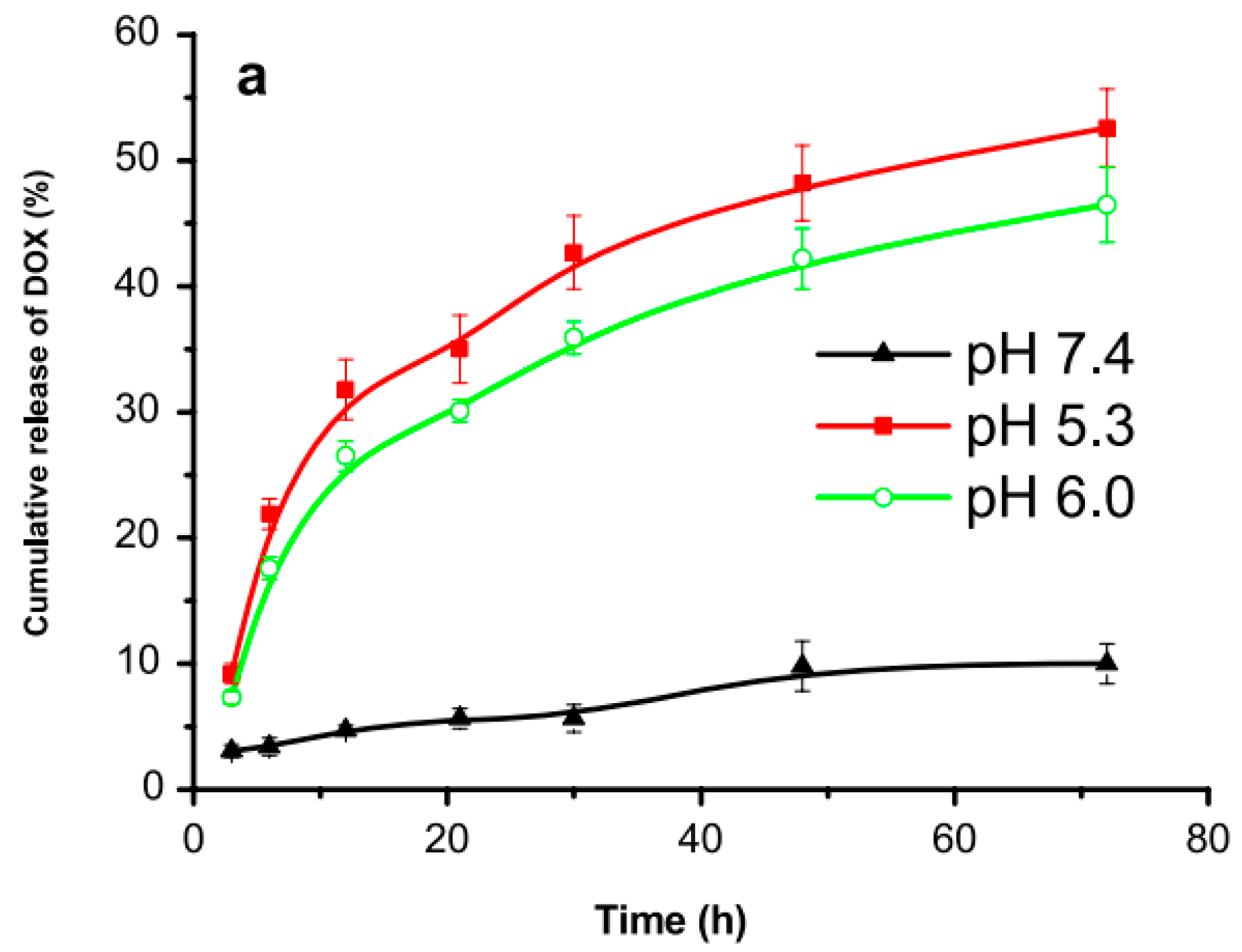
- Zahiri, M.; Falsafi, M.; Lamei, K.; Abnous, K.; Taghdisi, S. M.; Ramezani, M.; Alibolandi, M. Targeted biomimetic hollow mesoporous organosilica nanoparticles for delivery of doxorubicin to colon adenocarcinoma: In vitro and in vivo evaluation. Micropor. Mesopor. Mat. 2022, 335, 111841. [Google Scholar] [CrossRef]
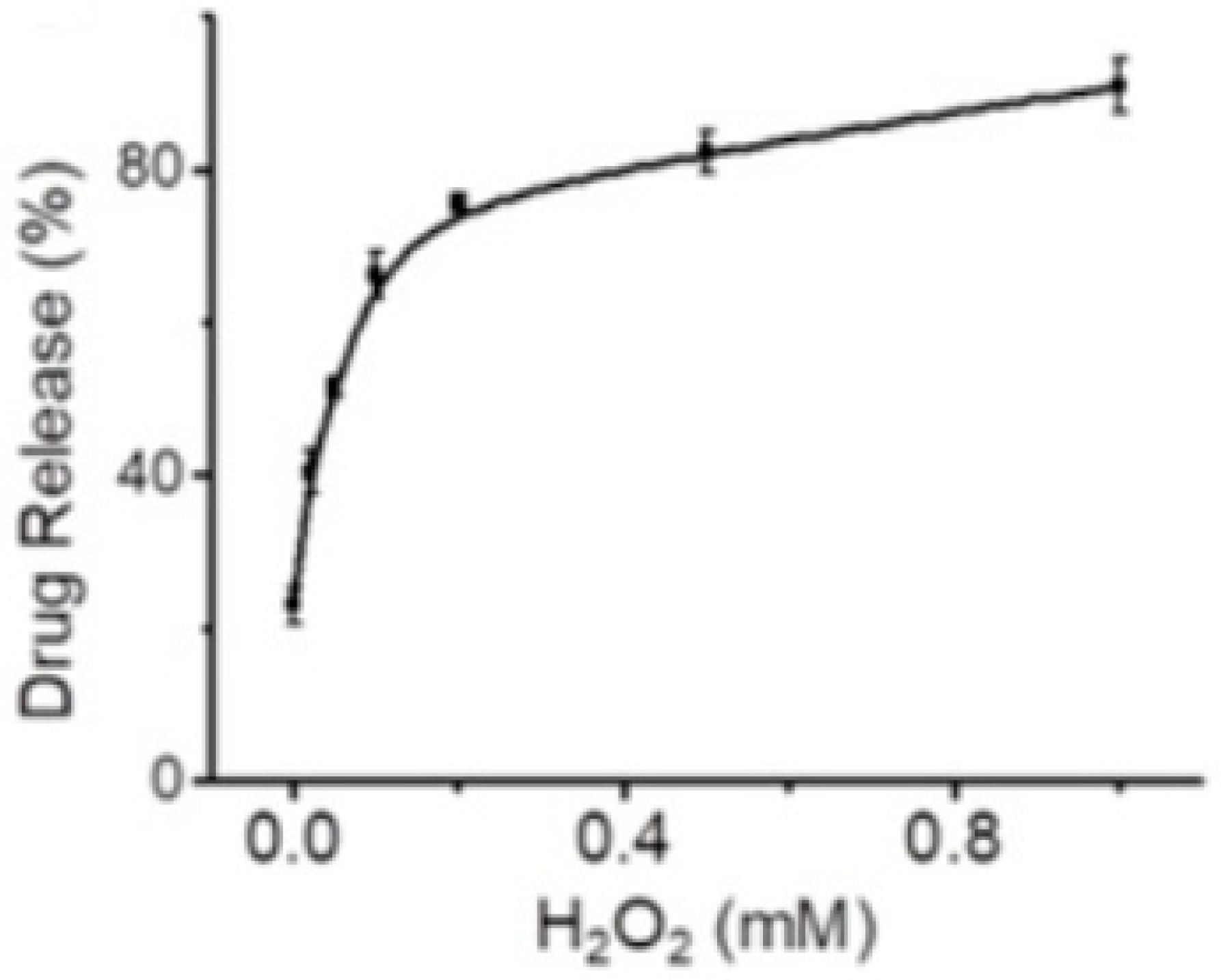
- Xu, Y.; Shi, W.; Li, H.; Li, X.; Ma, H. H2O2-Responsive Organosilica-Doxorubicin Nanoparticles for Targeted Imaging and Killing of Cancer Cells Based on a Synthesized Silane-Borate Precursor. ChemMedChem 2019, 14, 1079–1085. [Google Scholar] [CrossRef]
- Fan, J.; Fang, G.; Wang, X.; Zeng, F.; Xiang, Y.; Wu, S. Targeted anticancer prodrug with mesoporous silica nanoparticles as vehicles. Nanotechnol. 2011, 22, 455102. [Google Scholar] [CrossRef]
- Begines, B.; Ortiz, T.; Pérez-Aranda, M.; Martínez, G.; Merinero, M.; Argüelles-Arias, F.; Alcudia, A. Polymeric nanoparticles for drug delivery: Recent developments and future prospects. Nanomater. 2020, 10, 1403. [Google Scholar] [CrossRef]
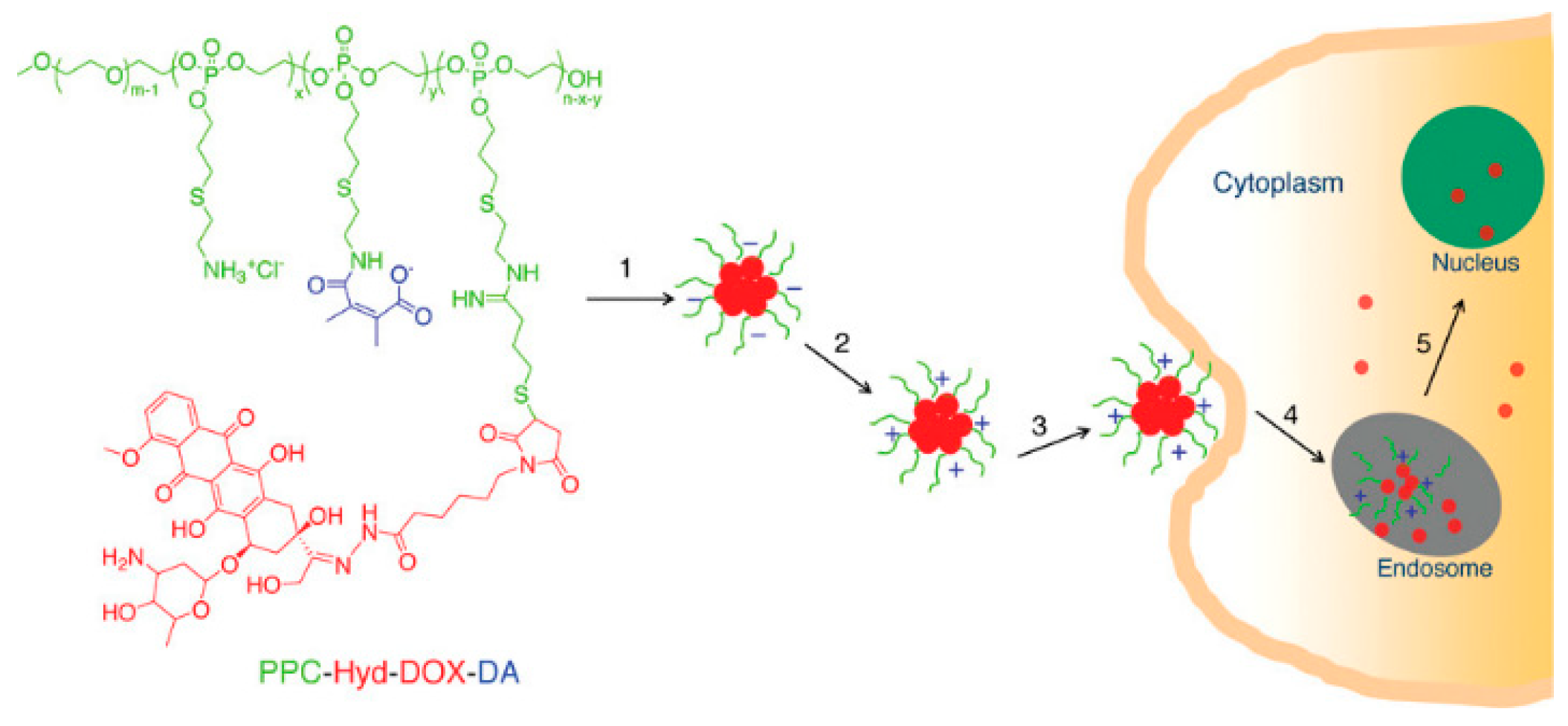
- Du, J. Z.; Du, X. J.; Mao, C. Q.; Wang, J. Tailor-made dual pH-sensitive polymer–doxorubicin nanoparticles for efficient anticancer drug delivery. J. Am. Chem. Soc. 2011, 133, 17560–17563. [Google Scholar] [CrossRef] [PubMed]
- Ak, G. Covalently coupling doxorubicin to polymeric nanoparticles as potential inhaler therapy: In vitro studies. Pharm. Dev. Technol. 2021, 26, 890–898. [Google Scholar] [CrossRef] [PubMed]
- Abd Ellah, N. H.; Abouelmagd, S. A. Surface functionalization of polymeric nanoparticles for tumor drug delivery: approaches and challenges. Expert Opin. Drug Deliv. 2017, 14, 201–214. [Google Scholar] [CrossRef] [PubMed]
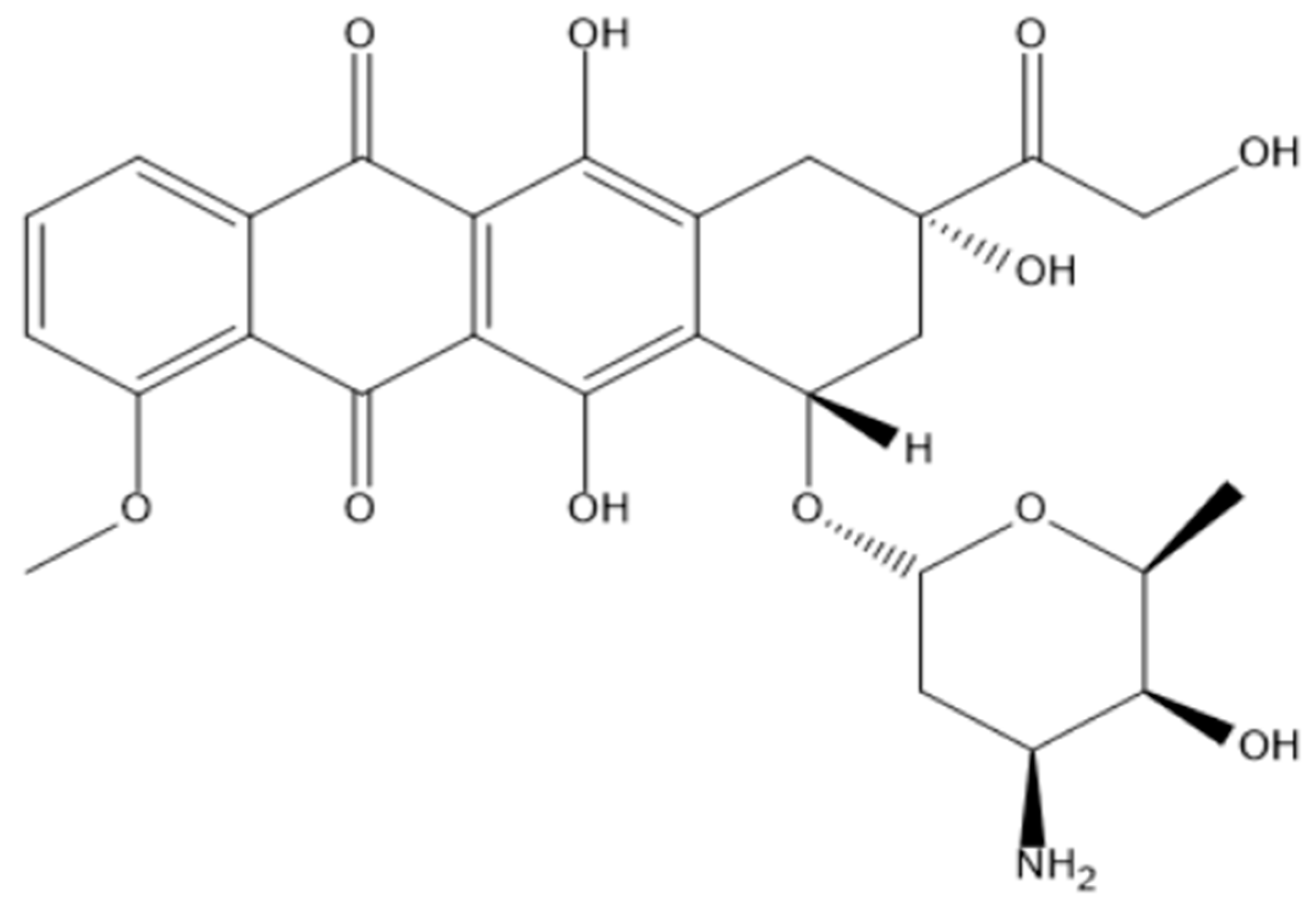
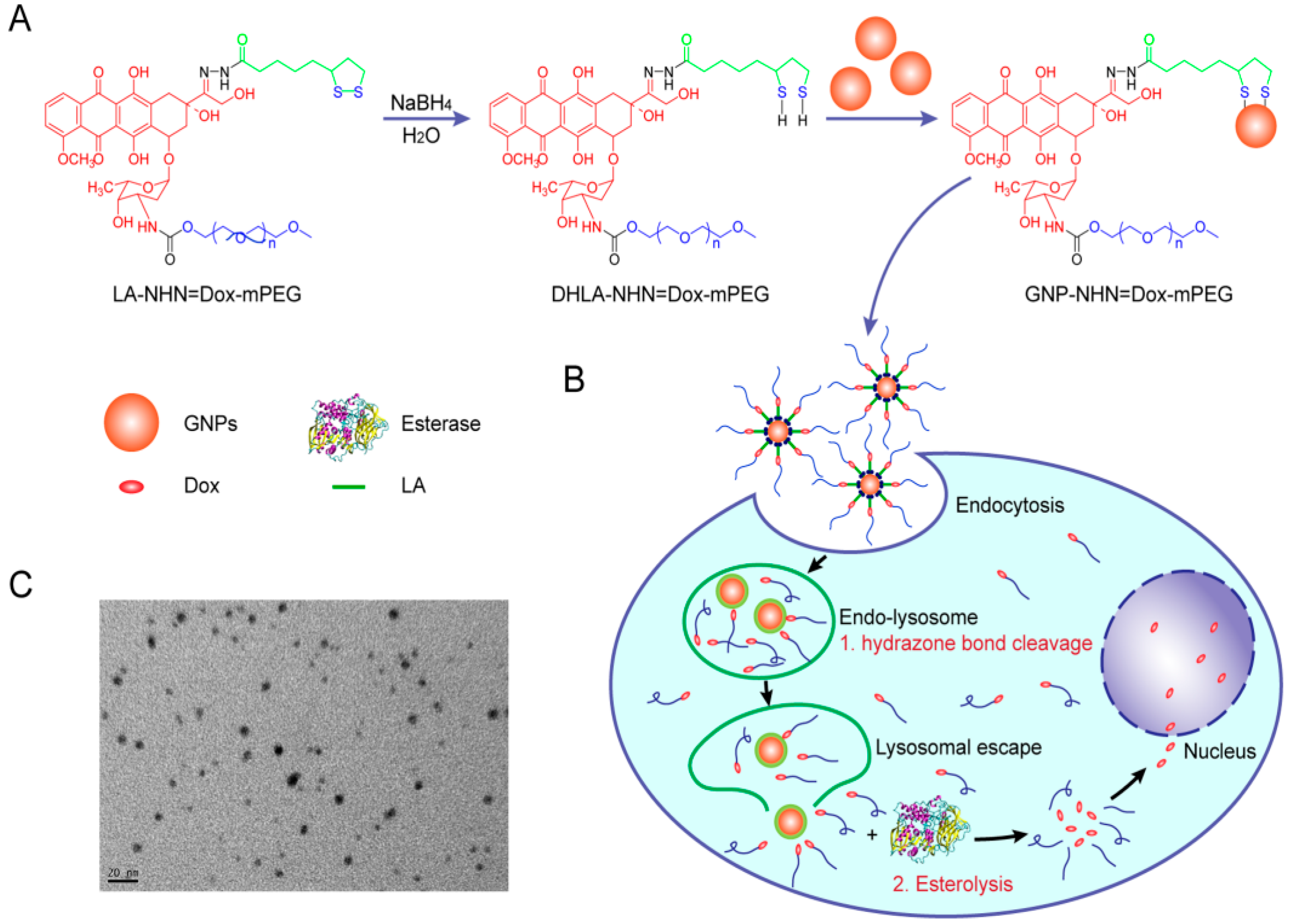
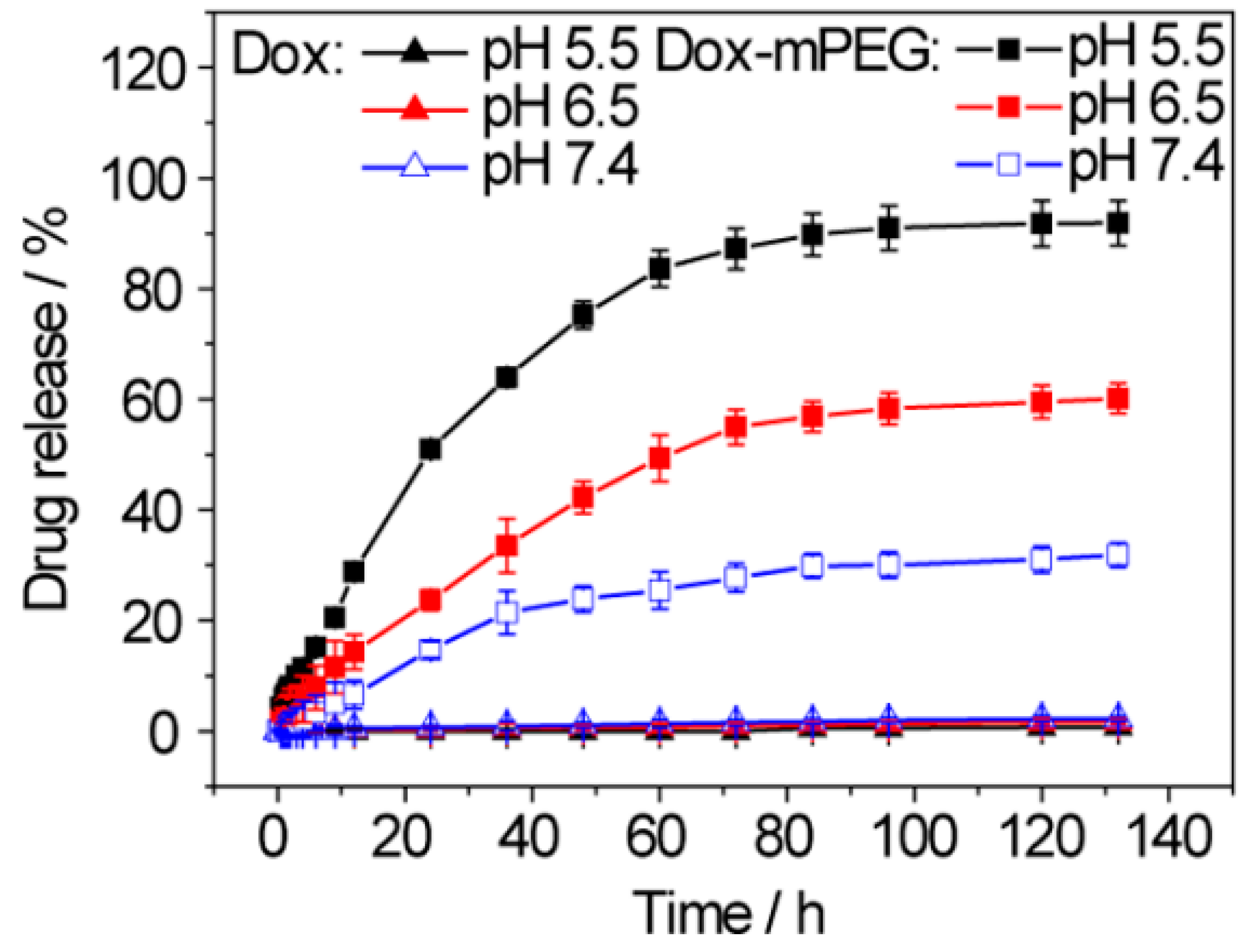
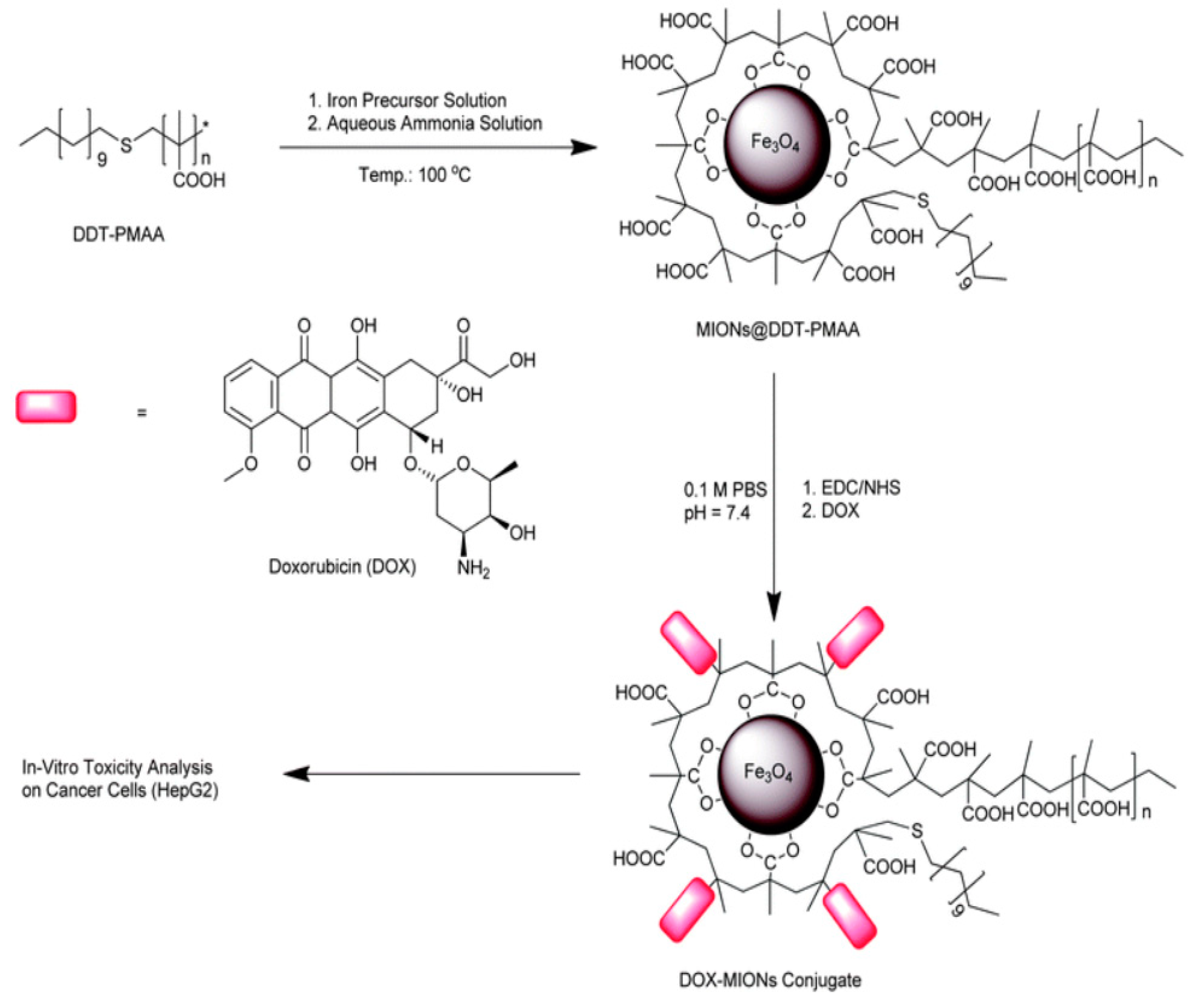
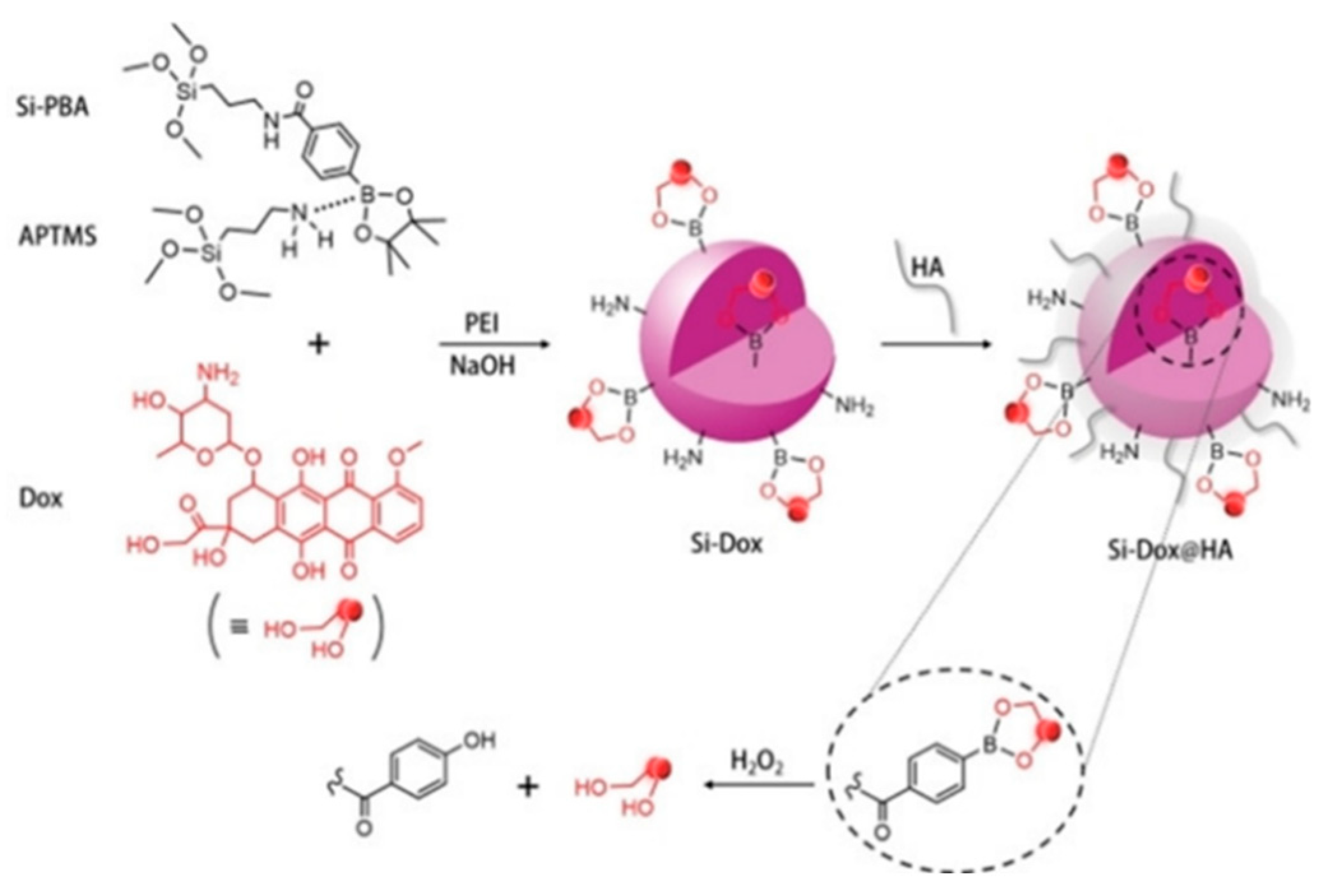

| No. | Si-PBA [mM] |
Dox [mM] |
APTMS [mM] |
NaOH [mM] |
PEI [mgmL-1] |
DLC [%] |
Diameter [nm] |
|---|---|---|---|---|---|---|---|
| 1 | 10 | 1 | 5 | 0.1 | 4 | 31.7 | 246±11 |
| 2 | 10 | 1 | 10 | 0.1 | 4 | 22.4 | 229±9 |
| 3 | 10 | 1 | 20 | 0.1 | 4 | 15.3 | 1352±162 |
| 4 | 10 | 1 | 10 | 0.1 | 2 | 40.0 | 410±12 |
| 5 | 10 | 1 | 10 | 0.1 | 8 | 14.0 | 290±3 |
| 6 | 10 | 1 | 10 | 0 | 4 | 22.6 | 517±33 |
| 7 | 10 | 1 | 10 | 1 | 4 | 17.1 | 519±20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




