Submitted:
02 December 2024
Posted:
04 December 2024
You are already at the latest version
Abstract
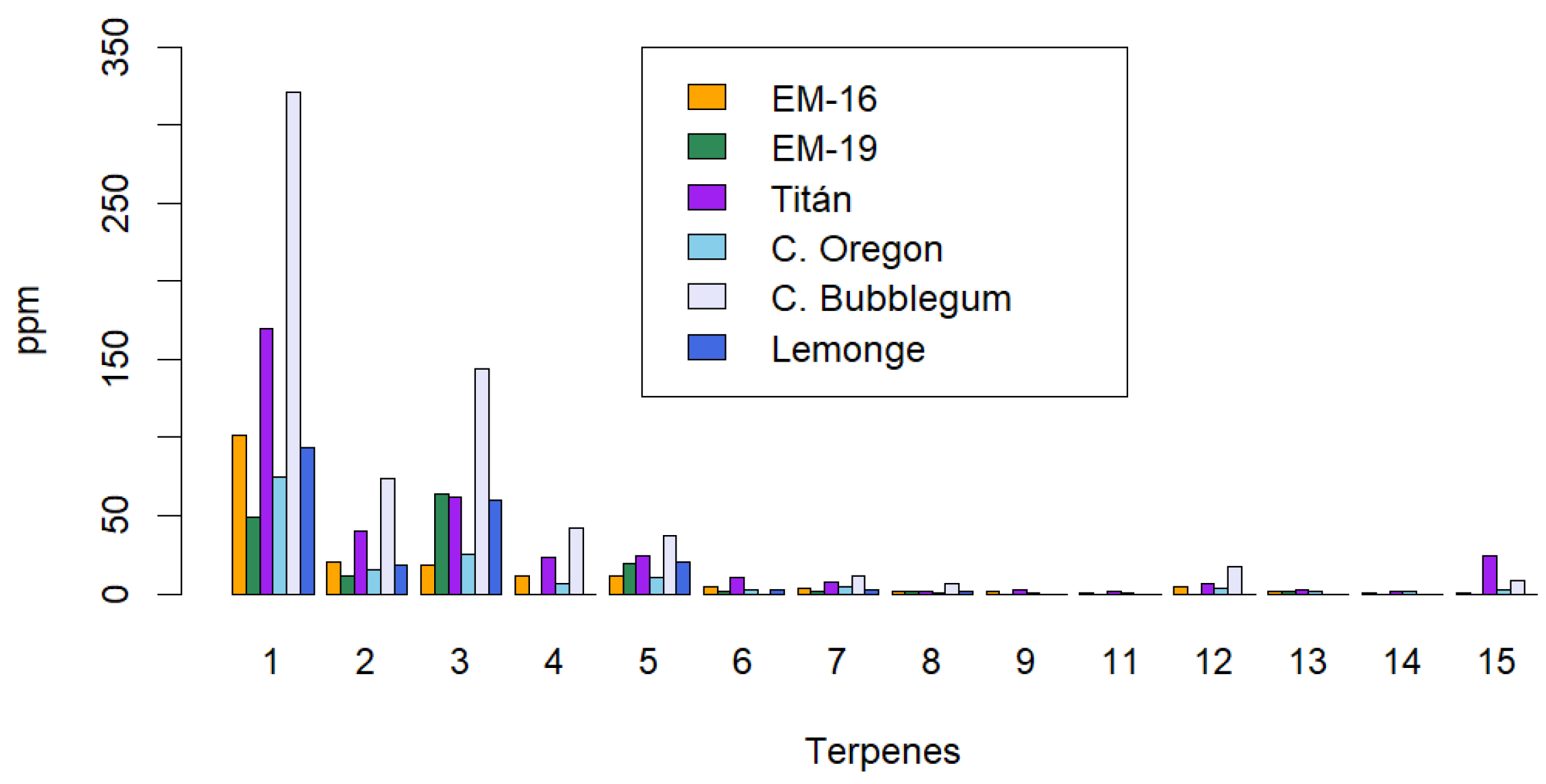
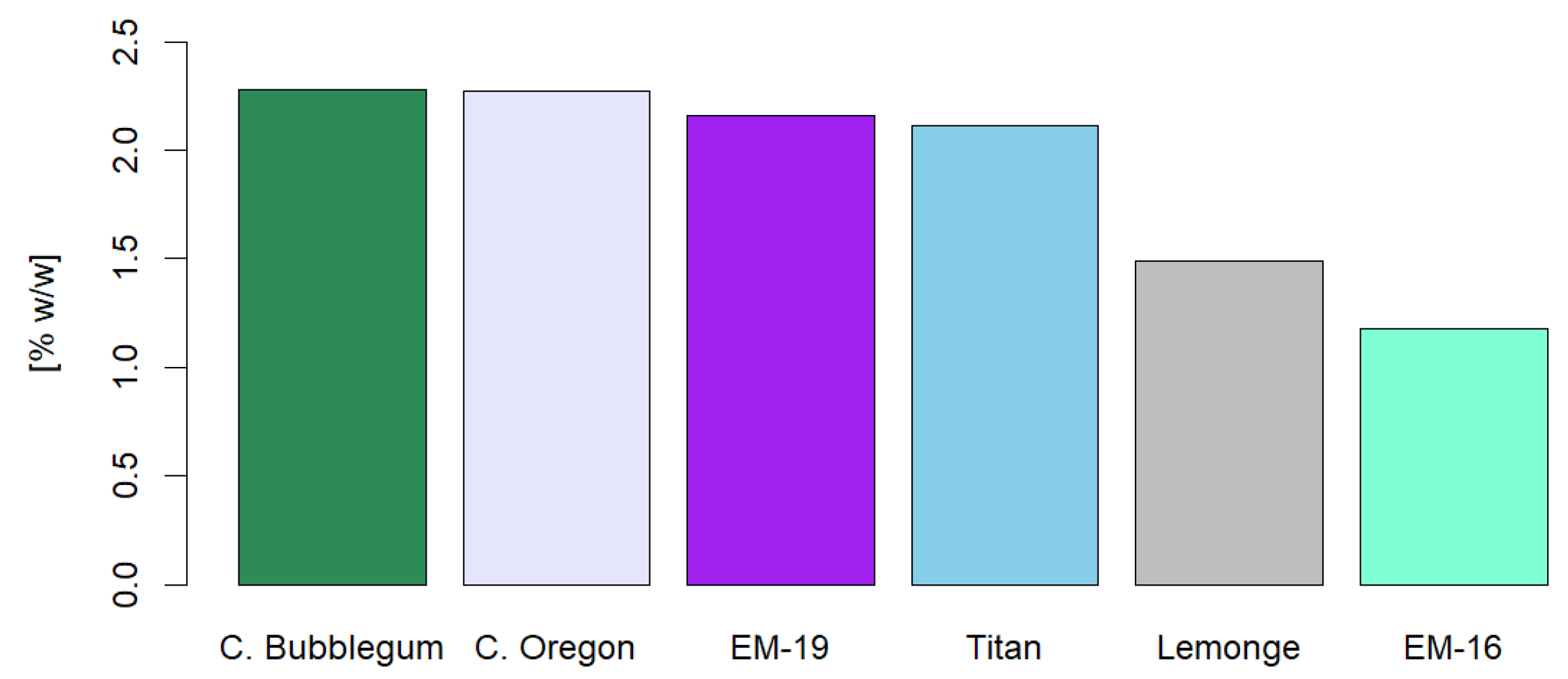
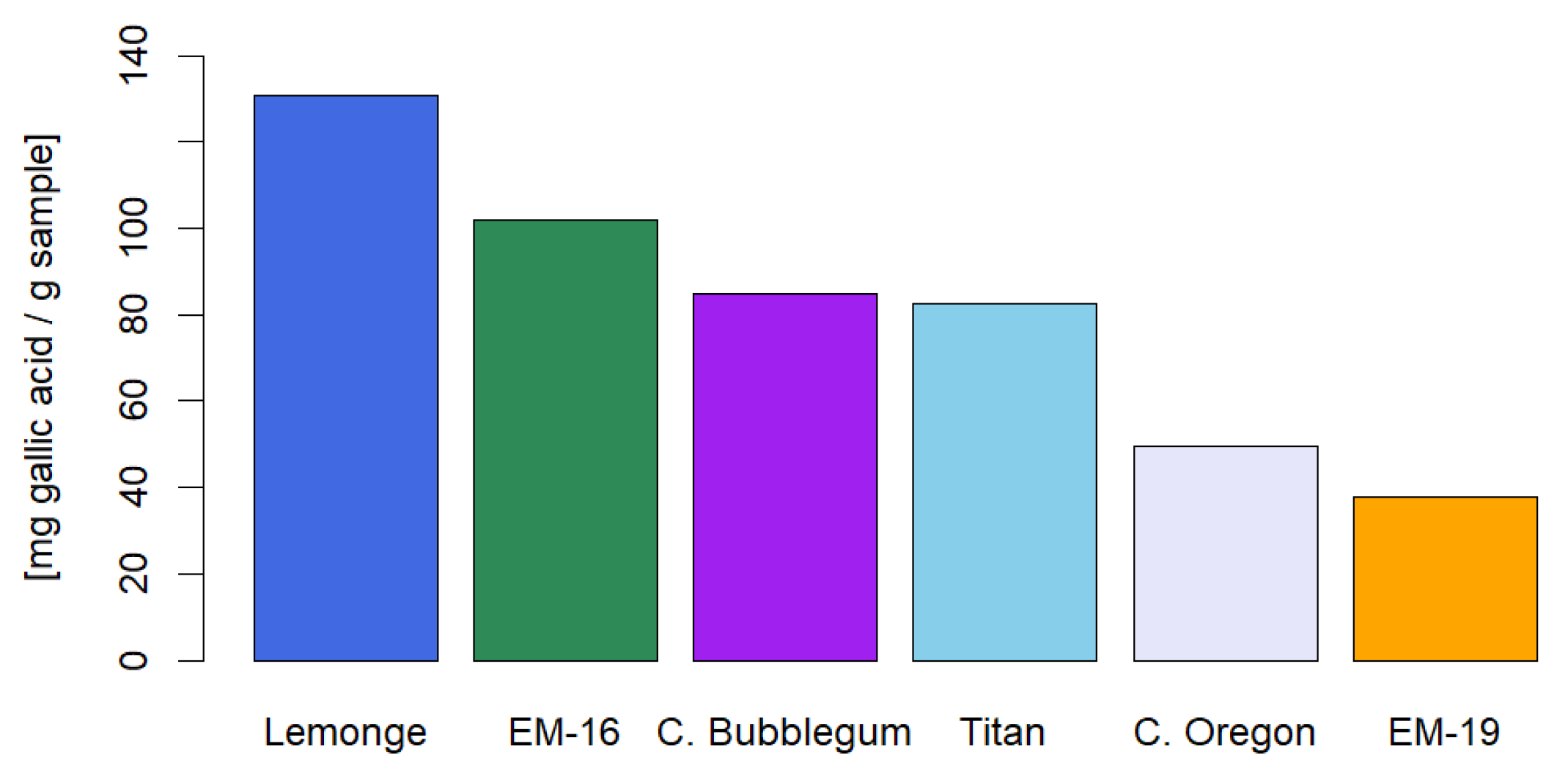
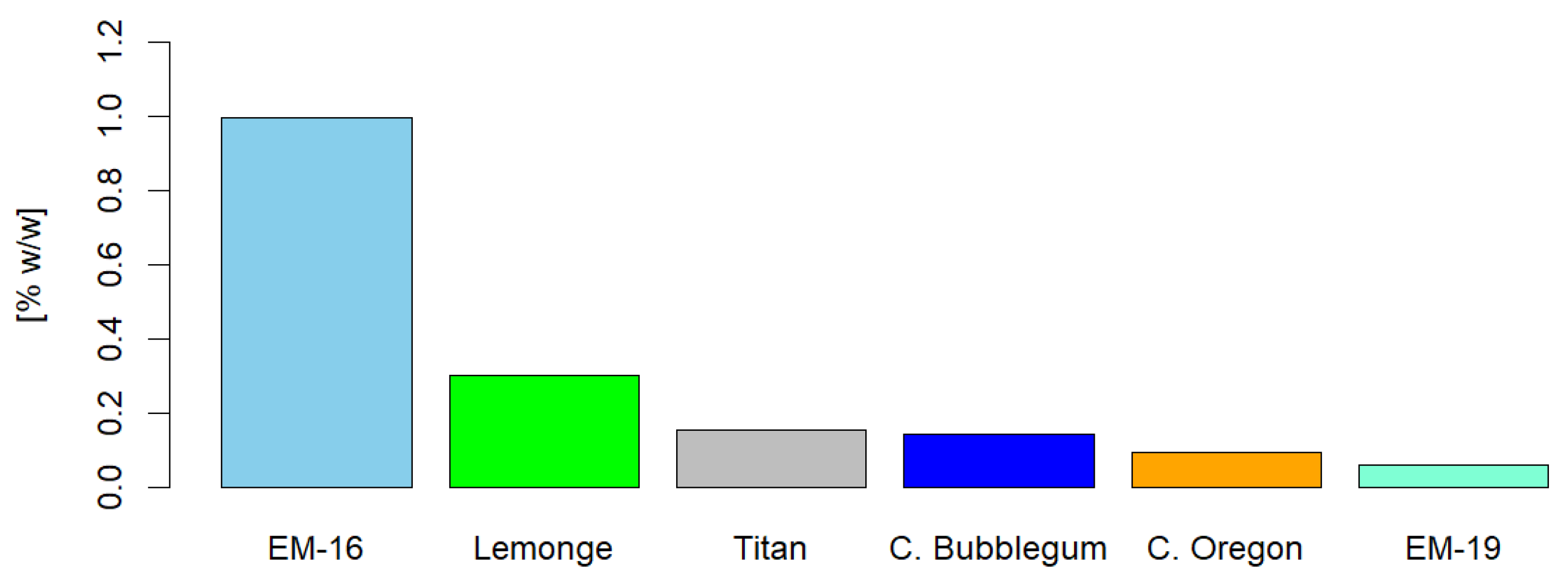
The present study focused on the extraction and characterization of phytochemical compounds from non-psychoactive varieties of Cannabis sativa L. (<1% w/w of Δ9-THC on a dry basis) cultivated in Ecuador, evaluating their therapeutic and food potential by different extraction methods. The analyses comprised six varieties: Cherry Oregon, Titan, Cherry Bubblegum, Lemonge, Medicinal Emerald 16, and Medicinal Emerald 19. The research included the identification of secondary metabolites such as cannabinoids, terpenes, flavonoids, polyphenols, and tannins, as well as a comprehensive analysis of heavy metals, microbiology, and pesticides. The extraction method and operating conditions significantly influenced the yield and quality of secondary metabolites. Three extraction methods were employed: maceration, ultrasound, and supercritical fluids with CO2. The ultrasonic extraction process presented the highest yield for the Titan variety (17.44% w/w ± 0.19%). The highest CBD concentration occurred with supercritical fluids in the Cherry Oregon variety (44.65% w/w ± 4.06%). In terms of terpenes, the Cherry Bubblegum variety presented the highest concentrations of caryophyllene (320.79 ppm), humulene (73.84 ppm), bisabolol (143.57 ppm), myrcene (42.17 ppm), and nerolidol (37.35 ppm) using supercritical fluids. Polyphenols, flavonoids, and total tannins reached their highest concentrations in Lemonge (130.91 mg/g), Cherry Bubblegum (2.28% w/w), and Medicinal Emerald 16 (0.99% w/w), respectively. Pesticide analysis confirmed the product's safety, suggesting adequate use of organic fertilizers. However, the concentration of heavy metals, specifically Pb, Cd, and Cr, exceeded the permitted limits in some varieties, highlighting the need for stricter crop controls. In conclusion, the research achieved a detailed characterization of non-psychoactive cannabis flower extracts grown at over 2450 m.a.s.l. in Ecuador, highlighting the variability in yield and composition of secondary metabolites according to the extraction method applied.
Keywords:
1. Introduction
2. Results and Discussion
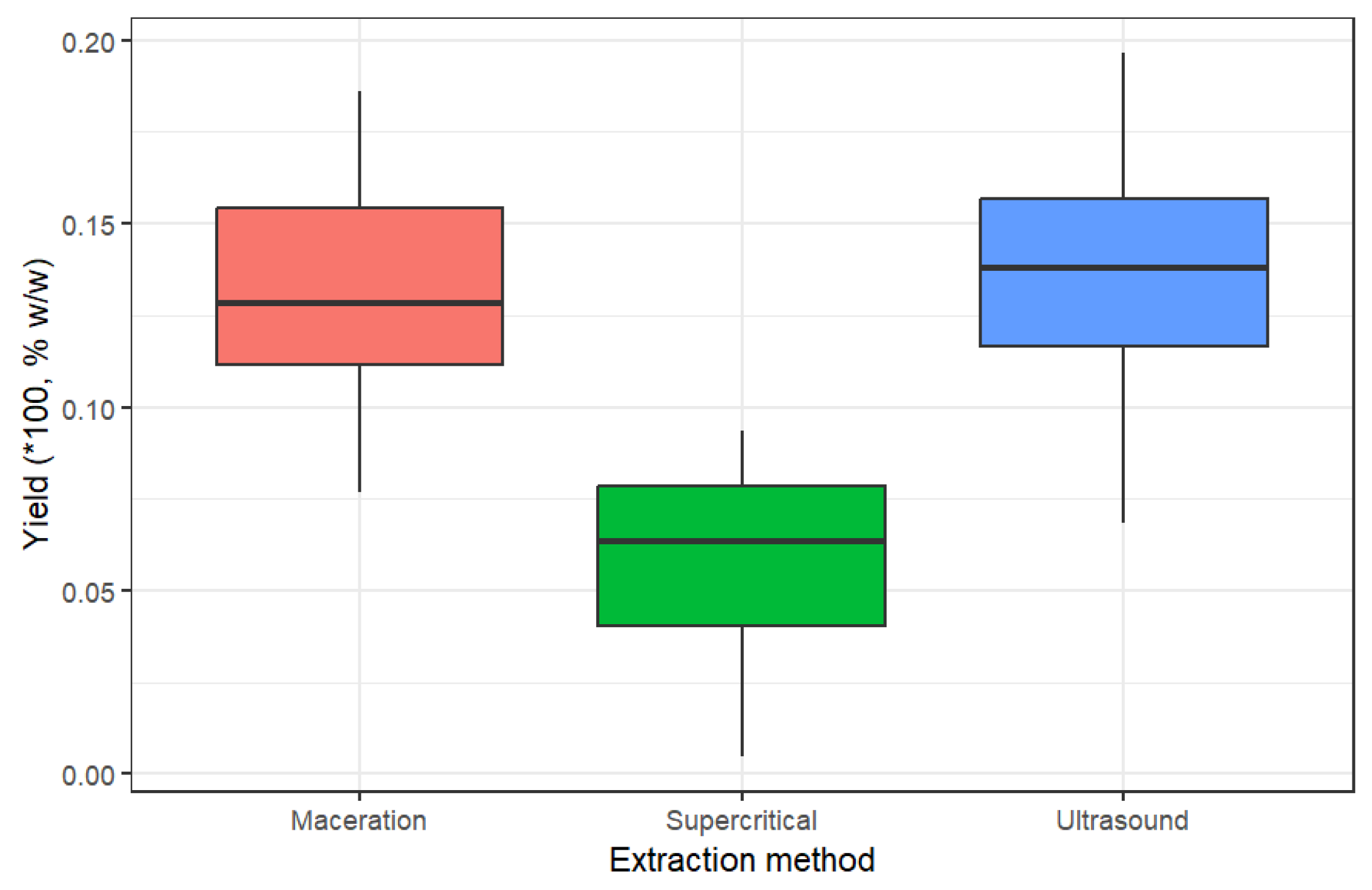
2.1. Extraction Yields for Non-Psychoactive Cannabis Flowers
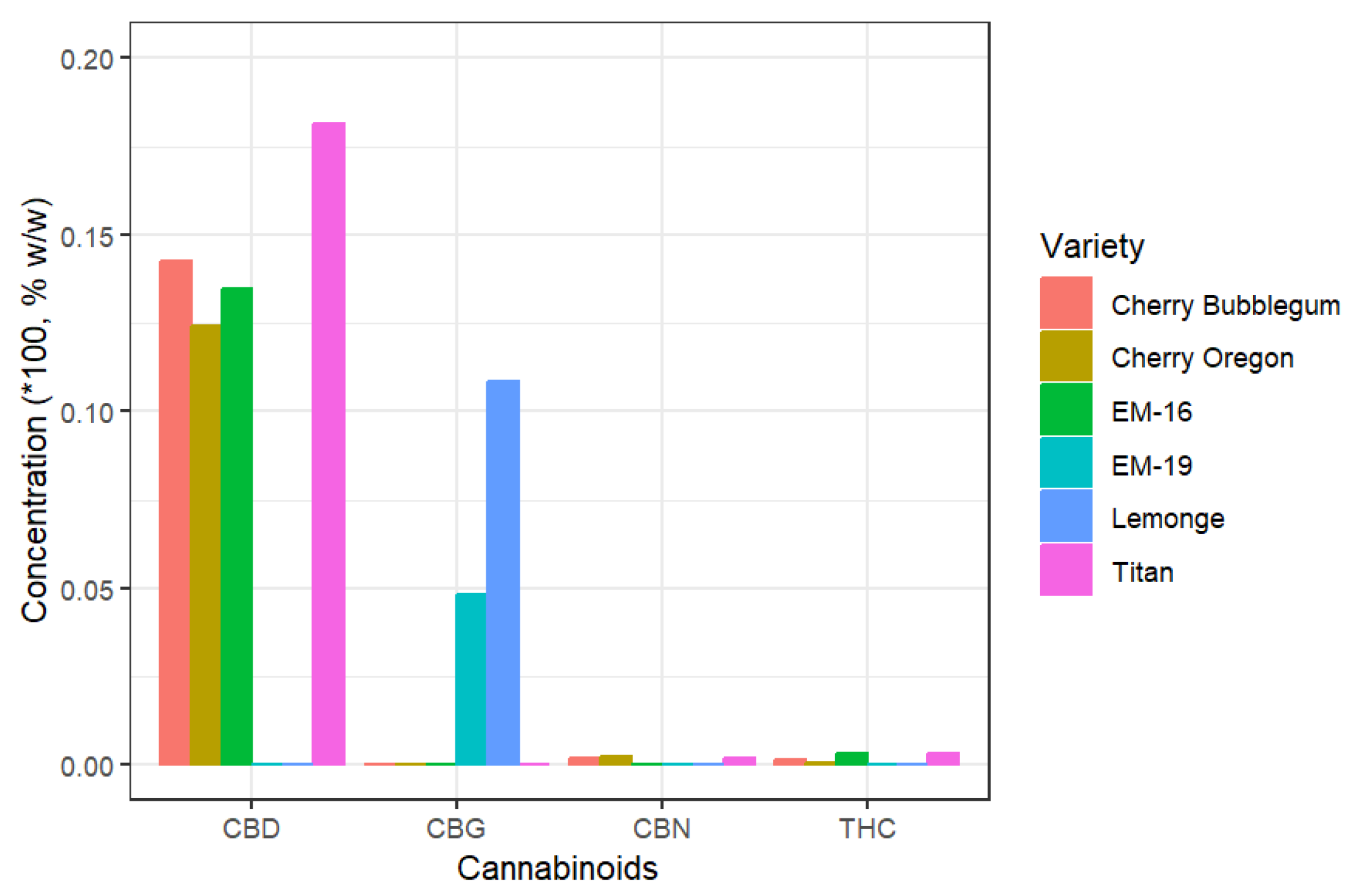
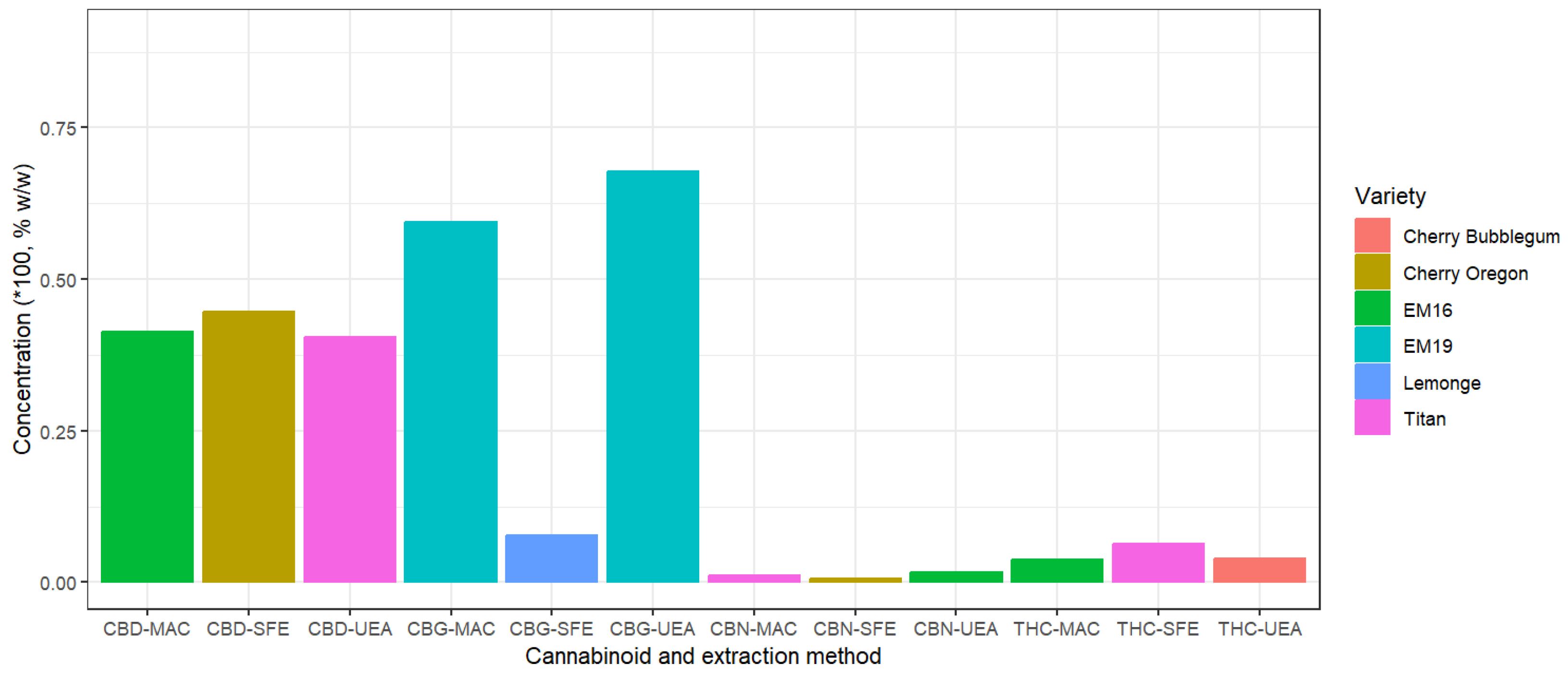
2.2. Cannabinoid Analysis in Non-Psychoactive Cannabis Flower Extracts
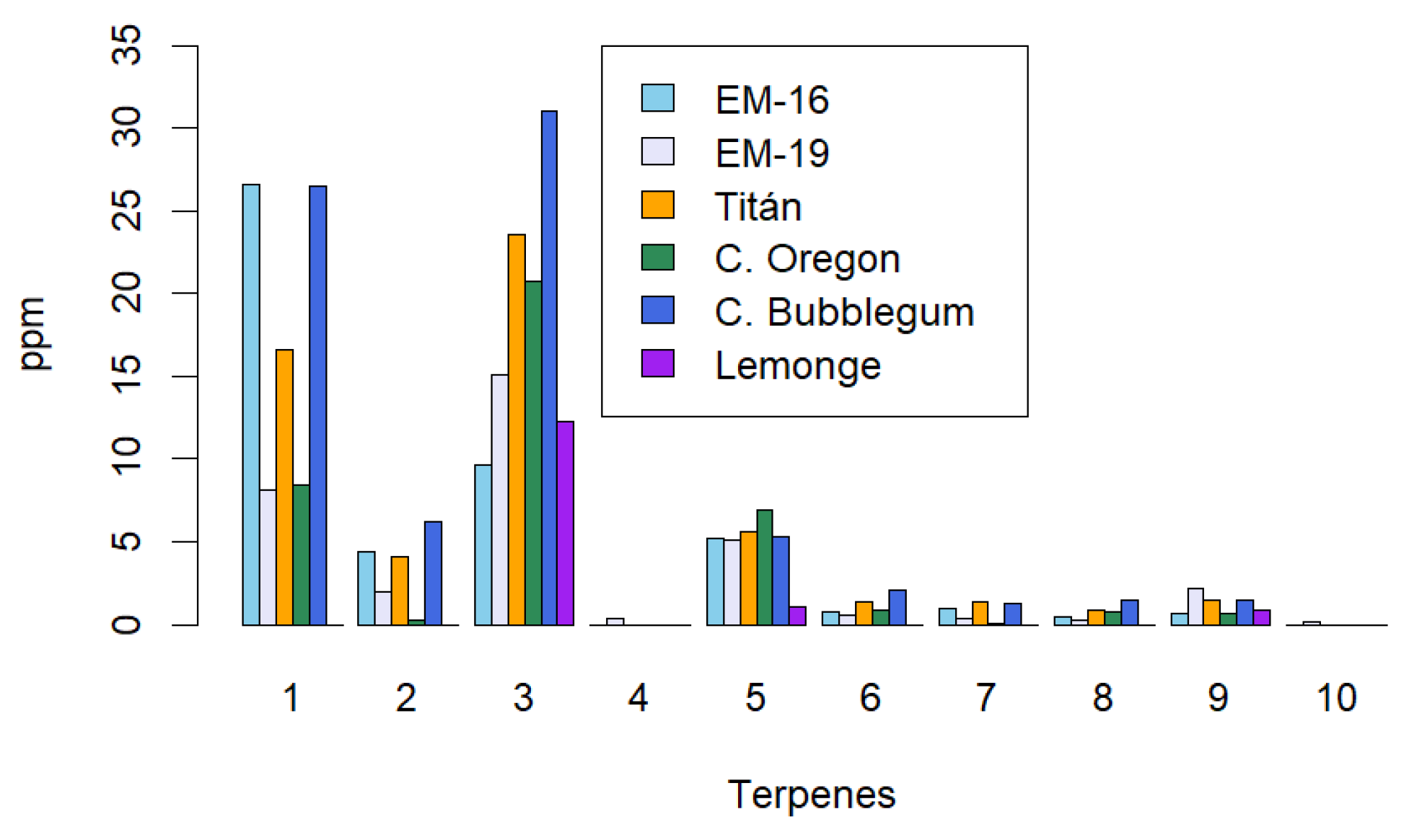
2.3. Identification of the Terpene Profile in Non-Psychoactive Cannabis Extract
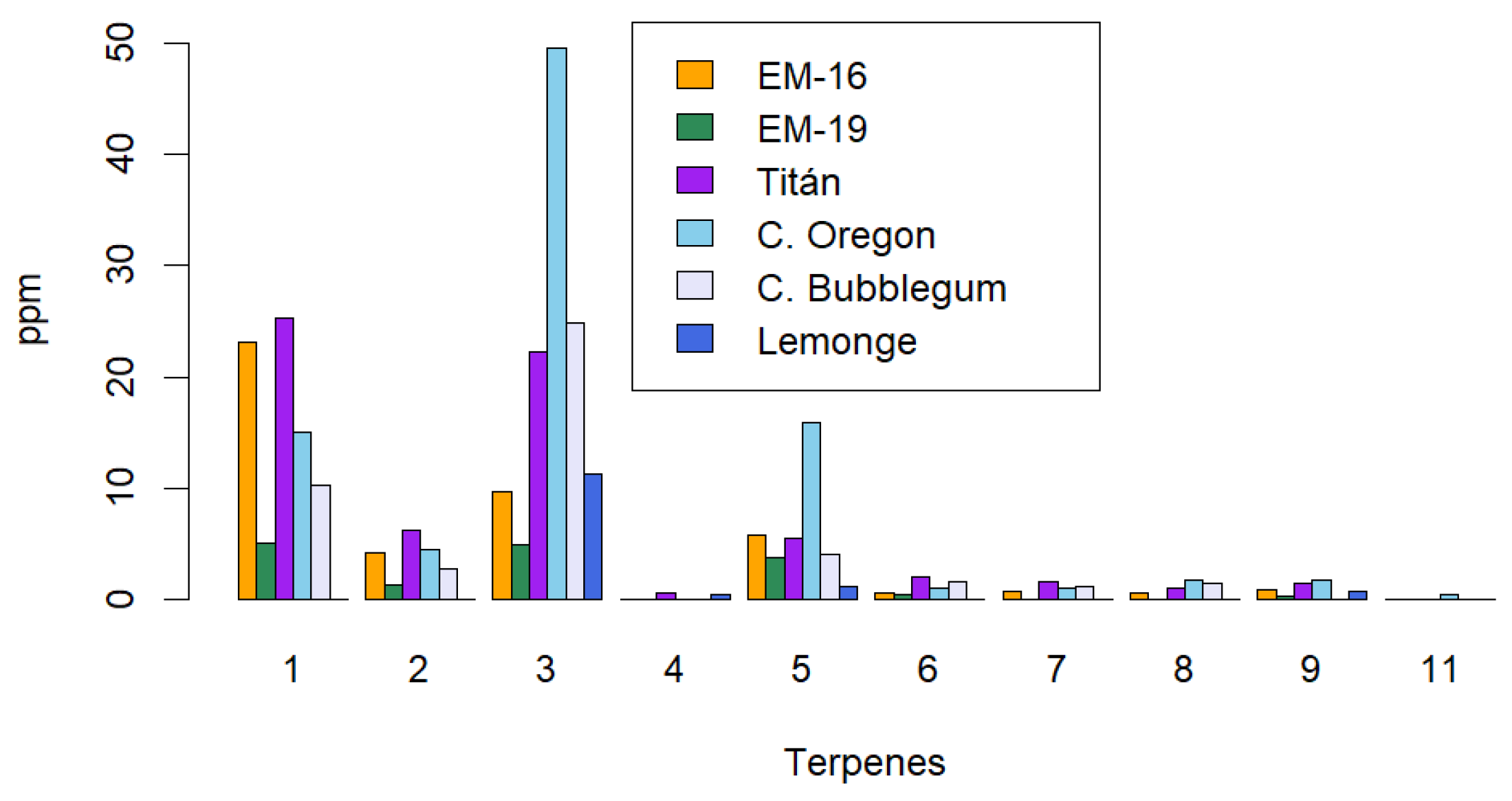
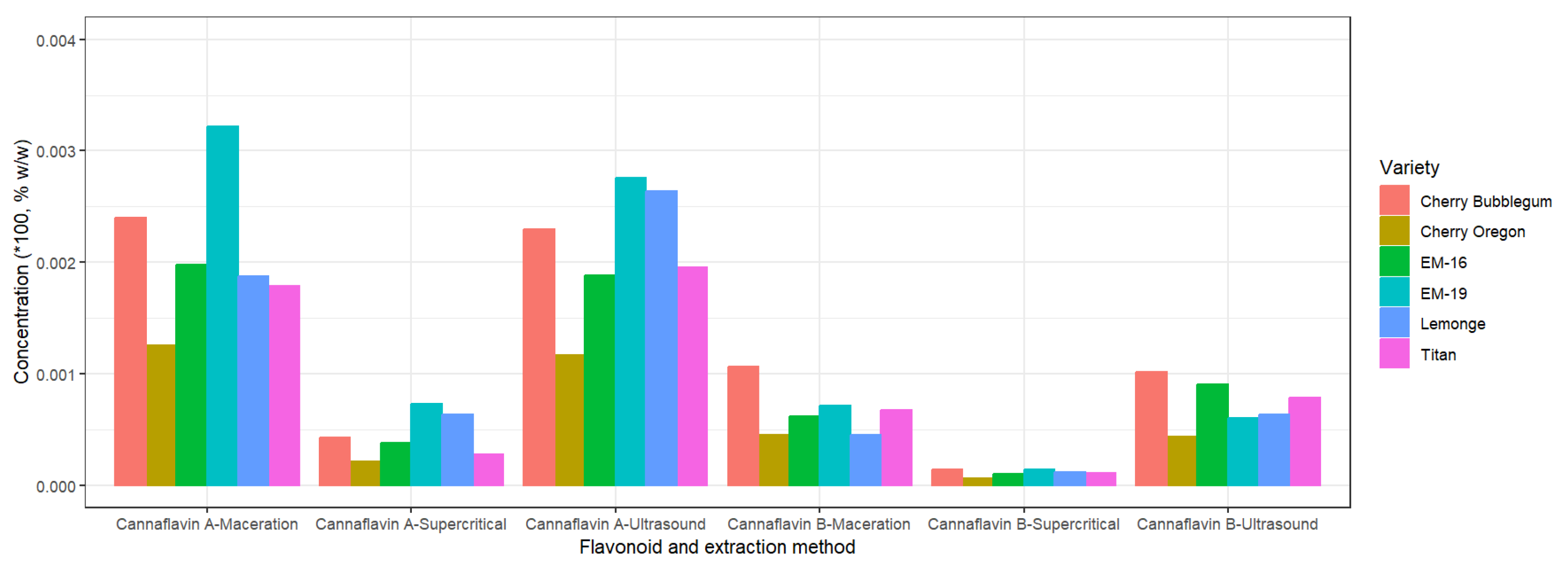
2.4. Identification of Cannaflavins, Total Flavonoids, Total Polyphenols, and Tannins
2.5. Microbiological Analysis of Non-Psychoactive Cannabis Inflorescences
| Sample | Heterotrophic bacteria (UFC/g) |
Escherichia coli (UFC/g) |
Total coliforms (UFC/g) |
Molds (UFC/g) |
Yeasts (UFC/g) |
|---|---|---|---|---|---|
| M1 | 2.50 x 1010 | 7.30 x 107 | >1.70 x 1011 | <10 | 1.50 x 1010 |
| M2 | 1.17 x 1010 | <10 | 6.00 x 1010 | <10 | 1.80 x 1010 |
| M3 | 1.50 x 1010 | <10 | 5.20 x 1010 | <10 | 1.10 x 1010 |
| M4 | 1.00 x 109 | <10 | 3.00 x 108 | <10 | 9.00 x 108 |
| M5 | 8.00 x 109 | <10 | 4.10 x 1010 | <10 | 1.90 x 1010 |
| M6 | 6.20 x 109 | <10 | 2.00 x 1010 | <10 | 5.90 x 109 |
2.6. Pesticide Analysis in Non-Psychoactive Cannabis Flowers
2.7. Identification of Heavy Metals in Non-Psychoactive Cannabis Flowers
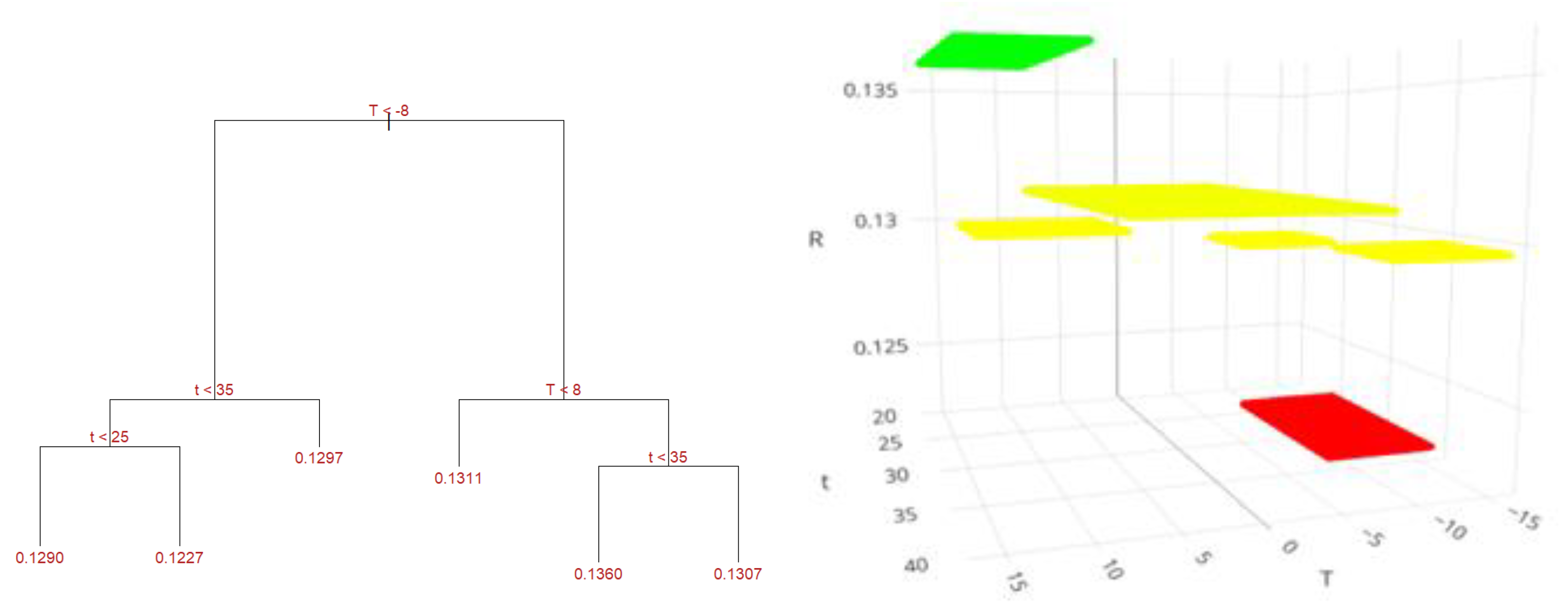
2.8. Machine Learning Predictions for Cannabis Flower Yield
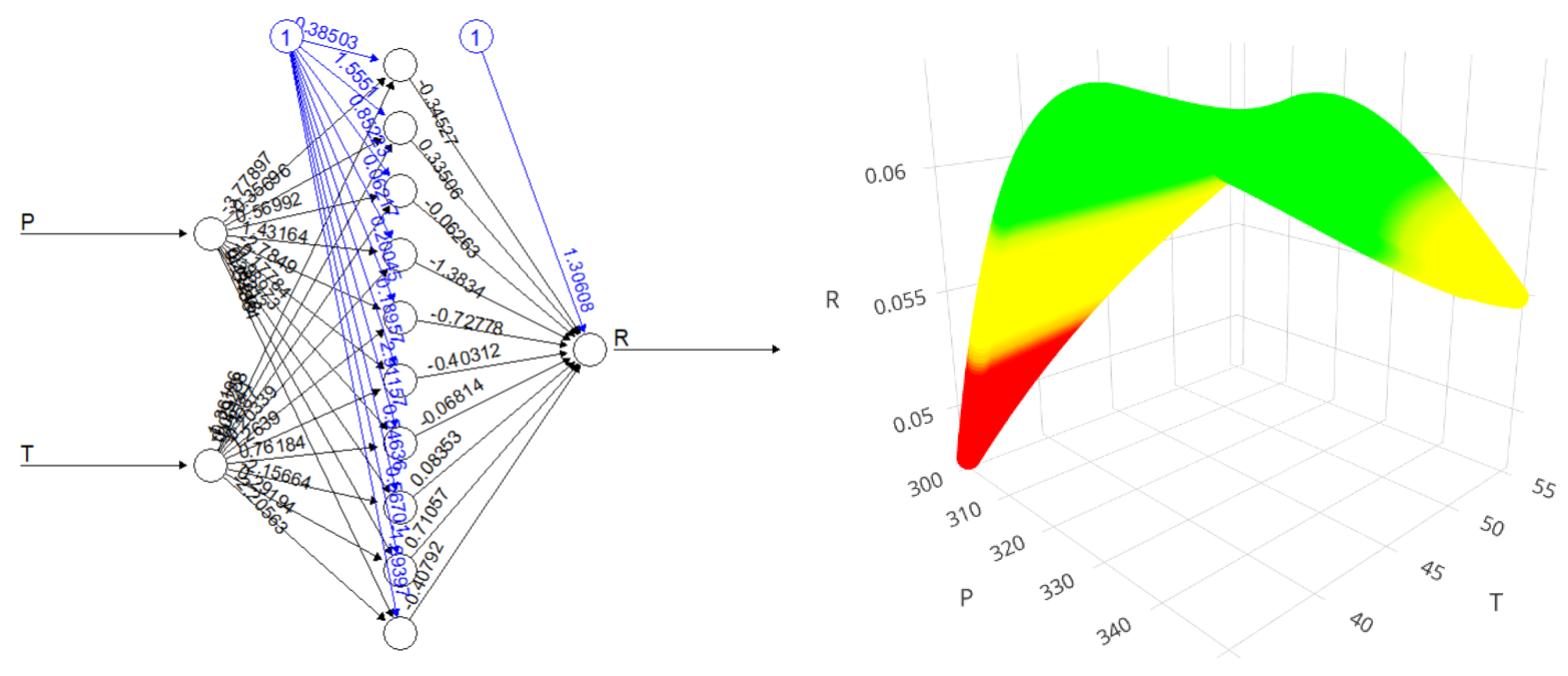
3. Materials and Methods
3.1. Materials
| Variety | Location | Tmax, °C | Tavg annual, °C | Tmin, °C | RH avg annual, % | Evapotranspiration, mm/year | Altitude, m.a.s.l. | Precipitation, mm/year |
|---|---|---|---|---|---|---|---|---|
| Cherry Oregon | Tabacundo | 20 | 13-14 | 11 | 82 | 600-650 | 2800 | 900-1000 |
| Cherry Bubblegum | Cotogchoa | 21 | 14-15 | 12 | 86 | 650-700 | 2614 | 1700-1800 |
| Titan | Cotogchoa | 21 | 14-15 | 12 | 86 | 650-700 | 2614 | 1700-1800 |
| Lemonge | Poaló | 18 | 13-14 | 9 | 82 | 650-700 | 2968 | 500-600 |
| Medicinal Emerald 16 (EM16) | San Antonio de Pichincha | 27 | 15-16 | 6 | 73 | 700-750 | 2577 | 500-600 |
| Medicinal Emerald 19 (EM19) | San Antonio de Pichincha | 27 | 15-16 | 6 | 73 | 700-750 | 2577 | 500-600 |
3.2. Methods
3.2.1. Phytocannabinoid Extraction Using Supercritical Fluid
3.2.2. Solid-Liquid Extraction (Maceration)
3.2.3. Ultrasonic-Assisted Extraction
3.2.4. Analytical Methods for Tannins, Total Flavonoids, and Total Polyphenols
3.2.5. Cannabinoids and Cannaflavins
3.2.6. Terpenes
3.2.7. Heavy Metals
3.2.8. Pesticides
3.2.9. Microbiological Testing
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- E. Small, “Classification of Cannabis sativa L.: In relation to agricultural, biotechnological, medical and recreational utilization,” in Cannabis sativa L. - Botany and Biotechnology, Springer International Publishing, 2017, pp. 1–62. [CrossRef]
- V. R. Preedy, Handbook of Cannabis and Related Pathologies: Biology, Pharmacology, Diagnosis, and Treatment. 2017.
- M. A. ElSohly, M. M. Radwan, W. Gul, S. Chandra, and A. Galal, “Phytochemistry of Cannabis sativa L,” Jan. 01, 2017. [CrossRef]
- M. M. Radwan, S. Chandra, S. Gul, and M. A. Elsohly, “Cannabinoids, phenolics, terpenes and alkaloids of cannabis,” Molecules, vol. 26, no. 9, 2021. [CrossRef]
- W. Leonard, P. Zhang, D. Ying, and Z. Fang, “Hempseed in food industry: Nutritional value, health benefits, and industrial applications,” Compr Rev Food Sci Food Saf, vol. 19, no. 1, pp. 282–308, Jan. 2020. [CrossRef]
- F. Potin and R. Saurel, “Hemp Seed as a Source of Food Proteins,” 2020. [CrossRef]
- C. Chen and Z. Pan, “Cannabidiol and terpenes from hemp – ingredients for future foods and processing technologies,” Journal of Future Foods, vol. 1, no. 2, pp. 113–127, Dec. 2021. [CrossRef]
- R. Ransing et al., “Current state of cannabis use, policies, and research across sixteen countries: cross-country comparisons and international perspectives,” 2022. [CrossRef]
- COIP, “Ley Orgánica Reformatoria Al Código Orgánico Integral Penal,” Asamblea Nacional del Ecuador, p. 24, 2019, [Online]. Available: https://www.asambleanacional.gob.ec/es/leyes-aprobadas?leyes-aprobadas=All&title=&fecha=.
- H. Gallegos, “Aplicaciones de cáñamo como alternativa rentable a la reactivación económica de Ecuador tras la pandemia de Covid-19,” Perfiles, vol. 1, no. 25, pp. 45–53, Jan. 2021. [CrossRef]
- Ministerio de Agricultura y Ganadería, “Acuerdo Ministerial No. 109,” Quito, Oct. 2020. Accessed: Oct. 22, 2021. [Online]. Available: https://www.agricultura.gob.ec/wp-content/uploads/2020/10/109-2020-1.pdf.
- ARCSA, “Normativa técnica sanitaria para regulación y control de productos terminados de uso y consumo humano que contengan cannabis no psicoactivo o cáñamo, o derivados de cannabis no psi-coactivo o cáñamo,” Quito, 2021. [Online]. Available: https://www.controlsanitario.gob.ec/wp-content/uploads/downloads/2021/02/Resolucion-ARCSA-DE-002-2021-MAFG_Normativa-Tecnica-Sanitaria-para-la-regulacion-y-control-de-productos-terminados-de-uso-y-consumo-humano-que-contengan-Cannabis-No-Psicoactivo-o-Can.
- E. Rodríguez and J. Fontaine, “Situación actual de Cannabis sativa, beneficios terapéuticos y reacciones adversas | Rodríguez-Venegas | Revista Habanera de Ciencias Médicas,” Revista Habanera de Ciencias Médicas, vol. 19, no. 6, pp. 1–17, Nov. 2020, Accessed: Oct. 22, 2021. [Online]. Available: http://www.revhabanera.sld.cu/index.php/rhab/article/view/2992.
- D. Scublinsky, “Uso de la marihuana medicinal en enfermedades neurológicas,” Rev Argent Reumatol, vol. 28, no. 3, pp. 7–11, 2017, [Online]. Available: http://revistasar.org.ar/revistas/2017/n3/3_actualizacion.pdf%0Ahttp://www.scielo.org.ar/scielo.php?script=sci_arttext&pid=S2362-36752017000300003&lng=es&nrm=iso&tlng=.
- J. M. Andrade, E. Ramírez, J. D. Cedeño, N. A. Ardila, and A. L. López, “Avances científicos y tecno-lógicos del cannabis en el campo medicinal,” Universitaria, Corporacion Dios, Minuto De, vol. 39, no. November, pp. 20–34, 2018, Accessed: Oct. 22, 2021. [Online]. Available: http://www.revistaespacios.com/a18v39n39/a18v39n39p20.pdf.
- S. O. Aloo, G. Mwiti, L. W. Ngugi, and D. H. Oh, “Uncovering the secrets of industrial hemp in food and nutrition: The trends, challenges, and new-age perspectives,” 2022. [CrossRef]
- R. Mechoulam and R. Gallily, “Cannabidiol: An Overview of Some Pharmacological Aspects,” The Journal of Clinical Pharmacology, vol. 42, no. 11, pp. 11–19, 2010. [CrossRef]
- B. F. Thomas and M. ElSholy, The Analytical Chemistry of Cannabis. Quality Assessment, Assurance, and Regulation of Medicinal Marijuana and Cannabinoid Preparations. Amsterdam: Elsevier B.V. Book Aid International, 2016.
- T. J. Jackson and S. Chakraborty, “The Cannabis sativa genetics and therapeutics relationship network: automatically associating cannabis-related genes to therapeutic properties through chemicals from cannabis literature,” J Cannabis Res, vol. 5, no. 1, Mar. 2023. [CrossRef]
- M. Monroe, “Factores que influyen en la producción de THC,” Cannabis Magazine, pp. 1–1, Sep. 2014, Accessed: Oct. 23, 2021. [Online]. Available: https://www.cannabismagazine.net/factores-que-influyen-en-la-produccion-de-thc/.
- F. Siano et al., “Comparative study of chemical, biochemical characteristic and ATR-FTIR analysis of seeds, oil and flour of the edible Fedora cultivar hemp (Cannabis sativa L.),” Molecules, vol. 24, no. 1, 2019. [CrossRef]
- R. A. Burton, M. Andres, M. Cole, J. M. Cowley, and M. A. Augustin, “Industrial hemp seed: from the field to value-added food ingredients,” Dec. 01, 2022, BioMed Central Ltd. [CrossRef]
- H. M. S. Al Ubeed, D. J. Bhuyan, M. A. Alsherbiny, A. Basu, and Q. V. Vuong, “A Comprehensive Re-view on the Techniques for Extraction of Bioactive Compounds from Medicinal Cannabis,” Molecules, vol. 27, no. 3, pp. 1–18, 2022. [CrossRef]
- M. Radoiu, H. Kaur, A. Bakowska-Barczak, and S. Splinter, “Microwave-Assisted Industrial Scale Cannabis Extraction,” Technologies (Basel), vol. 8, no. 3, pp. 1–16, 2020. [CrossRef]
- S. Jokić et al., “Terpenes and Cannabinoids in Supercritical CO2 Extracts of Industrial Hemp Inflorescences: Optimization of Extraction, Antiradical and Antibacterial Activity,” Pharmaceuticals, vol. 15, no. 9, 2022. [CrossRef]
- C. López-Olmos, M. T. García-Valverde, J. Hidalgo, C. Ferrerio-Vera, and V. Sánchez de Medina, “Comprehensive comparison of industrial cannabinoid extraction techniques: Evaluation of the most relevant patents and studies at pilot scale,” Frontiers in Natural Products, vol. 1, 2022. [CrossRef]
- L. J. Rovetto and N. V. Aieta, “Supercritical carbon dioxide extraction of cannabinoids from Cannabis sativa L.,” Journal of Supercritical Fluids, vol. 129, pp. 16–27, Nov. 2017. [CrossRef]
- J. Wilson, T. Simpson, and K. Spelman, “Total cannabidiol (CBD) concentrations and yields from traditional extraction methods: Percolation vs. maceration,” Front Pharmacol, vol. 13, Oct. 2022. [CrossRef]
- M. Gallo, A. Formato, M. Ciaravolo, G. Formato, and D. Naviglio, “Study of the kinetics of extraction process for the production of hemp inflorescences extracts by means of conventional maceration (CM) and rapid solid-liquid dynamic extraction (RSLDE),” Separations, vol. 7, no. 2, 2020. [CrossRef]
- M. M. Lewis, Y. Yang, E. Wasilewski, H. A. Clarke, and L. P. Kotra, “Chemical Profiling of Medical Cannabis Extracts,” ACS Omega, vol. 2, no. 9, pp. 6091–6103, 2017. [CrossRef]
- L. Nahar, A. Onder, and S. D. Sarker, “A review on the recent advances in HPLC, UHPLC and UPLC analyses of naturally occurring cannabinoids (2010–2019),” Phytochemical Analysis, vol. 31, no. 4, pp. 413–457, 2020. [CrossRef]
- United Nations, “RECOMMENDED METHODS FOR THE IDENTIFICATION AND ANALYSIS OF CANNABIS AND CANNABIS PRODUCTS NATIONAL DRUG ANALYSIS LABORATORIES,” Viena, Mar. 2022.
- M. D. Kleinhenz et al., “Nutrient concentrations, digestibility, and cannabinoid concentrations of industrial hemp plant components,” Applied Animal Science, vol. 36, no. 4, 2020. [CrossRef]
- L. S. Jiménez Álvarez, N. Vega, E. D. Capa Mora, N. del C. Fierro Jaramillo, and P. Q. Miguitama, “Learning teaching styles and strategies of University Students of Soil Science,” Revista Electronica de Investigacion Educativa, vol. 21, no. 1, 2019. [CrossRef]
- C. Scholz, M. M. Madry, T. Kraemer, and M. R. Baumgartner, “LC–MS-MS Analysis of ∆9-THC, CBN and CBD in Hair: Investigation of Artifacts,” J Anal Toxicol, vol. 46, no. 5, 2022. [CrossRef]
- J. W. King, “The relationship between cannabis/hemp use in foods and processing methodology,” 2019. [CrossRef]
- M. Valizadehderakhshan, A. Shahbazi, M. Kazem-Rostami, M. S. Todd, Bhowmik, and Arnab, “Ex-traction of Cannabinoids fromCannabis sativaL.(Hemp)—Review,” Agriculture, vol. 11, no. 384, 2021.
- D. R. Grijó, I. A. Vieitez Osorio, and L. Cardozo-Filho, “Supercritical extraction strategies using CO2 and ethanol to obtain cannabinoid compounds from Cannabis hybrid flowers,” Journal of CO2 Utilization, vol. 28, no. May, pp. 174–180, 2018. [CrossRef]
- D. De Vita et al., “Comparison of different methods for the extraction of cannabinoids from cannabis,” Nat Prod Res, vol. 34, no. 20, pp. 2952–2958, 2020. [CrossRef]
- S. R. Sommano, C. Chittasupho, W. Ruksiriwanich, and P. Jantrawut, “The Cannabis Terpenes,” Dec. 01, 2020, MDPI. [CrossRef]
- D. Jin, K. Dai, Z. Xie, and J. Chen, “Secondary Metabolites Profiled in Cannabis Inflorescences, Leaves, Stem Barks, and Roots for Medicinal Purposes,” Sci Rep, vol. 10, no. 1, 2020. [CrossRef]
- J. Bueno, E. Leuer, M. Kearney, E. H. Green, and E. A. Greenbaum, “The preservation and augmentation of volatile terpenes in cannabis inflorescence,” J Cannabis Res, vol. 2, no. 1, 2020. [CrossRef]
- R. Ascrizzi, M. Iannone, G. Cinque, A. Marianelli, L. Pistelli, and G. Flamini, “‘Hemping’ the drinks: Aromatizing alcoholic beverages with a blend of Cannabis sativa L. flowers,” Food Chem, vol. 325, 2020. [CrossRef]
- L. B. Eddin et al., “Health Benefits, Pharmacological Effects, Molecular Mechanisms, and Therapeutic Potential of α-Bisabolol,” 2022. [CrossRef]
- H. Sovová, R. P. Stateva, and M. Koptová, “Measurement and correlation of α-bisabolol solubility in near critical carbon dioxide,” J Chem Eng Data, vol. 58, no. 5, 2013. [CrossRef]
- E. D. Rocha et al., “Qualitative terpene profiling of Cannabis varieties cultivated for medical purposes,” Rodriguesia, vol. 7, 2020. [CrossRef]
- D. Y. Hoo et al., “Ultrasonic cavitation: An effective cleaner and greener intensification technology in the extraction and surface modification of nanocellulose,” 2022. [CrossRef]
- T. Nuutinen, “Medicinal properties of terpenes found in Cannabis sativa and Humulus lupulus,” Sep. 05, 2018, Elsevier Masson SAS. [CrossRef]
- D. Cox-Georgian, N. Ramadoss, C. Dona, and C. Basu, “Therapeutic and medicinal uses of terpenes,” in Medicinal Plants: From Farm to Pharmacy, 2019. [CrossRef]
- M. L. Barrett, A. M. Scutt, and F. J. Evans, “Cannflavin A and B, prenylated flavones from Cannabis sativa L.,” Experientia, vol. 42, no. 4, 1986. [CrossRef]
- R. Pavlovic et al., “Phytochemical and Ecological Analysis of Two Varieties of Hemp (Cannabis sativa L.) Grown in a Mountain Environment of Italian Alps,” Front Plant Sci, vol. 10, 2019. [CrossRef]
- L. Barrientos Ramírez et al., “Contenido de polifenoles y capacidad antioxidante de Physalis cheno-podifolia Lam. silvestre y cultivo,” Rev Mex Cienc For, vol. 10, no. 51, 2019. [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. “Flavonoids: An overview,” Jan. 08, 2016, Cambridge University Press. [CrossRef]
- K. Murti, M. A. Panchal, V. Gajera, and J. Solanki, “Pharmacological Properties of Matricaria recutita: A Review,” Pharmacologia, vol. 3, no. 8, 2012. [CrossRef]
- Tomko, A.M.; Whynot, E.G.; Ellis, L.D.; Dupré, D.J. “Anti-cancer potential of cannabinoids, terpenes, and flavonoids present in cannabis,” Jul. 01, 2020, MDPI AG. [CrossRef]
- M. Quiñones, M. Miguel, and A. Aleixandre, “The polyphenols, naturally occurring compounds with beneficial effects on cardiovascular disease.,” 2012.
- J. J. Buenrostro-Figueroa et al., “Improved Extraction of High Value-Added Polyphenols from Pomegranate Peel by Solid-State Fermentation,” Fermentation, vol. 9, no. 6, 2023. [CrossRef]
- Z. Tong, W. He, X. Fan, and A. Guo, “Biological Function of Plant Tannin and Its Application in Animal Health,” 2022. [CrossRef]
- L. U. Thompson, “Potential health benefits and problems associated with antinutrients in foods*,” 1993.
- N. Allocati, M. Masulli, M. F. Alexeyev, and C. Di Ilio, “Escherichia coli in Europe: An overview,” 2013. [CrossRef]
- E. Aysanew and D. Alemayehu, “Integrated management of ginger bacterial wilt (Ralstonia solanacearum) in Southwest Ethiopia,” Cogent Food Agric, vol. 8, no. 1, 2022. [CrossRef]
- M. Espín, “Evaluación de los efectos de la contaminación ambiental, en la productividad de los cultivos agrícolas, en la parroquia de Tumbaco.,” Investigación y Desarrollo, Quito, Jul. 2016.
- C. Taco, G. Vistín, V. Rosero, O. López, and W. Fonseca, “Las actividades productivas y su relación con la contaminación del agua de la Microcuenca Negroyacu, en Guaranda, Ecuador.,” Revista Ciencia UNEMI, vol. No. 10, pp. 88–97, 2017.
- D. Caplan, M. Dixon, and Y. Zheng, “Optimal rate of organic fertilizer during the vegetative-stage for cannabis grown in two coir-based substrates,” HortScience, vol. 52, no. 9, 2017. [CrossRef]
- M. Kuhn and K. Johnson, Applied Predictive Modeling. New York: Springer, 2013. [CrossRef]
- V. R. Benalcázar-Rojas, W. J. Yambay-Vallejo, and E. P. Herrera-Granda, “Multivariate Analysis for Prediction of Splitting Tensile Strength in Concrete Paving Blocks,” Applied Sciences (Switzerland), vol. 13, no. 19, 2023. [CrossRef]
- S. Sheather, A Modern Approach to Regression with R. in Springer Texts in Statistics. New York, NY: Springer New York, 2009. [CrossRef]
- Géron, A. Hands-On Machine Learning with Scikit-Learn, Keras, and TensorFlow, Second. Canada: Kristen Brown, 2019. [Online]. Available: http://oreilly.com.
- M. Wang et al., “Decarboxylation Study of Acidic Cannabinoids: A Novel Approach Using Ul-tra-High-Performance Supercritical Fluid Chromatography/Photodiode Array-Mass Spectrometry,” Cannabis Cannabinoid Res, vol. 1, no. 1, pp. 262–271, Jan. 2016. [CrossRef]
- S. Rochfort et al., “Utilisation of Design of Experiments Approach to Optimise Supercritical Fluid Ex-traction of Medicinal Cannabis,” Sci Rep, vol. 10, no. 1, pp. 1–7, 2020. [CrossRef]
- P. W. Addo et al., “Microwave- and Ultrasound-Assisted Extraction of Cannabinoids and Terpenes from Cannabis Using Response Surface Methodology,” Molecules, vol. 27, no. 24, Mar. 2022. [CrossRef]
- I. A. Herrera-Fuentes, K. L. Quimis-Ponce, N. A. Sorroza-Rojas, F. S. García-Larreta, W. Mariscal-Santi, and R. E. Mariscal-Garcia, “Determinación de Taninos y Cumarinas presente en la planta tres filos,” Polo del Conocimiento, vol. 2, no. 7, 2017. [CrossRef]
- W. Ortíz Fernández, Y. Aguilera, J. Rodríguez, D. M. Guzmán Mayancha, H. M. Cobo Salinas, and L. R. Bravo Sánchez, “Desarrollo y Validación de Técnicas Espectrofotométricas para la Determinación de Flavonoides Totales, Basada en Quercetina, en las Hojas de Psidium guajava L.,” Revista Amazónica. Ciencia y Tecnología, vol. 5, no. 3, 2016. [CrossRef]
- E. García, I. Fernández, and A. Fuentes, “Determinación de polifenoles totales por el método de Folin- Ciocalteu,” Etsiamn, 2015.
- L. Vaclavik et al., “Quantitation of cannabinoids in cannabis dried plant materials, concentrates, and oils using liquid chromatography-diode array detection technique with optional mass spectrometric detection: Single-laboratory validation study, first action 2018.11,” J AOAC Int, vol. 102, no. 6, 2019. [CrossRef]
- P. Lapwibunsuk, P. Wongphichai, y S. Lapwibunsuk, "Determinación del contenido de cannabinoides en bebidas de cáñamo mediante la técnica de cromatografía líquida de ultra alto rendimiento y espectrometría de masas en tándem", Metri, Journal of Applied Science. Departamento de Servicios Científicos, vol. 12, no. 1, 2023. [CrossRef]
- K. Rome and A. McIntyre, “Intelligent use of relative response factors in gas chromatography-flame ionisation detection.,” Chromatography Today, Macclesfield, UK, pp. 52–56, Jun. 2012. Accessed: May 12, 2024. [Online]. Available: https://www.chromatographytoday.com/article/gc-mdgc/32/astrazeneca/intelligent-use-of-relative-responsenbspfactors-in-gas-chromatography-flame-ionisation-detection/1205.
- AOAC, “AOAC Official Method 999.10 Lead, Cadmium, Zinc, Cooper, and Iron in Foods,” Official methods of analysis of AOAC International, 2008.
- Principle, A. “AOAC Official Method 2015.01 Heavy Metals in Food,” J AOAC Int, 2015.
- M. E. Calcina-Benique, L. E. Calcina-Rondán, F. R. Huaraya-Chambi, A. R. Salas-Camargo, and K. Tejada-Meza, “Arsenic in groundwater of the Callacame river basin, and its impact on agricultural soils in Desaguadero, Puno-Perú,” DYNA (Colombia), vol. 89, no. 221, 2022. [CrossRef]
- Caruso, A.; Santoro, M. “Detection of Organochlorine Pesticides by GC-ECD Following U.S. EPA Method 8081,” Detection of Organochlorine Pesticides by GC-ECD Following U.S. EPA Method 8081, 2014.
- D. Schwantes, A. Celso Gonçalves, É. Conradi Junior, M. A. Campagnolo, and J. Zimmermann, “De-termination of CHLORPYRIFOS by GC/ECD in water and its sorption mechanism study in a RHODIC FERRALSOL,” J Environ Health Sci Eng, vol. 18, no. 1, 2020. [CrossRef]
- L. I. N. Ezemonye, “Concentrations and distribution of Polychlorinated biphenyls (PCBs) in aquatic environment of Niger Delta of Nigeria (water and sediments),” Discov Innov, vol. 18, no. 2, 2006. [CrossRef]
- AOAC, Association of official analytical chemists., G. W. Latimer, and William. Horwitz, Official methods of analysis of AOAC international. AOAC International, 2006.
- I. Standard, “ISO 7218,” Reference number ISO, 2007.
- H. Kodaka, S. Mizuochi, H. Teramura, and T. Nirazuka, “Comparison of the Compact Dry EC with the most probable number method (AOAC official method 966.24) for enumeration of Escherichia coll and coliform bacteria in raw meats Performance-Tested MethodSM 110402,” J AOAC Int, vol. 89, no. 1, 2006. [CrossRef]
- B. O. Obong’O, G. Ayodo, F. Kawaka, and M. K. Adalla, “Fungi and Aflatoxin Levels in Traditionally Processed Cassava (Manihot esculenta Crantz) Products in Homa Bay County, Kenya,” Int J Microbiol, vol. 2020, 2020. [CrossRef]
- Geron, A. Hands-On Machine Learning with Scikit-Learn, Keras, and TensorFlow: Concepts, Tools, and Techniques to Build Intelligent Systems, 2nd ed. O’Reilly Media, Inc., 2019.
- M. Kuhn and K. Johnson, Applied Predictive Modeling. 2013. [CrossRef]
- Breiman L, Friedman JH, Olshen RA, and Stone CJ, Classification and regression trees. 1984.
- M. Mielsen, Neural Networks and Deep Learning. 2019.
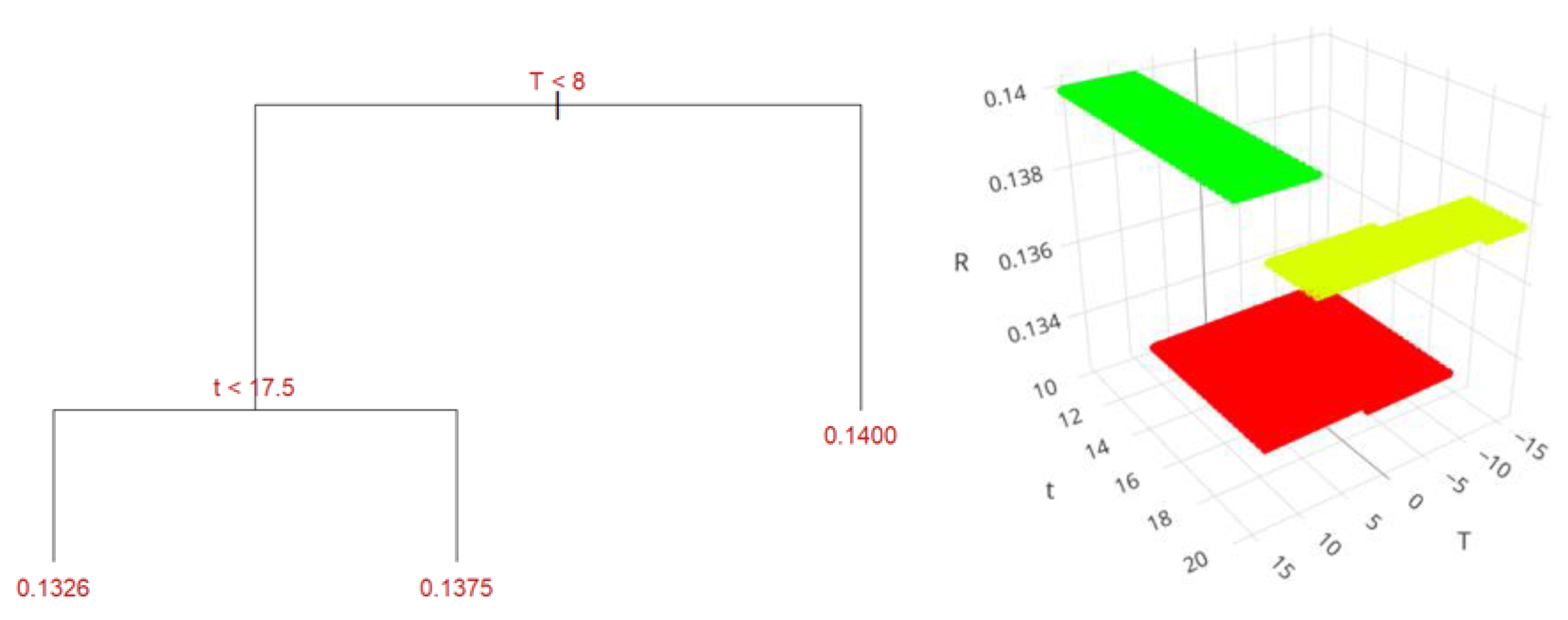
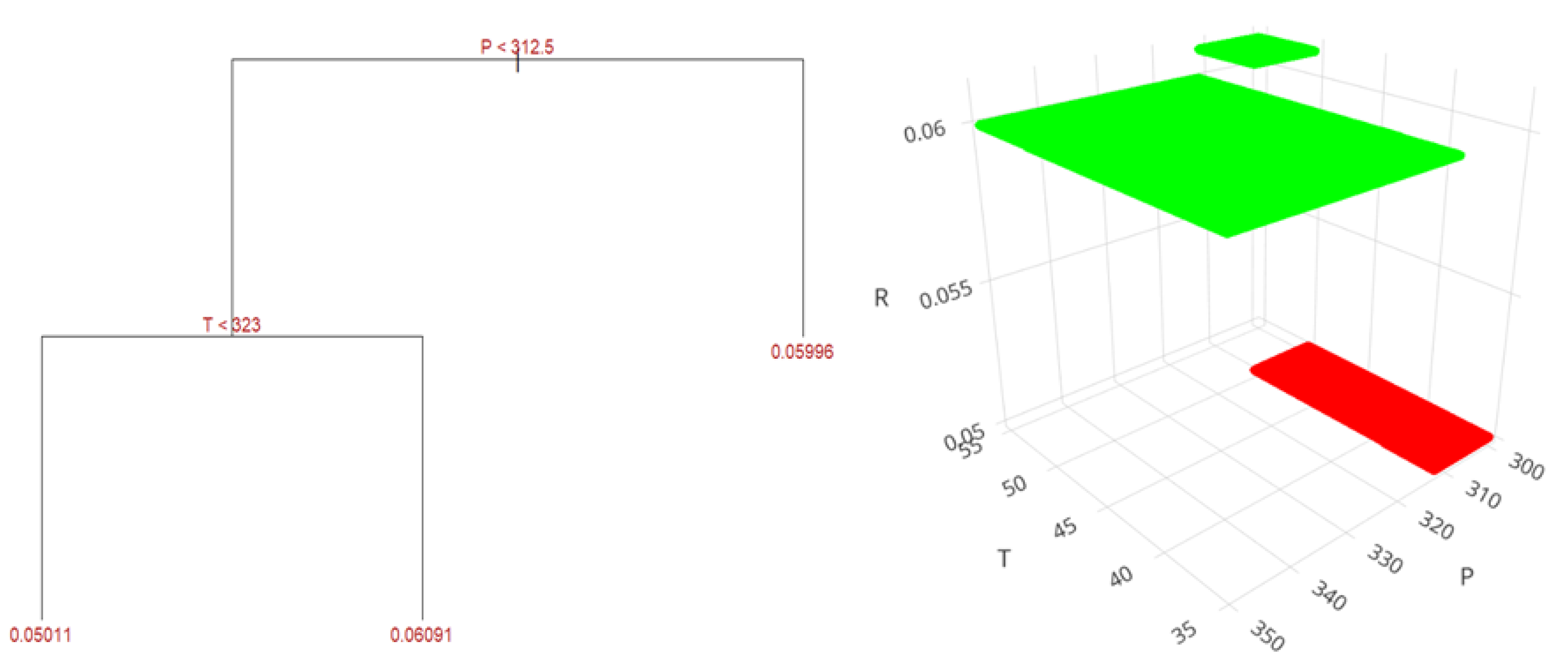
| Variable | UEA | MAC | SFE |
|---|---|---|---|
| Main Effects | P-value | P-value | P-value |
| A: Variety | 0.0000 | 0.0000 | 0.0000 |
| B: Time | 0.2122 | 0.7602 | 0.5982 1 |
| C: Temperature | 0.0001 | 0.0232 | 0.5982 |
| Interactions | - | - | - |
| Variable | UEA | MAC | SFE | |||
|---|---|---|---|---|---|---|
| Main Effects | Min | Max | Min | Max | Min | Max |
| A: Variety | Lemonge | Titan | Lemonge | Titan | Lemonge | Titan |
| B: Time, min | - | - | - | - | - | - |
| C: Temperature, °C | -16 | 16 | -16 | 16 | - | - |
| Yield, % | 9.26 ± 0.19 | 17.44 ± 0.18 | 9.46 ± 0.21 | 16.89 ± 0.20 | 1.88 ± 0.35 | 8.11 ± 0.35 |
| Method | Pressure, bar | Temperature, °C | Time, min |
|---|---|---|---|
| MAC | - | 16 | 20 |
| UEA | - | 16 | 10 |
| SFE (CBD, THC, CBN) |
325 | 45 | - |
| SFE (CBG) |
300 | 35 | - |
| Cannabinoids | Value | F | GL Num |
GL Denom |
p |
|---|---|---|---|---|---|
| Maceration | |||||
| Variety | 0.01884 | 15.829 | 20 | 136 | 0.000 |
| Time | 0.87636 | 0.699 | 8 | 82 | 0.691 |
| Temperature | 0.80341 | 1.185 | 8 | 82 | 0.318 |
| Ultrasound | |||||
| Variety | 0.01721 | 16.455 | 20 | 136 | 0.000 |
| Time | 0.86861 | 0.748 | 8 | 82 | 0.649 |
| Temperature | 0.68956 | 2.093 | 8 | 82 | 0.046 |
| Supercritical Fluids | |||||
| Variety | 0.00701 | 23.700 | 20 | 136 | 0.000 |
| Pressure | 0.89317 | 0.596 | 8 | 82 | 0.779 |
| Temperature | 0.81850 | 1.080 | 8 | 82 | 0.386 |
| Variety | Unit | CBDt / CBDv | THC / THCv | CBN / CBNv | CBG / CBGv |
|---|---|---|---|---|---|
| Cherry Bubblegum | % w/w | 13.84 / 14.27 | 0.55 / 0.16 | 0.00 / 0.19 | 0.00 / 0.00 |
| Cherry Oregon | % w/w | 10.10 / 12.42 | 0.55 / 0.06 | 0.00 / 0.21 | 0.00 / 0.00 |
| Titan | % w/w | 10.59 / 18.15 | 0.36 / 0.34 | 0.00 / 0.19 | 0.00 / 0.00 |
| Esmeralda 16 | % w/w | 16.78 / 13.46 | 0.55 / 0.33 | 0.00 / 0.00 | 0.00 / 0.00 |
| Esmeralda 19 | % w/w | 0.00 / 0.00 | 0.10 / 0.00 | 0.00 / 0.00 | 15.00 / 4.81 |
| Lemonge | % w/w | 0.00 / 0.00 | 0.10 / 0.00 | 0.00 / 0.00 | 17.00 / 10.83 |
| Method | CBD [% w/w] | THC [% w/w] | CBN [% w/w] | CBG [% w/w] |
|---|---|---|---|---|
| MAC | 41.32 ± 1.85 (EM-16) |
3.73 ± 0.15 (EM-16) |
1.12 ± 0.14 (Titan) |
59.36 ± 2.95 (EM-19) |
| UEA | 40.48 ± 2.52 (Titan) |
3.81 ± 0.35 (Cherry Bubblegum) |
1.54 ± 0.36 (EM-16) |
67.77 ± 3.43 (EM-19) |
| SFE | 44.65 ± 4.06 (Cherry Oregon) |
6.32 ± 0.36 (Titan) |
0.64 ± 0.06 (Cherry Oregon) |
7.68 ± 0.63 (Lemonge) |
| Laboratory coding | Sample information |
|---|---|
| M1 | EM-19 flower |
| M2 | EM-16 flower |
| M3 | Titan flower |
| M4 | Cherry Bubblegum flower |
| M5 | Cherry Oregon flower |
| M6 | Lemonge flower |
| Morphology | Number of axenic isolates |
|---|---|
| Gram positive cocci | 6 |
| Gram-negative bacilli | 12 |
| Gram-positive bacilli | 10 |
| Total, isolates | 28 |
| Samples | Unit | Organochlorines | Organophosphates |
|---|---|---|---|
| Cherry Bubblegum | µg/kg | < 4 | < 10 |
| Cherry Oregon | µg/kg | < 4 | < 10 |
| Titan | µg/kg | < 4 | < 10 |
| Lemonge | µg/kg | < 4 | < 10 |
| EM-19 | µg/kg | < 4 | < 10 |
| EM-16 | µg/kg | < 4 | < 10 |
| Variety | Unit | Cadmium, Cd | Chromium, Cr | Mercury, Hg | Arsenic, As | Lead, Pb |
|---|---|---|---|---|---|---|
| Cherry Bubblegum | ppm | 0.20 | < 0.05 | < 0.02 | < 0.025 | 1.70 |
| Cherry Oregon | ppm | 0.20 | 2.60 | < 0.02 | < 0.025 | 2.63 |
| EM-16 | ppm | < 0.001 | < 0.05 | < 0.02 | 0.051 | 3.52 |
| EM-19 | ppm | < 0.001 | < 0.05 | < 0.02 | 0.034 | 3.39 |
| Lemonge | ppm | 2.09 | < 0.05 | < 0.02 | < 0.025 | 2.84 |
| Titan | ppm | 0.60 | 1.40 | < 0.02 | < 0.025 | 2.01 |
| Time, min | Mobile phase A, % | Mobile phase B, % | Flow, ml/min | |
|---|---|---|---|---|
| 0.00 | 40 | 60 | 0.60 | |
| 6.00 | 5 | 95 | 0.60 | |
| 12.01 | 5 | 95 | 0.20 | |
| 14.00 | 0 | 100 | 0.20 | |
| 18.00 | 40 | 60 | 0.20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




