Submitted:
02 December 2024
Posted:
03 December 2024
You are already at the latest version
Abstract
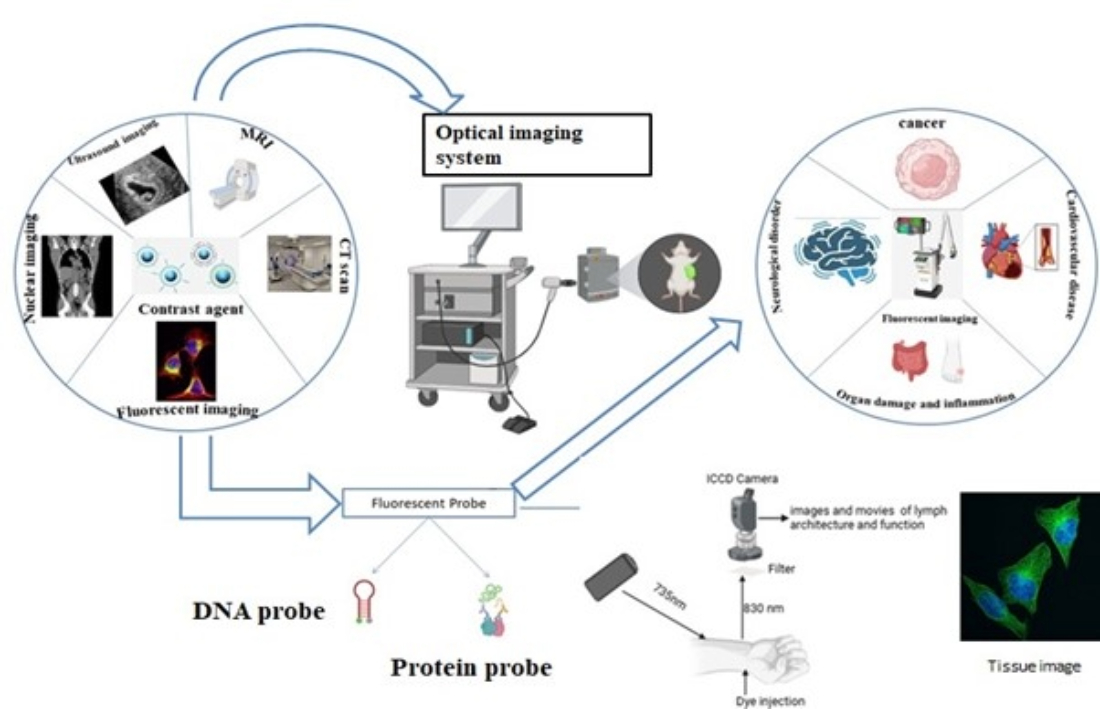
Keywords:
1. Introduction
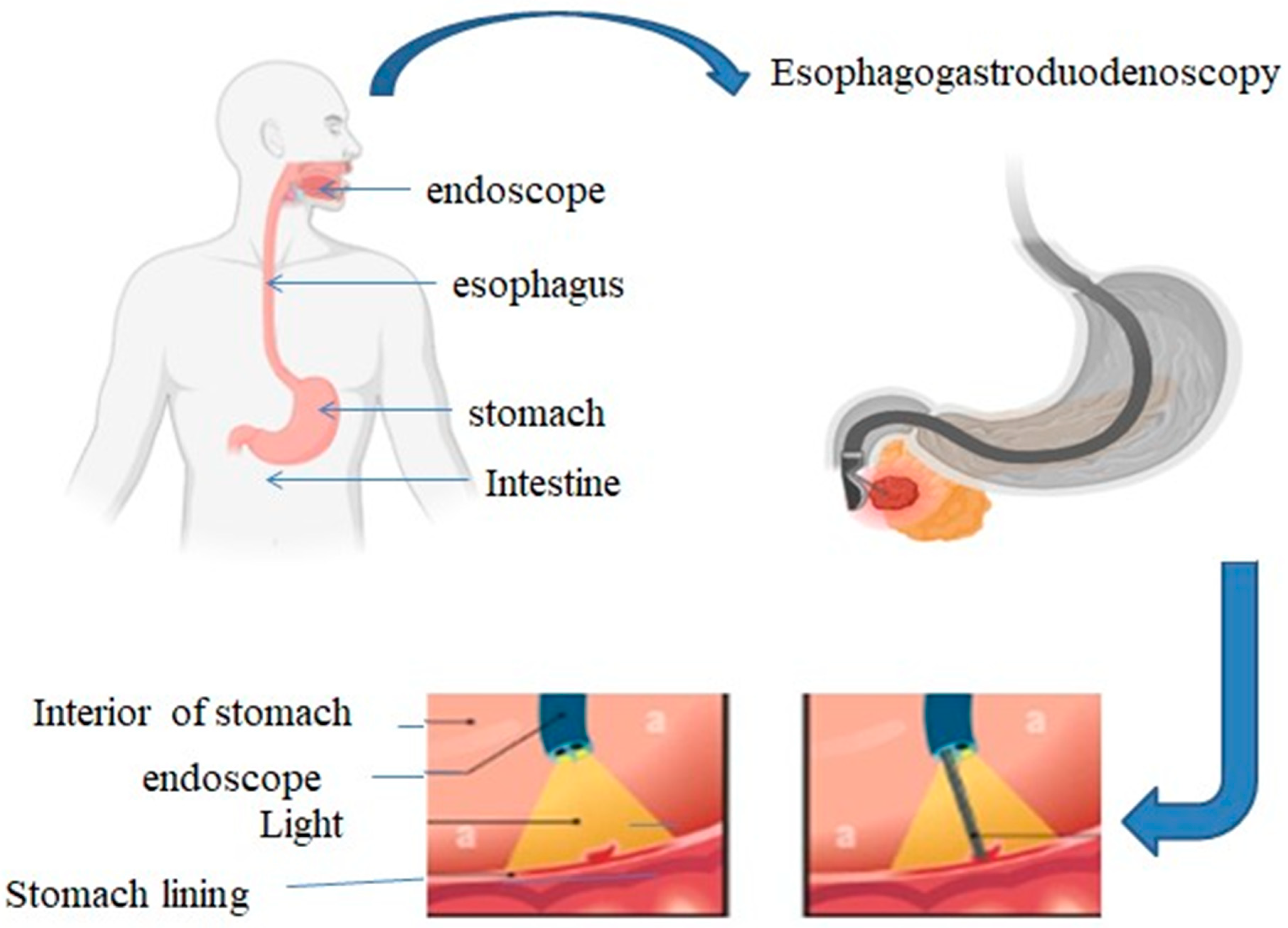
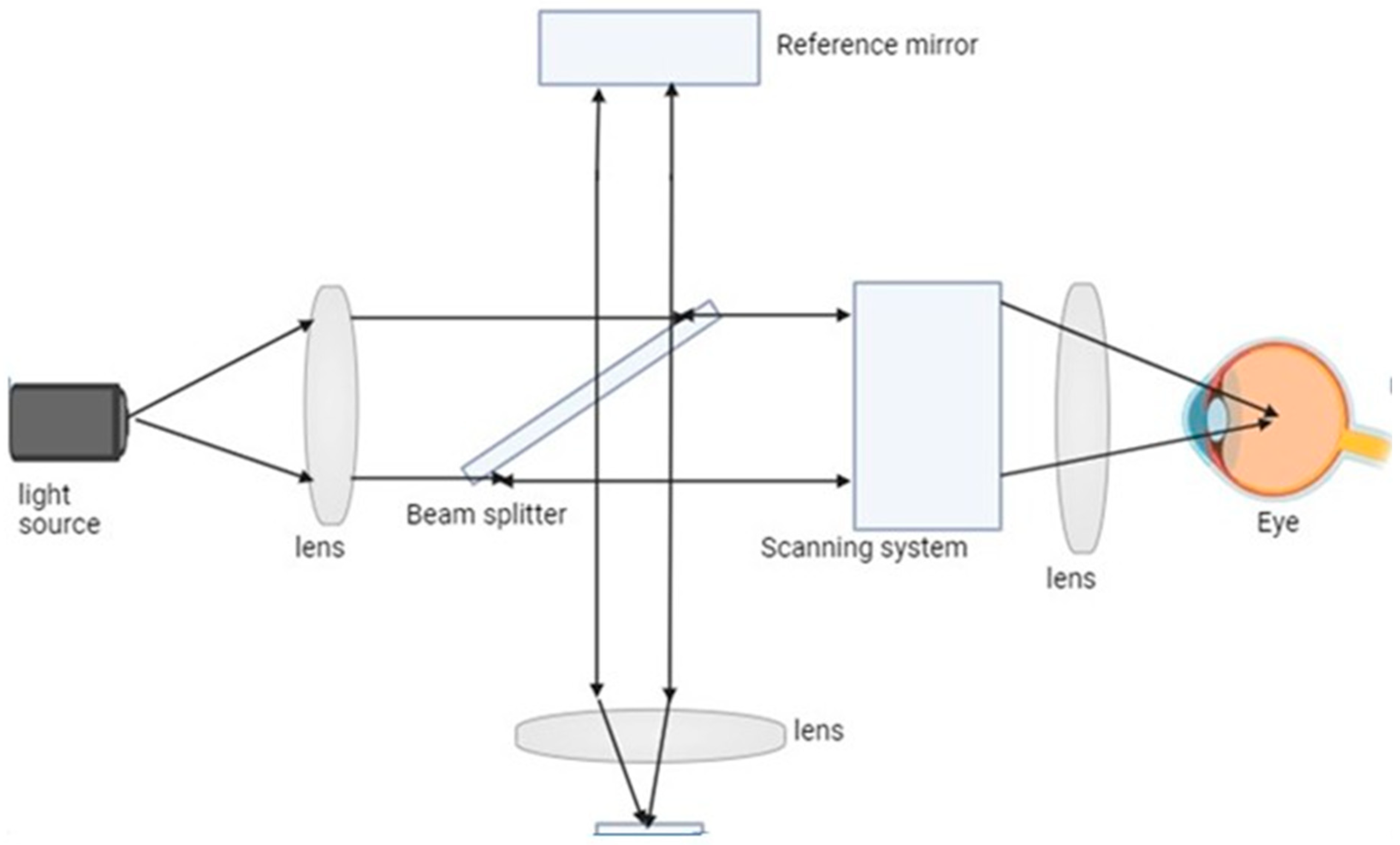
2. Principle of Optical Imaging
3. Contrast Agents
4. Fluorescence Imaging
5. Parts of Fluorescence Imaging
6. Types of Fluorescence Imaging
6.1. Widefield Fluorescence Imaging
6.2. Confocal Fluorescence Imaging
6.3. Multiphoton Fluorescence Imaging
6.4. Fluorescence Lifetime Imaging Microscopy (FLIM)
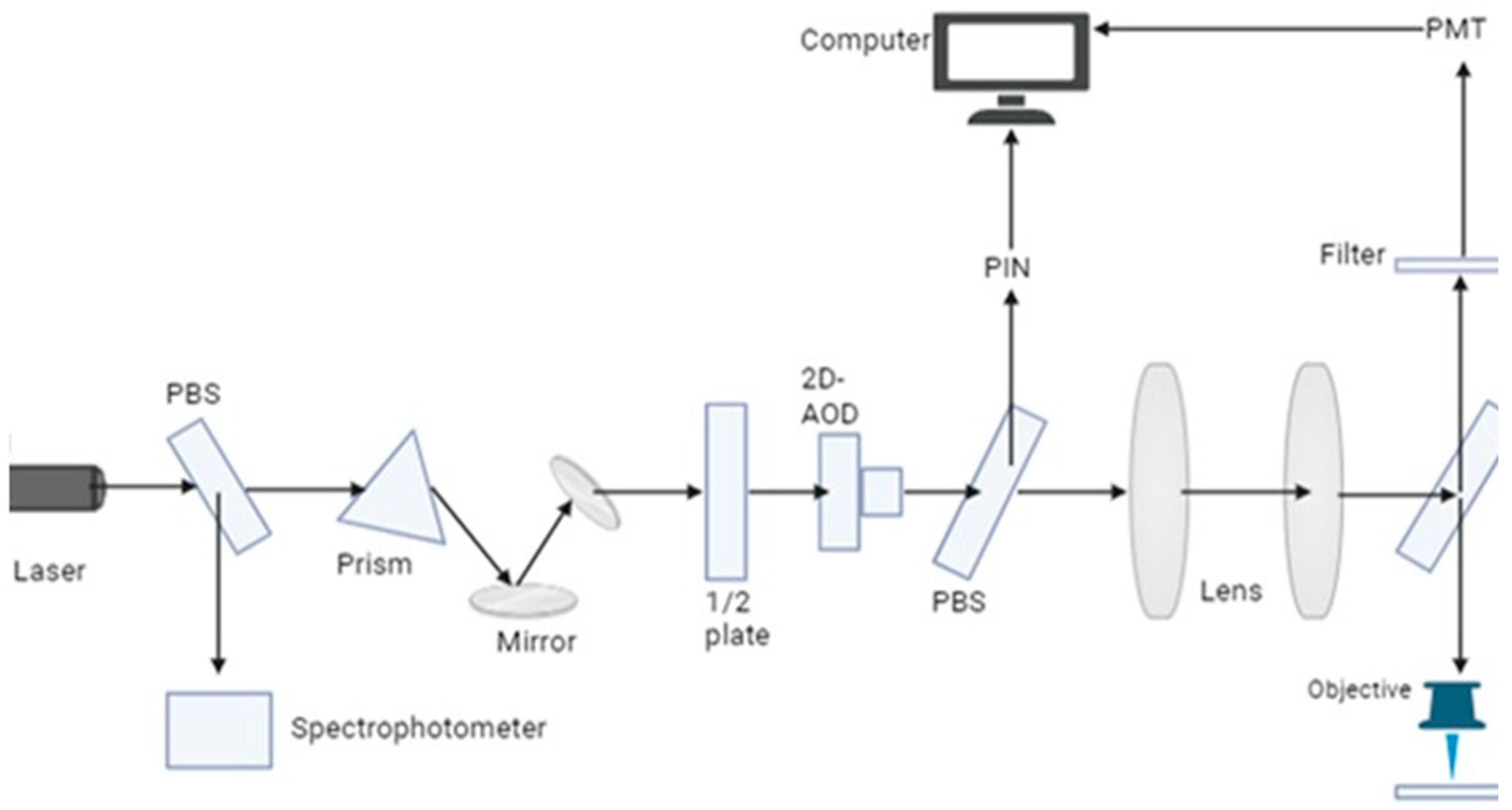
6.5. Super-Resolution Fluorescence Imaging
6.7. Vivo Fluorescence Imaging
7. Fluorescent Probe Designing and Synthesis
8. Applications of Fluorescence Probes
8.1. Molecular Imaging and Cancer Detection
8.2. Brain and Cardiovascular Imaging
| Application | Description |
|---|---|
| Fluorescence molecular imaging | A non-invasive technique for tracking illnesses, researching biological processes, and learning about how drugs work. |
| Cancer identification | Improves tumor border delineation with sophisticated imaging techniques; uses tumor-avid probes for high specificity and sensitivity in detecting malignancies. |
| Development of probes | The development of fluorescent probes that glow in the far-red to near-infrared spectrum and are sensitive to specific targets like HOCl has allowed for deep tissue penetration and high sensitivity. |
| Real-time Imaging | With the use of visualization techniques and adaptive procedures to improve accuracy, fluorescence imaging is being used in clinical settings more and more. |
| Surgical Guidance | Increases endoscopic and surgical imaging by continuously providing feedback during the procedure; motion artifacts are minimized by using methods such optical flow correction. |
| Near–infrared imaging | Provides superior real-time display and spatial resolution for cancer diagnosis, making up for the drawbacks of conventional imaging modalities in a range of applications. |
| Brain imaging | Enables the cellular and molecular analysis of brain activity; using specialized optics and fluorescent markers to examine neurotransmission and synaptic communication. |
| Vascular Imaging | Non-invasive cerebral vasculature observation is vital to comprehending disorders such as stroke since it tracks anomalies in blood vessels in real time. |
| Cardiovascular imaging | To assess vascular anatomy and detect cerebrovascular diseases, employ near-infrared fluorescence imaging, which offers deep tissue penetration and great spatial resolution.. |
| Benefits | Difficulties |
|---|---|
| Real-time imaging: Fluorescent probes make it possible to see physiological conditions and real-time cellular operations.[93] | Background signals: Fluorescence signals resulting from natural cofactors within living cells might provide a problem for imaging research.[94] |
| High sensitivity: The detection of particular biomolecules is made possible by the high sensitivity and specificity of fluorescent probes.[94] | Elevated background signals: They can diminish signal contrast in intact tissue and multi-cell systems.[94] |
| Non-invasive Imaging: Highly precise non-invasive imaging of cellular events is made possible by tiny fluorophores.[93] | Challenges with in vivo cancer imaging: Creating fluorescent nanoparticle probes presents difficulties. |
| Increased functionality: Optimal optical characteristics for certain subcellular locations are provided by small-molecule fluorescent probes. | Complex photo physical schemes: The complex photo physical schemes of certain fluorescent probes influence their bio-analytical responses. |
| Targeted therapy: Fluorescent probes can help medications be delivered in a specific manner in targeted therapy. | Inadequate signal-to-background ratios hinder the clinical application of optical molecular imaging. |
Difficulties with Tissue Auto-Fluorescence and Depth Penetration
8.3. Clinical Translation
Conclusion
References
- Licha, K. (2002). Contrast Agents for Optical Imaging. In: Krause, W. (eds) Contrast Agents II. Topics in Current Chemistry, vol 222. Springer, Berlin, Heidelberg. [CrossRef]
- https://www.nibib.nih.gov/science-education/science-topics/optical-imaging.
- Bremer, C., Ntziachristos, V. & Weissleder, R. Optical-based molecular imaging: contrast agents and potential medical applications. Eur Radiol 13, 231–243 (2003). [CrossRef]
- https://www.msdmanuals.com/en-in/professional/gastrointestinal-disorders/diagnostic-and-therapeutic-gastrointestinal-procedures/endoscopy.
- https://www.healthline.com/health/endoscopy.
- https://www.medicalnewstoday.com/articles/153737.
- Wu, Y., , Zeng, F., , Zhao, Y., , & Wu, S., (2021). Emerging contrast agents for multispectral optoacoustic imaging and their biomedical applications. Chemical Society reviews, 50(14), 7924–7940. [CrossRef]
- Ehlers, J. P., Gupta, P. K., Farsiu, S., Maldonado, R., Kim, T., Toth, C. A., & Mruthyunjaya, P. (2010). Evaluation of contrast agents for enhanced visualization in optical coherence tomography. Investigative ophthalmology & visual science, 51(12), 6614–6619. [CrossRef]
- Nguyen, V. P., Hu, J., Zhe, J., Ramasamy, S., Ahmed, U., & Paulus, Y. M. (2024). Advanced nanomaterials for imaging of eye diseases. ADMET & DMPK, 12(2), 269–298. [CrossRef]
- Cho, N., & Shokeen, M. (2019). Changing landscape of optical imaging in skeletal metastases. Journal of bone oncology, 17, 100249. [CrossRef]
- Nioka, S., & Chen, Y. (2011). Optical tecnology developments in biomedicine: history, current and future. Translational medicine @ UniSa, 1, 51–150.
- Hadjipanayis, C. G., Jiang, H., Roberts, D. W., & Yang, L. (2011). Current and future clinical applications for optical imaging of cancer: from intraoperative surgical guidance to cancer screening. Seminars in oncology, 38(1), 109–118. [CrossRef]
- Hintz, S. R., Cheong, W. F., van Houten, J. P., Stevenson, D. K., & Benaron, D. A. (1999). Bedside imaging of intracranial hemorrhage in the neonate using light: comparison with ultrasound, computed tomography, and magnetic resonance imaging. Pediatric research, 45(1), 54–59. [CrossRef]
- Wang, L., Zhu, W., Zhang, Y., Chen, S., & Yang, D. (2021). Harnessing the Power of Hybrid Light Propagation Model for Three-Dimensional Optical Imaging in Cancer Detection. Frontiers in oncology, 11, 750764. [CrossRef]
- Moriyama, E. H., Zheng, G., & Wilson, B. C. (2008). Optical molecular imaging: from single cell to patient. Clinical pharmacology and therapeutics, 84(2), 267–271. [CrossRef]
- Kaller, M. O., & An, J. (2023). Contrast Agent Toxicity. In StatPearls. StatPearls Publishing.
- https://www.msdmanuals.com/home/special-subjects/common-imaging-tests/angiography.
- Chen Y. (2022). Recent Advances in Excimer-Based Fluorescence Probes for Biological Applications. Molecules (Basel, Switzerland), 27(23), 8628. [CrossRef]
- Zhu, H., Fan, J., Du, J., & Peng, X. (2016). Fluorescent Probes for Sensing and Imaging within Specific Cellular Organelles. Accounts of chemical research, 49(10), 2115–2126. [CrossRef]
- Liu, Z., Liu, J., Wang, X., Mi, F., Wang, D., & Wu, C. (2020). Fluorescent Bioconjugates for Super-Resolution Optical Nanoscopy. Bioconjugate chemistry, 31(8), 1857–1872. [CrossRef]
- Dean, K. M., & Palmer, A. E. (2014). Advances in fluorescence labeling strategies for dynamic cellular imaging. Nature chemical biology, 10(7), 512–523. [CrossRef]
- Pirovano, G., Roberts, S., Kossatz, S., & Reiner, T. (2020). Optical Imaging Modalities: Principles and Applications in Preclinical Research and Clinical Settings. Journal of nuclear medicine : official publication, Society of Nuclear Medicine, 61(10), 1419–1427. [CrossRef]
- Wang, D., & Xia, J. (2019). Optics based biomedical imaging: Principles and applications. Journal of Applied Physics.
- Lin, L., Jiang, P., Bao, Z., Pang, W., Ding, S., Yin, M. J., Li, P., & Gu, B. (2021). Fundamentals of Optical Imaging. Advances in experimental medicine and biology, 3233, 1–22. [CrossRef]
- Han, S., Lee, D., Kim, S., Kim, H. H., Jeong, S., & Kim, J. (2022). Contrast Agents for Photoacoustic Imaging: A Review Focusing on the Wavelength Range. Biosensors, 12(8), 594. [CrossRef]
- Hadzima, M., Faucher, F. F., Blažková, K., Yim, J. J., Guerra, M., Chen, S., Woods, E. C., Park, K. W., Šácha, P., Šubr, V., Kostka, L., Etrych, T., Majer, P., Konvalinka, J., & Bogyo, M. (2024). Polymer-Tethered Quenched Fluorescent Probes for Enhanced Imaging of Tumor-Associated Proteases. ACS sensors, 9(7), 3720–3729. [CrossRef]
- Park, J. H., , Dumani, D. S., , Arsiwala, A., , Emelianov, S., , & Kane, R. S., (2018). Tunable aggregation of gold-silica janus nanoparticles to enable contrast-enhanced multiwavelength photoacoustic imaging in vivo. Nanoscale, 10(32), 15365–15370. [CrossRef]
- Miao, Q., & Pu, K. (2018). Organic Semiconducting Agents for Deep-Tissue Molecular Imaging: Second Near-Infrared Fluorescence, Self-Luminescence, and Photoacoustics. Advanced materials (Deerfield Beach, Fla.), 30(49), e1801778. [CrossRef]
- Gottschalk, H. M., Wecker, T., Khattab, M. H., Fischer, C. V., Callizo, J., Rehfeldt, F., Lubjuhn, R., Russmann, C., Hoerauf, H., & van Oterendorp, C. (2019). Lipid Emulsion-Based OCT Angiography for Ex Vivo Imaging of the Aqueous Outflow Tract. Investigative ophthalmology & visual science, 60(1), 397–406. [CrossRef]
- Hui, X., Malik, M. O. A., & Pramanik, M. (2022). Looking deep inside tissue with photoacoustic molecular probes: a review. Journal of biomedical optics, 27(7), 070901. [CrossRef]
- Nguyen, V. P., Qian, W., Wang, X., & Paulus, Y. M. (2021). Functionalized contrast agents for multimodality photoacoustic microscopy, optical coherence tomography, and fluorescence microscopy molecular retinal imaging. Methods in enzymology, 657, 443–480. [CrossRef]
- Lemaster, J. E., & Jokerst, J. V. (2017). What is new in nanoparticle-based photoacoustic imaging?. Wiley interdisciplinary reviews. Nanomedicine and nanobiotechnology, 9(1), 10.1002/wnan.1404. [CrossRef]
- Contrast Agents Market Size by Segments, Share, Regulatory, Reimbursement and Forecast to 2033. (2024). Market Research Reports & Consulting | GlobalData UK Ltd. https://www.globaldata.com/store/report/contrast-agents-devices-market-analysis/.
- Contrast Media Market Size, Share & Growth Report, 2030. (2024). grandviewresearch.com. https://www.grandviewresearch.com/industry-analysis/contrast-media-contrast-agents-market.
- https://www.fnfresearch.com/news/global-mri-contrast-media-agents-market.
- Lee, J. H., Park, G., Hong, G. H., Choi, J., & Choi, H. S. (2012). Design considerations for targeted optical contrast agents. Quantitative imaging in medicine and surgery, 2(4), 266–273. [CrossRef]
- Nakata, E., Gerelbaatar, K., Komatsubara, F., & Morii, T. (2022). Stimuli-Responsible SNARF Derivatives as a Latent Ratiometric Fluorescent Probe. Molecules (Basel, Switzerland), 27(21), 7181. [CrossRef]
- Guo, L., Liu, F., Cai, C., Liu, J., & Zhang, G. (2019). 3D deep encoder-decoder network for fluorescence molecular tomography. Optics letters, 44(8), 1892–1895. [CrossRef]
- Zhu, L., Zhang, L., Zhou, M., & Alifu, N. (2021). Sheng wu gong cheng xue bao = Chinese journal of biotechnology, 37(8), 2678–2687. [CrossRef]
- Boutorine, A. S., Novopashina, D. S., Krasheninina, O. A., Nozeret, K., & Venyaminova, A. G. (2013). Fluorescent probes for nucleic Acid visualization in fixed and live cells. Molecules (Basel, Switzerland), 18(12), 15357–15397. [CrossRef]
- Chen, G., Li, C., Zhang, Y., & Wang, Q. (2021). Whole-Body Fluorescence Imaging in the Near-Infrared Window. Advances in experimental medicine and biology, 3233, 83–108. [CrossRef]
- Heeman, W., Vonk, J., Ntziachristos, V., Pogue, B. W., Dierckx, R. A. J. O., Kruijff, S., & van Dam, G. M. (2022). A Guideline for Clinicians Performing Clinical Studies with Fluorescence Imaging. Journal of nuclear medicine : official publication, Society of Nuclear Medicine, 63(5), 640–645. [CrossRef]
- Si, K., Fiolka, R., & Cui, M. (2012). Fluorescence imaging beyond the ballistic regime by ultrasound pulse guided digital phase conjugation. Nature photonics, 6(10), 657–661. [CrossRef]
- Nguyen, J. Q. M., McWade, M., Thomas, G., Beddard, B. T., Herington, J. L., Paria, B. C., Schwartz, H. S., Halpern, J. L., Holt, G. E., & Mahadevan-Jansen, A. (2018). Development of a modular fluorescence overlay tissue imaging system for wide-field intraoperative surgical guidance. Journal of medical imaging (Bellingham, Wash.), 5(2), 021220. [CrossRef]
- Prakriti Karki. (2024). Fluorescence Microscope: Principle, Parts, Uses, Examples. Microbe Notes. https://microbenotes.com/fluorescence-microscope-principle-instrumentation-applications-advantages-limitations/.
- Mubaid, F., Kaufman, D., Wee, T. L., Nguyen-Huu, D. S., Young, D., Anghelopoulou, M., & Brown, C. M. (2019). Fluorescence microscope light source stability. Histochemistry and cell biology, 151(4), 357–366. [CrossRef]
- Fluorescence Microscopy - Anatomy of the Fluorescence Microscope | Olympus LS. (2024). olympus-lifescience.com. https://www.olympus-lifescience.com/de/microscope-resource/primer/techniques/fluorescence/anatomy/fluoromicroanatomy/.
- Fluorescence Microscopy - Explanation and Labelled Images. (2024). New York Microscope Company. https://microscopeinternational.com/fluorescence-microscopy/.
- Introduction to Fluorescence Microscopy. (2024). Nikon’s MicroscopyU. https://www.microscopyu.com/techniques/fluorescence/introduction-to-fluorescence-microscopy.
- Agard, D. A., Hiraoka, Y., & Sedat, J. W. (1988). Three-dimensional light microscopy of diploid Drosophila chromosomes. Cell motility and the cytoskeleton, 10(1-2), 18–27. [CrossRef]
- Park, S., Zhang, J., Reyer, M. A., Zareba, J., Troy, A. A., & Fei, J. (2018). Conducting Multiple Imaging Modes with One Fluorescence Microscope. Journal of visualized experiments : JoVE, (140), 58320. [CrossRef]
- Ishikawa-Ankerhold, H. C., Ankerhold, R., & Drummen, G. P. (2012). Advanced fluorescence microscopy techniques--FRAP, FLIP, FLAP, FRET and FLIM. Molecules (Basel, Switzerland), 17(4), 4047–4132. [CrossRef]
- Schäferling M. (2012). The art of fluorescence imaging with chemical sensors. Angewandte Chemie (International ed. in English), 51(15), 3532–3554. [CrossRef]
- Emptage N. J. (2001). Fluorescent imaging in living systems. Current opinion in pharmacology, 1(5), 521–525. [CrossRef]
- Ntziachristos, V., Turner, G., Dunham, J., Windsor, S., Soubret, A., Ripoll, J., & Shih, H. A. (2005). Planar fluorescence imaging using normalized data. Journal of biomedical optics, 10(6), 064007. [CrossRef]
- van der Valk, M. J. M., Handgraaf, H. J. M., Sibinga-Mulder, B. G., Hilling, D. E., van Dam, G. M., & Vahrmeijer, A. L. (2018). Intraoperatieve beeldvorming met fluorescentie, 5 jaar later [Intraoperative imaging using fluorescence, 5 years later]. Nederlands tijdschrift voor geneeskunde, 162, D2067.
- Li, C., Gao, L., Liu, Y., & Wang, L. V. (2013). Optical sectioning by wide-field photobleaching imprinting microscopy. Applied physics letters, 103(18), 183703. [CrossRef]
- Forsgren, E., Edlund, C., Oliver, M., Barnes, K., Sjögren, R., & Jackson, T. R. (2022). High-throughput widefield fluorescence imaging of 3D samples using deep learning for 2D projection image restoration. PloS one, 17(5), e0264241. [CrossRef]
- Xiao, D., Zang, Z., Wang, Q., Jiao, Z., Rocca, F. M. D., Chen, Y., & Li, D. D. U. (2022). Smart Wide-field Fluorescence Lifetime Imaging System with CMOS Single-photon Avalanche Diode Arrays. Annual International Conference of the IEEE Engineering in Medicine and Biology Society. IEEE Engineering in Medicine and Biology Society. Annual International Conference, 2022, 1887–1890. [CrossRef]
- Xiao, D., Zang, Z., Wang, Q., Jiao, Z., Rocca, F. M. D., Chen, Y., & Li, D. D. U. (2022). Smart Wide-field Fluorescence Lifetime Imaging System with CMOS Single-photon Avalanche Diode Arrays. Annual International Conference of the IEEE Engineering in Medicine and Biology Society. IEEE Engineering in Medicine and Biology Society. Annual International Conference, 2022, 1887–1890. [CrossRef]
- Crowe, S. E., & Ellis-Davies, G. C. (2014). Longitudinal in vivo two-photon fluorescence imaging. The Journal of comparative neurology, 522(8), 1708–1727. [CrossRef]
- Wolenski, J. S., & Julich, D. (2014). Fluorescence microscopy gets faster and clearer: roles of photochemistry and selective illumination. The Yale journal of biology and medicine, 87(1), 21–32.
- Pacheco, S., Wang, C., Chawla, M. K., Nguyen, M., Baggett, B. K., Utzinger, U., Barnes, C. A., & Liang, R. (2017). High resolution, high speed, long working distance, large field of view confocal fluorescence microscope. Scientific reports, 7(1), 13349. [CrossRef]
- Botchway, S. W., Scherer, K. M., Hook, S., Stubbs, C. D., Weston, E., Bisby, R. H., & Parker, A. W. (2015). A series of flexible design adaptations to the Nikon E-C1 and E-C2 confocal microscope systems for UV, multiphoton and FLIM imaging. Journal of microscopy, 258(1), 68–78. [CrossRef]
- Yan, W., Peng, X., Qi, J., Gao, J., Fan, S., Wang, Q., Qu, J., & Niu, H. (2014). Dynamic fluorescence lifetime imaging based on acousto-optic deflectors. Journal of biomedical optics, 19(11), 116004. [CrossRef]
- Datta, R., Heaster, T. M., Sharick, J. T., Gillette, A. A., & Skala, M. C. (2020). Fluorescence lifetime imaging microscopy: fundamentals and advances in instrumentation, analysis, and applications. Journal of biomedical optics, 25(7), 1–43. [CrossRef]
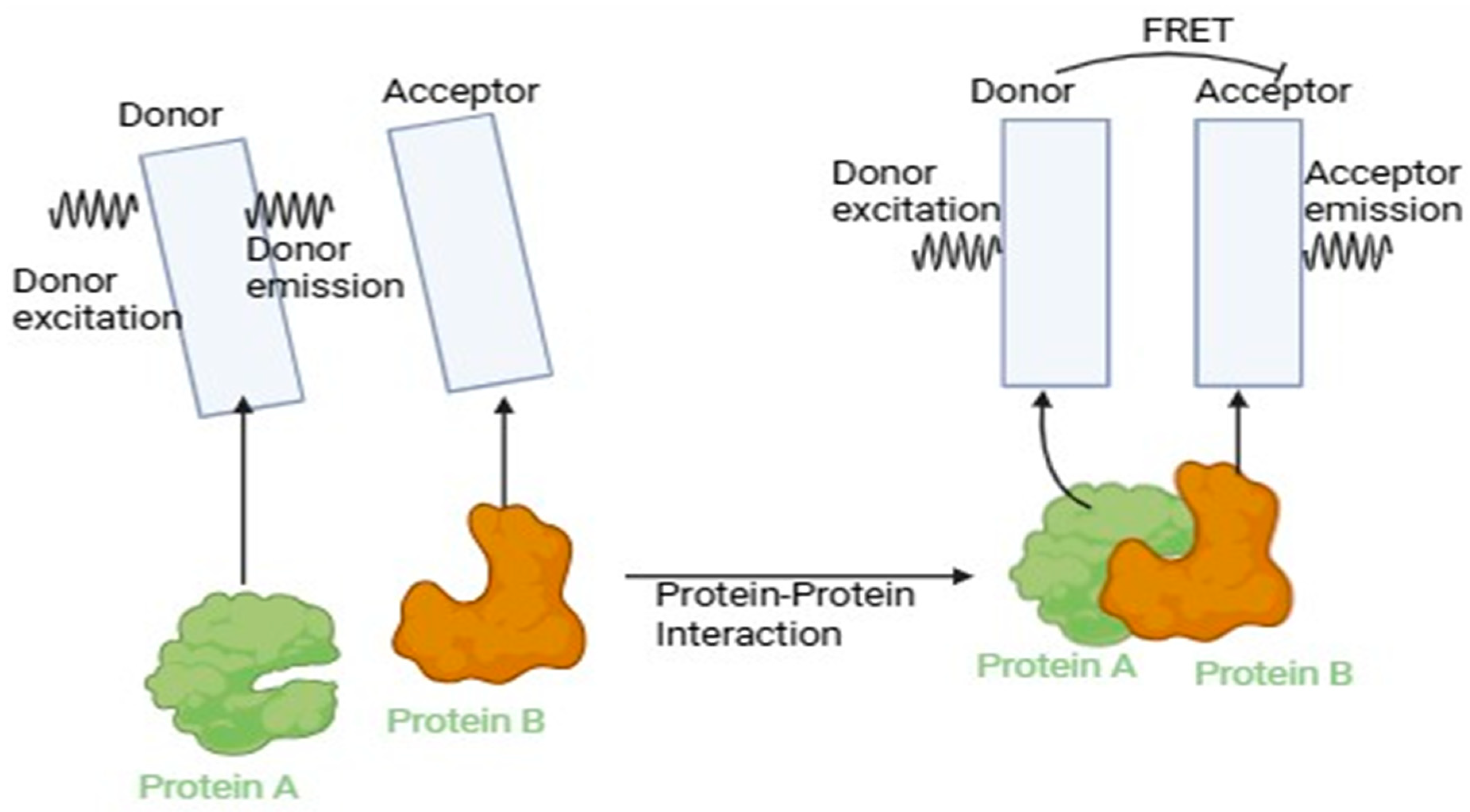
- Stanisavljevic, M., Krizkova, S., Vaculovicova, M., Kizek, R., & Adam, V. (2015). Quantum dots-fluorescence resonance energy transfer-based nanosensors and their application. Biosensors & bioelectronics, 74, 562–574. [CrossRef]
- Balconi, M., & Crivelli, D. (2010). Veridical and false feedback sensitivity and punishment-reward system (BIS/BAS): ERP amplitude and theta frequency band analysis. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology, 121(9), 1502–1510. [CrossRef]
- https://www.gu.se/en/core-facilities/fluorescence-resonance-energy-transfer.
- Kumar, N., Bhalla, V., & Kumar, M. (2014). Resonance energy transfer-based fluorescent probes for Hg2+, Cu2+ and Fe2+/Fe3+ ions. The Analyst, 139(3), 543–558. [CrossRef]
- Zhu, F. Y., Mei, L. J., Tian, R., Li, C., Wang, Y. L., Xiang, S. L., Zhu, M. Q., & Tang, B. Z. (2024). Recent advances in super-resolution optical imaging based on aggregation-induced emission. Chemical Society reviews, 53(7), 3350–3383. [CrossRef]
- Hugelier, S., Colosi, P. L., & Lakadamyali, M. (2023). Quantitative Single-Molecule Localization Microscopy. Annual review of biophysics, 52, 139–160. [CrossRef]
- Yu, J., Jiang, G., & Wang, J. (2023). In Vivo Fluorescence Imaging-Guided Development of Near-Infrared AIEgens. Chemistry, an Asian journal, 18(5), e202201251. [CrossRef]
- Yu, M., Ward, M. B., Franke, A., Ambrose, S. L., Whaley, Z. L., Bradford, T. M., Gorden, J. D., Beyers, R. J., Cattley, R. C., Ivanović-Burmazović, I., Schwartz, D. D., & Goldsmith, C. R. (2017). Adding a Second Quinol to a Redox-Responsive MRI Contrast Agent Improves Its Relaxivity Response to H2O2. Inorganic chemistry, 56(5), 2812–2826. [CrossRef]
- He, G., Liu, C., Liu, X., Wang, Q., Fan, A., Wang, S., & Qian, X. (2017). Design and synthesis of a fluorescent probe based on naphthalene anhydride and its detection of copper ions. PloS one, 12(10), e0186994. [CrossRef]
- Mao, G., Liu, C., Yang, N., Yang, L., & He, G. (2021). Design and Synthesis of a Fluorescent Probe Based on Copper Complex for Selective Detection of Hydrogen Sulfide. Journal of Sensors. [CrossRef]
- Li, L., Wang, J., Xu, S., Li, C., & Dong, B. (2022). Recent Progress in Fluorescent Probes For Metal Ion Detection. Frontiers in Chemistry. [CrossRef]
- Jacoby-Morris, K., & Patterson, G. H. (2021). Choosing Fluorescent Probes and Labeling Systems. Methods in molecular biology (Clifton, N.J.), 2304, 37–64. [CrossRef]
- Kobayashi, H., Ogawa, M., Alford, R., Choyke, P. L., & Urano, Y. (2010). New strategies for fluorescent probe design in medical diagnostic imaging. Chemical reviews, 110(5), 2620–2640. [CrossRef]
- Sun, J., Miller, J. P., Hathi, D., Zhou, H., Achilefu, S., Shokeen, M., & Akers, W. J. (2016). Enhancing in vivo tumor boundary delineation with structured illumination fluorescence molecular imaging and spatial gradient mapping. Journal of biomedical optics, 21(8), 80502. [CrossRef]
- Natarajan, V., Thirumalaivasan, N., Wu, S. P., & Sivan, V. (2019). A far-red to NIR emitting ultra-sensitive probe for the detection of endogenous HOCl in zebrafish and the RAW 264.7 cell line. Organic & biomolecular chemistry, 17(14), 3538–3544. [CrossRef]
- Glatz, J., Symvoulidis, P., Garcia-Allende, P. B., & Ntziachristos, V. (2014). Robust overlay schemes for the fusion of fluorescence and color channels in biological imaging. Journal of biomedical optics, 19(4), 040501. [CrossRef]
- Cheng, H., Xu, H., Peng, B., Huang, X., Hu, Y., Zheng, C., & Zhang, Z. (2024). Illuminating the future of precision cancer surgery with fluorescence imaging and artificial intelligence convergence. Npj Precision Oncology. [CrossRef]
- Malik, M. M. U. D., Alqahtani, M. M., Hadadi, I., Kanbayti, I., Alawaji, Z., & Aloufi, B. A. (2024). Molecular Imaging Biomarkers for Early Cancer Detection: A Systematic Review of Emerging Technologies and Clinical Applications. Diagnostics. [CrossRef]
- Schouw, H. M., Huisman, L. A., Janssen, Y. F., Slart, R. H. J. A., Borra, R. J. H., Willemsen, A. T. M., Brouwers, A. H., van Dijl, J. M., Dierckx, R. A., van Dam, G. M., Szymanski, W., Boersma, H. H., & Kruijff, S. (2021). Targeted optical fluorescence imaging: a meta-narrative review and future perspectives. European Journal of Nuclear Medicine and Molecular Imaging. [CrossRef]
- Okubo Y. (2022). Investigation of Brain Functions with Fluorescence Imaging Techniques. Juntendo Iji zasshi = Juntendo medical journal, 68(2), 157–162. [CrossRef]
- Hong, G., Diao, S., Chang, J., Antaris, A. L., Chen, C., Zhang, B., Zhao, S., Atochin, D. N., Huang, P. L., Andreasson, K. I., Kuo, C. J., & Dai, H. (2014). Through-skull fluorescence imaging of the brain in a new near-infrared window. Nature photonics, 8(9), 723–730. [CrossRef]
- Kim, T. H., & Schnitzer, M. J. (2022). Fluorescence imaging of large-scale neural ensemble dynamics. Cell. [CrossRef]
- Mashalchi, S., Pahlavan, S., & Hejazi, M. (2021). A novel fluorescent cardiac imaging system for preclinical intraoperative angiography. BMC Medical Imaging. [CrossRef]
- Sosnovik, D. E., Nahrendorf, M., & Weissleder, R. (2008). Targeted imaging of myocardial damage. Nature Clinical Practice Cardiovascular Medicine. [CrossRef]
- Duprée, A., Rieß, H., Detter, C., Debus, E. S., & Wipper, S. H. (2018). Utilization of indocynanine green fluorescent imaging (ICG-FI) for the assessment of microperfusion in vascular medicine. Innovative Surgical Sciences. [CrossRef]
- Che, F., Zhao, X., Wang, X., Li, P., & Tang, B. (2023). Fluorescent Imaging Agents for Brain Diseases. Targets. [CrossRef]
- Yu, X., Feng, Z., Cai, Z., Jiang, M., Xue, D., Zhu, L., Zhang, Y., Liu, J., Que, B., Yang, W., Xi, W., Zhang, D., Qian, J., & Li, G. (2019). Deciphering of cerebrovasculatures via ICG-assisted NIR-II fluorescence microscopy. Journal of materials chemistry. B, 7(42), 6623–6629. [CrossRef]
- de Moliner, F., Nadal-Bufi, F., & Vendrell, M. (2024). Recent advances in minimal fluorescent probes for optical imaging. Current opinion in chemical biology, 80, 102458. [CrossRef]
- Huang, J., & Pu, K. (2020). Activatable Molecular Probes for Second Near-Infrared Fluorescence, Chemiluminescence, and Photoacoustic Imaging. Angewandte Chemie (International ed. in English), 59(29), 11717–11731. [CrossRef]
- Sun, Y., Zhong, X., & Dennis, A. M. (2023). Minimizing near-infrared autofluorescence in preclinical imaging with diet and wavelength selection. Journal of biomedical optics, 28(9), 094805. [CrossRef]
- Dang, X., Bardhan, N. M., Qi, J., Gu, L., Eze, N. A., Lin, C. W., Kataria, S., Hammond, P. T., & Belcher, A. M. (2019). Deep-tissue optical imaging of near cellular-sized features. Scientific reports, 9(1), 3873. [CrossRef]
- Seah, D., Cheng, Z., & Vendrell, M. (2023). Fluorescent Probes for Imaging in Humans: Where Are We Now?. ACS nano, 17(20), 19478–19490. [CrossRef]
- Taruttis, A., & Ntziachristos, V. (2012). Translational optical imaging. AJR. American journal of roentgenology, 199(2), 263–271. [CrossRef]
- Zhou, L., Gan, Y., Wu, Y., Xue, D., Hu, J., Zhang, Y., Liu, Y., Ma, S., Zhou, J., Luo, G., Peng, D., & Qian, W. (2023). Indocyanine Green Fluorescence Imaging in the Surgical Management of Skin Squamous Cell Carcinoma. Clinical, cosmetic and investigational dermatology, 16, 3309–3320. [CrossRef]
- Achterberg, F. B., Deken, M. M., Meijer, R. P. J., Mieog, J. S. D., Burggraaf, J., van de Velde, C. J. H., Swijnenburg, R. J., & Vahrmeijer, A. L. (2020). Clinical translation and implementation of optical imaging agents for precision image-guided cancer surgery. European Journal of Nuclear Medicine and Molecular Imaging. [CrossRef]
- Rosenthal, E. L., Warram, J. M., de Boer, E., Basilion, J. P., Biel, M. A., Bogyo, M., Bouvet, M., Brigman, B. E., Colson, Y. L., DeMeester, S. R., Gurtner, G. C., Ishizawa, T., Jacobs, P. M., Keereweer, S., Liao, J. C., Nguyen, Q. T., Olson, J. M., Paulsen, K. D., Rieves, D., … van Dam, G. M. (2015). Successful Translation of Fluorescence Navigation During Oncologic Surgery: A Consensus Report. Journal of Nuclear Medicine. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




