1. Introduction
Bacillus bacteria such are soil-borne bacteria and are often contaminated final food products through the air and raw agricultural products such as grain, meat, milk and vegetable in food processing plants [
1]. Normally, almost bacteria contaminating in the food products are killed during heat cooking [
2]. However, since
Bacillus bacteria form heat tolerance spore,
Bacillus spore survives even through heat cooking and deteriorates the food products [
1,
2,
3,
4]. To extend shelf life of the food products, control of
Bacillus spore is important. Generally, to lose a risk of food deterioration caused by
Bacillus spore, retort sterilization above at 100 °C is subjected to the food products [
1,
2,
3,
4]. However, extensive heat treatment causes a deterioration of food quality such as taste, color, flavor, and nutritional value [
5]. Thus, novel technology which can kill
Bacillus spore at lower temperature of heat treatment has been demanded to minimize heat deterioration of food quality.
Although
Bacillus spore has high resistant to a various stress such as not only heat but also pressure, UV-C, oxidant and more, the germinated spore loses all of the high resistant [
1,
3,
4,
6,
7]. In this study, medium high hydrostatic pressure (MHHP) treatment at 100 MPa is focused as a nonthermal inducible method of
Bacillus spore germination. Many researchers have investigated MHHP germination induction of
B. subtilis spore and clarified a mechanism partially [
6,
7,
8,
9,
10,
11,
12]. In MHHP germination induction of
B. subtilis spore similarly to a path way of nutritional germination, germination receptors are activated and the spore begin to germinate when the spores are subjected to the MHHP treatment [
6,
7,
8,
9,
10,
11,
12]. The germinated spore by MHHP treatment lose the high resistant, which can be killed immediately even by heat treatment at 60-70 °C [
7,
9,
10,
11]. Thus, the MHHP germination technology can minimize a heat deterioration of the food quality. However, in the past study, it was clarified that the MHHP germination technology cannot germinate all of the spore, which ungerminated spore after MHHP treatment is called a super-dormant spore [
7,
9,
10,
12]. Therefore, to kill all of the spore, MHHP treatment and post-heat treatment at high temperature which can kill the spore must be conducted. Additionally, in general food processing, heat cooking is conducted before pasteurization process. However, an effect of pre-heating on pasteurization of MHHP treatment has been unclear.
In these backgrounds, this study aims to investigate pasteurization of Bacillus subtilis spore suspended in soy milk as a food model by combination of MHHP treatment with pre-/post-heat treatment and to clarify effect of post-/pre-heat treatment on pasteurization of B. subtilis spore by MHHP treatment via MHHP germination induction.
2. Materials and Methods
2.1. Bacterial Strain and Growth Condition
Bacillus subtilis NBRC 111470 as known 168 strain obtained from National Institute of Technology and Evaluation was used in this study. The strain was maintained at -80 °C in 0.9 % saline solution containing 20 % glycerol. After thawing, bacterial solution was inoculated in 20 ml of sporulation medium (pH 7.4) which contained 0.8 % Nutrient Broth (Difco TM Nutrient Broth, Becton, Dickinson and Company, U.S.A), 2.0 mM MgCl2, 1 mM CaCl2, 0.01 mM MnCl2, 1 mM FeCl3, 27.0 mM KCl (Wako Pure Chemical Industries, Ltd., Osaka, Japan). The culture was incubated at 37 °C with 130 rpm of agitation for 24 hours.
2.2. Plant Material Sample Preparation
After incubation, the culture was heated at 65 ˚C for 30 min in order to killing a vegetative cell. Spores were collected by centrifugation (3500 rpm, 10 min, 25 ˚C), and the resulting pellet was then resuspended in 40 ml of sterile distilled water. This procedure was repeated twice. After three times of centrifugation, the resulting pellet was resuspended in 5 ml of sterile distilled water. Then, the resuspension was heated at 65 ˚C for 30 min for killing vegetative cells. Subsequently, the spore resuspension was inoculated with 9-fold amount of soy milk which was purchased in a retail location. The spore solution of 1.5 ml was heat-sealed into each of sterile polystyrene bags (80 mm×80 mm) in order to utilize for MHHP treatment and pre-/post-heat treatment.
2.3. Pre-/Post-Heat Treatment
Each of the sample bag was subjected to immersing in a water bath (SB-1300, Tokyo Rikakikai Co., Ltd., Tokyo, Japan) at 80-90 ˚C and boiling to heat at 100 ˚C for performing pre-/post-heat treatment. After pre-/post-heat treatment, the heated sample was immediately chilled by tap water.
2.4. Medium High Hydrostatic Pressure Treatment
MHHP treatment was performed by a high-pressure food processor (Marugoto Ekisu EFS-0.5L, Toyo Kouatsu, Ltd., Hiroshima, Japan) using water as a pressure medium. Pressure medium temperature was maintained at 65 ˚C by inside heater of a pressure chamber (Φ 80 mm × D 140 mm). Untreated sample and pre-heated sample were immersed in the pressure chamber and pressurized up to 100 MPa at 1.1 MPa/sec, and this pressurization was maintained for 30 minutes. Then, the bag at 100 MPa was depressurized until ambient pressure at 10 MPa/sec. After decompression, the MHHP-treated sample was immediately chilled by tap water.
2.5. Bacterial Count and Calculation of D Value
The population of B. subtilis spores in all of the sample was determined by a direct plating method respectively. Samples serially diluted in sterile saline water, then, diluted solution of 0.1 ml was plated in duplicate on standard method agar (Nissui Pharmaceutical Co., Ltd., Tokyo, Japan), and the plates were incubated at 37 °C for 24 hours. The population of B. subtilis spore was calculated from a number of colony formation on the plate.
For calculating D value as known heat tolerance of bacteria, bacterial count of the four samples for different heat treatment time was approximated to a common logarithmic decrease of straight line utilizing least squares method. For calculating Z value, D value at each of heat treatment temperature was approximated to a common logarithmic decrease of straight line. Additionally, for calculating activation energy (
Ea (J·mol
-1)) in Arrhenius equation as shown Eq (1), rate constant of bacterial inactivation (
k) at each of heat treatment temperature was approximated to a natural logarithm decrease of straight line.
which
R is gas constant and 8.3 J·mol
-1·K
-1,
T is absolute temperature of heat treatment (K) and
a is frequency factor.
3. Results and Discussions
3.1. Pasteurization of B. subtilis Spore by Combination of Post-Heat Treatment with and Without MHHP Treatment
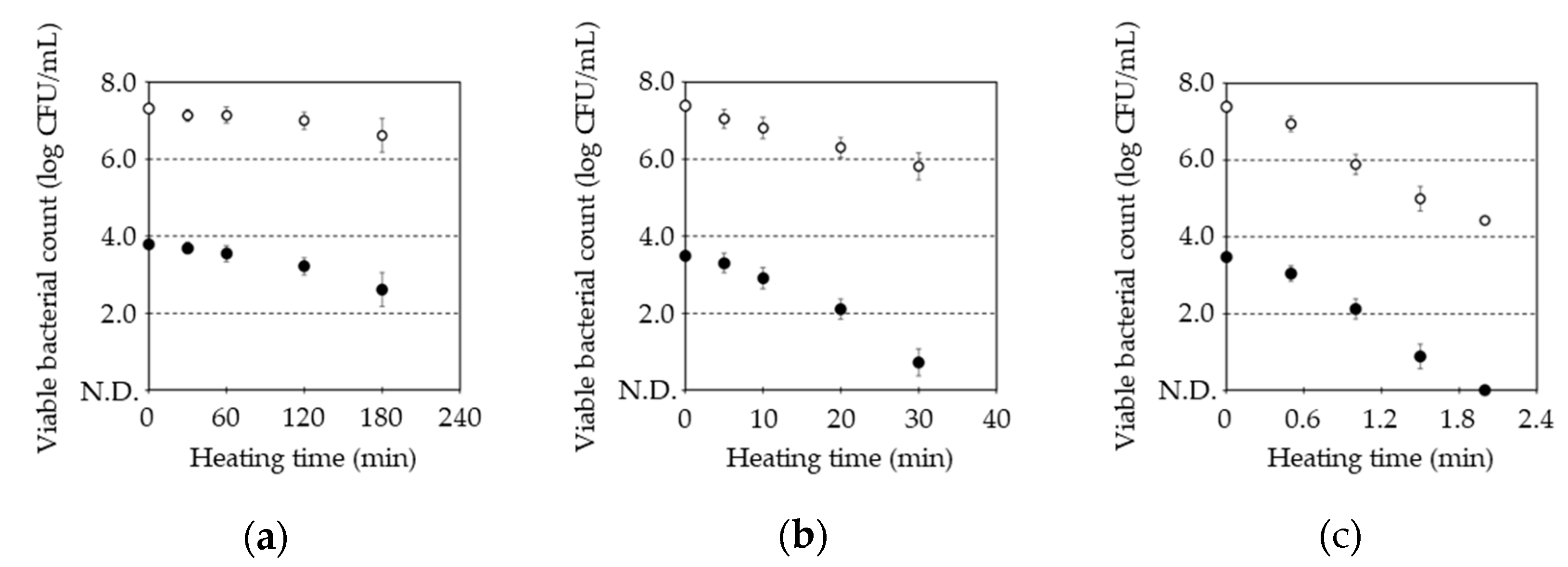
Figure 1 shows pasteurization of
B. subtilis spore by combination of post-heat treatment at 80 °C, 87 °C, 100 °C with and without MHHP treatment. Initial bacterial count of
B. subtilis spore treated with and without MHHP treatment for 30 min was 7.4±0.1 and 3.6±0.2 log CFU/mL. Heat treatment at 80 °C from 30 min to 180 min decreased from 7.1±0.1 to 6.6±0.1 log CFU/mL of the untreated spore and from 3.7±0.1 to 2.6±0.3 log CFU/mL of the MHHP-treated spore as shown in
Figure 1a, respectively. Heat treatment at 87 °C from 5 min to 30 min decreased from 7.1±0.1 to 5.9±0.1 log CFU/mL of the untreated spore and from 3.4±0.2 to 1.1±0.5 log CFU/mL of the MHHP-treated spore as shown in
Figure 1b, respectively. Heat treatment at 100 °C from 0.5 min to 2 min decreased from 6.9±0.0 to 4.4±0.1 log CFU/mL of the untreated spore and from 3.0±0.1 log CFU/mL to no detection level of the MHHP-treated spore as shown in
Figure 1c, respectively. Decrease in the bacterial count was linear line with extension of heating time regardless of heating temperature, respectively.
Table 1 shows D value, Z value and activation energy for heat pasteurization of
B. subtilis spore with and without MHHP treatment. D value of the untreated spore was significant less than that of the MHHP-treated spore, respectively. Whereas, Z value and activation energy had no significant difference between the untreated and the MHHP-treated spore. Ogino and Nishiumi reported that MHHP treatment at 200 MPa penetrated moisture into the
Bacillus subtilis spore [
13]. Thus, MHHP at 100 MPa could penetrate moisture into the spore, which resulted in increase in heat conductivity of the spore and decrease in the D value. On the other hand, since heat conductivity to the spore only increased, activation energy required for killing the spore could be maintained.
3.2. Germination Induction Effect of Combination of MHHP Treatment With and Without Pre-Heat Treatment on B. subtilis Spore
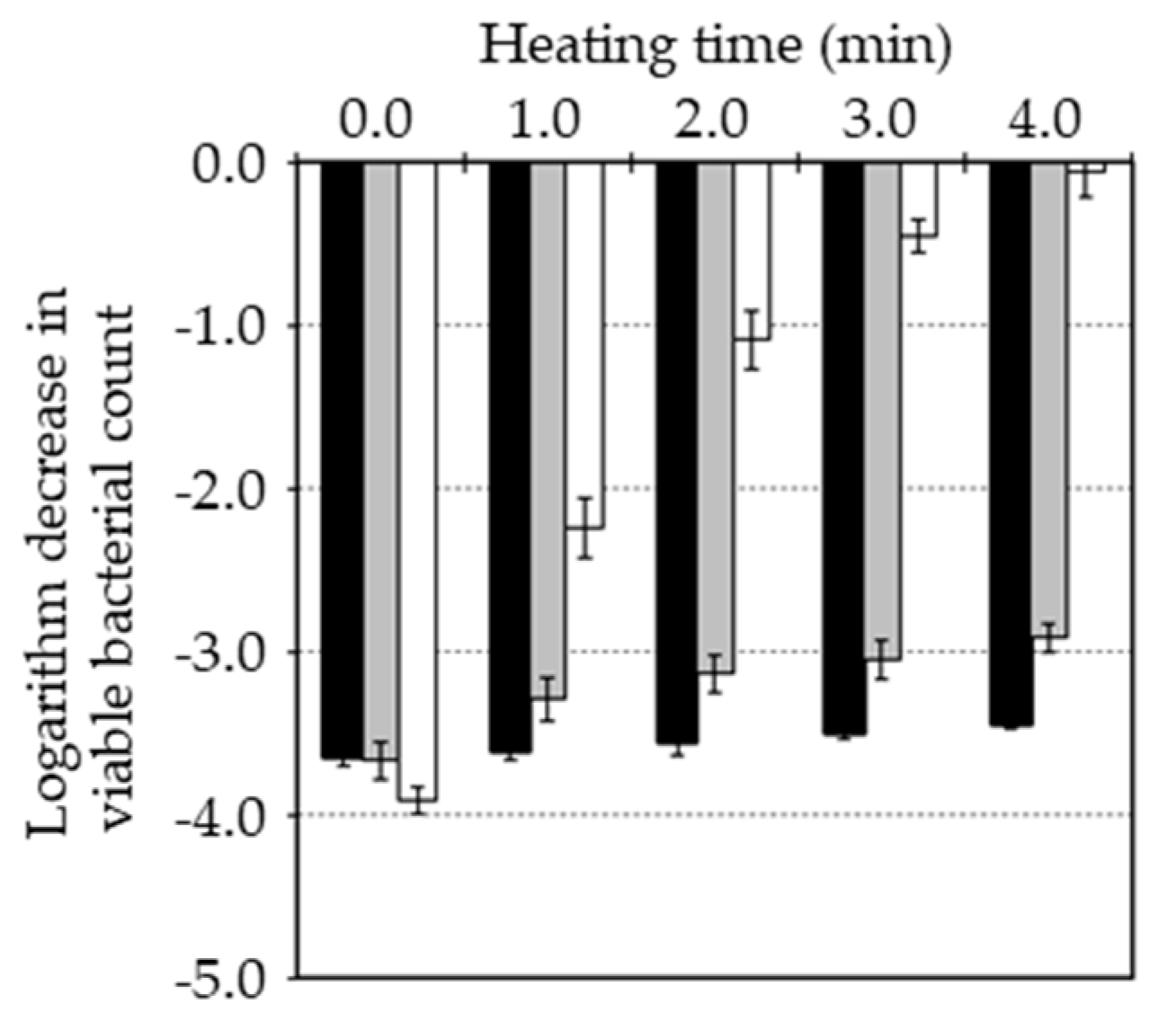
Figure 2 shows pasteurization of
B. subtilis spore after combination of MHHP treatment with and without pre-heat treatment at 80 °C, 90 °C, 100 °C for 1-4 min. Pasteurization effect of MHHP treatment on the untreated spore was 3.9 of logarithm reduction. Pasteurization effect of MHHP treatment on the pre-heated spore at 80 °C from 1 min to 4min was from 3.9 to 3.1 of logarithm reduction. Pasteurization effect of MHHP treatment on the pre-heated spore at 90 °C from 1 min to 4min was from 3.4 to 3.1 of logarithm reduction. Pasteurization effect of MHHP treatment on the pre-heated spore at 100 °C from 1 min to 4min was from 2.0 to 0.0 of logarithm reduction. Therefore, pre-heating and extending pre-heating time suppressed pasteurization effect of MHHP treatment. Especially, pre-heating at 100 °C suppressed the pasteurization effect considerably and finally to lose. However, since pre-heating at 100 °C inactivated the spore considerably, inactivation of spore might have an effect on the pasteurization by MHHP treatment. Thus, when inactivation of the spore after pre-heating was equivalent, pasteurization of
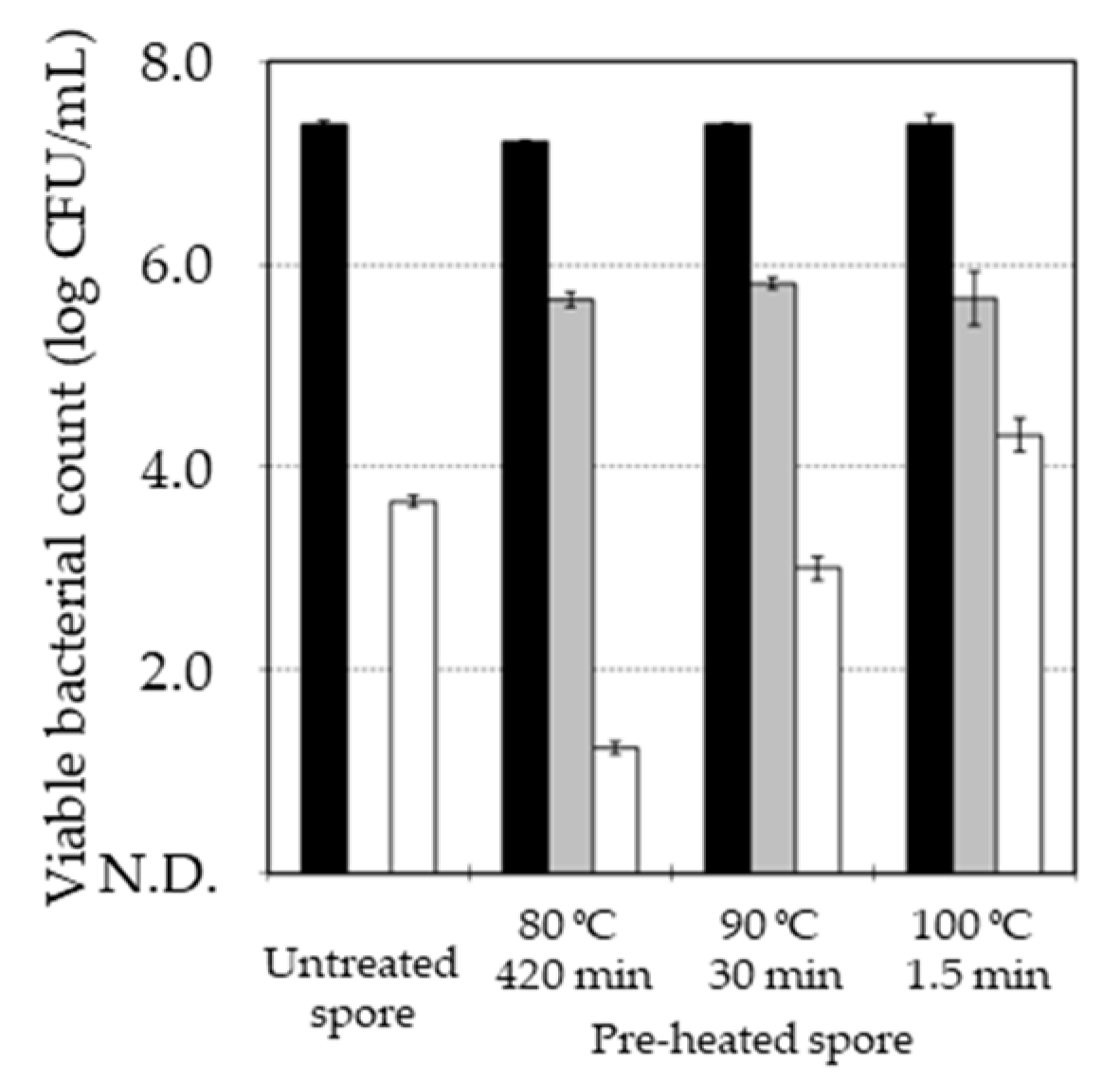
B. subtilis spore after combination of MHHP treatment was compared among the pre-heated spore at 80 °C, 90 °C, 100 °C as shown in
Figure 3. Pre-heating decreased from 7.4±0.1 log CFU/ml to 5.6±0.1 log CFU/ml at 80 °C for 420 min, to 5.8±0.1 log CFU/ml at 90 °C for 30 min and to 5.7±0.5 log CFU/ml at 100 °C for 1.5 min. Pasteurization effect of MHHP treatment was 4.5, 2.8, 1.4 of logarithm reduction for the pre-heated spore at 80 °C, 90 °C, 100 °C. Therefore, since pasteurization effect was 3.7 of logarithm reduction for the untreated spore, pre-heat treatment at 80 °C increased the pasteurization by MHHP treatment while pre-heating at 90 °C and 100 °C decreased that. The pasteurization by MHHP treatment can be achieved via MHHP germination induction of the spore[
6,
7,
8,
9,
10,
11,
12]. In process of MHHP germinate induction of the spore, MHHP treatment can firstly stimulate germination receptors and the germination can be advanced [
6,
7,
8,
9,
10,
11,
12]. Additionally, heating at 60-80 °C can activate germination receptors and promote germination of the spore while heating at higher temperature can inactivate germination receptors and suppress germination of the spore [
14,
15,
16]. Therefore, a difference among effect of pre-heating temperature on MHHP pasteurization could be caused by activation/inactivation of germination receptors.
3.3. Effect of Pre-Heat Treatment on Decrease in Heat Tolerance of B. subtilis Spore by MHHP Treatment
Table 2 shows effect of pre-heat treatment at 100 °C on decrease in heat tolerance of
B. subtilis spore at 87 °C by MHHP treatment. Although pre-heat treatment at 100 °C suppressed pasteurization of
B. subtilis spore by MHHP treatment via MHHP germination, it was unclear and investigated that the pre-heat treatment could suppress decrease in heat tolerance of
B. subtilis spore by MHHP treatment. D value of the untreated spore, the pre-heated spore, the MHHP-treated spore and the pre-heated and MHHP-treated spore was 23±4 min, 29±4 min, 11±2 min and 14±1 min. Significant difference was observed between the spore with and without MHHP treatment regardless of pre-heat treatment (
p<0.05). Thus, pre-heat treatment could have a significant effect on decrease in heat tolerance of
B. subtilis spore by MHHP treatment. While, the pre-heated spores tended to increase the D value regardless of MHHP treatment. Although heat shock at 100 °C might increase the heat tolerance at 87 °C of the spore, the mechanism was unclear. Further study focusing on effect of heat shock on heat tolerance of
B. subtilis spore is required.
4. Conclusion
In this study, effect of post-/pre-heat treatment on pasteurization of B. subtilis spore by MHHP treatment via MHHP germination induction was investigated. When the MHHP-treated spore was subjected to post-heat treatment, D value, as shown indicator of heat tolerance, of the ungerminated spore after MHHP treatment was decreased in comparison with the untreated spore. Whereas, Z value and activation energy of the untreated and the MHHP-treated spore were equivalent, which indicated that MHHP treatment might increase heat conductivity of the ungerminated spore. Therefore, MHHP treatment could have the effect of not only loss of heat tolerance of the spore via MHHP germination induction but also decrease in heat tolerance. When the spore was subjected to pre-heat treatment and subsequently to MHHP treatment, pasteurization effect of MHHP treatment was differed to the heating temperature. Pre-heat treatment at 80 °C promote the pasteurization while that at 90-100 °C, which might be caused by heat activation/inactivation of germination receptors. From these results, combination of MHHP treatment with post-/pre-heat treatment could be effective for pasteurization of B. subtilis in soy milk although pre-heat treatment was limited lower temperature than 80 °C.
Author Contributions
Conceptualization, M.K.; methodology, M.K.; software, M.K.; validation, M.K.; formal analysis, M.K.; investigation, M.K.; resources, M.K.; data curation, M.K.; writing—original draft preparation, M.K.; writing—review and editing, M.K.; visualization, M.K.; supervision, M.K.; project ad-ministration, M.K.; funding acquisition, M.K.
Funding
This research was funded by the Tojuro Iijima Foundation for Food Science and Technology, H27-41 and JSPS KAKENHI Grant Number JP 22K05909. The APC was funded by grant from Ehime University.
Data Availability Statement
Data are contained within the article.
Acknowledgments
I am thankful to Toyo Koatsu Co., Ltd. For their technical assistance in MHHP treatment.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Andersson, A.; Rönner, U.; Granum, P. E. What problems does the food industry have with the spore-forming pathogens Bacillus cereus and Clostridium perfringens?. Int. J. Food Microbiol. 1995, 28, 145-155.
- Guillermo, C.; Santiago, C.; Pilar, M. Physiology of the inactivation of vegetative bacteria by thermal treatments: mode of action, influence of environmental factors and inactivation kinetics. Foods 2017, 6, 107.
- Nicholson, W.L.; Munakata, N.; Horneck, G.; Melosh, H.J.; Setlow, P. Resistance of Bacillus endospores to extreme terrestrial and extraterrestrial environments. Microbiol. Mol. Biol. Rev. 2000, 64, 548-572.
- Setlow, P. Spores of Bacillus subtilis: their resistance to and killing by radiation, heat and chemicals. J. Appl. Microbiol. 2006, 101, 514-525.
- Ross, A.I.V.; Griffiths, M.W.; Mittal, G.S.; Deeth, H.C. Combining nonthermal technologies to control foodborne microorganisms. Int. J. Food Microbiol. 2003, 89, 125-138.
- Black, E.P.; Koziol-Dube, K.; Guan, D.; Wei, J.; Setlow, B.; Cortezzo, D.E.; Hoover, D.G.; Setlow, P. Factors influencing germination of Bacillus subtilis spores via activation of nutrient receptors by high pressure. Appl. Environ. Microbiol. 2005, 71, 5879-5887.
- Black, E.P.; Setlow, P.; Hocking, A.D.; Stewart, C.M.; Kelly, A.L.; Hoover, D.G. Response of spores to high-pressure processing. Compr. Rev. Food Sci. Food Saf. 2007, 6, 103-119.
- Paidhungat, M.; Setlow, B.; Daniels, W.B.; Hoover, D.; Papafragkou, E.; Setlow, P. Mechanisms of induction of germination of Bacillus subtilis spores by high pressure. Appl. Environ. Microbiol. 2002, 68. 3172-3175.
- Reineke, K; Doehner, I.; Schlumbach, K.; Baier, D.; Mathys, A.; Knorr, D. The different pathways of spore germination and inactivation in dependence of pressure and temperature. Innov. Food Sci. Emerg. Technol. 2012, 13, 31-41.
- Reinek, K.; Mathys, A.; Heinz, V.; Knorr, D. Mechanisms of endospore inactivation under high pressure. Trends Microbiol. 2013, 21, 296-304.
- Wuytack, E.Y.; Boven, S.; Michiels, C.W. Comparative study of pressure-induced germination of Bacillus subtilis spores at low and high pressures. Appl. Environ. Microbiol. 1998, 64, 3220-3224.
- Wuytack, E.Y.; Soons, J.; Poschet, F.; Michiels, C.W. Comparative study of pressure- and nutrient-induced germination of Bacillus subtilis spores. Appl. Environ. Microbiol. 2000, 66, 257-261.
- Ogino, H.; Nishiumi, T. Sterilization of heat-resistant spores by a combination of high-pressure and subsequent heat treatment (in Japanese). Rev. High Press. Sci. Technol. 2015. 25, 334-342.
- Berg, R.W.; Sandine, W.E. Activation of bacterial spores. A review. J. Food Prot. 1970. 33, 435-441.
- Luu, S.; Cruz-Mora, J.; Setlow, B.; Feeherry, F.E.; Doona, C.J; Setlow, P. The effects of heat activation on Bacillus spore germination, with nutrients or under high pressure, with or without various germination proteins. Appl. Environ. Microbiol. 2002, 68. 3172-3175.
- Wen, J; Smelt, J.P.P.M; Vischer, N.O.E.; Vos, A.L.; Setlow, P.; Brul, S. Heat activation and inactivation of bacterial spores: Is there an overlap?. Appl. Environ. Microbiol. 2022, 88, e02324-21.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).