Submitted:
29 November 2024
Posted:
02 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
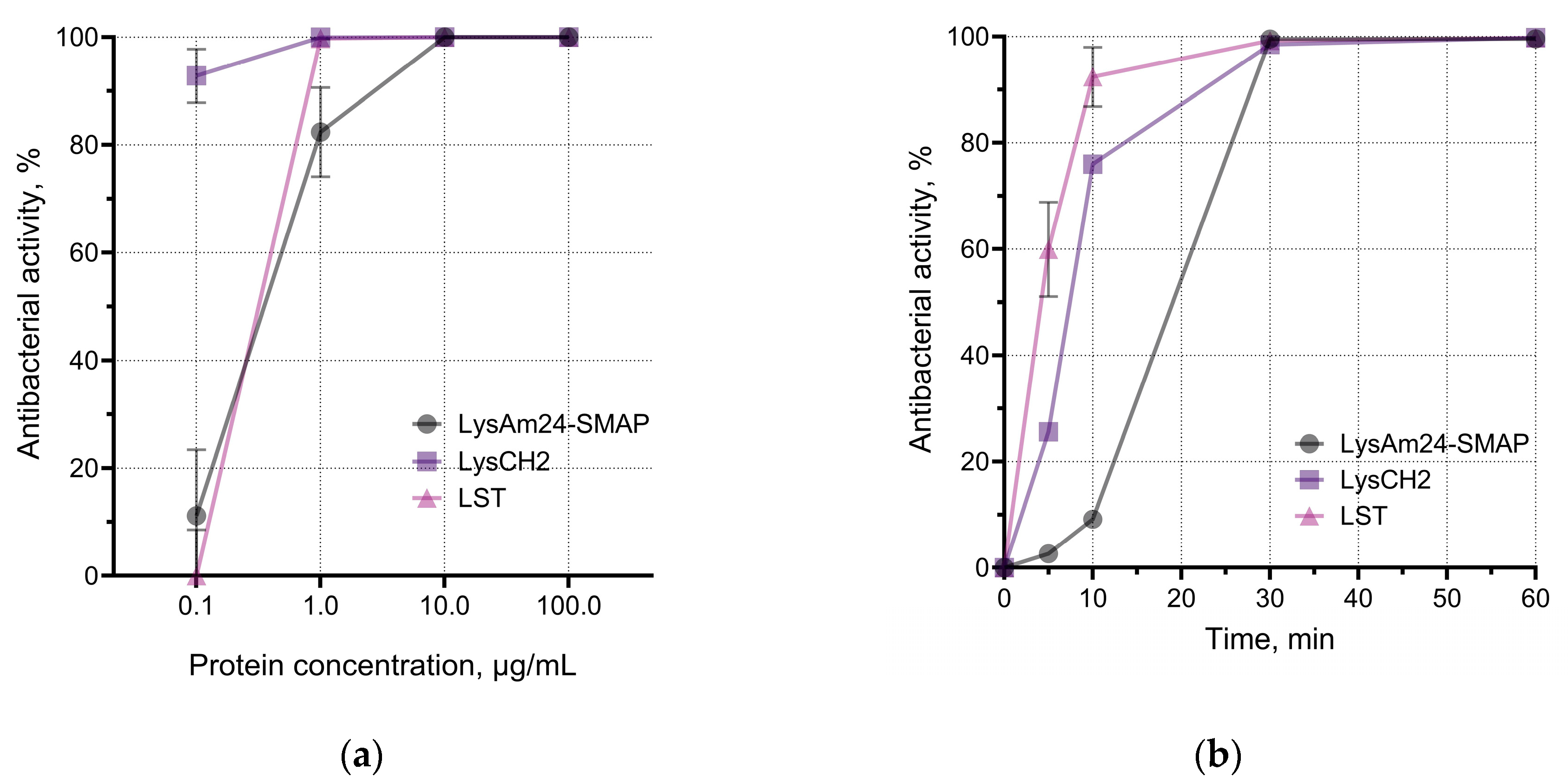
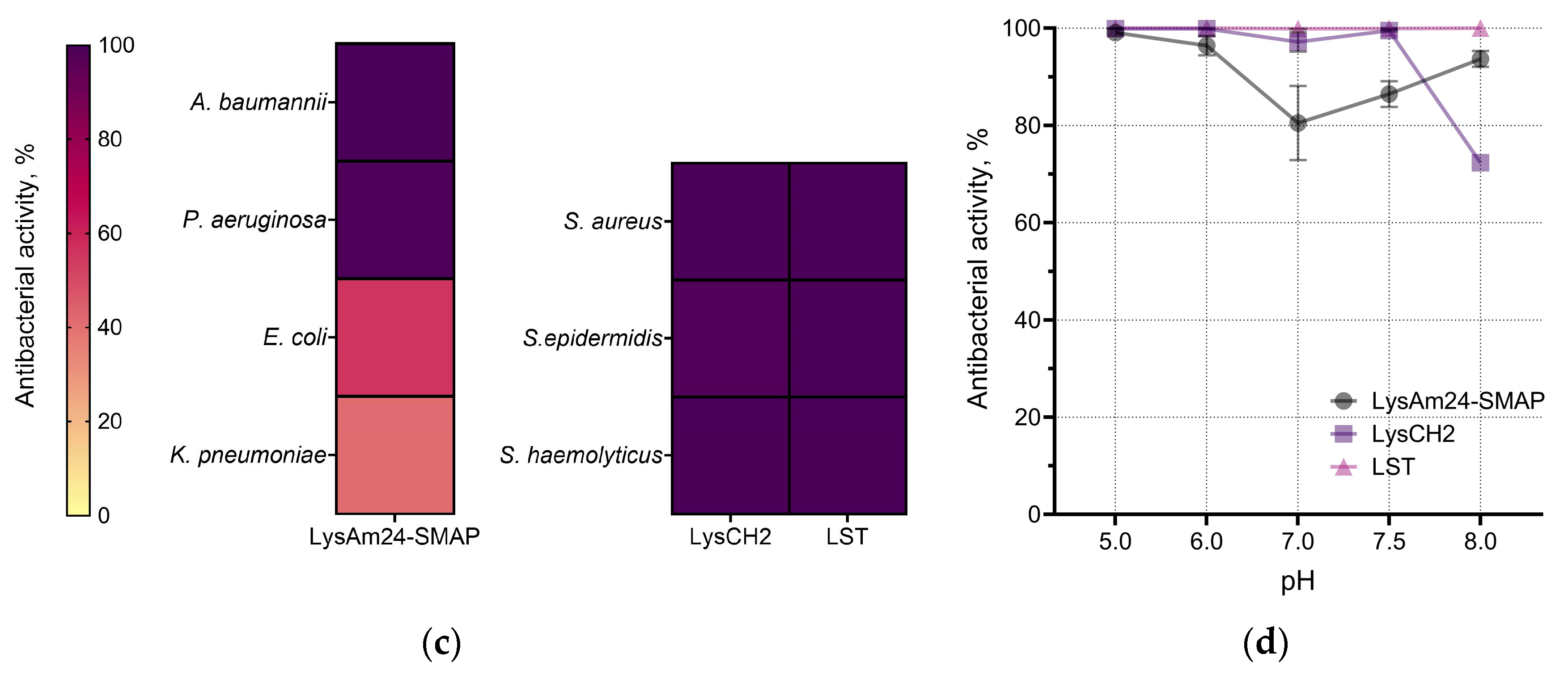
2.1. Antibacterial Properties of Bacteriolytic Enzymes
2.2. Enzymes Combinations Effects on Planktonic Bacteria and Biofilms
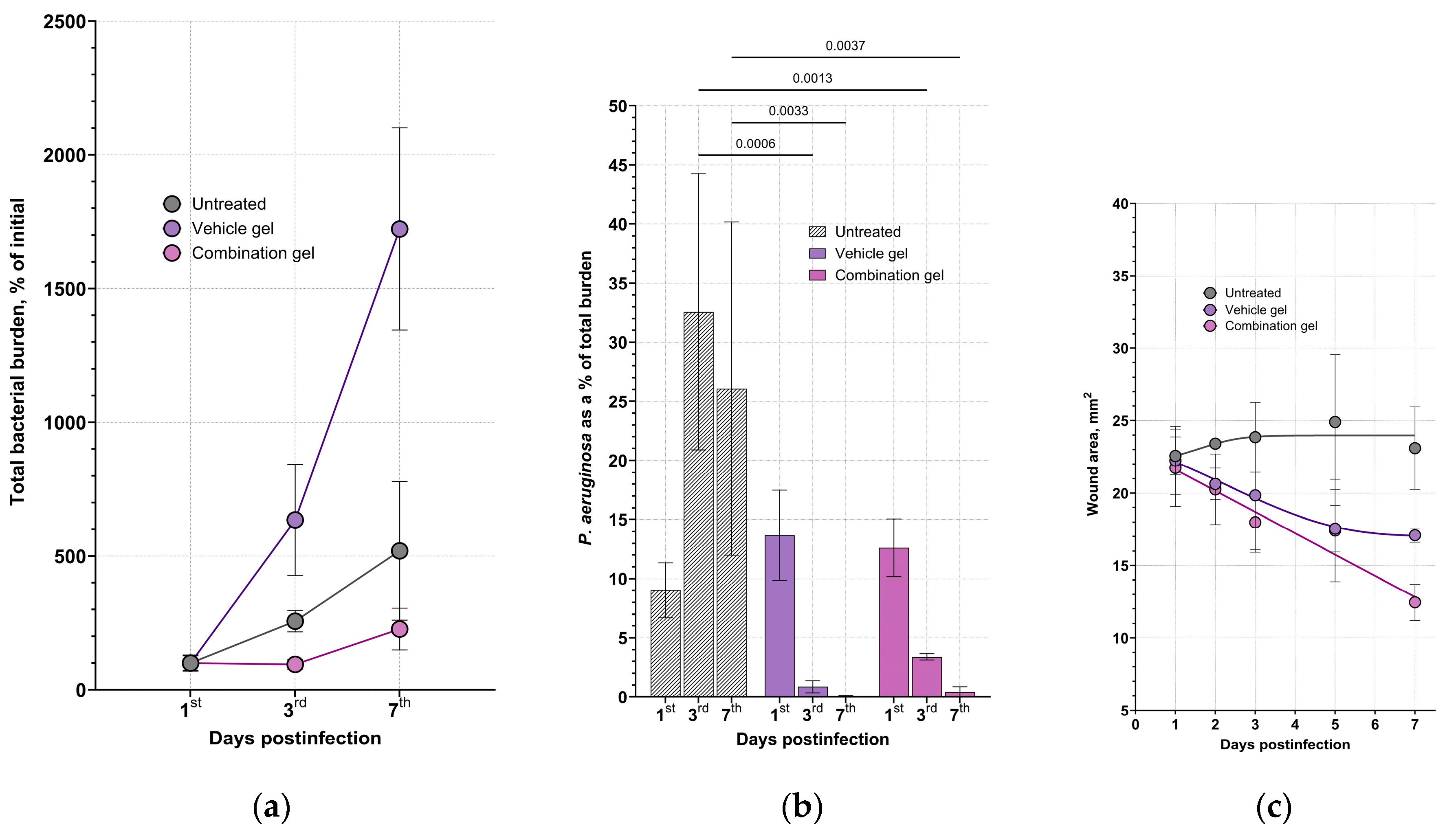
2.3. In Vivo Evaluation of Combination Against Biofilm Wound Infection
3. Materials and Methods
3.1. Recombinant Proteins Expression and Purification
3.2. Bacterial Strains
3.3. Enzymes Activity Assay
3.4. Minimum Inhibitory Concentrations Estimation and Checkerboard Assay
3.5. Biofilms Formation and Enzymes Antibiofilm Activity
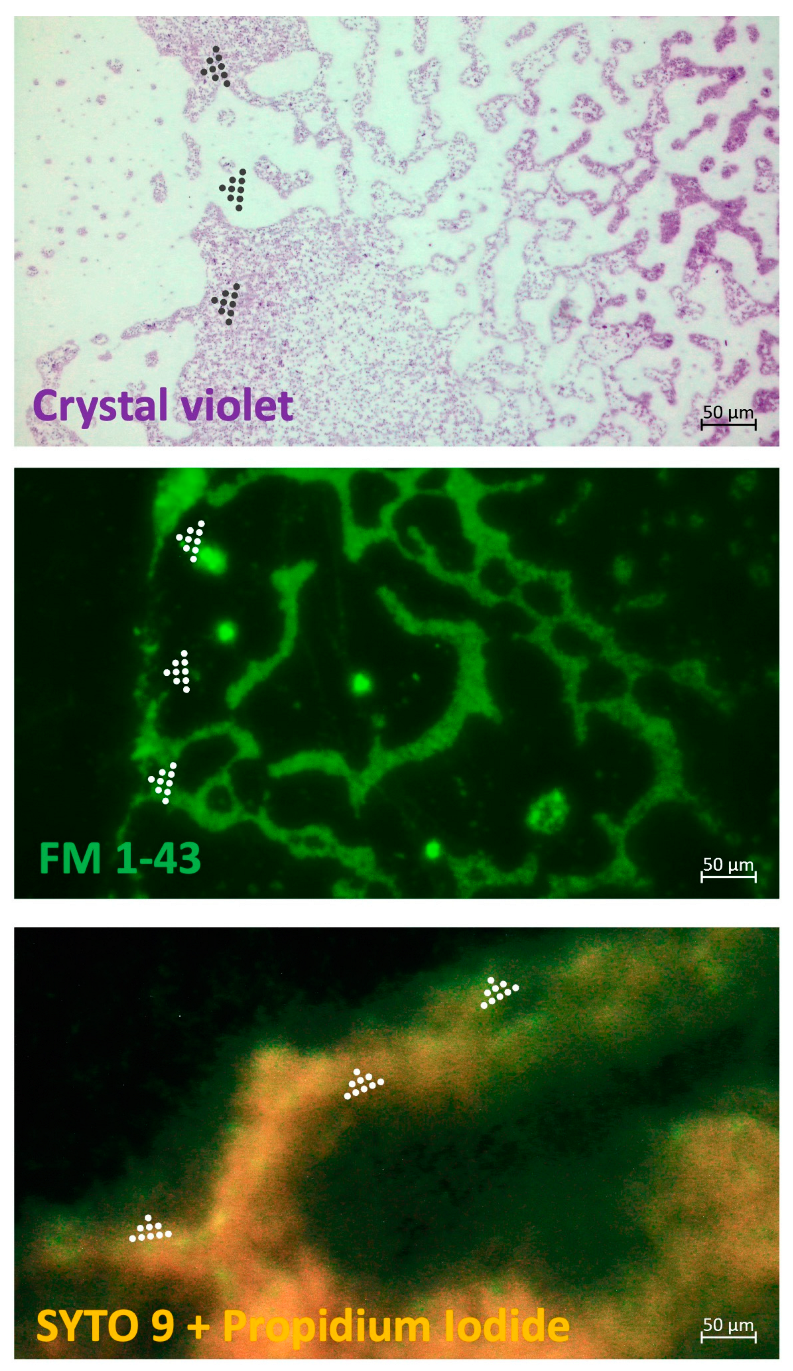
3.6. Microscopy
3.7. In Vivo Assessment of Enzymes Combination Using Polymicrobial Biofilm Wound Infection Model
4. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Global Action Plan on Antimicrobial Resistance. World Health Organization, Geneva, 2015.
- WHO Bacterial Priority Pathogens List, 2024: Bacterial Pathogens of Public Health Importance to Guide Research, Development and Strategies to Prevent and Control Antimicrobial Resistance; World Health Organization, Geneva, 2024.
- Antibacterial Agents in Clinical and Preclinical Development an Overview and Analysis; World Health Organization, Geneva, 2024.
- Liu, H.; Hu, Z.; Li, M.; Yang, Y.; Lu, S.; Rao, X. Therapeutic Potential of Bacteriophage Endolysins for Infections Caused by Gram-Positive Bacteria. J Biomed Sci 2023, 30. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Das, R.; Chavan, B.; Bajpai, U.; Hanif, S.; Ahmed, S. Beyond Antibiotics: Phage-Encoded Lysins against Gram-Negative Pathogens. Front Microbiol 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Hibbert, T.; Krpetic, Z.; Latimer, J.; Leighton, H.; McHugh, R.; Pottenger, S.; Wragg, C.; James, C.E. Antimicrobials: An Update on New Strategies to Diversify Treatment for Bacterial Infections. Adv Microb Physiol 2024, 84, 135–241. [Google Scholar] [CrossRef]
- Gontijo, M.; Teles, M.P.; Correia, H.M.; Jorge, G.P.; Rodrigues Santos Goes, I.C.; Flores, A.J.F.; Braz, M.; de Moraes Ceseti, L.; Ramos, P.Z.; E Silva, I.R.; et al. Combined Effect of SAR-Endolysin LysKpV475 with Polymyxin B and Salmonella Bacteriophage PhSE-5. Microbiology (United Kingdom) 2024, 170. [Google Scholar] [CrossRef]
- Lu, H.; Li, Z.; Elbaz, A.; Ni, S.Q. Synergistic Action of Phages and Lytic Proteins with Antibiotics: A Combination Strategy to Target Bacteria and Biofilms. BMC Microbiol 2023, 23. [Google Scholar] [CrossRef]
- Hong, H.W.; Kim, Y.D.; Jang, J.; Kim, M.S.; Song, M.; Myung, H. Combination Effect of Engineered Endolysin EC340 With Antibiotics. Front Microbiol 2022, 13. [Google Scholar] [CrossRef]
- Blasco, L.; Ambroa, A.; Trastoy, R.; Bleriot, I.; Moscoso, M.; Fernández-Garcia, L.; Perez-Nadales, E.; Fernández-Cuenca, F.; Torre-Cisneros, J.; Oteo-Iglesias, J.; et al. In Vitro and in Vivo Efficacy of Combinations of Colistin and Different Endolysins against Clinical Strains of Multi-Drug Resistant Pathogens. Sci Rep 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.H.; Park, W.B.; Cho, J.E.; Choi, Y.J.; Choi, S.J.; Jun, S.Y.; Kang, C.K.; Song, K.H.; Choe, P.G.; Bang, J.H.; et al. Effects of Phage Endolysin SAL200 Combined with Antibiotics on Staphylococcus Aureus Infection. Antimicrob Agents Chemother 2018, 62, e00731–18. [Google Scholar] [CrossRef]
- Daniel, A.; Euler, C.; Collin, M.; Chahales, P.; Gorelick, K.J.; Fischetti, V.A. Synergism between a Novel Chimeric Lysin and Oxacillin Protects against Infection by Methicillin-Resistant Staphylococcus Aureus. Antimicrob Agents Chemother 2010, 54, 1603–1612. [Google Scholar] [CrossRef] [PubMed]
- Fowler, V.G.; Das, A.F.; Lipka-Diamond, J.; Ambler, J.E.; Schuch, R.; Pomerantz, R.; Cassino, C.; Jáuregui-Peredo, L.; Moran, G.J.; Rupp, M.E.; et al. Exebacase in Addition to Standard-of-Care Antibiotics for Staphylococcus Aureus Bloodstream Infections and Right-Sided Infective Endocarditis: A Phase 3, Superiority-Design, Placebo-Controlled, Randomized Clinical Trial (DISRUPT). Clinical Infectious Diseases 2024, 78, 1473–1481. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, X.; Liang, S.; Wang, J.; Zhu, Y.; Zhang, W.; Liu, S.; Schwarz, S.; Xie, F. Bactericidal Synergism between Phage Endolysin Ply2660 and Cathelicidin LL-37 against Vancomycin-Resistant Enterococcus Faecalis Biofilms. NPJ Biofilms Microbiomes 2023, 9. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, J.L.; Gupta, P.; Ghate, M.M.; Kumar, D.; Poluri, K.M. Assessing the Synergistic Potential of Bacteriophage Endolysins and Antimicrobial Peptides for Eradicating Bacterial Biofilms. Arch Microbiol 2024, 206. [Google Scholar] [CrossRef] [PubMed]
- Loeffler, J.M.; Fischetti, V.A. Synergistic Lethal Effect of a Combination of Phage Lytic Enzymes with Different Activities on Penicillin-Sensitive and -Resistant Streptococcus Pneumoniae Strains. Antimicrob Agents Chemother 2003, 47, 375–357. [Google Scholar] [CrossRef]
- Becker, S.C.; Foster-Frey, J.; Donovan, D.M. The Phage K Lytic Enzyme LysK and Lysostaphin Act Synergistically to Kill MRSA. FEMS Microbiol Lett 2008, 287, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Vasina, D. V.; Antonova, N.P.; Shidlovskaya, E. V.; Kuznetsova, N.A.; Grishin, A. V.; Akoulina, E.A.; Trusova, E.A.; Lendel, A.M.; Mazunina, E.P.; Kozlova, S.R.; et al. Alginate Gel Encapsulated with Enzybiotics Cocktail Is Effective against Multispecies Biofilms. Gels 2024, 10. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Robles Aguilar, G.; Gray, A.; Han, C.; Bisignano, C.; Rao, P.; Wool, E.; et al. Global Burden of Bacterial Antimicrobial Resistance in 2019: A Systematic Analysis. The Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- Danis-Wlodarczyk, K.M.; Wozniak, D.J.; Abedon, S.T. Treating Bacterial Infections with Bacteriophage-Based Enzybiotics: In Vitro, in Vivo and Clinical Application. Antibiotics 2021, 10. [Google Scholar] [CrossRef]
- Antonova, N.P.; Vasina, D. V.; Lendel, A.M.; Usachev, E. V.; Makarov, V. V.; Gintsburg, A.L.; Tkachuk, A.P.; Gushchin, V.A. Broad Bactericidal Activity of the Myoviridae Bacteriophage Lysins LysAm24, LysECD7, and LysSi3 against Gram-Negative ESKAPE Pathogens. Viruses 2019, 11. [Google Scholar] [CrossRef]
- Vasina, D. V.; Antonova, N.P.; Grigoriev, I. V.; Yakimakha, V.S.; Lendel, A.M.; Nikiforova, M.A.; Pochtovyi, A.A.; Remizov, T.A.; Usachev, E. V.; Shevlyagina, N. V.; et al. Discovering the Potentials of Four Phage Endolysins to Combat Gram-Negative Infections. Front Microbiol 2021, 12. [Google Scholar] [CrossRef]
- O’Flaherty, S.; Coffey, A.; Meaney, W.; Fitzgerald, G.F.; Ross, R.P. The Recombinant Phage Lysin LysK Has a Broad Spectrum of Lytic Activity against Clinically Relevant Staphylococci, Including Methicillin-Resistant Staphylococcus Aureus. J Bacteriol 2005, 187, 7161–7164. [Google Scholar] [CrossRef]
- Horgan, M.; O’Flynn, G.; Garry, J.; Cooney, J.; Coffey, A.; Fitzgerald, G.F.; Paul Ross, R.; McAuliffe, O. Phage Lysin LysK Can Be Truncated to Its CHAP Domain and Retain Lytic Activity against Live Antibiotic-Resistant Staphylococci. Appl Environ Microbiol 2009, 75, 872–874. [Google Scholar] [CrossRef] [PubMed]
- Bastos, M. do C. de F.; Coutinho, B.G.; Coelho, M.L.V. Lysostaphin: A Staphylococcal Bacteriolysin with Potential Clinical Applications. Pharmaceuticals 2010, 3, 1139–1161. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, H.; São-José, C.; Azeredo, J. Phage-Derived Peptidoglycan Degrading Enzymes: Challenges and Future Prospects for in Vivo Therapy. Viruses 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Lendel, A.M.; Antonova, N.P.; Grigoriev, I. V.; Usachev, E. V.; Gushchin, V.A.; Vasina, D. V. Biofilm-Disrupting Effects of Phage Endolysins LysAm24, LysAp22, LysECD7, and LysSi3: Breakdown the Matrix. World J Microbiol Biotechnol 2024, 40. [Google Scholar] [CrossRef] [PubMed]
- Vasina, D. V.; Antonova, N.P.; Gushchin, V.A.; Aleshkin, A. V.; Fursov, M. V.; Fursova, A.D.; Gancheva, P.G.; Grigoriev, I. V.; Grinkevich, P.; Kondratev, A. V.; et al. Development of Novel Antimicrobials with Engineered Endolysin LysECD7-SMAP to Combat Gram-Negative Bacterial Infections. J Biomed Sci 2024, 31. [Google Scholar] [CrossRef] [PubMed]
- European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Dieases (ESCMID). EUCAST Definitive Document E.Def 1.2, May 2000: Terminology relating to methods for the determination of susceptibility of bacteria to antimicrobial agents. Clin Microbiol Infect 2000, 6, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Iaconis, A.; De Plano, L.M.; Caccamo, A.; Franco, D.; Conoci, S. Anti-Biofilm Strategies: A Focused Review on Innovative Approaches. Microorganisms 2024, 12. [Google Scholar] [CrossRef]
- Lin, Q.; Sheng, M.; Tian, Y.; Li, B.; Kang, Z.; Yang, Y.; Xu, Z.; Soteyome, T.; Guo, L.; Sun, H.; et al. Antibiofilm Activity and Synergistic Effects of DNase I and Lysostaphin against Staphylococcus Aureus Biofilms. Food Quality and Safety 2024, 8. [Google Scholar] [CrossRef]
- Vandecasteele, S.J.; Peetermans, W.E.; Merckx, R.; Van Eldere, J. Expression of Biofilm-Associated Genes in Staphylococcus Epidermidis during in Vitro and in Vivo Foreign Body Infections. J Infect Dis 2003, 188, 730–737. [Google Scholar] [CrossRef]
- Vandecasteele, S.J.; Peetermans, W.E.; Merckx, R.; Van Eldere, J. Quantification of Expression of Staphylococcus Epidermidis Housekeeping Genes with Taqman Quantitative PCR during in Vitro Growth and under Different Conditions. J Bacteriol 2001, 183, 7094–7101. [Google Scholar] [CrossRef]
- Wiegand, I.; Hilpert, K.; Hancock, R.E.W. Agar and Broth Dilution Methods to Determine the Minimal Inhibitory Concentration (MIC) of Antimicrobial Substances. Nat Protoc 2008, 3, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Stepanovic, S.; Vukovic, D.; Hola, V.; Bonaventura, G. DI; Djukic, S.; Ruzicka, F.; Bonaventura, D.G. Quantification of Biofilm in Microtiter Plates: Overview of Testing Conditions and Practical Recommendations for Assessment of Biofilm Production by Staphylococci. APMIS 2007, 115, 891–900. [Google Scholar] [CrossRef] [PubMed]
- Dalton, T.; Dowd, S.E.; Wolcott, R.D.; Sun, Y.; Watters, C.; Griswold, J.A.; Rumbaugh, K.P. An in Vivo Polymicrobial Biofilm Wound Infection Model to Study Interspecies Interactions. PLoS One 2011, 6. [Google Scholar] [CrossRef] [PubMed]
| Bacterial strain |
S. aureus ATCC 29213 |
S. aureus ATCC 25923 |
A. baumannii ATCC 19606 |
P. aeruginosa АТСС 9027 |
K. pneumoniae ATCC 10031 |
E. coli ATCC 25922 |
| MIC | ||||||
| LysAm24-SMAP | - | - | 64 µg/mL | n/a | n/a | n/a |
| LysCH2 | n/a | n/a | - | - | - | - |
| LST | 0.5 µg/mL | n/a | - | - | - | - |
| MBC | ||||||
| LysAm24-SMAP | - | - | 64 µg/mL | n/a | n/a | 1024 µg/mL |
| LysCH2 | n/a | 128 µg/mL | - | - | - | - |
| LST | n/a | n/a | - | - | - | - |
| ΣFBCmin | ||||||
|
LysAm24-SMAP + LysCH2 |
0.53 (ADD) | 0.53 (ADD) | 1.03 (IND) | n/a | 0.75 (ADD) | 0.63 (ADD) |
|
LysAm24-SMAP + LST |
0.15 (SYN) | 0.06 (SYN) | 1.03 (IND) | n/a | n/a | 1.5 (IND) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




