1. Introduction
Bacteria are often regarded as highly adaptable to extreme environmental changes, including rising temperatures driven by global warming. However, this perception oversimplifies the complexity of bacterial adaptation and does not account for the millions of years it has taken for the diversity of bacteria to adapt to particular niches. This highlights that their adaptive capacity is deeply rooted in long evolutionary processes.
Understanding bacterial responses to heat stress is critical, given their central role in nutrient cycling and ecosystem stability, especially as global temperatures are projected to increase by 2 to 4°C [
1,
2]. The impact of rising temperatures extends beyond bacterial physiology, with significant consequences for ecosystems and biogeochemical cycles. For example, elevated ocean temperatures alter nitrogen-fixing microbial populations, threatening the nitrogen cycle in coral reefs [
3]. Similarly, warming freshwater ecosystems decreases bacterial diversity, disrupting ecological balance [
2]. Rising temperatures are also linked to the spread of antibiotic resistance [
4,
5] and increased abundance of pathogens like
Vibrio cholerae in coastal waters [
6]. These examples underscore the urgency of understanding bacterial thermal adaptation in the context of global climate change.
Phenotypic plasticity—the ability of organisms to adjust their traits in response to changing environmental conditions [
7,
8]—allows species to buffer against environmental fluctuations. This adaptability can extend an organism’s ecological range, potentially leading to genetic assimilation, where initially flexible traits become genetically stabilized [
9]. Nevertheless, plasticity has inherent limitations and costs, especially under sustained stress [
10]. Reaction norms, which describe how a genotype's phenotype changes across environmental gradients, are valuable tools for assessing thermal limits and understanding adaptive strategies. We can learn about a given organism's thermal niche through reaction norms. (Wolterek, 1909 [
11], cited by Stearns[
12]). Mesophilic bacteria, such as
Escherichia coli [
13] and some
Bacillus species [
14], typically thrive between 27°C and 40°C.
When exposed to higher temperatures, mesophilic bacteria activate a heat-shock response (HSR), which involves the rapid production of heat-shock proteins that stabilize cellular components and temporarily protect against heat stress [
15,
16]. However, these adaptations are generally short-lived and insufficient for prolonged survival under extreme temperatures.
Escherichia coli, as a well-established model organism, has been extensively studied for its physiological responses to thermal stress. Unlike thermal tolerance, which temporarily protects cells from heat damage by activating heat shock proteins [
17], thermal adaptation requires genetic changes that can withstand and sustain growth at higher temperatures beyond the initial threshold [
18].
Experimental evolution is a powerful tool for gaining insights into the capacity of bacteria to adapt to specific biotic and abiotic challenges and understanding the types of mutations that shape or enable such adaptations to changing environments [
18,
19]
. It allows researchers to address a key question: whether bacteria can evolve similar phenotypic outcomes through different genetic pathways—a concept known as convergent evolution, a central theme in evolutionary biology. Previous experiments have documented a rapid adaptive response of
E. coli to high temperatures [
20], with evidence that the genetic response can be diverse [
21]. Similarly, Tenaillon et al. [
22] took a whole-genome approach to estimate the extent of evolutionary convergence that occurs during adaptation in 115 populations of
E. coli at a high temperature of 42.2°C, analyzing the overall genetic basis of adaptation. Their findings highlighted convergence among genes, with the most mutated gene being
rpoB, along with proteins that regulate the
rpoS stress response (RSS).
Although numerous studies have explored the ability of
E. coli to evolve at different temperatures [
23], few studies have aimed to evolve bacteria to expand their thermal niche. In this context, Murata
et al. [
24] subjected an
E. coli strain to 43 °C and identified 51 genes crucial for thermotolerance, highlighting the diverse genetic pathways involved in developing heat tolerance. Murata introduced the term "critically high temperatures" (CHT) to describe temperatures beyond the thermal limit.
Bennett and Lenski [
13] sought to isolate evolved lines capable of surpassing the upper thermal limit and found that this limit did not significantly increase in any of the derived lines, even those that evolved at 41-42°C. Exposure to 44°C proved lethal, except for one thermotolerant mutant, to which they referred to as "Lazarus," that managed to grow at 44°C. Mongold et al. [
25] further investigated evolutionary adaptation to what they termed a "lethal thermal environment" by directly shifting evolution to 44°C. Their findings revealed that three thermotolerant mutants were derived from two progenitor lines that had previously adapted to 41-42°C, suggesting that these lines extended their upper thermal limit in a stepwise manner relative to their progenitors. Additionally, they noted that not all evolved lines exhibited trade-offs, such as a loss of fitness at lower temperatures.
Bacillus strains are particularly important to study in the context of prolonged survival at temperatures exceeding their niche range because they are ubiquitous and ecologically significant, playing key roles in decomposing organic matter and nutrient cycling. Although
Bacillus can form heat-tolerant spores that remain dormant for decades, their heterotrophic functions occur only in the vegetative state. Therefore, understanding whether
Bacillus can evolve more heat-tolerant vegetative cells is crucial for predicting how these bacteria will respond to global warming. Recent research has shown that wild
Bacillus strains exhibit considerable variation in thermal tolerance, influenced by genetic background [
14], emphasizing that temperature is a critical factor affecting bacterial growth and survival. Despite extensive research on acute bacterial responses, the long-term evolution of bacterial thermotolerance and the upper thermal limits they can achieve remain poorly understood and has not been done in
Bacillus lineages.
This study addresses this knowledge gap by investigating whether wild Bacillus strains from two distinct lineages can evolve heat tolerance in their vegetative cells through experimental evolution. We aim to determine whether gradual temperature increases enable these bacteria to develop the plasticity needed to survive projected global temperature rises of 2 to 4°C and to identify the genetic mechanisms underlying their adaptations. By focusing on different lineages and genetic backgrounds, our research explores the constraints and potential pathways for bacterial adaptation to extreme heat, providing insights into their evolutionary resilience and broader ecosystem impacts.
The main conclusions of this work are: 1. The observed lineage-specific outcomes and convergence within strains underscore the critical role of genetic background in shaping adaptive responses. B. cereus and B. subtilis have different plasticity and B. cereus lineages, contrary to those of B. subtilis, were not able to increase thermal tolerance relative to ancestral lines. Only one evolved B. subtilis line successfully extended its thermal niche. 2. Tolerance to higher temperatures was observed in B. subtilis lines evolved constantly at 37°C, indicating a metabolic adjustment driven by continuous growth conditions and the growth medium rather than selective pressure from increased temperatures. 3. Convergent mutations in genes regulating c-di-AMP synthesis uncovers, for the first time, its critical role in thermal tolerance. This discovery links molecular mechanisms to thermal adaptation across lineages. 4. Finally, our findings warn about bacteria's inability to evolve significant thermal tolerance, even under gradual selection. This raises concerns about bacterial communities' resilience to climate change and the potential cascading effects on biogeochemical cycles and ecosystem stability.
4. Discussion
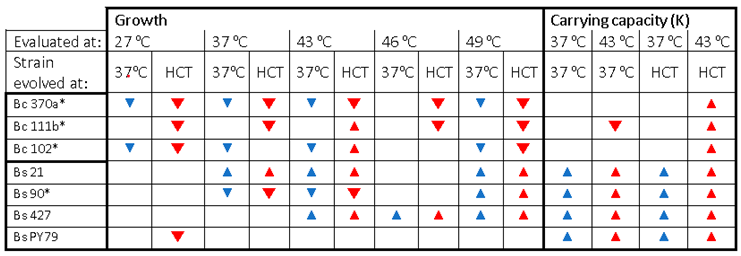
Our study highlights lineage-specific strategies by using experimental evolution with gradual temperature increases. It reveals significant differences in how Bacillus subtilis and Bacillus cereus lineages evolved thermal tolerance when subjected to gradual temperature increases beyond their thermal niche. B. subtilis strains displayed minimal changes in reaction norms, reflecting their greater inherent phenotypic plasticity, while B. cereus strains showed clear trade-offs, such as reduced growth rates at lower and even higher temperatures, indicating that high-temperature selection imposed a substantial evolutionary cost. These results suggest that larger initial plasticity might buffer against rapid adaptation under short-term experimental conditions.
In our study, the
B. cereus strains initially struggled under high-temperature stress but managed to stabilize and recover growth within weeks, although several times we had to stop the regime of increasing temperature until the final 300 generations of constant high-temperature selection. These strains accumulated more mutations than those of
B. subtilis lineage. This is similar to what Mongold et al. [
25] observed for
E. coli growth at the limit of their niche temperature, and suggests that evolutionary rescue mechanisms were at play, allowing the strains to persist despite the harsh conditions. The concept of evolutionary rescue is particularly relevant to our observations. Evolutionary rescue occurs when adaptive genetic changes enable populations to recover from near extinction under severe environmental stress [
38,
39].
While
B. subtilis benefited from its broad thermal tolerance,
B. cereus needed substantial genetic adjustments to cope with high temperatures. This observation raises critical questions about the role of plasticity in evolutionary dynamics. Does higher plasticity hinder adaptation by reducing selective pressure, or does it enable immediate survival, allowing gradual genetic changes? Our data support the notion that short-term evolution favors species with inherent plasticity, while long-term selection can drive adaptive genetic changes in species with less initial plasticity.
B. subtilis can be considered to possess more thermal plasticity compared to
B. cereus. This conclusion aligns with previous findings by Hurtado et al. [
14], which highlighted differences between the
B. cereus and
B. subtilis lineages in their upper tolerance limits, comparing strains from a thermal spring to those from a temperate lagoon. In this study, only the
B. subtilis evolve lines exhibited better growth than the parental strain at the HCT. At the same time, three of four
B. subtilis lineages surpassed their ancestral strain´s tolerance limit even at 49
oC. However, it is essential to note that the
B. subtilis lines evolved at 37
oC were also able to increase their thermal limit. The pressure of maintaining continuous growth in this particular medium impacted the metabolism that favored thermal tolerance. This is concordant with the change in carrying capacity that resulted from the experimental evolution, particularly for the
B. subtilis lines, for which higher carrying capacity changes occur in all lines, whether evolved at 37
oC or at high temperature. This contrasts with
B. cereus lines, for which only the lines evolved at higher temperatures exhibit increased carrying capacity under heat stress. These results suggest a shift in metabolism. Accordingly, the expression of proteins that provide stress tolerance or the capacity to adapt to new environmental conditions rapidly is expected to be at the expense of growth-related proteins, reducing the instantaneous growth rate [
40].
Finally, adaptation at high temperatures came with trade-offs, as evidenced by poorer growth at lower temperatures. This was observed for
B. cereus lines but not for
B. subtilis. Our results showed decreased fitness of some of the evolved
B. cereus lines (Bc111 and Bc370) at what were the ancestral strain´s optimal growth temperature, as a trade-off for adapting to high temperatures This supports evolutionary theory that suggests pleiotropic effects, where genes affecting multiple traits may constrain adaptation and result in maladaptation [
41].
Genetic analysis revealed that mutations in genes involved in osmolyte metabolism and heat-shock responses played critical roles in thermal tolerance. For instance, arginine and trehalose metabolism mutations were associated with improved thermal resilience. Arginine biosynthesis is known to be induced under heat stress [
32], and trehalose has been suggested to act as a thermoprotectant, stabilizing proteins and membranes [
42]. These mutations differ from those described in [
43] from an experimental evolution experiment with
B. subtilis, with a temperature regime of 50
oC. Sequencing revealed mutations in genes encoding proteins associated with pathways know to be involved in heat stress response: (
hrcA, ftsH) and other stress proteins (
relA and
sigW). We do not expect to find mutations in genes involved in the thermal response, such as those described by Hecker et al. (1996) [
33] and Helmann et al. (2001)[
32], which, if mutated, would reduce tolerance to temperature.
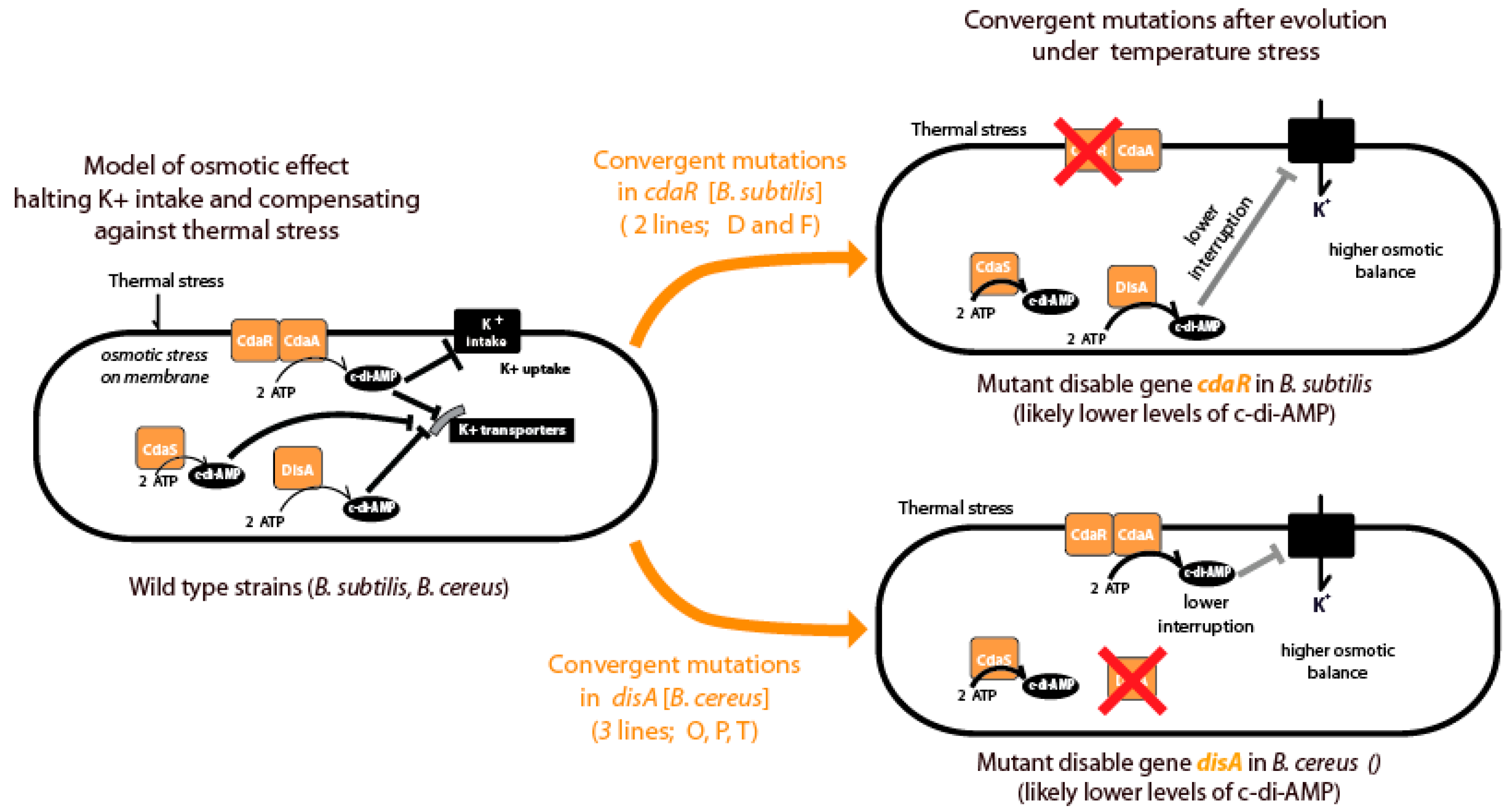
A novel and significant finding of our study was the convergence of mutations in DAC genes in several strains, in both lineages. Dac genes are involved in the synthesize c-di-AMP. Both
B. cereus and
B. subtilis possess three genes for the synthesis of c-di-AMP, DisA and CdaA function in the vegetative phase, while CdaS is sporulation specific [
44]. Most studies have been carried out in
B. subtilis, and it is known that the activity of CdaA is modulated by CdaR. c-di-AMP is essential for the growth of
B. subtilis and DisA and CdaA contribute to modulate different DNA damage responses during exponential growth [
45]. The parallel occurrence of mutations in DAC genes (
cdaR in
B. subtilis and
disA in
B. cereus, Figure 10) is intriguing. Several lines of evidence suggest that the diadenylate cyclase CdaA is part of the conserved essential
cda-glm module involved in cell wall metabolism. Notably, this molecule regulates potassium transport and osmotic balance [
46]; when bacteria are challenged with high osmolarity, they acquire increased resistance to high temperature and oxidative stresses. The high-osmolarity-dependent increase in thermotolerance has two manifestations: elevation of the upper limit of the growth temperature and enhanced survival at otherwise lethal high temperatures [
47]. Although c-di-AMP is involved in many essential pathways, to participate in virulence and osmotic stress, it had not been implicated in thermal stress. The parallel occurrence of mutations in DAC genes (
cdaR in
B. subtilis and
disA in
B. cereus, Figure 10) Our results suggest a lineage-specific evolution in the modulation of the c-di-AMP regulation. In either case, potassium uptake is likely involved in mitigating membrane destabilization caused by high temperatures. In
B. subtilis and many other bacteria, a sudden increase of the intracellular potassium concentration is the first response to osmotic stress [
48]. Thus, the control of potassium homeostasis by c-di-AMP might also be part of a larger picture of the regulation of osmoadaptation. It has been shown that a variety of compatible solutes (ie glycine betaine, proline) serve as heat protectants for
B. subtilis.
B subtilis has several redundant transporters, such that a mutation is one of the transporters has no impact on heart stress [
49] about molecular adaptation is whether it restores physiological and molecular processes from a stressed state back toward the unstressed, wild-type state or whether it instead tends to drive the evolution of novelty [
50]. Previous studies have suggested the former, because studies have shown that
E. coli adapts to high-temperature stress (42.2 C) by restoring both gene expression [
51,
52] and phenotypic characteristics [
53] toward that of the unstressed ancestor. We think that expanding the thermal niche requires both adapting gene expression to new threshold, as the previous level is inadequate for the permanent extralimited temperature stress and novelty to withstand a permanent stress. In this sense, we suggest that cells regulate the levels of c-di-AMP to fit the novel stressful conditions. These findings open up new avenues for understanding the role of c-di-AMP in bacterial heat stress response.
Our experimental design, starting with two species and multiple strains, allowed us to explore the potential for parallel evolution [
54] and identify multiple pathways leading to thermal tolerance. Parallel evolution, defined as the independent evolution of the same phenotype or genotype in response to identical selection pressures, provides insights into the predictability and diversity of evolutionary outcomes. In this study, we observed that while mutations affecting c-di-AMP synthesis emerged in all three
B. cereus lineages, only one
B. subtilis lineage (Bs_90) evolved a similar mutation. This finding suggests that while modulation of c-di-AMP synthesis is a common and potentially "go-to" solution for thermal tolerance, it is not the only pathway. The presence of c-di-AMP mutations in multiple lineages across species highlights its broad utility, yet, the absence of these mutations in some evolved lines underscores the diversity of evolutionary strategies. This observation supports the notion that standing genetic variation provides multiple routes to equivalent phenotypes, with natural selection shaping the most accessible solutions within specific genetic and physiological contexts.
Multiple pathways can lead to thermal tolerance, even under identical selective pressures, emphasizing the power of parallel evolution to reveal evolutionary constraints and opportunities. These findings align with the idea that evolution often proceeds along a single pathway within a given lineage but that alternative routes remain viable under different genetic or environmental conditions. By leveraging two species and multiple strains, our study underscores the importance of comparative approaches in experimental evolution for uncovering shared and lineage-specific adaptation mechanisms.
Our findings suggest that while different genetic pathways can lead to thermal tolerance, these pathways may converge on similar metabolic routes, such as osmotic protection. Mutations in genes regulating c-di-AMP synthesis highlight one such path, given its role in potassium transport and osmotic balance under stress. However, it is important to acknowledge a significant caveat in our analysis: genome sequencing identifies intragenic mutations that could be associated with the phenotype but cannot infer the impact of mutations in intergenic regions. Additionally, when multiple mutations occur within a genome, it is challenging to discern which specific mutations are responsible for the observed phenotypes. When working with wild strains, it is more challenging to reintroduce single mutations to validate the phenotype. Transcriptomic analyses would be necessary to uncover the overarching metabolic strategies and determine how these mutations integrate into the broader cellular response to thermal stress. Such analyses could reveal differential gene expression patterns and regulatory networks contributing to adaptation, providing a more comprehensive understanding of the metabolic adjustments underlying thermal tolerance.
The limited thermal tolerance observed in our study, even after experimental evolution, raises concerns about the capacity of microbial communities to adapt to climate change. Given the vital roles of Bacillus species in biogeochemical cycles, constraints on their thermal adaptability could have cascading effects on ecosystem functions, nutrient cycling, and soil health. Our findings suggest that while some bacterial populations may experience evolutionary rescue, the associated trade-offs could limit their ecological success and impact global processes.
It is notable that the three strains within each Bacillus lineage (B. subtilis and B. cereus) exhibited consistent, lineage-specific outcomes. Furthermore, the three independently evolved lines for each strain displayed similar trends in temperature tolerance, despite differences in the underlying mutations driving their responses. This result suggests a strong influence of both genetic background and lineage-specific traits in shaping the adaptive responses to temperature stress in Bacillus species. The consistency in outcomes within each lineage (B. subtilis and B. cereus) highlights that certain inherent genetic or physiological characteristics predispose these lineages to follow specific evolutionary trajectories.
The observed lineage-specific outcomes and convergence within strains underscore the critical role of genetic background in shaping adaptive responses. However, the diversity of underlying mutations leading to similar phenotypic outcomes highlights the unpredictability of evolutionary pathways. This variability suggests that microbial responses to environmental stressors, such as climate change, may be highly context-dependent and difficult to generalize. Each genetic background provides a distinct set of constraints and opportunities, leading to diverse adaptive trajectories even under similar selective pressures.
Consequently, this result makes it more challenging to predict how microbial communities, composed of diverse genetic lineages, will respond to global warming. While some lineages may adapt via predictable mechanisms, others may follow entirely different and unforeseen evolutionary paths. This underscores the need for further studies that explore the interplay between genetic background, evolutionary dynamics, and environmental change to better understand and anticipate microbial responses at both the species and community levels.
The experimental constraints of this study, which included duration of 1,000 generations and laboratory-based conditions, may not fully reflect the complexities of long-term evolutionary dynamics or the ecological interactions that occur in natural settings. Additionally, while we identified several mutations, especially those related to di-adenylate cyclase (DAC) genes, more studies would be necessary to understand their roles. This could involve transcriptomic analysis to better understand gene expression patterns in response to thermal stress. Furthermore, exploring the broader ecological context of these findings, such as how they translate to natural microbial populations and how bacteria respond to other environmental stressors like desiccation or nutrient scarcity, will be essential to evaluate bacterial adaptability and resilience comprehensively.
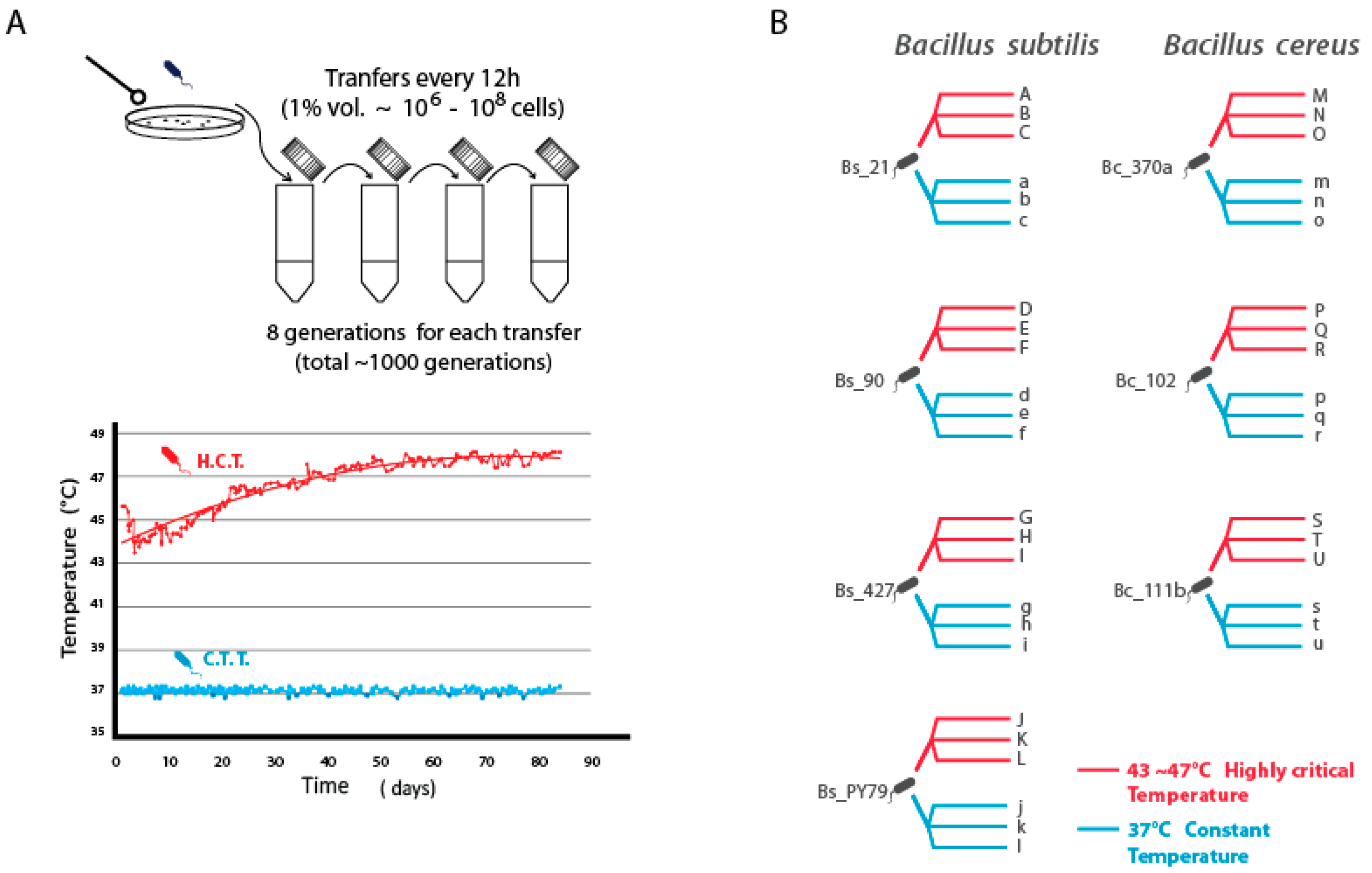
Figure 1.
Experimental Setup and Lineage Descent. (A) Inoculation and transfers were performed every 12 hours until reaching approximately 1000 generations (8 generations per transfer). The plot illustrates the measured temperature profiles of the incubator over 1000 generations both for the population evolved under highly critical temperature (HCT) and the constant (lower) temperature treatment (CTT) at 37 °C. The mathematical model for HCT is represented as 4.04 + 0.59(t^0.5), with an R^2 value of 0.8685. (B) The figure depicts the four parental strains from the B. subtilis lineage (left) and their respective descending lines labeled from A to L (HCT) and a to l (CTT_37 °C). Similarly, three parental strains from the B. cereus lineage (right) are depicted along with their corresponding descending lines named from A to L (HCT) and a to l (CTT_37 °C).
Figure 1.
Experimental Setup and Lineage Descent. (A) Inoculation and transfers were performed every 12 hours until reaching approximately 1000 generations (8 generations per transfer). The plot illustrates the measured temperature profiles of the incubator over 1000 generations both for the population evolved under highly critical temperature (HCT) and the constant (lower) temperature treatment (CTT) at 37 °C. The mathematical model for HCT is represented as 4.04 + 0.59(t^0.5), with an R^2 value of 0.8685. (B) The figure depicts the four parental strains from the B. subtilis lineage (left) and their respective descending lines labeled from A to L (HCT) and a to l (CTT_37 °C). Similarly, three parental strains from the B. cereus lineage (right) are depicted along with their corresponding descending lines named from A to L (HCT) and a to l (CTT_37 °C).
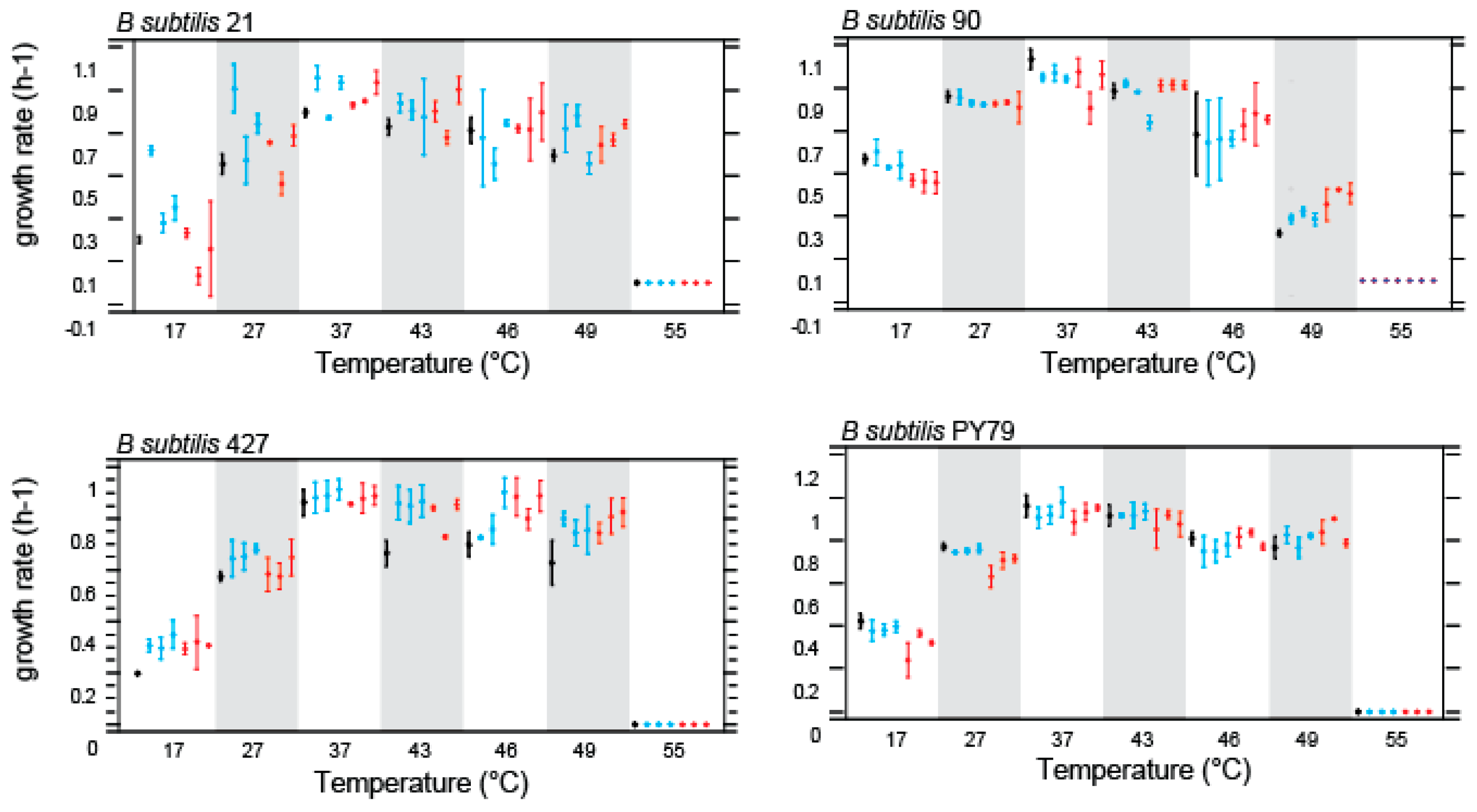
Figure 2.
Norms of reaction Bacillus subtilis parental strains (black) and their respective evolved lines at HCT treatment (red) and CTT treatment (37°C) (blue). Bars are confidence intervals qt 95% (ANOVA analysis, p=0.05).
Figure 2.
Norms of reaction Bacillus subtilis parental strains (black) and their respective evolved lines at HCT treatment (red) and CTT treatment (37°C) (blue). Bars are confidence intervals qt 95% (ANOVA analysis, p=0.05).
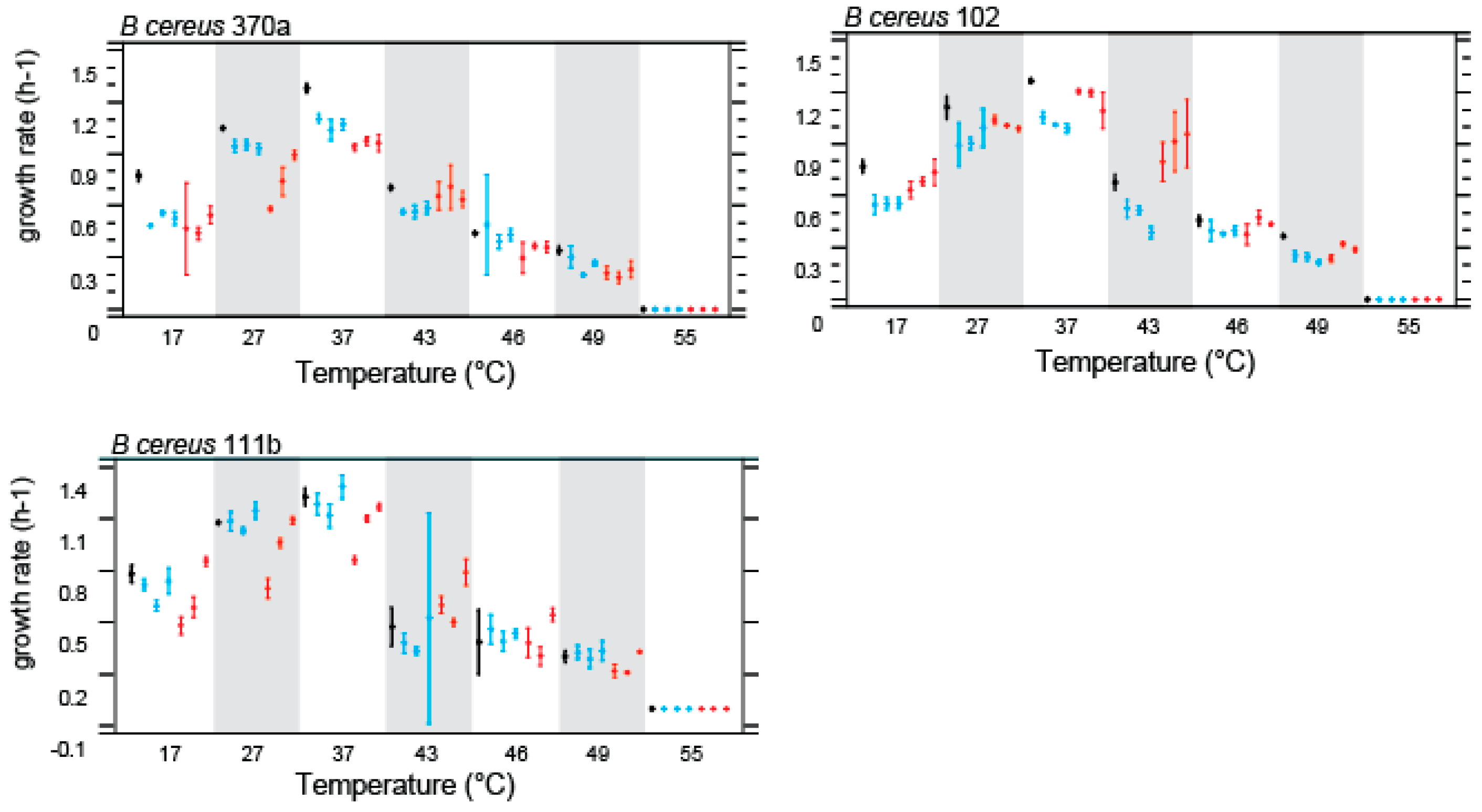
Figure 3.
Norms of reaction Bacillus cereus parental strains (black) and their respective evolved lines at HCT treatment (red) and CTT treatment (37°C) (blue). Bars are confidence intervals qt 95% (ANOVA analysis, p=0.05).
Figure 3.
Norms of reaction Bacillus cereus parental strains (black) and their respective evolved lines at HCT treatment (red) and CTT treatment (37°C) (blue). Bars are confidence intervals qt 95% (ANOVA analysis, p=0.05).
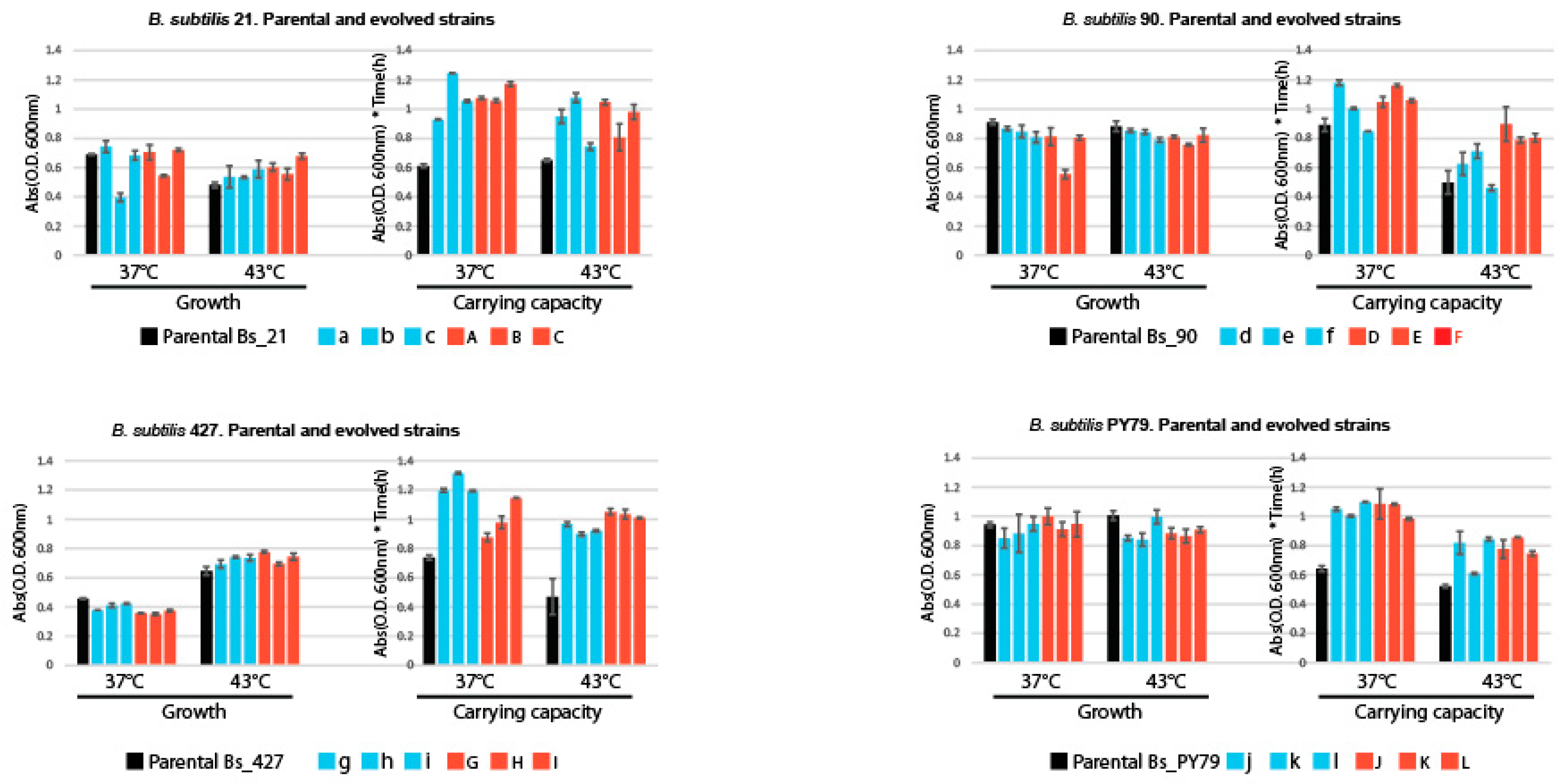
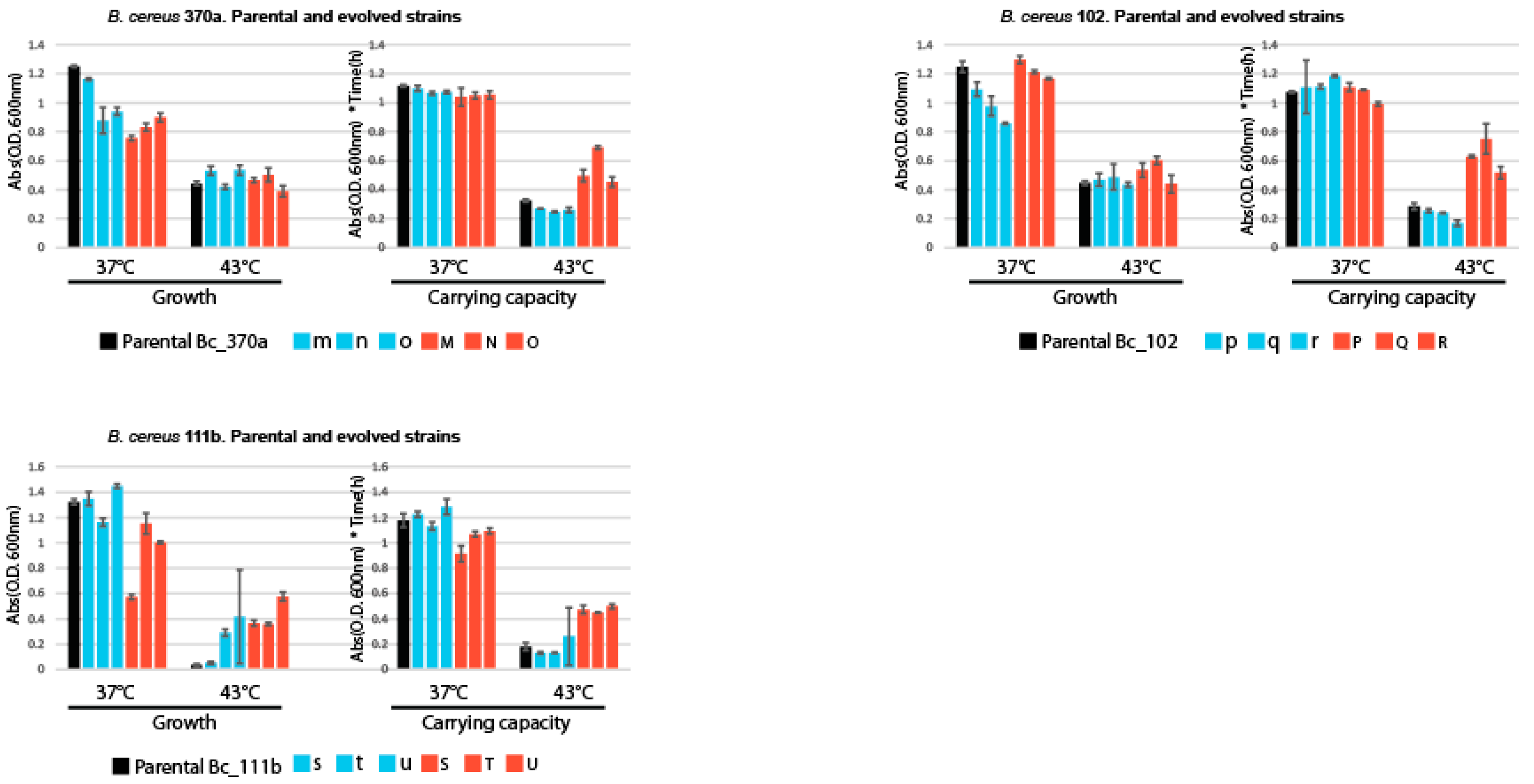
Figure 4.
Growth and Carrying Capacity in Parental and Experimentally Evolved Lines.
Figure 4.
Growth and Carrying Capacity in Parental and Experimentally Evolved Lines.
Figure 5.
Growth and Carrying Capacity in Parental and Experimentally Evolved Lines.
Figure 5.
Growth and Carrying Capacity in Parental and Experimentally Evolved Lines.
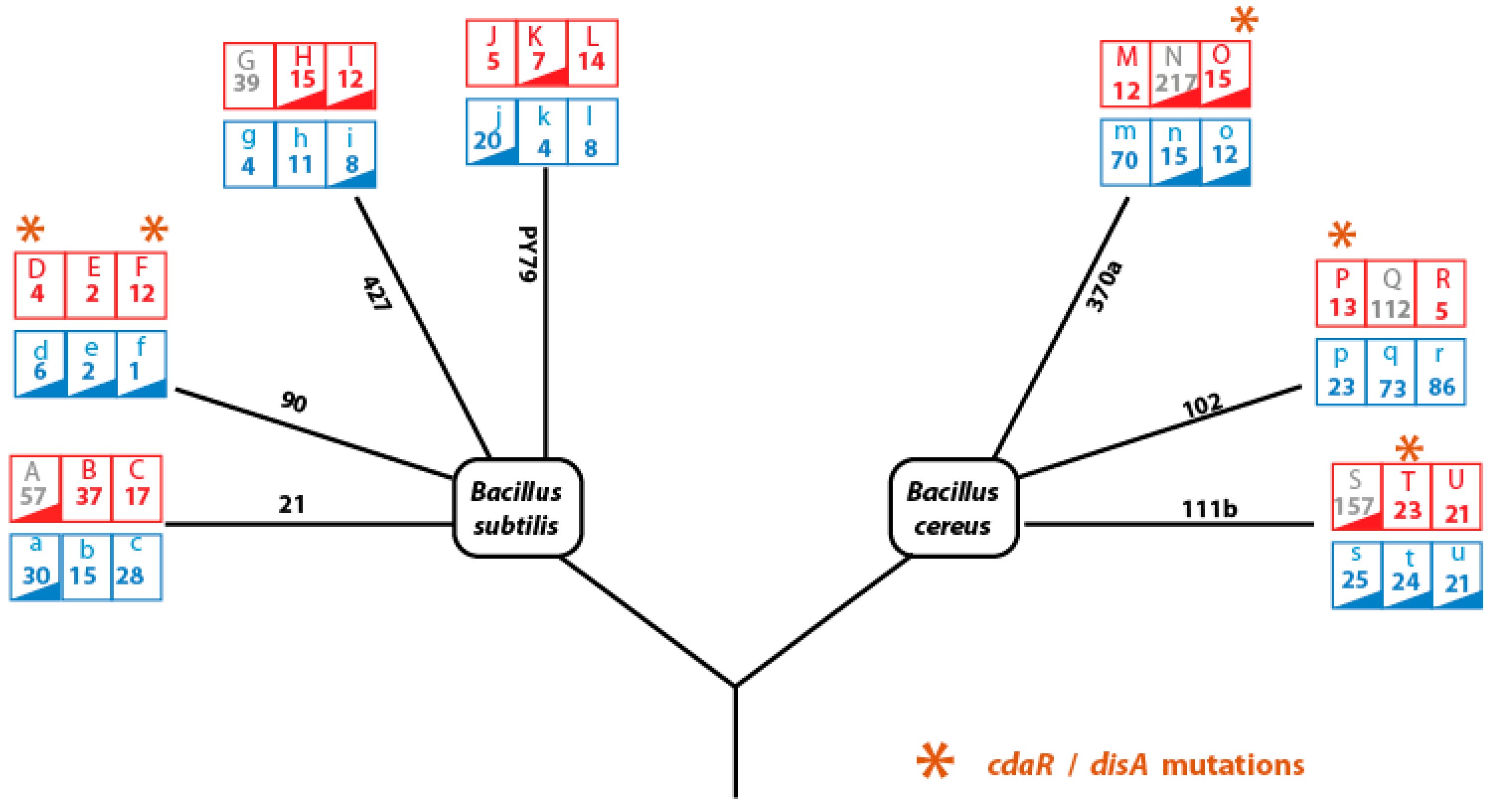
Figure 6.
Mutation Analysis of strain evolved from the Bacillus subtilis and Bacillus cereus lineages. Each lineage underwent highly critical temperature (HCT, red) treatment and constant temperature treatment (CTT, blue) with three replicates. The diagram illustrates the treatment conditions for each lineage and the total number of mutations observed after genome sequencing of a colony from the final evolution transfer. Loss of sporulation in evolved lines occurred in strains marked with red or blue triangles. Lines in grey represent hypermutant phenotypes, which were excluded from our analyses to reduce noise in data correlation. Orange asterisks denote convergent mutations at the gene level in the cyclic–d-AMP synthase gene (cdaR or disA), found in independent evolving lines under highly critical temperatures, supporting evidence of positive selection (statistical analysis in the results section).
Figure 6.
Mutation Analysis of strain evolved from the Bacillus subtilis and Bacillus cereus lineages. Each lineage underwent highly critical temperature (HCT, red) treatment and constant temperature treatment (CTT, blue) with three replicates. The diagram illustrates the treatment conditions for each lineage and the total number of mutations observed after genome sequencing of a colony from the final evolution transfer. Loss of sporulation in evolved lines occurred in strains marked with red or blue triangles. Lines in grey represent hypermutant phenotypes, which were excluded from our analyses to reduce noise in data correlation. Orange asterisks denote convergent mutations at the gene level in the cyclic–d-AMP synthase gene (cdaR or disA), found in independent evolving lines under highly critical temperatures, supporting evidence of positive selection (statistical analysis in the results section).
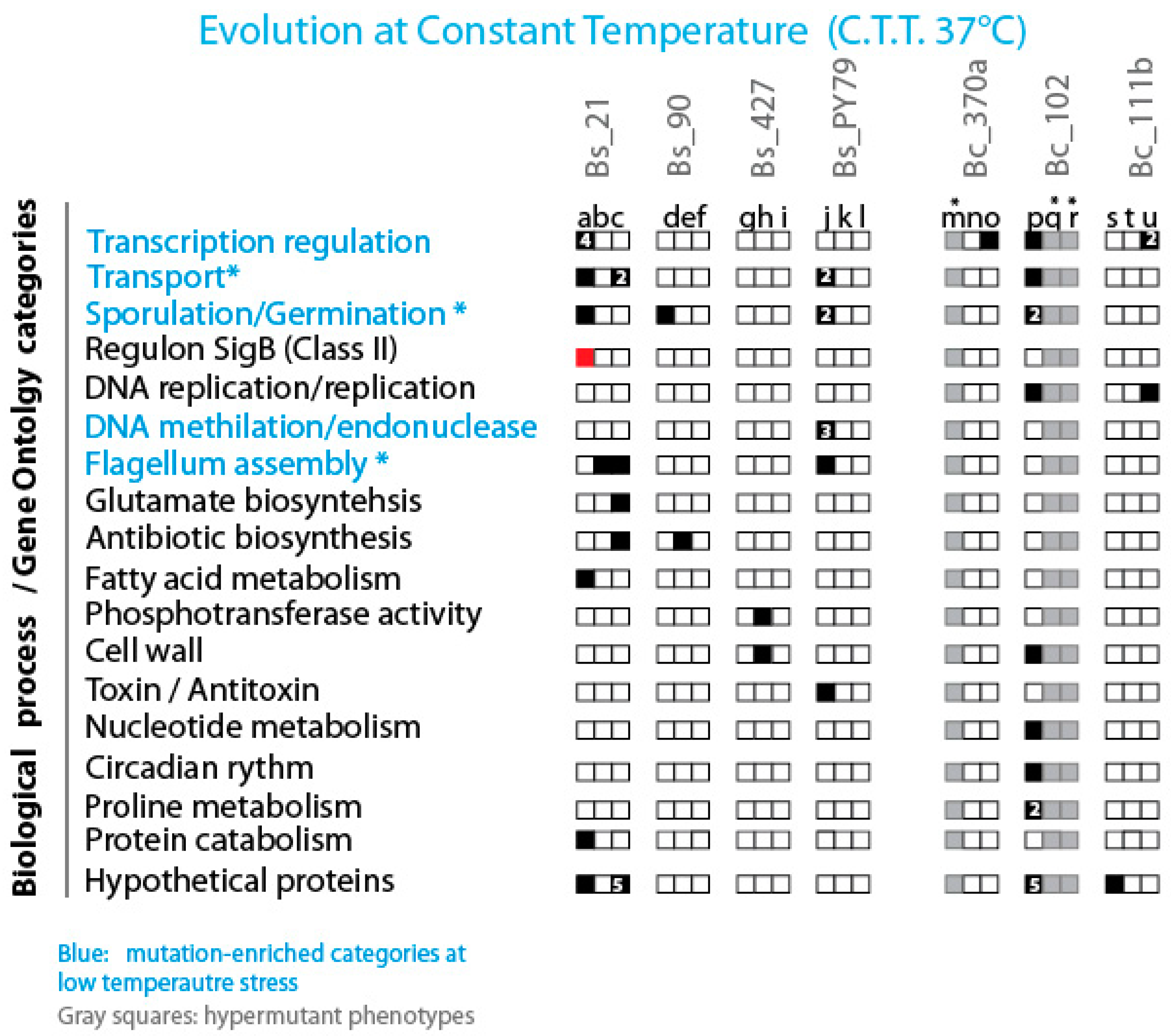
Figure 7.
Distribution of Mutations in Lines Evolved at Constant Low Temperature (37 °C) Across Gene Functional Categories. The figure displays the distribution of mutations observed in lines evolved at a constant low temperature (37 °C), categorized by gene functional categories. Black squares indicate genes with at least one mutation in the depicted category, with the number within the black squares representing the number of mutated genes within that category. White squares denote the absence of mutations in the respective gene functional categories. Grey squares are used for all hypermutant strains that were not analyzed. Additionally, red squares highlight genes that have been described as associated with the observed phenotype.
Figure 7.
Distribution of Mutations in Lines Evolved at Constant Low Temperature (37 °C) Across Gene Functional Categories. The figure displays the distribution of mutations observed in lines evolved at a constant low temperature (37 °C), categorized by gene functional categories. Black squares indicate genes with at least one mutation in the depicted category, with the number within the black squares representing the number of mutated genes within that category. White squares denote the absence of mutations in the respective gene functional categories. Grey squares are used for all hypermutant strains that were not analyzed. Additionally, red squares highlight genes that have been described as associated with the observed phenotype.
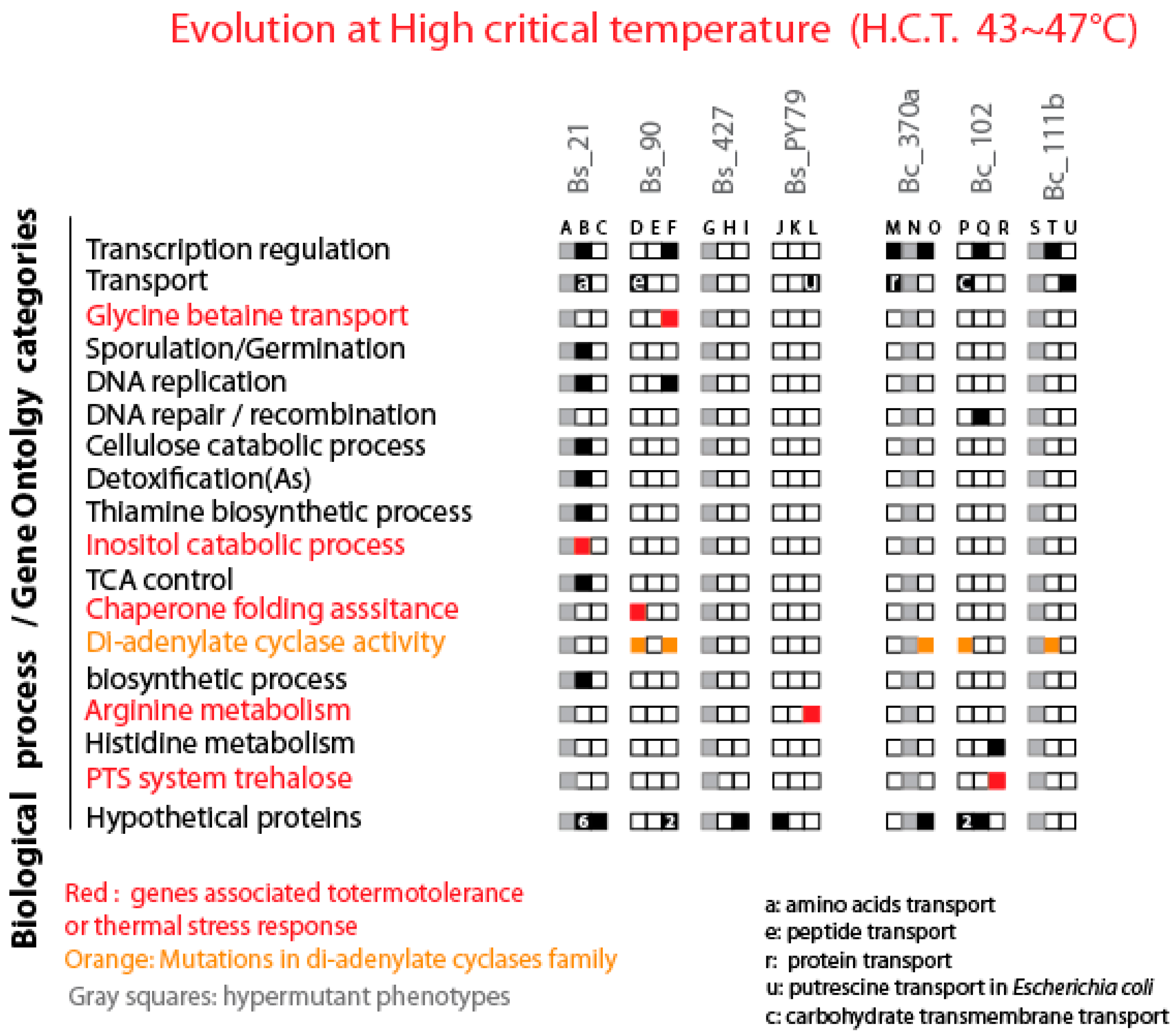
Figure 8.
Distribution of Mutations in Lines Evolved at Highly Critical Temperature Treatment (HCT) Across Gene Functional Categories. The figure illustrates the distribution of mutations observed in lines evolved under highly critical temperature treatment (HCT), categorized by gene functional categories. Black squares represent genes with at least one mutation in the depicted category, with the number within the black squares indicating the number of mutated genes within that category. White squares indicate the absence of mutations in the respective gene functional categories. Grey squares denote hypermutant strains that were not analyzed. Additionally, red squares highlight genes associated with the observed phenotype. An orange square specifically highlights diadenylate synthase encoding genes.
Figure 8.
Distribution of Mutations in Lines Evolved at Highly Critical Temperature Treatment (HCT) Across Gene Functional Categories. The figure illustrates the distribution of mutations observed in lines evolved under highly critical temperature treatment (HCT), categorized by gene functional categories. Black squares represent genes with at least one mutation in the depicted category, with the number within the black squares indicating the number of mutated genes within that category. White squares indicate the absence of mutations in the respective gene functional categories. Grey squares denote hypermutant strains that were not analyzed. Additionally, red squares highlight genes associated with the observed phenotype. An orange square specifically highlights diadenylate synthase encoding genes.
Figure 9.
Convergent Evolution in Lines Evolved at Critically High Temperature Revealed Cyclic-di-AMP as a Key Player. The figure illustrates convergent evolution observed in lines evolved at critically high temperature (CHT), highlighting Cyclic-di-AMP as a significant factor. Both Bacillus subtilis and Bacillus cereus possess three Cyclic-di-AMP synthase genes, with two expressed during the vegetative phase and the third restricted to sporulation. In B. subtilis, the mutated gene was cdaR, a regulator of the synthetase gene, while in B. cereus, mutations impacted the disA gene, the synthetase itself. Cyclic-di-AMP plays a crucial role in regulating K+ uptake, a key osmolite balance player, suggesting its involvement in heat stress adaptation.
Figure 9.
Convergent Evolution in Lines Evolved at Critically High Temperature Revealed Cyclic-di-AMP as a Key Player. The figure illustrates convergent evolution observed in lines evolved at critically high temperature (CHT), highlighting Cyclic-di-AMP as a significant factor. Both Bacillus subtilis and Bacillus cereus possess three Cyclic-di-AMP synthase genes, with two expressed during the vegetative phase and the third restricted to sporulation. In B. subtilis, the mutated gene was cdaR, a regulator of the synthetase gene, while in B. cereus, mutations impacted the disA gene, the synthetase itself. Cyclic-di-AMP plays a crucial role in regulating K+ uptake, a key osmolite balance player, suggesting its involvement in heat stress adaptation.
Table 1.
Summary of temperature plasticity gained on growth and carrying capacity by the evolved lines.
Table 1.
Summary of temperature plasticity gained on growth and carrying capacity by the evolved lines.