1. Introduction
Lung cancer (LC) is the second most common neoplastic disease worldwide, accounting for 18% of all cancer deaths [
1]. Non-small cell LC (NSCLC) is often diagnosed at advanced stages and presents significant treatment challenges due to its complexity and resistance to traditional therapies [
2,
3,
4]. While the emergence of targeted therapy and immunotherapy has transformed the treatment of NSCLC, resulting in significant enhancements in survival rates, patients with stage IV oncogene-negative, PDL1-negative NSCLC continue to experience a grim prognosis [
4]. The role of ablation treatment, including surgery and radiotherapy (RT), has been assessed in the current scenario. Several studies have shown a 5-year overall survival (OS) rate of 29% to 45% for patients with limited metastases who underwent surgery or ablation at all disease sites [
5,
6]. Consequently, to date, treatment strategies for these patients should be performed, and the use of potentially curative treatment, rather than merely palliative, should be increased.
The present case report delves into such a scenario, demonstrating a durable complete metabolic response in a patient with brain and bone metastases from NSCLC who was administered immune checkpoint inhibition (ICI) with chemotherapy, stereotactic radiotherapy (SRT) for brain metastases, and hypofractionated RT (hypo-RT) for lung tumor.
2. Case Report
A 71-year-old ex-smoker male patient with a history of hypertension and bladder cancer (pT1a in 2016) presented at our hospital in October 2021 with headache, language and memory difficulties, and tinnitus. He presented a nonproductive night cough without dyspnea.
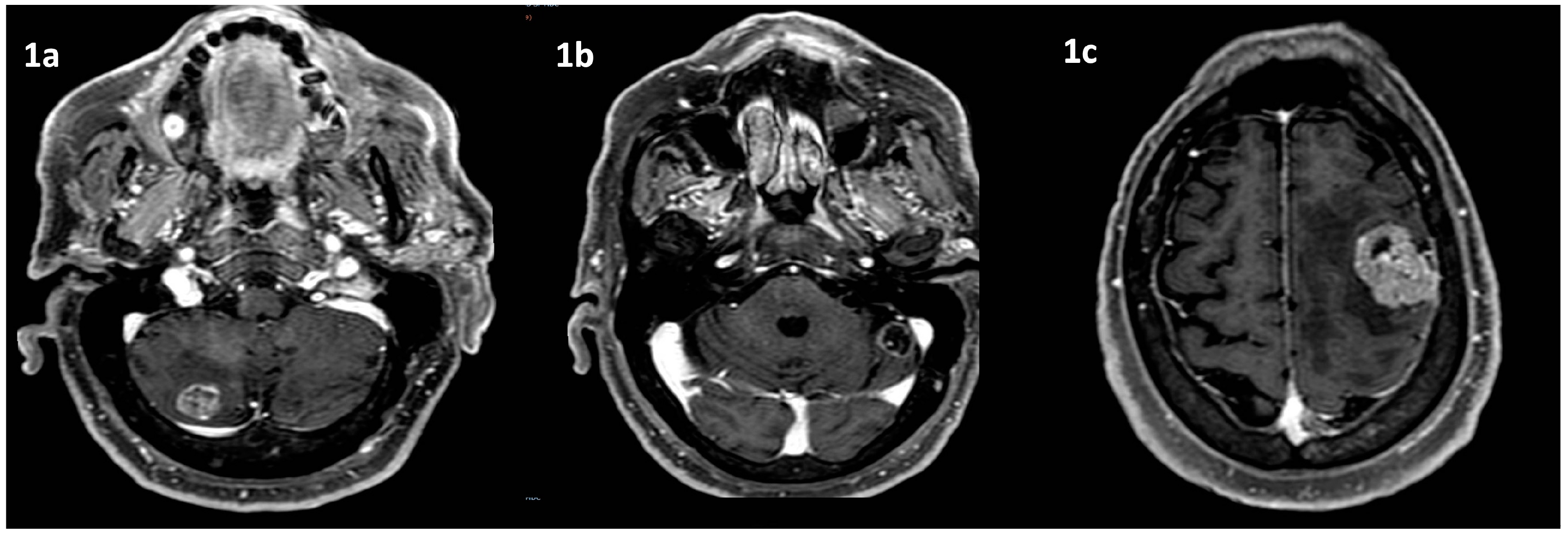
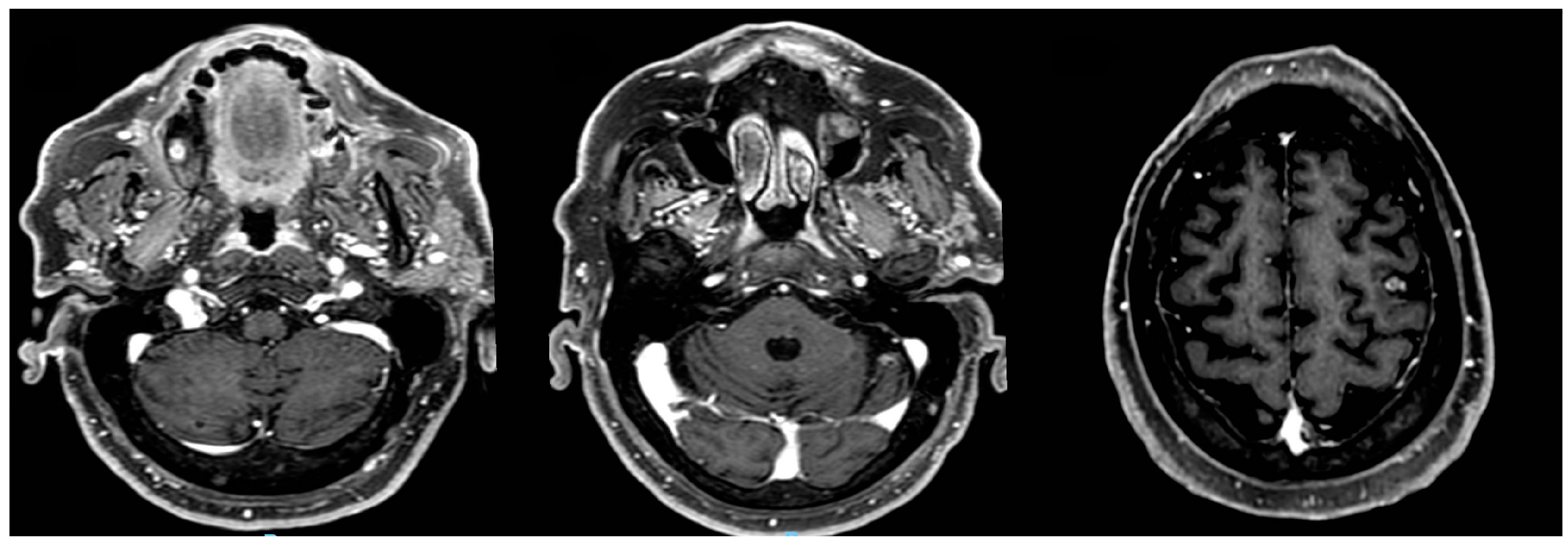
Magnetic resonance (MRI) of the brain was performed, reporting the presence of 3 brain lesions (one in the left frontal lobe of about 4 cm, the other 2 in the cerebellum right and left of about 2 cm) with oedema (
Figure 1a-b-c). Moreover, an occipital bone lesion was reported.
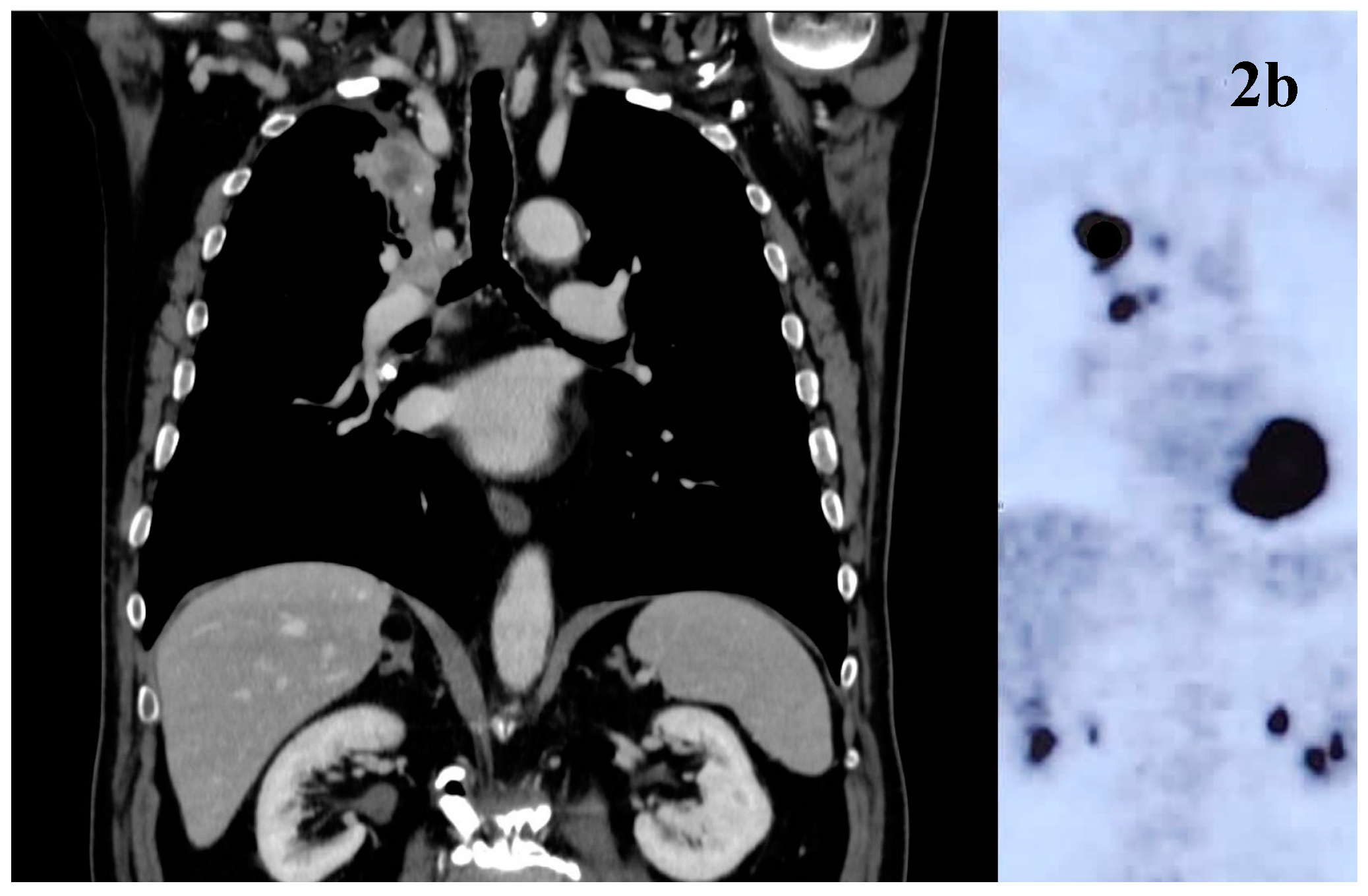
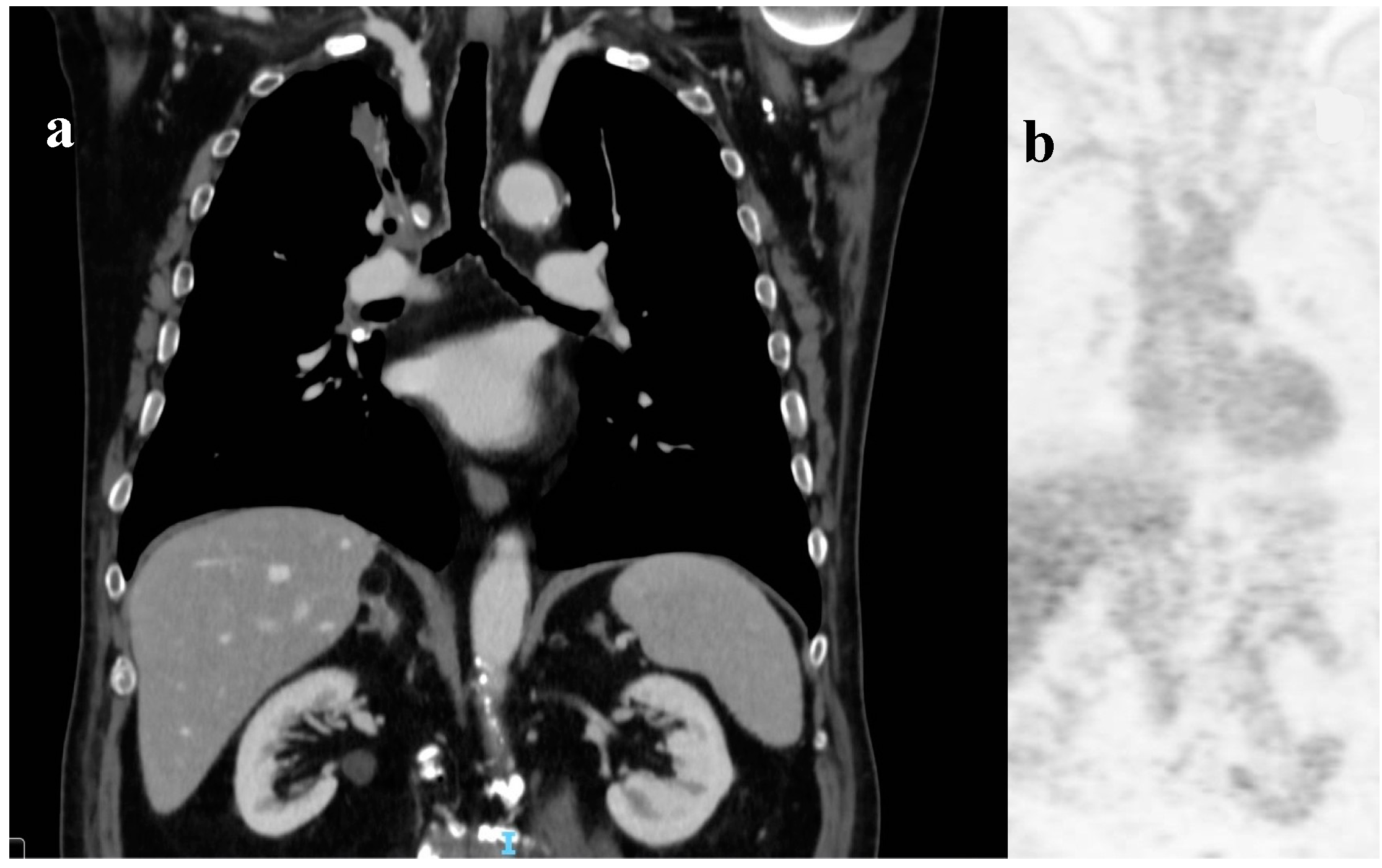
After one week, a total body computed tomography scan (TB CT scan) was performed, revealing a solid nodular, expansive mass (39 mm) in the right upper lobe of the lung and multiple lymphadenopathies in the mediastinum (paratracheal and subcarinal) (
Figure 2a).
Fluorodeoxyglucose positron emission tomography (FDG PET) (
Figure 2b) confirmed the presence of multiple hyper-capturing lesions consistent with the CT scan findings: right superior lobe (SUV 16.8), mediastinal nodes, plus lesion on the occipital bone. A cT2N2M1c stage IV lung cancer was diagnosed with CEA and Ca19.9 of 15.4 ng/mL (<6) and 65.6 UI/mL (<37), respectively.
A CT-guided biopsy was performed on the lung mass, and a poorly differentiated neoplasm suggestive of NSCLC adenocarcinoma was diagnosed. PDL1, ROS, EGFR, and ALK were negative.
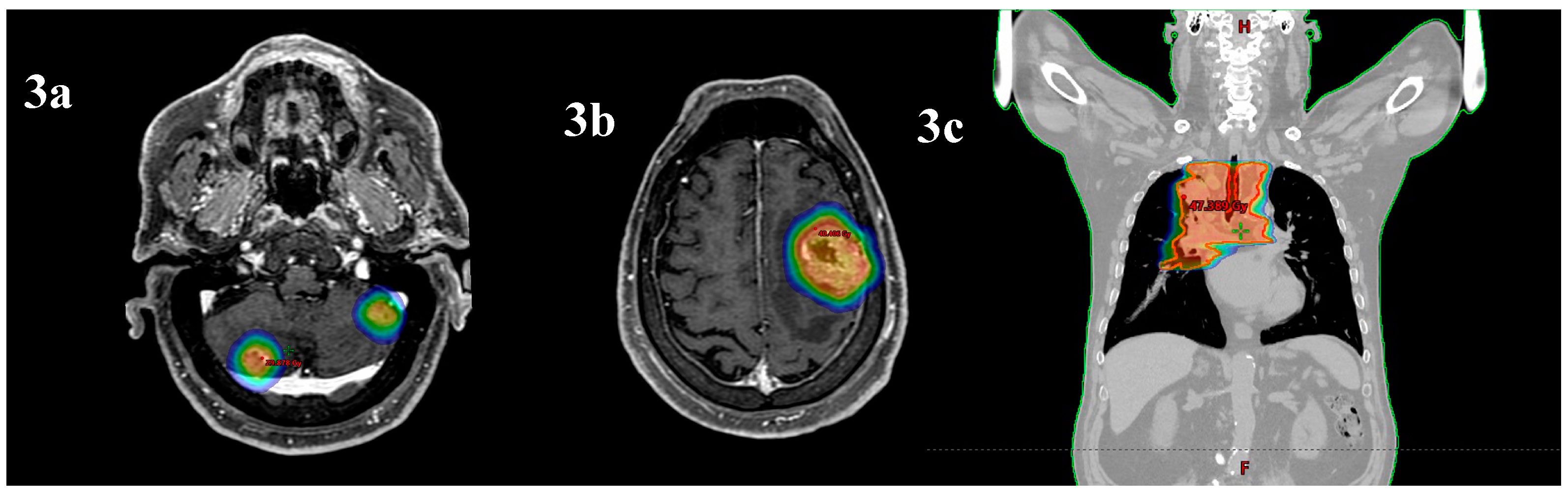
He started SRT: the frontal right lesion received 36 Gy/6 fractions and the cerebellum lesions received 24Gy/3 fractions (
Figure 3a-b) [
6,
7,
8].
In November 2021, he started a chemotherapy (CHT) regimen with cisplatin (75 mg/m2 every 21 days) and pemetrexed (500 mg/m2 every 21 days), and ICI with Azetolizumab (1200 mg every 21 days).
After 4 CHT and ICI cycles, blood tests showed a favorable response in cancer markers: CEA decreased from 15 ng/mL to 7 ng/mL, and Ca19.9 decreased from 65.5 UI/mL to 41.
Subsequent CT scans in January 2022 showed a positive response to brain metastases and a reduction in the size of the lung tumor from 39 mm to 29 mm, as well as improvements in thoracic lymphadenopathies.
From February 2022, he continued with only Atezolizumab (10 cycles).
In June 2022, imaging showed a growth in lung mass and mediastinal nodal size. Brain MRI indicated a positive response to SRT, and no other brain lesions were found.
In July 2022, patients received hypo-RT (45 Gy/16 fractions to lung mass and PET + nodes) (
Figure 3c).
From 29.8.2022 to 2.2.2023, Atezolizumab was performed with a total of 17 cycles.
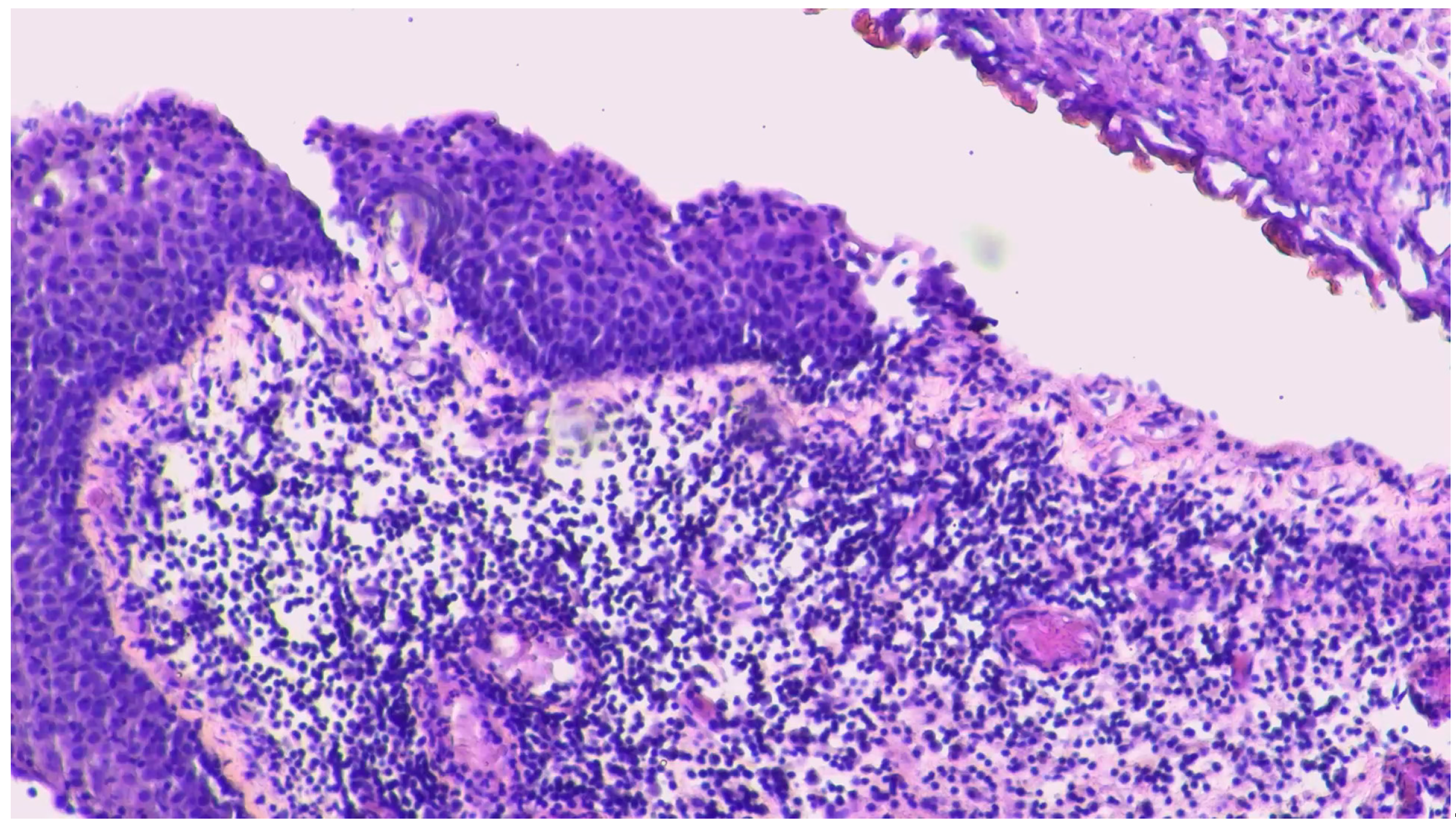
ICI was discontinued, as agreed with the patient, considering his adverse reactions. The patient developed hypothyroidism (TSH 64 UI/ml, range 0.27-4.2) with heart failure treated with levothyroxine, Grade 4 dermatitis from psoriasis, and pruritus unresponsive to antihistamines, which was managed with glucocorticoids. The patient resolves the side effects 3-4 months after the Atezolizumab interruption. Moreover, patients reported chronic cystitis with hematuria and, in July 2024, underwent a cystoscopy and a biopsy was performed. The result was bladder fragments with massive ulcer-necrotic phenomena and chronic granulomatous, giant cell and histiocytic inflammatory process (CKAE1/AE3 negative,
Figure 4).
Over the following months, regular monitoring with CEA (6.5 ng/ml), Ca19.9 (27UI/mL), CT-PET scans, and brain MRI indicated a complete response 35 months after diagnosis (
Figure 5 and
Figure 6).
3. Discussion
The management of patients with oncogene-negative and PDL1-negative NSCLC poses a significant clinical challenge [
4]. Furthermore, there is increasing evidence of an oligometastatic state in NSCLC in terms of association with local treatment and immunotherapy [
9]. In addition, the role of PD-L1 expression is intricate and multifaceted [
10,
11,
12]. It is important to note that cases where metastatic patients experience a complete response to treatment and remain disease-free for over two years are still uncommon and anecdotally in NSCLC [
11,
12,
13]. Our patient's prognosis was poor due to the presence of synchronous brain metastasis (BM), with a median OS of less than six months in untreated individuals [
14,
15].
The impressive and long-lasting metabolic complete response seen in this stage IV lung adenocarcinoma patient with BM, despite the absence of mutations (PDL1, ROS, ALK-negative), deserves careful consideration. This case provides valuable insights into the changing landscape of LC treatment.
3.1. The Role of RT in Oligometastatic NSCLC
The role of ablation treatment, including surgery and RT, has been assessed in the current scenario in a systematic review [
3]. The latter review evaluated 49 relevant studies involving 2176 patients. These studies focused on oligometastatic (1 to 5 lesions) patients underwent surgical metastasectomy and stereotactic ablative radiotherapy (SBRT). Sixty percent of all the studies included patients with exclusive brain metastases. In 82% of patients the primary tumor was controlled, with a median OS of 14.8 months for all patients (range 5.9-52) and of 19 months among patients with a controlled primary tumor [
3].
The integration of SBRT for persistent lesions highlights the potential of multimodality treatment. It offers high-precision, localized treatment that could benefit specific lesions and increase overall survival. The SABR-COMET phase II trial included patients with various types of primary tumors, with stage IV NSCLC being one of the most common, accounting for 18% of the total. Ninety-nine patients with con-trolled primary tumors and five or fewer metastatic lesions were eligible. They were divided into two groups for comparison: 66 in the SABR-plus-SOC arm and 33 in the palliative-SOC arm. Long-term follow-up showed a remarkable 5-year OS rate of 42% (95% CI, 28-56) in the SABR-plus-SOC arm com-pared to 18% (95% CI, 6-34; P .006) in the palliative-SOC arm [
14].
Based on the latter analysis, the present case was treated by SBRT for all the 3 BM.
Radiation can have both suppressive and stimulating effects on the immune system. The stimulating effects, such as releasing tumor antigens when radiation kills tumor cells, may help trigger an immune response against the tumor throughout the body. However, these responses are rare. To improve this effect, some suggest combining radiation with immune checkpoint inhibitor therapy for stage IV NSCLC. These are areas of ongoing preclinical and clinical investigation [
9,
15].
Atezolizumab is an immune checkpoint inhibitor, a type of targeted therapy drug. It is a monoclonal antibody that works by binding to the protein PD-L1 on the surface of some cancer cells. This prevents cancer cells from suppressing the immune system, allowing the immune system to attack and kill the cells. Data regarding the BBB penetration of ICIs are limited [
16,
17]. The most compelling evidence supporting the effectiveness of immune checkpoint inhibitor (ICI) therapy for active BM comes from a phase II trial of pembrolizumab conducted by Goldberg et al. This study involved 42 patients with advanced non-oncogene-driven NSCLC and untreated, asymptomatic BM ranging in size from 5 to 20 mm. Patients with PD-L1 ≥1% had a 29.7% intracranial objective response rate (ORR), with seven patients showing partial response on subsequent imaging and four achieving complete response. Their median OS was 9.9 months (cohort 1). In contrast, responses in brain metasta-ses of patients with PD-L1 < 1% were either not present or could not be evaluated (cohort 2) [
16].
3.2. The Role of Hypo-RT or Ablative RT in Antitumor Immunity
Emerging evidence suggests that hypo-RT may have a solid anti-tumor effect by causing tumor cell death through antitumor immunity and vascular damage [
15]. The RT not only directly impacts on the irradiated area but also stimulates the immune response indirectly because of the low irradiation doses outside the target area. Several studies have shown that hypo-RT can boost the body's anti-tumor immune responses; this happens because hypo-RT cause tumor cell death, normalize the abnormal blood vessel growth, and release tumor-associated antigens and pro-inflammatory cytokines.
However, some investigation in animal models has advised that the tumor microenvironment (TME) changes created by RT may develop an immunosuppressive TME, which could promote tumor invasion and spread in some cases.
After hypo-RT, tumor cells expose more specific molecules on their surfaces recognized by the immune system, with increased sensitivity to T cell-mediated cell death; also, hypo-RT increases pro-inflammatory molecules and danger signals in the TME. Consequently, the CD8+ T and dendritic cells are triggered and recruited into the tumor; in this context these immune cells play a key role in anti-cancer immune responses [
18].
Moreover, for the present case, local hypo-RT was chosen.
3.3. The Volume Reduction of RT Increases the Immunity Response
The lymph node controls interactions and movement of various immune cells, including T, B, and anti-gen-presenting cells. RT may enhance the ability of cytotoxic T cells to kill tumors in the TME by possibly increasing the expression of primary histocompatibility complex class I in tumor cells. However, tumor-draining lymph nodes (TDLN) are crucial for RT-induced immune stimulation and abscopal effects. Phenotypically, dysfunction of the TDLN caused by surgery, irradiation, pharmacological inhibitors, or genetic ablation significantly reduces the anti-tumor effects of RT and immunotherapy [
19]. Marciscano et al. [
20] showed that irradiating elective TDLN reduced immune-RT infiltration by regulating chemokine signaling. This led to an unfavorable TME in the primary tumor.
Thus, the RT volume reduction (involved field RT) was utilized in the present case.
3.4. The Timing ICI/RT
The first demonstration of a clinical benefit for combination treatment of ICI and RT was evidenced in retrospective analysis of highly immunogenic cancer lesions (in particular metastatic melanoma, NSCLC, and renal cells carcinoma) in which were compared the efficacy of RT alone or RT in combination with ICI [
21]. The evaluation of the optimal timing strategy was analyzed in a comprehensive meta-analysis of numerous clinical evidence that retrospectively suggested the superiority of concurrent ICI and RT treatment over sequential treatment.
In this case, SBRT was the first weapon used to treat BM, and then hypo-RT was performed between the 10 and 11 atezolizumab cycles.
3.5. The Role of ICI Acute Toxicity
ICIs induce a high risk of developing immune-related adverse events (irAEs) [
22,
23]. Different reports indicate that the incidence of irAEs can range from 60% to 85%, depending on the use of mono- or combination immunotherapy, affecting mostly the skin, endocrine glands, gastrointestinal tract, lungs, and liver. Hypophysitis and thyroid dysfunctions, as in the present case, are the most common endocrine side effects of immune checkpoint inhibitors, while Type 1 diabetes mellitus and adrenal insufficiency are less common side effects. Most patients usually recover from pituitary-thyroid and pituitary-gonadal axes disfunctions, while improvement of the pituitary-adrenal axis has been rarely observed.
On the other hand, limited literature exists on the incidence, time of onset, and risk factors for multiorgan systems irAEs, which occurred in about 5% of ICI-treated patients. It is still debated whether there is a direct correlation between ICI effectiveness and the degree of treatment-induced toxicity. Reports showed that irAEs are strongly correlated with better survival and higher response rates in patients with melanoma, advanced gastric cancer, and NSCLC receiving anti-PD-1 therapy.
A recent analysis was conducted using data from four clinical trials of atezolizumab in patients with NSCLC. The study found that 5.4% of patients experienced multiorgan irAEs, with "Skin " or "laboratory" being the most common. However, it was also observed that patients with multiorgan irAEs had improved OS compared to those without irAEs (hazard ratio, 0.47; 95% CI, 0.28–0.78; P < 0.0001). There was no significant difference in progression-free survival between the two groups (hazard ratio, 0.92; 95% CI, 0.62–1.35; P = 0.74).
Our case reported multiorgan irAEs (psoriasis, hypothyroidism, and cystitis with ulcer-necrotic bladder), showing an extended response 19 months after the interruption of ICI.
4. Conclusions
In the literature, few cases with metastases of adenocarcinoma of the lung are reported with a median OS of more than 24 months [
12]. Only one case, with a diagnosis of NSCLC and multiple brain metastases, was published in 2020 [
24]. He was diagnosed with pulmonary adenocarcinoma with EGFR exon 19 deletion mutation. Analysis based on genome sequencing revealed MGMT gene mutation in exon1, exon4, and exon5, and was treated with Temozolomide (150 mg/m2/d for five days every 28-day cycle) and RT, achieving an overall OS of 55 months. No data with prolonged survival was published for metastatic oncogene-negative, PDL1-negative NSCLC.
In conclusion, this case report illustrated the potential role and efficacy of combining ICI with chemotherapy and the addition of RT in treating oncogene-negative, PDL1-negative NSCLC.
Patients with metastatic NSCLC have a short life expectancy despite the use of advanced medical and pharmaceutical technologies. In this situation, it is increasingly important to collect and to study cases of patients with prolonged survival to examine their immunological characteristics. This will help search for improved treatment options for this group of patients. Further research is needed to improve the outcomes of this population. It's essential to understand how local therapy, such as RT, affects tumors and metastases and the immune effects of hypofractionated or SBRT on the TME. Figuring out the best timing for combining immunotherapy and RT may improve treatment outcomes and help us discover new combination therapy strategies.
Author Contributions
Conceptualization, all authors; methodology, MC, AF, FCDG, VD; software, AS, AF.; validation, IB, MPC, RC,RC,ANG, NS, GS, IB, and AF.; formal analysis, AF; investigation, AF, GS.; resources, MC.; da-ta curation, MC, AF; writing—original draft preparation, AF.; writing—review and editing, AS,RC, FCDG, VD; visualization, MC.; supervision, AF, IB, GS; All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
No available.
Informed Consent Statement
Written informed consent has been obtained from the patient to publish this paper.
Data Availability Statement
data could be required to corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Siegel, R.; Miller, KD.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Hendriks LE, Kerr KM, Menis J, et al. Non-oncogene-addicted metastatic non-small-cell lung cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34(4):358–76.
- Ashworth A, Rodrigues G, Boldt G, et al. Is there an oligometastatic state in non-small cell lung cancer? A systematic review of the literature. Lung Cancer. 2013 ;82:197-203. [CrossRef]
- Ashworth AB, Senan S, Palma DA, et al. An individual patient data metanalysis of outcomes and prognostic factors after treatment of oligometastatic non-small-cell lung cancer. Clin Lung Cancer. 2014;15: 346-55.
- Isbell JM, Li BT, Gomez DR. The emerging role of local therapy in oligometastatic non–small cell lung cancer. J Thorac Cardiovasc Surg. 2022 Mar;163(3):819-825. [CrossRef]
- Mazzola R, Corradini S, Gregucci F et al. Role of Radiosurgery/Stereotactic Radiotherapy in Oligometastatic Disease: Brain Oligometastases. Front. Oncol. 2019, 9, 206. [CrossRef]
- Alongi F, Fiorentino A, Gregucci F, et al First experience and clinical results using a new non-coplanar mono-isocenter technique (HyperArc™) for Linac-based VMAT radiosurgery in brain metastases. J. Cancer Res. Clin. Oncol 2019 145, 193–200. [CrossRef]
- Gregucci F, Bonaparte I, Surgo A, et al. Brain Linac-based Radiation Therapy: “Test Drive” of New Immobilization Solution and Surface Guided Radiation Therapy. J. Pers. Med. 2021 11, 1351. [CrossRef]
- Isbell JM, Li BT, Gomez DR. The emerging role of local therapy in oligometastatic non–small cell lung cancer J Thorac Cardiovasc Surg. 2022 Mar;163(3):819-825.
- Paz-Ares L, Ciuleanu TE, Cobo M, et al. First-line nivolumab plus ipilimumab combined with two cycles of chemotherapy in patients with non-small-cell lung cancer (CheckMate 9LA): an international, randomized, open-label, phase 3 trial [published correction appears in Lancet Oncol. 2021 Mar;22(3): e92]. Lancet Oncol. 2021;22(2):198–211. [CrossRef]
- Gamez Casado S, Benitez Fuentes JD, Álvarez Rodríguez B, et al. Achieving complete metabolic response in stage IV lung adenocarcinoma with chemotherapy, nivolumab, ipilimumab, and salvage SBRT: A case report. Respirol Case Rep. 2024 Apr 24;12(4): e01362.
- De Intinis C, Izzo P, Codacci-Pisanelli M, et al. A Rare Case of Complete-Immunotherapy- Responsive Metastatic Non-Small Cell Lung Cancer with Long Lasting Progression-Free Survival: A Case Report. Curr. Oncol. 2024, 31, 723–732. [CrossRef]
- Ali A, Goffin JR, Arnold A, et al. Survival of patients with non-small-cell lung cancer after a diagnosis of brain metastases. Curr Oncol. 2013 20(4): e300-6. [CrossRef]
- Palma DA, Olson R, Harrow S, et al. Stereotactic ablative radiotherapy for the comprehensive treatment of oligometastatic cancers: long-term results of the SABR-COMET phase II randomized trial. J Clin Oncol. 2020 ;38:2830-8. [CrossRef]
- Gregucci F, Beal K, Knisely JPS, et al. Biological Insights and Radiation–Immuno–Oncology Developments in Primary and Secondary Brain Tumors. Cancers, 2024: 16(11), 2047. [CrossRef]
- Goldberg SB, Schalper KA, Gettinger SN, Mahajan A, Herbst RS, Chiang AC, et al. Pembrolizumab for management of patients with NSCLC and brain metastases: long-term results and biomarker analysis from a non-randomised, open-label, phase 2 trial. Lancet Oncol. (2020) 21:655–63. [CrossRef]
- Wu W, Guo J, He L, Deng Q and Huang X (2024) Case report: Long-term intracranial effect of zimberelimab monotherapy following surgical resection of high PD-L1- expressing brain metastases from NSCLC. Front. Oncol. 14:1390343. [CrossRef]
- Wang, Y. Advances in Hypofractionated Irradiation-Induced Immunosuppression of Tumor Microenvironment. Front Immunol. 2021 Jan 25;11:612072. [CrossRef]
- Liu Z, Yu Z, Chen D, et al. Pivotal roles of tumor-draining lymph nodes in the abscopal effects from combined immunotherapy and radiotherapy Cancer Commun (Lond). 2022 Oct;42(10):971-986. [CrossRef]
- Marciscano AE, Ghasemzadeh A, Nirschl TR, et al. Elective Nodal Irradiation Attenuates the Combinatorial Efficacy of Stereotactic Radia- tion Therapy and Immunotherapy. Clin Cancer Res. 2018;24(20): 5058-71.
- Lehrer EJ, Peterson J, Brown PD, et al. Treatment of brain metastases with stereotactic radiosurgery and immune checkpoint inhibitors: An international meta-analysis of individual patient data. Radiother Oncol (2019) 130:104–12. [CrossRef]
- Squicciarini T, Villani R, Apollonio B, et al. Case report: Is severe toxicity the price to pay for high sensitivity to checkpoint inhibitors immunotherapy in desmoplastic melanoma? Front Immunol. 2024 ;15:1369531. 10 May. [CrossRef] [PubMed]
- Kichenadasse G, Miners JO, Mangoni AA, Rowland A, Hopkins AM, Sorich MJ. Multiorgan Immune-Related Adverse Events During Treatment With Atezolizumab. J Natl Compr Canc Netw. 2020 Sep;18(9):1191-1199. [CrossRef] [PubMed]
- Yang, Y.; Pu, Y.; Dai, N.; Wang, D.; Xu, M.M. Complete response of radioresistant brain metastases from non-small cell lung cancer with temozolomide. Medicine 2020, 99, e23592. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).