Submitted:
07 November 2024
Posted:
11 November 2024
Read the latest preprint version here
Abstract
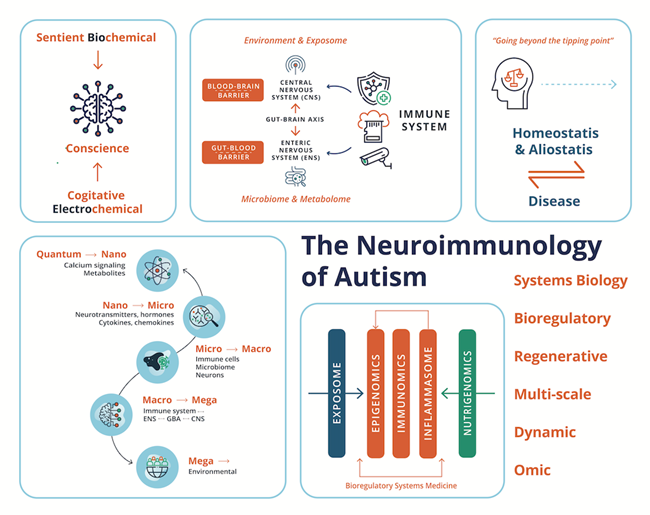
Keywords:
1. Introduction
2. Epidemiological Preambles of ASD
3. Underpinnings of ASD: From Genetic to Environmental Etiologies
4. ASD from Biochemical & Psychiatric Standpoints to Correlates of Consciousness
5. The Immune System: A System of Relations
6. “The ASD Bone Is Connected to the Immune Bone, the Digestive Bone & the Brain Bone”
7. The Pathophysiology of ASD
7.1. ASD, Inflammation & Oxidative Stress
7.2. Innate Immune Deregulation, Encephalitis & ASD
7.2.1. The Pathophysiology of Glial Cells in ASD
7.2.2. The Pathophysiology of NK Cells in ASD
7.2.3. The Pathophysiology of Mast Cells & Dendritic Cells in ASD
7.2.4. The Pathophysiology of Platelets in ASD
7.3. Adaptive Immune Dysfunction, Autoimmune Conditions, & the Pathophysiology of ASD
7.4. Autoimmune Encephalitis, N-Methyl-D-Aspartate (NMDAR) Encephalitis & ASD
7.5. GI Pathology, the Immune System & Pathophysiology of ASD
7.6. Sex Hormones, the Pathogenesis of ASD & the Immune System
8. Future Directions & Therapeutic Modalities for ASD
8.1. ASD: Viewpoints from Environmental Exposomes, Omics Technologies & BrSYS Medicine
8.2. ASD Therapeutic Modalities: Standard of Care & State of the ART
8.3. ASD Therapeutics: Personalized & Precision Nutrition and Mind-Body Modalities
9. Concluding Remarks
Funding
Data Availability
Acknowledgments
Competing Interests
Credit Authorship Contribution Statement
References
- Abbaoui, A., Fatoba, O., & Yamashita, T. (2023). Meningeal T cells function in the central nervous system homeostasis and neurodegenerative diseases. Frontiers in Cellular Neuroscience, 17. [CrossRef]
- Abbott, N. J., Patabendige, A. A. K., Dolman, D. E. M., Yusof, S. R., & Begley, D. J. (2010). Structure and function of the blood–brain barrier. Special Issue: Blood Brain Barrier, 37(1), 13–25. [CrossRef]
- Abnave, P., & Ghigo, E. (2019). Role of the immune system in regeneration and its dynamic interplay with adult stem cells. Planarian Regeneration, 87, 160–168. [CrossRef]
- Abo-Shaban, T., Sharna, S. S., Hosie, S., Lee, C. Y. Q., Balasuriya, G. K., & McKeown, S. J. (2023). Issues for patchy tissues: defining roles for gut-associated lymphoid tissue in neurodevelopment and disease. Journal of Neural Transmission, 130(3), 269–280. [CrossRef]
- Ackerman, L. S. (2006). Sex Hormones and the Genesis of Autoimmunity. Archives of Dermatology, 142(3), 371–376. [CrossRef]
- Adams, J. B., Borody, T. J., Kang, D.-W., Khoruts, A., Krajmalnik-Brown, R., & Sadowsky, M. J. (2019). Microbiota transplant therapy and autism: lessons for the clinic. Expert Review of Gastroenterology & Hepatology, 13(11), 1033–1037. [CrossRef]
- Ahmad, S. F., Ansari, M. A., Nadeem, A., Bakheet, S. A., AL-Ayadhi, L. Y., & Attia, S. M. (2019). Elevated IL-16 expression is associated with development of immune dysfunction in children with autism. Psychopharmacology, 236(2), 831–838. [CrossRef]
- Ahmad, S. F., Nadeem, A., Ansari, M. A., Bakheet, S. A., Attia, S. M., Zoheir, K. M. A., et al. (2017). Imbalance between the anti- and pro-inflammatory milieu in blood leukocytes of autistic children. Molecular Immunology, 82, 57–65. [CrossRef]
- Ahmad, S. F., Zoheir, K. M. A., Ansari, M. A., Nadeem, A., Bakheet, S. A., AL-Ayadhi, L. Y., et al. (2017). Dysregulation of Th1, Th2, Th17, and T regulatory cell-related transcription factor signaling in children with autism. Molecular Neurobiology, 54(6), 4390–4400. [CrossRef]
- Aideyan, B., Martin, G. C., & Beeson, E. T. (2020). A Practitioner’s Guide to Breathwork in Clinical Mental Health Counseling. Journal of Mental Health Counseling, 42(1), 78–94. [CrossRef]
- Akdis, C. A. (2021). Does the epithelial barrier hypothesis explain the increase in allergy, autoimmunity and other chronic conditions? Nature Reviews Immunology, 21(11), 739–751. [CrossRef]
- Akers, J. S., Davis, T. N., Gerow, S., & Avery, S. (2020). Decreasing motor stereotypy in individuals with autism spectrum disorder: A systematic review. Research in Autism Spectrum Disorders, 77, 101611. [CrossRef]
- Albert, L. J., & Inman, R. D. (2000). Gram-negative pathogens and molecular mimicry: is there a case for mistaken identity? Trends in Microbiology, 8(10), 444–445. [CrossRef]
- Albert L. J. & Inman R. D. (1999). Molecular Mimicry and Autoimmunity. New England Journal of Medicine, 341(27), 2068–2074. [CrossRef]
- Allen, M., Huang, B. S., Notaras, M. J., Lodhi, A., Barrio-Alonso, E., Lituma, P. J., et al. (2022). Astrocytes derived from ASD individuals alter behavior and destabilize neuronal activity through aberrant Ca2+ signaling. Molecular Psychiatry, 27(5), 2470–2484. [CrossRef]
- Almahayni, O., & Hammond, L. (2024). Does the Wim Hof Method have a beneficial impact on physiological and psychological outcomes in healthy and non-healthy participants? A systematic review. PLOS One, 19(3), e0286933. [CrossRef]
- Alsayouf, H. A. (2024). Growing evidence of pharmacotherapy effectiveness in managing attention-deficit/hyperactivity disorder in young children with or without autism spectrum disorder: a minireview. Frontiers in Psychiatry, 15. [CrossRef]
- American Psychiatric Association, D., & American Psychiatric Association, D. (2013). Diagnostic & statistical manual of mental disorders: DSM-5 (Vol. 5). American Psychiatric Association Washington, DC.
- American Psychiatric Association Staff, & Spitzer, R. L. (1980). Diagnostic & Statistical Manual of Mental Disorders (DSM-III). American Psychiatric Publishing.
- Amor, S., Puentes, F., Baker, D., & Van Der Valk, P. (2010). Inflammation in neurodegenerative diseases. Immunology, 129(2), 154–169. [CrossRef]
- An, J., Liu, Y., Wang, Y., Fan, R., Hu, X., Zhang, F., et al. (2022). The Role of Intestinal Mucosal Barrier in Autoimmune Disease: A Potential Target. Frontiers in Immunology, 13. [CrossRef]
- Anderson, M., Hooker, B. S., & Herbert, M. (2008). Bridging from cells to cognition in autism pathophysiology: biological pathways to defective brain function and plasticity. American Journal of Biochemistry & Biotechnology, 4 (2): 167-176, 4(PNNL-SA-54664). [CrossRef]
- Angajala, A., Lim, S., Phillips, J. B., Kim, J.-H., Yates, C., You, Z., & Tan, M. (2018). Diverse roles of mitochondria in immune responses: novel insights into immuno-metabolism. Frontiers in Immunology, 9, 1605. [CrossRef]
- Angelidou, A., Alysandratos, K.-D., Asadi, S., Zhang, B., Francis, K., Vasiadi, M., et al. (2011). Brief report:“allergic symptoms” in children with autism spectrum disorders. More than meets the eye? Journal of Autism & Developmental Disorders, 41, 1579–1585. [CrossRef]
- Ardalan, M., & Mallard, C. (2024). From hormones to behavior through microglial mitochondrial function. Brain, Behavior, & Immunity, 117, 471–472. [CrossRef]
- Ashwood, P., & Van de Water, J. (2004). A review of autism and the immune response. Clinical & Developmental Immunology, 11(2), 165.
- Asperger, H. (1944). Die „Autistischen psychopathen” im kindesalter. Archiv für Psychiatrie und Nervenkrankheiten, 117(1), 76–136. [CrossRef]
- Azzalini, D., Rebollo, I., & Tallon-Baudry, C. (2019). Visceral Signals Shape Brain Dynamics and Cognition. Trends in Cognitive Sciences, 23(6), 488–509. [CrossRef]
- Bach, J.-F. (2018). The hygiene hypothesis in autoimmunity: the role of pathogens and commensals. Nature Reviews Immunology, 18(2), 105–120. [CrossRef]
- Badimon, L., Vilahur, G., & Padro, T. (2017). Systems biology approaches to understand the effects of nutrition and promote health. British Journal of Clinical Pharmacology, 83(1), 38–45. [CrossRef]
- Balcells, C., Xu, Y., Gil-Solsona, R., Maitre, L., Gago-Ferrero, P., & Keun, H. C. (2024). Blurred lines: Crossing the boundaries between the chemical exposome and the metabolome. Current Opinion in Chemical Biology, 78, 102407. [CrossRef]
- Baquero, F., Coque, T. M., Galán, J. C., & Martinez, J. L. (2021). The Origin of Niches and Species in the Bacterial World. Frontiers in Microbiology, 12. [CrossRef]
- Barbaro, J., & Dissanayake, C. (2009). Autism spectrum disorders in infancy and toddlerhood: a review of the evidence on early signs, early identification tools, and early diagnosis. Journal of Developmental & Behavioral Pediatrics, 30(5), 447–459. [CrossRef]
- Barlattani, T., D’Amelio, C., Cavatassi, A., De Luca, D., Di Stefano, R., Di Berardo, A., et al. (2023). Autism spectrum disorders and psychiatric comorbidities: a narrative review. Journal of Psychopathology. [CrossRef]
- Baron-Cohen, S., Ring, H. A., Bullmore, E. T., Wheelwright, S., Ashwin, C., & Williams, S. C. R. (2000). The amygdala theory of autism. Neuroscience & Biobehavioral Reviews, 24(3), 355–364. [CrossRef]
- Baron-Cohen, S. (2000). Theory of mind and autism: A review. In International Review of Research in Mental Retardation (Vol. 23, pp. 169–184). Academic Press. [CrossRef]
- Baron-Cohen, S. (2004). The cognitive neuroscience of autism. Journal of Neurology, Neurosurgery & Psychiatry, 75(7), 945–948. [CrossRef]
- Basheer, S., Venkataswamy, M. M., Christopher, R., Van Amelsvoort, T., Srinath, S., Girimaji, S. C., & Ravi, V. (2018). Immune aberrations in children with Autism Spectrum Disorder: a case-control study from a tertiary care neuropsychiatric hospital in India. Psychoneuroendocrinology, 94, 162–167. [CrossRef]
- Baumann, N., & Pham-Dinh, D. (2001). Biology of oligodendrocyte and myelin in the mammalian central nervous system. Physiological Reviews, 81(2), 871–927. [CrossRef]
- Bauman, M. L. (2010). Medical comorbidities in autism: challenges to diagnosis and treatment. Neurotherapeutics, 7, 320–327. [CrossRef]
- Becker, E. B. E., & Stoodley, C. J. (2013). Chapter One - Autism Spectrum Disorder and the Cerebellum. In G. Konopka (Ed.), International Review of Neurobiology (Vol. 113, pp. 1–34). Academic Press. [CrossRef]
- Bei, R., Masuelli, L., Palumbo, C., Modesti, M., & Modesti, A. (2009). A common repertoire of autoantibodies is shared by cancer and autoimmune disease patients: Inflammation in their induction and impact on tumor growth. Cancer Letters, 281(1), 8–23. [CrossRef]
- Benakis, C., Martin-Gallausiaux, C., Trezzi, J.-P., Melton, P., Liesz, A., & Wilmes, P. (2020). The microbiome-gut-brain axis in acute and chronic brain diseases. Neurobiology of Disease, 61, 1–9. [CrossRef]
- Beopoulos, A., Gea, M., Fasano, A., & Iris, F. (2021). Autonomic nervous system neuroanatomical alterations could provoke and maintain gastrointestinal dysbiosis in autism spectrum disorder (ASD): a novel microbiome–host interaction mechanistic hypothesis. Nutrients, 14(1), 65. [CrossRef]
- Bernard, S., Enayati, A., Redwood, L., Roger, H., & Binstock, T. (2001). Autism: a novel form of mercury poisoning. Medical Hypotheses, 56(4), 462–471. [CrossRef]
- Berthoud, H.-R., & Neuhuber, W. L. (2000). Functional and chemical anatomy of the afferent vagal system. Autonomic Neuroscience: Basic & Clinical, 85(1), 1–17. [CrossRef]
- Bethell, C. D., Kogan, M. D., Strickland, B. B., Schor, E. L., Robertson, J., & Newacheck, P. W. (2011). A national and state profile of leading health problems and health care quality for US children: key insurance disparities and across-state variations. Academic Pediatrics, 11(3), S22–S33. [CrossRef]
- Bhagavati, S. (2021). Autoimmune disorders of the nervous system: pathophysiology, clinical features, and therapy. Frontiers in Neurology, 12, 664664. [CrossRef]
- Bien, C. G., & Elger, C. E. (2007). Limbic encephalitis: A cause of temporal lobe epilepsy with onset in adult life. Epilepsy & Behavior, 10(4), 529–538. [CrossRef]
- Bjørklund, G., Meguid, N. A., El-Bana, M. A., Tinkov, A. A., Saad, K., Dadar, M., et al. (2020). Oxidative Stress in Autism Spectrum Disorder. Molecular Neurobiology, 57(5), 2314–2332. [CrossRef]
- Bjørklund, G., Mkhitaryan, M., Sahakyan, E., Fereshetyan, K., Meguid, N. A., Hemimi, M., et al. (2024). Linking Environmental Chemicals to Neuroinflammation and Autism Spectrum Disorder: Mechanisms and Implications for Prevention. Molecular Neurobiology, 61(9), 6328–6340. [CrossRef]
- Blaxill, M. F., Redwood, L., & Bernard, S. (2004). Thimerosal and autism? A plausible hypothesis that should not be dismissed. Medical Hypotheses, 62(5), 788–794. [CrossRef]
- Bleuler, E. (1911). Dementia praecox: oder Gruppe der Schizophrenien. F. Deuticke.
- Bleuler, E. (1951). Autistic thinking. In Organization & pathology of thought: Selected sources (pp. 399–437). Columbia University Press.
- Bojarskaite, L., Bjørnstad, D. M., Pettersen, K. H., Cunen, C., Hermansen, G. H., & Åbjørsbråten, K. S. (2020). Astrocytic Ca2+ signaling is reduced during sleep and is involved in the regulation of slow wave sleep. Nature Communications, 11(1), 3240. [CrossRef]
- Bordenstein, S. R., & Theis, K. R. (2015). Host Biology in Light of the Microbiome: Ten Principles of Holobionts and Hologenomes. PLOS Biology, 13(8), e1002226. [CrossRef]
- Breece, E., Paciotti, B., Nordahl, C. W., Ozonoff, S., Van de Water, J. A., Rogers, S. J., et al. (2013). Myeloid dendritic cells frequencies are increased in children with autism spectrum disorder and associated with amygdala volume and repetitive behaviors. Inflammation & Mental Health, 31, 69–75. [CrossRef]
- Brincker, M., & Torres, E. B. (2013). Noise from the periphery in autism. Frontiers in Integrative Neuroscience, 7. [CrossRef]
- Brown, A. C., & Mehl-Madrona, L. (2011). Autoimmune and gastrointestinal dysfunctions: does a subset of children with autism reveal a broader connection? Expert Review of Gastroenterology & Hepatology, 5(4), 465–477. [CrossRef]
- Buie, T., Campbell, D. B., Fuchs, G. J., III, Furuta, G. T., Levy, J., VandeWater, J., et al. (2010). Evaluation, Diagnosis, and Treatment of Gastrointestinal Disorders in Individuals with ASDs: A Consensus Report. Pediatrics, 125(Supplement_1), S1–S18. [CrossRef]
- Burnouf, T., & Walker, T. L. (2022). The multifaceted role of platelets in mediating brain function. Blood, 140(8), 815–827. [CrossRef]
- Burns-Naas, L. A., Dearman, R. J., Germolec, D. R., Kaminski, N. E., Kimber, I., Ladics, G. S., et al. (2006). “Omics” Technologies and the Immune System. Toxicology Mechanisms & Methods, 16(2–3), 101–119. [CrossRef]
- Cakir, J., Frye, R. E., & Walker, S. J. (2020). The lifetime social cost of autism: 1990–2029. Research in Autism Spectrum Disorders, 72, 101502. [CrossRef]
- Calçada, D., Vianello, D., Giampieri, E., Sala, C., Castellani, G., de Graaf, A., et al. (2014). The role of low-grade inflammation and metabolic flexibility in aging and nutritional modulation thereof: a systems biology approach. Mechanisms of Ageing & Development, 136, 138–147. [CrossRef]
- Cantando, I., Centofanti, C., D’Alessandro, G., Limatola, C., & Bezzi, P. (2024). Metabolic dynamics in astrocytes and microglia during post-natal development and their implications for autism spectrum disorders. Frontiers in Cellular Neuroscience, 18, 1354259. [CrossRef]
- Carré, A., Chevallier, C., Robel, L., Barry, C., Maria, A.-S., Pouga, L., et al. (2015). Tracking social motivation systems deficits: the affective neuroscience view of autism. Journal of Autism & Developmental Disorders, 45, 3351–3363. [CrossRef]
- CDC. (2021). Managing Chronic Health Conditions. CDC Healthy Schools. https://www.cdc.gov/healthyschools/chronicconditions.htm. Accessed 1 June 2024.
- Cekici, H., & Sanlier, N. (2019). Current nutritional approaches in managing autism spectrum disorder: A review. Nutritional Neuroscience, 22(3), 145–155. [CrossRef]
- Cellot, G., & Cherubini, E. (2014). GABAergic Signaling as Therapeutic Target for Autism Spectrum Disorders. Frontiers in Pediatrics, 2. [CrossRef]
- Chambers, C., & Schaefer, C. (2015). 2.10 - Epilepsy and antiepileptic medications. In C. Schaefer, P. Peters, & R. K. Miller (Eds.), Drugs During Pregnancy and Lactation (Third Edition) (pp. 251–291). San Diego: Academic Press. [CrossRef]
- Chavez, J. A., & Zappaterra, M. (2023). Can Wim Hof Method breathing induce conscious metabolic waste clearance of the brain? Medical Hypotheses, 177, 111118. [CrossRef]
- Chen, C.-C., & Chiu, C.-H. (2022). Current and future applications of fecal microbiota transplantation for children. Biomedical Journal, 45(1), 11–18. [CrossRef]
- Chia, S. L., Kapoor, S., Carvalho, C., Bajénoff, M., & Gentek, R. (2023). Mast cell ontogeny: From fetal development to life-long health and disease. Immunological Reviews, 315(1), 31–53. [CrossRef]
- Chirumbolo, S., & Bjørklund, G. (2017). PERM hypothesis: the fundamental machinery able to elucidate the role of xenobiotics and hormesis in cell survival and homeostasis. International Journal of Molecular Sciences, 18(1), 165. [CrossRef]
- Choi, H., Kim, J. H., Yang, H. S., Kim, J. Y., Cortese, S., Smith, L., et al. (2024). Pharmacological and non-pharmacological interventions for irritability in autism spectrum disorder: a systematic review and meta-analysis with the GRADE assessment. Molecular Autism, 15(1), 7. [CrossRef]
- Ciéslińska, A., Kostyra, E., & Savelkoul, H. F. (2017). Treating autism spectrum disorder with gluten-free and casein-free diet: the underlying microbiota-gut-brain axis mechanisms. HSOA Journal of Clinical Immunology & Immunotherapy, 3.
- Clappison, E., Hadjivassiliou, M., & Zis, P. (2020). Psychiatric Manifestations of Coeliac Disease, a Systematic Review and Meta-Analysis. Nutrients, 12(1). [CrossRef]
- Copf, T. (2016). Impairments in dendrite morphogenesis as etiology for neurodevelopmental disorders and implications for therapeutic treatments. Neuroscience & Biobehavioral Reviews, 68, 946–978. [CrossRef]
- Costello, E. K., Lauber, C. L., Hamady, M., Fierer, N., Gordon, J. I., & Knight, R. (2009). Bacterial Community Variation in Human Body Habitats Across Space and Time. Science, 326(5960), 1694–1697. [CrossRef]
- Coury, D. L., Anagnostou, E., Manning-Courtney, P., Reynolds, A., Cole, L., McCoy, R., et al. (2012). Use of Psychotropic Medication in Children and Adolescents with Autism Spectrum Disorders. Pediatrics, 130(Supplement_2), S69–S76. [CrossRef]
- Crépeaux, G., Authier, F.-J., Exley, C., Luján, L., & Gherardi, R. K. (2020). The role of aluminum adjuvants in vaccines raises issues that deserve independent, rigorous and honest science. Journal of Trace Elements in Medicine & Biology, 62, 126632. [CrossRef]
- Cryan, J. F., O’Riordan, K. J., Cowan, C. S. M., Sandhu, K. V., Bastiaanssen, T. F. S., Boehme, M., et al. (2019). The Microbiota-Gut-Brain Axis. Physiological Reviews, 99(4), 1877–2013. [CrossRef]
- Csaba, G. (2014). Hormones in the immune system and their possible role. A critical review. Acta Microbiologica et Immunologica Hungarica, 61(3), 241–260. [CrossRef]
- Cutolo, M., Capellino, S., Sulli, A., Serioli, B., Secchi, M. E., Villaggio, B., & Straub, R. H. (2006). Estrogens and autoimmune diseases. Annals of the New York Academy of Sciences, 1089(1), 538–547. [CrossRef]
- Cwik, J. C. (2021). Spiritual needs of people with autism spectrum disorder. In Spiritual Needs in Research and Practice: The Spiritual Needs Questionnaire as a Global Resource for Health and Social Care (pp. 265–280). Springer.
- Cruz-Machado, S. S., Campos, L. M. G., Fadini, C. C., Anderson, G., Markus, R. P., & Pinato, L. (2021). Disrupted nocturnal melatonin in autism: Association with tumor necrosis factor and sleep disturbances. Journal of Pineal Research, 70(3), e12715. [CrossRef]
- Daëron, M. (2022). The immune system as a system of relations. Frontiers in Immunology, 13, 984678. [CrossRef]
- D’Agostino, P. M., Gottfried-Blackmore, A., Anandasabapathy, N., & Bulloch, K. (2012). Brain dendritic cells: biology and pathology. Acta Neuropathologica, 124(5), 599–614. [CrossRef]
- Daly, E., D. Tricklebank, M., & Wichers, R. (2019). Chapter Two - Neurodevelopmental roles and the serotonin hypothesis of autism spectrum disorder. In M. D. Tricklebank & E. Daly (Eds.), The Serotonin System (pp. 23–44). Academic Press. [CrossRef]
- Davenport, P., & Sola-Visner, M. (2022). Platelets in the neonate: Not just a small adult. Research & Practice in Thrombosis & Haemostasis, 6(3), e12719. [CrossRef]
- Davies, S., Bishop, D., Manstead, A. S., & Tantam, D. (1994). Face perception in children with autism and Asperger’s syndrome. Journal of Child Psychology & Psychiatry, 35(6), 1033–1057. [CrossRef]
- D’Adamo, C. R., Nelson, J. L., Miller, S. N., Rickert Hong, M., Lambert, E., & Tallman Ruhm, H. (2024). Reversal of Autism Symptoms among Dizygotic Twins through a Personalized Lifestyle and Environmental Modification Approach: A Case Report and Review of the Literature. Journal of Personalized Medicine, 14(6), 641. [CrossRef]
- D’Angelo, E. (2018). Physiology of the cerebellum. Handbook of Clinical Neurology, 154, 85–108. [CrossRef]
- De Giorgio, R., Guerrini, S., Barbara, G., Stanghellini, V., De Ponti, F., Corinaldesi, R., et al. (2004). Inflammatory neuropathies of the enteric nervous system. Gastroenterology, 126(7), 1872–1883. [CrossRef]
- Delafield-Butt, J., & Ciaunica, A. (2024). The sensorimotor foundations of bodily self-consciousness in utero. [CrossRef]
- Dempsey, J. L., Little, M., & Cui, J. Y. (2019). Gut microbiome: An intermediary to neurotoxicity. NeuroToxicology, 75, 41–69. [CrossRef]
- Deng, W., Yi, P., Xiong, Y., Ying, J., Lin, Y., Dong, Y., et al. (2024). Gut Metabolites Acting on the Gut-Brain Axis: Regulating the Functional State of Microglia. Aging & Disease, 15(2), 480.
- Denman, A. M. (1991). Sex hormones, autoimmune diseases, and immune responses. BMJ, 303(6793), 2–3. [CrossRef]
- Deth, R. C. (2013). Autism: A redox/methylation disorder. Global Advances in Health & Medicine, 2(6), 68–73. [CrossRef]
- Dethlefsen, L., McFall-Ngai, M., & Relman, D. A. (2007). An ecological and evolutionary perspective on human–microbe mutualism and disease. Nature, 449(7164), 811–818. [CrossRef]
- Di Liberto, D., D’Anneo, A., Carlisi, D., Emanuele, S., De Blasio, A., Calvaruso, G., et al. (2020). Brain Opioid Activity and Oxidative Injury: Different Molecular Scenarios Connecting Celiac Disease and Autistic Spectrum Disorder. Brain Sciences, 10(7). [CrossRef]
- Dietz, P. M., Rose, C. E., McArthur, D., & Maenner, M. (2020). National and state estimates of adults with autism spectrum disorder. Journal of Autism & Developmental Disorders, 50, 4258–4266. [CrossRef]
- Dimmeler, S., Ding, S., Rando, T. A., & Trounson, A. (2014). Translational strategies and challenges in regenerative medicine. Nature Medicine, 20(8), 814–821. [CrossRef]
- D’Mello, A. M., & Stoodley, C. J. (2015). Cerebro-cerebellar circuits in autism spectrum disorder. Frontiers in Neuroscience, 9. [CrossRef]
- Doherty, M., Foley, K.-R., & Schloss, J. (2024). Complementary and Alternative Medicine for Autism – A Systematic Review. Journal of Autism & Developmental Disorders. [CrossRef]
- Donald, K., & Finlay, B. B. (2023). Early-life interactions between the microbiota and immune system: impact on immune system development and atopic disease. Nature Reviews Immunology, 23(11), 735–748. [CrossRef]
- Dong, Y., & Benveniste, E. N. (2001). Immune function of astrocytes. Glia, 36(2), 180–190. [CrossRef]
- Dukhinova, M., Kuznetsova, I., Kopeikina, E., Veniaminova, E., Yung, A. W. Y., Veremeyko, T., et al. (2018). Platelets mediate protective neuroinflammation and promote neuronal plasticity at the site of neuronal injury. Brain, Behavior, and Immunity, 74, 7–27. [CrossRef]
- Ebrahimi M, S., Rostam-Abadi, Y., & Rezaei, N. (2021). Autism spectrum disorders and natural killer cells: a review on pathogenesis and treatment. Expert Review of Clinical Immunology, 17(1), 27–35. [CrossRef]
- Edmiston, E., Ashwood, P., & Van de Water, J. (2017). Autoimmunity, Autoantibodies, and Autism Spectrum Disorder. Biological Psychiatry, 81(5), 383–390. [CrossRef]
- El-Ansary, A., Bhat, R. S., & Zayed, N. (2020). Gut Microbiome and Sex Bias in Autism Spectrum Disorders. Current Behavioral Neuroscience Reports, 7(1), 22–31. [CrossRef]
- Enstrom, A. M., Lit, L., Onore, C. E., Gregg, J. P., Hansen, R. L., Pessah, I. N., et al. (2009). Altered gene expression and function of peripheral blood natural killer cells in children with autism. Brain, Behavior, & Immunity, 23(1), 124–133. [CrossRef]
- Epel, E. S., McEwen, B. S., & Ickovics, J. R. (1998). Embodying Psychological Thriving: Physical Thriving in Response to Stress. Journal of Social Issues, 54(2), 301–322. [CrossRef]
- Estes, M. L., & McAllister, A. K. (2016). Maternal immune activation: Implications for neuropsychiatric disorders. Science, 353(6301), 772–777. [CrossRef]
- Exley, C. (2013). Human exposure to aluminium. Environmental Science: Processes & Impacts, 15(10), 1807–1816. [CrossRef]
- Exley, C., & Clarkson, E. (2020). Aluminium in human brain tissue from donors without neurodegenerative disease: A comparison with Alzheimer’s disease, multiple sclerosis and autism. Scientific Reports, 10(1), 7770. [CrossRef]
- Faas, M., & De Vos, P. (2020). Mitochondrial function in immune cells in health and disease. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 1866(10), 165845. [CrossRef]
- Hamidreza, F., & Morteza, K. (2018). Overview of the Recent Advances in Pathophysiology and Treatment for Autism. (Formerly Current Drug Targets - CNS & Neurological Disorders), 17(8), 590–594. [CrossRef]
- Fan, G., Ma, J., Ma, R., Suo, M., Chen, Y., Zhang, S., et al. (2023). Microglia modulate neurodevelopment in autism spectrum disorder and schizophrenia. International Journal of Molecular Sciences, 24(24), 17297. [CrossRef]
- Farmer, C. A., Thurm, A. E., Honnekeri, B., Kim, P., Swedo, S. E., & Han, J. C. (2021). The contribution of platelets to peripheral BDNF elevation in children with autism spectrum disorder. Scientific Reports, 11(1), 18158. [CrossRef]
- Fasano, A. (2012). Leaky Gut and Autoimmune Diseases. Clinical Reviews in Allergy & Immunology, 42(1), 71–78. [CrossRef]
- Fatemi, S. H., Halt, A. R., Realmuto, G., Earle, J., Kist, D. A., Thuras, P., & Merz, A. (2002). Purkinje Cell Size Is Reduced in Cerebellum of Patients with Autism. Cellular & Molecular Neurobiology, 22(2), 171–175. [CrossRef]
- Ferri, S. L., Abel, T., & Brodkin, E. S. (2018). Sex Differences in Autism Spectrum Disorder: a Review. Current Psychiatry Reports, 20(2), 9. [CrossRef]
- Filiano, A. J., Xu, Y., Tustison, N. J., Marsh, R. L., Baker, W., Smirnov, I., et al. (2016). Unexpected role of interferon-γ in regulating neuronal connectivity and social behaviour. Nature, 535(7612), 425–429. [CrossRef]
- Filipek, P. A., Juranek, J., Nguyen, M. T., Cummings, C., & Gargus, J. J. (2004). Relative carnitine deficiency in autism. Journal of Autism and Developmental Disorders, 34, 615–623. [CrossRef]
- Fincham, G. W., Strauss, C., Montero-Marin, J., & Cavanagh, K. (2023). Effect of breathwork on stress and mental health: A meta-analysis of randomised-controlled trials. Scientific Reports, 13(1), 432. [CrossRef]
- Fleming, D. (1984). Walter B. Cannon and Homeostasis. Social Research, 51(3), 609–640. http://www.jstor.org/stable/40970955.
- Folstein, S., & Rutter, M. (1977). Genetic influences and infantile autism. Nature, 265(5596), 726–728. [CrossRef]
- Forbes, J. D., Van Domselaar, G., & Bernstein, C. N. (2016). The Gut Microbiota in Immune-Mediated Inflammatory Diseases. Frontiers in Microbiology, 7. [CrossRef]
- Fraguas, D., Díaz-Caneja, C., Pina-Camacho, L., Moreno, C., Durán Cutilla, M., Ayora, M., et al. (2019). Dietary Interventions for Autism Spectrum Disorder: A Meta-analysis. Pediatrics, 144, e20183218. [CrossRef]
- Frith, U. (1994). Autism and theory of mind in everyday life. Social Development, 3(2), 108–124. [CrossRef]
- Frith, U., & Happé, F. (1994). Autism: beyond “theory of mind.” Cognition, 50(1), 115–132. [CrossRef]
- Frith, U., & Happé, F. (1999). Theory of Mind and Self-Consciousness: What Is It Like to Be Autistic? Mind & Language, 14(1), 82–89. [CrossRef]
- Frye, R. E., Rincon, N., McCarty, P. J., Brister, D., Scheck, A. C., & Rossignol, D. A. (2024). Biomarkers of mitochondrial dysfunction in autism spectrum disorder: A systematic review and meta-analysis. Neurobiology of Disease, 106520. [CrossRef]
- Fu, J., Schroder, K., & Wu, H. (2024). Mechanistic insights from inflammasome structures. Nature Reviews Immunology, 24(7), 518–535. [CrossRef]
- Fuller, R., Rahona, E., Fisher, S., Caravanos, J., Webb, D., Kass, D., et al. (2018). Pollution and non-communicable disease: time to end the neglect. The Lancet Planetary Health, 2(3), e96–e98. [CrossRef]
- Furness, J. B., Callaghan, B. P., Rivera, L. R., & Cho, H.-J. (2014). The Enteric Nervous System and Gastrointestinal Innervation: Integrated Local and Central Control. In M. Lyte & J. F. Cryan (Eds.), Microbial Endocrinology: The Microbiota-Gut-Brain Axis in Health and Disease (pp. 39–71). New York, NY: Springer New York. [CrossRef]
- Gable, M. S., Sheriff, H., Dalmau, J., Tilley, D. H., & Glaser, C. A. (2012). The Frequency of Autoimmune N-Methyl-D-Aspartate Receptor Encephalitis Surpasses That of Individual Viral Etiologies in Young Individuals Enrolled in the California Encephalitis Project. Clinical Infectious Diseases, 54(7), 899–904. [CrossRef]
- Gabriele, S., Sacco, R., & Persico, A. M. (2014). Blood serotonin levels in autism spectrum disorder: A systematic review and meta-analysis. European Neuropsychopharmacology, 24(6), 919–929. [CrossRef]
- Galvez-Contreras, A. Y., Zarate-Lopez, D., Torres-Chavez, A. L., & Gonzalez-Perez, O. (2020). Role of oligodendrocytes and myelin in the pathophysiology of autism spectrum disorder. Brain Sciences, 10(12), 951. [CrossRef]
- Garg, D., Mohammad, S. S., & Sharma, S. (2020). Autoimmune Encephalitis in Children: An Update. Indian Pediatrics, 57(7), 662–670. [CrossRef]
- Gaugler, T., Klei, L., Sanders, S. J., Bodea, C. A., Goldberg, A. P., Lee, A. B., et al. (2014). Most genetic risk for autism resides with common variation. Nature Genetics, 46(8), 881–885. [CrossRef]
- Geier, D. A., Kern, J. K., & Geier, M. R. (2010). The biological basis of autism spectrum disorders: Understanding causation and treatment by clinical geneticists. Acta neurobiologiae experimentalis, 70(2), 209–226. [CrossRef]
- Geier, D. A., King, P. G., Hooker, B. S., Dórea, J. G., Kern, J. K., Sykes, L. K., & Geier, M. R. (2015). Thimerosal: clinical, epidemiologic and biochemical studies. Clinica Chimica Acta, 444, 212–220. [CrossRef]
- Geng, Z.-H., Zhu, Y., Li, Q.-L., Zhao, C., & Zhou, P.-H. (2022). Enteric Nervous System: The Bridge Between the Gut Microbiota and Neurological Disorders. Frontiers in Aging Neuroscience, 14. [CrossRef]
- Genuis, S. J. (2012). What′s Out There Making Us Sick? Journal of Environmental & Public Health, 2012(1), 605137. [CrossRef]
- Gershon, M. D. (1999). The Enteric Nervous System: A Second Brain. Hospital Practice, 34(7), 31–52. [CrossRef]
- Gershon, M. D. (2022). The Shaggy Dog Story of Enteric Signaling: Serotonin, a Molecular Megillah. In N. J. Spencer, M. Costa, & S. M. Brierley (Eds.), The Enteric Nervous System II (pp. 307–318). Cham: Springer International Publishing.
- Ghanizadeh, A., Akhondzadeh, S., Hormozi, M., Makarem, A., Abotorabi-Zarchi, M., & Firoozabadi, A. (2012). Glutathione-related factors and oxidative stress in autism, a review. Current Medicinal Chemistry, 19(23), 4000–4005. [CrossRef]
- Ghaziuddin, M., & Al-Owain, M. (2013). Autism spectrum disorders and inborn errors of metabolism: an update. Pediatric Neurology, 49(4), 232–236. [CrossRef]
- Ghosh, A., Michalon, A., Lindemann, L., Fontoura, P., & Santarelli, L. (2013). Drug discovery for autism spectrum disorder: challenges and opportunities. Nature Reviews Drug Discovery, 12(10), 777–790. [CrossRef]
- Gianchecchi, E., Delfino, D. V., & Fierabracci, A. (2018). NK cells in autoimmune diseases: Linking innate and adaptive immune responses. Autoimmunity Reviews, 17(2), 142–154. [CrossRef]
- Gładysz, D., Krzywdzińska, A., & Hozyasz, K. K. (2018). Immune abnormalities in autism spectrum disorder—could they hold promise for causative treatment? Molecular Neurobiology, 55, 6387–6435. [CrossRef]
- Gobin, V., Van Steendam, K., Denys, D., & Deforce, D. (2014). Selective serotonin reuptake inhibitors as a novel class of immunosuppressants. International Immunopharmacology, 20(1), 148–156. [CrossRef]
- Goldberg, W. A., Osann, K., Filipek, P. A., Laulhere, T., Jarvis, K., Modahl, C., et al. (2003). Language and other regression: assessment and timing. Journal of Autism & Developmental Disorders, 33, 607–616. [CrossRef]
- Goldman, A. W., Burmeister, Y., Cesnulevicius, K., Herbert, M., Kane, M., Lescheid, D., et al. (2015). Bioregulatory systems medicine: an innovative approach to integrating the science of molecular networks, inflammation, and systems biology with the patient’s autoregulatory capacity? Frontiers in Physiology, 6. [CrossRef]
- Goubau, C., Buyse, G. M., Van Geet, C., & Freson, K. (2014). The contribution of platelet studies to the understanding of disease mechanisms in complex and monogenetic neurological disorders. Developmental Medicine & Child Neurology, 56(8), 724–731. [CrossRef]
- Graeber, M. B., & Streit, W. J. (2010). Microglia: biology and pathology. Acta Neuropathologica, 119, 89–105. [CrossRef]
- Graeber, M. B., & Stre’rt, W. J. (1990). Microglia: immune network in the CNS. Brain Pathology, 1(1), 2–5. [CrossRef]
- Green, D. J., Park, K., Bhatt-Mehta, V., Snyder, D., & Burckart, G. J. (2021). Regulatory Considerations for the Mother, Fetus and Neonate in Fetal Pharmacology Modeling. Frontiers in Pediatrics, 9. [CrossRef]
- Greenberg, G., & Tobach, E. (2014). Behavioral Evolution & Integrative Levels: The T.C. Schneirla Conferences Series, Volume 1. Taylor & Francis.
- Gropman, A., & Sadle, C. J. (2024). Epigenetics of autism spectrum disorder. In Neuropsychiatric Disorders & Epigenetics (pp. 81–102). Elsevier. [CrossRef]
- Gupta, N., & Gupta, M. (2024). Off-label psychopharmacological interventions for autism spectrum disorders: strategic pathways for clinicians. CNS Spectrums, 29(1), 10–25. [CrossRef]
- Gzielo, K., & Nikiforuk, A. (2021). Astroglia in Autism Spectrum Disorder. International Journal of Molecular Sciences, 22(21). [CrossRef]
- Hacohen, Y., Wright, S., Gadian, J., Vincent, A., Lim, M., Wassmer, E., & Lin, J.-P. (2016). N-methyl-d-aspartate (NMDA) receptor antibodies encephalitis mimicking an autistic regression. Developmental Medicine & Child Neurology, 58(10), 1092–1094. [CrossRef]
- Hafizi, S., Tabatabaei, D., & Lai, M.-C. (2019). Review of Clinical Studies Targeting Inflammatory Pathways for Individuals With Autism. Frontiers in Psychiatry, 10. [CrossRef]
- Hallmayer, J., Cleveland, S., Torres, A., Phillips, J., Cohen, B., Torigoe, T., et al. (2011). Genetic heritability and shared environmental factors among twin pairs with autism. Archives of General Psychiatry, 68(11), 1095–1102. [CrossRef]
- Hameroff, S. (2010). The “conscious pilot”—dendritic synchrony moves through the brain to mediate consciousness. Journal of Biological Physics, 36(1), 71–93. [CrossRef]
- Hampe, C. S., & Mitoma, H. (2022). A Breakdown of Immune Tolerance in the Cerebellum. Brain Sciences, 12(3). [CrossRef]
- Han, A., Peng, T., Xie, Y., Zhang, W., Sun, W., Xie, Y., et al. (2023). Mitochondrial-regulated Tregs: potential therapeutic targets for autoimmune diseases of the central nervous system. Frontiers in Immunology, 14. [CrossRef]
- Hanaford, A., & Johnson, S. C. (2022). The immune system as a driver of mitochondrial disease pathogenesis: a review of evidence. Orphanet Journal of Rare Diseases, 17(1), 335. [CrossRef]
- Happé, F. G. E. (1994). Wechsler IQ Profile and Theory of Mind in Autism: A Research Note. Journal of Child Psychology & Psychiatry, 35(8), 1461–1471. [CrossRef]
- Hardy, D. (2022). Autoimmune Encephalitis in Children. Pediatric Neurology, 132, 56–66. [CrossRef]
- Harkey, J. (1983). The epidemiology of selected chronic childhood health conditions. Children’s Health Care, 12(2), 62. [CrossRef]
- Harris, J. (2018). Leo Kanner and autism: a 75-year perspective. International Review of Psychiatry, 30(1), 3–17. [CrossRef]
- Haruwaka, K., Ikegami, A., Tachibana, Y., Ohno, N., Konishi, H., Hashimoto, A., et al. (2019). Dual microglia effects on blood brain barrier permeability induced by systemic inflammation. Nature Communications, 10(1), 5816. [CrossRef]
- Hasib, R. A., Ali, Md. C., Rahman, M. H., Ahmed, S., Sultana, S., Summa, S. Z., et al. (2023). Integrated gene expression profiling and functional enrichment analyses to discover biomarkers and pathways associated with Guillain-Barré syndrome and autism spectrum disorder to identify new therapeutic targets. Journal of Biomolecular Structure and Dynamics, 1–23. [CrossRef]
- Hawgood, S., Hook-Barnard, I. G., O’Brien, T. C., & Yamamoto, K. R. (2015). Precision medicine: Beyond the inflection point. Science Translational Medicine, 7(300), 300ps17-300ps17. [CrossRef]
- Hazan, S., Spradling-Reeves, K. D., Papoutsis, A., & Walker, S. J. (2020). Shotgun metagenomic sequencing identifies dysbiosis in triplet sibling with gastrointestinal symptoms and ASD. Children, 7(12), 255. [CrossRef]
- Heberling, C. A., Dhurjati, P. S., & Sasser, M. (2013). Hypothesis for a systems connectivity model of autism spectrum disorder pathogenesis: Links to gut bacteria, oxidative stress, and intestinal permeability. Medical Hypotheses, 80(3), 264–270. [CrossRef]
- Heemskerk, V. H., Daemen, M. A., & Buurman, W. A. (1999). Insulin-like growth factor-1 (IGF-1) and growth hormone (GH) in immunity and inflammation. Cytokine & Growth Factor Reviews, 10(1), 5–14. [CrossRef]
- Heiss, C. N., & Olofsson, L. E. (2019). The role of the gut microbiota in development, function and disorders of the central nervous system and the enteric nervous system. Journal of Neuroendocrinology, 31(5), e12684. [CrossRef]
- Hendriksen, E., van Bergeijk, D., Oosting, R. S., & Redegeld, F. A. (2017). Mast cells in neuroinflammation and brain disorders. Neuroscience & Biobehavioral Reviews, 79, 119–133. [CrossRef]
- Heneka, M. T., Carson, M. J., El Khoury, J., Landreth, G. E., Brosseron, F., Feinstein, D. L., et al. (2015). Neuroinflammation in Alzheimer’s disease. The Lancet Neurology, 14(4), 388–405. [CrossRef]
- Heppner, F. L., Ransohoff, R. M., & Becher, B. (2015). Immune attack: the role of inflammation in Alzheimer disease. Nature Reviews Neuroscience, 16(6), 358–372. [CrossRef]
- Herberman, R. B., & Ortaldo, J. R. (1981). Natural killer cells: their roles in defenses against disease. Science, 214(4516), 24–30. [CrossRef]
- Herbert, M. R. (2012). 10 Autism: From Static Genetic Brain Defect to Dynamic Gene-Environment-Modulated Pathophysiology. In Genetic explanations: Sense & nonsense (pp. 122–146). Harvard University Press.
- Herbert, M. R. (2014). Translational implications of a whole-body approach to brain health in autism: how transduction between metabolism and electrophysiology points to mechanisms for neuroplasticity. In Frontiers in Autism Research: New Horizons for Diagnosis & Treatment, 515–556. [CrossRef]
- Herbert, M. R., & Sage, C. (2013a). Autism and EMF? Plausibility of a pathophysiological link – Part I. Pathophysiology, 20(3), 191–209. [CrossRef]
- Herbert, M. R., & Sage, C. (2013b). Autism and EMF? Plausibility of a pathophysiological link part II. Pathophysiology, 20(3), 211–234. [CrossRef]
- Herbert, M., & Weintraub, K. (2013). The Autism Revolution: Whole-Body Strategies for Making Life All It Can Be. Random House Publishing Group.
- Hertz-Picciotto, I., Schmidt, R. J., & Krakowiak, P. (2018). Understanding environmental contributions to autism: Causal concepts and the state of science. Autism Research, 11(4), 554–586. [CrossRef]
- Hickey, A., Crabtree, J., & Stott, J. (2018). ‘Suddenly the first fifty years of my life made sense’: Experiences of older people with autism. Autism, 22(3), 357–367. [CrossRef]
- Higdon, R., Earl, R. K., Stanberry, L., Hudac, C. M., Montague, E., Stewart, E., et al. (2015). The Promise of Multi-Omics and Clinical Data Integration to Identify and Target Personalized Healthcare Approaches in Autism Spectrum Disorders. OMICS: A Journal of Integrative Biology, 19(4), 197–208. [CrossRef]
- Hill, E. L. (2004a). Executive dysfunction in autism. Trends in Cognitive Sciences, 8(1), 26–32. [CrossRef]
- Hill, E. L. (2004b). Evaluating the theory of executive dysfunction in autism. Developmental Review, 24(2), 189–233. [CrossRef]
- Hill, K. (1990). The decline of childhood mortality.
- Hiller-Sturmhöfel, S., & Bartke, A. (1998). The endocrine system - An overview. Alcohol health & Research World, 22, 153–64.
- Hirahara, K., & Nakayama, T. (2016). CD4+ T-cell subsets in inflammatory diseases: beyond the T h 1/T h 2 paradigm. International Immunology, 28(4), 163–171. [CrossRef]
- Ho, P., & Ross, D. A. (2017). More than a gut feeling: the implications of the gut microbiota in psychiatry. Biological Psychiatry, 81(5), e35–e37. [CrossRef]
- Hooker, B., Kern, J., Geier, D., Haley, B., Sykes, L., King, P., & Geier, M. (2014). Methodological issues and evidence of malfeasance in research purporting to show Thimerosal in vaccines is safe. BioMed Research International, 2014(1), 247218. [CrossRef]
- Horlin, C., Falkmer, M., Parsons, R., Albrecht, M. A., & Falkmer, T. (2014). The cost of autism spectrum disorders. PLOS One, 9(9), e106552. [CrossRef]
- Hridi, S. U., Barbour, M., Wilson, C., Franssen, A. J., Harte, T., Bushell, T. J., & Jiang, H.-R. (2021). Increased Levels of IL-16 in the Central Nervous System during Neuroinflammation Are Associated with Infiltrating Immune Cells and Resident Glial Cells. Biology, 10(6). [CrossRef]
- Huang, J., Li, X., Chen, Z., Zou, L., Healy, S., Tse, C. Y. A., & Li, C. (2023). Effects of Mind-Body Exercises on Health-related Outcomes in Children and Adolescents with Autism Spectrum Disorder: A Systematic Review. Review Journal of Autism & Developmental Disorders. [CrossRef]
- Hughes, H. K., Mills Ko, E., Rose, D., & Ashwood, P. (2018). Immune dysfunction and autoimmunity as pathological mechanisms in autism spectrum disorders. Frontiers in Cellular Neuroscience, 12, 405. [CrossRef]
- Hughes, H. K., Rose, D., & Ashwood, P. (2018). The gut microbiota and dysbiosis in autism spectrum disorders. Current Neurology & Neuroscience Reports, 18, 1–15. [CrossRef]
- Hurley-Hanson, A. E., Giannantonio, C. M., Griffiths, A. J., Hurley-Hanson, A. E., Giannantonio, C. M., & Griffiths, A. J. (2020). The costs of autism. Springer. [CrossRef]
- Hussaini, S. M., & Jang, M. H. (2018). New roles for old glue: astrocyte function in synaptic plasticity and neurological disorders. International Neurourology Journal, 22(Suppl 3), S106.
- Ichim, T. E., Solano, F., Glenn, E., Morales, F., Smith, L., Zabrecky, G., & Riordan, N. H. (2007). Stem Cell Therapy for Autism. Journal of Translational Medicine, 5(1), 30. [CrossRef]
- Irwin, J. K., MacSween, J., & Kerns, K. A. (2011). History and evolution of the autism spectrum disorders. In International Handbook of Autism & Pervasive Developmental disorders (pp. 3–16). Springer.
- Jaga, K., & Dharmani, C. (2007). The interrelation between organophosphate toxicity and the epidemiology of depression and suicide. Reviews on Environmental Health, 22(1), 57–74. [CrossRef]
- Jarrold, C., & Russell, J. (1997). Counting Abilities in Autism: Possible Implications for Central Coherence Theory. Journal of Autism & Developmental Disorders, 27(1), 25–37. [CrossRef]
- Jaswal, V. K., Wayne, A., & Golino, H. (2020). Eye-tracking reveals agency in assisted autistic communication. Scientific reports, 10(1), 7882. [CrossRef]
- Jaswal, V. K., Lampi, A. J., & Stockwell, K. M. (2024). Literacy in nonspeaking autistic people. Autism, 13623613241230709. [CrossRef]
- Jenne, C. N., Urrutia, R., & Kubes, P. (2013). Platelets: bridging hemostasis, inflammation, and immunity. International Journal of Laboratory Hematology, 35(3), 254–261. [CrossRef]
- Jiang, N. M., Cowan, M., Moonah, S. N., & Petri, W. A. (2018). The impact of systemic inflammation on neurodevelopment. Trends in Molecular Medicine, 24(9), 794–804. [CrossRef]
- Jones, J. P., Williamson, L., Konsoula, Z., Anderson, R., Reissner, K. J., & Parker, W. (2024). Evaluating the Role of Susceptibility Inducing Cofactors and of Acetaminophen in the Etiology of Autism Spectrum Disorder. Life, 14(8). [CrossRef]
- Jung, C., Hugot, J.-P., & Barreau, F. (2010). Peyer′s Patches: The Immune Sensors of the Intestine. International Journal of Inflammation, 2010(1), 823710. [CrossRef]
- Jyonouchi, H. (2009). Food allergy and autism spectrum disorders: Is there a link? Current Allergy and Asthma Reports, 9(3), 194–201. [CrossRef]
- Jyonouchi, H. (2024). Autism spectrum disorder and a possible role of anti-inflammatory treatments: experience in the pediatric allergy/immunology clinic. Frontiers in Psychiatry, 15. [CrossRef]
- Kang, D.-W., Adams, J. B., Coleman, D. M., Pollard, E. L., Maldonado, J., McDonough-Means, S., et al. (2019). Long-term benefit of Microbiota Transfer Therapy on autism symptoms and gut microbiota. Scientific Reports, 9(1), 5821. [CrossRef]
- Kanner, L. (1943). Autistic disturbances of affective contact. Nervous Child, 2(3), 217–250.
- Karhu, E., Zukerman, R., Eshraghi, R. S., Mittal, J., Deth, R. C., Castejon, A. M., et al. (2020). Nutritional interventions for autism spectrum disorder. Nutrition Reviews, 78(7), 515–531. [CrossRef]
- Karl, J. P., Hatch, A. M., Arcidiacono, S. M., Pearce, S. C., Pantoja-Feliciano, I. G., Doherty, L. A., & Soares, J. W. (2018). Effects of Psychological, Environmental and Physical Stressors on the Gut Microbiota. Frontiers in Microbiology, 9. [CrossRef]
- Karlsson, O. (2023). Chemical safety and the exposome. Emerging Contaminants, 9(2), 100225. [CrossRef]
- Kaur, I., Behl, T., Aleya, L., Rahman, M. H., Kumar, A., Arora, S., & Akter, R. (2021). Role of metallic pollutants in neurodegeneration: effects of aluminum, lead, mercury, and arsenic in mediating brain impairment events and autism spectrum disorder. Environmental Science & Pollution Research, 28, 8989–9001. [CrossRef]
- Kayser, M. S., & Dalmau, J. (2016). Anti-NMDA receptor encephalitis, autoimmunity, and psychosis. Oxidative Stress & Inflammation in Schizophrenia, 176(1), 36–40. [CrossRef]
- Keating, B. A., Lees, J. G., & Moalem-Taylor, G. (2019). The Roles of Regulatory T Cells in Central Nervous System Autoimmunity. In H. Mitoma & M. Manto (Eds.), Neuroimmune Diseases: From Cells to the Living Brain (pp. 167–193). Cham: Springer International Publishing. [CrossRef]
- Keller, A., Rimestad, M. L., Friis Rohde, J., Holm Petersen, B., Bruun Korfitsen, C., Tarp, S., et al. (2021). The Effect of a Combined Gluten- and Casein-Free Diet on Children and Adolescents with Autism Spectrum Disorders: A Systematic Review and Meta-Analysis. Nutrients, 13(2). [CrossRef]
- Kelly, L. S., Apple, C. G., Gharaibeh, R., Pons, E. E., Thompson, C. W., Kannan, K. B., et al. (2021). Stress-related changes in the gut microbiome after trauma. Journal of Trauma & Acute Care Surgery, 91(1).
- Kepser, L.-J., & Homberg, J. R. (2015). The neurodevelopmental effects of serotonin: A behavioural perspective. Special Issue: Serotonin, 277, 3–13. [CrossRef]
- Kern, J., Geier, D., Audhya, T., King, P., Sykes, L., & Geier, M. (2012). Evidence of parallels between mercury intoxication and the brain pathology in autism. Acta Neurobiologiae Experimentalis, 72(2), 113–153. [CrossRef]
- Kern, J. K., Geier, D. A., Deth, R. C., Sykes, L. K., Hooker, B. S., Love, J. M., et al. (2017). Systematic assessment of research on autism spectrum disorder (ASD) and mercury reveals conflicts of interest and the need for transparency in autism research. Science & Engineering ethics, 23, 1691–1718. [CrossRef]
- Kern, J. K., Geier, D. A., Sykes, L. K., & Geier, M. R. (2016). Relevance of Neuroinflammation and Encephalitis in Autism. Frontiers in Cellular Neuroscience, 9. [CrossRef]
- Khachadourian, V., Mahjani, B., Sandin, S., Kolevzon, A., Buxbaum, J. D., Reichenberg, A., & Janecka, M. (2023). Comorbidities in autism spectrum disorder and their etiologies. Translational Psychiatry, 13(1), 71. [CrossRef]
- Khan, M. F., & Wang, G. (2018). Environmental agents, oxidative stress and autoimmunity. Oxidative Toxicology: Role of Reactive Oxygen Species (ROS) in Health & Disease: Mechanisms, Target Organ Toxicities, & Biomarkers, 7, 22–27. [CrossRef]
- Kharrazian, D., Herbert, M., & Lambert, J. (2023). The Relationships between Intestinal Permeability and Target Antibodies for a Spectrum of Autoimmune Diseases. International Journal of Molecular Sciences, 24(22). [CrossRef]
- Khetrapal, N. (2008). The framework for disturbed affective consciousness in autism. Neuropsychiatric Disease & Treatment, 4(3), 531–533.
- Khundakji, Y., Masri, A., & Khuri-Bulos, N. (2018). Anti-NMDA receptor encephalitis in a toddler: A diagnostic challenge. International Journal of Pediatrics & Adolescent Medicine, 5(2). [CrossRef]
- Kim, D.-Y., & Camilleri, M. (2000). Serotonin: A Mediator of The Brain–Gut Connection. Official journal of the American College of Gastroenterology ACG, 95(10). [CrossRef]
- Kim, H., Cho, M., Shim, W., Kim, J., Jeon, E., Kim, D., & Yoon, S. (2017). Deficient autophagy in microglia impairs synaptic pruning and causes social behavioral defects. Molecular psychiatry, 22(11), 1576–1584. [CrossRef]
- King, B. H., Hollander, E., Sikich, L., McCracken, J. T., Scahill, L., Bregman, J. D., et al. (2009). Lack of efficacy of citalopram in children with autism spectrum disorders and high levels of repetitive behavior: citalopram ineffective in children with autism. Archives of General Psychiatry, 66(6), 583–590. [CrossRef]
- Kissane, D. W. (2012). The relief of existential suffering. Archives of Internal Medicine, 172(19), 1501–1505. [CrossRef]
- Kleine, B., & Rossmanith, W. (2016). Hormones & the Endocrine System. [CrossRef]
- Knapp, M., Romeo, R., & Beecham, J. (2009). Economic cost of autism in the UK. Autism, 13(3), 317–336. [CrossRef]
- Knudsen, G. P. (2009). Gender bias in autoimmune diseases: X chromosome inactivation in women with multiple sclerosis. Journal of the Neurological Sciences, 286(1), 43–46. [CrossRef]
- Knuesel, I., Chicha, L., Britschgi, M., Schobel, S. A., Bodmer, M., Hellings, J. A., et al. (2014). Maternal immune activation & abnormal brain development across CNS disorders. Nature Reviews Neurology, 10(11), 643–660. [CrossRef]
- Ko, C.-L., Lin, C.-K., & Lin, C.-L. (2024). Relationship between executive function and autism symptoms in preschoolers with autism spectrum disorder. Research in Developmental Disabilities, 147, 104692. [CrossRef]
- Kobayashi, N., Takahashi, D., Takano, S., Kimura, S., & Hase, K. (2019). The Roles of Peyer’s Patches and Microfold Cells in the Gut Immune System: Relevance to Autoimmune Diseases. Frontiers in Immunology, 10. [CrossRef]
- Koh, C.-H., Lee, S., Kwak, M., Kim, B.-S., & Chung, Y. (2023). CD8 T-cell subsets: heterogeneity, functions, and therapeutic potential. Experimental & Molecular Medicine, 55(11), 2287–2299. [CrossRef]
- Konteh, F. H. (2009). Urban sanitation and health in the developing world: Reminiscing the nineteenth century industrial nations. Health & Place, 15(1), 69–78. [CrossRef]
- Kovacheva, E., Gevezova, M., Maes, M., & Sarafian, V. (2024). Mast Cells in Autism Spectrum Disorder—The Enigma to Be Solved? International Journal of Molecular Sciences, 25(5), 2651. [CrossRef]
- Koyama, R., & Ikegaya, Y. (2015). Microglia in the pathogenesis of autism spectrum disorders. Neuroscience research, 100, 1–5. [CrossRef]
- Krausová, M., Braun, D., Buerki-Thurnherr, T., Gundacker, C., Schernhammer, E., Wisgrill, L., & Warth, B. (2023). Understanding the Chemical Exposome During Fetal Development and Early Childhood: A Review. Annual Review of Pharmacology & Toxicology. Annual Reviews. [CrossRef]
- Kushak I. R., & Winter S. H. (2020). Gut Microbiota and Gender in Autism Spectrum Disorders. Current Pediatric Reviews, 16(4), 249–254. [CrossRef]
- Lacivita, E., Perrone, R., Margari, L., & Leopoldo, M. (2017). Targets for Drug Therapy for Autism Spectrum Disorder: Challenges and Future Directions. Journal of Medicinal Chemistry, 60(22), 9114–9141. [CrossRef]
- Lahiri, D. K., Maloney, B., Wang, R., Sokol, D. K., Rogers, J. T., & Westmark, C. J. (2021). How autism and Alzheimer’s disease are TrAPPed. Molecular Psychiatry, 26(1), 26–29. [CrossRef]
- Lai, Y., Dhingra, R., Zhang, Z., Ball, L. M., Zylka, M. J., & Lu, K. (2022). Toward Elucidating the Human Gut Microbiota–Brain Axis: Molecules, Biochemistry, and Implications for Health and Diseases. Biochemistry, 61(24), 2806–2821. [CrossRef]
- Lam, K. S., Aman, M. G., & Arnold, L. E. (2006). Neurochemical correlates of autistic disorder: a review of the literature. Research in Developmental Disabilities, 27(3), 254–289. [CrossRef]
- Lange, K. W., Hauser, J., & Reissmann, A. (2015). Gluten-free and casein-free diets in the therapy of autism. Current Opinion in Clinical Nutrition & Metabolic Care, 18(6). [CrossRef]
- Lebow, M., Kuperman, Y., & Chen, A. (2024). Prenatal-induced psychopathologies: All roads lead to microglia. In Stress: Immunology & Inflammation (pp. 199–214). Elsevier.
- Lederberg, J. (2000). Infectious History. Science, 288(5464), 287–293. [CrossRef]
- Leiter, O., & Walker, T. L. (2019). Platelets: The missing link between the blood and brain? Progress in Neurobiology, 183, 101695. [CrossRef]
- Leo, E. E. M., & Campos, M. R. S. (2020). Effect of ultra-processed diet on gut microbiota and thus its role in neurodegenerative diseases. Nutrition, 71, 110609. [CrossRef]
- Lerer, L., & Varia, J. (2022). A long trip into the universe: Psychedelics and space travel. Frontiers in Space Technologies, 3, 899159. [CrossRef]
- Lesch, K.-P., & Waider, J. (2012). Serotonin in the Modulation of Neural Plasticity and Networks: Implications for Neurodevelopmental Disorders. Neuron, 76(1), 175–191. [CrossRef]
- Leventhal, J., Miller, J., Abecassis, M., Tollerud, D. J., & Ildstad, S. T. (2013). Evolving Approaches of Hematopoietic Stem Cell–Based Therapies to Induce Tolerance to Organ Transplants: The Long Road to Tolerance. Clinical Pharmacology & Therapeutics, 93(1), 36–45. [CrossRef]
- Li, X., Chauhan, A., Sheikh, A. M., Patil, S., Chauhan, V., Li, X.-M., et al. (2009). Elevated immune response in the brain of autistic patients. Journal of Neuroimmunology, 207(1), 111–116. [CrossRef]
- Lilienfeld, S. O., Marshall, J., Todd, J. T., & Shane, H. C. (2014). The persistence of fad interventions in the face of negative scientific evidence: Facilitated communication for autism as a case example. Evidence-Based Communication Assessment and Intervention, 8(2), 62–101. [CrossRef]
- Lindhard, T. (2019). Consciousness from the Outside-In and Inside-Out Perspective. Journal of Consciousness Exploration & Research, 10, 135–150.
- Liu, G., Pearl, A. M., Kong, L., Leslie, D. L., & Murray, M. J. (2017). A Profile on Emergency Department Utilization in Adolescents and Young Adults with Autism Spectrum Disorders. Journal of Autism & Developmental Disorders, 47(2), 347–358. [CrossRef]
- Liu, M., Liang, S., & Zhang, C. (2021). NK cells in autoimmune diseases: protective or pathogenic? Frontiers in Immunology, 12, 624687. [CrossRef]
- Liu, S.-H., Shi, X.-J., Fan, F.-C., & Cheng, Y. (2021). Peripheral blood neurotrophic factor levels in children with autism spectrum disorder: a meta-analysis. Scientific Reports, 11(1), 15. [CrossRef]
- Lleo, A. (2014). Chapter 2 - What Is an Autoantibody? In Y. Shoenfeld, P. L. Meroni, & M. E. Gershwin (Eds.), Autoantibodies (Third Edition) (pp. 13–20). San Diego: Elsevier. [CrossRef]
- Lleo, A., Invernizzi, P., Gao, B., Podda, M., & Gershwin, M. E. (2010). Definition of human autoimmunity — autoantibodies versus autoimmune disease. Special Issue on The Environment Geoepidemiology & Autoimmune Diseases, 9(5), A259–A266. [CrossRef]
- López-Cacho, J. M., Gallardo, S., Posada, M., Aguerri, M., Calzada, D., Mayayo, T., et al. (2016). Characterization of immune cell phenotypes in adults with autism spectrum disorders. Journal of Investigative Medicine, 64(7), 1179–1185. [CrossRef]
- López-Varela, S., González-Gross, M., & Marcos, A. (2002). Functional foods and the immune system: a review. European Journal of Clinical Nutrition, 56(3), S29–S33. [CrossRef]
- Lord, C., Shulman, C., & DiLavore, P. (2004). Regression and word loss in autistic spectrum disorders. Journal of Child Psychology & Psychiatry, 45(5), 936–955. [CrossRef]
- Lou, H. C. (2012). Paradigm shift in consciousness research: the child′s self-awareness and abnormalities in autism, ADHD and schizophrenia. Acta Paediatrica, 101(2), 112–119. [CrossRef]
- Luo, Y., & Wang, Z. (2024). The impact of Microglia on Neurodevelopment and Brain function in Autism. Biomedicines, 12(1), 210. [CrossRef]
- Luvián-Morales, J., Varela-Castillo, F. O., Flores-Cisneros, L., Cetina-Pérez, L., & Castro-Eguiluz, D. (2022). Functional foods modulating inflammation and metabolism in chronic diseases: a systematic review. Critical Reviews in Food Science & Nutrition, 62(16), 4371–4392. [CrossRef]
- Luyster, R., Richler, J., Risi, S., Hsu, W.-L., Dawson, G., Bernier, R., et al. (2005). Early regression in social communication in autism spectrum disorders: A CPEA study. Developmental Neuropsychology, 27(3), 311–336. [CrossRef]
- Lynch S. V., & Pedersen O. (2016). The Human Intestinal Microbiome in Health and Disease. New England Journal of Medicine, 375(24), 2369–2379. [CrossRef]
- Machado, A. P., Ratliff, H., Abdelwahab, A., Vohra, M. H., Kuang, A., Shatila, M., et al. (2023). The Safety of Immunosuppressants Used in the Treatment of Immune-Related Adverse Events due to Immune Checkpoint Inhibitors: a Systematic Review. Journal of Cancer, 14(16), 2956–2963. [CrossRef]
- Macpherson, A. J., Pachnis, V., & Prinz, M. (2023). Boundaries and integration between microbiota, the nervous system, and immunity. Immunity, 56(8), 1712–1726. [CrossRef]
- Maenner, M. J. (2021). Prevalence and characteristics of autism spectrum disorder among children aged 8 years—autism and developmental disabilities monitoring network, 11 sites, United States, 2018. MMWR. Surveillance Summaries, 70. [CrossRef]
- Maier, S. F., & Watkins, L. R. (1999). Bidirectional communication between the brain and the immune system: implications for behaviour. Animal Behaviour, 57(4), 741–751. [CrossRef]
- Maitre, L., Bustamante, M., Hernández-Ferrer, C., Thiel, D., Lau, C.-H. E., Siskos, A. P., et al. (2022). Multi-omics signatures of the human early life exposome. Nature Communications, 13(1), 7024. [CrossRef]
- Mallozzi, M., Bordi, G., Garo, C., & Caserta, D. (2016). The effect of maternal exposure to endocrine disrupting chemicals on fetal and neonatal development: A review on the major concerns. Birth Defects Research Part C: Embryo Today: Reviews, 108(3), 224–242. [CrossRef]
- Manji, H., Kato, T., Di Prospero, N. A., Ness, S., Beal, M. F., Krams, M., & Chen, G. (2012). Impaired mitochondrial function in psychiatric disorders. Nature Reviews Neuroscience, 13(5), 293–307. [CrossRef]
- Margulis, L., & Fester, R. (1991). Symbiosis as a Source of Evolutionary Innovation: Speciation & Morphogenesis. MIT Press.
- Marotta, R., Risoleo, M. C., Messina, G., Parisi, L., Carotenuto, M., Vetri, L., & Roccella, M. (2020). The neurochemistry of autism. Brain Sciences, 10(3), 163. [CrossRef]
- Marques, R. E., Marques, P. E., Guabiraba, R., & Teixeira, M. M. (2016). Exploring the Homeostatic and Sensory Roles of the Immune System. Frontiers in Immunology, 7. [CrossRef]
- Marshall, L., & Born, J. (2002). Brain-Immune interactions in sleep. In International Review of Neurobiology (Vol. 52, pp. 93–131). Academic Press. [CrossRef]
- Martin, A., Scahill, L., Anderson, G. M., Aman, M., Arnold, L. E., McCracken, J., et al. (2004). Weight and Leptin Changes Among Risperidone-Treated Youths With Autism: 6-Month Prospective Data. American Journal of Psychiatry, 161(6), 1125–1127. [CrossRef]
- Martínez-Cerdeño, V. (2017). Dendrite and spine modifications in autism and related neurodevelopmental disorders in patients and animal models. Developmental Neurobiology, 77(4), 393–404. [CrossRef]
- Marwaha, A., Leung, N., McMurchy, A. N., & Levings, M. (2012). TH17 Cells in Autoimmunity and Immunodeficiency: Protective or Pathogenic? Frontiers in Immunology, 3. [CrossRef]
- Masi, A., DeMayo, M. M., Glozier, N., & Guastella, A. J. (2017). An overview of autism spectrum disorder, heterogeneity and treatment options. Neuroscience Bulletin, 33, 183–193. [CrossRef]
- Matcovitch-Natan, O., Winter, D. R., Giladi, A., Vargas Aguilar, S., Spinrad, A., Sarrazin, S., et al. (2016). Microglia development follows a stepwise program to regulate brain homeostasis. Science, 353(6301), aad8670. [CrossRef]
- Mauri, C., & Bosma, A. (2012). Immune Regulatory Function of B Cells. Annual Review of Immunology. Annual Reviews. [CrossRef]
- Mayer, E. A. (2011). Gut feelings: the emerging biology of gut–brain communication. Nature Reviews Neuroscience, 12(8), 453–466. [CrossRef]
- Mayer, E. A., Nance, K., & Chen, S. (2022). The Gut–Brain Axis. Annual Review of Medicine. Annual Reviews. [CrossRef]
- McCarthy, M. M., & Wright, C. L. (2017). Convergence of Sex Differences and the Neuroimmune System in Autism Spectrum Disorder. Biological Psychiatry, 81(5), 402–410. [CrossRef]
- McCormick, C. M., Furey, B. F., Child, M., Sawyer, M. J., & Donohue, S. M. (1998). Neonatal sex hormones have `organizational’ effects on the hypothalamic-pituitary-adrenal axis of male rats. Developmental Brain Research, 105(2), 295–307. [CrossRef]
- McCracken, J. T., Anagnostou, E., Arango, C., Dawson, G., Farchione, T., Mantua, V., et al. (2021). Drug development for Autism Spectrum Disorder (ASD): Progress, challenges, and future directions. Opportunities & Challenges in Drug Development for ASD: Product of ISCTM/ECNP Joint Working Group, 48, 3–31. [CrossRef]
- McCruden, A. B., & Stimson, W. H. (1991). Sex Hormones and Immune Function. In R. ADER, D. L. FELTEN, & N. COHEN (Eds.), Psychoneuroimmunology (pp. 475–493). Academic Press. [CrossRef]
- McEwen, B. S. (1998a). Protective and damaging effects of stress mediators. New England Journal of Medicine, 338(3), 171–179. [CrossRef]
- McEwen, B. S. (1998b). Stress, adaptation, and disease: Allostasis and allostatic load. Annals of the New York academy of sciences, 840(1), 33–44. [CrossRef]
- Mead, J., & Ashwood, P. (2015). Evidence supporting an altered immune response in ASD. Immunology Letters, 163(1), 49–55. [CrossRef]
- Megremi, A. S. (2013). Is fever a predictive factor in the autism spectrum disorders? Medical Hypotheses, 80(4), 391–398. [CrossRef]
- Mezzacappa, A., Lasica, P.-A., Gianfagna, F., Cazas, O., Hardy, P., Falissard, B., et al. (2017). Risk for Autism Spectrum Disorders According to Period of Prenatal Antidepressant Exposure: A Systematic Review and Meta-analysis. JAMA Pediatrics, 171(6), 555–563. [CrossRef]
- Miclotte, L., & Van de Wiele, T. (2020). Food processing, gut microbiota and the globesity problem. Critical Reviews in Food Science & Nutrition, 60(11), 1769–1782. [CrossRef]
- Miller, H., Zhang, J., Kuolee, R., Patel, G., & Chen, W. (2007). Intestinal M cells: The fallible sentinels? World Journal of Gastroenterology : WJG, 13, 1477–86.
- Mills, E. L., Kelly, B., & O’Neill, L. A. (2017). Mitochondria are the powerhouses of immunity. Nature Immunology, 18(5), 488–498. [CrossRef]
- Misquita, A. G., da Silva, C. N. F., Souza, D. R. O., dos Santos, D. N. B., Brito, I. de S. M., Ribeiro, J. de O. S., et al. (2024). Cognitive behavioral therapy: A valuable intervention in the autistic universe. International Seven Journal of Health Research, 3(2), 754–760. [CrossRef]
- Mitsea, E., Drigas, A., & Skianis, C. (2022). Cutting-Edge Technologies in Breathwork for Learning Disabilities in Special Education. Technium Social Sciences Journal, 34, 136–157. [CrossRef]
- Mitsea, E., Drigas, A., & Skianis, C. (2022). Cutting-Edge Technologies in Breathwork for Learning Disabilities in Special Education. Technium Social Sciences Journal, 34, 136–157. [CrossRef]
- Nordin, A. M., Ismail, J., & Nor, N. K. (2021). Motor development in children with autism spectrum disorder. Frontiers in Pediatrics, 9, 598276. [CrossRef]
- Molloy, C. A., Morrow, A. L., Meinzen-Derr, J., Schleifer, K., Dienger, K., Manning-Courtney, P., et al. (2006). Elevated cytokine levels in children with autism spectrum disorder. Journal of Neuroimmunology, 172(1), 198–205. [CrossRef]
- Moncrieff, J. (2006). Why is it so difficult to stop psychiatric drug treatment? It may be nothing to do with the original problem. Medical Hypotheses, 67(3), 517–523. [CrossRef]
- Moncrieff, J., Cohen, D., & Porter, S. (2013). The Psychoactive Effects of Psychiatric Medication: The Elephant in the Room. Journal of Psychoactive Drugs, 45(5), 409–415. [CrossRef]
- Money, J., Bobrow, N. A., & Clarke, F. C. (1971). Autism and autoimmune disease: A family study. Journal of Autism & Childhood Schizophrenia, 1(2), 146–160. [CrossRef]
- Monteiro, M., Santos, A., Gomes, L., & Rito, R. (2020). Autism Spectrum Disorder: A Systematic Review About Nutritional Interventions. Revista Paulista de Pediatria, 38. [CrossRef]
- Montgomery, S. L., & Bowers, W. J. (2012). Tumor Necrosis Factor-alpha and the Roles it Plays in Homeostatic and Degenerative Processes Within the Central Nervous System. Journal of Neuroimmune Pharmacology, 7(1), 42–59. [CrossRef]
- Moore, J. B., & Weeks, M. E. (2011). Proteomics and systems biology: current and future applications in the nutritional sciences. Advances in Nutrition, 2(4), 355–364. [CrossRef]
- Mordelt, A., & de Witte, L. D. (2023). Microglia-mediated synaptic pruning as a key deficit in neurodevelopmental disorders: Hype or hope? Current Opinion in Neurobiology, 79, 102674. [CrossRef]
- Gianluca, M., Giovanni, C., Devis, B., & Armando., G. (2017). Biologics in Inflammatory Immune-mediated Systemic Diseases. Current Pharmaceutical Biotechnology, 18(12), 1008–1016. [CrossRef]
- Moroni, L., Bianchi, I., & Lleo, A. (2012). Geoepidemiology, gender and autoimmune disease. Special Issue on Gender, Sex Hormones, Pregnancy & Autoimmunity, 11(6), A386–A392. [CrossRef]
- Mosconi, M. W., Wang, Z., Schmitt, L. M., Tsai, P., & Sweeney, J. A. (2015). The role of cerebellar circuitry alterations in the pathophysiology of autism spectrum disorders. Frontiers in Neuroscience, 9. [CrossRef]
- Mulder, S. J., & Mulder-Bos, G. C. (2006). Most probable origin of coeliac disease is low immune globulin A in the intestine caused by malfunction of Peyer’s patches. Medical Hypotheses, 66(4), 757–762. [CrossRef]
- Nadeem, A., Ahmad, S. F., Al-Harbi, N. O., Al-Ayadhi, L. Y., Sarawi, W., Attia, S. M., et al. (2022). Imbalance in pro-inflammatory and anti-inflammatory cytokines milieu in B cells of children with autism. Molecular Immunology, 141, 297–304. [CrossRef]
- Nelissen, S., Lemmens, E., Geurts, N., Kramer, P., Maurer, M., Hendriks, J., & Hendrix, S. (2013). The role of mast cells in neuroinflammation. Acta Neuropathologica, 125, 637–650. [CrossRef]
- Nelson, A. D., & Bender, K. J. (2021). Dendritic Integration Dysfunction in Neurodevelopmental Disorders. Developmental Neuroscience, 43(3–4), 201–221. [CrossRef]
- Netea, M. G., Joosten, L. A., Latz, E., Mills, K. H., Natoli, G., Stunnenberg, H. G., et al. (2016). Trained immunity: a program of innate immune memory in health and disease. Science, 352(6284), aaf1098. [CrossRef]
- evison, C., Blaxill, M., & Zahorodny, W. (2018). California autism prevalence trends from 1931 to 2014 and comparison to national ASD data from IDEA and ADDM. Journal of Autism & Developmental Disorders, 48, 4103–4117. [CrossRef]
- Nevison, C. D., & Blaxill, M. (2017). Diagnostic substitution for intellectual disability: A flawed explanation for the rise in autism. Journal of Autism & Developmental Disorders, 47, 2733–2742. [CrossRef]
- Ngo, S. T., Steyn, F. J., & McCombe, P. A. (2014). Gender differences in autoimmune disease. Sex Differences in Neurological & Psychiatric Disorders, 35(3), 347–369. [CrossRef]
- Niciu, M. J., Kelmendi, B., & Sanacora, G. (2012). Overview of glutamatergic neurotransmission in the nervous system. Glutamate Receptors, 100(4), 656–664. [CrossRef]
- Nie, Z.-Q., Han, D., Zhang, K., Li, M., Kwon, H.-K., Im, S.-H., et al. (2023). TH1/Treg ratio may be a marker of autism in children with immune dysfunction. Research in Autism Spectrum Disorders, 101, 102085. [CrossRef]
- NIEHS. (2024). Autoimmune Diseases. https://www.niehs.nih.gov/health/topics/conditions/autoimmune. Accessed 1 May 2024.
- Niesler, B., Kuerten, S., Demir, I. E., & Schäfer, K.-H. (2021). Disorders of the enteric nervous system—a holistic view. Nature reviews Gastroenterology & Hepatology, 18(6), 393–410. [CrossRef]
- NIMH. (2024). Autism Spectrum Disorder. https://www.nimh.nih.gov/health/topics/autism-spectrum-disorders-asd. Accessed 16 July 2024.
- Niyozovh, S., Djurabekova, A., & Igamova, S. (2020). Complication of encephalitis in children, an innovative approach for the treatment. International Journal of Pharmaceutical Research (09752366), 12(1). [CrossRef]
- Njotto, L. L., Simin, J., Fornes, R., Odsbu, I., Mussche, I., Callens, S., et al. (2023). Maternal and early-life exposure to antibiotics and the risk of autism and Attention-Deficit Hyperactivity Disorder in childhood: A Swedish population-based cohort study. Drug Safety, 46(5), 467–478. [CrossRef]
- Nyati, K. K., & Prasad, K. N. (2014). Role of Cytokines and Toll-Like Receptors in the Immunopathogenesis of Guillain-Barré Syndrome. Mediators of Inflammation, 2014(1), 758639. [CrossRef]
- Obata, Y., & Pachnis, V. (2016). The effect of microbiota and the immune system on the development and organization of the enteric nervous system. Gastroenterology, 151(5), 836–844. [CrossRef]
- Odenwald, M. A., & Turner, J. R. (2017). The intestinal epithelial barrier: a therapeutic target? Nature Reviews Gastroenterology & Hepatology, 14(1), 9–21. [CrossRef]
- Onore, C., Careaga, M., & Ashwood, P. (2012). The role of immune dysfunction in the pathophysiology of autism. Brain, Behavior, & Immunity, 26(3), 383–392. [CrossRef]
- Ozonoff, S., Iosif, A.-M., Baguio, F., Cook, I. C., Hill, M. M., Hutman, T., et al. (2010). A prospective study of the emergence of early behavioral signs of autism. Journal of the American Academy of Child & Adolescent Psychiatry, 49(3), 256–266. [CrossRef]
- Padmakumar, M., Van Raes, E., Van Geet, C., & Freson, K. (2019). Blood platelet research in autism spectrum disorders: In search of biomarkers. Research & Practice in Thrombosis & Haemostasis, 3(4), 566–577. [CrossRef]
- Palmieri, L., & Persico, A. M. (2010). Mitochondrial dysfunction in autism spectrum disorders: cause or effect? Biochimica et Biophysica Acta (BBA)-Bioenergetics, 1797(6–7), 1130–1137. [CrossRef]
- Panksepp, J. (1979). A neurochemical theory of autism. Trends in Neurosciences, 2, 174–177. [CrossRef]
- Paoletti, P., Bellone, C., & Zhou, Q. (2013). NMDA receptor subunit diversity: impact on receptor properties, synaptic plasticity and disease. Nature Reviews Neuroscience, 14(6), 383–400. [CrossRef]
- Pardo, C. A., & Eberhart, C. G. (2007). The neurobiology of autism. Brain Pathology, 17(4), 434–447. [CrossRef]
- Pardo, C. A., Vargas, D. L., & Zimmerman, A. W. (2005). Immunity, neuroglia and neuroinflammation in autism. International Review of Psychiatry, 17(6), 485–495. [CrossRef]
- Park, K. M., & Bowers, W. J. (2010). Tumor necrosis factor-alpha mediated signaling in neuronal homeostasis and dysfunction. Cellular Signalling, 22(7), 977–983. [CrossRef]
- Pathak, D., & Sriram, K. (2023). Neuron-astrocyte omnidirectional signaling in neurological health and disease. Frontiers in Molecular Neuroscience, 16, 1169320. [CrossRef]
- Peper, E., & Shufor, J. (2024). Reflections on the Increase in Autism, ADHD, Anxiety, and Depression: Part 2 – Exposure to Neurotoxins and Ultraprocessed Foods. NeuroRegulation, 11, 219–228. [CrossRef]
- Peralta-Marzal, L. N., Rojas-Velazquez, D., Rigters, D., Prince, N., Garssen, J., Kraneveld, A. D., et al. (2024). A robust microbiome signature for autism spectrum disorder across different studies using machine learning. Scientific Reports, 14(1), 814. [CrossRef]
- Pereira, A. (2020). Chapter Seven - Classical-quantum interfaces in living neural tissue supporting conscious functions. In R. R. Poznański & E. J. Brändas (Eds.), Advances in Quantum Chemistry (Vol. 82, pp. 213–252). Academic Press. [CrossRef]
- Pereira, A., & Furlan, F. A. (2009). On the role of synchrony for neuron–astrocyte interactions and perceptual conscious processing. Journal of Biological Physics, 35, 465–480. [CrossRef]
- Pereira, A., & Furlan, F. A. (2010). Astrocytes and human cognition: Modeling information integration and modulation of neuronal activity. Progress in Neurobiology, 92(3), 405–420. [CrossRef]
- Pereira, J. A. (2020). The Role of Sentience in the Theory of Consciousness and Medical Practice. [CrossRef]
- Pereira, J. A., & Wang, F. (2016). Neuromodulation, Emotional Feelings and Affective Disorders. Mens Sana Monographs, 0. [CrossRef]
- Perez-Muñoz, M. E., Arrieta, M.-C., Ramer-Tait, A. E., & Walter, J. (2017). A critical assessment of the “sterile womb” and “in utero colonization” hypotheses: implications for research on the pioneer infant microbiome. Microbiome, 5(1), 48. [CrossRef]
- Perrin, J. M., Anderson, L. E., & Van Cleave, J. (2014). The rise in chronic conditions among infants, children, and youth can be met with continued health system innovations. Health Affairs, 33(12), 2099–2105. [CrossRef]
- Petrelli, F., Pucci, L., & Bezzi, P. (2016). Astrocytes and microglia and their potential link with autism spectrum disorders. Frontiers in Cellular Neuroscience, 10, 21. [CrossRef]
- Ploog, B. O. (2023). Theory of Mind in Autism. In A. El Idrissi & D. McCloskey (Eds.), Neurobiology of Autism Spectrum Disorders (pp. 23–35). Cham: Springer International Publishing. [CrossRef]
- Pociūtė, A., Pivoriūnas, A., & Verkhratsky, A. (2024). Astrocytes dynamically regulate the blood-brain barrier in the healthy brain. Neural Regeneration Research, 19(4), 709–710.
- Politte, L. C., Howe, Y., Nowinski, L., Palumbo, M., & McDougle, C. J. (2015). Evidence-Based Treatments for Autism Spectrum Disorder. Current Treatment Options in Psychiatry, 2(1), 38–56. [CrossRef]
- Pourgholaminejad, A., & Tahmasebinia, F. (2019). The Role of Th17 Cells in Immunopathogenesis of Neuroinflammatory Disorders. In H. Mitoma & M. Manto (Eds.), Neuroimmune Diseases: From Cells to the Living Brain (pp. 83–107). Cham: Springer International Publishing. [CrossRef]
- Pretzsch, C. M., Schäfer, T., Lombardo, M. V., Warrier, V., Mann, C., Bletsch, A., et al. (2022). Neurobiological Correlates of Change in Adaptive Behavior in Autism. American Journal of Psychiatry, 179(5), 336–349. [CrossRef]
- Prüss, H. (2021). Autoantibodies in neurological disease. Nature Reviews Immunology, 21(12), 798–813. https://doi.org/10.1038/s41577-021-00543-w. [CrossRef]
- Pu, Y., Yang, J., Chang, L., Qu, Y., Wang, S., Zhang, K., et al. (2020). Maternal glyphosate exposure causes autism-like behaviors in offspring through increased expression of soluble epoxide hydrolase. Proceedings of the National Academy of Sciences, 117(21), 11753–11759. [CrossRef]
- Pulikkan, J., Mazumder, A., & Grace, T. (2019). Role of the gut microbiome in autism spectrum disorders. Reviews on Biomarker Studies in Psychiatric & Neurodegenerative disorders, 253–269. [CrossRef]
- Puricelli, C., Rolla, R., Gigliotti, L., Boggio, E., Beltrami, E., Dianzani, U., & Keller, R. (2022). The Gut-Brain-Immune Axis in Autism Spectrum Disorders: A State-of-Art Report. Frontiers in Psychiatry, 12. [CrossRef]
- Qi, H. (2023). Neuroimmunology: reviews and perspectives on recent advances. Cellular & Molecular Immunology, 20(11), 1257–1258. [CrossRef]
- Rabinovici, G. D., Stephens, M. L., & Possin, K. L. (2015). Executive Dysfunction. CONTINUUM: Lifelong Learning in Neurology, 21(3). [CrossRef]
- Ramautar, R., Berger, R., van der Greef, J., & Hankemeier, T. (2013). Human metabolomics: strategies to understand biology. Current Opinion in Chemical Biology, 17(5), 841–846. [CrossRef]
- Ramsay, D. S., & Woods, S. C. (2014). Clarifying the roles of homeostasis and allostasis in physiological regulation. Psychological Review, 121(2), 225–247. [CrossRef]
- Randolph-Gips, M., & Srinivasan, P. (2012). Modeling autism: a systems biology approach. Journal of Clinical Bioinformatics, 2(1), 17. [CrossRef]
- Reemst, K., Noctor, S. C., Lucassen, P. J., & Hol, E. M. (2016). The indispensable roles of microglia and astrocytes during brain development. Frontiers in Human Neuroscience, 10, 566. [CrossRef]
- Reid, G., Younes, J. A., Van der Mei, H. C., Gloor, G. B., Knight, R., & Busscher, H. J. (2011). Microbiota restoration: natural and supplemented recovery of human microbial communities. Nature Reviews Microbiology, 9(1), 27–38. [CrossRef]
- Renz, H., Holt, P. G., Inouye, M., Logan, A. C., Prescott, S. L., & Sly, P. D. (2017). An exposome perspective: Early-life events and immune development in a changing world. Journal of Allergy & Clinical Immunology, 140(1), 24–40. [CrossRef]
- Réthelyi, J. M., Vincze, K., Schall, D., Glennon, J., & Berkel, S. (2023). The role of insulin/IGF1 signalling in neurodevelopmental and neuropsychiatric disorders–evidence from human neuronal cell models. Neuroscience & Biobehavioral Reviews, 105330. [CrossRef]
- Ribatti, D. (2015). The crucial role of mast cells in blood–brain barrier alterations. Experimental Cell Research, 338(1), 119–125. [CrossRef]
- Ribeiro, S. (2018). Whole Organisms or Pure Compounds? Entourage Effect Versus Drug Specificity. In B. C. Labate & C. Cavnar (Eds.), Plant Medicines, Healing & Psychedelic Science: Cultural Perspectives (pp. 133–149). Cham: Springer International Publishing. [CrossRef]
- Riikonen, R. (2017). Insulin-like growth factors in the pathogenesis of neurological diseases in children. International Journal of Molecular Sciences, 18(10), 2056. [CrossRef]
- Robertson, J. M. (2002). The Astrocentric Hypothesis: proposed role of astrocytes in consciousness and memory formation. Journal of Physiology-Paris, 96(3), 251–255. [CrossRef]
- Rogers, S. J., Cook, I., & Meryl, A. (2005). Imitation and play in autism. In Handbook of Autism a& Pervasive Developmental Disorders (Vol. 1, pp. 382–405). Wiley Online Library.
- Romagnani, S. (1999). Th1/Th2 Cells. Inflammatory Bowel Diseases, 5(4), 285–294. [CrossRef]
- Rook, G. A. W. (2012). Hygiene Hypothesis and Autoimmune Diseases. Clinical Reviews in Allergy & Immunology, 42(1), 5–15. [CrossRef]
- Rosen, N. E., Lord, C., & Volkmar, F. R. (2021). The diagnosis of autism: From Kanner to DSM-III to DSM-5 and beyond. Journal of Autism & Developmental Disorders, 51, 4253–4270. [CrossRef]
- Rosin, J. M., & Kurrasch, D. M. (2019). Emerging roles for hypothalamic microglia as regulators of physiological homeostasis. Frontiers in Neuroendocrinology, 54, 100748. [CrossRef]
- Rossignol, D A., & Frye, R. E. (2012). A review of research trends in physiological abnormalities in autism spectrum disorders: immune dysregulation, inflammation, oxidative stress, mitochondrial dysfunction and environmental toxicant exposures. Molecular Psychiatry, 17(4), 389–401. [CrossRef]
- Rossignol, D. A., & Frye, R. E. (2014). Evidence linking oxidative stress, mitochondrial dysfunction, and inflammation in the brain of individuals with autism. Frontiers in Physiology, 5, 150. [CrossRef]
- Rucklidge, J. J., Johnstone, J. M., & Kaplan, B. J. (2021). Nutrition Provides the Essential Foundation for Optimizing Mental Health. Evidence-Based Practice in Child & Adolescent Mental Health, 6(1), 131–154. [CrossRef]
- Rueda-Ruzafa, L., Cruz, F., Roman, P., & Cardona, D. (2019). Gut microbiota and neurological effects of glyphosate. NeuroToxicology, 75, 1–8. [CrossRef]
- Saad, K., Zahran, A. M., Elsayh, K. I., Abdel-Rahman, A. A., Al-Atram, A. A., Hussein, A., & El-Gendy, Y. G. (2017). Frequency of Dendritic Cells and Their Expression of Costimulatory Molecules in Children with Autism Spectrum Disorders. Journal of Autism & Developmental Disorders, 47(9), 2671–2678. [CrossRef]
- Safari-Alighiarloo, N., Taghizadeh, M., Rezaei tavirani, M., Goliaei, B., & Peyvandi, A. (2014). Protein-Protein Interaction Networks (PPI) and complex diseases. Gastroenterology & Hepatology From Bed to Bench, 7, 17–31.
- Salmond, C., De Haan, M., Friston, K., Gadian, D., & Vargha-Khadem, F. (2003). Investigating individual differences in brain abnormalities in autism. Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences, 358(1430), 405–413. [CrossRef]
- Salter, M. W., & Stevens, B. (2017). Microglia emerge as central players in brain disease. Nature Medicine, 23(9), 1018–1027. [CrossRef]
- Samanipour, S., Barron, L. P., van Herwerden, D., Praetorius, A., Thomas, K. V., & O’Brien, J. W. (2024). Exploring the Chemical Space of the Exposome: How Far Have We Gone? JACS Au, 4(7), 2412–2425. [CrossRef]
- Samsam, M., Ahangari, R., & Naser, S. A. (2014). Pathophysiology of autism spectrum disorders: revisiting gastrointestinal involvement and immune imbalance. World journal of gastroenterology: WJG, 20(29), 9942.
- Samsel, A., & Seneff, S. (2013). Glyphosate’s suppression of cytochrome P450 enzymes and amino acid biosynthesis by the gut microbiome: pathways to modern diseases. Entropy, 15(4), 1416–1463. [CrossRef]
- Saresella, M., Piancone, F., Marventano, I., Zoppis, M., Hernis, A., Zanette, M., et al. (2016). Multiple inflammasome complexes are activated in autistic spectrum disorders. Brain, Behavior, & Immunity, 57, 125–133. [CrossRef]
- Schroder, K., & Tschopp, J. (2010). The Inflammasomes. Cell, 140(6), 821–832. [CrossRef]
- Schwartz, M., & Cahalon, L. (2022). The vicious cycle governing the brain–immune system relationship in neurodegenerative diseases. Current Opinion in Immunology, 76, 102182. [CrossRef]
- Segal, Y., & Shoenfeld, Y. (2018). Vaccine-induced autoimmunity: the role of molecular mimicry and immune crossreaction. Cellular & Molecular Immunology, 15(6), 586–594. [CrossRef]
- Seneff, S., Kyriakopoulos, A. M., & Nigh, G. (2024). Is autism a PIN1 deficiency syndrome? A proposed etiological role for glyphosate. Journal of Neurochemistry, n/a(n/a). [CrossRef]
- Serrano-Pozo, A. (2018). Encephalopathy. In The Wiley Handbook on the Aging Mind and Brain (pp. 553–590). [CrossRef]
- Shah, P. A., Park, C. J., Shaughnessy, M. P., & Cowles, R. A. (2021). Serotonin as a Mitogen in the Gastrointestinal Tract: Revisiting a Familiar Molecule in a New Role. Cellular & Molecular Gastroenterology & Hepatology, 12(3), 1093–1104. [CrossRef]
- Shahar, O., Botvinnik, A., Shwartz, A., Lerer, E., Golding, P., Buko, A., et al. (2024). Effect of chemically synthesized psilocybin and psychedelic mushroom extract on molecular and metabolic profiles in mouse brain. Molecular Psychiatry. [CrossRef]
- Shahbazi, R., Sharifzad, F., Bagheri, R., Alsadi, N., Yasavoli-Sharahi, H., & Matar, C. (2021). Anti-Inflammatory and Immunomodulatory Properties of Fermented Plant Foods. Nutrients, 13(5). [CrossRef]
- Shajib, Md. S., Baranov, A., & Khan, W. I. (2017). Diverse Effects of Gut-Derived Serotonin in Intestinal Inflammation. ACS Chemical Neuroscience, 8(5), 920–931. [CrossRef]
- Shaw, C. A., Seneff, S., Kette, S. D., Tomljenovic, L., Oller Jr, J. W., & Davidson, R. M. (2014). Aluminum-Induced Entropy in Biological Systems: Implications for Neurological Disease. Journal of Toxicology, 2014(1), 491316. [CrossRef]
- Shaw, J. C., Crombie, G. K., Palliser, H. K., & Hirst, J. J. (2021). Impaired oligodendrocyte development following preterm birth: promoting GABAergic action to improve outcomes. Frontiers in Pediatrics, 9, 618052. [CrossRef]
- Shin, Y.-W., Lee, S.-T., Park, K.-I., Jung, K.-H., Jung, K.-Y., Lee, S. K., & Chu, K. (2018). Treatment strategies for autoimmune encephalitis. Therapeutic Advances in Neurological Disorders, 11, 1756285617722347. [CrossRef]
- Siegel, M., Milligan, B., Chemelski, B., Payne, D., Ellsworth, B., Harmon, J., et al. (2014). Specialized inpatient psychiatry for serious behavioral disturbance in autism and intellectual disability. Journal of Autism & Developmental Disorders, 44, 3026–3032. [CrossRef]
- Sies, H. (2015). Oxidative stress: a concept in redox biology and medicine. Redox Biology, 4, 180–183. [CrossRef]
- Gonzalez, S. M., Ni, G., Lam, V., & Demopoulos, C. (2024). Beyond words: an investigation of fine motor skills and the verbal communication spectrum in autism. Frontiers in Psychiatry, 15, 1379307. [CrossRef]
- Simons, M., Levin, J., & Dichgans, M. (2023). Tipping points in neurodegeneration. Neuron, 111. [CrossRef]
- Singh, A., Kukreti, R., Saso, L., & Kukreti, S. (2019). Oxidative Stress: A Key Modulator in Neurodegenerative Diseases. Molecules, 24(8). [CrossRef]
- Siniscalco, D., Bradstreet, J. J., & Antonucci, N. (2013). Therapeutic Role of Hematopoietic Stem Cells in Autism Spectrum Disorder-Related Inflammation. Frontiers in Immunology, 4. [CrossRef]
- Sloan, S. A., & Barres, B. A. (2014). Mechanisms of astrocyte development and their contributions to neurodevelopmental disorders. Current Opinion in Neurobiology, 27, 75–81. [CrossRef]
- Smyth, M. J., Godfrey, D. I., & Trapani, J. A. (2001). A fresh look at tumor immunosurveillance and immunotherapy. Nature Immunology, 2(4), 293–299. [CrossRef]
- Ssucharewa, G. E. (1926). Die schizoiden Psychopathien im Kindesalter. (Part 1 of 2). European Neurology, 60(3–4), 235–247. [CrossRef]
- Stein, Y., Hänninen, O., Huttunen, P., Ahonen, M., & Ekman, R. (2015). Electromagnetic Radiation and Health: Human Indicators. In R. H. Armon & O. Hänninen (Eds.), Environmental Indicators (pp. 1025–1046). Dordrecht: Springer Netherlands. [CrossRef]
- Steinman, G. (2015). Plausible etiology of brain dysconnectivity in autism – Review and prospectus. Medical Hypotheses, 85(4), 405–407. [CrossRef]
- Steinman, G., & Mankuta, D. (2013). Insulin-like growth factor and the etiology of autism. Medical Hypotheses, 80(4), 475–480. [CrossRef]
- Stilling, R. M., Bordenstein, S. R., Dinan, T. G., & Cryan, J. F. (2014). Friends with social benefits: host-microbe interactions as a driver of brain evolution and development? Frontiers in Cellular & Infection Microbiology, 4. [CrossRef]
- Stinson, L. F., Boyce, M. C., Payne, M. S., & Keelan, J. A. (2019). The Not-so-Sterile Womb: Evidence That the Human Fetus Is Exposed to Bacteria Prior to Birth. Frontiers in Microbiology, 10. [CrossRef]
- Stinson, L. F., Payne, M. S., & Keelan, J. A. (2017). Planting the seed: Origins, composition, and postnatal health significance of the fetal gastrointestinal microbiota. Critical Reviews in Microbiology, 43(3), 352–369. [CrossRef]
- Storch, E. A., Arnold, E. B., Lewin, A. B., Nadeau, J. M., Jones, A. M., De Nadai, A. S., et al. (2013). The Effect of Cognitive-Behavioral Therapy Versus Treatment as Usual for Anxiety in Children With Autism Spectrum Disorders: A Randomized, Controlled Trial. Journal of the American Academy of Child & Adolescent Psychiatry, 52(2), 132-142.e2. [CrossRef]
- Strandwitz, P. (2018). Neurotransmitter modulation by the gut microbiota. Where the gut meets the brain, 1693, 128–133. [CrossRef]
- Strati, F., Cavalieri, D., Albanese, D., De Felice, C., Donati, C., Hayek, J., et al. (2017). New evidences on the altered gut microbiota in autism spectrum disorders. Microbiome, 5, 1–11. [CrossRef]
- Streng, M. L., & Krook-Magnuson, E. (2021). The cerebellum and epilepsy. Epilepsy & Behavior, 121. [CrossRef]
- Stubbs, E. G., Crawford, M. L., Burger, D. R., & Vandenbark, A. A. (1977). Depressed lymphocyte responsiveness in autistic children. Journal of Autism & Childhood Schizophrenia, 7(1), 49–55. [CrossRef]
- Su, L.-D., Xu, F.-X., Wang, X.-T., Cai, X.-Y., & Shen, Y. (2021). Cerebellar Dysfunction, Cerebro-cerebellar Connectivity and Autism Spectrum Disorders. Neuroscience, 462, 320–327. [CrossRef]
- Sweeney, M. D., Zhao, Z., Montagne, A., Nelson, A. R., & Zlokovic, B. V. (2019). Blood-Brain Barrier: From Physiology to Disease and Back. Physiological Reviews, 99(1), 21–78. [CrossRef]
- Sweeten, T. L., Posey, D. J., Shekhar, A., & McDougle, C. J. (2002). The amygdala and related structures in the pathophysiology of autism. Functional Role of Specific Systems Within the Extended Amygdala & Hypothalamus, 71(3), 449–455. [CrossRef]
- Tahmasebinia, F., & Pourgholaminejad, A. (2017). The role of Th17 cells in auto-inflammatory neurological disorders. Progress in Neuro-Psychopharmacology & Biological Psychiatry, 79, 408–416. [CrossRef]
- Taniya, M. A., Chung, H.-J., Al Mamun, A., Alam, S., Aziz, M. A., Emon, N. U., et al. (2022). Role of gut microbiome in autism spectrum disorder and its therapeutic regulation. Frontiers in Cellular & Infection Microbiology, 12, 915701. [CrossRef]
- Tarabeux, J., Kebir, O., Gauthier, J., Hamdan, F. F., Xiong, L., Piton, A., et al. (2011). Rare mutations in N-methyl-D-aspartate glutamate receptors in autism spectrum disorders and schizophrenia. Translational Psychiatry, 1(11), e55–e55. [CrossRef]
- Tartaglione, A. M., Camoni, L., Calamandrei, G., Chiarotti, F., & Venerosi, A. (2024). The contribution of environmental pollutants to the risk of autism and other neurodevelopmental disorders: A systematic review of case-control studies. Neuroscience & Biobehavioral Reviews, 164, 105815. [CrossRef]
- Tarnowska, K., Gruczyńska–Sękowska, E., Kowalska, D., Majewska, E., Kozłowska, M., & Winkler, R. (2023). The opioid excess theory in autism spectrum disorders - is it worth investigating further? Critical Reviews in Food Science & Nutrition, 63(19), 3980–3993. [CrossRef]
- Terrabuio, E., Zenaro, E., & Constantin, G. (2023). The role of the CD8+ T cell compartment in ageing and neurodegenerative disorders. Frontiers in Immunology, 14, 1233870. [CrossRef]
- Theofilopoulos, A. N., Kono, D. H., & Baccala, R. (2017). The multiple pathways to autoimmunity. Nature Immunology, 18(7), 716–724. [CrossRef]
- Theoharides, T. C., Angelidou, A., Alysandratos, K.-D., Zhang, B., Asadi, S., Francis, K., et al. (2012). Mast cell activation and autism. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 1822(1), 34–41. [CrossRef]
- Theoharides, T. C., & Doyle, R. (2008). Autism, gut-blood-brain barrier, and mast cells. Journal of Clinical Psychopharmacology, 28(5), 479–483. [CrossRef]
- Theoharides, T. C., Kavalioti, M., & Tsilioni, I. (2019). Mast cells, stress, fear and autism spectrum disorder. International Journal of Molecular Sciences, 20(15), 3611. [CrossRef]
- Theoharides, T. C., Stewart, J. M., Panagiotidou, S., & Melamed, I. (2016). Mast cells, brain inflammation and autism. European Journal of Pharmacology, 778, 96–102. [CrossRef]
- Theoharides, T., Tsilioni, I., Patel, A., & Doyle, R. (2016). Atopic diseases and inflammation of the brain in the pathogenesis of autism spectrum disorders. Translational Psychiatry, 6(6), e844–e844. [CrossRef]
- Tillmann, T., Gibson, A. R., Scott, G., Harrison, O., Dominiczak, A., & Hanlon, P. (2015). Systems Medicine 2.0: Potential Benefits of Combining Electronic Health Care Records with Systems Science Models. J Med Internet Res, 17(3), e64. [CrossRef]
- Toledano, M. P., Sean J. (2015). Autoimmune Epilepsy. Seminars in Neurology, 35(03), 245–258. [CrossRef]
- Tomova, A., Husarova, V., Lakatosova, S., Bakos, J., Vlkova, B., Babinska, K., & Ostatnikova, D. (2015). Gastrointestinal microbiota in children with autism in Slovakia. Physiology & Behavior, 138, 179–187. [CrossRef]
- Toomey, R., Kang, H. K., Karlinsky, J., Baker, D. G., Vasterling, J. J., Alpern, R., et al. (2007). Mental health of US Gulf War veterans 10 years after the war. British Journal of Psychiatry, 190(5), 385–393. [CrossRef]
- Tordjman, S., Celume, M. P., Denis, L., Motillon, T., & Keromnes, G. (2019). Reframing schizophrenia and autism as bodily self-consciousness disorders leading to a deficit of theory of mind and empathy with social communication impairments. Neuroscience & Biobehavioral Reviews, 103, 401–413. [CrossRef]
- Tordjman, S., Somogyi, E., Coulon, N., Kermarrec, S., Cohen, D., Bronsard, G., et al. (2014). Gene× Environment interactions in autism spectrum disorders: role of epigenetic mechanisms. Frontiers in Psychiatry, 5, 53. [CrossRef]
- Torres, E. B., & Denisova, K. (2016). Motor noise is rich signal in autism research and pharmacological treatments. Scientific Reports, 6(1), 37422. [CrossRef]
- Tuchman, R., Moshé, S. L., & Rapin, I. (2009). Convulsing toward the pathophysiology of autism. Brain & Development, 31(2), 95–103. [CrossRef]
- Tzang, R.-F., Chang, C.-H., Chang, Y.-C., & Lane, H.-Y. (2019). Autism Associated With Anti-NMDAR Encephalitis: Glutamate-Related Therapy. Frontiers in Psychiatry, 10. [CrossRef]
- Üçok, A., & Gaebel, W. (2008). Side effects of atypical antipsychotics: a brief overview. World Psychiatry, 7(1), 58–62. [CrossRef]
- Ullah, F., & Kaelber, D. C. (2021). Using large aggregated de-identified electronic health record data to determine the prevalence of common chronic diseases in pediatric patients who visited primary care clinics. Academic Pediatrics, 21(6), 1084–1093. [CrossRef]
- Usui, N., Kobayashi, H., & Shimada, S. (2023). Neuroinflammation and oxidative stress in the pathogenesis of autism spectrum disorder. International Journal of Molecular Sciences, 24(6), 5487. [CrossRef]
- Vakilzadeh, G., & Martinez-Cerdeño, V. (2023). Pathology and astrocytes in autism. Neuropsychiatric Disease & Treatment, 841–850. [CrossRef]
- van den Brink, W., van Bilsen, J., Salic, K., Hoevenaars, F. P., Verschuren, L., Kleemann, R., et al. (2019). Current and future nutritional strategies to modulate inflammatory dynamics in metabolic disorders. Frontiers in Nutrition, 6, 129. [CrossRef]
- van Ommen, B., & Stierum, R. (2002). Nutrigenomics: exploiting systems biology in the nutrition and health arena. Current Opinion in Biotechnology, 13(5), 517–521. [CrossRef]
- Vargas, D. L., Nascimbene, C., Krishnan, C., Zimmerman, A. W., & Pardo, C. A. (2005). Neuroglial activation and neuroinflammation in the brain of patients with autism. Annals of Neurology: Official Journal of the American Neurological Association & the Child Neurology Society, 57(1), 67–81. [CrossRef]
- Venkatasubramanian, G. (2015). Understanding Schizophrenia as a Disorder of Consciousness: Biological Correlates and Translational Implications from Quantum Theory Perspectives. Clinical Psychopharmacology & Neuroscience, 13(1), 36–47. [CrossRef]
- Verkhratsky, A., & Nedergaard, M. (2018). Physiology of astroglia. Physiological Reviews, 98(1), 239–389. [CrossRef]
- Vermeulen, R., Schymanski, E. L., Barabási, A.-L., & Miller, G. W. (2020). The exposome and health: Where chemistry meets biology. Science, 367(6476), 392–396. [CrossRef]
- Vicedo, M. (2024). Moving beyond the search for the first discoverer of autism. Frontiers in Psychiatry, 15, 1266486. [CrossRef]
- Victoria, H. K., & Caldwell, C. (2013). Breathwork in body psychotherapy: Clinical applications. Body, Movement & Dance in Psychotherapy, 8(4), 216–228. [CrossRef]
- Vineis, P., Robinson, O., Chadeau-Hyam, M., Dehghan, A., Mudway, I., & Dagnino, S. (2020). What is new in the exposome? Environment International, 143, 105887. [CrossRef]
- Vivier, E., Raulet, D. H., Moretta, A., Caligiuri, M. A., Zitvogel, L., Lanier, L. L., et al. (2011). Innate or adaptive immunity? The example of natural killer cells. Science, 331(6013), 44–49. [CrossRef]
- Vivier, E., Tomasello, E., Baratin, M., Walzer, T., & Ugolini, S. (2008). Functions of natural killer cells. Nature Immunology, 9(5), 503–510. [CrossRef]
- Vojdani, A., Mumper, E., Granpeesheh, D., Mielke, L., Traver, D., Bock, K., et al. (2008). Low natural killer cell cytotoxic activity in autism: the role of glutathione, IL-2 and IL-15. Journal of Neuroimmunology, 205(1–2), 148–154. [CrossRef]
- Volkmar, F. R., & Nelson, D. S. (1990). Seizure Disorders in Autism. Journal of the American Academy of Child & Adolescent Psychiatry, 29(1), 127–129. [CrossRef]
- Volterra, A., & Meldolesi, J. (2005). Astrocytes, from brain glue to communication elements: the revolution continues. Nature Reviews Neuroscience, 6(8), 626–640. [CrossRef]
- Wakefield, A. (2002). The Gut–Brain Axis in Childhood Developmental Disorders. Journal of Pediatric Gastroenterology & Nutrition, 34 Suppl 1, S14-7. [CrossRef]
- Wakefield, A. J., Puleston, J. M., Montgomery, S. M., Anthony, A., O’Leary, J. J., & Murch, S. H. (2002). The concept of entero-colonic encephalopathy, autism and opioid receptor ligands. Alimentary Pharmacology & Therapeutics, 16(4), 663–674. [CrossRef]
- Walsh, L., Hill, C., & Ross, R. P. (2023). Impact of glyphosate (RoundupTM) on the composition and functionality of the gut microbiome. Gut Microbes, 15(2), 2263935. [CrossRef]
- Wan, M., Ding, L., Wang, D., Han, J., & Gao, P. (2020). Serotonin: A Potent Immune Cell Modulator in Autoimmune Diseases. Frontiers in Immunology, 11. [CrossRef]
- Wang, T., Sternes, P. R., Guo, X.-K., Zhao, H., Xu, C., & Xu, H. (2024). Autoimmune diseases exhibit shared alterations in the gut microbiota. Rheumatology, 63(3), 856–865. [CrossRef]
- Ward, J. H., Weir, E., Allison, C., & Baron-Cohen, S. (2023). Increased rates of chronic physical health conditions across all organ systems in autistic adolescents and adults. Molecular Autism, 14(1), 35. [CrossRef]
- Warren, R. P., Foster, A., & Margaretten, N. C. (1987). Reduced natural killer cell activity in autism. Journal of the American Academy of Child & Adolescent Psychiatry, 26(3), 333–335. [CrossRef]
- Wegiel, J., Chadman, K., London, E., Wisniewski, T., & Wegiel, J. (2024). Contribution of the serotonergic system to developmental brain abnormalities in autism spectrum disorder. Autism Research, 17(7), 1300–1321. [CrossRef]
- Weinberg, S. E., Sena, L. A., & Chandel, N. S. (2015). Mitochondria in the regulation of innate and adaptive immunity. Immunity, 42(3), 406–417.
- Weitlauf, A. S., Gotham, K. O., Vehorn, A. C., & Warren, Z. E. (2014). Brief report: DSM-5 “levels of support:” A comment on discrepant conceptualizations of severity in ASD. Journal of Autism & Developmental Disorders, 44(2), 471–476. [CrossRef]
- Wen, Y., Alshikho, M. J., & Herbert, M. R. (2016). Pathway network analyses for autism reveal multisystem involvement, major overlaps with other diseases and convergence upon MAPK and calcium signaling. PLOS One, 11(4), e0153329. [CrossRef]
- Wen, Y., & Herbert, M. R. (2017). Connecting the dots: Overlaps between autism and cancer suggest possible common mechanisms regarding signaling pathways related to metabolic alterations. Medical Hypotheses, 103, 118–123. [CrossRef]
- Werling, D. M. (2016). The role of sex-differential biology in risk for autism spectrum disorder. Biology of Sex Differences, 7(1), 58. [CrossRef]
- Werner, E., & Dawson, G. (2005). Validation of the phenomenon of autistic regression using home videotapes. Archives of general psychiatry, 62(8), 889–895. [CrossRef]
- West, L., Brunssen, S. H., & Waldrop, J. (2009). Review of the Evidence for Treatment of Children with Autism with Selective Serotonin Reuptake Inhibitors. Journal for Specialists in Pediatric Nursing, 14(3), 183–191. [CrossRef]
- West, L., Waldrop, J., & Brunssen, S. (2009). Pharmacologic Treatment for the Core Deficits and Associated Symptoms of Autism in Children. Journal of Pediatric Health Care, 23(2), 75–89. [CrossRef]
- White, J. F. (2003). Intestinal pathophysiology in autism. Experimental Biology & Medicine, 228(6), 639–649. [CrossRef]
- Whiteley, P., Carr, K., & Shattock, P. (2019). Is autism inborn and lifelong for everyone? Neuropsychiatric disease & Treatment, 2885–2891. [CrossRef]
- Wild, C. P. (2005). Complementing the Genome with an “Exposome”: The Outstanding Challenge of Environmental Exposure Measurement in Molecular Epidemiology. Cancer Epidemiology, Biomarkers & Prevention, 14(8), 1847–1850. [CrossRef]
- Willison, H. J., Jacobs, B. C., & van Doorn, P. A. (2016). Guillain-Barré syndrome. The Lancet, 388(10045), 717–727. [CrossRef]
- Wills, S., Cabanlit, M., Bennett, J., Ashwood, P., Amaral, D. G., & Van de Water, J. (2009). Detection of autoantibodies to neural cells of the cerebellum in the plasma of subjects with autism spectrum disorders. Brain, Behavior, & Immunity, 23(1), 64–74. [CrossRef]
- Witkamp, R. F., & van Norren, K. (2018). Let thy food be thy medicine…. when possible. European Journal of Pharmacology, 836, 102–114. [CrossRef]
- Wood, J. D., Alpers, D. H., & Andrews, P. L. R. (1999). Fundamentals of neurogastroenterology. Gut, 45(suppl 2), II6. [CrossRef]
- Wood, J. J., Kendall, P. C., Wood, K. S., Kerns, C. M., Seltzer, M., Small, B. J., et al. (2020). Cognitive Behavioral Treatments for Anxiety in Children With Autism Spectrum Disorder: A Randomized Clinical Trial. JAMA Psychiatry, 77(5), 474–483. [CrossRef]
- Wu, M., Zheng, W., Song, X., Bao, B., Wang, Y., Ramanan, D., et al. (2024). Gut complement induced by the microbiota combats pathogens and spares commensals. Cell, 187. [CrossRef]
- Xiong, Y., Chen, J., & Li, Y. (2023). Microglia and astrocytes underlie neuroinflammation and synaptic susceptibility in autism spectrum disorder. Frontiers in Neuroscience, 17, 1125428. [CrossRef]
- Xu, G., Snetselaar, L. G., Jing, J., Liu, B., Strathearn, L., & Bao, W. (2018). Association of Food Allergy and Other Allergic Conditions with Autism Spectrum Disorder in Children. JAMA Network Open, 1(2), e180279–e180279. [CrossRef]
- Yavuz, B. R., Arici, M. K., Demirel, H. C., Tsai, C.-J., Jang, H., Nussinov, R., & Tuncbag, N. (2023). Neurodevelopmental disorders and cancer networks share pathways, but differ in mechanisms, signaling strength, and outcome. NPI Genomic Medicine, 8(1), 37. [CrossRef]
- Yeung, A. W. K., Heinrich, M., Kijjoa, A., Tzvetkov, N. T., & Atanasov, A. G. (2020). The ethnopharmacological literature: An analysis of the scientific landscape. Journal of Ethnopharmacology, 250, 112414. [CrossRef]
- Yngve, M., & Lidström, H. (2024). Implementation of information and communication technology to facilitate participation in high school occupations for students with neurodevelopmental disorders. Disability & Rehabilitation: Assistive Technology, 19(5), 2017–2025. [CrossRef]
- Yong, V. W., & Rivest, S. (2009). Taking Advantage of the Systemic Immune System to Cure Brain Diseases. Neuron, 64(1), 55–60. [CrossRef]
- Zanchi, M. M., Marins, K., & Zamoner, A. (2023). Could pesticide exposure be implicated in the high incidence rates of depression, anxiety and suicide in farmers? A systematic review. Environmental Pollution, 331, 121888. [CrossRef]
- Zandman-Goddard, G., Peeva, E., & Shoenfeld, Y. (2007). Gender and autoimmunity. Autoimmunity Reviews, 6(6), 366–372. [CrossRef]
- Zarate-Lopez, D., Torres-Chávez, A. L., Gálvez-Contreras, A. Y., & Gonzalez-Perez, O. (2023). Three decades of valproate: a current model for studying autism spectrum disorder. Current Neuropharmacology, 22(2), 260.
- Zhang, J.-M., & An, J. (2007). Cytokines, inflammation, and pain. International Anesthesiology Clinics, 45(2), 27–37.
- Zhang, S., Han, F., Wang, Q., & Fan, F. (2024). Probiotics and Prebiotics in the Treatment of Autism Spectrum Disorder: A Narrative Review. JIN, 23(1), 20-null. [CrossRef]
- Zhu, J., & Paul, W. E. (2008). CD4 T cells: fates, functions, and faults. Blood, 112(5), 1557–1569. [CrossRef]
- Ziv, Y., & Schwartz, M. (2008). Immune-based regulation of adult neurogenesis: Implications for learning and memory. Brain, Behavior, & Immunity, 22(2), 167–176. [CrossRef]
- Zota, A. R., Riederer, A. M., Ettinger, A. S., Schaider, L. A., Shine, J. P., Amarasiriwardena, C. J., et al. (2016). Associations between metals in residential environmental media and exposure biomarkers over time in infants living near a mining-impacted site. Journal of Exposure Science & Environmental Epidemiology, 26(5), 510–519. [CrossRef]
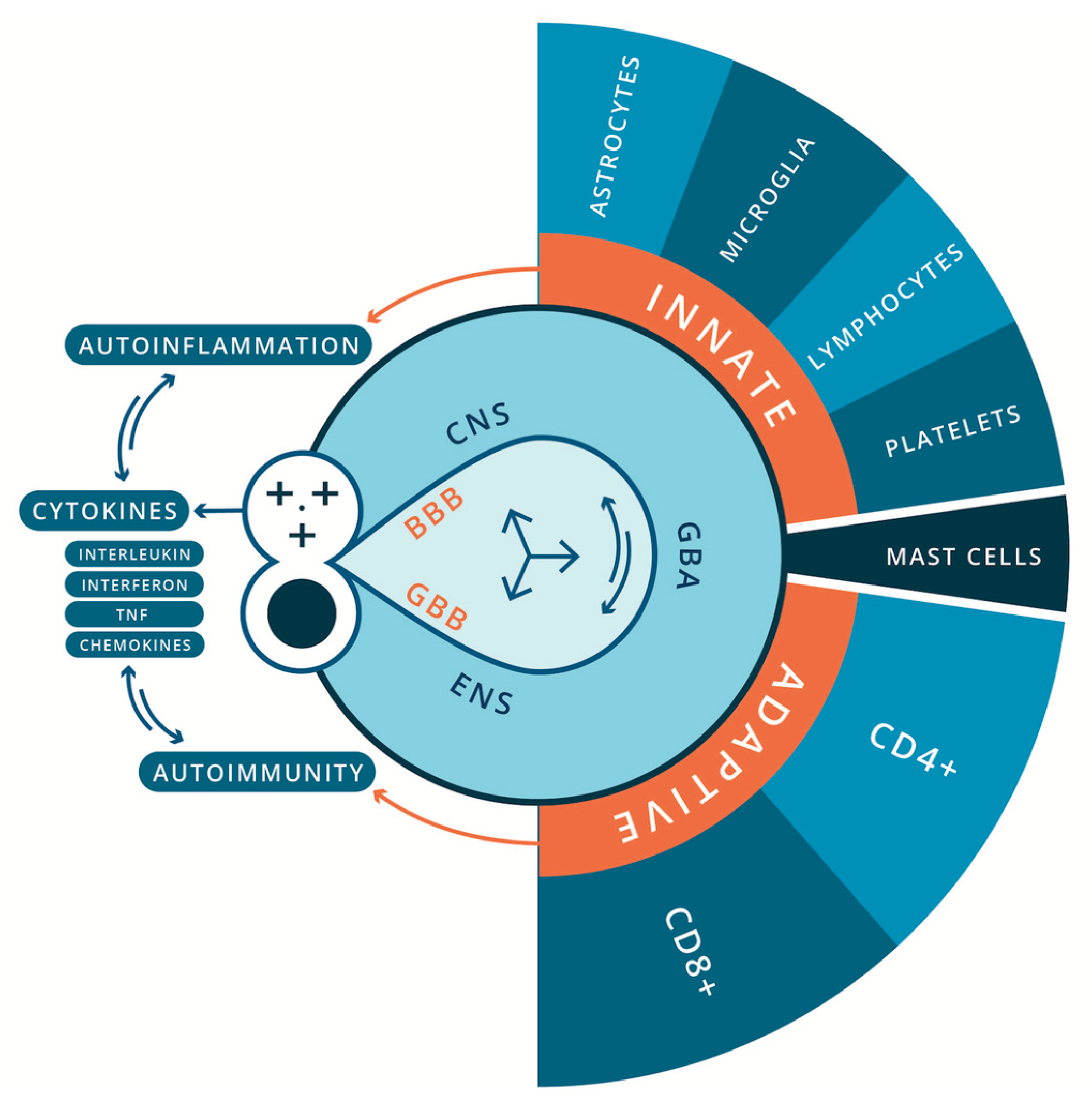
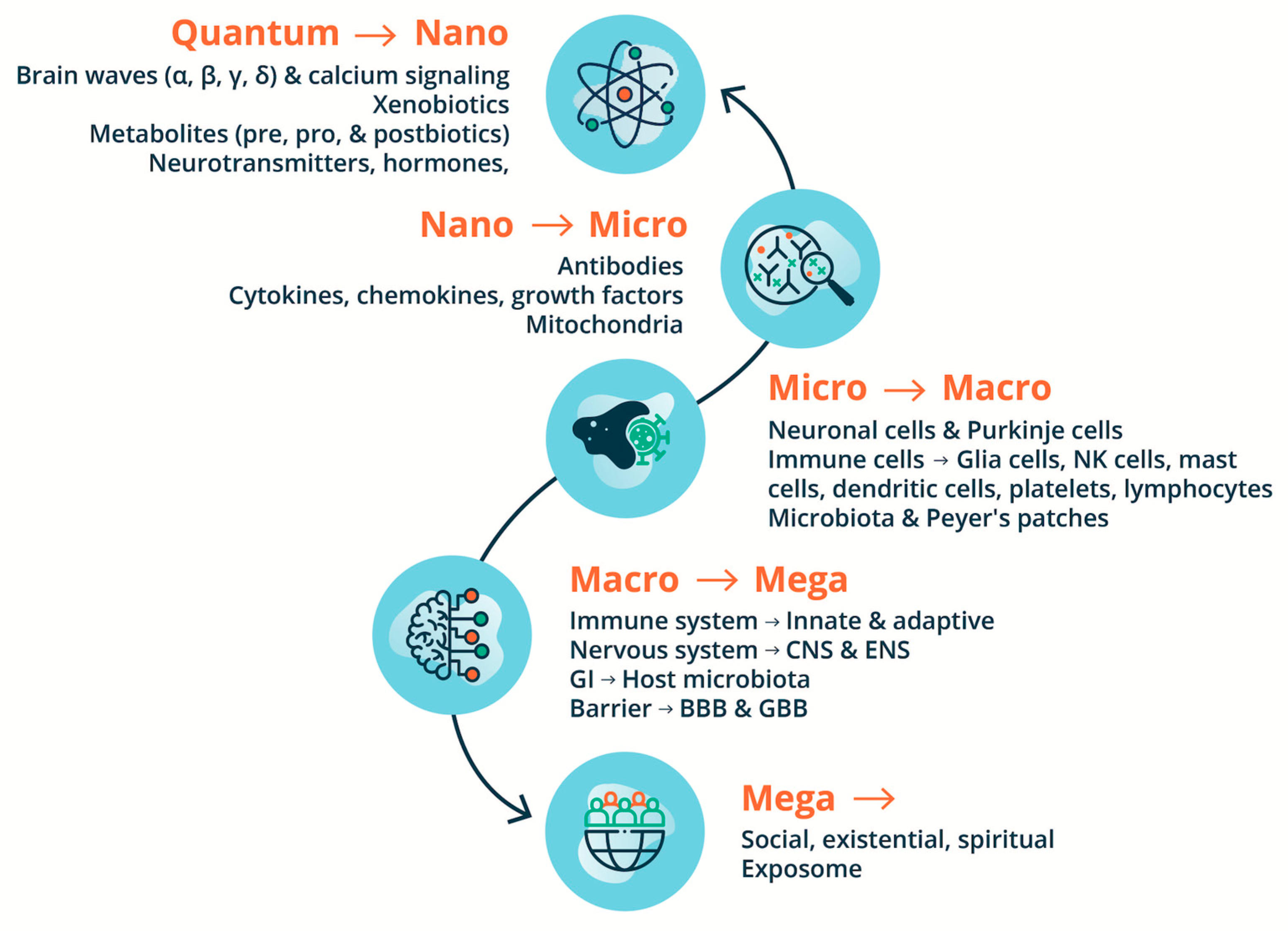
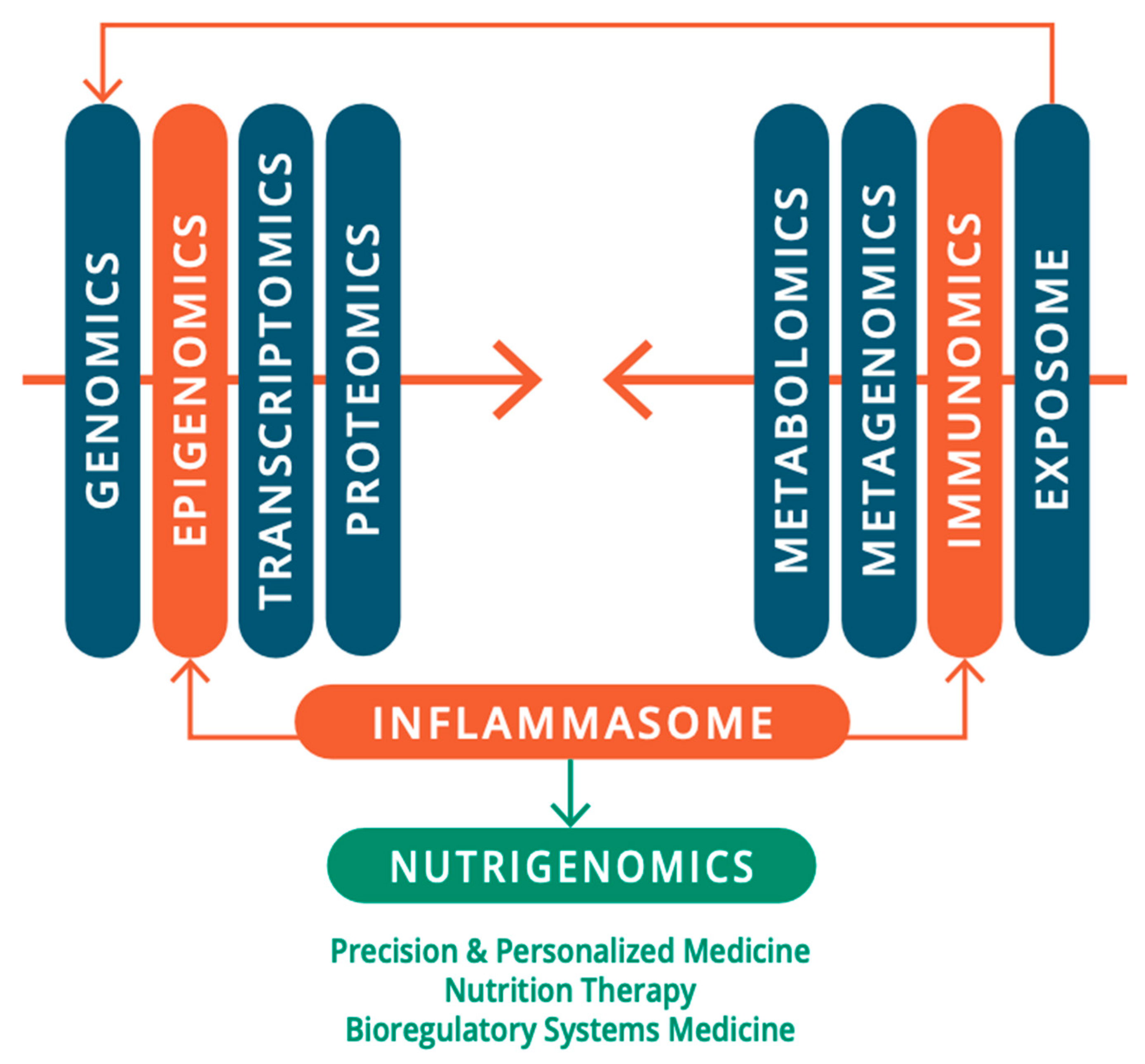
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



