Submitted:
06 November 2024
Posted:
06 November 2024
You are already at the latest version
Abstract
Keywords:
Introduction
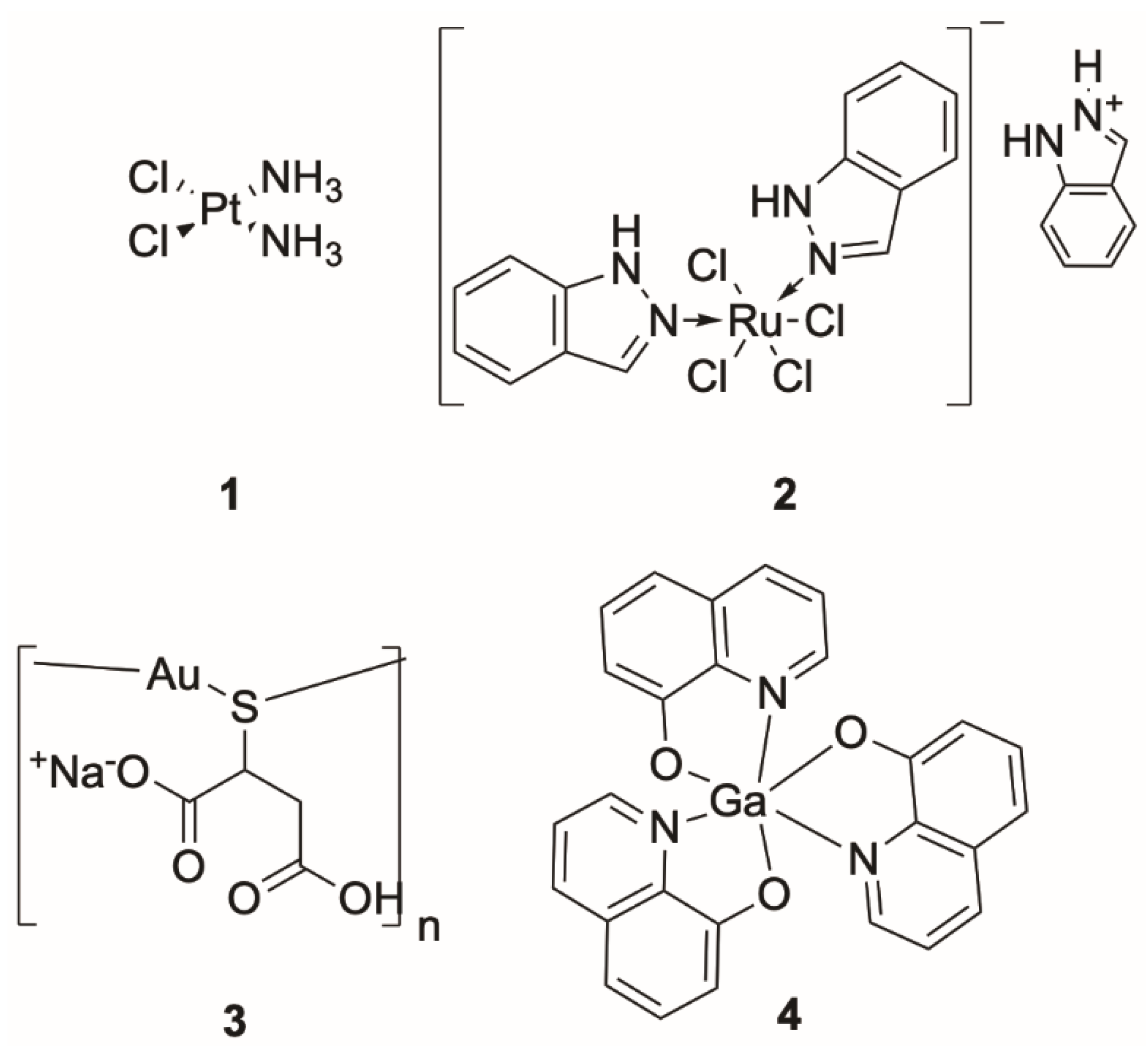
Scorpionate Chemistry
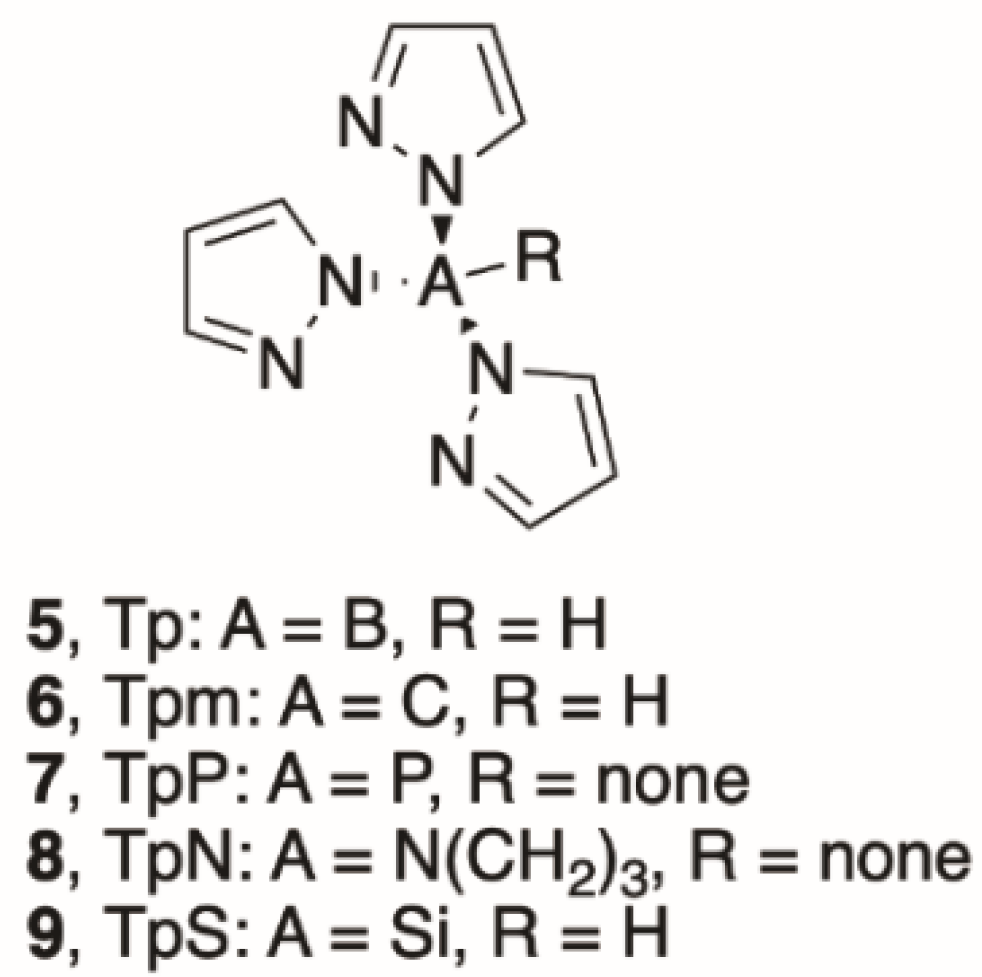
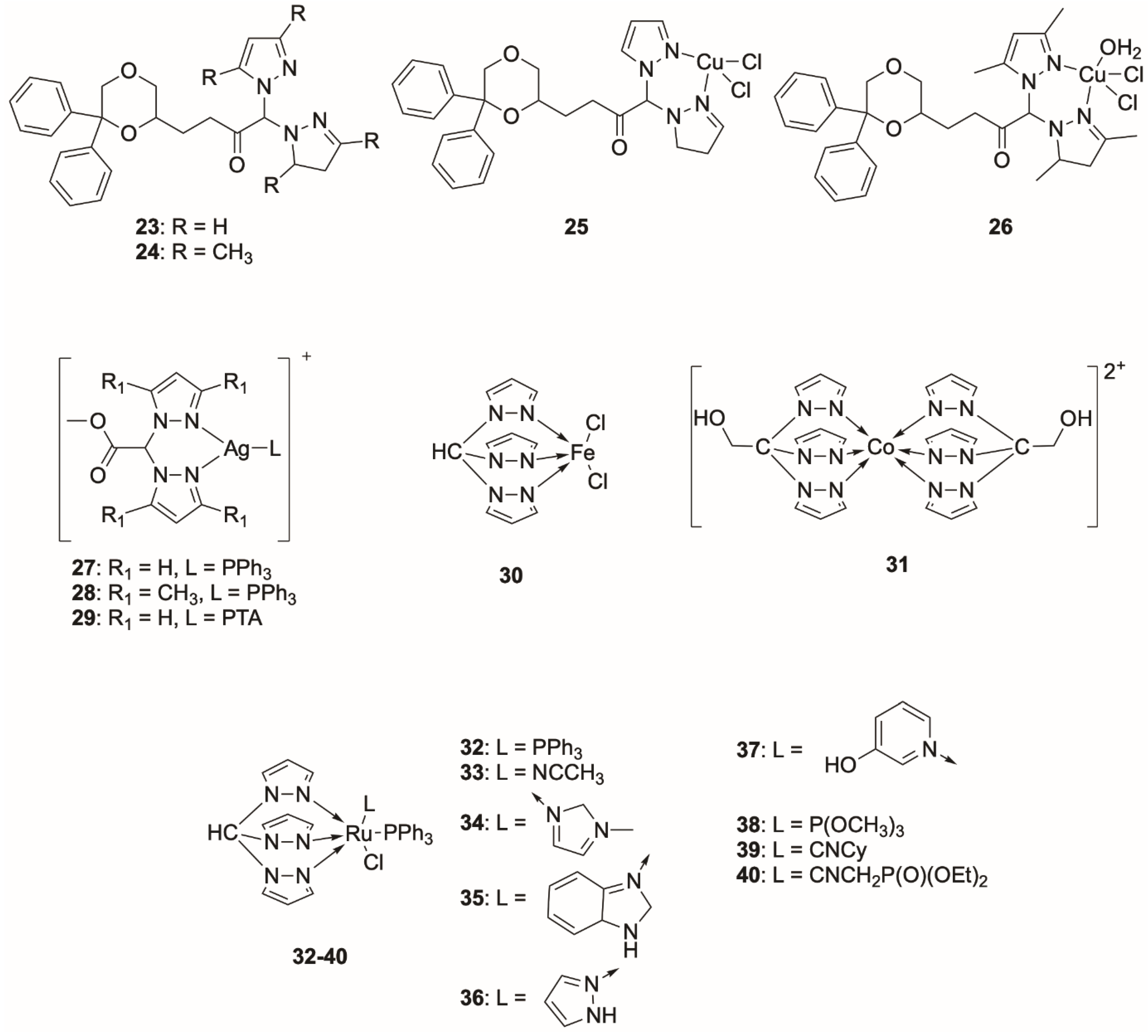
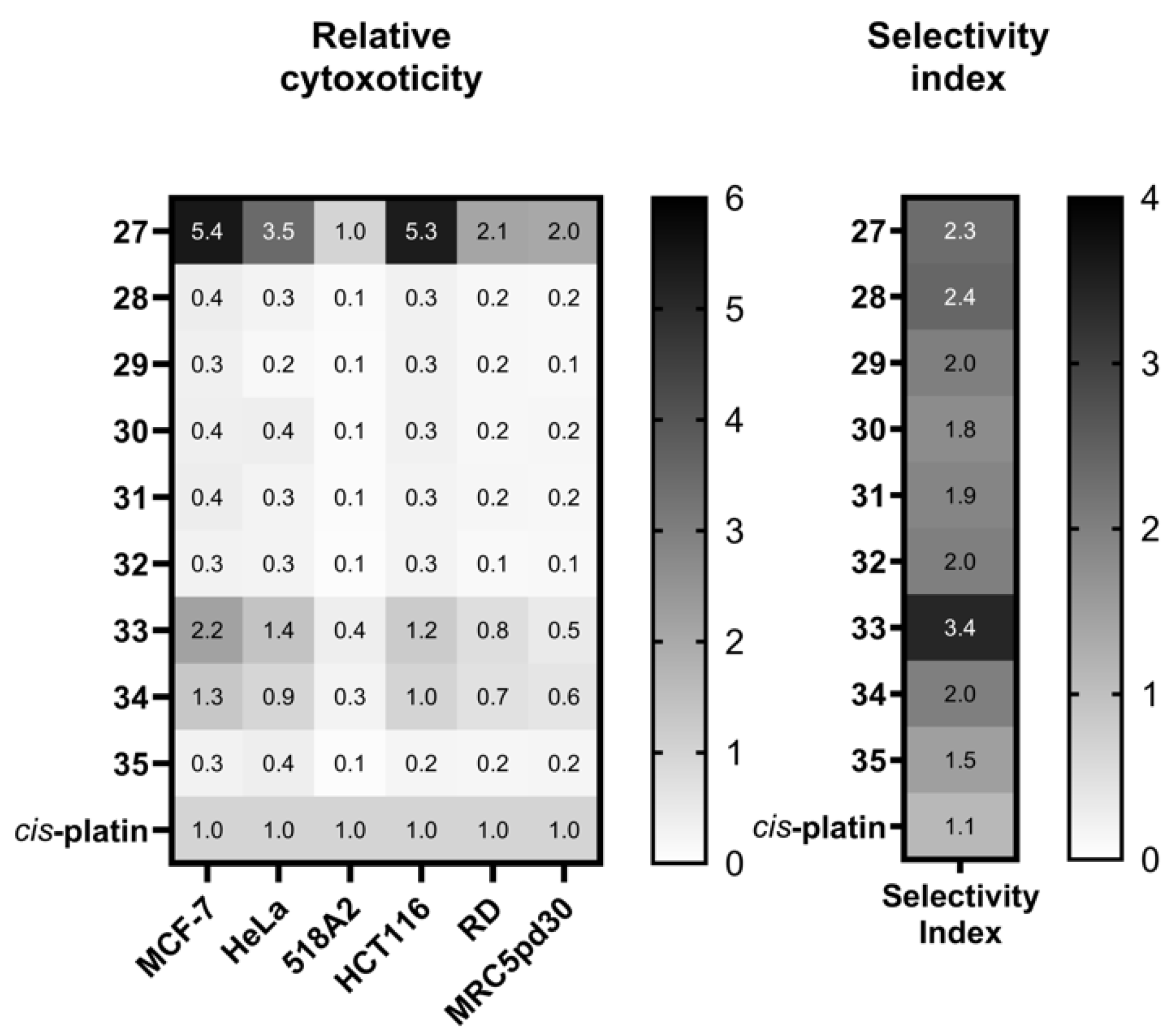
Poly(pyrazol-1-yl)borate Complexes
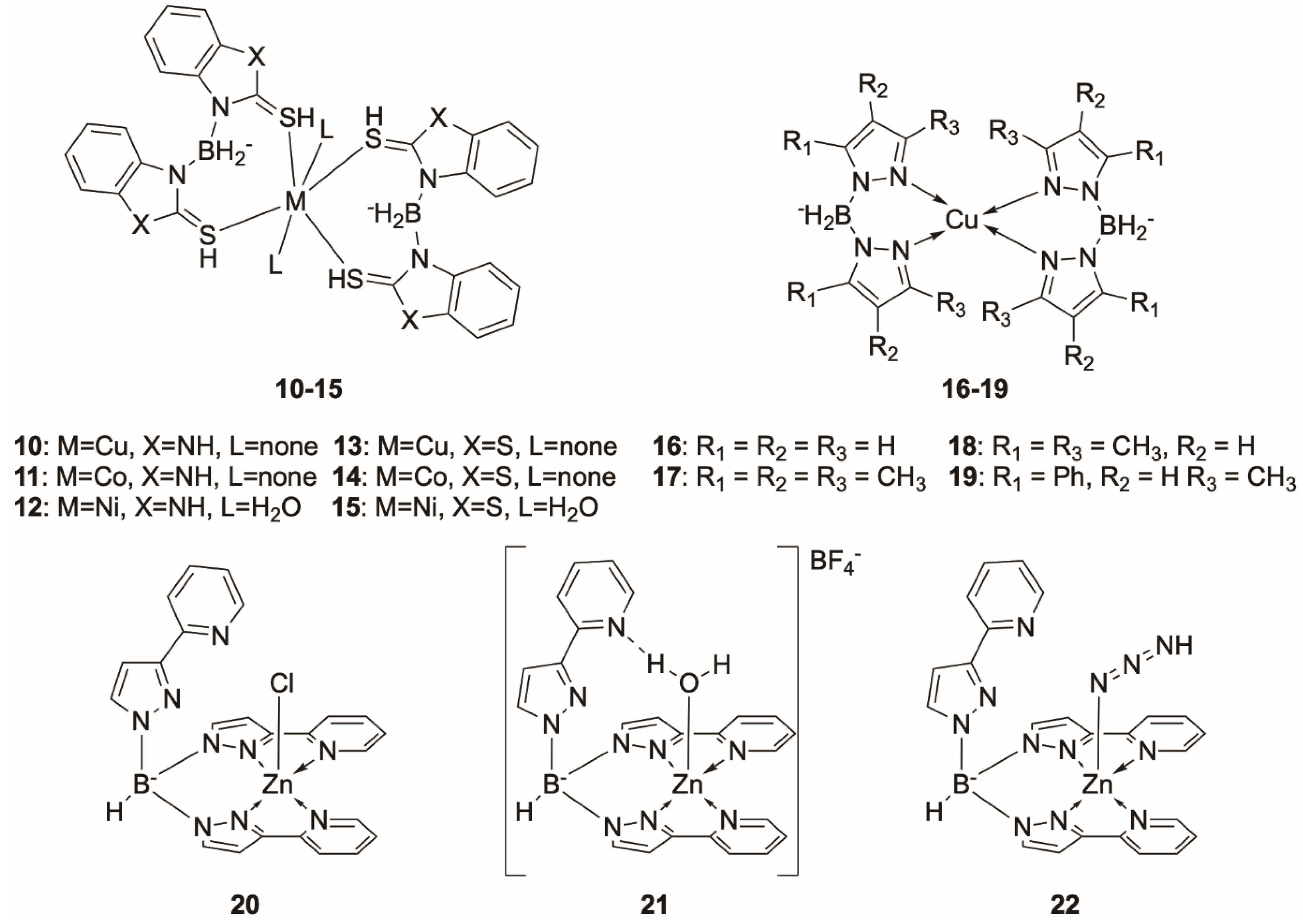
Poly(pyrazol-1-yl)methane Complexes
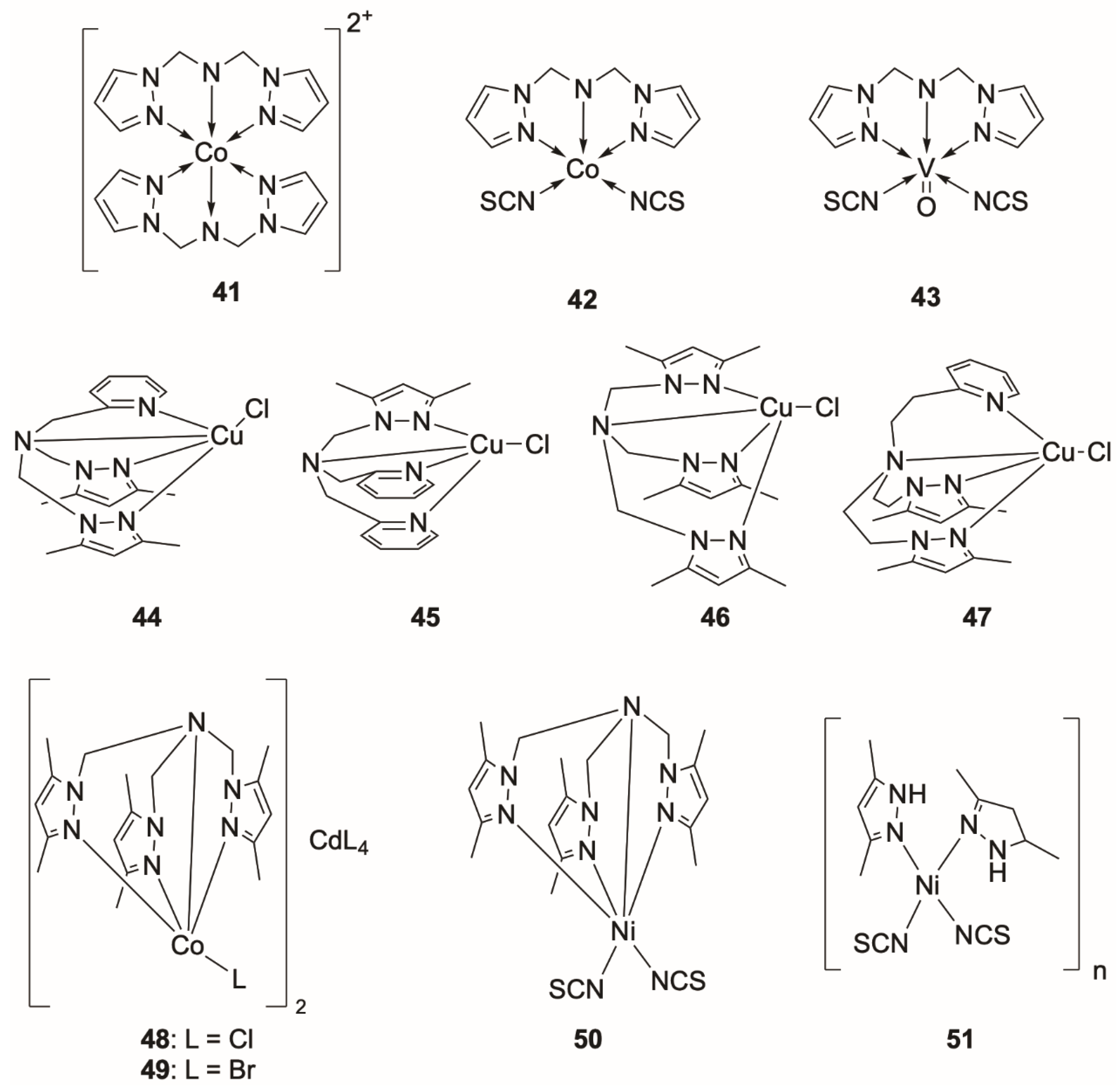
Poly(pyrazol-1-ylmethyl)amine Complexes
Conclusion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviation List
| 2008 | human ovarian carcinoma cell line |
| 518A2 | human metastatic melanoma cell line |
| A431 | human cervical carcinoma cell line |
| A549 | human lung adenocarcinoma epithelial cell line |
| A2780 | human ovarian cancer cell line |
| A2780R | cisplatin resistant human ovarian cancer cell line |
| B16 | mouse melanoma cell line |
| BJ | human fibroblast cell line |
| C13 | cisplatin resistant human ovarian cancer cell line |
| CaCo-2 | human colorectal adenocarcinoma cell line |
| Caki-2 | human renal carcinoma cell line |
| CHO-K1 | chinese hamster ovary cell line |
| H460 | human lung carcinoma cell line |
| HaCaT | human immortalized keratinocytes cell line |
| HCC1937 | human breast cancer cell line |
| HCT116 | human colon cancer cell line |
| HCT-15 | human colorectal cancer cell line |
| HeLa | human cervical cancer cell line |
| HepG2 | human hepatocellular carcinoma cell line |
| HOS | human osteosarcoma cell line |
| Hs 578T | human breast cancer cell line |
| MCF-7 | human breast cancer cell line |
| MDA-MB-231 | human breast cancer cell line |
| MDA-MB-468 | human breast cancer cell line |
| MRC5pd30 | Medical Research Council human fibroblast cell line 5 |
| NMDA | N-methyl-D-aspartate |
| PCy3 | tricyclohexylphosphine |
| PPh3 | triphenylphosphine |
| PTA | 1,3,5-triaza-7-phosphadamantane |
| PSN-1 | human breast cancer cell line |
| RD | human rhabdomyosarcoma cell line |
| ROS | reactive oxygen species |
| SKBR3 | human breast cancer cell line |
| SW620 | human colon cancer cell line |
| SW480 | human colon cancer cell line |
| SW116 | human colorectal cancer cell line |
| T24 | human bladder cancer cell line |
| TNBC | triple-negative breast cancer |
| Tp | tris(pyrazol-1-yl)borate |
| TrxR | thioredoxin reductase |
| U1285 | human breast cancer cell line |
| PC3 | human prostate cancer cell line |
References
- Anand, U.; Dey, A.; Chandel, A.K.S.; Sanyal, R.; Mishra, A.; Pandey, D.K.; De Falco, V.; Upadhyay, A.; Kandimalla, R.; Chaudhary, A. , et al. Cancer chemotherapy and beyond: Current status, drug candidates, associated risks and progress in targeted therapeutics. Genes Dis 2023, 10, 1367–1401. [Google Scholar] [CrossRef] [PubMed]
- Tilsed, C.M.; Fisher, S.A.; Nowak, A.K.; Lake, R.A.; Lesterhuis, W.J. Cancer chemotherapy: insights into cellular and tumor microenvironmental mechanisms of action. Front Oncol 2022, 12, 960317. [Google Scholar] [CrossRef] [PubMed]
- Safa, A.R. Drug and apoptosis resistance in cancer stem cells: a puzzle with many pieces. Cancer Drug Resist 2022, 5, 850–872. [Google Scholar] [CrossRef] [PubMed]
- Nurgali, K.; Jagoe, R.T.; Abalo, R. Editorial: Adverse Effects of Cancer Chemotherapy: Anything New to Improve Tolerance and Reduce Sequelae? Frontiers in Pharmacology 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Crawford, S. Is it time for a new paradigm for systemic cancer treatment? Lessons from a century of cancer chemotherapy. Frontiers in Pharmacology 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Min, H.Y.; Lee, H.Y. Molecular targeted therapy for anticancer treatment. Exp Mol Med 2022, 54, 1670–1694. [Google Scholar] [CrossRef]
- Abdolmaleki, S.; Khaksar, S.; Aliabadi, A.; Panjehpour, A.; Motieiyan, E.; Marabello, D.; Faraji, M.H.; Beihaghi, M. Cytotoxicity and mechanism of action of metal complexes: An overview. Toxicology 2023, 492, 153516. [Google Scholar] [CrossRef]
- Florea, A.-M.; Büsselberg, D. Cisplatin as an Anti-Tumor Drug: Cellular Mechanisms of Activity, Drug Resistance and Induced Side Effects. Cancers 2011, 3, 1351–1371. [Google Scholar] [CrossRef]
- Wu, S.; Wu, Z.; Ge, Q.; Zheng, X.; Yang, Z. Antitumor activity of tridentate pincer and related metal complexes. Org Biomol Chem 2021, 19, 5254–5273. [Google Scholar] [CrossRef]
- Andrade, M.A.; Martins, L. Novel Chemotherapeutic Agents - The Contribution of Scorpionates. Curr Med Chem 2019, 26, 7452–7475. [Google Scholar] [CrossRef]
- Trofimenko, S. Polypyrazolylborates: Scorpionates. Journal of Chemical Education 2005, 82, 1715. [Google Scholar] [CrossRef]
- Pricker, S.P. Medical uses of gold compounds: Past, present and future. Gold Bulletin 1996, 29, 53–60. [Google Scholar] [CrossRef]
- Frezza, M.; Hindo, S.; Chen, D.; Davenport, A.; Schmitt, S.; Tomco, D.; Dou, Q.P. Novel metals and metal complexes as platforms for cancer therapy. Curr Pharm Des 2010, 16, 1813–1825. [Google Scholar] [CrossRef]
- Das, K.; Beyene, B.B.; Datta, A.; Garribba, E.; Roma-Rodrigues, C.; Silva, A.; Fernandes, A.R.; Hung, C.-H. EPR and electrochemical interpretation of bispyrazolylacetate anchored Ni(ii) and Mn(ii) complexes: cytotoxicity and anti-proliferative activity towards human cancer cell lines. New Journal of Chemistry 2018, 42, 9126–9139. [Google Scholar] [CrossRef]
- Maroń, A.; Czerwińska, K.; Machura, B.; Raposo, L.; Roma-Rodrigues, C.; Fernandes, A.R.; Małecki, J.G.; Szlapa-Kula, A.; Kula, S.; Krompiec, S. Spectroscopy, electrochemistry and antiproliferative properties of Au(iii), Pt(ii) and Cu(ii) complexes bearing modified 2,2′:6′,2′′-terpyridine ligands. Dalton Transactions 2018, 47, 6444–6463. [Google Scholar] [CrossRef]
- Berasaluce, I.; Cseh, K.; Roller, A.; Hejl, M.; Heffeter, P.; Berger, W.; Jakupec, M.A.; Kandioller, W.; Malarek, M.S.; Keppler, B.K. The First Anticancer Tris(pyrazolyl)borate Molybdenum(IV) Complexes: Tested in Vitro and in Vivo-A Comparison of O,O-, S,O-, and N,N-Chelate Effects. Chemistry 2020, 26, 2211–2221. [Google Scholar] [CrossRef]
- Tyszka-Czochara, M.; Adach, A.; Grabowski, T.; Konieczny, P.; Pasko, P.; Ortyl, J.; Świergosz, T.; Majka, M. Selective Cytotoxicity of Complexes with N,N,N-Donor Dipodal Ligand in Tumor Cells. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Narwane, M.; Dorairaj, D.P.; Chang, Y.L.; Karvembu, R.; Huang, Y.H.; Chang, H.W.; Hsu, S.C.N. Tris-(2-pyridyl)-pyrazolyl Borate Zinc(II) Complexes: Synthesis, DNA/Protein Binding and In Vitro Cytotoxicity Studies. Molecules 2021, 26. [Google Scholar] [CrossRef]
- Trofimenko, S. Boron-pyrazole chemistry. II. Poly(1-pyrazolyl)-borates. Journal of the American Chemical Society 1967, 89, 3170–3177. [Google Scholar] [CrossRef]
- Tăbăcaru, A.; Khan, R.A.; Lupidi, G.; Pettinari, C. Synthesis, Characterization and Assessment of the Antioxidant Activity of Cu(II), Zn(II) and Cd(II) Complexes Derived from Scorpionate Ligands. Molecules 2020, 25, 5298. [Google Scholar] [CrossRef]
- Silva, F.; Fernandes, C.; Campello, M.P.C.; Paulo, A. Metal complexes of tridentate tripod ligands in medical imaging and therapy. Polyhedron 2017, 125, 186–205. [Google Scholar] [CrossRef]
- Tazelaar, C.G.J.; Slootweg, J.C.; Lammertsma, K. Coordination chemistry of tris(azolyl)phosphines. Coordination Chemistry Reviews 2018, 356, 115–126. [Google Scholar] [CrossRef]
- Bitto, F.; Wagler, J.; Kroke, E. 3,5-Dimethylpyrazole Derivatives of (Hydrido)chlorosilanes. European Journal of Inorganic Chemistry 2012, 2012, 2402–2408. [Google Scholar] [CrossRef]
- Adach, A. Review: an overview of recent developments in coordination chemistry of polypyrazolylmethylamines. Complexes with N-scorpionate ligands created in situ from pyrazole derivatives and zerovalent metals. Journal of Coordination Chemistry 2017, 70, 757–779. [Google Scholar] [CrossRef]
- Banerjee, A.; Li, J.; Easley, C.R.; Brennessel, W.W.; Loloee, R.; Chavez, F.A. Synthesis, structure, and characterization of tris(1-ethyl-4-isopropyl-imidazolyl-қN)phosphine nickel(II) complexes. Inorganica Chimica Acta 2019, 489, 170–179. [Google Scholar] [CrossRef]
- Bartholomew, A.K.; Guard, L.M.; Hazari, N.; Luzik, E.D. Synthesis of Mg Complexes Supported by Tris-(1-pyrazolyl)phosphine. Australian Journal of Chemistry 2013, 66, 1455–1458. [Google Scholar] [CrossRef]
- Pullen, E.E.; Rheingold, A.L.; Rabinovich, D. Methyltris(pyrazolyl)silanes: new tripodal nitrogen-donor ligands. Inorganic Chemistry Communications 1999, 2, 194–196. [Google Scholar] [CrossRef]
- Pullen, E.E.; Rabinovich, D.; Incarvito, C.D.; Concolino, T.E.; Rheingold, A.L. Syntheses and Structures of Methyltris(pyrazolyl)silane Complexes of the Group 6 Metals. Inorganic Chemistry 2000, 39, 1561–1567. [Google Scholar] [CrossRef]
- Karam, A.; Tenia, R.; Martínez, M.; López-Linares, F.; Albano, C.; Diaz-Barrios, A.; Sánchez, Y.; Catarí, E.; Casas, E.; Pekerar, S. , et al. Iron(II) and cobalt(II) tris(2-pyridyl)phosphine and tris(2-pyridyl)amine catalysts for the ethylene polymerization. Journal of Molecular Catalysis A: Chemical 2007, 265, 127–132. [Google Scholar] [CrossRef]
- Setifi, Z.; Cubillán, N.; Glidewell, C.; Gil, D.M.; Torabi, E.; Morales-Toyo, M.; Dege, N.; Setifi, F.; Mirzaei, M. A combined experimental, Hirshfeld surface analysis, and theoretical study on fac-[tri(azido)(tris(2-pyridyl)amine)iron(III)]. Polyhedron 2023, 233, 116320. [Google Scholar] [CrossRef]
- Mautner, F.A.; Albering, J.H.; Vicente, R.; Louka, F.R.; Gallo, A.A.; Massoud, S.S. Copper(II) complexes derived from tripodal tris[(2-ethyl-(1-pyrazolyl)]amine. Inorganica Chimica Acta 2011, 365, 290–296. [Google Scholar] [CrossRef]
- Martins, L.M.D.R.S.; Pombeiro, A.J.L. Tris(pyrazol-1-yl)methane metal complexes for catalytic mild oxidative functionalizations of alkanes, alkenes and ketones. Coordination Chemistry Reviews 2014, 265, 74–88. [Google Scholar] [CrossRef]
- Santini, C.; Pellei, M.; Lobbia, G.G.; Papini, G. Synthesis and Properties of Poly(pyrazolyl)borate and Related Boron-Centered Scorpionate Ligands. Part A: Pyrazole-Based Systems. Mini-Reviews in Organic Chemistry 2010, 7, 84–124. [Google Scholar] [CrossRef]
- Antunes, M.A.; Domingos, Â.; Santos, I.C.d.; Marques, N.; Takats, J. Synthesis and characterization of uranium(III) compounds supported by the hydrotris(3,5-dimethyl-pyrazolyl)borate ligand: Crystal structures of [U(TpMe2)2(X)] complexes (X=OC6H2-2,4,6-Me3, dmpz, Cl). Polyhedron 2005, 24, 3038–3045. [Google Scholar] [CrossRef]
- Saliu, K.O.; Maunder, G.H.; Ferguson, M.J.; Sella, A.; Takats, J. Synthesis and structure of heteroleptic ytterbium (II) tetrahydroborate complexes. Inorganica Chimica Acta 2009, 362, 4616–4622. [Google Scholar] [CrossRef]
- Liang, S.; Jensen, M.P. Half-Sandwich Scorpionates as Nitrene Transfer Catalysts. Organometallics 2012, 31, 8055–8058. [Google Scholar] [CrossRef]
- Otero, A.; Fernández-Baeza, J.; Lara-Sánchez, A.; Sánchez-Barba, L.F. Metal complexes with heteroscorpionate ligands based on the bis(pyrazol-1-yl)methane moiety: Catalytic chemistry. Coordination Chemistry Reviews 2013, 257, 1806–1868. [Google Scholar] [CrossRef]
- John, R.; Mark, D.S. The Bioinorganic Chemistry of Methimazole Based Soft Scorpionates. Current Bioactive Compounds 2009, 5, 264–276. [Google Scholar] [CrossRef]
- Pellei, M.; Lobbia, G.G.; Santini, C.; Spagna, R.; Camalli, M.; Fedeli, D.; Falcioni, G. Synthesis, characterization and antioxidant activity of new copper(i) complexes of scorpionate and water soluble phosphane ligands. Dalton Transactions 2004. [CrossRef]
- Berasaluce, I.; Cseh, K.; Roller, A.; Hejl, M.; Heffeter, P.; Berger, W.; Jakupec, M.A.; Kandioller, W.; Malarek, M.S.; Keppler, B.K. The First Anticancer Tris(pyrazolyl)borate Molybdenum(IV) Complexes: Tested in Vitro and in Vivo—A Comparison of O,O-, S,O-, and N,N-Chelate Effects. Chemistry – A European Journal 2020, 26, 2211–2221. [Google Scholar] [CrossRef] [PubMed]
- Faghih, Z.; Neshat, A.; Mastrorilli, P.; Gallo, V.; Faghih, Z.; Gilanchi, S. Cu(II), Ni(II) and Co(II) complexes with homoscorpionate Bis(2-Mercaptobenzimidazolyl) and Bis(2-Mercaptobenzothiazolyl)borate ligands: Synthesis and in vitro cytotoxicity studies. Inorganica Chimica Acta 2020, 512, 119896. [Google Scholar] [CrossRef]
- Azam, M.A.; Suresh, B. Biological activities of 2-mercaptobenzothiazole derivatives: a review. Sci Pharm 2012, 80, 789–823. [Google Scholar] [CrossRef] [PubMed]
- Shah, K.; Chhabra, S.; Shrivastava, S.K.; Mishra, P. Benzimidazole: a promising pharmacophore. Medicinal Chemistry Research 2013, 22, 5077–5104. [Google Scholar] [CrossRef]
- Ghorbanpour, M.; Soltani, B.; Molavi, O.; Shayanfar, A.; Mehdizadeh Aghdam, E.; Ziegler, C.J. Copper (II) complexes based bis(pyrazolyl)borate derivatives as efficient anticancer agents: synthesis, characterization, X-ray structure, cytotoxicity, molecular docking and QSAR studies. Chemical Papers 2022, 76, 7343–7356. [Google Scholar] [CrossRef]
- Maret, W. Zinc in Cellular Regulation: The Nature and Significance of "Zinc Signals". Int J Mol Sci 2017, 18. [Google Scholar] [CrossRef]
- Chen, B.; Yu, P.; Chan, W.N.; Xie, F.; Zhang, Y.; Liang, L.; Leung, K.T.; Lo, K.W.; Yu, J.; Tse, G.M.K. , et al. Cellular zinc metabolism and zinc signaling: from biological functions to diseases and therapeutic targets. Signal Transduction and Targeted Therapy 2024, 9, 6. [Google Scholar] [CrossRef]
- Morelli, M.B.; Amantini, C.; Santoni, G.; Pellei, M.; Santini, C.; Cimarelli, C.; Marcantoni, E.; Petrini, M.; Del Bello, F.; Giorgioni, G. , et al. Novel antitumor copper(ii) complexes designed to act through synergistic mechanisms of action, due to the presence of an NMDA receptor ligand and copper in the same chemical entity. New Journal of Chemistry 2018, 42, 11878–11887. [Google Scholar] [CrossRef]
- Pellei, M.; Santini, C.; Bagnarelli, L.; Caviglia, M.; Sgarbossa, P.; De Franco, M.; Zancato, M.; Marzano, C.; Gandin, V. Novel Silver Complexes Based on Phosphanes and Ester Derivatives of Bis(pyrazol-1-yl)acetate Ligands Targeting TrxR: New Promising Chemotherapeutic Tools Relevant to SCLC Management. International Journal of Molecular Sciences 2023, 24, 4091. [Google Scholar] [CrossRef]
- Silva, P.M.G.; Pinheiro, P.F.; Camões, S.P.; Ribeiro, A.P.C.; Martins, L.; Miranda, J.P.G.; Justino, G.C. Exploring the Mechanisms behind the Anti-Tumoral Effects of Model C-Scorpionate Complexes. Molecules 2023, 28. [Google Scholar] [CrossRef]
- Cervinka, J.; Gobbo, A.; Biancalana, L.; Markova, L.; Novohradsky, V.; Guelfi, M.; Zacchini, S.; Kasparkova, J.; Brabec, V.; Marchetti, F. Ruthenium(II)-Tris-pyrazolylmethane Complexes Inhibit Cancer Cell Growth by Disrupting Mitochondrial Calcium Homeostasis. J Med Chem 2022, 65, 10567–10587. [Google Scholar] [CrossRef]
- North, W.G.; Gao, G.; Memoli, V.A.; Pang, R.H.; Lynch, L. Breast cancer expresses functional NMDA receptors. Breast Cancer Res Treat 2010, 122, 307–314. [Google Scholar] [CrossRef]
- Hanson, S.; Dharan, A.; P, V.J.; Pal, S.; Nair, B.G.; Kar, R.; Mishra, N. Paraptosis: a unique cell death mode for targeting cancer. Front Pharmacol 2023, 14, 1159409. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, F.; Soltani, A.; Ghahremanloo, A.; Javid, H.; Hashemy, S.I. The thioredoxin system and cancer therapy: a review. Cancer Chemother Pharmacol 2019, 84, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Massoud, S.S.; Louka, F.R.; Ducharme, G.T.; Fischer, R.C.; Mautner, F.A.; Vančo, J.; Herchel, R.; Dvořák, Z.; Trávníček, Z. Copper(II) complexes based on tripodal pyrazolyl amines: Synthesis, structure, magnetic properties and anticancer activity. Journal of Inorganic Biochemistry 2018, 180, 39–46. [Google Scholar] [CrossRef]
- Adach, A.; Daszkiewicz, M.; Tyszka-Czochara, M. Comparative X-ray, vibrational, theoretical and biological studies of new in situ formed [CoLSX]2[CdX4] halogenocadmate(II) complexes containing N-scorpionate ligand. Polyhedron 2020, 175, 114229. [Google Scholar] [CrossRef]
- Adach, A.; Tyszka-Czochara, M.; Bukowska-Strakova, K.; Rejnhardt, P.; Daszkiewicz, M. In situ synthesis, crystal structure, selective anticancer and proapoptotic activity of complexes isolated from the system containing zerovalent nickel and pyrazole derivatives. Polyhedron 2022, 223, 115943. [Google Scholar] [CrossRef]
- Pfeffer, C.M.; Singh, A.T.K. Apoptosis: A Target for Anticancer Therapy. Int J Mol Sci 2018, 19. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).