Submitted:
25 November 2024
Posted:
27 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Testing Antibodies to Treat Neurological Disorders
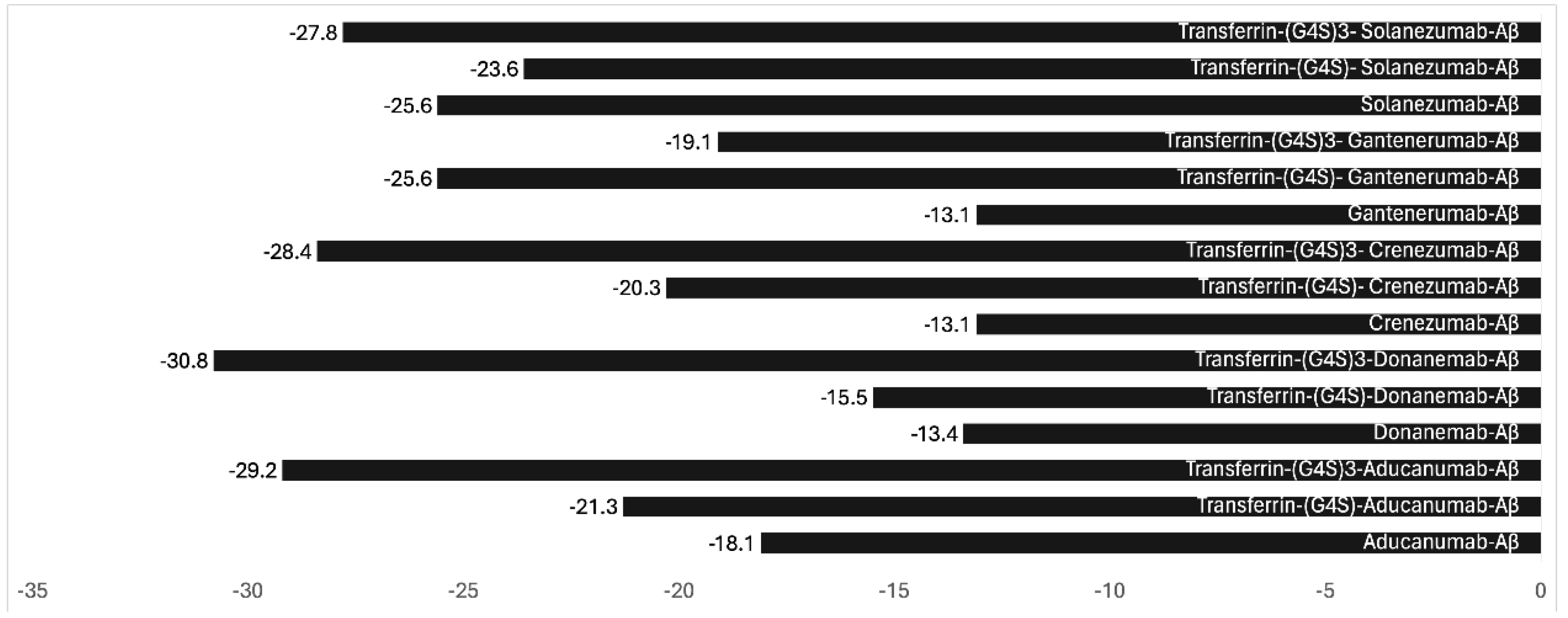
3. Future
5. Conclusion
Funding
Acknowledgments
Conflicts of Interest
References
- Kessler WBaC. What Drugmakers Did Not Tell Volunteers in Alzheimer’s Trials https://www.nytimes.com/2024/10/23/health/alzheimers-drug-brain-bleeding.html: The New York Times: 2024.
- Giarratana AO, Zheng C, Reddi S, Teng SL, Berger D, Adler D, et al. APOE4 genetic polymorphism results in impaired recovery in a repeated mild traumatic brain injury model and treatment with Bryostatin-1 improves outcomes. Scientific Reports. 2020;10(1):19919. [CrossRef]
- Huang LK, Kuan YC, Lin HW, Hu CJ. Clinical trials of new drugs for Alzheimer disease: a 2020-2023 update. J Biomed Sci. 2023;30(1):83. [CrossRef]
- FDA. FDA Grants Accelerated Approval for Alzheimer’s Disease Treatment https://www.fda.gov/news-events/press-announcements/fda-grants-accelerated-approval-alzheimers-disease-treatment2023 [.
- EMA. Refusal of the marketing authorisation for Leqembi (lecanemab) https://www.ema.europa.eu/en/documents/smop-initial/questions-answers-refusal-marketing-authorisation-leqembi-lecanemab_en.pdf2024 [.
- TGA-Australia. TGA's decision to not register lecanemab (LEQEMBI) https://www.tga.gov.au/news/news/tgas-decision-not-register-lecanemab-leqembi2024 [.
- Mo JJ, Li JY, Yang Z, Liu Z, Feng JS. Efficacy and safety of anti-amyloid-β immunotherapy for Alzheimer's disease: a systematic review and network meta-analysis. Ann Clin Transl Neurol. 2017;4(12):931-42.
- van Dyck CH, Swanson CJ, Aisen P, Bateman RJ, Chen C, Gee M, et al. Lecanemab in Early Alzheimer's Disease. N Engl J Med. 2023;388(1):9-21. [CrossRef]
- Baggett D, Olson A, Parmar MS. Novel approaches targeting α-Synuclein for Parkinson's Disease: Current progress and future directions for the disease-modifying therapies. Brain Disorders. 2024;16:100163.
- Sevigny J, Chiao P, Bussière T, Weinreb PH, Williams L, Maier M, et al. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature. 2016;537(7618):50-6.
- Salloway S, Sperling R, Fox NC, Blennow K, Klunk W, Raskind M, et al. Two phase 3 trials of bapineuzumab in mild-to-moderate Alzheimer's disease. N Engl J Med. 2014;370(4):322-33.
- Sigurdsson EM. Tau Immunotherapies for Alzheimer's Disease and Related Tauopathies: Progress and Potential Pitfalls. J Alzheimers Dis. 2018;64(s1):S555-s65. [CrossRef]
- Manoutcharian K, Gevorkian G. Recombinant Antibody Fragments for Immunotherapy of Parkinson's Disease. BioDrugs. 2024;38(2):249-57. [CrossRef]
- Cummings J, Lee G, Ritter A, Zhong K. Alzheimer's disease drug development pipeline: 2018. Alzheimers Dement (N Y). 2018;4:195-214.
- Ostrowitzki S, Lasser RA, Dorflinger E, Scheltens P, Barkhof F, Nikolcheva T, et al. A phase III randomized trial of gantenerumab in prodromal Alzheimer's disease. Alzheimers Res Ther. 2017;9(1):95. [CrossRef]
- Rosenfeld PJ, Berger B, Reichel E, Danis RP, Gress A, Ye L, et al. A Randomized Phase 2 Study of an Anti–Amyloid β Monoclonal Antibody in Geographic Atrophy Secondary to Age-Related Macular Degeneration. Ophthalmology Retina. 2018;2(10):1028-40. [CrossRef]
- Landen JW, Andreasen N, Cronenberger CL, Schwartz PF, Börjesson-Hanson A, Östlund H, et al. Ponezumab in mild-to-moderate Alzheimer's disease: Randomized phase II PET-PIB study. Alzheimer's & Dementia: Translational Research & Clinical Interventions. 2017;3(3):393-401.
- Teng E, Manser PT, Pickthorn K, Brunstein F, Blendstrup M, Sanabria Bohorquez S, et al. Safety and Efficacy of Semorinemab in Individuals With Prodromal to Mild Alzheimer Disease: A Randomized Clinical Trial. JAMA Neurol. 2022;79(8):758-67.
- Doody RS, Thomas RG, Farlow M, Iwatsubo T, Vellas B, Joffe S, et al. Phase 3 trials of solanezumab for mild-to-moderate Alzheimer's disease. N Engl J Med. 2014;370(4):311-21.
- Krämer J, Wiendl H. What Have Failed, Interrupted, and Withdrawn Antibody Therapies in Multiple Sclerosis Taught Us? Neurotherapeutics. 2022;19(3):785-807.
- Niazi SK. Bioavailability as Proof to Authorize the Clinical Testing of Neurodegenerative Drugs—Protocols and Advice for the FDA to Meet the ALS Act Vision. International Journal of Molecular Sciences. 2024;25(18):10211. [CrossRef]
- Kouhi A, Pachipulusu V, Kapenstein T, Hu P, Epstein AL, Khawli LA. Brain Disposition of Antibody-Based Therapeutics: Dogma, Approaches and Perspectives. Int J Mol Sci. 2021;22(12). [CrossRef]
- Dumitriu A, Popescu BO. Placebo effects in neurological diseases. J Med Life. 2010;3(2):114-21.
- Oken BS. Placebo effects: clinical aspects and neurobiology. Brain. 2008;131(11):2812-23. [CrossRef]
- Peciña M, Zubieta J-K. Molecular Mechanisms of Placebo Responses In Humans. Molecular psychiatry. 2014;20:416 - 23.
- Niazi SK. A Modern View of Placebo Interventions: From Comparative Clinical Trials to Novel Therapies to Quantum Tunnelling. Preprints: Preprints; 2024.
- Niazi SK, Mariam Z, Magoola M. Engineered Antibodies to Improve Efficacy against Neurodegenerative Disorders. International Journal of Molecular Sciences. 2024;25(12):6683. [CrossRef]
- Niazi SK. Non-Invasive Drug Delivery across the Blood–Brain Barrier: A Prospective Analysis. Pharmaceutics. 2023;15(11):2599. [CrossRef]
- Tien J, Leonoudakis D, Petrova R, Trinh V, Taura T, Sengupta D, et al. Modifying antibody-FcRn interactions to increase the transport of antibodies through the blood-brain barrier. MAbs. 2023;15(1):2229098.
- Marathe PH, Shyu WC, Humphreys WG. The use of radiolabeled compounds for ADME studies in discovery and exploratory development. Curr Pharm Des. 2004;10(24):2991-3008. [CrossRef]
- Arndt JW, Qian F, Smith BA, Quan C, Kilambi KP, Bush MW, et al. Structural and kinetic basis for the selectivity of aducanumab for aggregated forms of amyloid-β. Scientific Reports. 2018;8(1):6412. [CrossRef]
- Nelke C, Spatola M, Schroeter CB, Wiendl H, Lünemann JD. Neonatal Fc Receptor-Targeted Therapies in Neurology. Neurotherapeutics. 2022;19(3):729-40. [CrossRef]
- Watanabe T, Arashida N, Fujii T, Shikida N, Ito K, Shimbo K, et al. Exo-Cleavable Linkers: Enhanced Stability and Therapeutic Efficacy in Antibody–Drug Conjugates. Journal of Medicinal Chemistry. 2024;67(20):18124-38. [CrossRef]
- Roopenian DC, Akilesh S. FcRn: the neonatal Fc receptor comes of age. Nat Rev Immunol. 2007;7(9):715-25. [CrossRef]
| Monoclonal Antibody | Target | Indication | Stages |
|---|---|---|---|
| Aducanumab [10] | Amyloid-β | Alzheimer's Disease | Initially approved, then withdrawn due to efficacy concerns and safety issues. |
| Bapineuzumab [11] | Amyloid-β | Alzheimer's Disease | Phase III trials did not demonstrate significant cognitive improvements; development discontinued. |
| Bepranemab [12] | Tau | Alzheimer's Disease | Phase II trials did not meet primary endpoints; development status uncertain. |
| Crenezumab [14] | Amyloid-β | Alzheimer's Disease | Phase III trials did not meet primary endpoints; development discontinued. |
| Gantenerumab [15], | Amyloid-β | Alzheimer's Disease | Phase III trials did not meet primary endpoints; development discontinued. |
| GSK933776 [16] | Amyloid-β | Alzheimer's Disease | Phase II trials did not meet primary endpoints; development discontinued. |
| Ponezumab [17] | Amyloid-β | Alzheimer's Disease | Phase II trials showed no significant efficacy; development halted. |
| Semorinema [18] | Tau | Alzheimer's Disease | Phase II trials did not meet primary endpoints; development status uncertain. |
| Solanezumab [19] | Amyloid-β | Alzheimer's Disease | Phase III trials failed to meet primary endpoints; development halted. |
| Tilavonemab [12] |
Tau | Alzheimer's Disease | Phase II trials did not meet primary endpoints; development status uncertain. |
| Atacicept [20] | BLyS and APRIL | Multiple Sclerosis | Phase II trials showed increased disease activity, leading to termination. |
| Daclizumab [20] | CD25 (IL-2 receptor α) | Multiple Sclerosis | Withdrawn due to serious inflammatory brain disorders and other immune-mediated conditions. |
| Opicinumab [20] | LINGO-1 | Multiple Sclerosis | Phase II trials did not meet endpoints for remyelination and functional improvement. |
| Tabalumab [20] | BAFF | Multiple Sclerosis | Phase II trials did not demonstrate efficacy in reducing disease activity. |
| Cinpanemab [13], | α-Synuclein | Parkinson's Disease | Phase II SPARK trial terminated due to lack of efficacy. |
| Prasinezumab | α-Synuclein | Parkinson's Disease | Phase II trials did not meet primary endpoints; development status uncertain. |
| Antibody | United States | European Union | United Kingdom | Japan | China | Australia | Canada |
|---|---|---|---|---|---|---|---|
| Aducanumab | Approved by FDA (2021) FDA | Rejected by EMA (2021) EMA | Not approved | Not approved | Not approved | Not approved | Not approved |
| Lecanemab | Approved by FDA (2023) FDA | Rejected by EMA (2023) Reuters | Approved by MHRA (2023); Not recommended by NICE Reuters | Approved (2023) | Approved (2023) | Not approved by TGA (2023) Herald Sun | Approved (2023) |
| Donanemab | Approved by FDA (2024) FDA | Under EMA review | Approved by MHRA (2024); Not recommended by NICE for NHS Reuters | Application submitted; pending decision | Application submitted; pending decision | Application submitted; pending decision | Application submitted; pending decision |
| Ocrelizumab | Approved by FDA (2017) FDA | Approved by EMA (2018) EMA | Approved in line with EMA decision | Approved (2019) | Approved (2021) | Approved (2017) | Approved (2017) |
| Ofatumumab | Approved by FDA (2020) FDA | Approved by EMA (2021) EMA | Approved in line with EMA decision | Approved (2021) | Approved (2021) | Approved (2021) | Approved (2021) |
| Alemtuzumab | Approved by FDA (2014) FDA | Approved by EMA (2013) EMA | Approved in line with EMA decision | Approved (2014) | Not approved | Approved (2013) | Approved (2013) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




