Submitted:
04 November 2024
Posted:
06 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
- The goal of this scientific research is to answer the explanation of the phenomenon of quantum leap and quantum entanglement and to add some modifications in the Bohr model.
2. Equations
3. These laws have been modified from the mix Planck laws
- How quantum entanglement occurs?
4. Method
- This part of the research will explain the spectrum of the hydrogen atom in a new way, as the results presented in these tables from previous research match the results extracted from the equation, and this is consistent with the validity of this equation. Because the new equation is consistent with the photon energy equation. We will discuss that part of the research in the results and discussion.
- Table 5 shows the measurement results tested.[3] (Nanni, 2015).
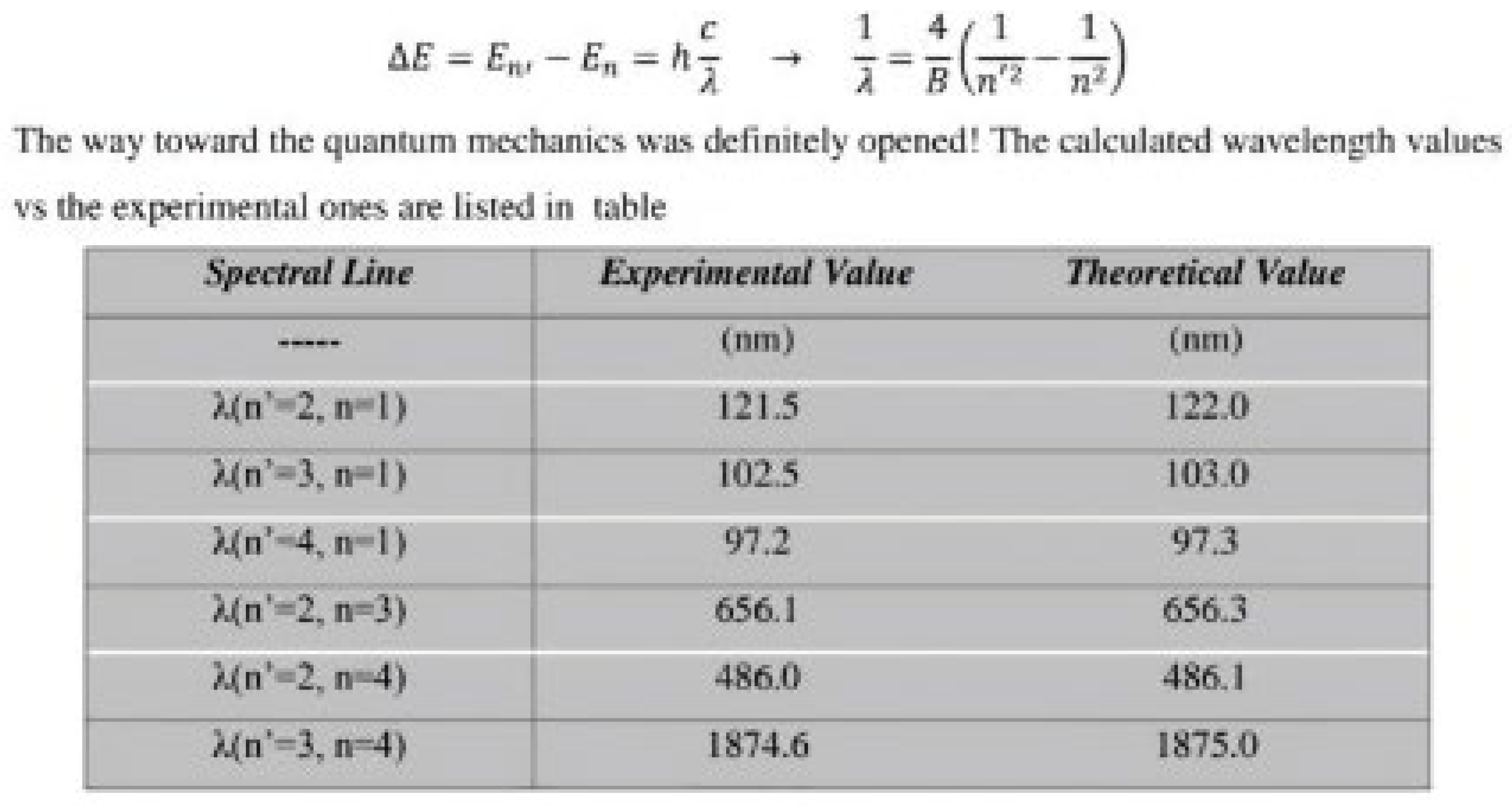
- Table 5 it represents the theoretical and experimental value of the hydrogen atom. Using the photon energy law mentioned above, this table.
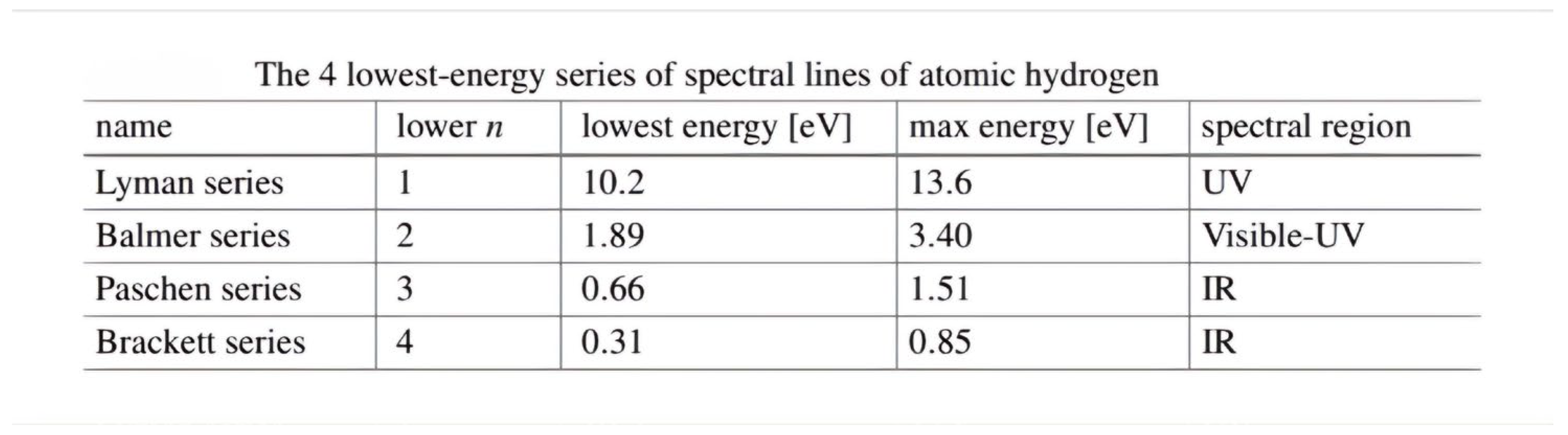 |
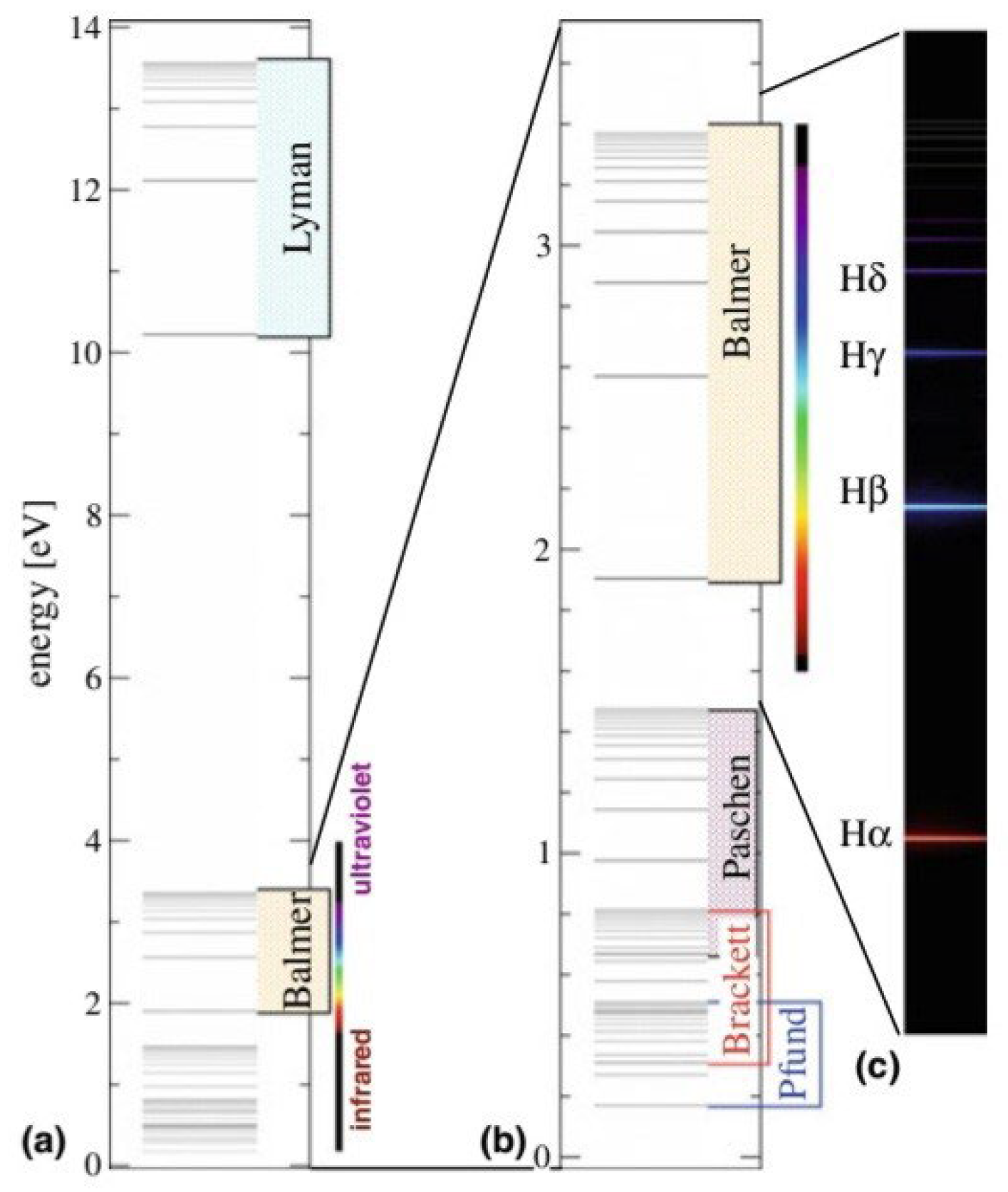
- This shape is a result of the fact that the electron, after a quantum leap occurred as a result of an interference between the orbital that it occupies and the energy level above it, was in an unstable state. Therefore, when the highest level of energy returns to its position, it releases energy in the form of spectral lines. These lines are determined according to the amount of energy, as shown in the picture.
5. Results Obtained
| Spectral Line | Energy | λ | λ | ||
|---|---|---|---|---|---|
| λ(n’=2, n=1) | 10.204269824 | eV | 121.50227268 | nm | 121.5 nm |
| λ(n’=3, n=1) | 12.093949421 | eV | 102.51754257 | nm | 102.5 nm |
| λ(n’=4, n=1) | 12.75533728 | eV | 97.20181814 | nm | 97.20 nm |
| λ(n’=3, n=2) | 1.8896795971 | eV | 656.11227245 | nm | 656.1 nm |
| λ(n’=4, n=2) | 2.5510674561 | eV | 486.0090907 | nm | 486.0 nm |
| λ(n’=4, n=3) | 0.66138785898 eV | 1874.6064927 | nm | 1874.6 nm | |
6. Conclusions
- 1)
- serving humanity in the advancement of scientific research.
- 2)
- using these equations to explore space and quantum world.
- 3)
- using these equations in developing communications machines.
References
- Svidzinsky, A.; Scully, M. Bohr's molecular model, a century later. Physics T 2014, 67, 33–39. [Google Scholar] [CrossRef]
- Udema, I. I. Renaissance of Bohr's model via derived alternative equation. American J. Mod. Phys 2017, 6, 23–31. [Google Scholar] [CrossRef]
- Nanni, L. The hydrogen atom: A review on the birth of modern quantum mechanics. Physics 2015. [CrossRef]
- Jordan, R. B. Principles of Inorganic Chemistry. Springer N, 2024; pp. 1–18. [Google Scholar] [CrossRef]
- Manini, N. Introduction to the physics of matter: basic atomic, molecular, and solid-state physics. Springer N, 2020; pp. 11–16. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




