Submitted:
28 October 2024
Posted:
29 October 2024
You are already at the latest version
Abstract
Keywords:
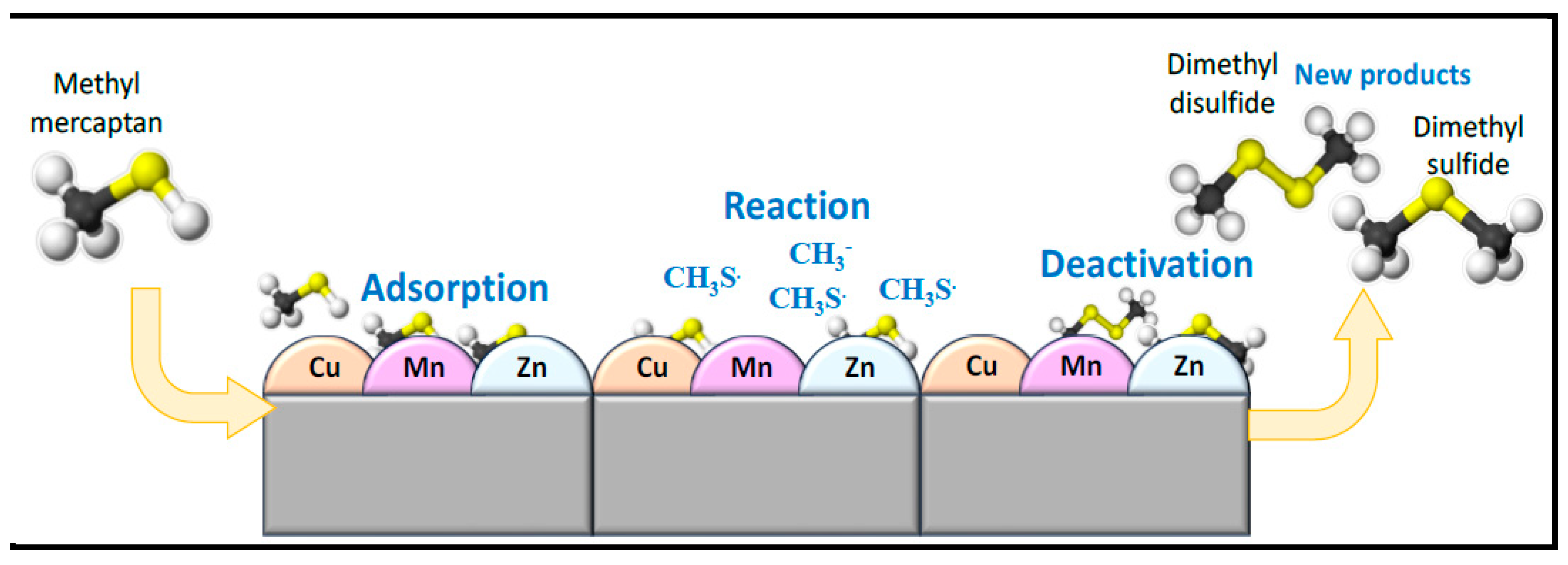
1. Introduction
2. Materials and Methods
2.1. Materials for Synthesis
2.1.1. Catalyst Preparation
2.1.2. System Calibration
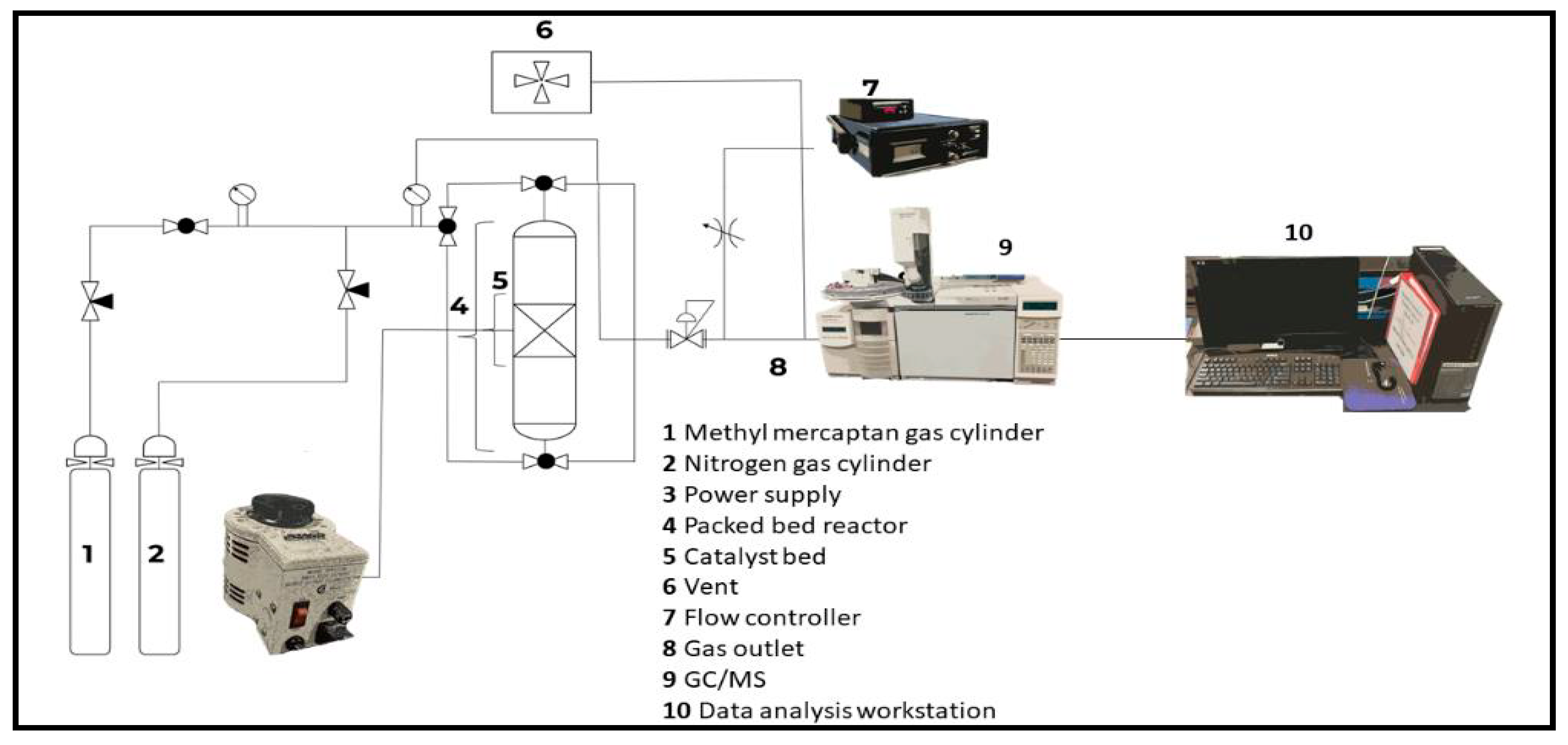
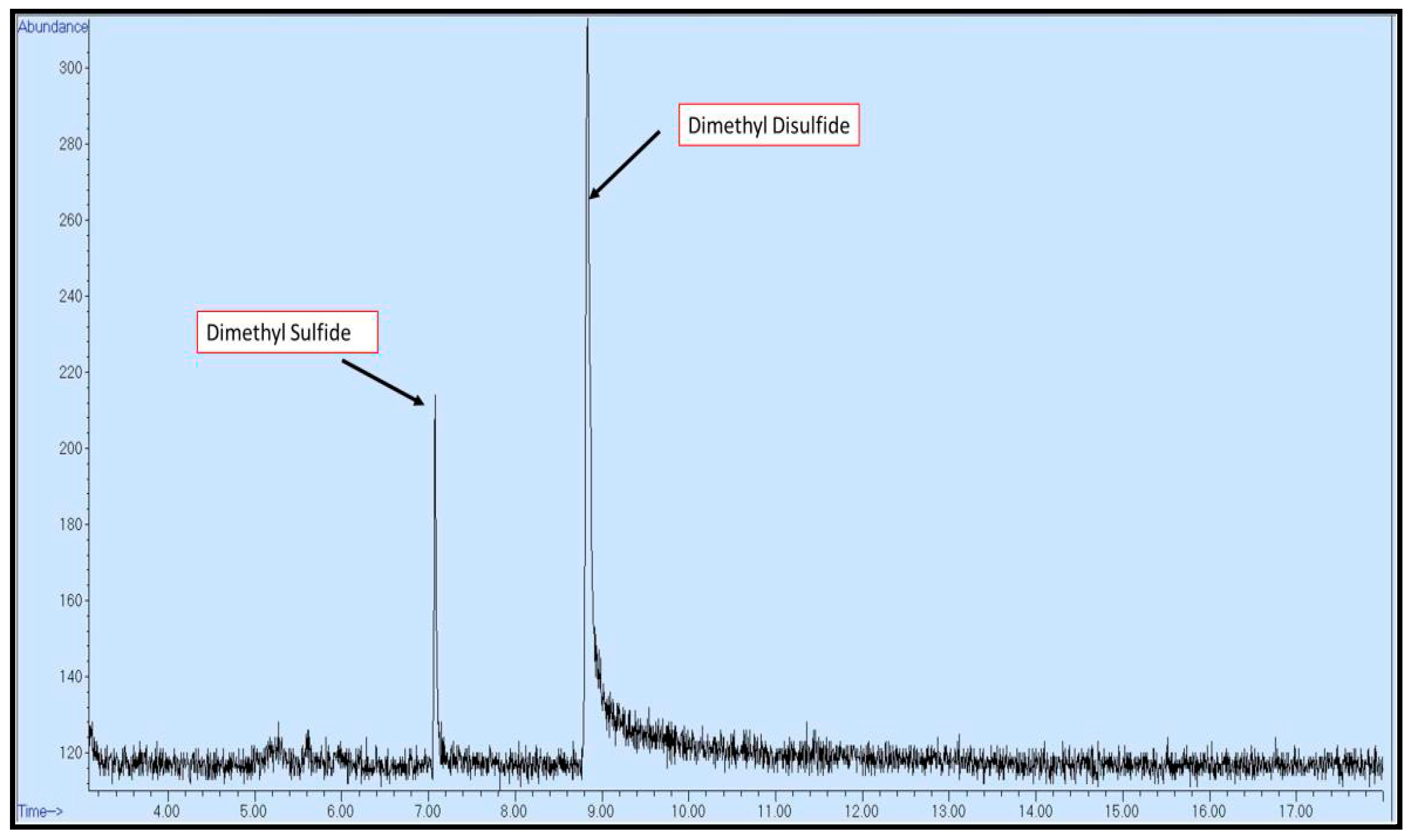
2.2. Methods and Procedure
3. Results and Discussions
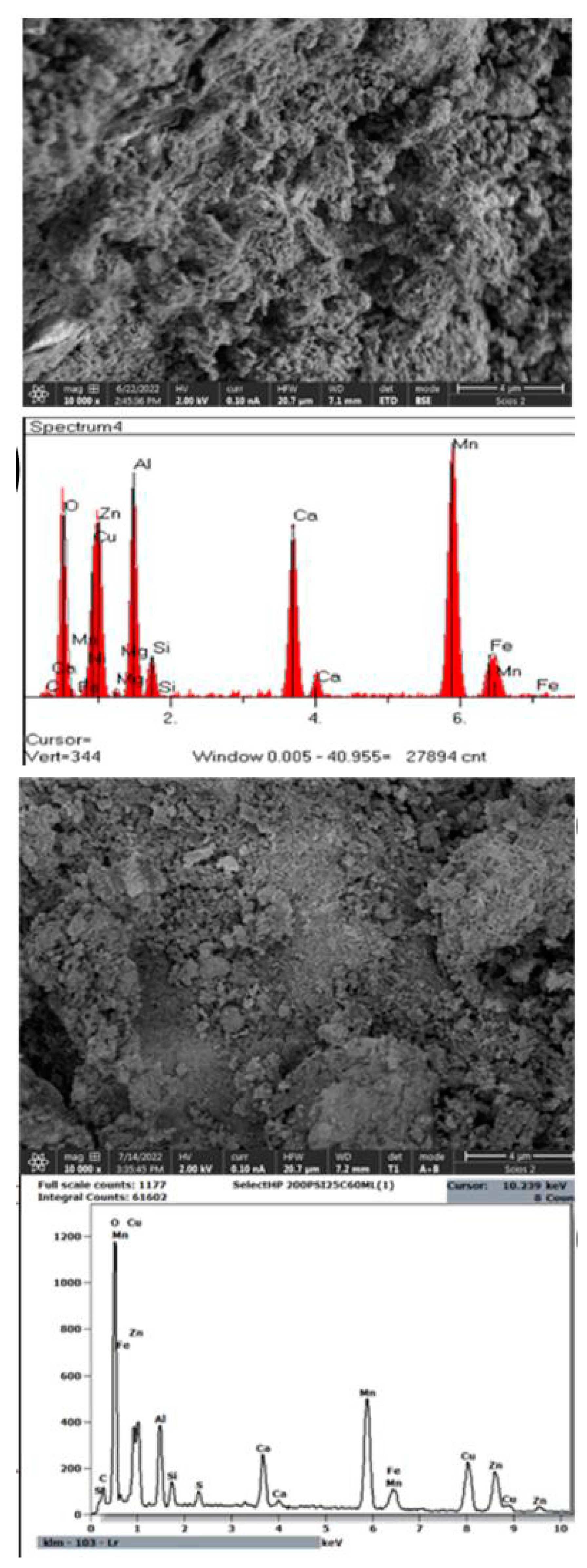
3.1. The Analysis of Surface Morphology
3.1.1. Commercial Catalysts
3.1.2. Synthesized Catalysts
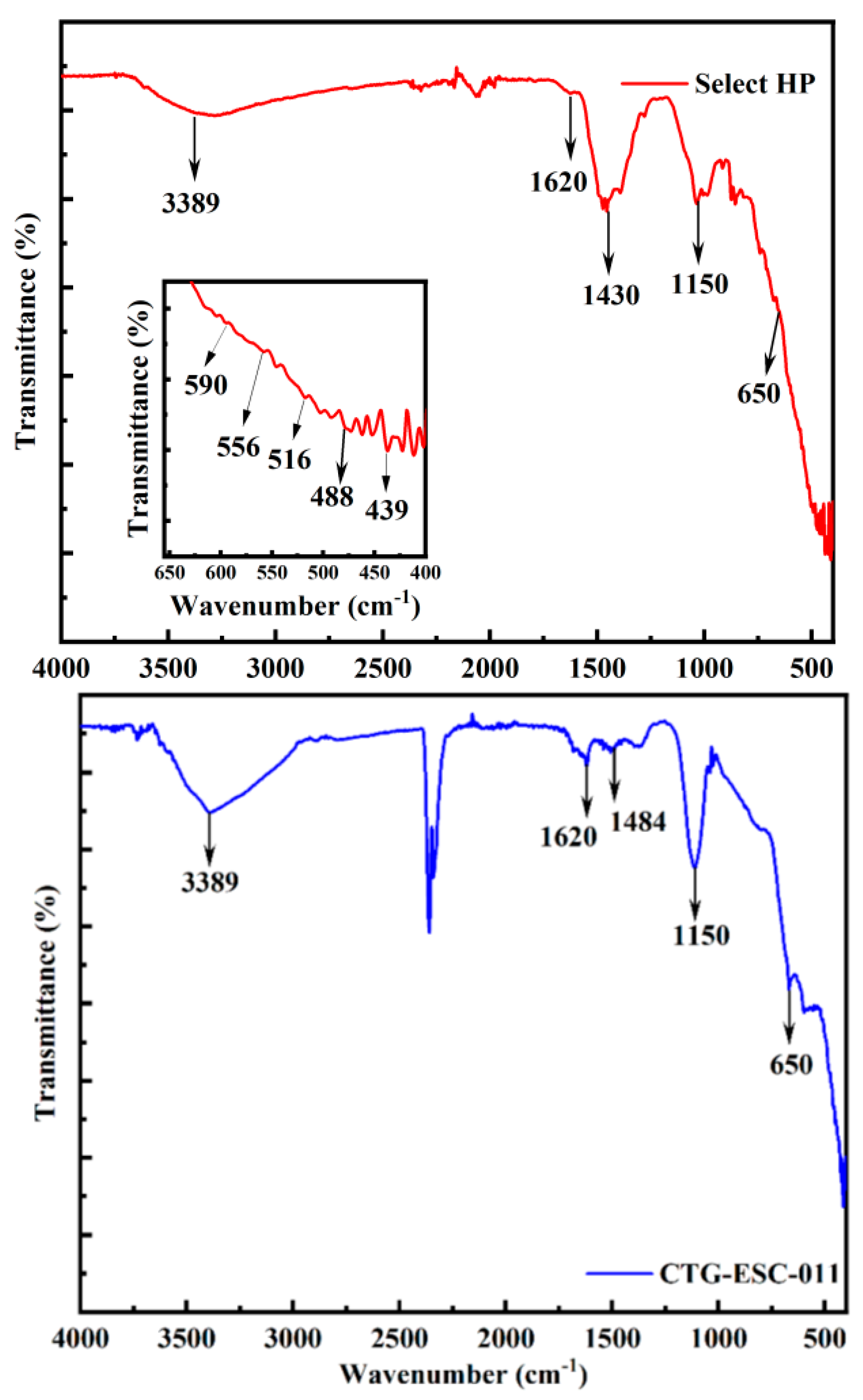
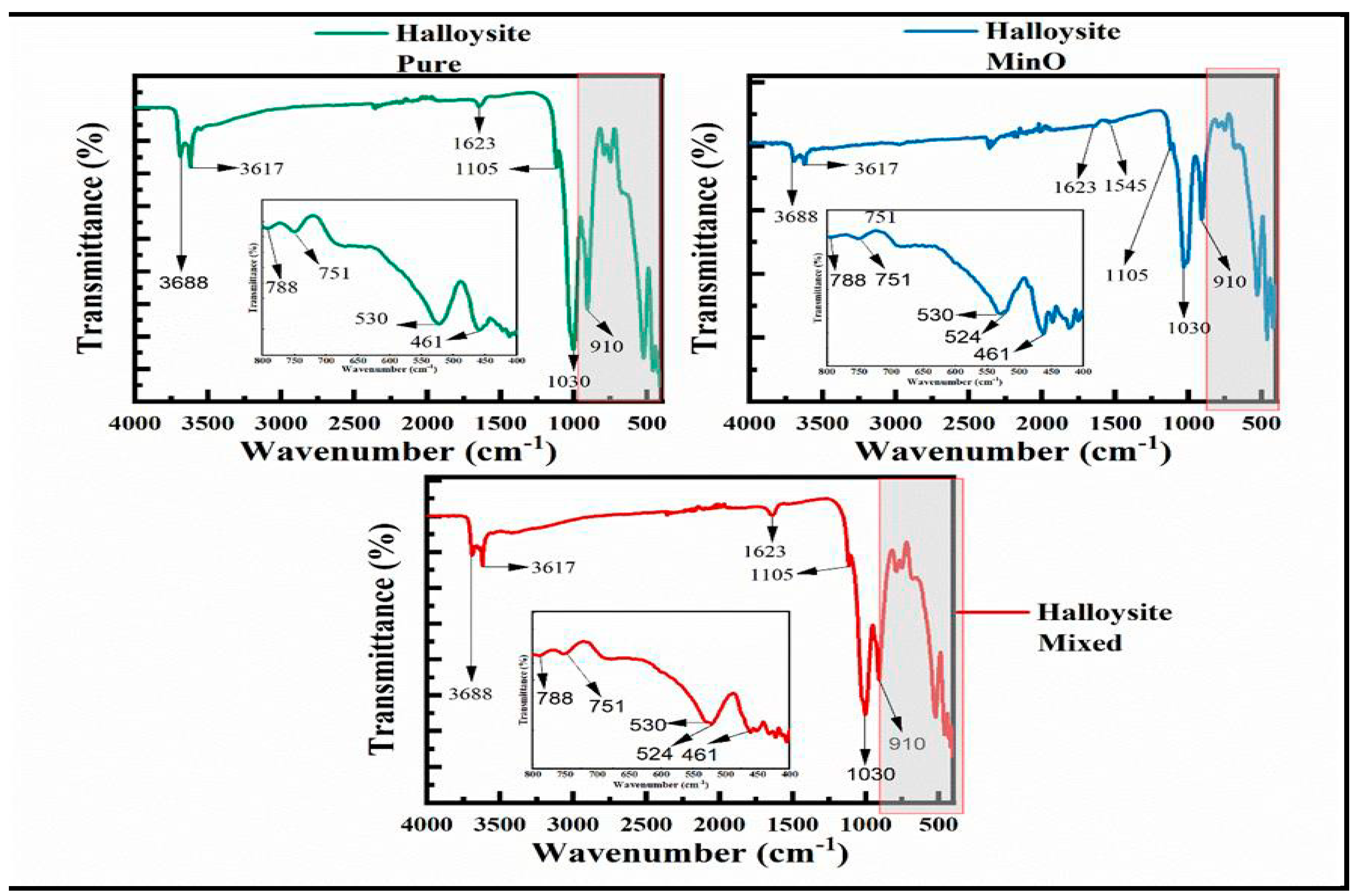
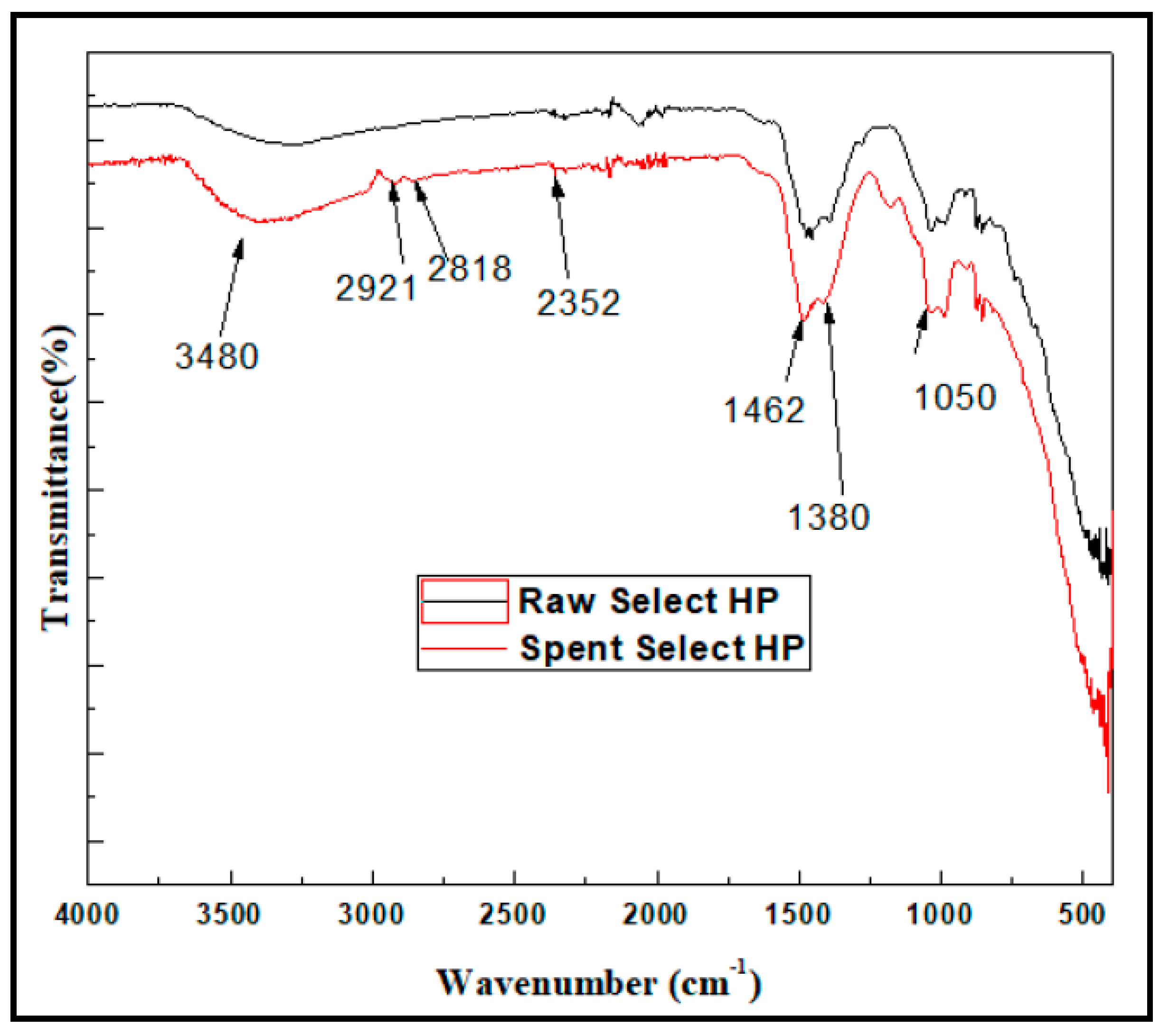
3.2. The Analysis of Functional Groups
3.3. The Analysis of Elemental Composition Verification
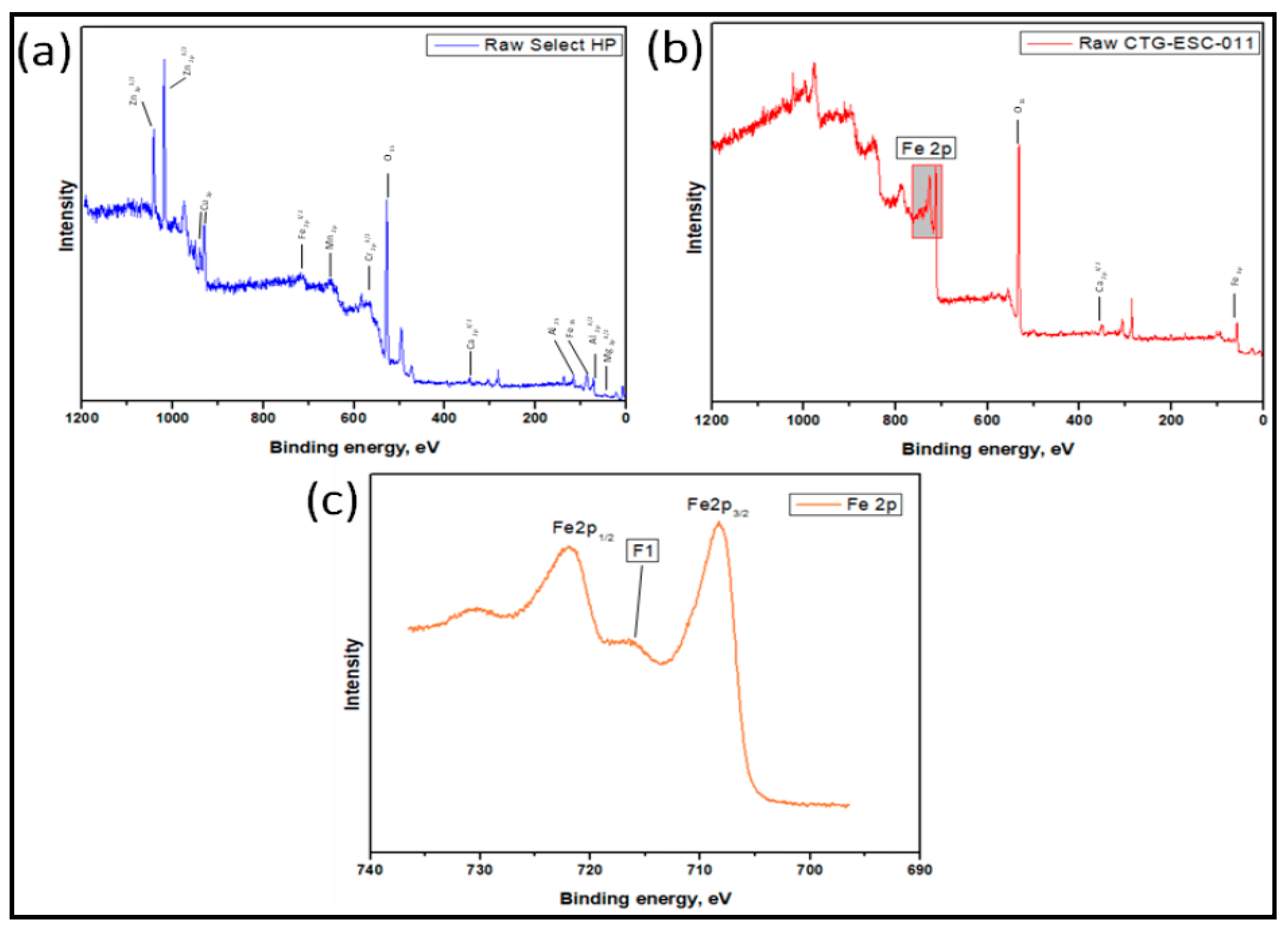
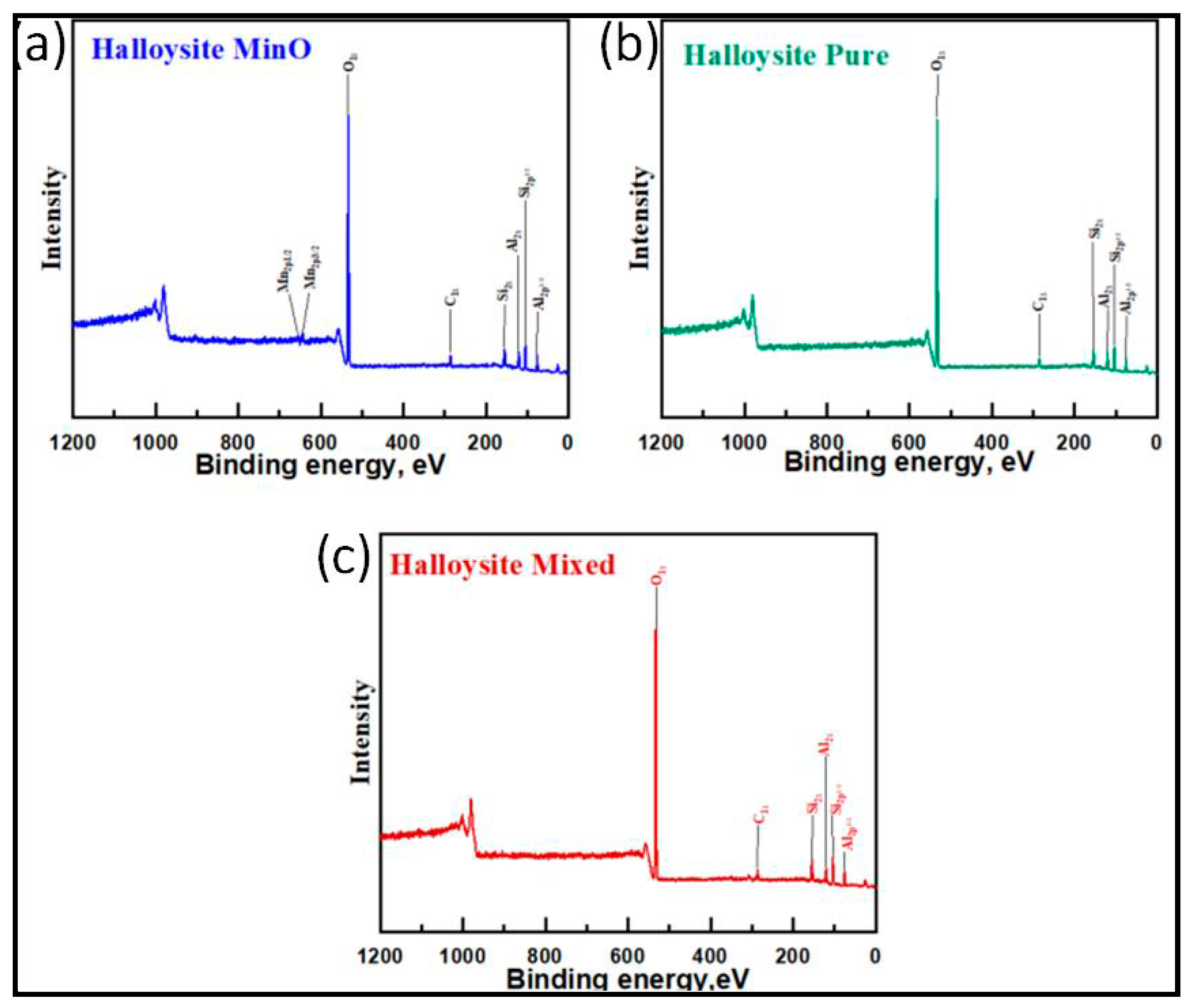
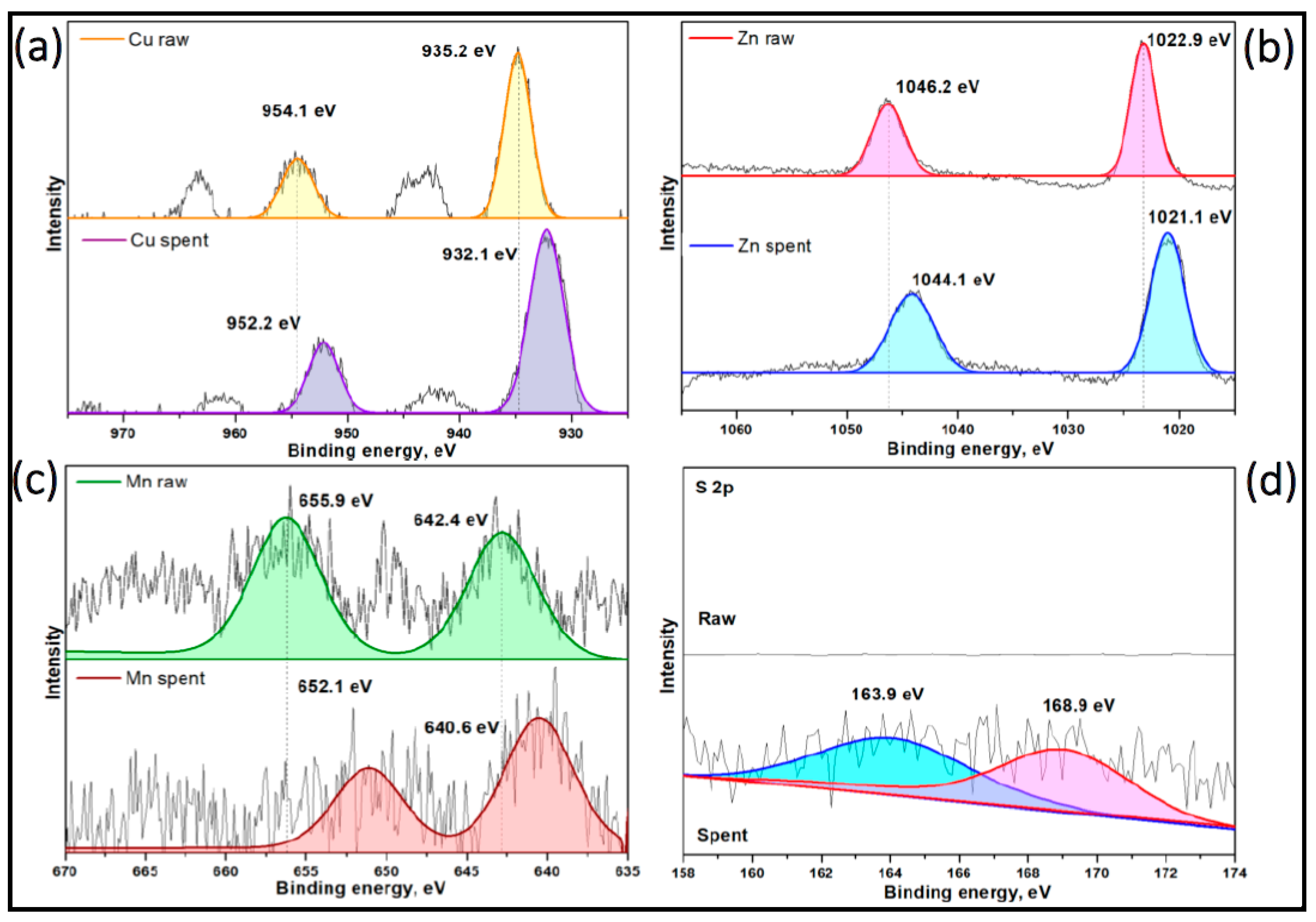
3.4. The Analysis of XPS Measurements
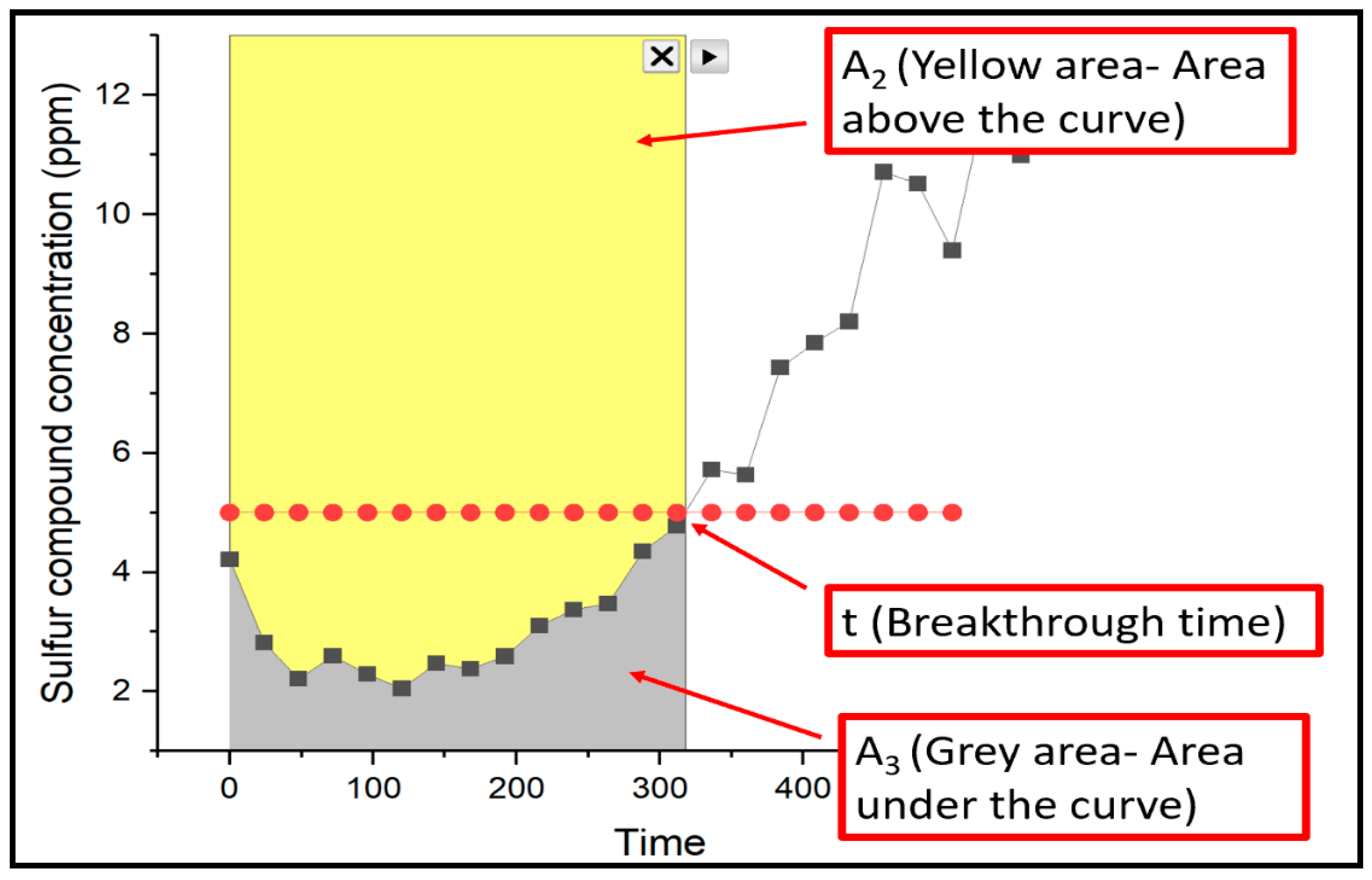
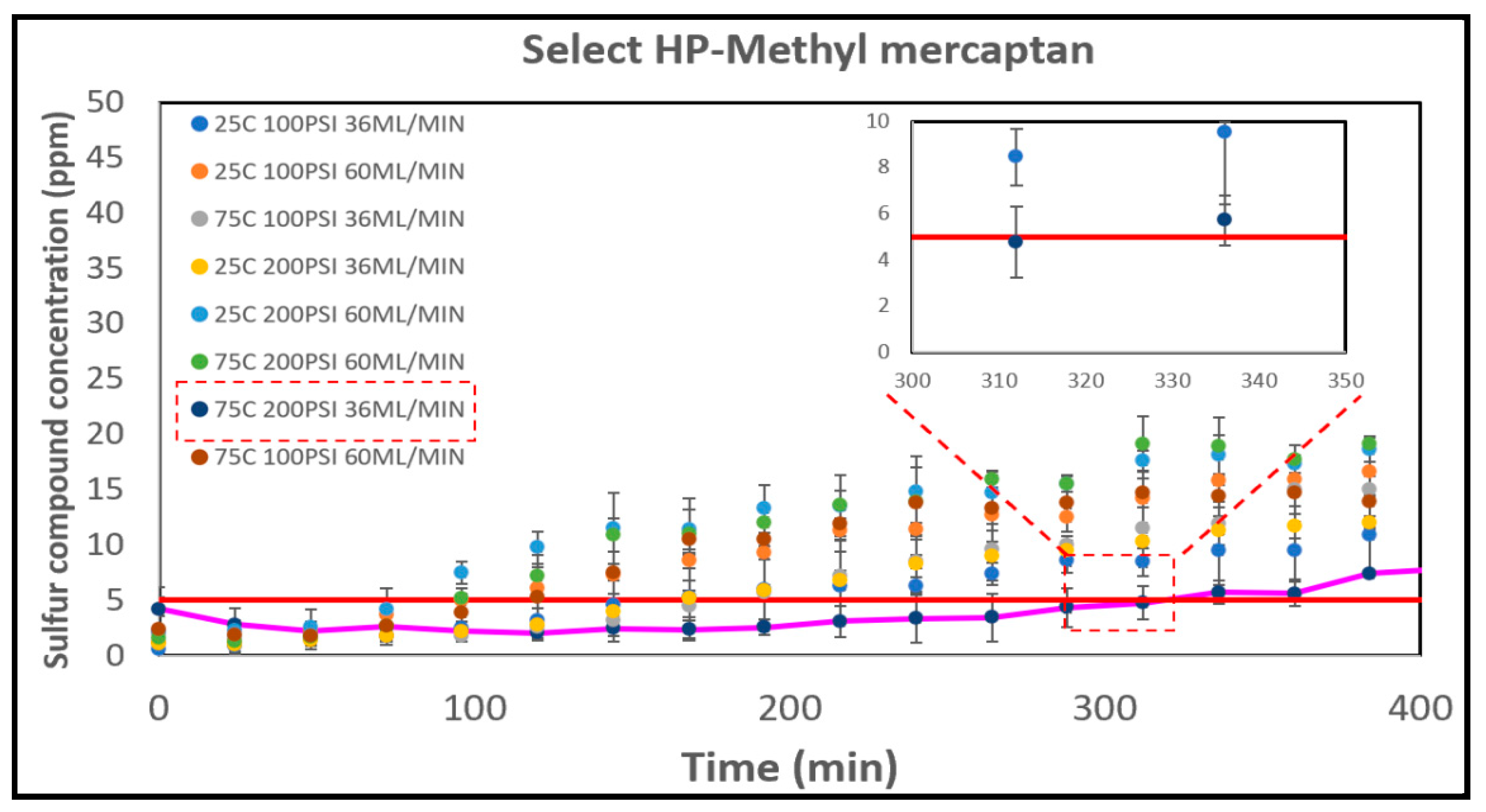
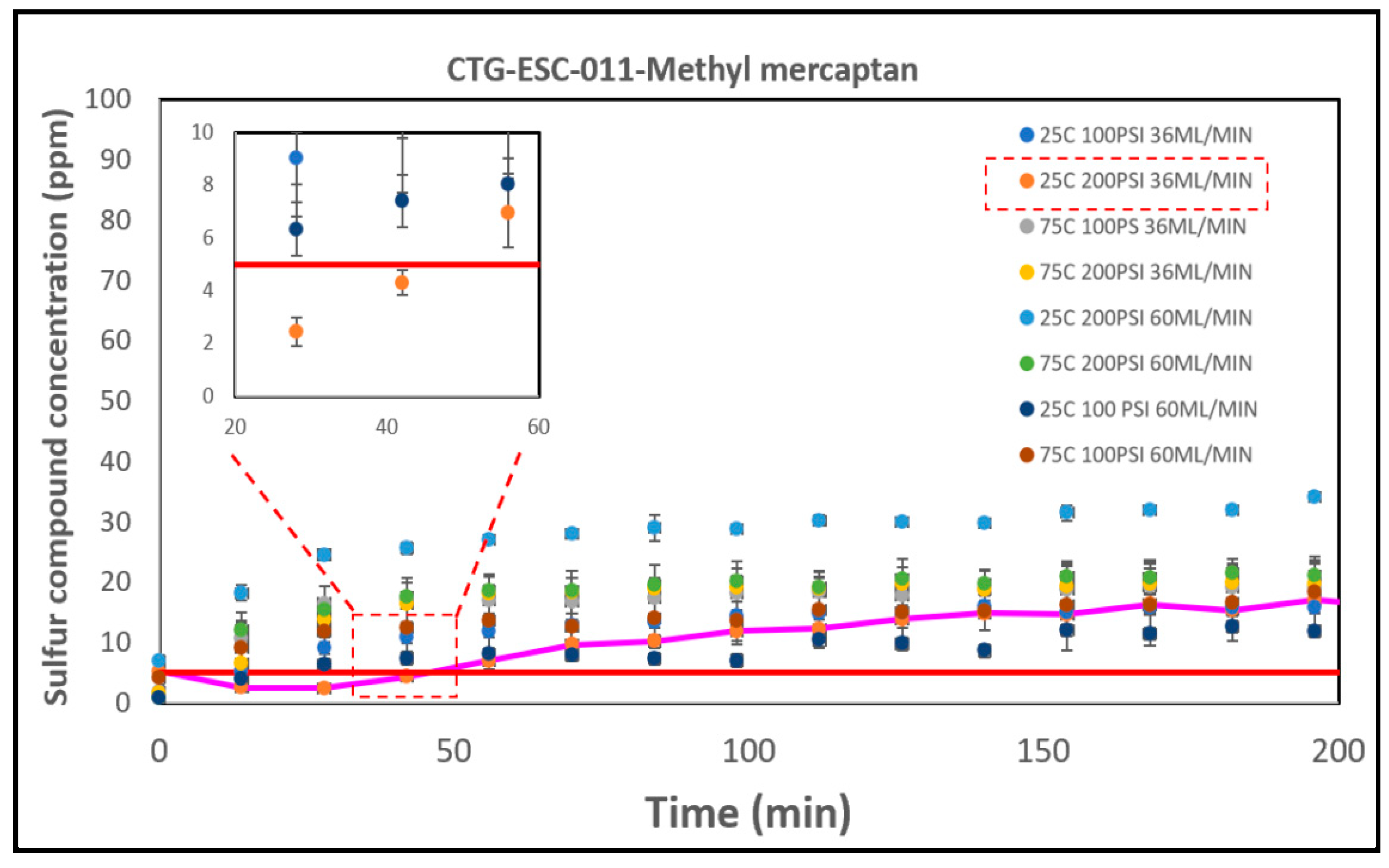
3.5. The Breakthrough Analysis – Commercial Catalysts
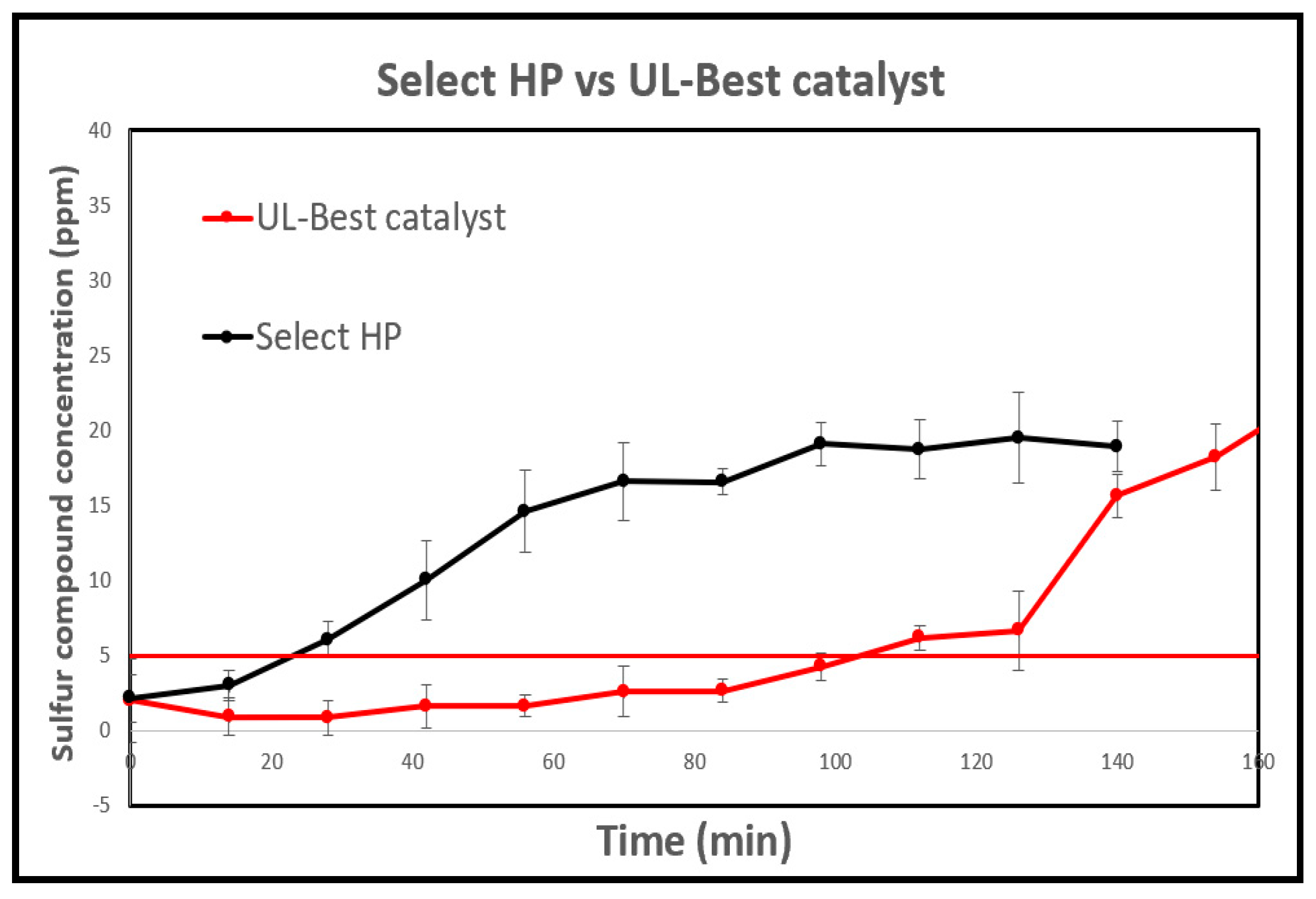
3.5.1. The Select HP
3.5.2. The CTG-ESC-011
3.5.3. The Halloysites
3.6. The Breakthrough Analysis– Novel Catalysts
3.7. The Performance Summary
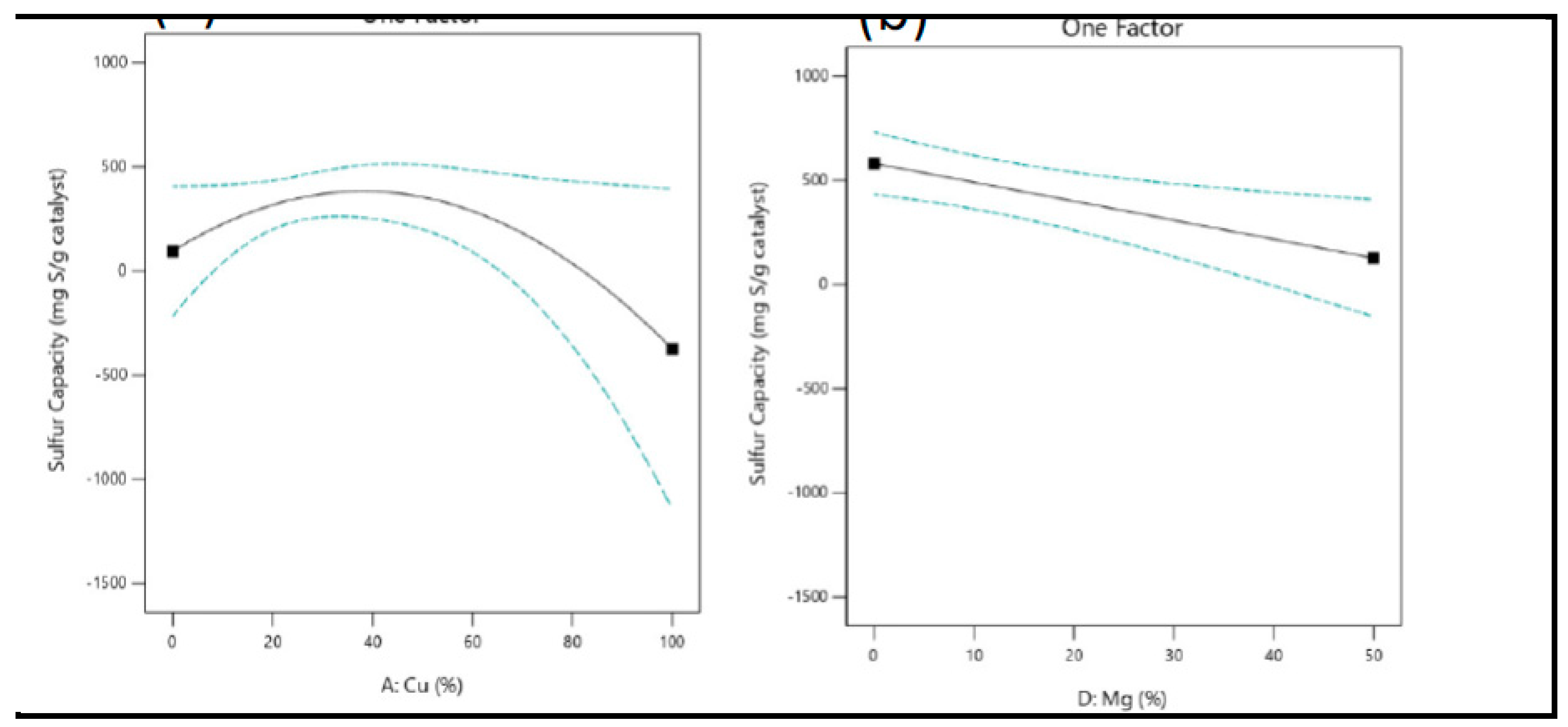
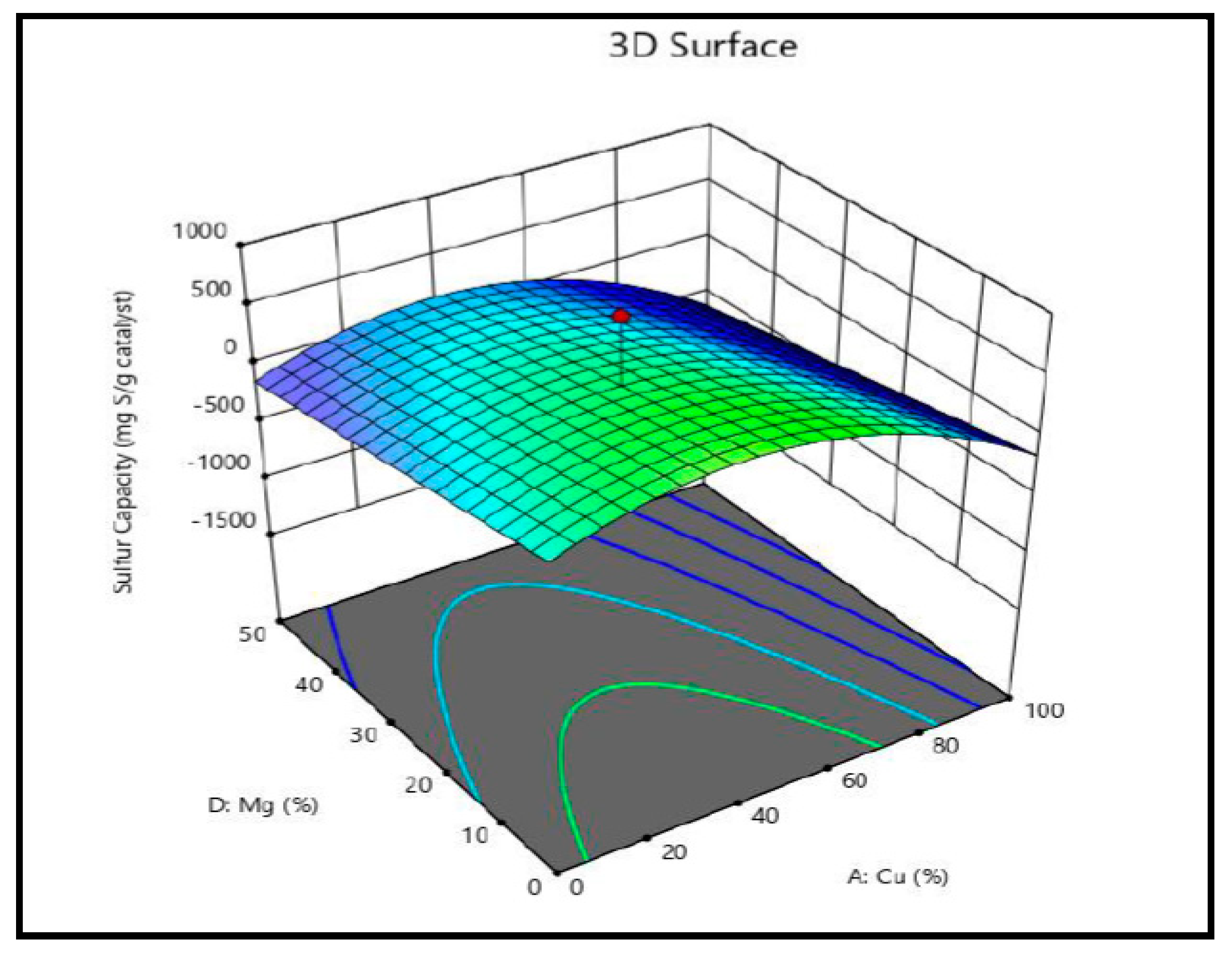
3.7.1. The Impact of Metal Composition on the Breakthrough Performance
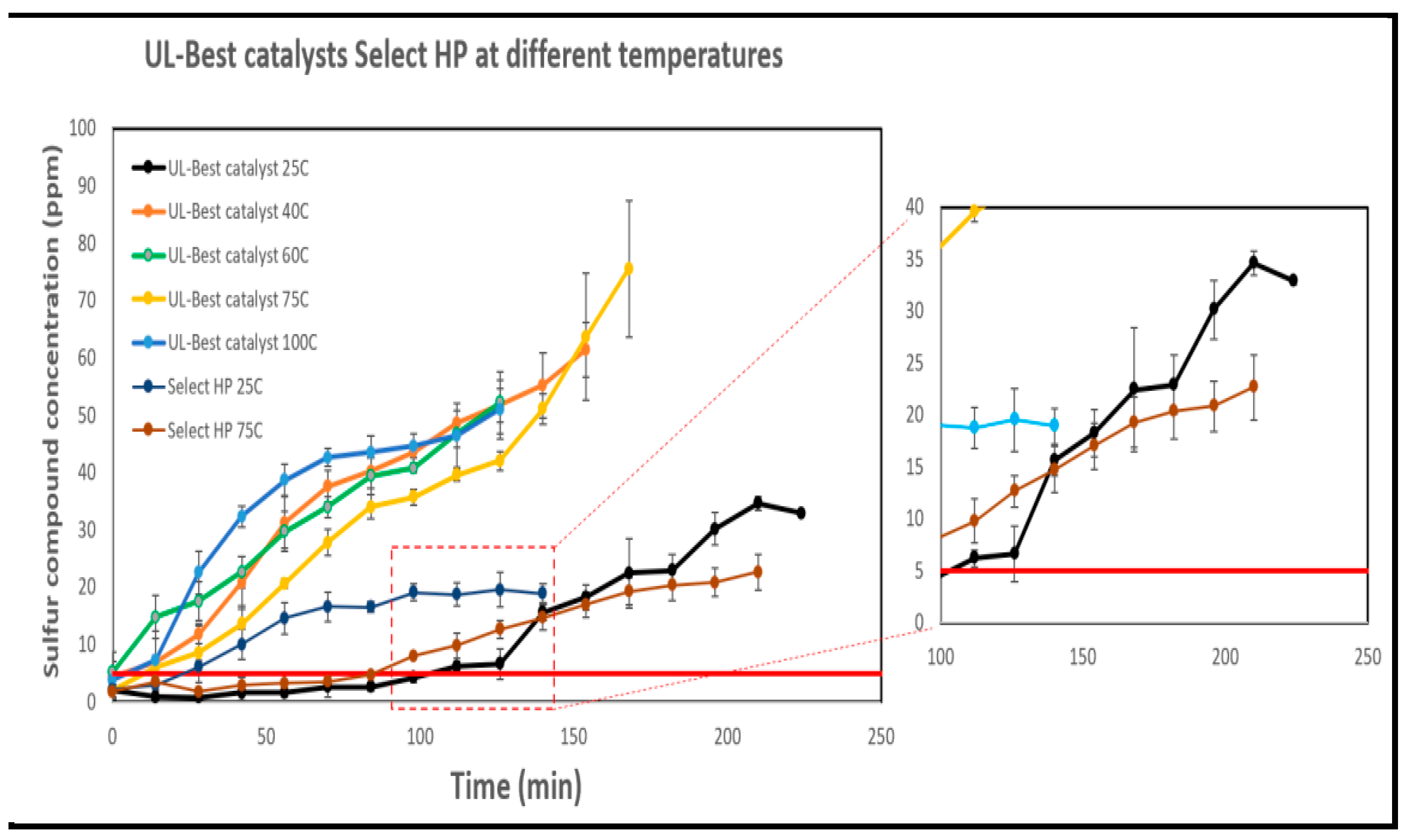
3.7.2. The Impact of Temperature on the Breakthrough Performance
3.7.3. General Comparison
4. Conclusions
Author Contributions
Funding
Acknowledgments
References
- Tim Gould, C. M. (2017). The environmental case for natural gas. International energy agency. https://www.iea.org/commentaries/the-environmental-case-for-natural-gas.
- Ratnasamy, C., Wagner, J. P., Spivey, S., & Weston, E. (2012). Removal of sulfur compounds from natural gas for fuel cell applications using a sequential bed system. Catalysis Today, 198(1), 233-238. [CrossRef]
- Tomei Torres, F. (2014). Addendum to the Toxicological Profile for Methyl Mercaptan. [CrossRef]
- Deshmukh, S., Jana, A., Bhattacharyya, N., Bandyopadhyay, R., & Pandey, R. A. (2014). Quantitative determination of pulp and paper industry emissions and associated odor intensity in methyl mercaptan equivalent using electronic nose. Atmospheric Environment, 82, 401-409. [CrossRef]
- Argyle, M. D., & Bartholomew, C. H. (2015). Heterogeneous Catalyst Deactivation and Regeneration: A Review. Catalysts, 5(1), 145-269.
- Luttrell, W. E., & Bobo, M. E. (2015). Methyl mercaptan. Journal of Chemical Health & Safety, 22(5), 37-39. [CrossRef]
- Yuan, Y., Gao, X., Zhou, J., Liu, G., Kuang, X., Yang, L., & Liao, R. (2022). A review: Research on corrosive sulphur in electrical power equipment. High Voltage, 7(2), 209-221. [CrossRef]
- Mahdipoor, H. R., & Dehghani Ashkezari, A. (2016). Feasibility study of a sulfur recovery unit containing mercaptans in lean acid gas feed. Journal of Natural Gas Science and Engineering, 31, 585-588. [CrossRef]
- Mokhatab, S., Poe, W. A., & Mak, J. Y. (2018). Handbook of natural gas transmission and processing: principles and practices. Gulf professional publishing.
- Tohidi, Z., Fatemi, S., & Qazvini, O. T. (2015). Mercaptan removal from natural gas by the efficient cyclic adsorption process; a simulation study. Journal of Natural Gas Science and Engineering, 26, 758-769.
- Gupta, A. K., Ibrahim, S., & Al Shoaibi, A. (2016). Advances in sulfur chemistry for treatment of acid gases. Progress in Energy and Combustion Science, 54, 65-92. [CrossRef]
- Ross, R. A., & Sood, S. P. (1977). Catalytic Oxidation of Methyl Mercaptan over Cobalt Molybdate. Product R&D, 16(2), 147-150. [CrossRef]
- Bashkova, S., Bagreev, A., & Bandosz, T. J. (2002). Adsorption of Methyl Mercaptan on Activated Carbons. Environmental Science & Technology, 36(12), 2777-2782. [CrossRef]
- Huguet, E., Coq, B., Durand, R., Leroi, C., Cadours, R., & Hulea, V. (2013). A highly efficient process for transforming methyl mercaptan into hydrocarbons and H2S on solid acid catalysts. Applied Catalysis B: Environmental, 134-135, 344-348. [CrossRef]
- Laosiripojana, N., & Assabumrungrat, S. (2011). Conversion of poisonous methanethiol to hydrogen-rich gas by chemisorption/reforming over nano-scale CeO2: The use of CeO2 as catalyst coating material. Applied Catalysis B: Environmental, 102(1), 267-275. [CrossRef]
- Cai, W., Lu, G., He, J., & Lan, Y. (2012). The adsorption feature and photocatalytic oxidation activity of K1−2xMxTiNbO5 (M=Mn, Ni) for methyl mercaptan in methane. Ceramics International, 38(4), 3167-3174. [CrossRef]
- Georgiadis, A. G., Charisiou, N. D., & Goula, M. A. (2020). Removal of Hydrogen Sulfide From Various Industrial Gases: A Review of The Most Promising Adsorbing Materials. Catalysts, 10(5).
- Thomas, B., & George, S. (2015). Production of Activated Carbon from Natural Sources. Trends in Green Chemistry, 1. [CrossRef]
- Xiao, Y., Wang, S., Wu, D., & Yuan, Q. (2008). Catalytic oxidation of hydrogen sulfide over unmodified and impregnated activated carbon. Separation and Purification Technology, 59(3), 326-332. [CrossRef]
- Bakshi H. V. (Jun, 2017). A Study on Synthesis of Synthetic Zeolites. Journal of Advances in Science and Technology, 14(1), 3. [CrossRef]
- Ozekmekci, M., Salkic, G., & Fellah, M. F. (2015). Use of zeolites for the removal of H2S: A mini-review. Fuel Processing Technology, 139, 49-60. [CrossRef]
- Vodyashkin, A. A., Sergorodceva, A. V., Kezimana, P., & Stanishevskiy, Y. M. (2023). Metal-Organic Framework (MOF)—A Universal Material for Biomedicine. International Journal of Molecular Sciences, 24(9).
- Yusuf, V. F., Malek, N. I., & Kailasa, S. K. (2022). Review on Metal–Organic Framework Classification, Synthetic Approaches, and Influencing Factors: Applications in Energy, Drug Delivery, and Wastewater Treatment. ACS Omega, 7(49), 44507-44531. [CrossRef]
- Jiao, L., Seow, J. Y. R., Skinner, W. S., Wang, Z. U., & Jiang, H.-L. (2019). Metal–organic frameworks: Structures and functional applications. Materials Today, 27, 43-68. [CrossRef]
- Gas, P.-Q. N. (2006). Natural gas processing: the crucial link between natural gas production and its transportation to market. Los Alamos, 20.
- He, H., Hu, L., Zeng, J., Huang, Y., He, C., Zhang, Q., Zhang, F., & Shu, D. (2020). Activation of persulfate by CuO-sludge-derived carbon dispersed on silicon carbide foams for odorous methyl mercaptan elimination: identification of reactive oxygen species. Environmental Science and Pollution Research, 27, 1224-1233.
- Saqer, S. M., Kondarides, D. I., & Verykios, X. E. (2011). Catalytic oxidation of toluene over binary mixtures of copper, manganese and cerium oxides supported on γ-Al2O3. Applied Catalysis B: Environmental, 103(3-4), 275-286.
- Su, H., Liu, J., Hu, Y., Ai, T., Gong, C., Lu, J., & Luo, Y. (2023). Comparative Study of α-and β-MnO2 on Methyl Mercaptan Decomposition: The Role of Oxygen Vacancies. Nanomaterials, 13(4), 775.
- Wen, Y., Zhao, S., Yi, H., Gao, F., Yu, Q., Liu, J., Tang, T., & Tang, X. (2022). Efficient catalytic oxidation of methyl mercaptan to sulfur dioxide with NiCuFe mixed metal oxides. Environmental Technology & Innovation, 26, 102252. [CrossRef]
- Yi, H., Tao, T., Zhao, S., Yu, Q., Gao, F., Zhou, Y., & Tang, X. (2020). Promoted adsorption of methyl mercaptan by γ -Al2O3 catalyst loaded with Cu/Mn. Environmental Technology & Innovation, 21, 101349. [CrossRef]
- Liu, W., Zhang, Y., Wang, S., Bai, L., Deng, Y., & Tao, J. (2021). Effect of Pore Size Distribution and Amination on Adsorption Capacities of Polymeric Adsorbents. Molecules, 26(17), 5267. https://www.mdpi.com/1420-3049/26/17/5267.
- Zhang, Y., Li, K., Sun, X., Song, X., Wang, F., Wang, C., Ning, P., & He, H. (2021). The role of H2O in the removal of methane mercaptan (CH3SH) on Cu/C-PAN catalyst. Applied Surface Science, 567, 150851. [CrossRef]
- Bashkova, S., Bagreev, A., & Bandosz, T. J. (2005). Catalytic properties of activated carbon surface in the process of adsorption/oxidation of methyl mercaptan. Catalysis Today, 99(3), 323-328. [CrossRef]
- Ma, X., Peng, S., Li, W., Liu, H., & Chen, Y. (2018). Efficient removal of low concentration methyl mercaptan by HKUST-1 membrane constructed on porous alumina granules. CrystEngComm, 20. [CrossRef]
- Juma, A. O., Arbab, E. A. A., Muiva, C. M., Lepodise, L. M., & Mola, G. T. (2017). Synthesis and characterization of CuO-NiO-ZnO mixed metal oxide nanocomposite. Journal of Alloys and Compounds, 723, 866-872. [CrossRef]
- Habibi, M. H., & Karimi, B. (2014). Application of impregnation combustion method for fabrication of nanostructure CuO/ZnO composite oxide: XRD, FESEM, DRS and FTIR study. Journal of Industrial and Engineering Chemistry, 20(4), 1566-1570. [CrossRef]
- Kang, L., Zhang, M., Liu, Z.-H., & Ooi, K. (2007). IR spectra of manganese oxides with either layered or tunnel structures. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 67(3), 864-869. [CrossRef]
- Wang, X., Jia, J., Zhao, L., & Sun, T. (2008). Mesoporous SBA-15 Supported Iron Oxide: A Potent Catalyst for Hydrogen Sulfide Removal. Water, Air, and Soil Pollution, 193(1), 247-257. [CrossRef]
- Zhao, J., Lin, Z., Zhang, J., Wu, J., Yue, Y., & Qian, G. (2022). Enhanced removal of low-concentration methyl mercaptan by synergetic effect between surface functional group and metallic site. Surfaces and Interfaces, 28, 101663. [CrossRef]
- Namduri, H., & Nasrazadani, S. (2008). Quantitative analysis of iron oxides using Fourier transform infrared spectrophotometry. Corrosion Science, 50(9), 2493-2497. [CrossRef]
- Xu, D., Jin, T., Song, L., Zhu, N., Han, L., & Hong, H. (2023). Synthesis of stable calcium carbonate nanoparticles for pH-responsive controlled drug release. Materials Letters, 333, 133635. [CrossRef]
- Zhang, Y., Ouyang, J., & Yang, H. (2014). Metal oxide nanoparticles deposited onto carbon-coated halloysite nanotubes. Applied Clay Science, 95, 252-259. [CrossRef]
- Szczepanik, B., Słomkiewicz, P., Garnuszek, M., Czech, K., Banaś, D., Kubala-Kukuś, A., & Stabrawa, I. (2015). The effect of chemical modification on the physico-chemical characteristics of halloysite: FTIR, XRF, and XRD studies. Journal of Molecular Structure, 1084, 16-22. [CrossRef]
- Bordeepong, S., Bhongsuwan, D., & Pungrassami, T. (2011). Characterization of halloysite from Thung Yai District, Nakhon Si Thammarat Province, in Southern Thailand.
- Kouser, S., Prabhu, A., Sheik, S., Prashantha, K., Nagaraja, G. K., Neetha D'souza, J., Navada, K. M., & Manasa, D. J. (2021). Chitosan functionalized halloysite nanotube/poly (caprolactone) nanocomposites for wound healing application. Applied Surface Science Advances, 6, 100158. [CrossRef]
- Bayiha, B. N., Billong, N., Yamb, E., Kaze, R. C., & Nzengwa, R. (2019). Effect of limestone dosages on some properties of geopolymer from thermally activated halloysite. Construction and Building Materials, 217, 28-35. [CrossRef]
- Kesavan, G., & Chen, S. M. (2021). Manganese oxide anchored on carbon modified halloysite nanotubes: An electrochemical platform for the determination of chloramphenicol. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 615, 126243. [CrossRef]
- Peña, D. A., Uphade, B. S., & Smirniotis, P. G. (2004). TiO2-supported metal oxide catalysts for low-temperature selective catalytic reduction of NO with NH3: I. Evaluation and characterization of first row transition metals. Journal of Catalysis, 221(2), 421-431. [CrossRef]
- Stranick, M. A. (1999). MnO2 by XPS. Surface Science Spectra, 6(1), 31-38. [CrossRef]
- Park, J. E., Jo, E. S., Lee, G. B., Lee, S. E., & Hong, B.-U. (2023). Adsorption Capacity and Desorption Efficiency of Activated Carbon for Odors from Medical Waste. Molecules, 28(2), 785. https://www.mdpi.com/1420-3049/28/2/785.
- Hadnadjev, M., Vulic, T., Marinkovic-Neducin, R., Suchorski, Y., & Weiss, H. (2008). The iron oxidation state in Mg–Al–Fe mixed oxides derived from layered double hydroxides: An XPS study. Applied Surface Science, 254(14), 4297-4302. [CrossRef]
- Smith, R. D. L., Prévot, M. S., Fagan, R. D., Trudel, S., & Berlinguette, C. P. (2013). Water Oxidation Catalysis: Electrocatalytic Response to Metal Stoichiometry in Amorphous Metal Oxide Films Containing Iron, Cobalt, and Nickel. Journal of the American Chemical Society, 135(31), 11580-11586. [CrossRef]
- Liu, S., Ay, A., Luo, Q., Hu, X., Białas, K., Dutta, G., Moschou, D., & Regoutz, A. (2022). Oxidation of copper electrodes on flexible polyimide substrates for non-enzymatic glucose sensing. Materials Research Express, 9(4), 045010. [CrossRef]
- Velu, S., Suzuki, K., Munusamy, V., Barman, S., & Gopinath, C. (2005). In situ XPS investigations of Cu1-xNixZnAl-mixed metal oxide catalysts used in the oxidative steam reforming of bio-ethanol. Applied Catalysis B: Environmental, 55, 287–299. [CrossRef]
- Al-Gaashani, R., Zakaria, Y., Gladich, I., Kochkodan, V., & Lawler, J. (2022). XPS, structural and antimicrobial studies of novel functionalized halloysite nanotubes. Scientific Reports, 12(1), 21633. [CrossRef]
- Sun, P., Liu, G., Lv, D., Dong, X., Wu, J., & Wang, D. (2015). Effective activation of halloysite nanotubes by piranha solution for amine modification via silane coupling chemistry [10.1039/C5RA04444H]. RSC Advances, 5(65), 52916-52925. [CrossRef]
- Liu, M., Guo, B., Du, M., Cai, X., & Jia, D. (2007). Properties of halloysite nanotube–epoxy resin hybrids and the interfacial reactions in the systems. Nanotechnology, 18, 455703. [CrossRef]
- Kloprogge, J. T., & Wood, B. J. (2015). CHEMICAL BONDING AND ELECTRONIC STRUCTURES OF THE Al_2Si_2O_5(OH)_4 POLYMORPHS KAOLINITE, DICKITE, NACRITE, AND HALLOYSITE BY X-RAY PHOTOELECTRON PECTROSCOPY. Clay Science, 19(2), 39-44. [CrossRef]
- Mokhatab, S., Poe, W. A., & Mak, J. Y. (2019). Chapter 4 - Basic Concepts of Natural Gas Processing. In S. Mokhatab, W. A. Poe, & J. Y. Mak (Eds.), Handbook of Natural Gas Transmission and Processing (Fourth Edition) (pp. 177-189). Gulf Professional Publishing. [CrossRef]
- Dhage, P., Samokhvalov, A., McKee, M., Duin, E., & Tatarchuk, B. (2013). Reactive adsorption of hydrogen sulfide by promoted sorbents Cu-ZnO/SiO2: Active sites by experiment and simulation. Surface and Interface Analysis, 45. [CrossRef]
- Alguacil, F. J. (2023). Recent Advances in H2S Removal from Gas Streams. Applied Sciences, 13(5), 3217. https://www.mdpi.com/2076-3417/13/5/3217.
- Bergel, M., & Tierno, I. (2009). Sweetening technologies-A look at the whole picture. 24th World Gas Conf. Argentina,.
- Khabazipour, M., & Anbia, M. (2019). Removal of Hydrogen Sulfide from Gas Streams Using Porous Materials: A Review. Industrial & Engineering Chemistry Research, 58(49), 22133-22164. [CrossRef]
- Xie, Y., Bao, J., Song, X., Sun, X., Ning, P., Wang, C., Wang, F., Ma, Y., Fan, M., & Li, K. (2023). Catalysts for gaseous organic sulfur removal. Journal of Hazardous Materials, 442, 130029. [CrossRef]
- Jiang, D., Su, L., Ma, L., Yao, N., Xu, X., Tang, H., & Li, X. (2010). Cu–Zn–Al mixed metal oxides derived from hydroxycarbonate precursors for H2S removal at low temperature. Applied Surface Science, 256(10), 3216-3223. [CrossRef]
- Nabipoor H. M., Hallajisani, A. (2020). Application of Molybdenum oxide nanoparticles in H2S removal from natural gas under different operational and geometrical conditions. Journal of Petroleum Science and Engineering, 190, 107131. [CrossRef]
- Weinlaender, C., Neubauer, R., & Hochenauer, C. (2016). Low-temperature H2S removal for solid oxide fuel cell application with metal oxide adsorbents. Adsorption Science & Technology, 35(1-2), 120-136. [CrossRef]
- Li, S., Wang, D., Wu, X., & Chen, Y. (2020). Recent advance on VOCs oxidation over layered double hydroxides derived mixed metal oxides. Chinese Journal of Catalysis, 41(4), 550-560. [CrossRef]
- Wang, L., Zhang, X., He, J., Zhu, J., & Hu, L. (2022). The removal of ethyl mercaptan by Fe2O3/HNb3O8-NS composite. Inorganic Chemistry Communications, 140, 109440. [CrossRef]
- Zhang, Z., Jiang, Z., & Shangguan, W. (2016). Low-temperature catalysis for VOCs removal in technology and application: A state-of-the-art review. Catalysis Today, 264, 270-278. [CrossRef]
- Liu, Q., Ke, M., Yu, P., Liu, F., Hu, H., & Li, C. (2018). High performance removal of methyl mercaptan on metal modified activated carbon. Korean Journal of Chemical Engineering, 35(1), 137-146. [CrossRef]
- West, J., Peter Williams, B., Young, N., Rhodes, C., & Hutchings, G. J. (2001). Ni- and Zn-promotion of γ-Al2O3 for the hydrolysis of COS under mild conditions. Catalysis Communications, 2(3), 135-138. [CrossRef]
- Yi, H., Zhang, X., Tang, X., Zhao, S., Ma, C., Han, W., & Song, L. (2019). Promotional Effects of Transition Metal Modification over Al2O3 for CH3SH Catalytic Oxidation. ChemistrySelect, 4(34), 9901-9907. [CrossRef]
- Li, Y., Wang, L.-J., Fan, H.-L., Shangguan, J., Wang, H., & Mi, J. (2015). Removal of Sulfur Compounds by a Copper-Based Metal Organic Framework under Ambient Conditions. Energy & Fuels, 29(1), 298-304. [CrossRef]
- Singh, A., Krishna, V., Angerhofer, A., Do, B., MacDonald, G., & Moudgil, B. (2010). Copper Coated Silica Nanoparticles for Odor Removal. Langmuir, 26(20), 15837-15844. [CrossRef]
- Liu, Q., Ke, M., Liu, F., Yu, P., Hu, H., & Li, C. (2017). High-performance removal of methyl mercaptan by nitrogen-rich coconut shell activated carbon [10.1039/C7RA03227G]. RSC Advances, 7(37), 22892-22899. [CrossRef]
- Zhao, S., Yi, H., Tang, X., Gao, F., Zhang, B., Wang, Z., & Zuo, Y. (2015). Methyl mercaptan removal from gas streams using metal-modified activated carbon. Journal of Cleaner Production, 87, 856-861. [CrossRef]
- Feng, Q., & Wen, S. (2017). Formation of zinc sulfide species on smithsonite surfaces and its response to flotation performance. Journal of Alloys and Compounds, 709, 602-608. [CrossRef]
- Su, H., Liu, J., Hu, Y., Ai, T., Gong, C., Lu, J., & Luo, Y. (2023). Comparative Study of α-and β-MnO2 on Methyl Mercaptan Decomposition: The Role of Oxygen Vacancies. Nanomaterials, 13(4), 775.
- Yao, X., Zhou, J., & Liu, Z. (2022). Study on adsorption of low-concentration methyl mercaptan by starch-based activated carbon. Chemosphere, 302, 134901. [CrossRef]
- Soni Castro, P., Martinez Zuniga, G., Holmes, W., Buchireddy, P. R., Gang, D. D., Revellame, E., Zappi, M., & Hernandez, R. (2023). Review of the adsorbents/catalysts for the removal of sulfur compounds from natural gas. Gas Science and Engineering, 115, 205004. [CrossRef]
- Wei, G., Liu, P., Chen, D., Chen, T., Liang, X., & Chen, H. (2019). Activity of manganese oxides supported on halloysite towards the thermal catalytic oxidation of formaldehyde: Constraint from the manganese precursor. Applied Clay Science, 182, 105280. [CrossRef]
- Kim, D. J., Lee, H. I., Yie, J. E., Kim, S.-J., & Kim, J. M. (2005). Ordered mesoporous carbons: Implication of surface chemistry, pore structure and adsorption of methyl mercaptan. Carbon, 43(9), 1868-1873. [CrossRef]
- Shakirullah, M., Ahmad, W., Ahmad, I., & Ishaq, M. (2010). Oxidative desulphurization study of gasoline and kerosene: Role of some organic and inorganic oxidants. Fuel Processing Technology, 91(11), 1736-1741. [CrossRef]
- Yi, D., Huang, H., Meng, X., & Shi, L. (2014). Adsorption–desorption behavior and mechanism of dimethyl disulfide in liquid hydrocarbon streams on modified Y zeolites. Applied Catalysis B: Environmental, 148-149, 377-386. [CrossRef]
- Tamai, H., Nakamori, M., Nishikawa, M., & Shiono, T. (2011). Activated Carbons Containing Dispersed Metal Oxide Particles for Removal of Methyl Mercaptan in Air. Materials Sciences and Applications, Vol.02No.01, 4, Article 3650. [CrossRef]
- Wang, L., Wang, X., Ning, P., Liu, W., Wang, F., & Ma, Y. (2017). Selective adsorption of CH3SH on cobalt-modified activated carbon with low oxygen concentration. Journal of the Taiwan Institute of Chemical Engineers, 75, 156-163. [CrossRef]
- Kim, Y.-H., Tuan, V. A., Park, M.-K., & Lee, C.-H. (2014). Sulfur removal from municipal gas using magnesium oxides and a magnesium oxide/silicon dioxide composite. Microporous and Mesoporous Materials, 197, 299-307. [CrossRef]
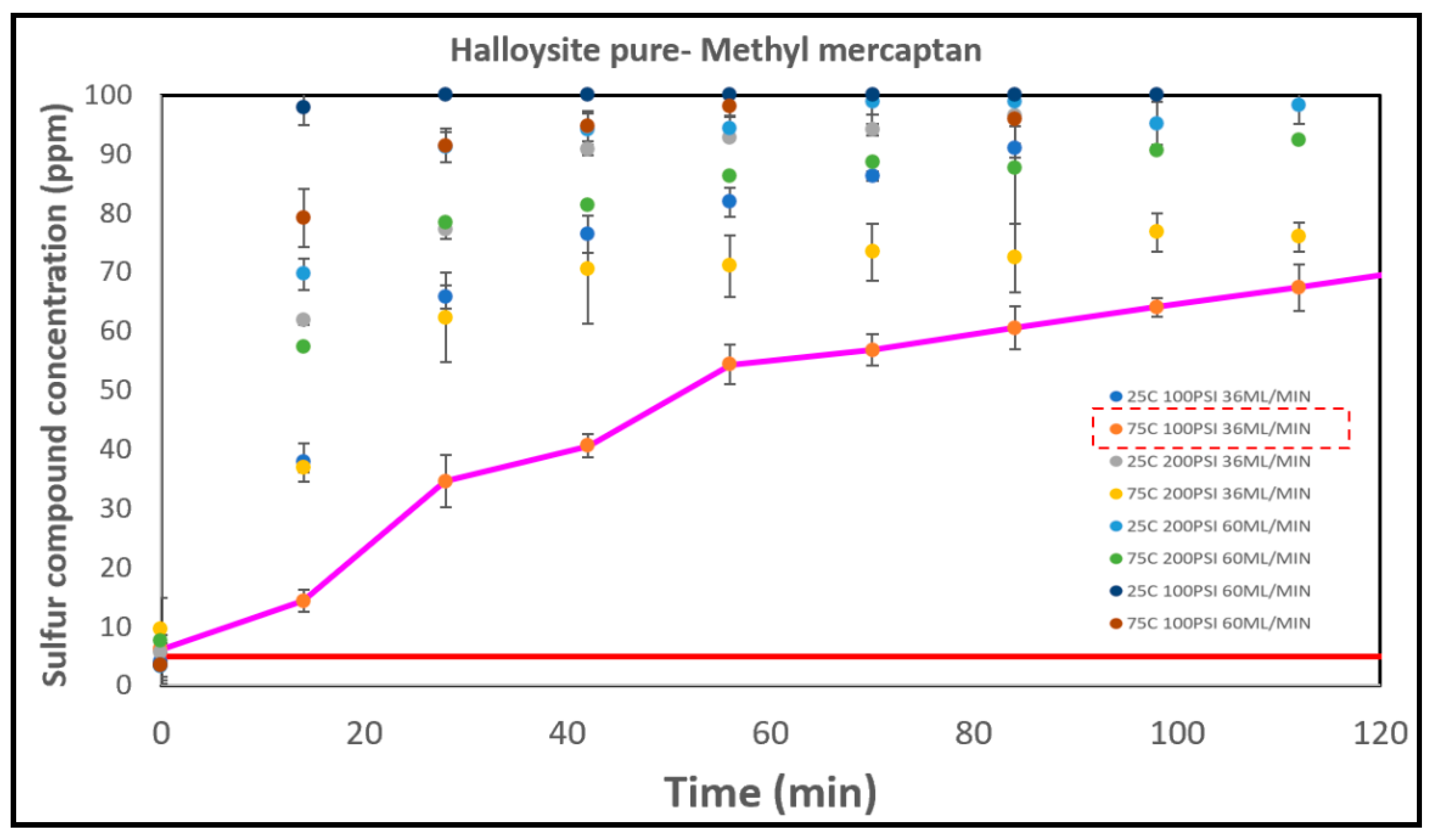
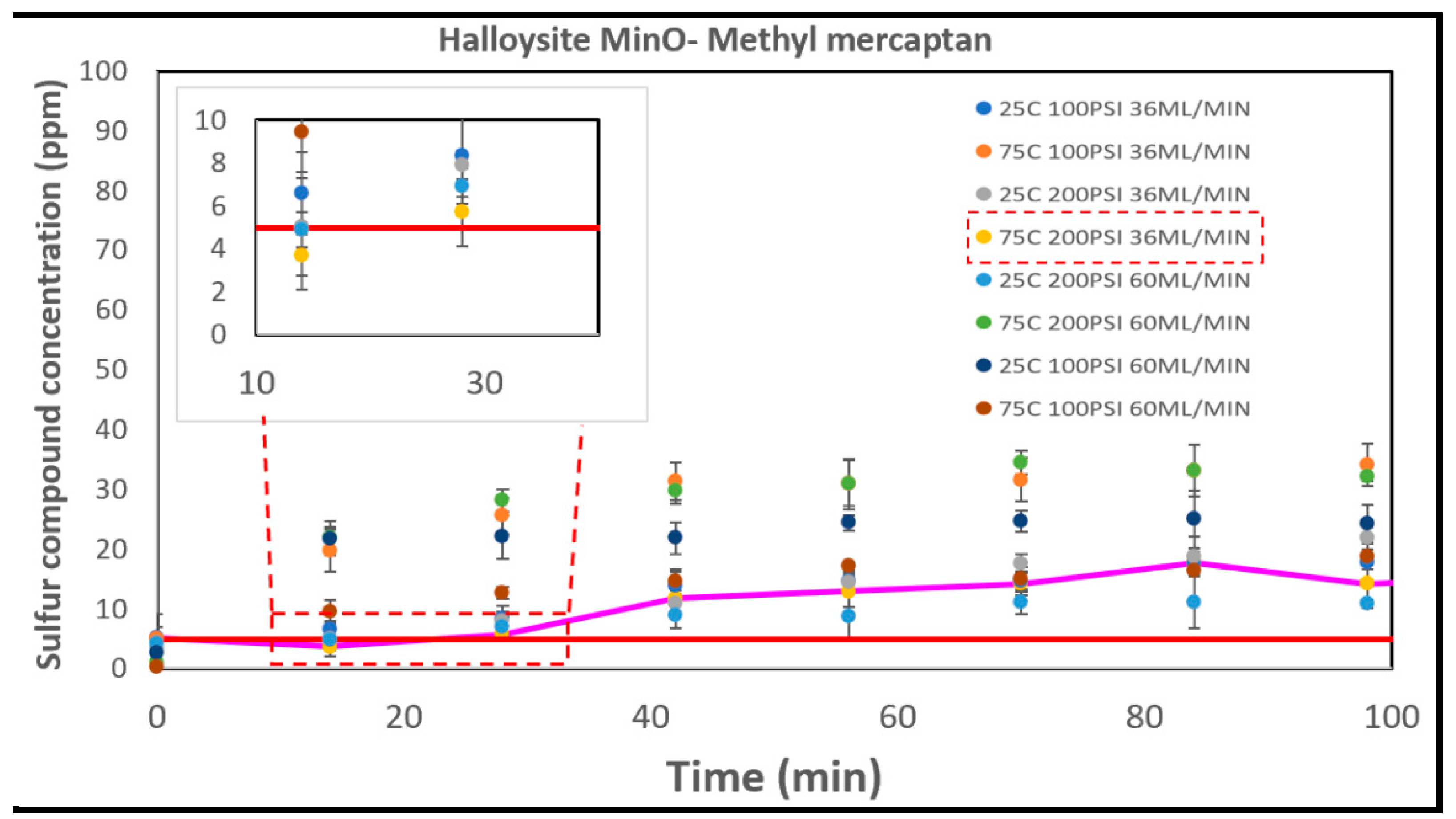
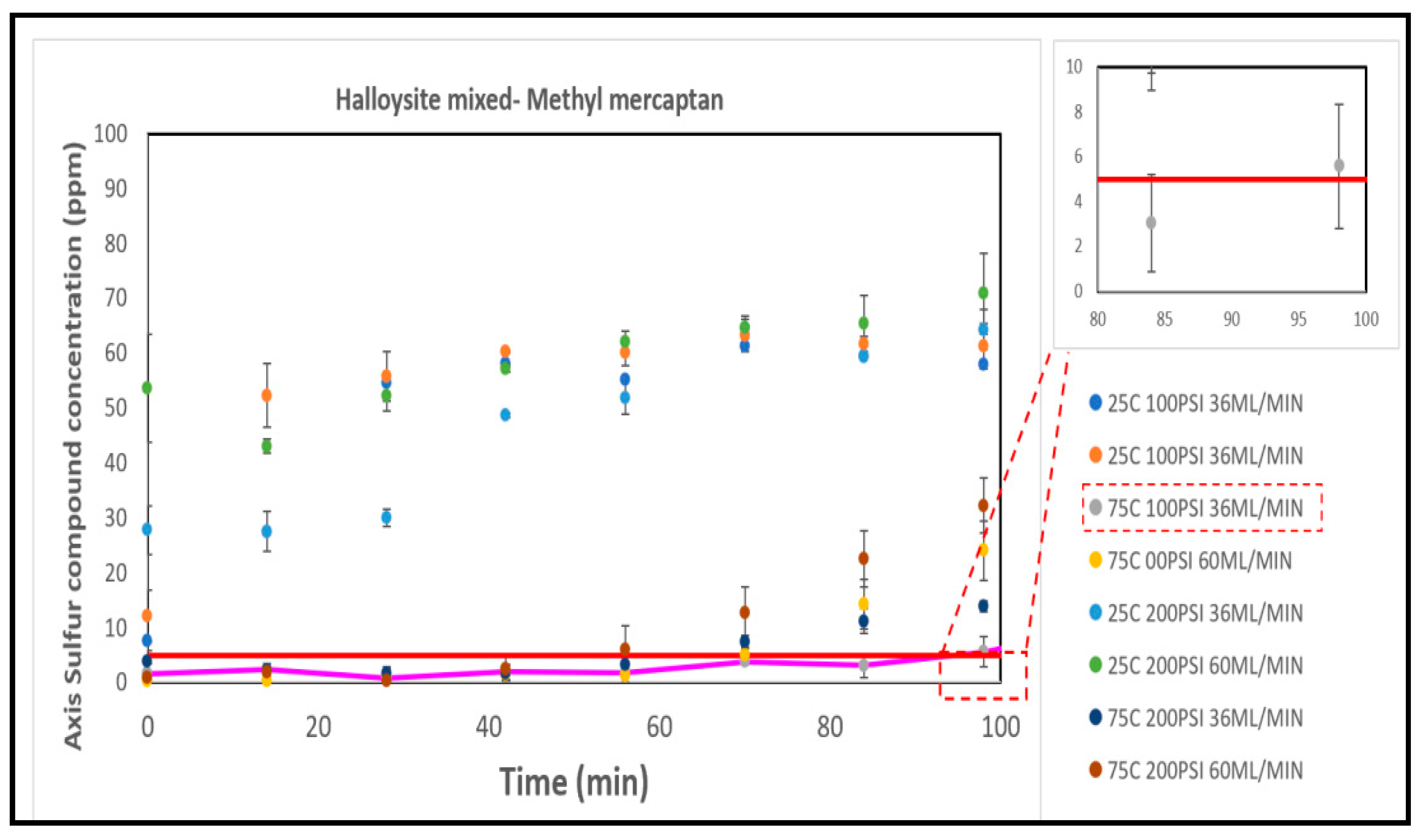
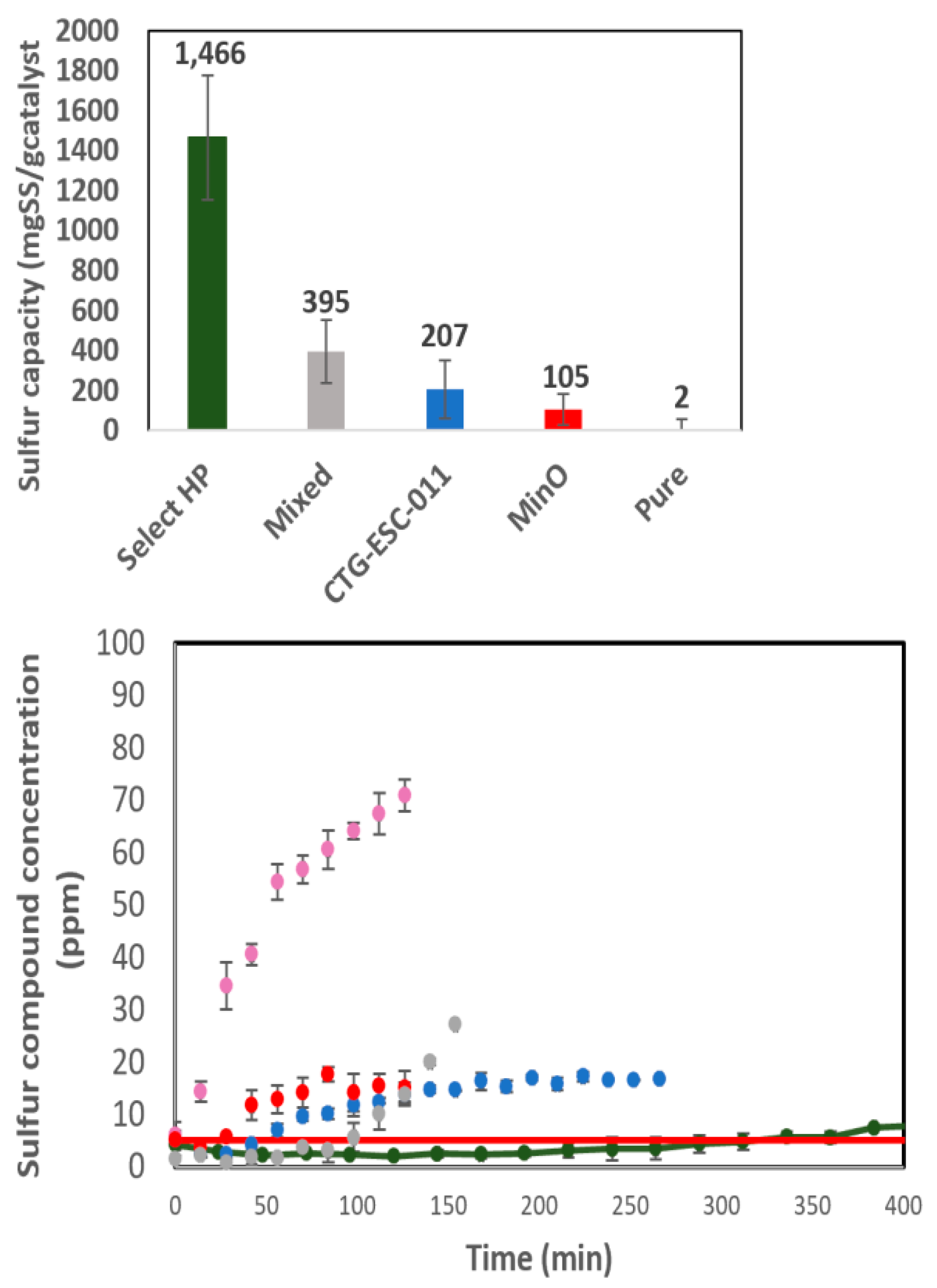
| Run | A (°C) | B (PSI) | C (ml/min) |
| 1 | 25 | 100 | 36 |
| 2 | 25 | 100 | 60 |
| 3 | 25 | 200 | 60 |
| 4 | 25 | 200 | 36 |
| 5 | 75 | 100 | 60 |
| 6 | 75 | 100 | 36 |
| 7 | 75 | 200 | 60 |
| 8 | 75 | 200 | 36 |
| Catalyst | SBET (m2/g) | vt (cm3/g) | Pore Size (A) |
|---|---|---|---|
| CTG-ESC-011 | 196.74 | 0.5 | 108.4 |
| Select HP | 88.82 | 0.27 | 137.7 |
| Halloysite pure | 51.18 | 0.21 | 163.9 |
| Halloysite mINo | 85.5 | 0.27 | 126.8 |
| Halloysite mixed | 27.63 | 0.089 | 129 |
| Catalyst | Theretical Chemical Composition | SBET (m2.g-1) | Vt(cm3.g-1) | Pore size (A) |
|---|---|---|---|---|
| Run 08 | .4Cu-.4Zn-.2Mg | 43.26 | 0.19 | 180.29 |
| Run 016 | .286Cu-.286Zn-.286Ni-.143Mg | 29.97 | 0.14 | 198.32 |
| Run 022 | .4Cu-.4Zn-.2Ni | 43.02 | 0.16 | 158.36 |
| Halloysite | Base | 51.18 | 0.21 | 163.9 |
| Compound [mass %] |
Select HP | CTG-ESG-011 | Halloysites | ||
|---|---|---|---|---|---|
| Pure | MinO | Mixed | |||
| Al | 11.4 | 0.7 | 23.5 | 23.2 | 23.9 |
| Si | 2 | 0.5 | 26.1 | 24.5 | 29.6 |
| Fe | 1.7 | 45.3 | 12.5 | 12.9 | 11.8 |
| Ca | 6.9 | 3.4 | 0.2 | 0.7 | 0.8 |
| Mg | 0.4 | 1.8 | 0 | 0 | 0.4 |
| K | 0 | 0 | 0.2 | 0 | 0.7 |
| Cl | 0 | 1.9 | 0 | 0 | 0 |
| Zn | 18.9 | 0.7 | 0 | 0 | 0 |
| Cu | 17.6 | 0 | 0 | 0 | 0 |
| Mn | 19.5 | 0.2 | 0 | 7.6 | 1.7 |
| O | 21.6 | 45.5 | 37.5 | 31.1 | 31.1 |
| Operating Conditions | Catalysts Breakthrough Time (mins) | ||||||
|---|---|---|---|---|---|---|---|
| Meal oxides | Halloysites | ||||||
| Temperature (OC) | Pressure (psi) | Flowrate (ml/min) | Select HP | CTG-ESC-011 | Pure | MinO | Mixed |
| 25 | 100 | 36 | 148.14 | 22.15 | 1.3 | 6.84 | 0 |
| 25 | 100 | 60 | 97.29 | 101.41 | 0.43 | 1.7 | 0 |
| 75 | 100 | 36 | 186.45 | 8.49 | 1 | 0.344 | 85.94 |
| 25 | 200 | 36 | 224.06 | 49.06 | 0 | 23.34 | 0 |
| 25 | 200 | 60 | 65.04 | 0 | 0.48 | 31.43 | 0 |
| 75 | 200 | 60 | 96.58 | 5.31 | 0 | 2.36 | 60.58 |
| 75 | 200 | 36 | 361.66 | 16.84 | 0 | 0 | 60.48 |
| 75 | 100 | 60 | 136.04 | 20.14 | 0.54 | 8.4 | 71.65 |
| Run | A:Cu (%) | B:Zn (%) | C:Ni (%) | D:Mg (%) | Bt time (minutes) | Sulfur Capacity (mg S/g catalyst) |
|---|---|---|---|---|---|---|
| 1 | 33.33 | 33.33 | 16.67 | 16.67 | 5.58 | 78.44 |
| 2 | 28.57 | 28.57 | 14.29 | 28.57 | 14.22 | 199.69 |
| 3 | 42.50 | 7.5 | 25.00 | 25 | 6 | 82.84 |
| 4 | 45.95 | 27.03 | 13.51 | 13.51 | 28.00 | 393.12 |
| 5 | 43.48 | 13.04 | 21.74 | 21.74 | 4.73 | 66.50 |
| 6 | 33.33 | 33.33 | 16.67 | 16.67 | 0.00 | 0 |
| 7 | 50.00 | 50 | 0 | 0 | 49 | 692.80 |
| 8 | 40.00 | 40 | 0 | 20 | 70 | 979.90 |
| 9 | 11.54 | 11.54 | 38.46 | 38.46 | 14.80 | 207.70 |
| 10 | 27.03 | 45.95 | 13.51 | 13.51 | 28.40 | 398.70 |
| 11 | 10 | 56.67 | 0 | 33.33 | 15.95 | 223.93 |
| 12 | 33.33 | 33.33 | 16.67 | 16.67 | 30.00 | 421.20 |
| 13 | 18.75 | 18.75 | 0 | 62.5 | 0.0 | 0 |
| 14 | 10 | 56.67 | 33.33 | 0 | 33 | 468.00 |
| 15 | 13.04 | 43.48 | 21.74 | 21.74 | 16.55 | 232.36 |
| 16 | 28.57 | 28.57 | 28.57 | 14.29 | 61.30 | 860.65 |
| 17 | 38.64 | 38.64 | 0 | 22.73 | 30.93 | 434.25 |
| 18 | 56.67 | 10 | 33.33 | 0 | 42 | 589.68 |
| 19 | 50 | 50 | 0 | 0 | 42 | 589.68 |
| 20 | 31.48 | 31.48 | 18.52 | 18.52 | 18.10 | 254.12 |
| 21 | 33.33 | 33.33 | 16.67 | 16.67 | 29.20 | 409.96 |
| 22 | 40 | 40 | 20 | 0 | 58 | 816.84 |
| 23 | 18.75 | 18.75 | 62.5 | 0 | 29 | 412.49 |
| 24 | 7.5 | 42.5 | 25 | 25 | 17 | 231.66 |
| 25 | 56.67 | 15 | 28.53 | 0 | 3 | 42.12 |
| 26 | 15.00 | 85 | 0 | 0 | 43 | 602.31 |
| 27 | 33.33 | 33.33 | 16.67 | 16.67 | 35.90 | 504.00 |
| 28 | 33.33 | 33.33 | 16.67 | 16.67 | 30.00 | 421.20 |
| 29 | 38.64 | 38.64 | 22.73 | 0 | 56 | 789 |
| 30 | 56.67 | 10 | 0 | 33.33 | 46.00 | 645.84 |
| Order | Adsorbent | Composition | ToC | P atm | Sulfur cap mgs/gCat | Reference |
|---|---|---|---|---|---|---|
| Zeolites | ||||||
| 1 | 13X(NaX) | 0-2000ppm CH3SH in CH4 | 25 | 1 | 182.8 | Taheri et al, 2017 |
| 2 | 5A | 0-2000ppm CH3SH in CH4 | 25 | 1 | 186.3 | Taheri et al, 2017 |
| 3 | Silica Allumina gel | 0-2000ppm CH3SH in CH4 | 25 | 1 | 24 | Taheri et al, 2017 |
| 4 | CaX | .05% CH3SH in CH4 | 25 | 1 | 140 | Ryzhikov et al, 2011 |
| 5 | MgNaX | .05% CH3SH in CH4 | 25 | 1 | 132 | Ryzhikov et al, 2011 |
| 6 | ZnNaX | .05% CH3SH in CH4 | 25 | 1 | 115 | Ryzhikov et al, 2011 |
| Activated carbon | ||||||
| 8 | HNO3-AC | 2000ppm CH4SH | 25 | 1 | 602.1 | Liu et al, 2017 |
| 9 | AC | 50 ppm of methyl mercaptan | 25 | 1 | 1.1 | Zhao et al, 2022 |
| 10 | aAC-Fe | 50 ppm of methyl mercaptan | 25 | 1 | 13.4 | Zhao et al, 2022 |
| MOF | ||||||
| 11 | MIL-53(Al) | CH3SCH3 in methane | 25 | 1 | 433 | Taheri et al, 2017 |
| 12 | MAC-2 | 600mgm-3.CH3SCH3 | 50 | _ | 85.3 | Shi et al, 2017 |
| 13 | MAC-1 | 600mgm-3.CH3SCH3 | 50 | _ | 66.3 | Shi et al, 2017 |
| 14 | MOF-199 | 600mgm-3.CH3SCH3 | 50 | _ | 60.1 | Shi et al, 2017 |
| 15 | MAC-3 | 600mgm-3.CH3SCH3 | 50 | _ | 58.1 | Shi et al, 2017 |
| Metal Oxides | ||||||
| 16 | Select HP | 200 ppm of CH3SCH3 in methane | 25 | 13.6 | 389.1 | This work |
| 17 | .33Mn-33Cu-33Zn | 200 ppm of CH3SCH3 in methane | 25 | 13.6 | 1213 | This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




